The Influence of Grain Legume and Tillage Strategies on CO2 and N2O Gas Exchange under Varied Environmental Conditions
Abstract
1. Introduction
2. Materials and Methods
2.1. Soils
2.2. Experimental Design
2.3. Plants
2.4. Climate Treatments
2.5. Measurements of Gas Concentrations: CO2, N2O
2.6. Temperature Campaign and Partial Biomass Harvest
2.7. Data Analyses
3. Results and Discussion
3.1. Ecosystem Respiration, Net Ecosystem Exchange, and Gross Ecosystem Photosynthesis
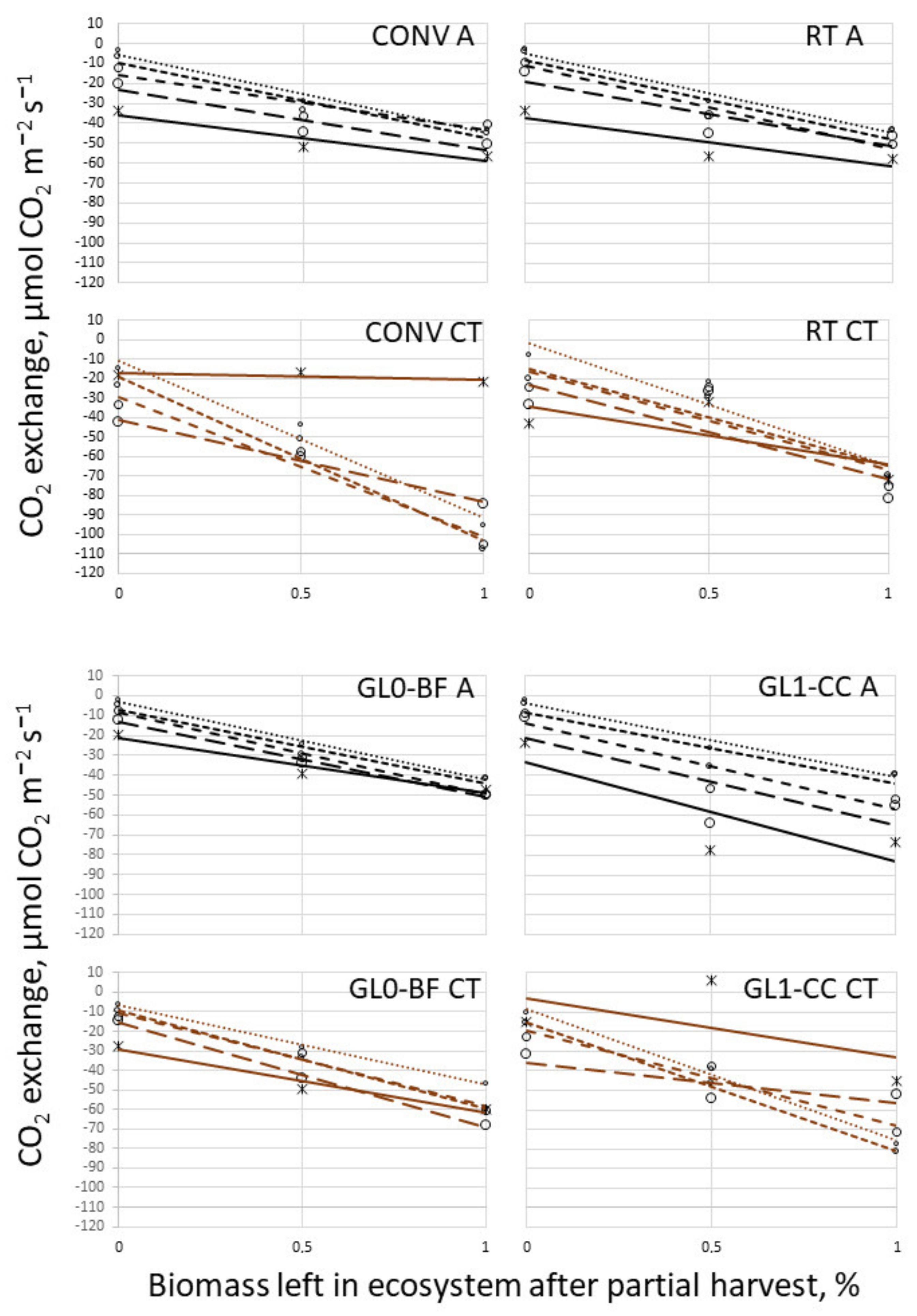
3.2. Respiration
3.3. Temperature Campaign and Biomass Cut
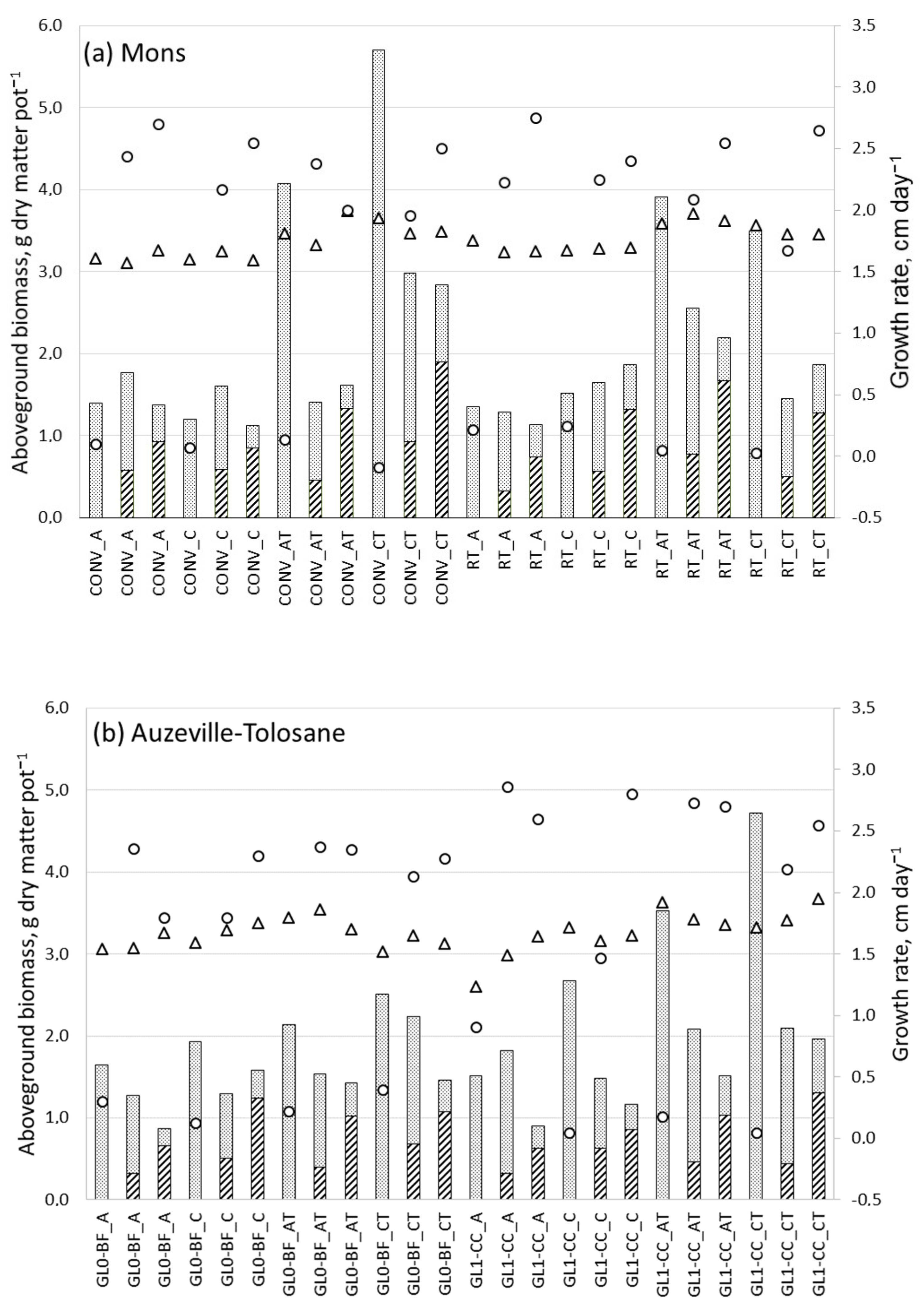
3.4. Barley Total Aboveground Dry Matter Production
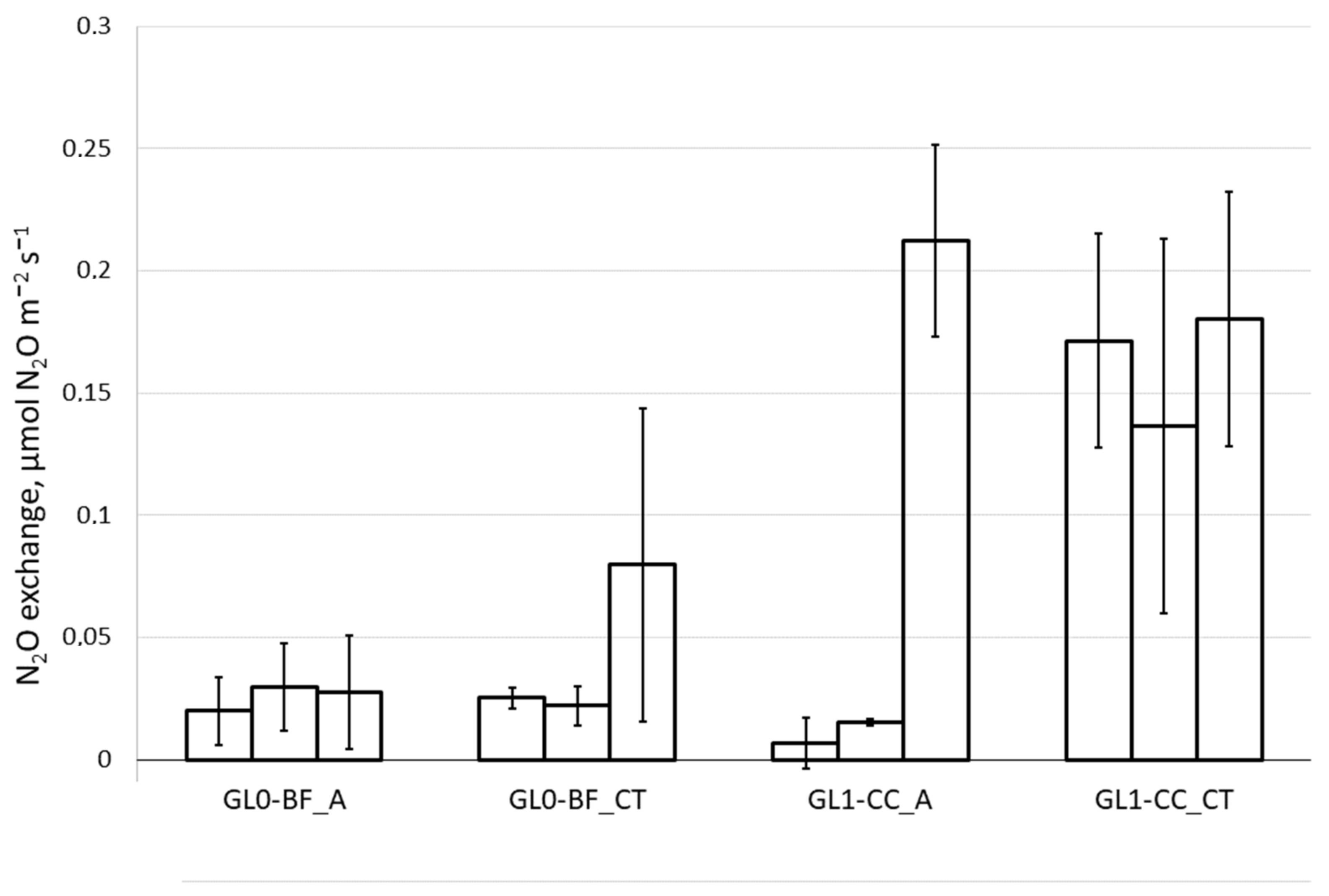
3.5. Nitrous Oxide
3.6. Climate Chambers vs. Field Conditions
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Köchy, M.; Don, A.; van der Molen, M.K.; Freibauer, A. Global distribution of soil organic carbon—Part 2: Certainty of changes related to land use and climate. Soil 2015, 1, 367–380. [Google Scholar] [CrossRef]
- Scharlemann, J.P.; Tanner, E.V.J.; Hiederer, R.; Kapos, V. Global soil carbon: Understanding and managing the largest terrestrial carbon pool. Carbon Manag. 2014, 5, 81–91. [Google Scholar] [CrossRef]
- Grace, P.R.; Colunga-Garcia, M.; Gage, S.H.; Robertson, G.P.; Safir, G.R. The potential impact of agricultural management and climate change on soil organic carbon of the North Central Region of the United States. Ecosystems 2006, 9, 816–827. [Google Scholar] [CrossRef]
- Guo, L.B.; Gifford, R.M. Soil carbon stocks and land use change: A meta analysis. Glob. Chang. Biol. 2002, 8, 345–360. [Google Scholar] [CrossRef]
- Lal, R. Soil carbon management and climate change. Carbon Manag. 2013, 4, 439–462. [Google Scholar] [CrossRef]
- Sanderman, J.; Hengl, T.; Fiske, G.J. Soil carbon debt of 12,000 years of human land use. Proc. Natl. Acad. Sci. USA 2017, 114, 9575–9580. [Google Scholar] [CrossRef]
- Brisson, N.; Gate, P.; Gouache, D.; Charmet, G.; Oury, F.X.; Huard, F. Why are wheat yields stagnating in Europe? A comprehensive data analysis for France. Field Crops Res. 2010, 119, 201–212. [Google Scholar] [CrossRef]
- Lal, R. Soil carbon sequestration impacts on global climate change and food security. Science 2004, 304, 1623–1627. [Google Scholar] [CrossRef]
- Six, J.; Ogle, S.M.; Jay breidt, F.; Conant, R.T.; Mosier, A.R.; Paustian, K. The potential to mitigate global warming with no-tillage management is only realized when practised in the long term. Glob. Chang. Biol. 2004, 10, 155–160. [Google Scholar] [CrossRef]
- Trivedi, P.; Delgado-Baquerizo, M.; Anderson, I.C.; Singh, B.K. Response of Soil Properties and Microbial Communities to Agriculture: Implications for Primary Productivity and Soil Health Indicators. Front. Plant Sci. 2016, 7, 990. [Google Scholar] [CrossRef]
- Altieri, M.A.; Nicholls, C.I.; Henao, A.; Lana, M.A. Agroecology and the design of climate change-resilient farming systems. Agron. Sustain. Dev. 2015, 35, 869–890. [Google Scholar] [CrossRef]
- Chatskikh, D.; Olesen, J.E. Soil tillage enhanced CO2 and N2O emissions from loamy sand soil under spring barley. Soil Tillage Res. 2007, 97, 5–18. [Google Scholar] [CrossRef]
- Jensen, E.S.; Peoples, M.B.; Boddey, R.M.; Gresshoff, P.M.; Henrik, H.N.; Alves, B.J.; Morrison, M.J. Legumes for mitigation of climate change and the provision of feedstock for biofuels and biorefineries. A review. Agron. Sustain. Dev. 2012. [Google Scholar] [CrossRef]
- Peoples, M.; Herridge, D.; Alves, R.; Urquiaga, S.; Boddey, R.; Dakora, F.; Bhattarai, S.; Maskey, S.; Sampet, C.; Rerkasem, B.; et al. The contributions of nitrogen-fixing crop legumes to the productivity of agricultural systems. Symbiosis 2009, 48, 1–17. [Google Scholar] [CrossRef]
- Rustad, L.E.; Huntington, T.G.; Boone, R.D. Controls on soil respiration: Implications for climate change. Biogeochemistry 2000, 48, 1–6. [Google Scholar] [CrossRef]
- Smith, K.A.; Ball, T.; Conen, F.; Dobbie, K.E.; Massheder, J.; Rey, A. Exchange of greenhouse gases between soil and atmosphere: Interactions of soil physical factors and biological processes. Eur. J. Soil Sci. 2018, 69, 10–20. [Google Scholar] [CrossRef]
- Kruse, J.; Simon, J.; Rennenberg, H. Soil Respiration and Soil Organic Matter Decomposition in Response to Climate Change. In Climate Change, Air Pollution and Global Challenges: Understanding and Perspectives from Forest Research; Matyssek, R., Clarke, N., Cudlin, P., Mikkelsen, T.N., Tuovinen, J.-P., Wieser, G., Paoletti, E., Eds.; Elsevier: Amsterdam, The Netherlands, 2013; Volume 13, pp. 131–149. [Google Scholar] [CrossRef]
- Ryan, M.G.; Law, B.E. Interpreting, measuring, and modeling soil respiration. Biogeochemistry 2005, 73, 3–27. [Google Scholar] [CrossRef]
- Xu, M.; Shang, H. Contribution of soil respiration to the global carbon equation. J. Plant Physiol. 2016, 203, 16–28. [Google Scholar] [CrossRef]
- Fang, C.; Moncrieff, J.B. The dependence of soil CO2 efflux on temperature. Soil Biol. Biochem. 2001, 33, 155–165. [Google Scholar] [CrossRef]
- Matschullat, J.; Zurba, K.; Zimmermann, F.; Erasmi, S.; Oertel, C. Greenhouse gas emissions from soils—A review. Chem. Erde Geochem. 2016, 76, 327–352. [Google Scholar] [CrossRef]
- Wiesmeier, M.; Poeplau, C.; Sierra, C.A.; Maier, H.; Frühauf, C.; Hübner, R.; Kühnel, A.; Spörlein, P.; Geuß, U.; Hangen, E.; et al. Projected loss of soil organic carbon in temperate agricultural soils in the 21st century: Effects of climate change and carbon input trends. Sci. Rep. 2016, 6, 32525. [Google Scholar] [CrossRef]
- Smith, P.; Lanigan, G.; Kutsch, W.L.; Buchmann, N.; Eugster, W.; Aubinet, M.; Ceschia, E.; Béziat, P.; Yeluripati, J.B.; Osborne, B.; et al. Measurements necessary for assessing the net ecosystem carbon budget of croplands. Agric. Ecosyst. Environ. 2010, 139, 302–315. [Google Scholar] [CrossRef]
- Cheng, W.; Gershenson, A. Carbon Fluxes in the Rhizosphere. In The Rhizosphere: An Ecological Perspective; Cardon, Z.G., Whitbeck, J.L., Eds.; Academic Press: Cambridge, MA, USA, 2007; pp. 31–56. [Google Scholar] [CrossRef]
- Clivot, H.; Mary, B.; Valé, M.; Cohan, J.P.; Champolivier, L.; Piraux, F.; Laurent, F.; Justes, E. Quantifying in situ and modeling net nitrogen mineralization from soil organic matter in arable cropping systems. Soil Biol. Biochem. 2017, 111, 44–59. [Google Scholar] [CrossRef]
- Krauss, M.; Ruser, R.; Müller, T.; Hansen, S.; Mäder, P.; Gattinger, A. Impact of reduced tillage on greenhouse gas emissions and soil carbon stocks in an organic grass-clover ley-winter wheat cropping sequence. Agric. Ecosyst. Environ. 2017, 239, 324–333. [Google Scholar] [CrossRef]
- Chen, S.; Wang, W.; Xu, W.; Wang, Y.; Wan, H.; Chen, D.; Tang, Z.; Tang, X.; Zhou, G.; Xie, Z.; et al. Plant diversity enhances productivity and soil carbon storage. Proc. Natl. Acad. Sci. USA 2018, 115, 4027–4032. [Google Scholar] [CrossRef]
- Kirkby, C.A.; Richardson, A.E.; Wade, L.J.; Batten, G.D.; Blanchard, C.; Kirkegaard, J.A. Carbon-nutrient stoichiometry to increase soil carbon sequestration. Soil Biol. Biochem. 2013, 60, 77–86. [Google Scholar] [CrossRef]
- Butterbach-Bahl, K.; Baggs, E.M.; Dannenmann, M.; Kiese, R.; Zechmeister-Boltenstern, S. Nitrous oxide emissions from soils: How well do we understand the processes and their controls? Philos. Trans. Biol. Sci. 2013, 368, 1–13. [Google Scholar] [CrossRef]
- Coudrain, V.; Hedde, M.; Chauvat, M.; Maron, P.-A.; Bourgeois, E.; Mary, B.; Léonard, J.; Ekelund, F.; Villenave, C.; Recous, S. Temporal differentiation of soil communities in response to arable crop management strategies. Agric. Ecosyst. Environ. 2016, 225, 12–21. [Google Scholar] [CrossRef]
- Plaza-Bonilla, D.; Nolot, J.-M.; Raffaillac, D.; Justes, E. Cover crops mitigate nitrate leaching in cropping systems including grain legumes: Field evidence and model simulations. Agric. Ecosyst. Environ. 2015, 212, 1–12. [Google Scholar] [CrossRef]
- Marschner, P.; Hatam, Z.; Cavagnaro, T.R. Soil respiration, microbial biomass and nutrient availability after the second amendment are influenced by legacy effects of prior residue addition. Soil Biol. Biochem. 2015, 88, 169–177. [Google Scholar] [CrossRef]
- Reichstein, M.; Falge, E.; Baldocchi, D.; Papale, D.; Aubinet, M.; Berbigier, P.; Bernhofer, C.; Buchmann, N.; Gilmanov, T.; Granier, A.A.; et al. On the separation of net ecosystem exchange into assimilation and ecosystem respiration: Review and improved algorithm. Glob. Chang. Biol. 2005, 11, 1424–1439. [Google Scholar] [CrossRef]
- Selsted, M.B.; Ambus, P.; Michelsen, A.; Van Der Linden, L.; Larsen, K.S.; Pilegaard, K.; Mikkelsen, T.N.; Beier, C. Measurement of carbon dioxide fluxes in a free-air carbon dioxide enrichment experiment using the closed flux chamber technique. Atmos. Environ. 2011, 45, 208–214. [Google Scholar] [CrossRef]
- Plaza-Bonilla, D.; Nolot, J.-M.; Passot, S.; Raffaillac, D.; Justes, E. Grain legume-based rotations managed under conventional tillage need cover crops to mitigate soil organic matter losses. Soil Tillage Res. 2016, 156, 33–43. [Google Scholar] [CrossRef]
- Sauvadet, M.; Lashermes, G.; Alavoine, G.; Recous, S.; Chauvat, M.; Maron, P.A.; Bertrand, I. High carbon use efficiency and low priming effect promote soil C stabilization under reduced tillage. Soil Biol. Biochem. 2018, 123, 64–73. [Google Scholar] [CrossRef]
- Ingvordsen, C.H.; Backes, G.; Lyngkjær, M.F.; Peltonen-Sainio, P.; Jensen, J.D.; Jalli, M.; Jahoor, A.; Rasmussen, M.; Mikkelsen, T.N.; Jørgensen, R.B.; et al. Significant decrease in yield under future climate conditions: Stability and production of 138 spring barley accessions. Eur. J. Agron. 2015, 63, 105–113. [Google Scholar] [CrossRef]
- MétéoFrance.com. Météo France [WWW Document]. Homepage. 2019. Available online: https://www.meteofrance.com/contact (accessed on 4 January 2019).
- Kovats, R.S.; Valentini, R.; Bouwer, L.M.; Georgopoulou, E.; Jacob, D.; Martin, E.; Rounsevell, M.; Soussana, J.-F. Europe. In Climate Change 2014: Impacts, Adaptation, and Vulnerability. Part B: Regional Aspects. Contribution of Working Group II to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Barros, V.R., Field, C.B., Dokken, D.J., Mastrandrea, M.D., Mach, K.J., Bilir, T.E., Chatterjee, M., Ebi, K.L., Estrada, Y.O., Genova, R.C., et al., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2014; pp. 1267–1326. [Google Scholar]
- Clough, T.J.; Rochette, P.; Thomas, S.M.; Pihlatie, M.; Christiansen, J.R.; Thorman, R.E. Global Research Alliance N2O chamber methodology guidelines: Design considerations. J. Environ. Qual. 2020, 49, 1081–1091. [Google Scholar] [CrossRef]
- Gao, X.; Thomas, B.W.; Beck, R.; Thompson, D.J.; Zhao, M.; Willms, W.D.; Hao, X. Long-Term Grazing Alters Soil Trace Gas Fluxes from Grasslands in the Foothills of the Rocky Mountains, Canada. Land Degrad. Dev. 2018, 29, 292–302. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2017. [Google Scholar]
- López-López, G.; Lobo, M.C.; Negre, A.; Colombàs, M.; Rovira, J.M.; Martorell, A.; Reolid, C.; Sastre-Conde, I. Impact of fertilisation practices on soil respiration, as measured by the metabolic index of short-term nitrogen input behaviour. J. Environ. Manag. 2012, 113, 517–526. [Google Scholar] [CrossRef]
- Hagedorn, F.; Hiltbrunner, D.; Streit, K.; Ekblad, A.; Lindahl, B.; Miltner, A.; Handa, T.; Hättenschwiler, S. Nine years of CO2 enrichment at the alpine treeline stimulates soil respiration but does not alter soil microbial communities. Soil Biol. Biochem. 2013, 57, 390–400. [Google Scholar] [CrossRef]
- Linn, D.M.; Doran, J.W. Effect of Water-Filled Pore Space on Carbon Dioxide and Nitrous Oxide Production in Tilled and Nontilled Soils1. Soil Sci. Soc. Am. J. 1984, 48, 1267. [Google Scholar] [CrossRef]
- Castro, H.F.; Classen, A.T.; Austin, E.E.; Norby, R.J.; Schadt, C.W. Soil microbial community responses to multiple experimental climate change drivers. Appl. Environ. Microbiol. 2010, 76, 999–1007. [Google Scholar] [CrossRef] [PubMed]
- Grünzweig, J.M.; Subke, J.-A.; Pumpanen, J.S.; Vogel, J.; Gonzalez-Meler, M.A.; Silva, L.B.C.; Dias-De-Oliveira, E.; Flower, C.E.; Martinez, C.A. Experimental Air Warming of a Stylosanthes capitata, Vogel Dominated Tropical Pasture Affects Soil Respiration and Nitrogen Dynamics. Front. Plant Sci. 2017, 8, 46. [Google Scholar] [CrossRef]
- Shaw, M.R.; Zavaleta, E.S.; Chiariello, N.R. Grassland Responses to Global Environmental Changes Suppressed by Elevated CO2. Science 2002, 298, 1987–1990. [Google Scholar] [CrossRef]
- Leakey, A.D.B.; Ainsworth, E.A.; Bernacchi, C.J.; Rogers, A.; Long, S.P.; Ort, D.R. Elevated CO2 effects on plant carbon, nitrogen, and water relations: Six important lessons from FACE. J. Exp. Bot. 2009, 60, 2859–2876. [Google Scholar] [CrossRef]
- Stagnari, F.; Maggio, A.; Galieni, A.; Pisante, M. Multiple benefits of legumes for agriculture sustainability: An overview. Chem. Biol. Technol. Agric. 2017, 4. [Google Scholar] [CrossRef]
- Kassam, A.; Friedrich, T.; Derpsch, R.; Kienzle, J. Overview of the Worldwide Spread of Conservation Agriculture. Field Actions Sci. Rep. 2015, 8. [Google Scholar]
- Drury, C.F.; McKenney, D.J.; Findlay, W.I. Relationships between denitrification, microbial biomass and indigenous soil properties. Soil Biol. Biochem. 1991, 23, 751–755. [Google Scholar] [CrossRef]
- Giltrap, D.L.; Berben, P.; Palmada, T.; Saggar, S. Understanding and analysing spatial variability of nitrous oxide emissions from a grazed pasture. Ecosyst. Environ. 2014, 186, 1–10. [Google Scholar] [CrossRef]
- Peyrard, C.; Mary, B.; Perrin, P.; Véricel, G.; Gréhan, E.; Justes, E.; Léonard, J. N2O emissions of low input cropping systems as affected by legume and cover crops use. Agric. Ecosyst. Environ. 2016, 224, 145–156. [Google Scholar] [CrossRef]
- Sanz-Cobena, A.; García-Marco, S.; Quemada, M.; Gabriel, J.L.; Almendros, P.; Vallejo, A. Do cover crops enhance N2O, CO2 or CH4 emissions from soil in Mediterranean arable systems? Sci. Total Environ. 2014, 466–467, 164–174. [Google Scholar] [CrossRef]
- Kamp, T.; Steindl, H.; Hantschel, R.E.; Beese, F. Nitrous oxide emissions from a fallow and wheat field as affected by increased soil temperatures. Biol. Fertil. Soils 1998, 3, 307–314. [Google Scholar] [CrossRef]
- Clausen, S.K.; Frenck, G.; Linden, L.G.; Mikkelsen, T.N.; Lunde, C.; Jørgensen, R.B. Effects of single and multifactor treatments with elevated temperature, CO2 and ozone on oilseed rape and barley. J. Agron. Crop Sci. 2011, 197, 442–453. [Google Scholar] [CrossRef]
- Poorter, H.; Fiorani, F.; Pieruschka, R.; Wojciechowski, T.; van der Putten, W.H.; Kleyer, M.; Schurr, U.; Postma, J. Pampered inside, pestered outside? Differences and similarities between plants growing in controlled conditions and in the field. New Phytol. 2016, 212, 838–855. [Google Scholar] [CrossRef]
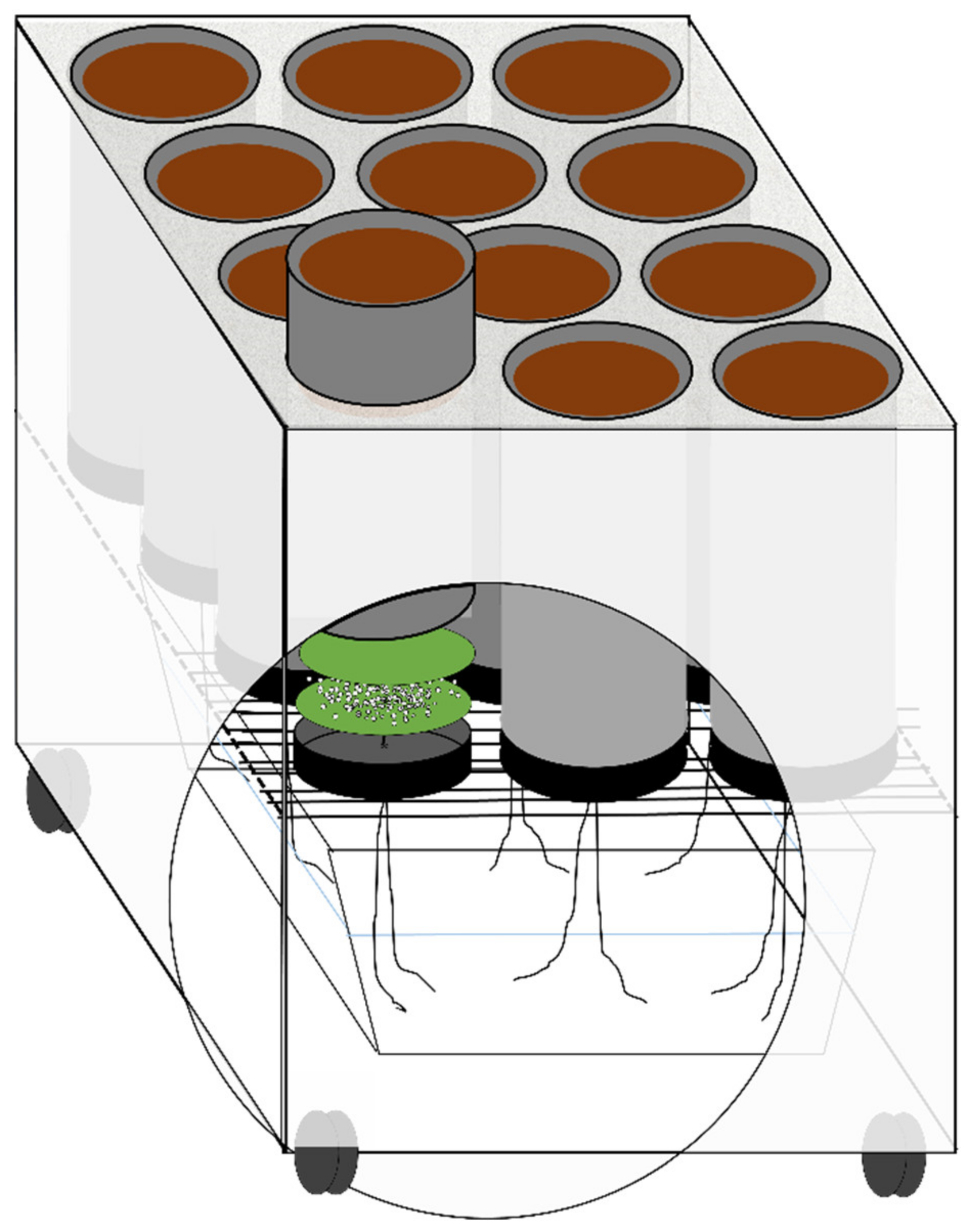


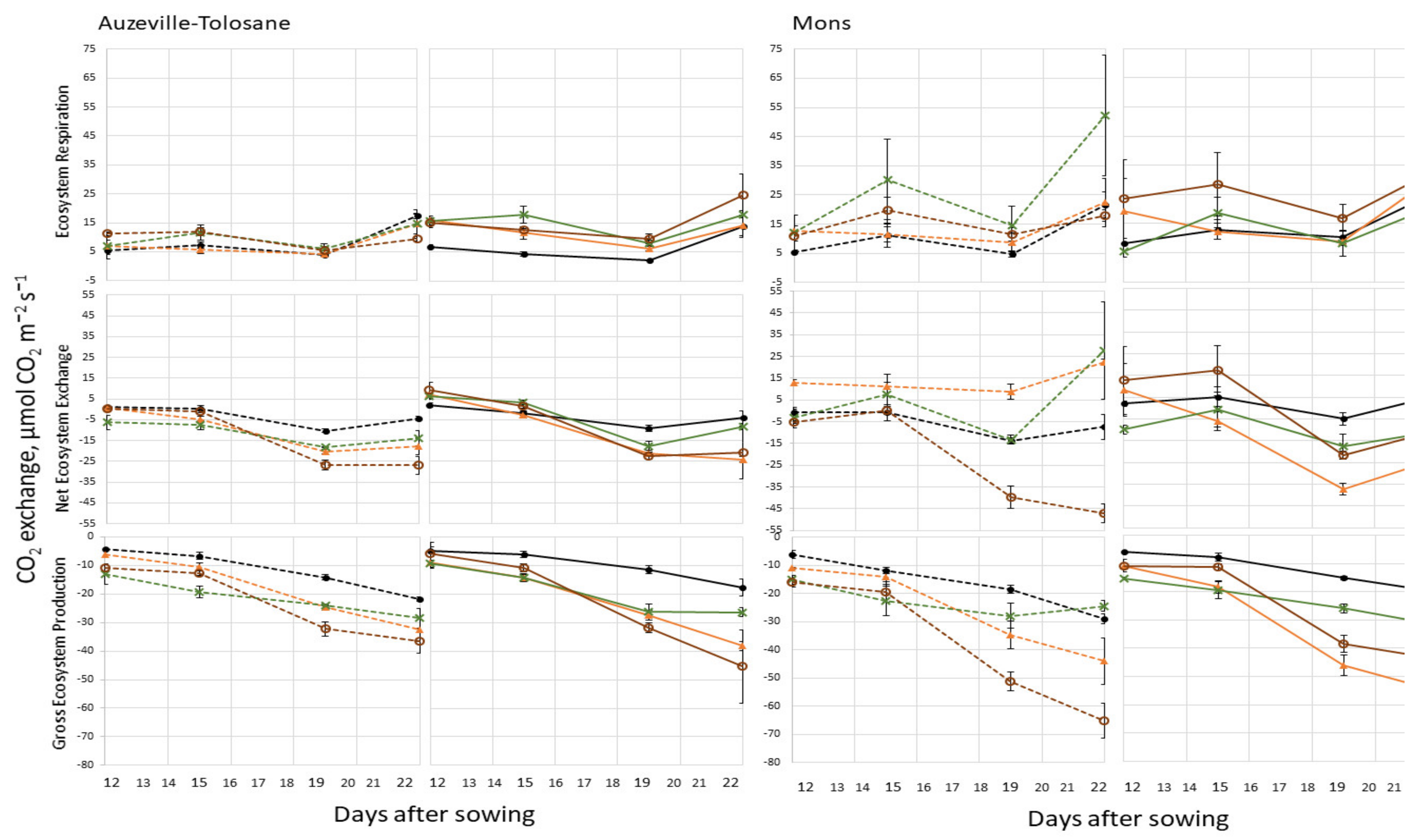
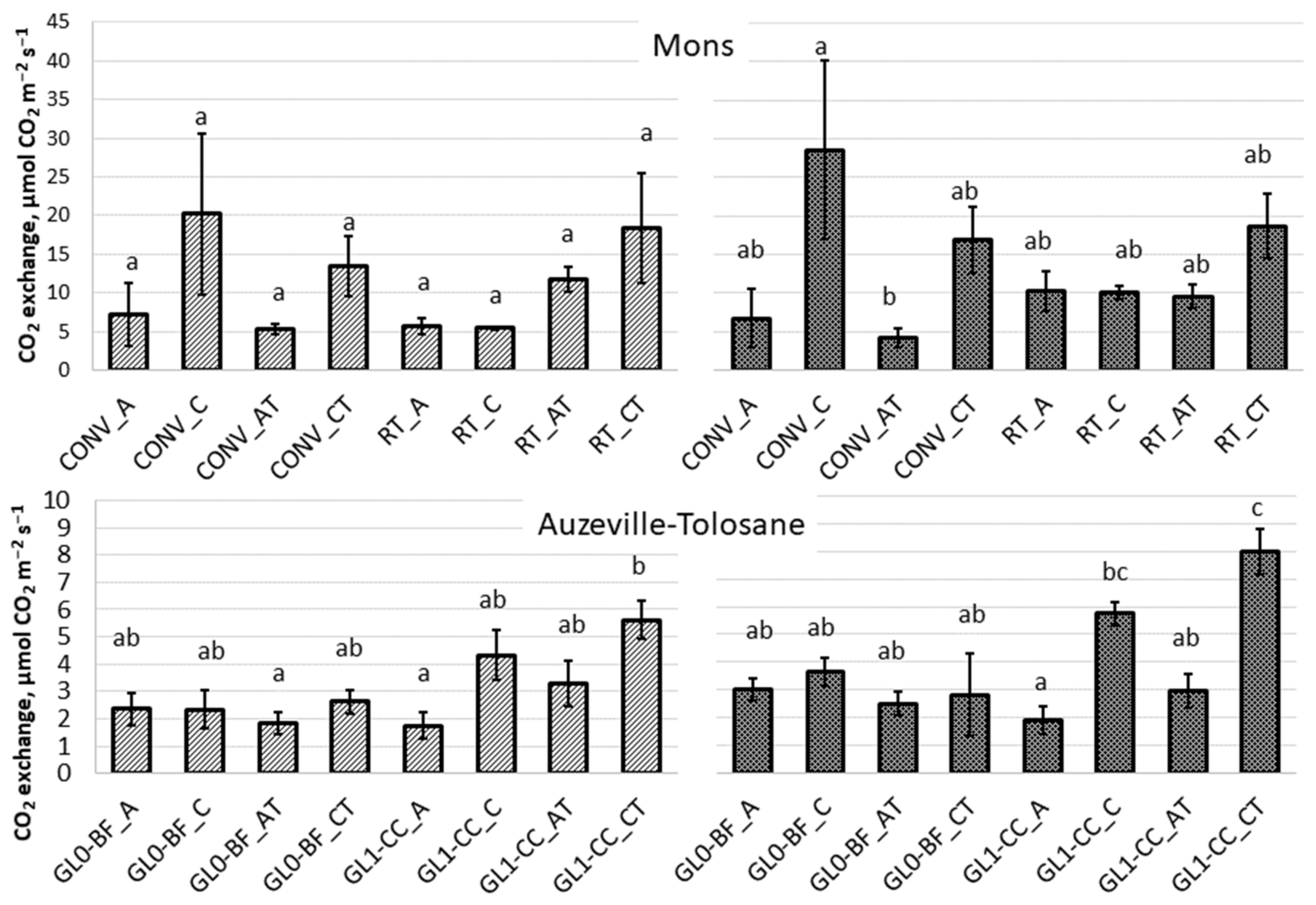
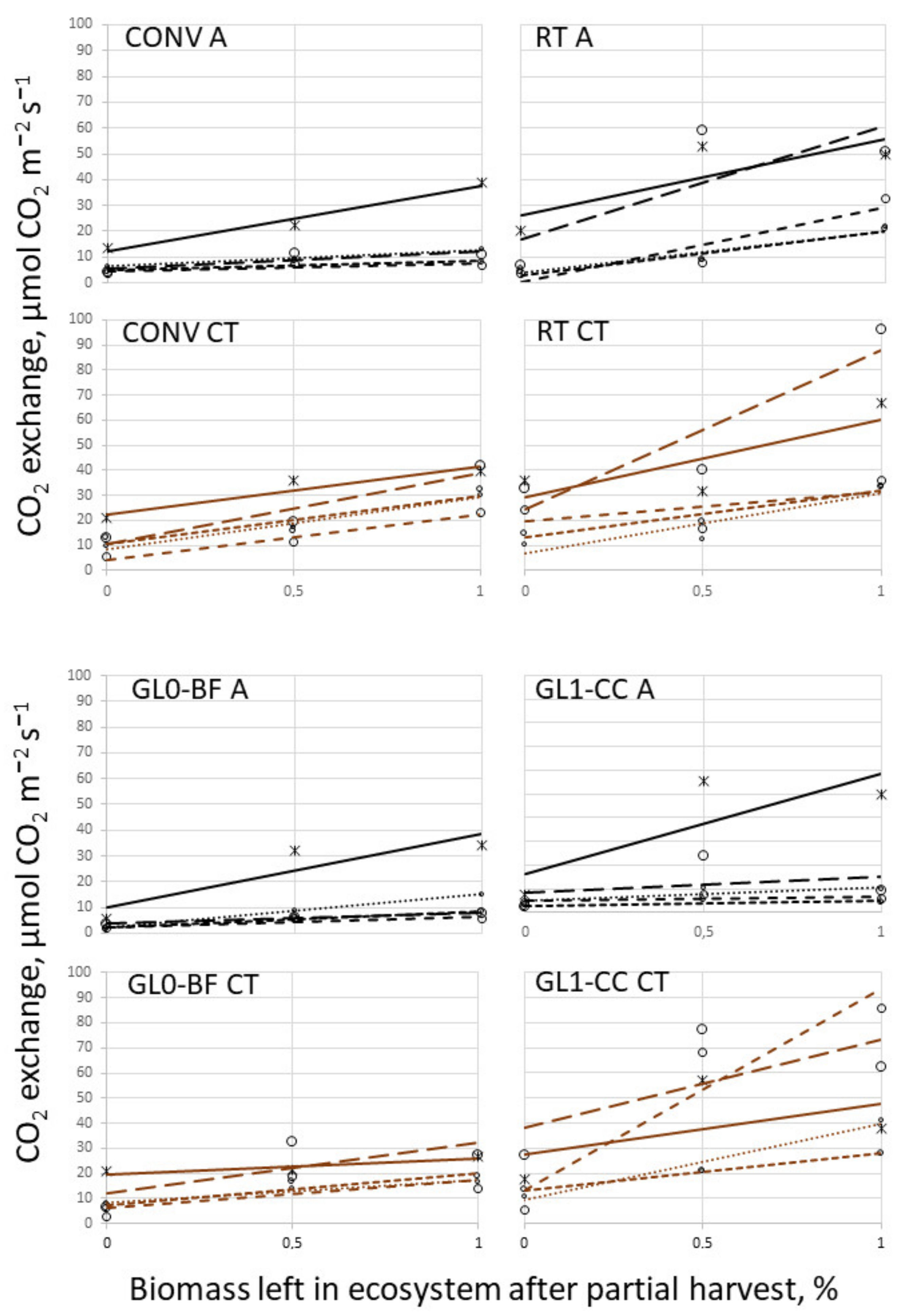
| Texture/Site | Auzeville-Tolosane 1 | Mons 2 | |||
|---|---|---|---|---|---|
| GL1-CC | GL0-BF | Before Exp. | RT | CONV | |
| Clay, % | 26 | 23.7 | 18.7 | ||
| Silt, % | 34.2 | 33 | 74.4 | ||
| Sand, % | 35.5 | 41.5 | 6.9 | ||
| Organic C, g C kg−1 | 8.9 | 7.4 | 10.4 | 12.0 ± 0.1 | 9.8 ± 0.1 |
| Total N, g N kg−1 | 1.05 | 0.82 | 1.0 | 1.4 ± 0.0 | 1.3 ± 0.0 |
| Mons | Auzeville-Tolosane | |||
|---|---|---|---|---|
| Low | High | Low | High | |
| January | 0.6 | 5.5 | 2.4 | 9.5 |
| February | 0.6 | 6.6 | 3 | 11.1 |
| March | 3 | 10.6 | 5 | 14.5 |
| April | 4.5 | 14 | 7.1 | 17 |
| May | 8.2 | 17.9 | 10.9 | 21 |
| June | 10.6 | 20.7 | 14.3 | 25.2 |
| July | 12.5 | 23.4 | 16.5 | 28 |
| August | 12.4 | 23.4 | 16.5 | 27.9 |
| September | 10.1 | 19.6 | 13.4 | 24.6 |
| October | 7.3 | 14.9 | 10.5 | 19.5 |
| November | 3.6 | 9.3 | 5.8 | 13.3 |
| December | 1.3 | 5.9 | 3.2 | 9.9 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hansen, E.M.Ø.; Hauggaard-Nielsen, H.; Justes, E.; Ambus, P.; Mikkelsen, T.N. The Influence of Grain Legume and Tillage Strategies on CO2 and N2O Gas Exchange under Varied Environmental Conditions. Agriculture 2021, 11, 464. https://doi.org/10.3390/agriculture11050464
Hansen EMØ, Hauggaard-Nielsen H, Justes E, Ambus P, Mikkelsen TN. The Influence of Grain Legume and Tillage Strategies on CO2 and N2O Gas Exchange under Varied Environmental Conditions. Agriculture. 2021; 11(5):464. https://doi.org/10.3390/agriculture11050464
Chicago/Turabian StyleHansen, Emilie Marie Øst, Henrik Hauggaard-Nielsen, Eric Justes, Per Ambus, and Teis Nørgaard Mikkelsen. 2021. "The Influence of Grain Legume and Tillage Strategies on CO2 and N2O Gas Exchange under Varied Environmental Conditions" Agriculture 11, no. 5: 464. https://doi.org/10.3390/agriculture11050464
APA StyleHansen, E. M. Ø., Hauggaard-Nielsen, H., Justes, E., Ambus, P., & Mikkelsen, T. N. (2021). The Influence of Grain Legume and Tillage Strategies on CO2 and N2O Gas Exchange under Varied Environmental Conditions. Agriculture, 11(5), 464. https://doi.org/10.3390/agriculture11050464






