The Impact of Dormancy Breakers on Hormone Profiles, Fruit Growth and Quality in Sweet Cherry
Abstract
:1. Introduction
2. Materials and Methods
2.1. Bud Sampling for Phenological Assessment and Hormone Analysis
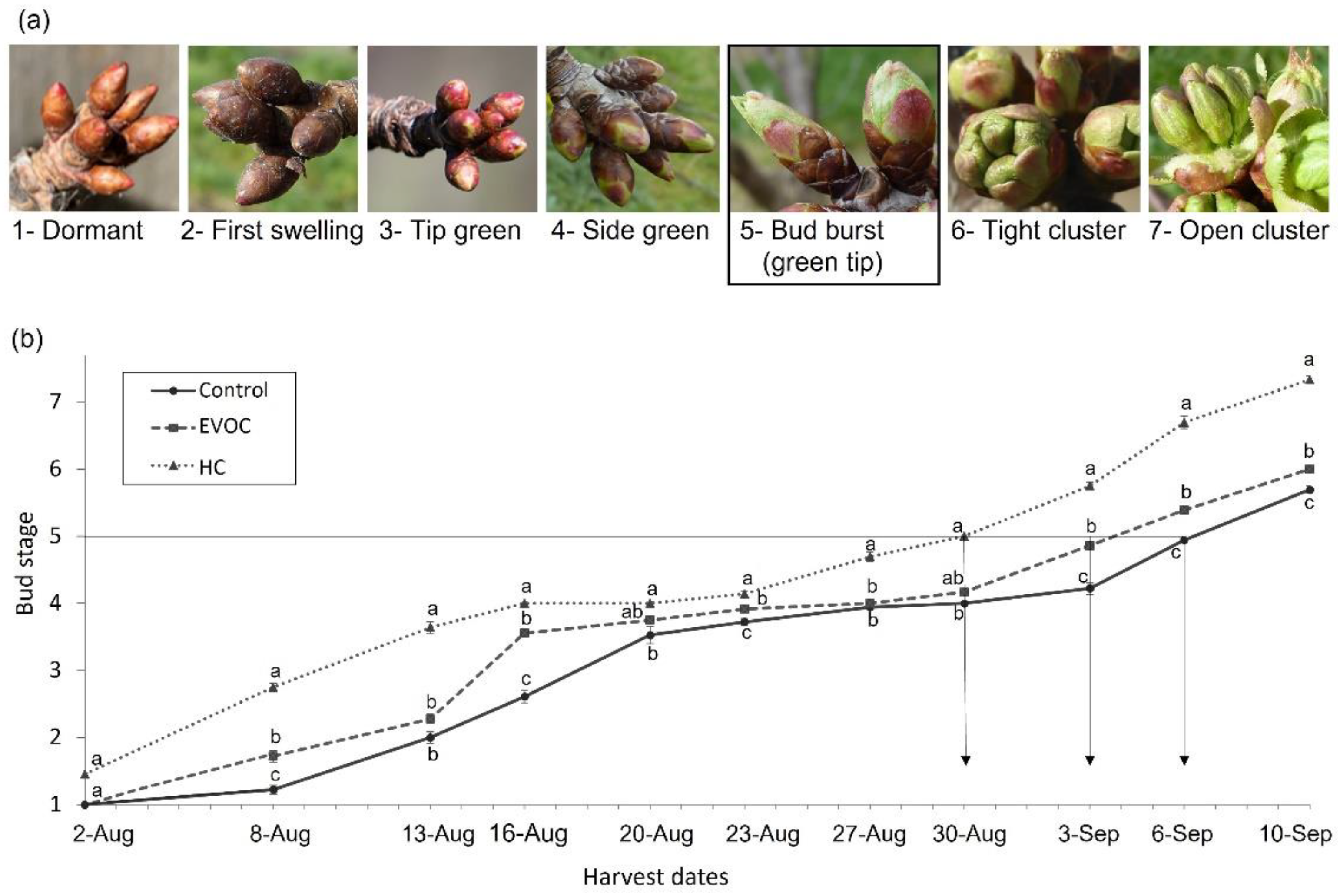
2.1.1. Bud Stage Assessment
2.1.2. Hormone Extraction
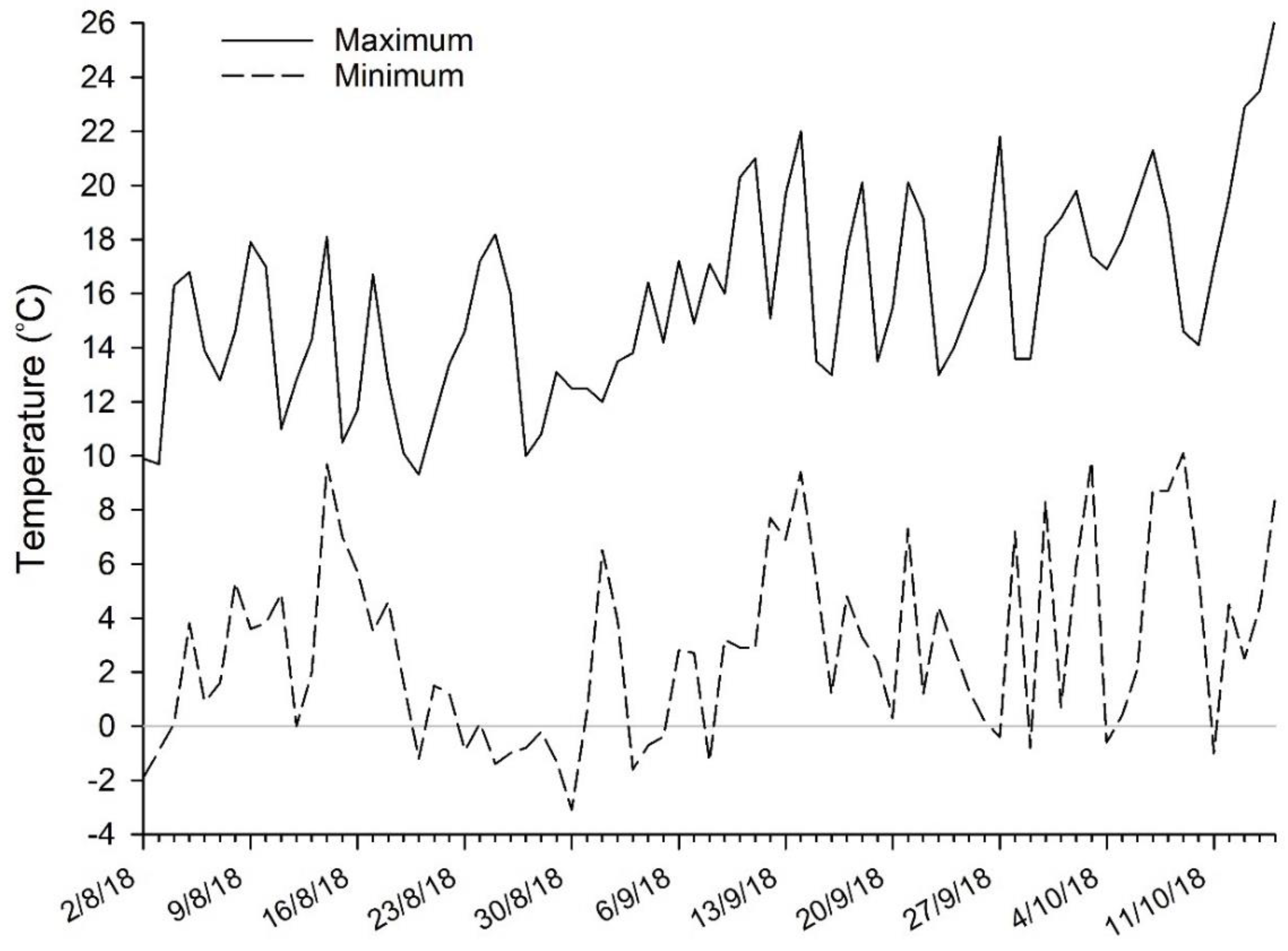
2.2. Field Assessment of Bud Development and Phenological Stage
2.3. Fruit Growth and Quality Sampling and Assessment
2.4. Statistical Analysis
3. Results
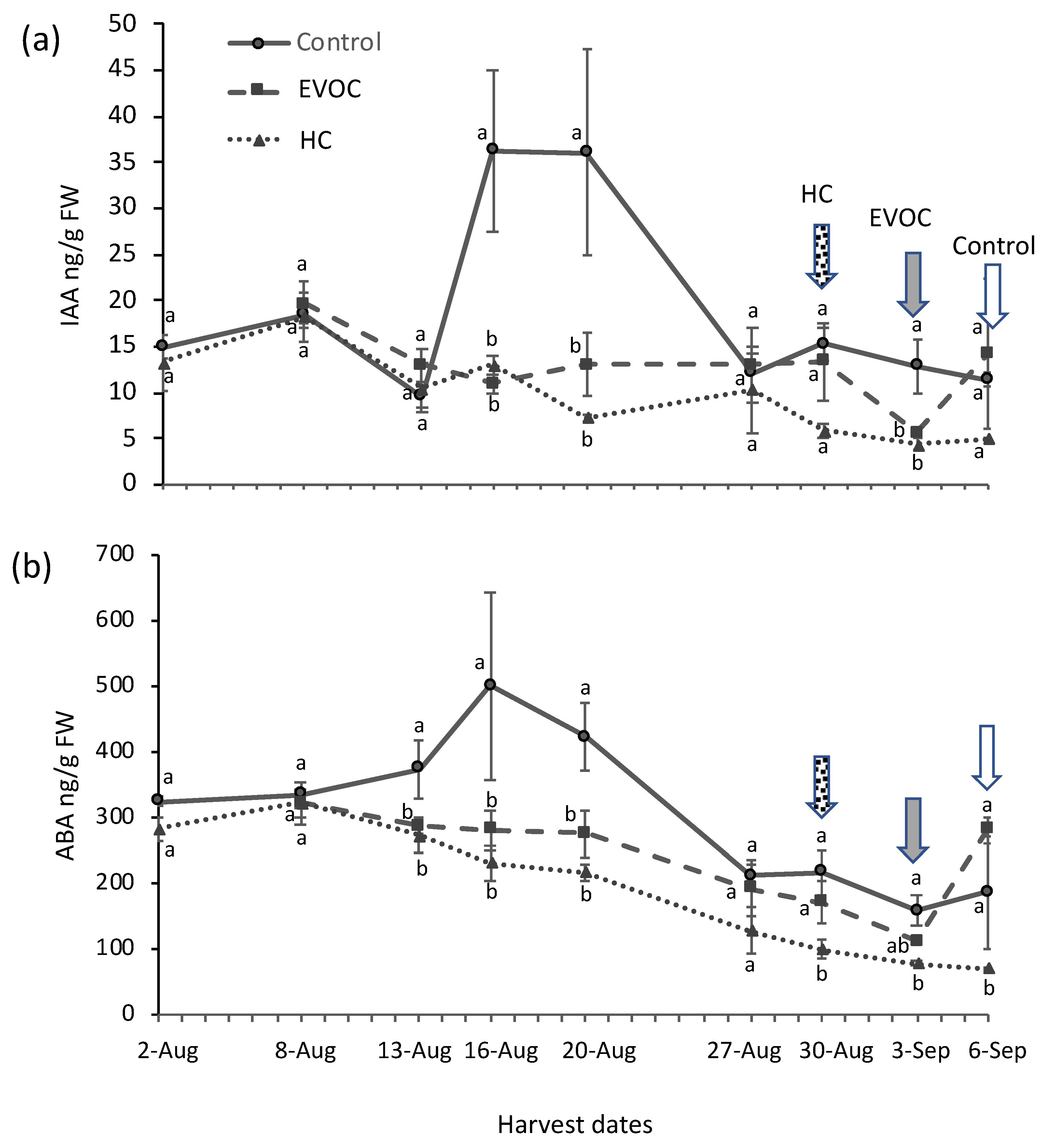
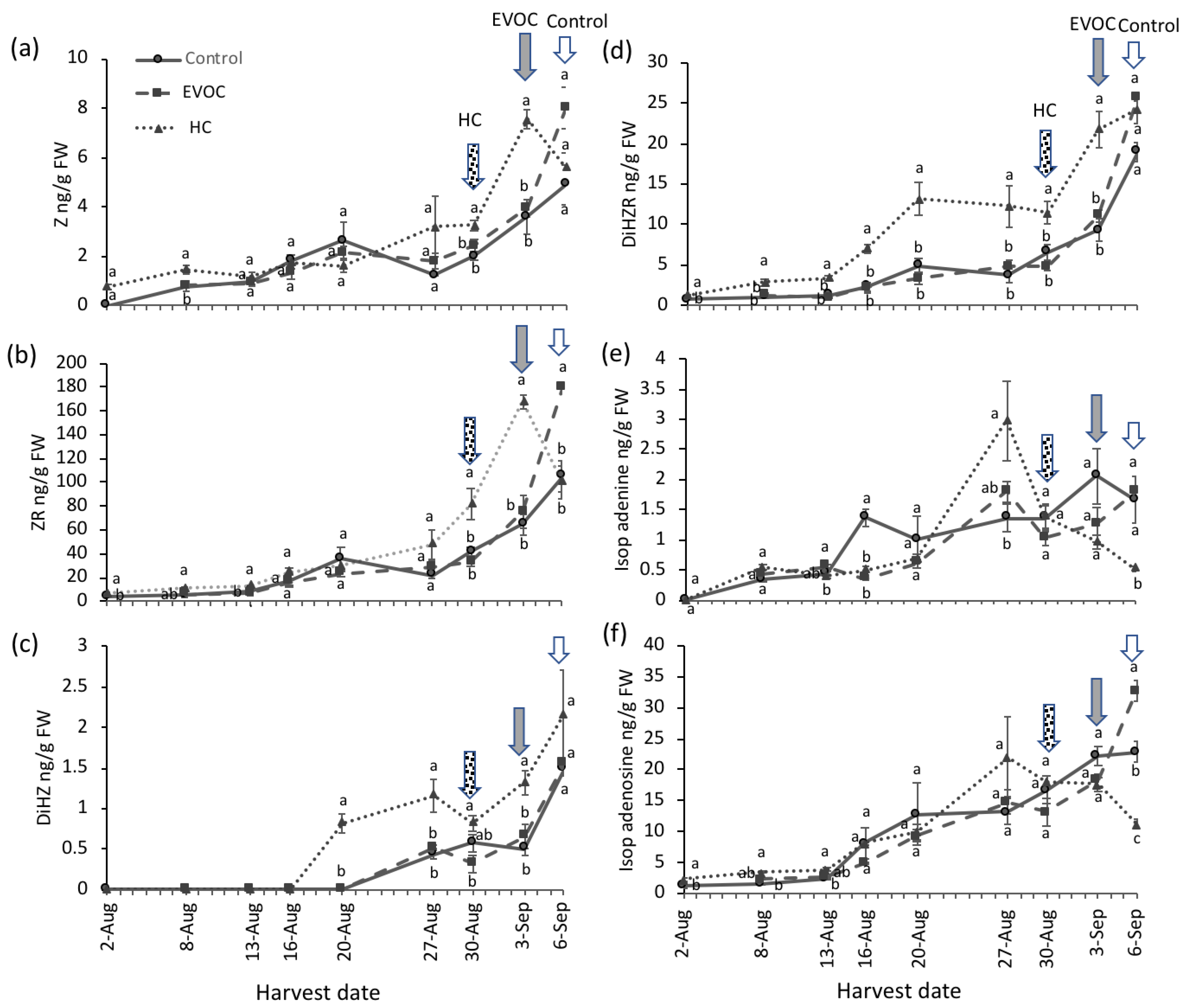
3.1. Bud Break and Hormone Analysis
3.2. Phenological Development
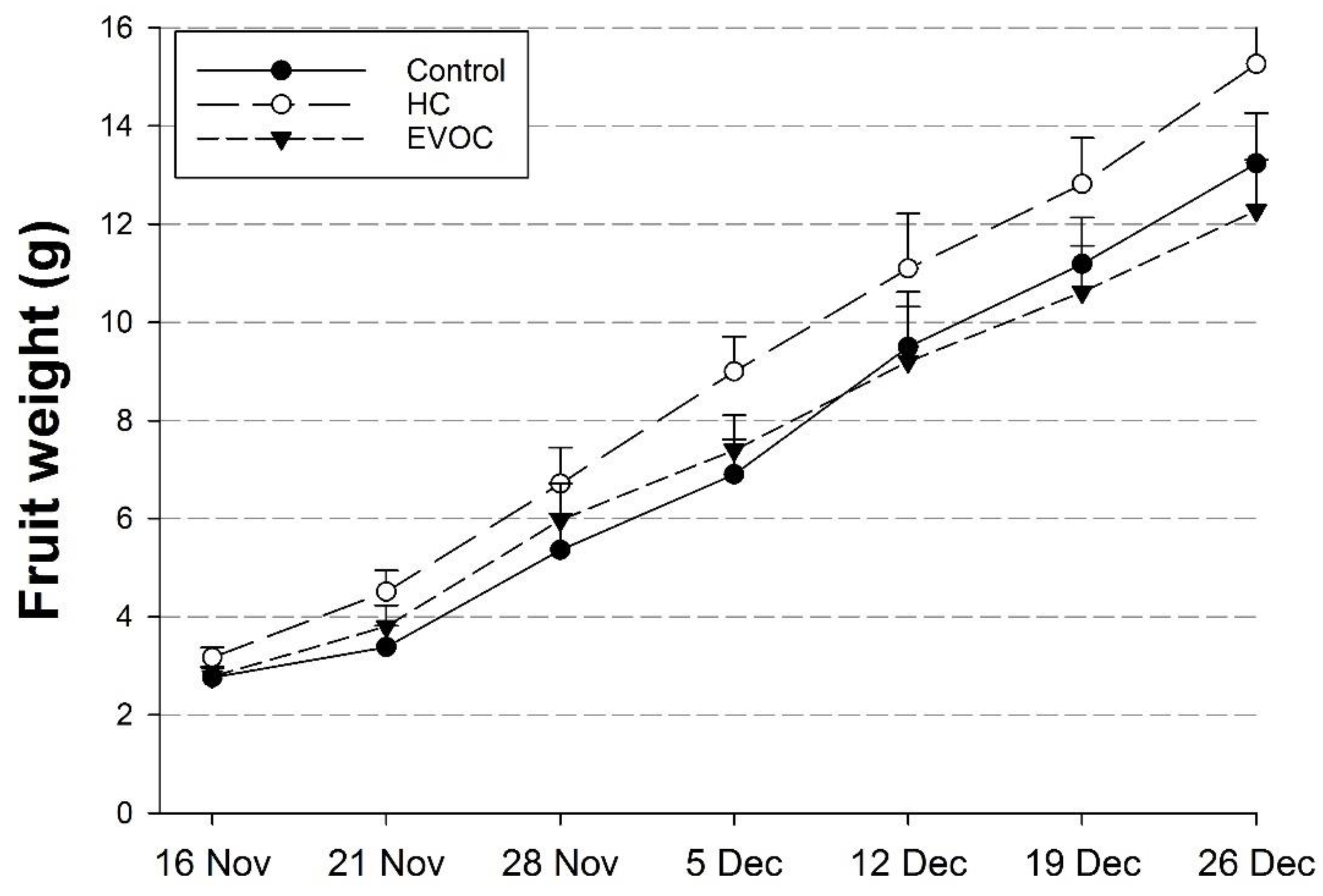
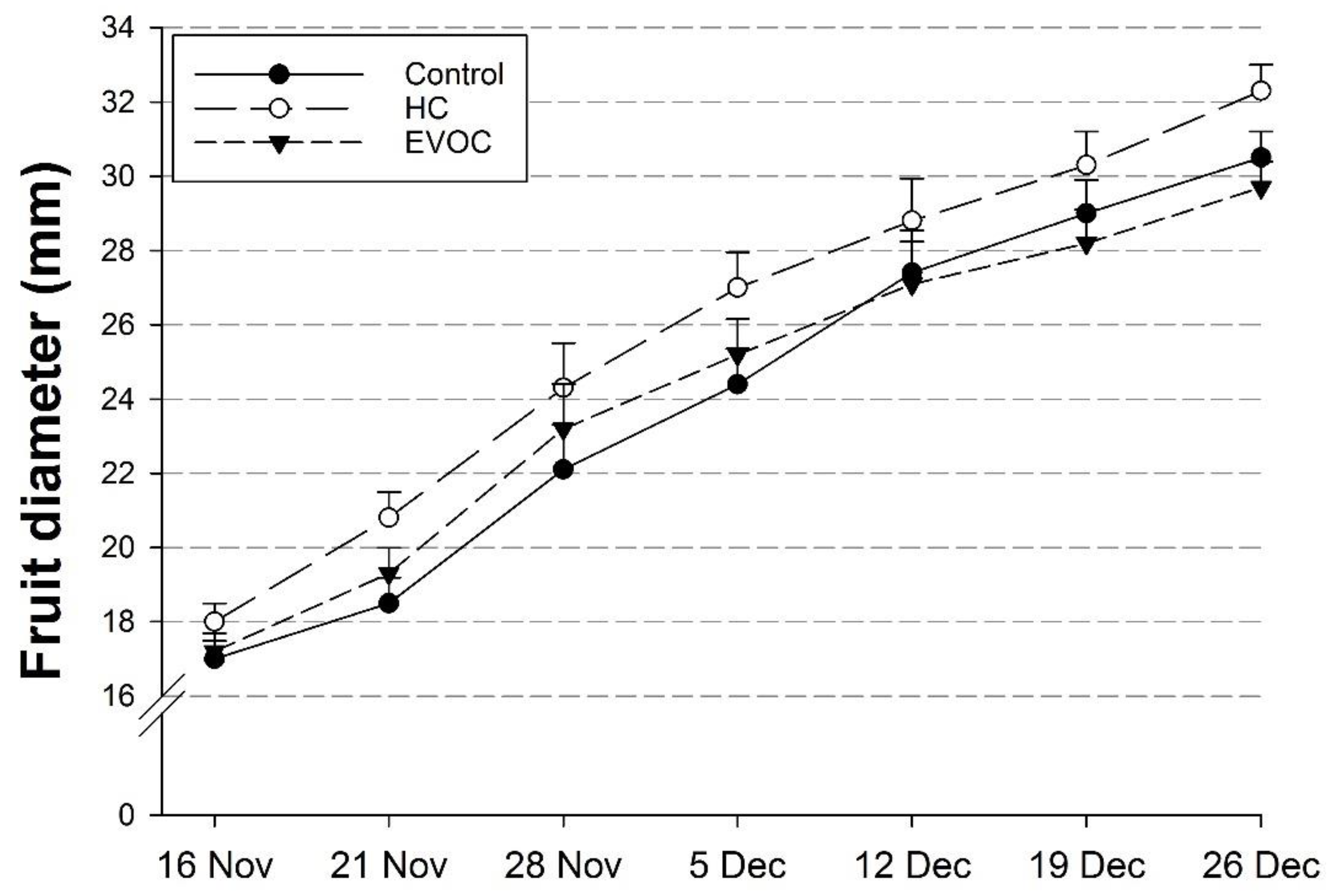
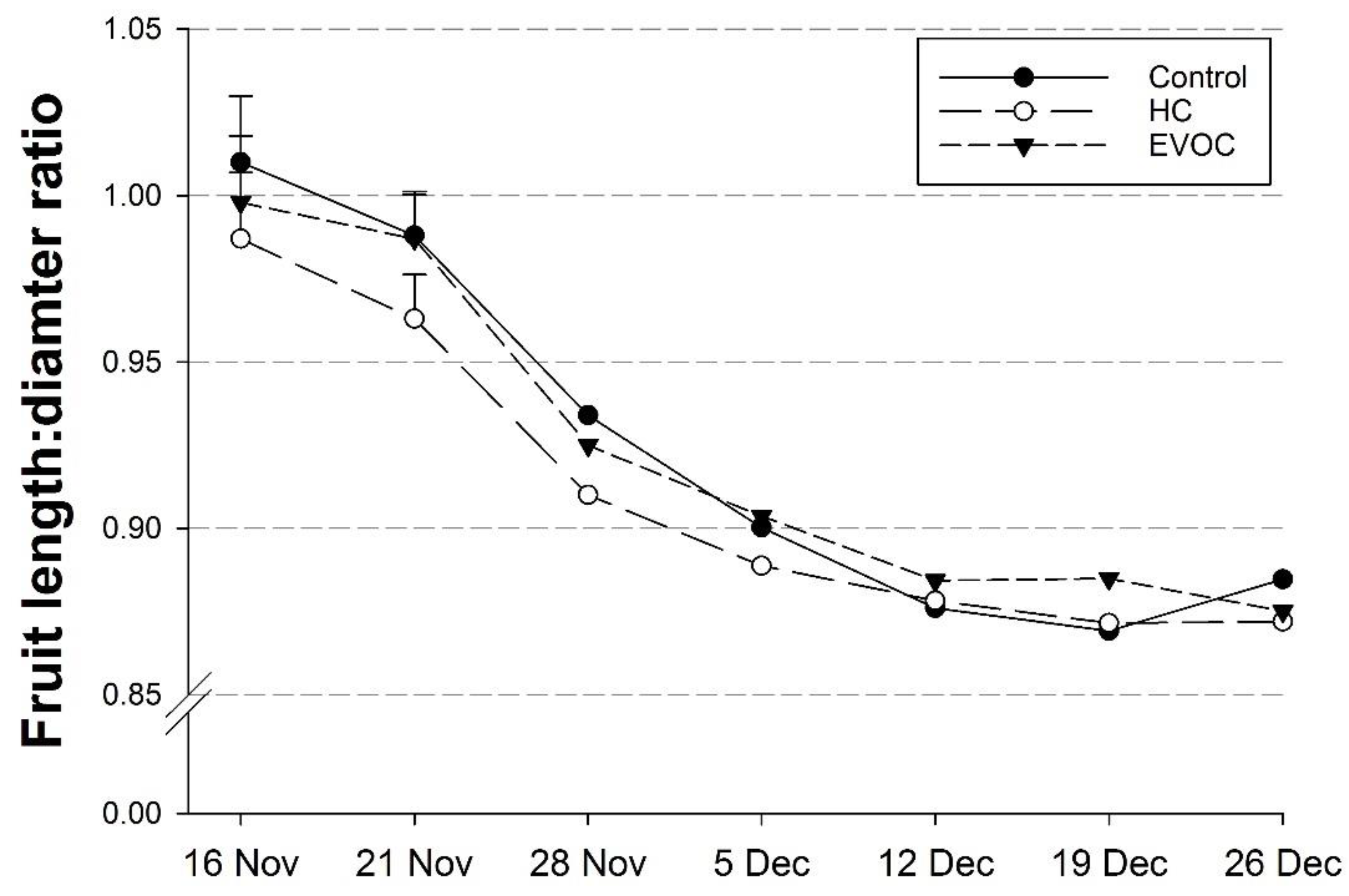
3.3. Fruit Growth
3.4. Harvest Packout and Fruit Quality
4. Discussion
4.1. Bud Break and Hormone Analysis
4.2. Phenological Development and Fruit Growth
4.3. Harvest Packout and Fruit Quality
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Mode | Analyte | Primary Transition | Approx. RT (min) |
|---|---|---|---|
| + | Indole-3-acetic acid | 176 to 130 m/z | 10.3 |
| + | [13C6] Indole-3-acetic acid | 182 to 136 m/z | 10.3 |
| − | Abscisic acid | 263 to 153 m/z | 12.4 |
| − | [D6] Abscisic acid | 269 to 159 m/z | 12.4 |
| + | Zeatin | 220 to 136 m/z | 7.0 |
| + | [D5] Zeatin | 225 to 137 m/z | 7.0 |
| + | Zeatin riboside | 352 to 220 m/z | 7.5 |
| + | [D5] Zeatin riboside | 357 to 225 m/z | 7.5 |
| + | di-hydrozeatin | 222 to 136 m/z | 7.3 |
| + | [D3] di-hydrozeatin | 225 to 136 m/z | 7.3 |
| + | di-hydrozeatin riboside | 354 to 222 m/z | 7.8 |
| + | [D3] di-hydrozeatin riboside | 357 to 225 m/z | 7.8 |
| + | isopentenyl adenine | 204 to 136 m/z | 12.2 |
| + | [D6] isopentenyl adenine | 210 to 137 m/z | 12.2 |
| + | Isopentenyl adenosine | 336 to 204 m/z | 11.8 |
| + | [D6] Isopentenyl adenosine | 342 to 210 m/z | 11.8 |
References
- Saure, M.C. Dormancy release in deciduous fruit trees. Hortic. Rev. 1985, 7, 239–300. [Google Scholar]
- Campoy, J.A.; Ruiz, D.; Egea, J. Dormancy in temperate fruit trees in a global warming context: A review. Sci. Hortic. 2011, 130, 357–372. [Google Scholar] [CrossRef]
- Luedeling, E.; Girvetz, E.H.; Semenov, M.A.; Brown, P.H. Climate change affects winter chill for temperate fruit and nut trees. PLoS ONE 2011, 6, e20155. [Google Scholar] [CrossRef] [PubMed]
- Ionescu, I.A.; Møller, B.L.; Sánchez-Pérez, R. Chemical control of flowering time. J. Exp. Bot. 2016, 68, 369–382. [Google Scholar] [CrossRef] [PubMed]
- Petri, J.L.; Leite, G.B.; Couto, M.; Gabardo, G.C.; Haveroth, F.J. Chemical induction of budbreak: New generation products to replace hydrogen cyanamide. Acta Hortic. 2014, 1042, 159–166. [Google Scholar] [CrossRef]
- Rademacher, W. Plant growth regulators: Backgrounds and uses in plant production. J. Plant Growth Regul. 2015, 34, 845–872. [Google Scholar] [CrossRef]
- Ardiles, M.; Ayala, M. An alternative dormancy-breaking agent to hydrogen cyanamide for sweet cherry (Prunus avium L.) under low chilling accumulation conditions in the Central Valley of Chile. Acta Hortic. 2017, 1161, 423–430. [Google Scholar] [CrossRef]
- Cai, B.; Wang, H.; Liu, T.; Zhuang, W.; Wang, Z.; Qu, S.; Qin, Y. Effects of gibberellins A 4 on budbreak, antioxidant enzymes’ activity and proline content of flower buds in sweet cherry (Prunus avium). Acta Physiol. Plant. 2019, 41, 88. [Google Scholar] [CrossRef]
- Bound, S.A.; Miller, P. Effects of Waiken® on flowering and spring growth in apple. Acta Hortic. 2006, 727, 167–174. [Google Scholar] [CrossRef]
- Bound, S.A.; Miller, P. Manipulation of bud break, flowering and crop development of sweet cherry with the dormancy breaker Waiken®. Acta Hortic. 2016, 1130, 285–291. [Google Scholar] [CrossRef]
- Bound, S.A.; Jones, K.M. Hydrogen cyanamide impacts on flowering, crop load, and fruit quality of red ‘Fuji’ apple (Malus domestica). N. Z. J. Crop. Hortic. Sci. 2004, 32, 227–234. [Google Scholar] [CrossRef]
- Beauvieux, R.; Wenden, B.; Dirlewanger, E. Bud dormancy in perennial fruit tree species: A pivotal role for oxidative cues. Front. Plant Sci. 2018, 9, 657. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Li, Y.; Xin, D.; Chen, W.; Shao, X.; Wang, Y.; Guo, W. RNA-seq-based transcriptome analysis of dormant flower buds of Chinese cherry (Prunus pseudocerasus). Gene 2015, 555, 362–376. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhuo, X.; Zhao, K.; Zheng, T.; Han, Y.; Yuan, C.; Zhang, Q. Transcriptome profiles reveal the crucial roles of hormone and sugar in the bud dormancy of Prunus mume. Sci. Rep. 2018, 8, 5090. [Google Scholar] [CrossRef] [Green Version]
- Wen, L.H.; Zhong, W.J.; Huo, X.M.; Zhuang, W.B.; Ni, Z.J.; Gao, Z.H. Expression analysis of ABA-and GA-related genes during four stages of bud dormancy in Japanese apricot (Prunus mume Sieb. et Zucc). J. Hortic. Sci. Biotechnol. 2016, 91, 362–369. [Google Scholar] [CrossRef]
- Khalil-Ur-Rehman, M.; Dong, Y.; Faheem, M.; Zeng, J.; Wang, W.; Tao, J. Expression profiling of ABA and GA signaling cascades regulating bud dormancy in grape. Sci. Hortic. 2019, 246, 44–50. [Google Scholar] [CrossRef]
- Ionescu, I.A.; López-Ortega, G.; Burow, M.; Bayo-Canha, A.; Junge, A.; Gericke, O.; Møller, B.L.; Sánchez-Pérez, R. Transcriptome and metabolite changes during hydrogen cyanamide-induced floral bud break in sweet cherry. Front. Plant Sci. 2017, 8, 1233. [Google Scholar] [CrossRef]
- Chmielewski, F.; Götz, K.; Homann, T.; Huschek, G.; Rawel, H. Identification of endodormancy release for cherries (Prunus Avium, L.) by abscisic acid and sugars. J. Hortic. 2017, 4, 585. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, C.; Huang, J.; Zhu, L.; Yu, X.; Li, J.; Lou, Y.; Xu, W.; Wang, S.; Ma, C. Hydrogen cyanamide improves endodormancy release and blooming associated with endogenous hormones in ‘Summit’ sweet cherry trees. N. Z. J. Crop. Hortic. Sci. 2017, 45, 14–28. [Google Scholar] [CrossRef]
- El-Yazal, M.A.; El-Yazal, S.A.; Rady, M.M. Exogenous dormancy-breaking substances positively change endogenous phytohormones and amino acids during dormancy release in ‘Anna’ apple trees. Plant Growth Regul. 2014, 72, 211–220. [Google Scholar] [CrossRef]
- Hoad, G.V. Hormonal regulation of fruit-bud formation in fruit trees. Acta Hortic. 1984, 149, 13–24. [Google Scholar] [CrossRef]
- Lenahan, O.M.; Whiting, M.D.; Elfving, D.C. Gibberellic acid inhibits floral bud induction and improves ‘Bing’ sweet cherry fruit quality. HortScience 2006, 41, 654–659. [Google Scholar] [CrossRef] [Green Version]
- Zhuang, W.; Gao, Z.; Wang, L.; Zhong, W.; Ni, Z.; Zhang, Z. Comparative proteomic and transcriptomic approaches to address the active role of GA4 in Japanese apricot flower bud dormancy release. J. Exp. Bot. 2013, 64, 4953–4966. [Google Scholar] [CrossRef] [PubMed]
- Wood, B. Hydrogen cyanamide advances pecan budbreak and harvesting. J. Am. Soc. Hort. Sci. 1993, 118, 690–693. [Google Scholar] [CrossRef]
- Carreño, J.; Faraj, S.; Martínez, A. The effects of hydrogen cyanamide on budburst and fruit maturity of ‘Thompson Seedless’ grapevine. J. Hort. Sci. Biotechnol. 1999, 74, 426–429. [Google Scholar] [CrossRef]
- George, A.P.; Broadley, R.H.; Nissen, R.J.; Ward, G. Effects of new rest-breaking chemicals on flowering, shoot production and yield of subtropical tree crops. Acta Hortic. 2002, 575, 835–840. [Google Scholar] [CrossRef]
- Godini, A.; Palasciano, M.; Ferrara, G.; Camposeo, S.; Pacifico, A. On the advancement of bud break and fruit ripening induced by hydrogen cyanamide (Dormex®) in sweet cherry: A three-year study. Acta Hortic. 2008, 795, 469–477. [Google Scholar] [CrossRef]
- Raffoa, M.D.; Mañueco, L.; Candan, A.P.; Santagni, A.; Menni, F. Dormancy breaking and advancement of maturity induced by hydrogen cyanamide: A strategy to improve profits in sweet cherry production. Acta Hortic. 2014, 1020, 497–502. [Google Scholar] [CrossRef]
- Erez, A.; Fishman, S.; Linsley-Noakes, G.C.; Allan, P. The dynamic model for rest completion in peach buds. Acta Hortic. 1990, 276, 165–174. [Google Scholar] [CrossRef]
- Bennett, J.P. Temperature and bud rest period. Calif. Agric. 1949, 3, 9–12. [Google Scholar]
- Weinberger, J.H. Chilling requirements of peach varieties. Proc. Am. Soc. Hortic. Sci. 1950, 56, 122–128. [Google Scholar]
- Richardson, E.A.; Seeley, S.D.; Walker, D.R. A model for estimating the completion of rest for Redhaven and Elberta peach trees. HortScience 1974, 9, 331–332. [Google Scholar]
- Hack, H.; Bleiholder, H.; Buhr, L.; Meier, U.; Schnock-Fricke, U.; Weber, E.; Witzenberger, A. Einheitliche codierung der phänologischen entwicklungsstadien mono- und dikotyler pflanzen—Erweiterte BBCH-skala, allgemein. Nachr. Dtsch. Pflanzenschutzd. 1992, 44, 265–270. [Google Scholar]
- Meier, U. Growth Stages of Mono- and Dicotyledonous Plants: BBCH Monograph; Federal Biological Research Centre for Agriculture and Forestry: Berlin, Germany, 2001. [Google Scholar]
- Großkinsky, D.K.; Albacete, A.; Jammer, A.; Krbez, P.; van der Graaff, E.; Pfeifhofer, H.; Roitsch, T. A rapid phytohormone and phytoalexin screening method for physiological phenotyping. Mol. Plant 2014, 7, 1053–1056. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anonymous. Export packing. In Cherry Export Manual and Biosecurity Management Programme—Production Guide; Cherry Growers Australia Inc.: Tasmania, Australia, 2016. [Google Scholar]
- Murrell, T.D. How does one pH compare to another? Better Crops 2011, 95, 27. [Google Scholar]
- Sakakibara, H. Cytokinins: Activity, biosynthesis, and translocation. Annu. Rev. Plant Biol. 2006, 57, 431–449. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lombard, P.; Cook, N.; Bellstedt, D. Endogenous cytokinin levels of table grape vines during spring budburst as influenced by hydrogen cyanamide application and pruning. Scientia Hortic. 2006, 109, 92–96. [Google Scholar] [CrossRef]
- Upreti, K.K.; Reddy, Y.T.N.; Prasad, S.R.S.; Bindu, G.V.; Jayaram, H.L.; Rajan, S. Hormonal changes in response to paclobutrazol induced early flowering in mango. Scientia Hortic. 2013, 150, 414–418. [Google Scholar] [CrossRef]
- Zheng, C.; Acheampong, A.k.; Shi, Z.; Halaly, T.; Kamiya, Y.; Ophir, R.; Galbraith, D.W.; Or, E. Distinct gibberellin functions during and after grapevine bud dormancy release. J. Exp. Bot. 2018, 69, 1635–1648. [Google Scholar] [CrossRef] [Green Version]
- Brunt, C.; Darbyshire, R.; Nissen, R. Chill and heat requirements: From dormancy to flowering. In Australian Cherry Production Guide 2017; Cherry Growers Australia Inc.: Hobart, Australia, 2017. [Google Scholar]
- Sheard, A.G.; Johnson, S.D.; Cook, N.C. Effect of timing and concentration of rest breaking agents on budburst in ‘Bing’ sweet cherry under conditions of inadequate winter chilling in South Africa. S. Afr. J. Plant Soil 2009, 26, 73–79. [Google Scholar] [CrossRef] [Green Version]
- Measham, P.F.; Quentin, A.G.; MacNair, N. Climate, winter chill, and decision making in sweet cherry production. HortScience 2014, 49, 254–259. [Google Scholar] [CrossRef] [Green Version]
- Measham, P.F.; Bound, S.A.; Gracie, A.J.; Wilson, S.J. Crop load manipulation and fruit cracking in sweet cherry (Prunus avium L.). Adv. Hort. Sci. 2012, 26, 25–31. [Google Scholar]
- Petri, J.L. Interrupting the winter dormancy of apple trees. BASF Agric. News 1989, 2, 17–20. [Google Scholar]

| Label Recommendation (dBeBB) | Application Time | Accumulated Chill | ||||
|---|---|---|---|---|---|---|
| Date | dBeBB | Portions 1 | Hours 2 | Units 3 | ||
| HC | 30–45 | 28/07/2018 | 40 | 75.8 | 1137 | 1185 |
| EVOC | 20–50 | 02/08/2018 | 35 | 82.1 | 1278 | 1324 |
| Quality Attribute | Domestic | Export Finest |
|---|---|---|
| Skin colour | 3–5 | 3–5 |
| Fruit diameter (mm) | 22+, 24+, 26+, 28+, 30+, 32+ | 26+, 28+, 30+, 32+ |
| Compression firmness (g mm−2) | Minimum 250 | Minimum 300 |
| Stem pull force (g) | Minimum 500 | Minimum 500 |
| Total soluble solids (°Brix) | 16+ | 17+ |
| 6-Sep | 13-Sep | 17-Sep | 20-Sep | 24-Sep | 27-Sep | 1-Oct | 4-Oct | 8-Oct | 11-Oct | 15-Oct | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| (1) Percentage of buds ≥ open cluster | |||||||||||
| Control | 0 a | 64 a | 96 | 100 | |||||||
| HC | 10 b | 92 b | 95 | 97 | |||||||
| EVOC | 0 a | 80 ab | 98 | 100 | |||||||
| (2) Percentage of buds ≥ white bud (popcorn) | |||||||||||
| Control | - | 0 a | 49 | 81 | 98 | ||||||
| HC | - | 11 b | 62 | 93 | 100 | ||||||
| EVOC | - | 0 a | 63 | 91 | 98 | ||||||
| (3) Percentage of buds ≥ full bloom | |||||||||||
| Control | - | - | - | 9 a | 60 | 85 | 97 | ||||
| HC | - | - | - | 64 b | 95 | 97 | 99 | ||||
| EVOC | - | - | - | 6 a | 77 | 92 | 99 | ||||
| (4) Percentage of buds ≥ petal fall | |||||||||||
| Control | - | - | - | - | 1 | 2 a | 9 a | 59 | 73 a | 94 a | 98 a |
| HC | - | - | - | - | 2 | 6 b | 41 b | 89 | 95 b | 100 c | 100 b |
| EVOC | - | - | - | - | 0 | 2 a | 9 a | 70 | 83 a | 97 b | 100 b |
| (5) Percentage of buds ≥ shuck fall | |||||||||||
| Control | - | - | - | - | - | - | - | - | 9 a | 50 a | 92 a |
| HC | - | - | - | - | - | - | - | - | 28 b | 86 b | 100 b |
| EVOC | - | - | - | - | - | - | - | - | 11 a | 61 a | 98 b |
| Budburst | Open Cluster | White Bud | Full Bloom | Petal Fall | Shuck Fall | |
|---|---|---|---|---|---|---|
| Control | 4.8 a | 5.2 | 7.4 | 8.8 b | 12.7 b | 8.9 b |
| HC | 10.4 b | 6.3 | 4.5 | 6.7 a | 8.6 a | 6.5 a |
| EVOC | 4.5 a | 4.7 | 6.5 | 9.7 b | 12.5 b | 8.0 b |
| A-Grade Mean Fruit Weight (g) | A-Grade Fruit (%) | Reject Fruit (%) | Cracked Fruit (%) | |
|---|---|---|---|---|
| Control | 12.1 b | 78 a | 5 | 14 b |
| HC | 13.9 a | 69 b | 6 | 23 a |
| EVOC | 11.4 b | 81 a | 3 | 10 b |
| Compression Firmness (g mm−2) | Flesh Firmness (kg) | Skin Puncture Force (kg) | Stem Pull Force (g) | |
|---|---|---|---|---|
| Control | 325 a | 0.109 | 0.369 a | 581 a |
| HC | 322 a | 0.106 | 0.362 a | 577 a |
| EVOC | 307 b | 0.105 | 0.346 b | 529 b |
| Dry Matter Content (%) | Total Soluble Solids (°Brix) | Malic Acid Content (g L−1) | Juice pH | |
|---|---|---|---|---|
| Control | 19.0 | 18.0 | 5.2 | 3.93 a |
| HC | 18.6 | 17.2 | 5.5 | 3.87 b |
| EVOC | 18.7 | 17.4 | 5.1 | 3.95 a |
| Compression Firmness (g mm−2) | Flesh Firmness (kg) | Skin Puncture Force (kg) | Stem Pull Force (g) | |
|---|---|---|---|---|
| Control | 359 | 0.112 a | 0.380 a | 471 a |
| HC | 354 | 0.107 b | 0.373 a | 456 ab |
| EVOC | 349 | 0.105 b | 0.356 b | 435 b |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bound, S.A.; Foo, E.; Gélinas-Marion, A.; Nichols, D.S.; Nissen, R. The Impact of Dormancy Breakers on Hormone Profiles, Fruit Growth and Quality in Sweet Cherry. Agriculture 2022, 12, 270. https://doi.org/10.3390/agriculture12020270
Bound SA, Foo E, Gélinas-Marion A, Nichols DS, Nissen R. The Impact of Dormancy Breakers on Hormone Profiles, Fruit Growth and Quality in Sweet Cherry. Agriculture. 2022; 12(2):270. https://doi.org/10.3390/agriculture12020270
Chicago/Turabian StyleBound, Sally A., Eloise Foo, Ariane Gélinas-Marion, David S. Nichols, and Robert Nissen. 2022. "The Impact of Dormancy Breakers on Hormone Profiles, Fruit Growth and Quality in Sweet Cherry" Agriculture 12, no. 2: 270. https://doi.org/10.3390/agriculture12020270
APA StyleBound, S. A., Foo, E., Gélinas-Marion, A., Nichols, D. S., & Nissen, R. (2022). The Impact of Dormancy Breakers on Hormone Profiles, Fruit Growth and Quality in Sweet Cherry. Agriculture, 12(2), 270. https://doi.org/10.3390/agriculture12020270








