Irrigation Increases Crop Species Diversity in Low-Diversity Farm Regions of Mexico
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Sources and Variables
2.2. Quantile Regressions
3. Results
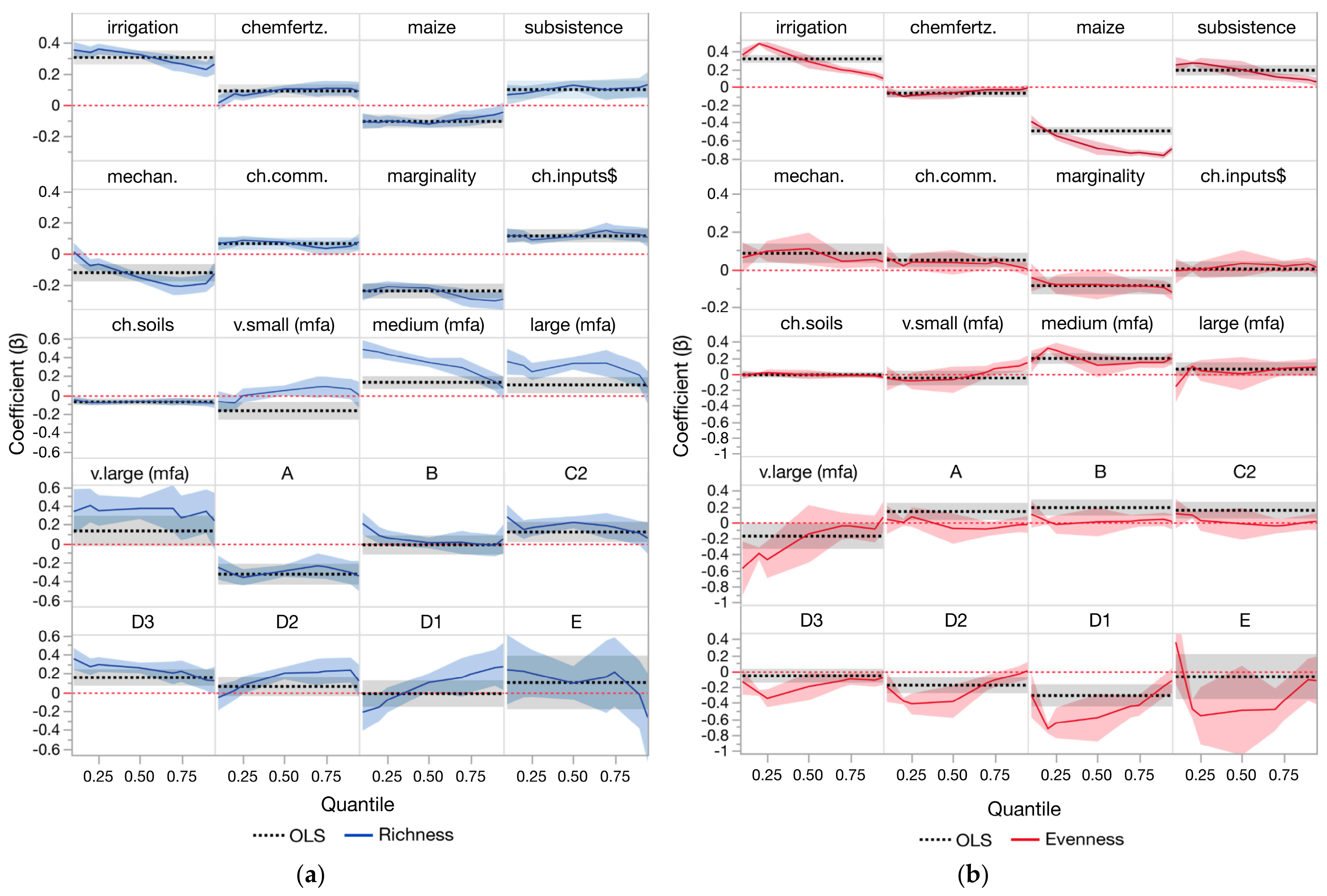
3.1. Model Results (OLS)
3.2. Model Results (Quantile)
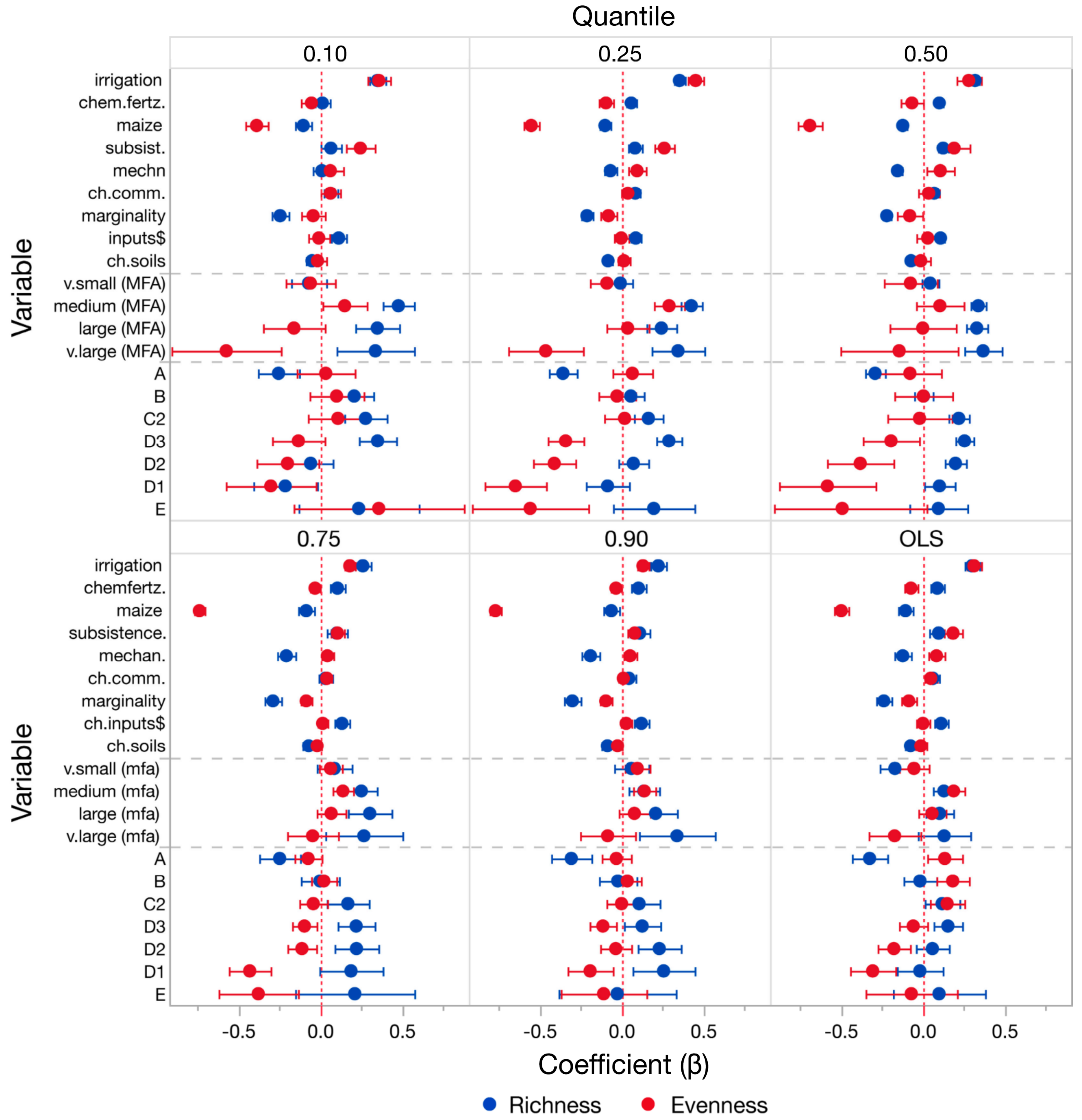
3.3. Forest Plot Comparisons
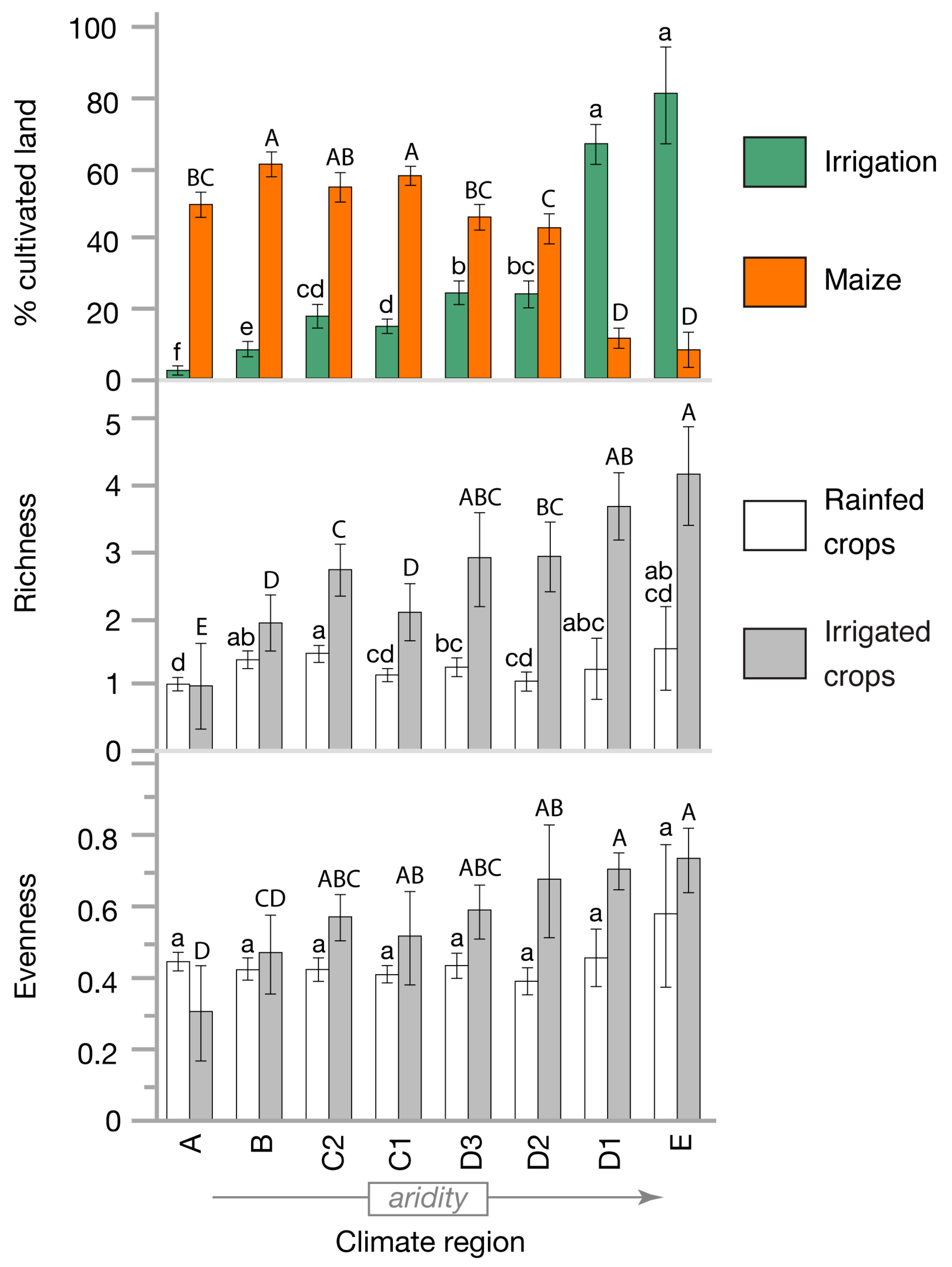
3.4. Rainfed and Irrigated Crop Diversity
4. Discussion
4.1. Irrigation Enhances Crop Species Richness and Evenness Diversity
4.2. Irrigation Has Stronger Effects in Regions of Low Crop Species Diversity
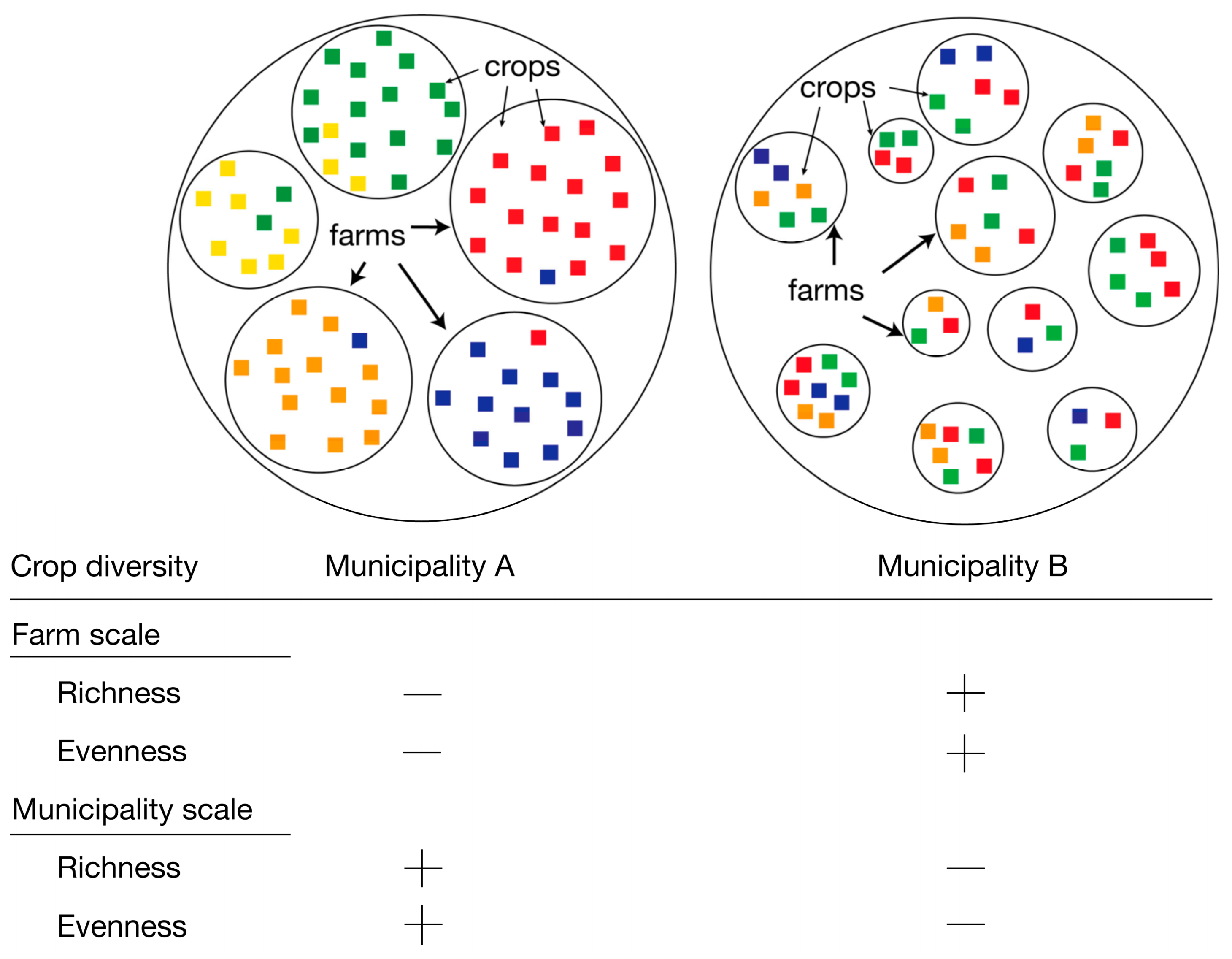
4.3. Crop Species Diversity and Scale: Important Distinctions
4.4. Other Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Variable | OLS | Richness (Margalef) | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Quantile Regression | ||||||||||||||||||
| 10th | 25th | 50th | 75th | 90th | ||||||||||||||
| β | SE | β | SE | β | SE | β | SE | β | SE | β | SE | |||||||
| irrigation | 0.30 | 0.02 | *** | 0.35 | 0.03 | *** | 0.35 | 0.02 | *** | 0.32 | 0.01 | *** | 0.26 | 0.02 | *** | 0.23 | 0.02 | *** |
| chemfertz. | 0.09 | 0.02 | *** | 0.01 | 0.02 | 0.06 | 0.02 | *** | 0.10 | 0.01 | *** | 0.11 | 0.02 | *** | 0.10 | 0.02 | *** | |
| maize | −0.11 | 0.02 | *** | −0.10 | 0.03 | *** | −0.10 | 0.17 | *** | −0.12 | 0.01 | *** | −0.09 | 0.03 | *** | −0.06 | 0.02 | ** |
| ch.inputs $ | 0.11 | 0.02 | *** | 0.11 | 0.02 | *** | 0.09 | 0.02 | *** | 0.11 | 0.01 | *** | 0.13 | 0.02 | *** | 0.12 | 0.02 | *** |
| ch.soils | −0.07 | 0.02 | *** | −0.05 | 0.02 | ** | −0.08 | 0.01 | *** | −0.07 | 0.01 | *** | −0.07 | 0.02 | *** | −0.09 | 0.02 | *** |
| ch.comm. | 0.06 | 0.02 | *** | 0.06 | 0.02 | ** | 0.08 | 0.02 | *** | 0.07 | 0.01 | *** | 0.03 | 0.02 | 0.05 | 0.02 | * | |
| mechan. | −0.12 | 0.03 | *** | 0.01 | 0.03 | −0.07 | 0.02 | *** | −0.15 | 0.01 | *** | −0.21 | 0.03 | *** | −0.19 | 0.03 | *** | |
| subsistance | 0.10 | 0.03 | *** | 0.06 | 0.03 | * | 0.08 | 0.02 | *** | 0.12 | 0.02 | *** | 0.10 | 0.03 | ** | 0.11 | 0.03 | *** |
| marginality | −0.24 | 0.02 | *** | −0.25 | 0.03 | *** | −0.21 | 0.02 | *** | −0.22 | 0.01 | *** | −0.29 | 0.03 | *** | −0.30 | 0.03 | *** |
| v.small (mfa) | −0.17 | 0.05 | *** | −0.07 | 0.05 | −0.01 | 0.04 | 0.04 | 0.03 | 0.09 | 0.05 | 0.06 | 0.05 | |||||
| med. (mfa) | 0.13 | 0.03 | *** | 0.48 | 0.05 | *** | 0.43 | 0.03 | *** | 0.34 | 0.02 | *** | 0.25 | 0.05 | *** | 0.14 | 0.05 | *** |
| Large (mfa) | 0.10 | 0.04 | ** | 0.35 | 0.07 | *** | 0.24 | 0.05 | *** | 0.33 | 0.03 | *** | 0.30 | 0.07 | *** | 0.21 | 0.07 | ** |
| v.large (mfa) | 0.13 | 0.08 | *** | 0.34 | 0.12 | ** | 0.35 | 0.08 | *** | 0.37 | 0.06 | *** | 0.27 | 0.12 | * | 0.34 | 0.12 | ** |
| A | −0.33 | 0.06 | *** | −0.26 | 0.06 | *** | −0.36 | 0.04 | *** | −0.29 | 0.03 | *** | −0.25 | 0.06 | *** | −0.31 | 0.06 | *** |
| B | −0.02 | 0.05 | 0.21 | 0.06 | *** | 0.06 | 0.04 | 0.00 | 0.03 | 0.00 | 0.06 | −0.02 | 0.06 | |||||
| C2 | 0.12 | 0.05 | * | 0.28 | 0.07 | *** | 0.16 | 0.05 | *** | 0.22 | 0.03 | *** | 0.17 | 0.07 | ** | 0.11 | 0.06 | |
| D3 | 0.15 | 0.04 | *** | 0.35 | 0.06 | *** | 0.29 | 0.04 | *** | 0.26 | 0.03 | *** | 0.22 | 0.06 | *** | 0.13 | 0.06 | * |
| D2 | 0.06 | 0.05 | −0.06 | 0.07 | 0.07 | 0.05 | 0.20 | 0.03 | *** | 0.22 | 0.07 | ** | 0.23 | 0.07 | *** | |||
| D1 | −0.02 | 0.07 | −0.21 | 0.10 | * | −0.09 | 0.07 | 0.10 | 0.05 | * | 0.19 | 0.10 | * | 0.26 | 0.10 | ** | ||
| E | 0.10 | 0.14 | 0.24 | 0.19 | 0.20 | 0.13 | 0.10 | 0.09 | 0.21 | 0.19 | −0.03 | 0.18 | ||||||
| Adj. R-sqr | 0.38 | |||||||||||||||||
| F-test | 73.29 | |||||||||||||||||
| Variable | OLS | Evenness (Simpson) | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Quantile Regression | ||||||||||||||||||
| 10th | 25th | 50th | 75th | 90th | ||||||||||||||
| β | SE | β | SE | β | SE | β | SE | β | SE | β | SE | |||||||
| irrigation | 0.31 | 0.02 | *** | 0.36 | 0.04 | *** | 0.35 | 0.02 | *** | 0.28 | 0.04 | *** | 0.18 | 0.02 | *** | 0.13 | 0.02 | *** |
| chemfertz. | −0.07 | 0.02 | *** | −0.05 | 0.03 | 0.06 | 0.02 | *** | −0.07 | 0.04 | −0.03 | 0.02 | * | −0.03 | 0.02 | * | ||
| maize | −0.50 | 0.02 | *** | −0.39 | 0.04 | *** | −0.10 | 0.02 | *** | −0.69 | 0.04 | *** | −0.74 | 0.02 | *** | −0.77 | 0.02 | *** |
| ch.inputs $ | 0.00 | 0.02 | −0.01 | 0.03 | 0.09 | 0.02 | *** | 0.03 | 0.04 | 0.01 | 0.02 | 0.03 | 0.02 | |||||
| ch.soils | −0.01 | 0.02 | −0.02 | 0.03 | −0.08 | 0.01 | *** | −0.01 | 0.03 | −0.02 | 0.01 | −0.02 | 0.01 | |||||
| ch.comm. | 0.05 | 0.02 | ** | 0.06 | 0.03 | * | 0.08 | 0.01 | *** | 0.04 | 0.03 | 0.04 | 0.01 | ** | 0.01 | 0.02 | ||
| mechan. | 0.08 | 0.03 | *** | 0.06 | 0.04 | −0.07 | 0.02 | *** | 0.11 | 0.04 | * | 0.04 | 0.02 | * | 0.05 | 0.02 | ** | |
| subsistance | 0.19 | 0.03 | *** | 0.25 | 0.04 | *** | 0.08 | 0.02 | *** | 0.19 | 0.05 | *** | 0.10 | 0.02 | *** | 0.08 | 0.02 | *** |
| marginality | −0.09 | 0.02 | *** | −0.04 | 0.04 | −0.21 | 0.02 | *** | −0.08 | 0.04 | * | −0.09 | 0.02 | *** | −0.10 | 0.02 | *** | |
| v.small (mfa) | −0.05 | 0.05 | −0.06 | 0.08 | −0.01 | 0.04 | −0.08 | 0.08 | 0.06 | 0.04 | 0.10 | 0.04 | * | |||||
| med. (mfa) | 0.19 | 0.03 | *** | 0.15 | 0.07 | * | 0.43 | 0.03 | *** | 0.10 | 0.07 | 0.14 | 0.03 | *** | 0.14 | 0.03 | *** | |
| Large (mfa) | 0.06 | 0.04 | −0.16 | 0.10 | 0.24 | 0.05 | *** | 0.00 | 0.10 | 0.07 | 0.05 | 0.08 | 0.05 | |||||
| v.large (mfa) | −0.17 | 0.08 | * | −0.58 | 0.17 | *** | 0.35 | 0.08 | *** | −0.14 | 0.18 | −0.05 | 0.08 | −0.08 | 0.09 | |||
| A | 0.13 | 0.05 | * | 0.03 | 0.09 | −0.36 | 0.04 | *** | −0.08 | 0.10 | −0.07 | 0.04 | −0.03 | 0.05 | ||||
| B | 0.18 | 0.05 | *** | 0.10 | 0.08 | 0.06 | 0.04 | 0.00 | 0.09 | 0.02 | 0.04 | 0.04 | 0.04 | |||||
| C2 | 0.15 | 0.05 | ** | 0.11 | 0.09 | 0.16 | 0.04 | *** | −0.02 | 0.10 | −0.04 | 0.04 | 0.00 | 0.05 | ||||
| D3 | −0.06 | 0.04 | −0.13 | 0.08 | 0.29 | 0.04 | *** | −0.19 | 0.09 | * | −0.10 | 0.04 | * | −0.11 | 0.04 | ** | ||
| D2 | −0.18 | 0.05 | *** | −0.20 | 0.10 | * | 0.07 | 0.05 | −0.38 | 0.10 | *** | −0.11 | 0.05 | * | −0.04 | 0.05 | ||
| D1 | −0.31 | 0.07 | *** | −0.30 | 0.14 | * | −0.09 | 0.07 | −0.58 | 0.15 | *** | −0.43 | 0.07 | *** | −0.19 | 0.07 | ** | |
| E | −0.07 | 0.14 | 0.36 | 0.27 | 0.20 | 0.13 | −0.49 | 0.29 | −0.38 | 0.12 | ** | −0.11 | 0.13 | |||||
| Adj. R-sqr | 0.39 | |||||||||||||||||
| F-test | 76.78 | |||||||||||||||||
References
- Zsögön, A.; Peres, L.E.P.; Xiao, Y.; Yan, J.; Fernie, A.R. Enhancing Crop Diversity for Food Security in the Face of Climate Uncertainty. Plant J. 2022, 109, 402–414. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, C.C.; Emery, B.F.; Niles, M.T. Global Relationships between Crop Diversity and Nutritional Stability. Nat. Commun. 2021, 12, 5310. [Google Scholar] [CrossRef] [PubMed]
- Keleman, A.; Rañó, H.G.; Hellin, J. Maize Diversity, Poverty, and Market Access: Lessons from Mexico. Dev. Pract. 2009, 19, 187–199. [Google Scholar] [CrossRef]
- Mesfin, W.; Fufa, B.; Haji, J. Pattern, Trend and Determinants of Crop Diversification: Empirical Evidence from Smallholders in Eastern Ethiopia. J. Econ. Sustain. Dev. 2011, 2, 78–89. [Google Scholar]
- Reidsma, P.; Ewert, F. Regional Farm Diversity Can Reduce Vulnerability of Food Production to Climate Change. Ecol. Soc. 2008, 13, 38. [Google Scholar] [CrossRef]
- Stefan, L.; Hartmann, M.; Engbersen, N.; Six, J.; Schöb, C. Positive Effects of Crop Diversity on Productivity Driven by Changes in Soil Microbial Composition. Front. Microbiol. 2021, 12, 808. [Google Scholar] [CrossRef]
- Renard, D.; Tilman, D. National Food Production Stabilized by Crop Diversity. Nature 2019, 571, 257–260. [Google Scholar] [CrossRef]
- Massawe, F.; Mayes, S.; Cheng, A. Crop Diversity: An Unexploited Treasure Trove for Food Security. Trends Plant Sci. 2016, 21, 365–368. [Google Scholar] [CrossRef]
- Gaudin, A.C.M.; Tolhurst, T.N.; Ker, A.P.; Janovicek, K.; Tortora, C.; Martin, R.C.; Deen, W. Increasing Crop Diversity Mitigates Weather Variations and Improves Yield Stability. PLoS ONE 2015, 10, e0113261. [Google Scholar] [CrossRef]
- Louhichi, K.; Ciaian, P.; Espinosa, M.; Colen, L.; Perni, A.; y Paloma, S.G. Does the Crop Diversification Measure Impact EU Farmers’ Decisions? An Assessment Using an Individual Farm Model for CAP Analysis (IFM-CAP). Land Use Policy 2017, 66, 250–264. [Google Scholar] [CrossRef]
- Fenzi, M.; Bonneuil, C. From “Genetic Resources” to “Ecosystems Services”: A Century of Science and Global Policies for Crop Diversity Conservation. Cult. Agric. Food Environ. 2016, 38, 72–83. [Google Scholar] [CrossRef]
- Liu, Z.; Tang, G.; Zhou, Y.; Sun, J.; Wu, W.; Yang, P. Why Can China Maintain a High Crop Diversity? A Spatial-Temporal Dynamic Analysis. Prog. Phys. Geogr. Earth Environ. 2021, 46, 030913332110558. [Google Scholar] [CrossRef]
- Agrawal, T.; Hirons, M.; Gathorne-Hardy, A. Understanding Farmers’ Cropping Decisions and Implications for Crop Diversity Conservation: Insights from Central India. Curr. Res. Environ. Sustain. 2021, 3, 100068. [Google Scholar] [CrossRef]
- Hufnagel, J.; Reckling, M.; Ewert, F. Diverse Approaches to Crop Diversification in Agricultural Research. A Review. Agron. Sustain. Dev. 2020, 40, 14. [Google Scholar] [CrossRef]
- Moss, C.; Lukac, M.; Harris, F.; Outhwaite, C.L.; Scheelbeek, P.F.D.; Green, R.; Berstein, F.M.; Dangour, A.D. The Effects of Crop Diversity and Crop Type on Biological Diversity in Agricultural Landscapes: A Systematic Review Protocol. Wellcome Open Res. 2020, 4, 101. [Google Scholar] [CrossRef]
- Khoury, C.K.; Brush, S.; Costich, D.E.; Curry, H.A.; de Haan, S.; Engels, J.M.M.; Guarino, L.; Hoban, S.; Mercer, K.L.; Miller, A.J.; et al. Crop Genetic Erosion: Understanding and Responding to Loss of Crop Diversity. New Phytol. 2022, 233, 84–118. [Google Scholar] [CrossRef]
- Sumasgutner, P.; Terraube, J.; Coulon, A.; Villers, A.; Chakarov, N.; Kruckenhauser, L.; Korpimäki, E. Landscape Homogenization Due to Agricultural Intensification Disrupts the Relationship between Reproductive Success and Main Prey Abundance in an Avian Predator. Front. Zool. 2019, 16, 31. [Google Scholar] [CrossRef]
- Matson, P.A.; Parton, W.J.; Power, A.G.; Swift, M.J. Agricultural Intensification and Ecosystem Properties. Science 1997, 277, 504–509. [Google Scholar] [CrossRef]
- Teixeira, H.M.; Van den Berg, L.; Cardoso, I.M.; Vermue, A.J.; Bianchi, F.J.J.A.; Peña-Claros, M.; Tittonell, P. Understanding Farm Diversity to Promote Agroecological Transitions. Sustainability 2018, 10, 4337. [Google Scholar] [CrossRef]
- Alvarez, S.; Timler, C.J.; Michalscheck, M.; Paas, W.; Descheemaeker, K.; Tittonell, P.; Andersson, J.A.; Groot, J.C.J. Capturing Farm Diversity with Hypothesis-Based Typologies: An Innovative Methodological Framework for Farming System Typology Development. PLoS ONE 2018, 13, e0194757. [Google Scholar] [CrossRef]
- Altieri, M.; Nicholls, C.; Montalba, R. Technological Approaches to Sustainable Agriculture at a Crossroads: An Agroecological Perspective. Sustainability 2017, 9, 349. [Google Scholar] [CrossRef]
- LaFevor, M.C.; Magliocca, N.R. Farmland Size, Chemical Fertilizers, and Irrigation Management Effects on Maize and Wheat Yield in Mexico. J. Land Use Sci. 2020, 15, 532–546. [Google Scholar] [CrossRef]
- Firbank, L.G.; Attwood, S.; Eory, V.; Gadanakis, Y.; Lynch, J.M.; Sonnino, R.; Takahashi, T. Grand Challenges in Sustainable Intensification and Ecosystem Services. Front. Sustain. Food Syst. 2018, 2, 7. [Google Scholar] [CrossRef]
- Weltin, M.; Zasada, I.; Piorr, A.; Debolini, M.; Geniaux, G.; Moreno Perez, O.; Scherer, L.; Tudela Marco, L.; Schulp, C.J.E. Conceptualising Fields of Action for Sustainable Intensification—A Systematic Literature Review and Application to Regional Case Studies. Agric. Ecosyst. Environ. 2018, 257, 68–80. [Google Scholar] [CrossRef]
- Cassman, K.G.; Grassini, P. A Global Perspective on Sustainable Intensification Research. Nat. Sustain. 2020, 3, 262–268. [Google Scholar] [CrossRef]
- Feliciano, D. A Review on the Contribution of Crop Diversification to Sustainable Development Goal 1 “No Poverty” in Different World Regions. Sustain. Dev. 2019, 27, 795–808. [Google Scholar] [CrossRef]
- Pretty, J.; Bharucha, Z.P. Sustainable Intensification in Agricultural Systems. Ann. Bot. 2014, 114, 1571–1596. [Google Scholar] [CrossRef]
- Gliessman, S. Scaling-out and Scaling-up Agroecology. Agroecol. Sustain. Food Syst. 2018, 42, 841–842. [Google Scholar] [CrossRef]
- Altieri, M.A.; Nicholls, C.I. Agroecology Scaling Up for Food Sovereignty and Resiliency. In Sustainable Agriculture Reviews; Lichtfouse, E., Ed.; Sustainable Agriculture Reviews; Springer: Dordrecht, The Netherlands, 2012; Volume 11, pp. 1–29. ISBN 978-94-007-5449-2. [Google Scholar]
- Wezel, A.; Goris, M.; Bruil, J.; Félix, G.; Peeters, A.; Bàrberi, P.; Bellon, S.; Migliorini, P. Challenges and Action Points to Amplify Agroecology in Europe. Sustainability 2018, 10, 1598. [Google Scholar] [CrossRef]
- Bernard, B.; Lux, A. How to Feed the World Sustainably: An Overview of the Discourse on Agroecology and Sustainable Intensification. Reg. Environ. Chang. 2017, 17, 1279–1290. [Google Scholar] [CrossRef]
- Beckmann, M.; Gerstner, K.; Akin-Fajiye, M.; Ceaușu, S.; Kambach, S.; Kinlock, N.L.; Phillips, H.R.P.; Verhagen, W.; Gurevitch, J.; Klotz, S.; et al. Conventional Land-Use Intensification Reduces Species Richness and Increases Production: A Global Meta-Analysis. Glob. Chang. Biol. 2019, 25, 1941–1956. [Google Scholar] [CrossRef] [PubMed]
- Winowiecki, L.A.; Bourne, M.; Magaju, C.; Neely, C.; Massawe, B.; Masikati, P.; Vågen, T.-G.; Musili, F.; Nabi, M.; Nguyo, A.; et al. Bringing Evidence to Bear for Negotiating Tradeoffs in Sustainable Agricultural Intensification Using a Structured Stakeholder Engagement Process. Int. J. Agric. Sustain. 2021, 19, 474–496. [Google Scholar] [CrossRef]
- Li, S.; Lei, Y.; Zhang, Y.; Liu, J.; Shi, X.; Jia, H.; Wang, C.; Chen, F.; Chu, Q. Rational Trade-Offs between Yield Increase and Fertilizer Inputs Are Essential for Sustainable Intensification: A Case Study in Wheat–Maize Cropping Systems in China. Sci. Total Environ. 2019, 679, 328–336. [Google Scholar] [CrossRef] [PubMed]
- Struik, P.C.; Kuyper, T.W. Sustainable Intensification in Agriculture: The Richer Shade of Green. A Review. Agron. Sustain. Dev. 2017, 37, 39. [Google Scholar] [CrossRef]
- Herzig, A.; Dymond, J.; Ausseil, A.-G. Exploring Limits and Trade-Offs of Irrigation and Agricultural Intensification in the Ruamahanga Catchment, New Zealand. N. Z. J. Agric. Res. 2016, 59, 216–234. [Google Scholar] [CrossRef]
- Fenzi, M.; Rogé, P.; Cruz-Estrada, A.; Tuxill, J.; Jarvis, D. Community Seed Network in an Era of Climate Change: Dynamics of Maize Diversity in Yucatán, Mexico. Agric. Hum. Values 2021, 39, 339–356. [Google Scholar] [CrossRef]
- Fenzi, M.; Jarvis, D.I.; Arias Reyes, L.M.; Latournerie Moreno, L.; Tuxill, J. Longitudinal Analysis of Maize Diversity in Yucatan, Mexico: Influence of Agro-Ecological Factors on Landraces Conservation and Modern Variety Introduction. Plant Genet. Resour. 2017, 15, 51–63. [Google Scholar] [CrossRef]
- Pérez-García, O.; del Castillo, R.F. The Decline of the Itinerant Milpa and the Maintenance of Traditional Agrobiodiversity: Crops and Weeds Coexistence in a Tropical Cloud Forest Area in Oaxaca, Mexico. Agric. Ecosyst. Environ. 2016, 228, 30–37. [Google Scholar] [CrossRef]
- LaFevor, M.C.; Frake, A.N.; Couturier, S. Targeting Irrigation Expansion to Address Sustainable Development Objectives: A Regional Farm Typology Approach. Water 2021, 13, 2393. [Google Scholar] [CrossRef]
- Arreguín-Cortes, F.I.A.; Villanueva, N.H.G.; Casillas, A.G.; Gonzalez, J.A.G. Reforms in the Administration of Irrigation Systems: Mexican Experiences. Irrig. Drain. 2019, 68, 6–19. [Google Scholar] [CrossRef]
- INEGI (Instituto Nacional de Estadística y Geografía El VIII Censo Agrícola). Ganadero y Forestal 2007: Aspectors Metodológicos y Principales Resultados. Available online: https://www.inegi.org.mx/programas/cagf/2007/ (accessed on 27 March 2020).
- CONAPO. Índices de Marginación|Consejo Nacional de Población CONAPO; Secretaría de Gobernación: Ciudad Juárez, Mexico, 2005. [Google Scholar]
- CONAZA; UACH. Escenarios Climatológicos de la República Mexicana Ante el Cambio Climático; Comisión Nacional de las Zonas Áridas: Aguascalientes, Mexico; Universidad Autónoma Chapingo: Texcoco, Mexico, 2003; ISBN 968-884-941-3. [Google Scholar]
- SIAP Estadística de la Producción Agrícola (2007); Servicio de Información Agroalimentaría y Pesquera; Secretaría de Agricultura y Desarrollo Rural: Mexico City, Mexico, 2007.
- Crossley, M.S.; Burke, K.D.; Schoville, S.D.; Radeloff, V.C. Recent Collapse of Crop Belts and Declining Diversity of US Agriculture since 1840. Glob. Chang. Biol. 2021, 27, 151–164. [Google Scholar] [CrossRef] [PubMed]
- Lazíková, J.; Bandlerová, A.; Rumanovská, Ľ.; Takáč, I.; Lazíková, Z. Crop Diversity and Common Agricultural Policy—The Case of Slovakia. Sustainability 2019, 11, 1416. [Google Scholar] [CrossRef]
- Martin, A.R.; Cadotte, M.W.; Isaac, M.E.; Milla, R.; Vile, D.; Violle, C. Regional and Global Shifts in Crop Diversity through the Anthropocene. PLoS ONE 2019, 14, e0209788. [Google Scholar] [CrossRef] [PubMed]
- Roberts, F.S. Measurement of Biodiversity: Richness and Evenness. In Mathematics of Planet Earth: Protecting Our Planet, Learning from the Past, Safeguarding for the Future; Springer: Berlin/Heidelberg, Germany, 2019; pp. 203–224. [Google Scholar]
- Morris, E.K.; Caruso, T.; Buscot, F.; Fischer, M.; Hancock, C.; Maier, T.S.; Meiners, T.; Müller, C.; Obermaier, E.; Prati, D.; et al. Choosing and Using Diversity Indices: Insights for Ecological Applications from the German Biodiversity Exploratories. Ecol. Evol. 2014, 4, 3514–3524. [Google Scholar] [CrossRef]
- Sauerbrei, W.; Perperoglou, A.; Schmid, M.; Abrahamowicz, M.; Becher, H.; Binder, H.; Dunkler, D.; Harrell, F.E.; Royston, P.; Heinze, G.; et al. State of the Art in Selection of Variables and Functional Forms in Multivariable Analysis—Outstanding Issues. Diagn. Progn. Res. 2020, 4, 3. [Google Scholar] [CrossRef]
- LaFevor, M.C.; Ponette-González, A.G.; Larson, R.; Mungai, L.M. Spatial Targeting of Agricultural Support Measures: Indicator-Based Assessment of Coverages and Leakages. Land 2021, 10, 740. [Google Scholar] [CrossRef]
- Galeana-Pizaña, J.M.; Couturier, S.; Figueroa, D.; Jiménez, A.D. Is Rural Food Security Primarily Associated with Smallholder Agriculture or with Commercial Agriculture?: An Approach to the Case of Mexico Using Structural Equation Modeling. Agric. Syst. 2021, 190, 103091. [Google Scholar] [CrossRef]
- Samberg, L.H.; Gerber, J.S.; Ramankutty, N.; Herrero, M.; West, P.C. Subnational Distribution of Average Farm Size and Smallholder Contributions to Global Food Production. Environ. Res. Lett. 2016, 11, 124010. [Google Scholar] [CrossRef]
- Lamu, A.N.; Olsen, J.A. The Relative Importance of Health, Income and Social Relations for Subjective Well-Being: An Integrative Analysis. Soc. Sci. Med. 2016, 152, 176–185. [Google Scholar] [CrossRef]
- Hao, L.; Naiman, D.Q. Quantile Regression; Quantitative Applications in the Social Sciences; Sage: Thousand Oaks, CA, USA, 2007; ISBN 1-4129-2628-9. [Google Scholar]
- John, O.O. Robustness of Quantile Regression to Outliers. Am. J. Appl. Math. Stat. 2015, 3, 86–88. [Google Scholar] [CrossRef]
- Cade, B.S.; Noon, B.R. A Gentle Introduction to Quantile Regression for Ecologists. Front. Ecol. Environ. 2003, 1, 412–420. [Google Scholar] [CrossRef]
- Alsayed, A.R.M.; Isa, Z.; Kun, S.S.; Manzi, G. Quantile Regression to Tackle the Heterogeneity on the Relationship Between Economic Growth, Energy Consumption, and CO2 Emissions. Environ. Model. Assess. 2020, 25, 251–258. [Google Scholar] [CrossRef]
- Waldmann, E. Quantile Regression: A Short Story on How and Why. Stat. Model. 2018, 18, 203–218. [Google Scholar] [CrossRef]
- Zahniser, S.; López, N.F.L.; Motamed, M.; Vargas, Z.Y.S.; Capehart, T. The Growing Corn Economies of Mexico and the United States; FDS-19f-01; US Department of Agriculture, Economic Research Service: Washington, DC, USA, 2019. [Google Scholar]
- Lerner, A.M.; Appendini, K. Dimensions of Peri-Urban Maize Production in the Toluca-Atlacomulco Valley, Mexico. J. Lat. Am. Geogr. 2011, 10, 87–106. [Google Scholar] [CrossRef]
- Boserup, E. The Conditions of Agricultural Growth: The Economics of Agrarian Change under Population Pressure; Aldine: Chicago, IL, USA, 1965. [Google Scholar]
- Turner, B.L.; Doolittle, W.E. The Concept and Measure of Agricultural Intensity. Prof. Geogr. 1978, 30, 297–301. [Google Scholar] [CrossRef]
- FAO. The Ethics of Sustainable Agricultural Intensification; Food Agriculture Organization: Rome, Italy, 2004; ISBN 978-92-5-105067-5. [Google Scholar]
- D’Odorico, P.; Davis, K.F.; Rosa, L.; Carr, J.A.; Chiarelli, D.; Dell’Angelo, J.; Gephart, J.; MacDonald, G.K.; Seekell, D.A.; Suweis, S.; et al. The Global Food-Energy-Water Nexus. Rev. Geophys. 2018, 56, 456–531. [Google Scholar] [CrossRef]
- MacDonald, G.K.; D’Odorico, P.; Seekell, D.A. Pathways to Sustainable Intensification through Crop Water Management. Environ. Res. Lett. 2016, 11, 091001. [Google Scholar] [CrossRef]
- Mukherjee, A. Evaluation of the Policy of Crop Diversification as a Strategy for Reduction of Rural Poverty in India. In Poverty Reduction Policies and Practices in Developing Asia; Economic Studies in Inequality, Social Exclusion and Well-Being; Heshmati, A., Maasoumi, E., Wan, G., Eds.; Spring: Singapore, 2015; pp. 125–143. ISBN 978-981-287-419-1. [Google Scholar]
- Zimmerer, K.S. Conserving Agrobiodiversity amid Global Change, Migration, and Nontraditional Livelihood Networks: The Dynamic Uses of Cultural Landscape Knowledge. E&S 2014, 19, art1. [Google Scholar] [CrossRef]
- Thapa, G.; Kumar, A.; Roy, D.; Joshi, P.K. Impact of Crop Diversification on Rural Poverty in Nepal. Can. J. Agric. Econ./Rev. Can. D’agroeconomie 2018, 66, 379–413. [Google Scholar] [CrossRef]
- Rahman, S.; Kazal, M.M.H. Determinants of Crop Diversity in the Regions of Bangladesh (1990–2008): Determinants of Crop Diversity in Bangladesh. Singap. J. Trop. Geogr. 2015, 36, 83–97. [Google Scholar] [CrossRef]
- Passarelli, S.; Mekonnen, D.; Bryan, E.; Ringler, C. Evaluating the Pathways from Small-Scale Irrigation to Dietary Diversity: Evidence from Ethiopia and Tanzania. Food Sec. 2018, 10, 981–997. [Google Scholar] [CrossRef]
- Domenech, L.; Ringler, C. Impact of Irrigation on Nutrition, Health and Gender: A Review Paper with Insights for Africa South of the Sahar; The International Food Policy Research Institute: Washington, DC, USA, 2013; p. 28. [Google Scholar]
- Aheibam, M.; Singh, R.; Feroze, S.M.; Singh, N.U.; Singh, R.J.; Singh, A.K. Identifying the Determinants and Extent of Crop Diversification at Household Level: An Evidence from Ukhrul District, Manipur. Econ. Aff. 2017, 62, 89. [Google Scholar] [CrossRef]
- Headey, D.D.; Hoddinott, J. Agriculture, Nutrition and the Green Revolution in Bangladesh. Agric. Syst. 2016, 149, 122–131. [Google Scholar] [CrossRef]
- Hossain, M.; Naher, F.; Shahabuddin, Q. Food Security and Nutrition in Bangladesh: Progress and Determinants. eJADE Electron. J. Agric. Dev. Econ. 2005, 2, 30. [Google Scholar]
- Cutforth, L.B.; Francis, C.A.; Lynne, G.D.; Mortensen, D.A.; Eskridge, K.M. Factors Affecting Farmers’ Crop Diversity Decisions: An Integrated Approach. Am. J. Altern. Agric. 2001, 16, 168–176. [Google Scholar] [CrossRef]
- Akanda, A.I. Rethinking Crop Diversification under Changing Climate, Hydrology and Food Habit in Bangladesh. J. Agric. Environ. Int. Dev. 2010, 104, 3–23. [Google Scholar] [CrossRef]
- Rosegrant, M.W.; Schleyer, R.G.; Yadav, S.N. Water Policy for Efficient Agricultural Diversification: Market-Based Approaches. Food Policy 1995, 20, 203–223. [Google Scholar] [CrossRef]
- Van Zonneveld, M.; Turmel, M.-S.; Hellin, J. Decision-Making to Diversify Farm Systems for Climate Change Adaptation. Front. Sustain. Food Syst. 2020, 4, 32. [Google Scholar] [CrossRef]
- Giordano, M.; Namara, R.; Bassini, E. The Impacts of Irrigation: A Review of Published Evidence; The World Bank: Washington, DC, USA, 2019; 46p. [Google Scholar]
- Goslee, S.C. Drivers of Agricultural Diversity in the Contiguous United States. Front. Sustain. Food Syst. 2020, 4, 75. [Google Scholar] [CrossRef]
- Mortensen, D.A.; Smith, R.G. Confronting Barriers to Cropping System Diversification. Front. Sustain. Food Syst. 2020, 4, 199. [Google Scholar] [CrossRef]
- Kissoly, L.D.; Karki, S.K.; Grote, U. Diversity in Farm Production and Household Diets: Comparing Evidence From Smallholders in Kenya and Tanzania. Front. Sustain. Food Syst. 2020, 4, 77. [Google Scholar] [CrossRef]
- Michler, J.D.; Josephson, A.L. To Specialize or Diversify: Agricultural Diversity and Poverty Dynamics in Ethiopia. World Dev. 2017, 89, 214–226. [Google Scholar] [CrossRef]
- Hartman, S.; Chiarelli, D.D.; Rulli, M.C.; D’Odorico, P. A Growing Produce Bubble: United States Produce Tied to Mexico’s Unsustainable Agricultural Water Use. Environ. Res. Lett. 2021, 16, 105008. [Google Scholar] [CrossRef]
- Turrent-Fernández, A.; Cortés-Flores, J.I.; Espinosa-Calderón, A.; Turrent-Thompson, C.; Mejía-Andrade, H. Cambio Climático y Algunas Estrategias Agrícolas Para Fortalecer La Seguridad Alimentaria de México. Rev. Mex. Cienc. Agrícolas 2016, 7, 1727–1739. [Google Scholar] [CrossRef][Green Version]
- Turrent-Fernández, A.T.; Gómez, R.C.; Luna, A.L.; Almaguer, M.C.; Silva, J.R.; Méndez, J.M.; Caballero, A.P. Producción de Maíz Bajo Riego En El Sur-Sureste de México: I. Análisis Agronómico. Agric. Técnica México 2004, 30, 153–167. [Google Scholar]
- CONAGUA. Estadísticas de Agua en México, Edición 2018; Comisión Nacional de Agua: Mexico City, Mexico, 2018. [Google Scholar]
- Rosa, L.; Chiarelli, D.D.; Rulli, M.C.; Dell’Angelo, J.; D’Odorico, P. Global Agricultural Economic Water Scarcity. Sci. Adv. 2020, 6, eaaz6031. [Google Scholar] [CrossRef]
- Wilder, M. Water Governance in Mexico: Political and Economic Aperatures and a Shifting State-Citizen Relationship. Ecol. Soc. 2010, 15, 22. [Google Scholar] [CrossRef]
- Rosa, L.; Chiarelli, D.D.; Sangiorgio, M.; Beltran-Peña, A.A.; Rulli, M.C.; D’Odorico, P.; Fung, I. Potential for Sustainable Irrigation Expansion in a 3 °C Warmer Climate. Proc. Natl. Acad. Sci. USA 2020, 117, 29526–29534. [Google Scholar] [CrossRef]
- Smith, J.C.; Ghosh, A.; Hijmans, R.J. Agricultural Intensification Was Associated with Crop Diversification in India (1947–2014). PLoS ONE 2019, 14, e0225555. [Google Scholar] [CrossRef]
- Merlos, F.A.; Hijmans, R.J. The Scale Dependency of Spatial Crop Species Diversity and Its Relation to Temporal Diversity. Proc. Natl. Acad. Sci. USA 2020, 117, 26176–26182. [Google Scholar] [CrossRef]
- Walters, K.E.; Martiny, J.B.H. Alpha-, Beta-, and Gamma-Diversity of Bacteria Varies across Habitats. PLoS ONE 2020, 15, e0233872. [Google Scholar] [CrossRef] [PubMed]
- Van de Wouw, M.; Kik, C.; van Hintum, T.; van Treuren, R.; Visser, B. Genetic Erosion in Crops: Concept, Research Results and Challenges. Plant Genet. Res. 2010, 8, 1–15. [Google Scholar] [CrossRef]
- Valentine, J.W. Determinants of Diversity in Higher Taxonomic Categories. Paleobiology 1980, 6, 444–450. [Google Scholar] [CrossRef]
- Lopez-Ridaura, S.; Barba-Escoto, L.; Reyna-Ramirez, C.A.; Sum, C.; Palacios-Rojas, N.; Gerard, B. Maize Intercropping in the Milpa System. Diversity, Extent and Importance for Nutritional Security in the Western Highlands of Guatemala. Sci. Rep. 2021, 11, 3696. [Google Scholar] [CrossRef]
- Orozco-Ramírez, Q.; Perales, H.; Hijmans, R.J. Geographical Distribution and Diversity of Maize (Zea mays L. Subsp. Mays) Races in Mexico. Genet. Resour. Crop. Evol. 2017, 64, 855–865. [Google Scholar] [CrossRef]
- Aguiar, S.; Texeira, M.; Garibaldi, L.A.; Jobbágy, E.G. Global Changes in Crop Diversity: Trade Rather than Production Enriches Supply. Glob. Food Secur. 2020, 26, 100385. [Google Scholar] [CrossRef]
- Beillouin, D.; Ben-Ari, T.; Malézieux, E.; Seufert, V.; Makowski, D. Positive but Variable Effects of Crop Diversification on Biodiversity and Ecosystem Services. Glob. Chang. Biol. 2021, 27, 4697–4710. [Google Scholar] [CrossRef]
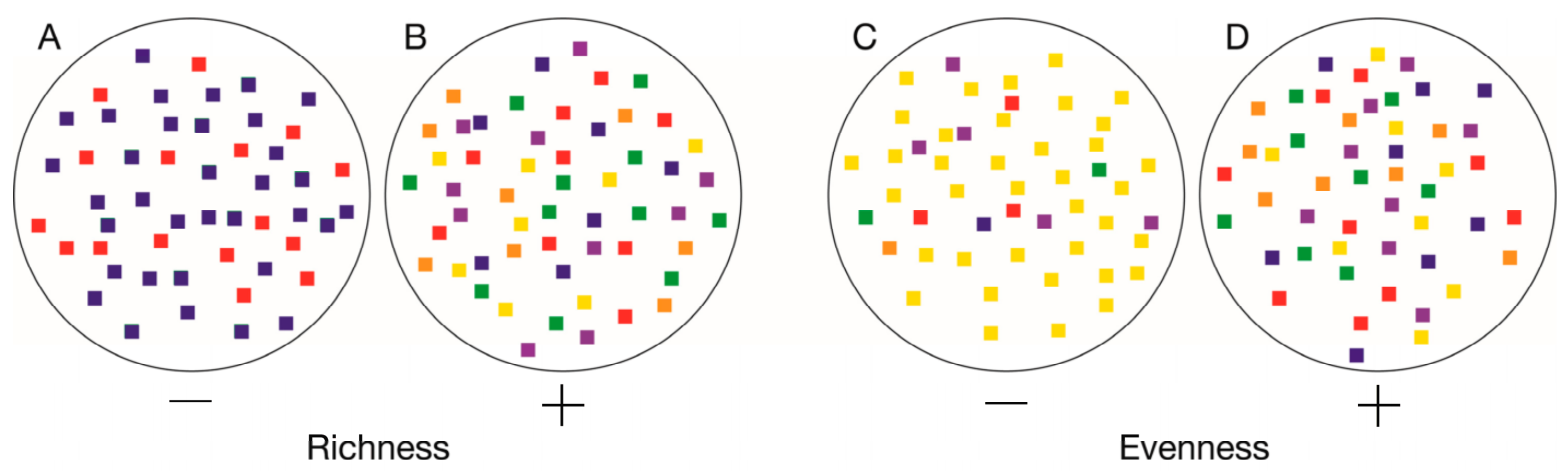
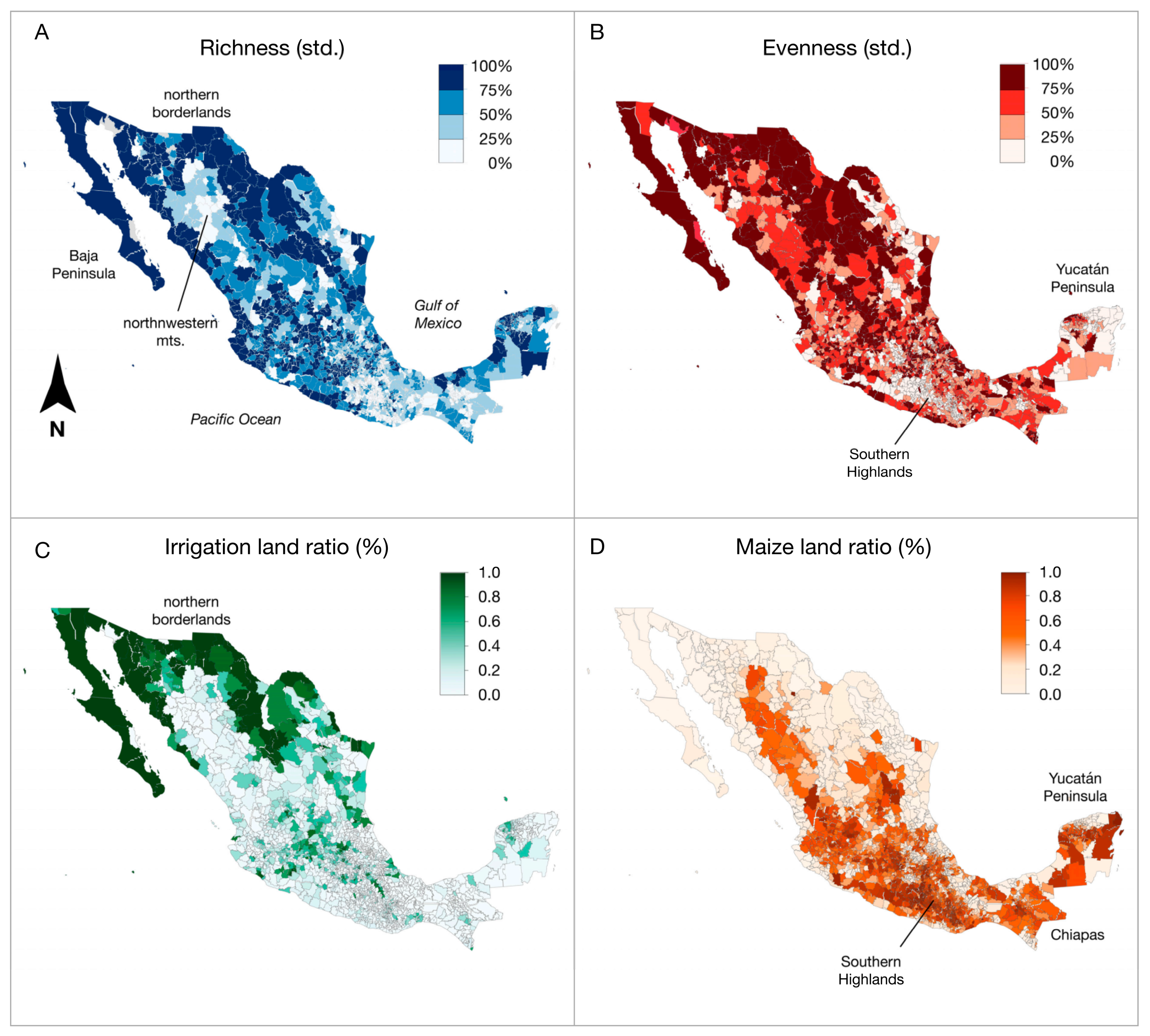
| Municipalities (N = 2455) | |||||
|---|---|---|---|---|---|
| Continuous | Variable | Unit | Code | Mean | SD |
| Land use | Irrigation land ratio | % cropland | Irrigation | 0.19 | 0.28 |
| Chemical fertilizer land ratio | chemfertz. | 0.28 | 0.27 | ||
| Maize land ratio | Maize | 0.49 | 0.32 | ||
| Farm challenges | Input costs | % farms | ch.inputs $ | 0.22 | 0.20 |
| Soil fertility | ch.soils | 0.23 | 0.20 | ||
| Commercialization | ch.comm. | 0.54 | 0.25 | ||
| Labor | Mechanized | mechan. | 0.32 | 0.31 | |
| Production | Subsistence | subsistence | 0.72 | 0.25 | |
| Socioeconomic | Marginalization | index (std.) | marginality | 0.00 | 1.00 |
| N | % | ||||
| Categorical | 2455 | 100 | |||
| Mean farm area (MFA) | Very small (0–2 ha) | v.small (mfa) | 421 | 17 | |
| Small (2–5 ha) * | small (mfa) | 871 | 35 | ||
| Medium (5–15 ha) | med. (mfa) | 727 | 30 | ||
| Large (15–50 ha) | large (mfa) | 350 | 14 | ||
| Very large (>50 ha) | v.large (mfa) | 86 | 4 | ||
| 2455 | 100 | ||||
| Climate region | Perhumid (A) | A | 334 | 14 | |
| Humid (B) | B | 333 | 14 | ||
| Moist subhumid (C2) | C2 | 246 | 10 | ||
| Dry subhumid (C1) * | C1 | 704 | 29 | ||
| Semiarid light (D3) | D3 | 388 | 16 | ||
| Semiarid moderate (D2) | D2 | 258 | 11 | ||
| Semiarid dry (D1) | D1 | 164 | 7 | ||
| Arid (E) | E | 28 | 1 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
LaFevor, M.C.; Pitts, A.K. Irrigation Increases Crop Species Diversity in Low-Diversity Farm Regions of Mexico. Agriculture 2022, 12, 911. https://doi.org/10.3390/agriculture12070911
LaFevor MC, Pitts AK. Irrigation Increases Crop Species Diversity in Low-Diversity Farm Regions of Mexico. Agriculture. 2022; 12(7):911. https://doi.org/10.3390/agriculture12070911
Chicago/Turabian StyleLaFevor, Matthew C., and Aoife K. Pitts. 2022. "Irrigation Increases Crop Species Diversity in Low-Diversity Farm Regions of Mexico" Agriculture 12, no. 7: 911. https://doi.org/10.3390/agriculture12070911
APA StyleLaFevor, M. C., & Pitts, A. K. (2022). Irrigation Increases Crop Species Diversity in Low-Diversity Farm Regions of Mexico. Agriculture, 12(7), 911. https://doi.org/10.3390/agriculture12070911






