Inhibitory Effects of Appropriate Addition of Zero-Valent Iron on NH3 and H2S Emissions during Sewage Sludge Composting
Abstract
:1. Introduction
2. Materials and Methods
2.1. Composting Materials
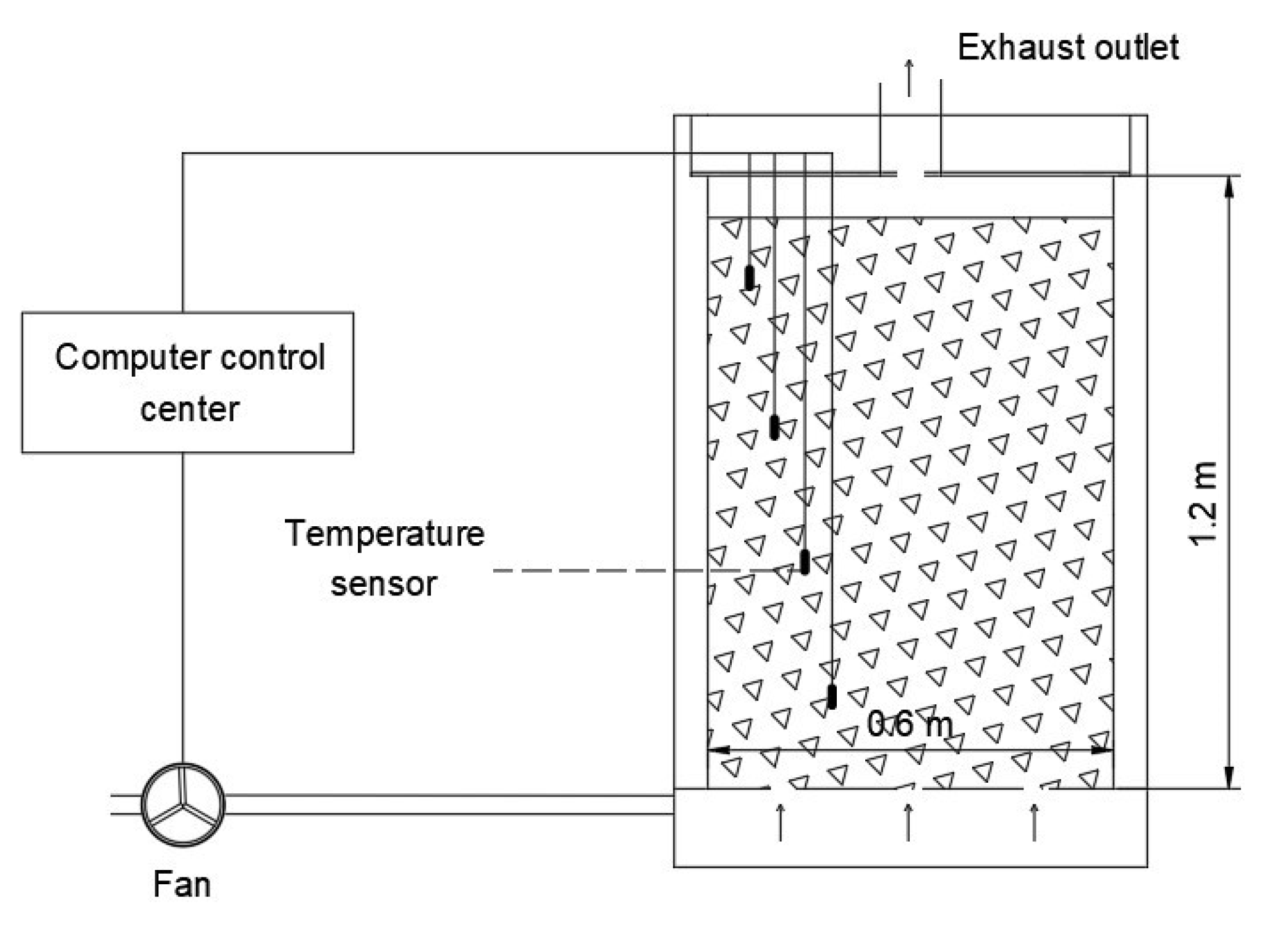
2.2. Composting Method and Device
2.3. Analytical Methods
2.4. Statistical Analysis
3. Results and Discussion
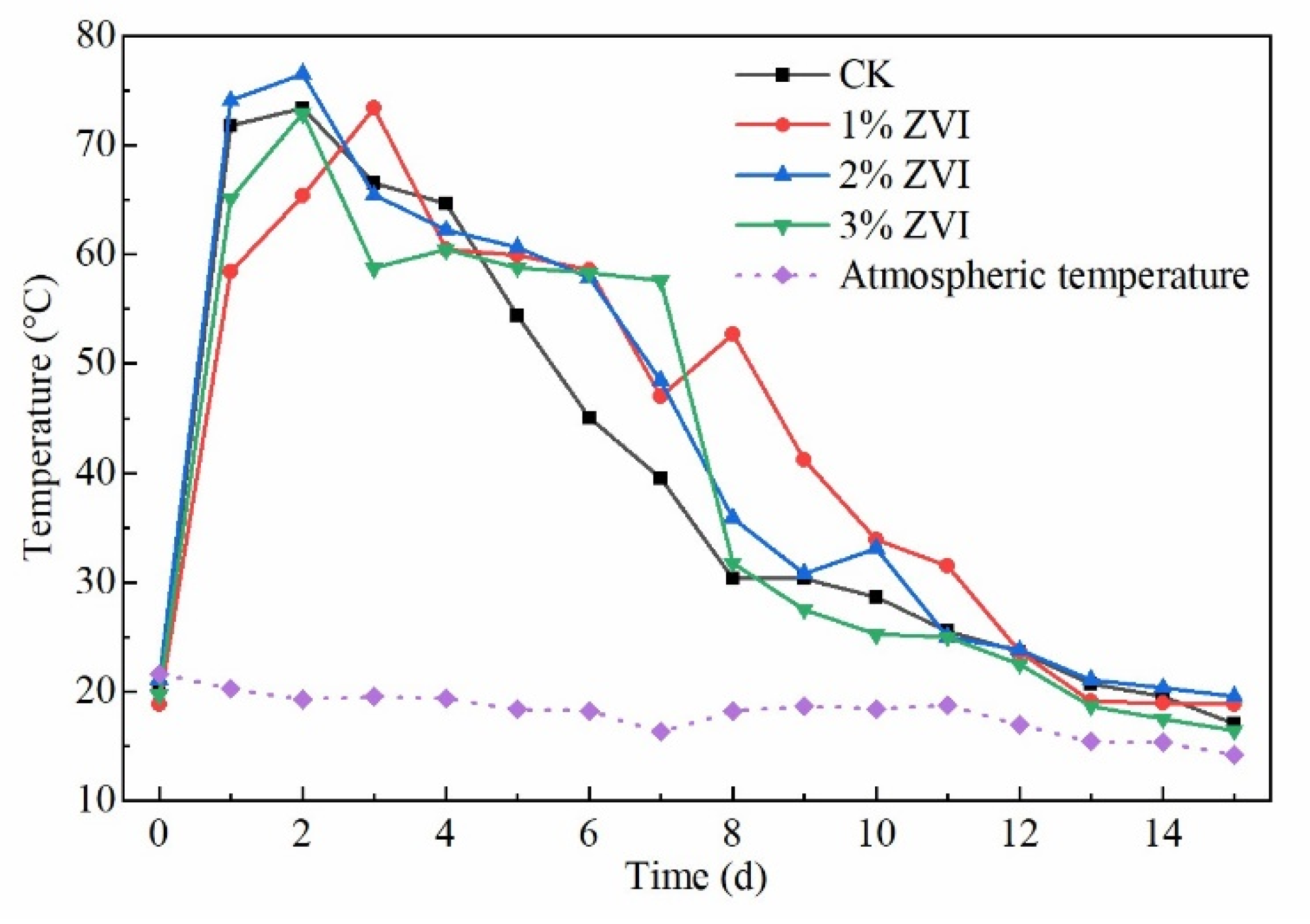
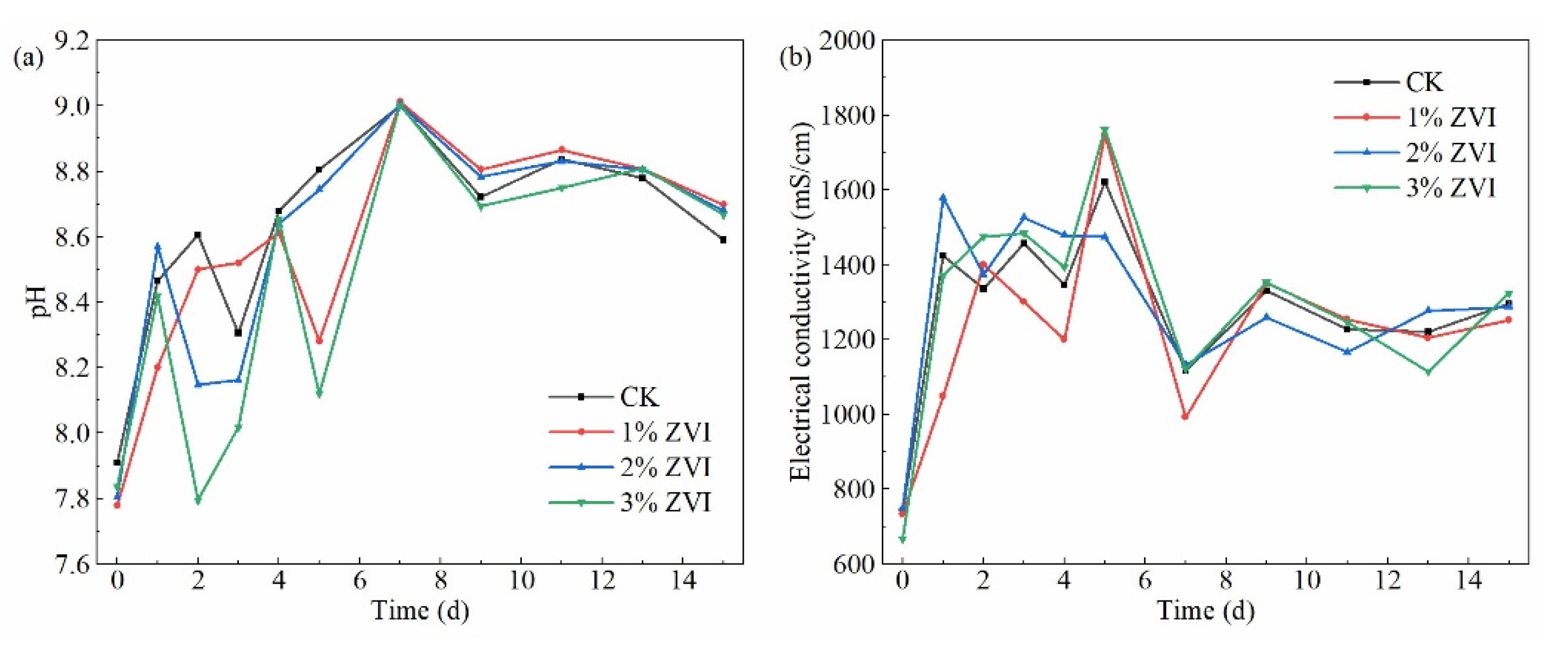
3.1. Effect of ZVI on the Temperature of the Composting Pile
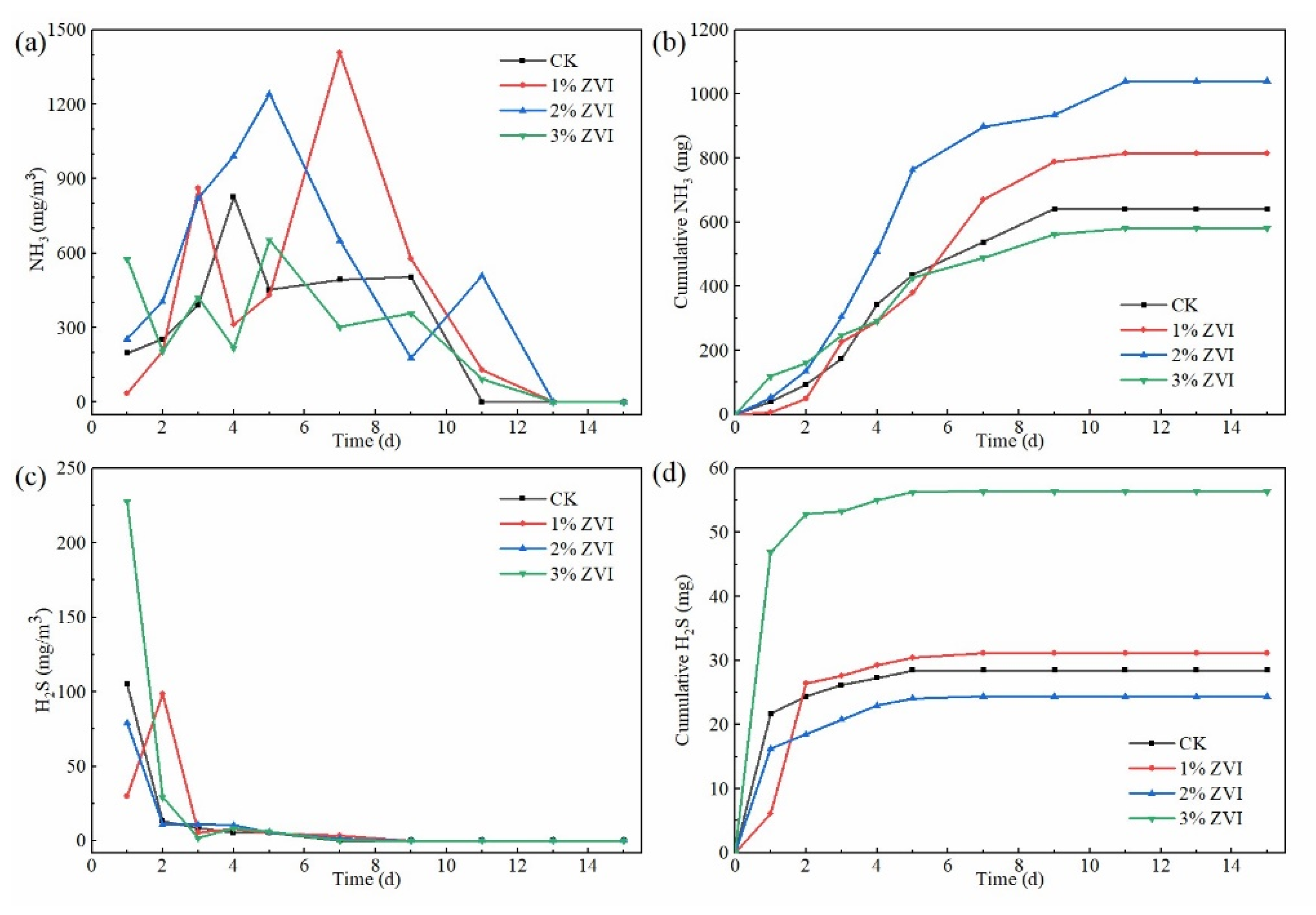
3.2. Effect of ZVI on NH3 Emission during the Composting Process
3.3. Effect of ZVI on H2S Emission during the Composting Process
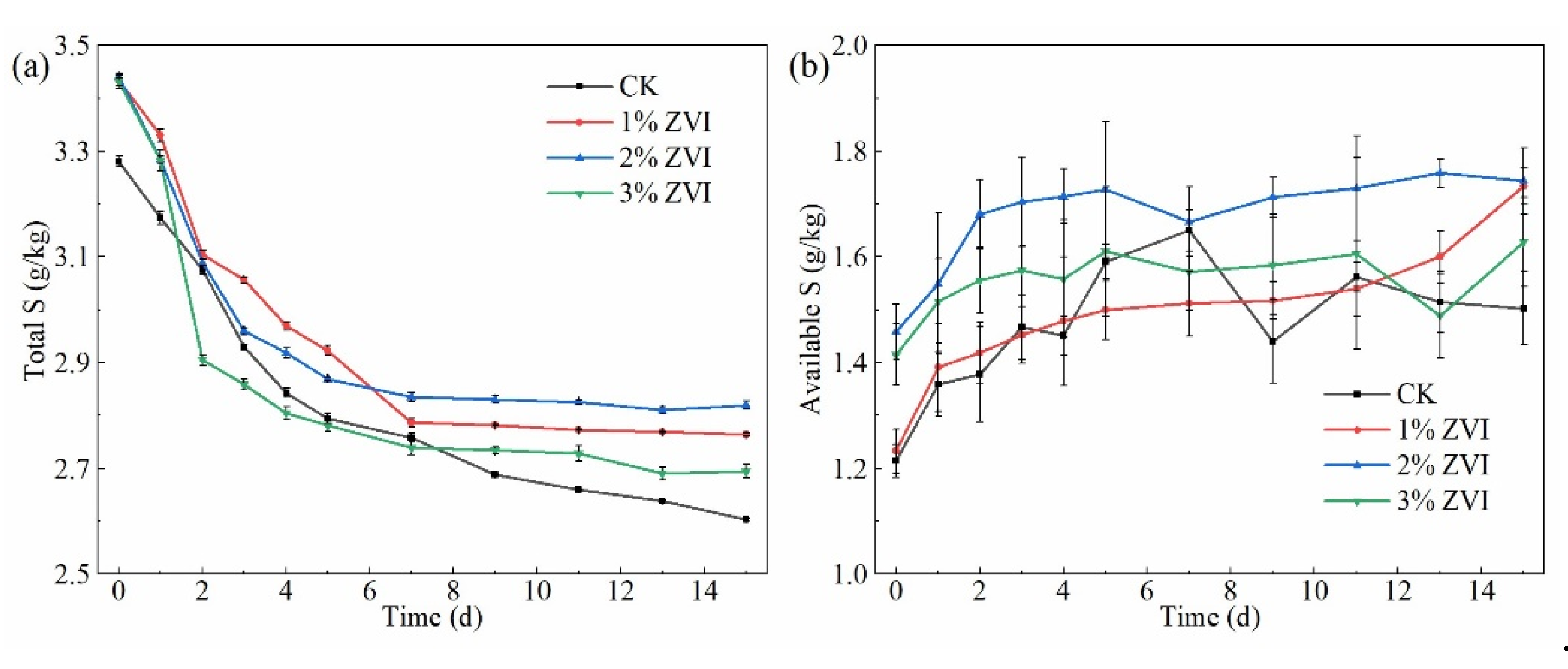
3.4. Effects of Adding ZVI on Total Sulfur and Available Sulfur Content
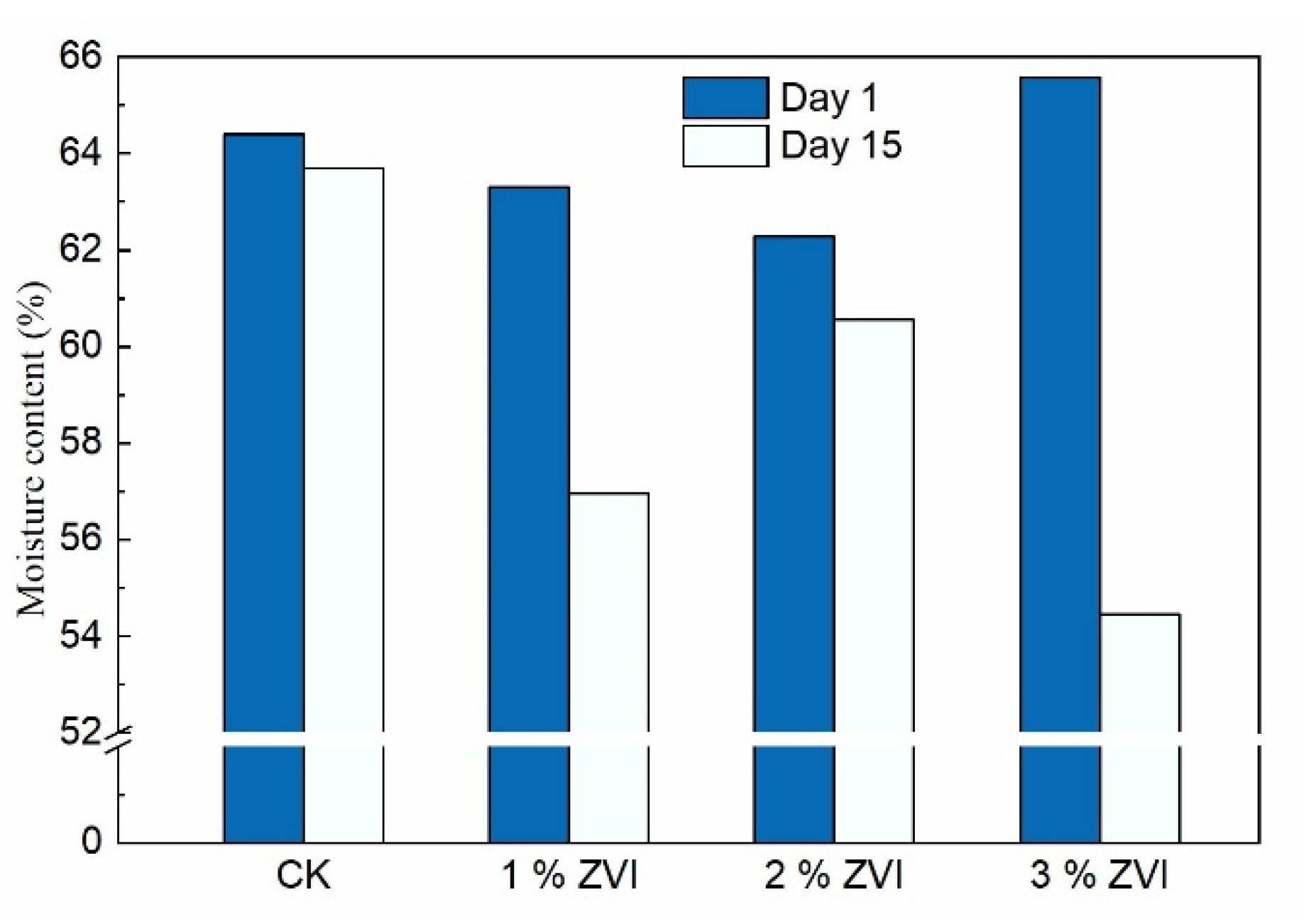
3.5. Effect of Adding ZVI on Dewatering
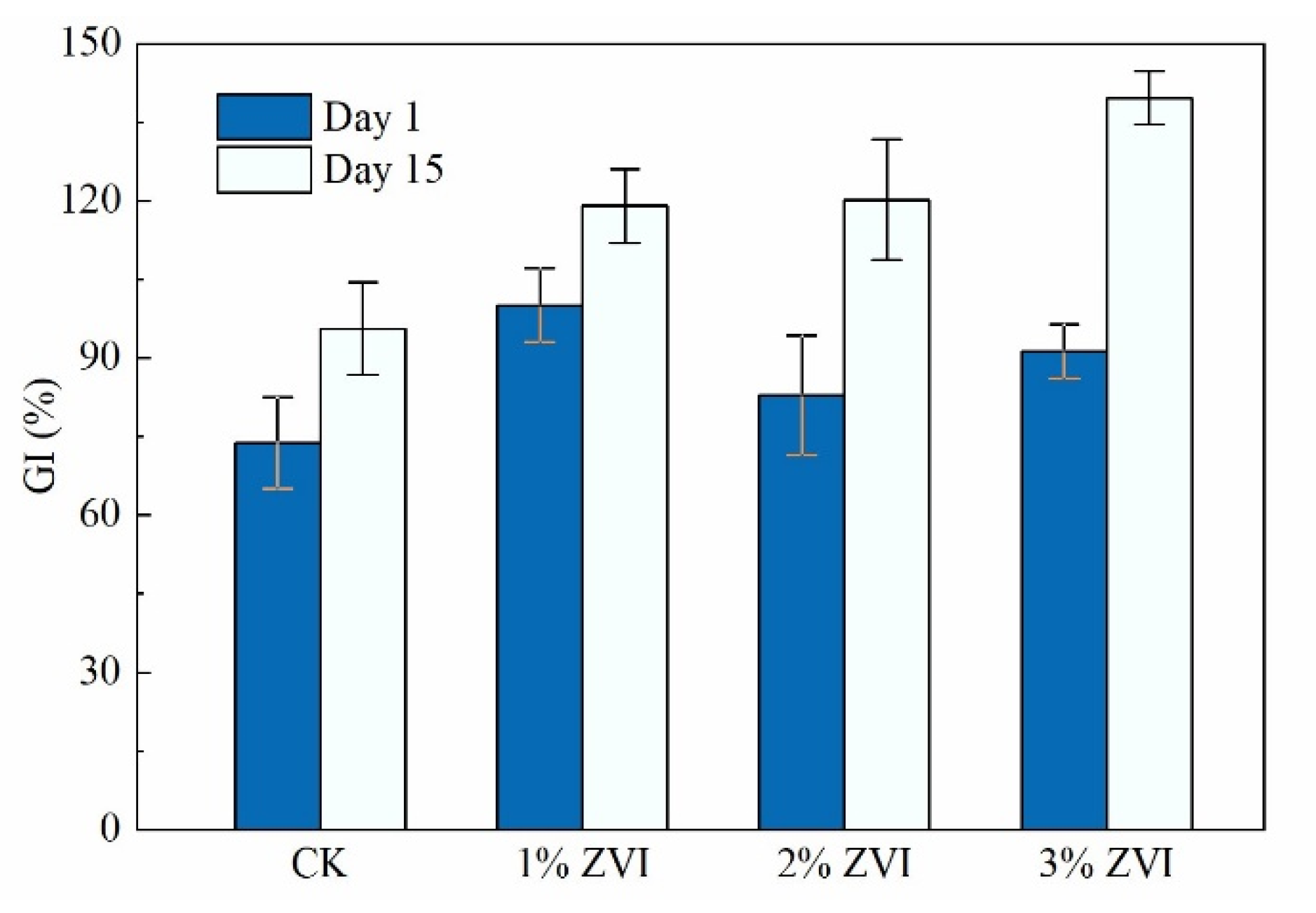
3.6. Effect of Adding ZVI on Maturity
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Shi, M.; Zhao, Y.; Zhu, L.; Song, X.; Tang, Y.; Qi, H.; Cao, H.; Wei, Z. Denitrification during composting: Biochemistry, implication and perspective. Int. Biodeterior. Biodegrad. 2020, 153, 105043. [Google Scholar] [CrossRef]
- Li, W.; Wu, C.; Wang, K.; Meng, L.; Lv, L. Nitrogen loss reduction by adding sucrose and beet pulp in sewage sludge composting. Int. Biodeterior. Biodegrad. 2017, 124, 297–303. [Google Scholar] [CrossRef]
- He, P.; Du, W.; Xu, X.; Zhang, H.; Shao, L.; Lü, F. Effect of biochemical composition on odor emission potential of biowaste during aerobic biodegradation. Sci. Total Environ. 2020, 727, 138285. [Google Scholar] [CrossRef] [PubMed]
- Andraskar, J.; Yadav, S.; Kapley, A. Challenges and control strategies of odor emission from composting operation. Appl. Biochem. Biotechnol. 2021, 193, 2331–2356. [Google Scholar] [CrossRef]
- Duan, Z.; Lu, W.; Mustafa, M.F.; Du, J.; Wen, Y. Odorous gas emissions from sewage sludge composting windrows affected by the turning operation and associated health risks. Sci. Total Environ. 2022, 839, 155996. [Google Scholar] [CrossRef] [PubMed]
- Shan, G.; Li, W.; Gao, Y.; Tan, W.; Xi, B. Additives for reducing nitrogen loss during composting: A review. J. Clean. Prod. 2021, 307, 127308. [Google Scholar] [CrossRef]
- Cui, G.Y.; Bhat, S.A.; Li, W.J.; Ishiguro, Y.; Wei, Y.F.; Li, F.S. H2S, MeSH, and NH3 emissions from activated sludge: An insight towards sludge characteristics and microbial mechanisms. Int. Biodeterior. Biodegrad. 2022, 166, 105331. [Google Scholar] [CrossRef]
- Liu, L.; Chen, T.; Zheng, G.; Gao, D.; Chen, J.; Zhang, J.; Lin, X.; Huang, Z. Odor production and treatment technologies in sewage sludge composting plant. China Water Wastewater 2010, 26, 120–124. (In Chinese) [Google Scholar] [CrossRef]
- Nie, E.; Gao, D.; Zheng, G. Effects of lactic acid on modulating the ammonia emissions in co-composts of poultry litter with slaughter sludge. Bioresour. Technol. 2020, 315, 123812. [Google Scholar] [CrossRef] [PubMed]
- Schiavon, M.; Martini, L.M.; Corra, C.; Scapinello, M.; Coller, G.; Tosi, P.; Ragazzi, M. Characterisation of volatile organic compounds (VOCs) released by the composting of different waste matrices. Environ. Pollut. 2017, 231, 845–853. [Google Scholar] [CrossRef]
- Shou, Z.; Zhu, N.; Yuan, H.; Dai, X.; Shen, Y. Buffering phosphate mitigates ammonia emission in sewage sludge composting: Enhanced organics removal coupled with microbial ammonium assimilation. J. Clean. Prod. 2019, 227, 189–198. [Google Scholar] [CrossRef]
- González, D.; Colón, J.; Gabriel, D.; Sánchez, A. The effect of the composting time on the gaseous emissions and the compost stability in a full-scale sewage sludge composting plant. Sci. Total Environ. 2019, 654, 311–323. [Google Scholar] [CrossRef]
- He, P.; Wei, S.; Shao, L.; Lü, F. Emission potential of volatile sulfur compounds (VSCs) and ammonia from sludge compost with different bio-stability under various oxygen levels. Waste Manag. 2018, 73, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhang, Y.; Ni, B. Zero valent iron simultaneously enhances methane production and sulfate reduction in anaerobic granular sludge reactors. Water Res. 2015, 75, 292–300. [Google Scholar] [CrossRef]
- Zhang, L.; Zheng, X.; She, Z.; Yang, J.; Xie, J. Inhibition effect of FeCl3 and AlCl3 on H2S from sludge mesotherm anaerobic digestion system. Chin. J. Environ. Eng. 2015, 9, 5907–5914. (In Chinese) [Google Scholar] [CrossRef]
- Zhao, Y.; Ren, N.; Wang, A.; Liu, Y. The influence of Fe elements on sulfate reduction process and the response of microbial community. China Environ. Sci. 2007, 27, 199–203. (In Chinese) [Google Scholar] [CrossRef]
- Jadhav, P.; Khalid, Z.B.; Zularisam, A.W.; Krishnan, S.; Nasrullah, M. The role of iron-based nanoparticles (Fe-NPs) on methanogenesis in anaerobic digestion (AD) performance. Environ. Res. 2022, 204, 112043–112056. [Google Scholar] [CrossRef] [PubMed]
- Shou, Z.; Yuan, H.; Shen, Y.; Liang, J.; Zhu, N.; Gu, L. Mitigating inhibition of undissociated volatile fatty acids (VFAs) for enhanced sludge-rice bran composting with ferric nitrate amendment. Bioresour. Technol. 2017, 244, 672–678. [Google Scholar] [CrossRef] [PubMed]
- Medina, J.; Calabi-Floody, M.; Aponte, H.; Santander, C.; Paneque, M.; Meier, S.; Panettieri, M.; Cornejo, P.; Borie, F.; Knicker, H. Utilization of inorganic nanoparticles and biochar as additives of agricultural waste composting: Effects of end-products on plant growth, C and nutrient stock in soils from a mediterranean region. Agronomy 2021, 11, 767–783. [Google Scholar] [CrossRef]
- Bao, Y.; Guan, L.; Zhou, Q.; Wang, H.; Yan, L. Various sulphur fractions changes during different manure composting. Bioresour. Technol. 2010, 101, 7841–7848. [Google Scholar] [CrossRef] [PubMed]
- Nasini, L.; De Luca, G.; Ricci, A.; Ortolani, F.; Caselli, A.; Massaccesi, L.; Regni, L.; Gigliotti, G.; Proietti, P. Gas emissions during olive mill waste composting under static pile conditions. Int. Biodeterior. Biodegrad. 2016, 107, 70–76. [Google Scholar] [CrossRef]
- Kianirad, M.; Muazardalan, M.; Savaghebi, G.; Farahbakhsh, M.; Mirdamadi, S. Effects of temperature treatment on corn cob composting and reducing of composting time: A comparative study. Waste Manag. Res. 2010, 28, 882–887. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Lin, X.; Li, G.; Guo, R. Effect of FeCl3 and Ca3(PO4)2 on composting maturity and study on synthesis of ternary compound substance. Ecol. Environ. Sci. 2010, 19, 1232–1237. (In Chinese) [Google Scholar] [CrossRef]
- Liu, J.; Li, X.; Liu, F.; Zhang, Y. Research advantages on molecular mechanisms of interfacial electron transfer between iron oxide and microbe. Bull. Mineral. Petrol. Geochem. 2018, 37, 39–47. (In Chinese) [Google Scholar] [CrossRef]
- Luo, J.; Feng, L.; Chen, Y.; Li, X.; Chen, H.; Xiao, N.; Wang, D. Stimulating short-chain fatty acids production from waste activated sludge by nano zero-valent iron. J. Biotechnol. 2014, 187, 98–105. [Google Scholar] [CrossRef]
- Zhang, L.; Hu, Y.; Huang, H.; Ren, L.; Zhang, J.; Yan, B.; Luo, L.; Liu, J.; Gu, S.J. Response of bacterial community to iron oxide nanoparticles during agricultural waste composting and driving factors analysis. Bioresour. Technol. 2022, 345, 126530. [Google Scholar] [CrossRef]
- Zhang, L.; Zhu, Y.; Zhang, J.; Zeng, G.; Dong, H.; Cao, W.; Fang, W.; Cheng, Y.; Wang, Y.; Ning, Q. Impacts of iron oxide nanoparticles on organic matter degradation and microbial enzyme activities during agricultural waste composting. Waste Manag. 2019, 95, 289–297. [Google Scholar] [CrossRef]
- Tang, J.; Xu, G.; Xiao, J.; Ludovico, S.; Li, G. The effect of Fe2(SO4)3 on microbial activity of activated sludge. J. Harbin Inst. Technol. 2012, 44, 1–5. (In Chinese) [Google Scholar] [CrossRef]
- Wu, D.; Zheng, S.; Ding, A.; Sun, G.; Yang, M. Performance of a zero valent iron-based anaerobic system in swine wastewater treatment. J. Hazard. Mater. 2015, 286, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Pagans, E.; Barrena, R.; Font, X.; Sánchez, A. Ammonia emissions from the composting of different organic wastes. dependency on process temperature. Chemosphere 2006, 62, 1534–1542. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lai, J.; Cheng, M.; Huang, R.; Yu, G.; Chong, Y.; Li, Y.; Zhong, Y. Mechanism of ammonium sharp increase during sediments odor control by calcium nitrate addition and an alternative control approach by subsurface injection. Environ. Res. 2020, 190, 109979. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Zeng, Y. Ammonia emission mitigation in food waste composting: A review. Bioresour. Technol. 2018, 248, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Xue, J.; Li, Y.; Chang, R.; Wang, J.; Peng, L. Nitrogen transformation and loss during co-composting of kitchen and garden wastes. Trans. Chin. Soc. Agric. Eng. 2021, 37, 192–197. (In Chinese) [Google Scholar] [CrossRef]
- Wan, L.; Liu, H.; Wang, X. Anaerobic ammonium oxidation coupled to Fe(III) reduction: Discovery, mechanism and application prospects in wastewater treatment. Sci. Total Environ. 2022, 818, 151687. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Hou, L.; Liu, M.; Zheng, Y.; Yin, G.; Lin, X.; Cheng, L.; Li, Y.; Hu, X. Evidence of nitrogen loss from anaerobic ammonium oxidation coupled with ferric iron reduction in an intertidal wetland. Environ. Sci. Technol. 2015, 49, 11560–11568. [Google Scholar] [CrossRef]
- Han, Z.; Qi, F.; Li, R.; Wang, H.; Sun, D. Health impact of odor from on-situ sewage sludge aerobic composting throughout different seasons and during anaerobic digestion with hydrolysis pretreatment. Chemosphere 2020, 249, 126077. [Google Scholar] [CrossRef]
- Zheng, G.; Liu, Y.; Li, Y.; Liu, J.; Yang, J. Inhibitory effects of the addition of KNO3 on volatile sulfur compound emissions during sewage sludge composting. Bioengineering 2022, 9, 258–269. [Google Scholar] [CrossRef]
- Zang, B.; Li, S.; Michel, F.; Li, G.; Luo, Y.; Zhang, D.; Li, Y. Effects of mix ratio, moisture content and aeration rate on sulfur odor emissions during pig manure composting. Waste Manag. 2016, 56, 498–505. [Google Scholar] [CrossRef]
- Zhou, C.; Peng, Y.; Deng, Y.; Yu, M.; Chen, L.; Zhang, L.; Xu, X.; Zhao, F.; Yan, Y.; Wang, G. Increasing sulfate concentration and sedimentary decaying cyanobacteria co-affect organic carbon mineralization in eutrophic lake sediments. Sci. Total Environ. 2022, 806, 151260. [Google Scholar] [CrossRef]
- Islam, S.; Redwan, A.; Millerick, K.; Filip, J.; Fan, L.; Yan, W. Effect of copresence of zerovalent iron and sulfate reducing bacteria on reductive dechlorination of trichloroethylene. Environ. Sci. Technol. 2021, 55, 4851–4861. [Google Scholar] [CrossRef]
- Xin, Y.; Yong, K.; Duujong, L.; Ying, F. Bioaugmented sulfate reduction using enriched anaerobic microflora in the presence of zero valent iron. Chemosphere 2008, 73, 1436–1441. [Google Scholar] [CrossRef] [PubMed]
- Choong, Y.Y.; Norli, I.; Abdullah, A.Z.; Yhaya, M.F. Impacts of trace element supplementation on the performance of anaerobic digestion process: A critical review. Bioresour. Technol. 2016, 209, 369–379. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.; Tang, L.; Feng, H.; Zeng, G.; Dong, H.; Zhang, C.; Huang, B.; Deng, Y.; Wang, J.; Zhou, Y. pH-dependent degradation of p-nitrophenol by sulfidated nanoscale zerovalent iron under aerobic or anoxic conditions. J. Hazard. Mater. 2016, 320, 581–590. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.; Li, Z.; Hou, Y.; Wang, A.; Liu, Q.; Huang, C. Effects of different carbon sources on the efficiency of sulfur-oxidizing denitrifying microorganisms. Environ. Res. 2022, 204, 111946. [Google Scholar] [CrossRef]
- Luo, K.; Pang, Y.; Yang, Q.; Wang, D.; Li, X.; Lei, M.; Huang, Q. A critical review of volatile fatty acids produced from waste activated sludge: Enhanced strategies and its applications. Environ. Sci. Pollut. Res. 2019, 26, 13984–13998. [Google Scholar] [CrossRef]
- Robledo-Mahón, T.; Martín, M.A.; Gutiérrez, M.C.; Toledo, M.; González, I.; Aranda, E.; Chica, A.F.; Calvo, C. Sewage sludge composting under semi-permeable film at full-scale: Evaluation of odour emissions and relationships between microbiological activities and physico-chemical variables. Environ. Res. 2019, 177, 108624. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Xiao, K.; Zhu, Y.; Sun, M.; Liang, S.; Hu, J.; Hou, H.; Liu, B.; Yang, J. Comparison of different valent iron on anaerobic sludge digestion: Focusing on oxidation reduction potential, dissolved organic nitrogen and microbial community. Front. Environ. Sci. Eng. 2022, 16, 80–92. [Google Scholar] [CrossRef]
- Ge, M.; Shen, Y.; Ding, J.; Meng, H.; Zhou, H.; Zhou, J.; Cheng, H.; Zhang, X.; Wang, J.; Wang, H.; et al. New insight into the impact of moisture content and pH on dissolved organic matter and microbial dynamics during cattle manure composting. Bioresour. Technol. 2022, 344, 126236. [Google Scholar] [CrossRef]
- Wu, J.; Zhang, A.; Li, G.; Wei, Y.; He, S.; Lin, Z.; Shen, X.; Wang, Q. Effect of different components of single superphosphate on organic matter degradation and maturity during pig manure composting. Sci. Total Environ. 2019, 646, 587–594. [Google Scholar] [CrossRef]
- Awasthi, M.K.; Pandey, A.K.; Bundela, P.S.; Khan, J. Co-composting of organic fraction of municipal solid waste mixed with different bulking waste: Characterization of physicochemical parameters and microbial enzymatic dynamic. Bioresour. Technol. 2015, 182, 200–207. [Google Scholar] [CrossRef]
- Sun, C.; Wei, Y.; Kou, J.; Han, Z.; Shi, Q.; Liu, L.; Sun, Z. Improve spent mushroom substrate decomposition, bacterial community and mature compost quality by adding cellulase during composting. J. Clean. Prod. 2021, 299, 126928. [Google Scholar] [CrossRef]
- Aslam, D.N.; Horwath, W.; Vandergheynst, J.S. Comparison of several maturity indicators for estimating phytotoxicity in compost-amended soil. Waste Manag. 2008, 28, 2070–2076. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Y.; Liu, J.; Zheng, G.; Yang, J.; Cheng, Y. Inhibitory Effects of Appropriate Addition of Zero-Valent Iron on NH3 and H2S Emissions during Sewage Sludge Composting. Agriculture 2022, 12, 2002. https://doi.org/10.3390/agriculture12122002
Liu Y, Liu J, Zheng G, Yang J, Cheng Y. Inhibitory Effects of Appropriate Addition of Zero-Valent Iron on NH3 and H2S Emissions during Sewage Sludge Composting. Agriculture. 2022; 12(12):2002. https://doi.org/10.3390/agriculture12122002
Chicago/Turabian StyleLiu, Yuan, Junwan Liu, Guodi Zheng, Junxing Yang, and Yuan Cheng. 2022. "Inhibitory Effects of Appropriate Addition of Zero-Valent Iron on NH3 and H2S Emissions during Sewage Sludge Composting" Agriculture 12, no. 12: 2002. https://doi.org/10.3390/agriculture12122002
APA StyleLiu, Y., Liu, J., Zheng, G., Yang, J., & Cheng, Y. (2022). Inhibitory Effects of Appropriate Addition of Zero-Valent Iron on NH3 and H2S Emissions during Sewage Sludge Composting. Agriculture, 12(12), 2002. https://doi.org/10.3390/agriculture12122002





