Impact of Seaweed Liquid Extract Biostimulant on Growth, Yield, and Chemical Composition of Cucumber (Cucumis sativus)
Abstract
1. Introduction
2. Materials and Methods
2.1. Seaweed Liquid Extract Methods
2.2. Cucumber Cucumis sativus Methods
2.2.1. Soil Analysis
2.2.2. Experimental Design
2.2.3. Tested Parameters
Cucumis sativus Growth Parameters
Cucumis sativus Yield and Fruit Parameters
Chlorophyll Content of Leaves
Dry Matter Content of Leaves and Fruits
Mineral Contents of Leaves and Fruits
2.3. Statistical Analysis
3. Results
3.1. Cucumber Cucumis sativus
3.1.1. Growth
3.1.2. Yield and Chemical Composition
4. Discussion
5. Conclusions
6. Patents
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Layek, J.; Das, A.; Idapuganti, R.G.; Sarkar, D.; Ghosh, A.; Zodape, S.T.; Meena, R.S. Seaweed extract as organic bio-stimulant improves productivity and quality of rice in eastern Himalayas. J. Appl. Phycol. 2018, 30, 547–558. [Google Scholar] [CrossRef]
- Shubha, K.; Mukherjee, A.; Kumari, M.; Tiwari, K.; Meena, V.S. Bio-stimulants: An Approach Towards the Sustainable Vegetable Production. In Agriculturally Important Microbes for Sustainable Agriculture; Springer: Singapore, 2017; pp. 259–277. [Google Scholar] [CrossRef]
- Hassan, S.; Ashour, M.; Soliman, A. Anticancer activity, antioxidant activity, mineral contents, vegetative and yield of Eruca sativa using foliar application of autoclaved cellular extract of Spirulina platensis extract, comparing to N-P-K fertilizers. J. Plant Prod. 2017, 8, 529–536. [Google Scholar] [CrossRef]
- Ashour, M.; El-Shafei, A.A.; Khairy, H.M.; Abd-Elkader, D.Y.; Mattar, M.M.; Alataway, A.; Hassan, S.M. Effect of Pterocladia capillacea seaweed extracts on growth parameters and biochemical constituents of Jew’s Mallow. Agronomy 2020, 10, 420. [Google Scholar] [CrossRef]
- Ashour, M.; Elshobary, M.E.; El-shenody, R.; Kamil, A.; Abomohra, A.E. Evaluation of a native oleaginous marine microalga Nannochloropsis oceanica for dual use in biodiesel production and aquaculture feed. Biomass Bioenergy 2019, 120, 439–447. [Google Scholar] [CrossRef]
- Heneash, A.; Ashour, M.; Mustafa, M. Effect of un-live microalgal diet Nannochloropsis oculata and Arthrospira (Spirulina) platensis, comparing to yeast on population of rotifer, Brachionus plicatilis. Mediterr. Aquac. J. 2015, 7, 48–54. [Google Scholar] [CrossRef]
- Zaki, M.A.; Ashour, M.; Heneash, A.M.M.; Mabrouk, M.M.; Alprol, A.E.; Khairy, H.M.; Nour, A.M.; Mansour, A.T.; Hassanien, H.A.; Gaber, A.; et al. Potential Applications of native cyanobacterium isolate (Arthrospira platensis NIOF17/003) for biodiesel production and utilization of its byproduct in marine rotifer (Brachionus plicatilis) production. Sustainability 2021, 13, 1769. [Google Scholar] [CrossRef]
- El-Shenody, R.A.; Ashour, M.; Ghobara, M.M.E. Evaluating the chemical composition and antioxidant activity of three Egyptian seaweeds Dictyota dichtoma, Turbinaria decurrens, and Laurencia obtuse. Braz. J. Food Technol. 2019, 22, e2018203. [Google Scholar] [CrossRef]
- Sharawy, Z.Z.; Ashour, M.; Abbas, E.; Ashry, O.; Helal, M.; Nazmi, H.; Kelany, M.; Kamel, A.; Hassaan, M.; Rossi, W.J.; et al. Effects of dietary marine microalgae, Tetraselmis suecica on production, gene expression, protein markers and bacterial count of Pacific white shrimp Litopenaeus vannamei. Aquac. Res. 2020, 51, 2216–2228. [Google Scholar] [CrossRef]
- Abo-Taleb, H.; Ashour, M.; El-Shafei, A.; Alataway, A.; Maaty, M.M. Biodiversity of Calanoida Copepoda in different habitats of the North-Western Red Sea (Hurghada Shelf). Water 2020, 12, 656. [Google Scholar] [CrossRef]
- Ashour, M. Bioactive Compounds Extracted from Marine Algae Improve the Growth and Immunity of Plants, Fish and Marine Crustaceans. Egypt Patent Application 2046, 23 December 2019. [Google Scholar]
- Abbas, E.M.; Ali, F.S.; Desouky, M.G.; Ashour, M.; El-Shafei, A.; Maaty, M.M.; Sharawy, Z.Z. Novel Comprehensive Molecular and Ecological Study Introducing Coastal Mud Shrimp (Solenocera Crassicornis) Recorded at the Gulf of Suez, Egypt. J. Mar. Sci. Eng. 2021, 9, 9. [Google Scholar] [CrossRef]
- Ashour, M.; Mabrouk, M.M.; Ayoub, H.F.; El-Feky, M.M.M.; Sharawy, Z.Z.; van Doan, H.; El-Haroun, E.; Goda, A.M.A.-S. Effect of Dietary Seaweed Extract Supplementation on Growth, Feed Utilization, Hematological Indices and Non-Specific immunity of Nile Tilapia, Oreochromis niloticus Challenged with Aeromonas hydrophila. J. Appl. Phycol. 2020, 32, 3467–3479. [Google Scholar] [CrossRef]
- Abo-Taleb, H.A.; Zeina, A.F.; Ashour, M.; Mabrouk, M.M.; Sallam, A.E.; El-feky, M.M.M. Isolation and cultivation of the freshwater amphipod Gammarus pulex (Linnaeus, 1758), with an evaluation of its chemical and nutritional content. Egypt. J. Aquat. Biol. Fish. 2020, 24, 69–82. [Google Scholar] [CrossRef]
- Tarakhovskaya, E.R.; Maslov, Y.I.; Shishova, M.F. Phytohormones in algae. Russ. J. Plant Physiol. 2007, 54, 163–170. [Google Scholar] [CrossRef]
- Xu, C.; Leskovar, D.I. Effects of Ascophyllum nodosum seaweed extracts on spinach growth, physiology and nutrition value under drought stress. Sci. Hortic. 2015, 183, 39–47. [Google Scholar] [CrossRef]
- Battacharyya, D.; Babgohari, M.Z.; Rathor, P.; Prithiviraj, B. Seaweed extracts as biostimulants in horticulture. Sci. Hortic. 2015, 196, 39–48. [Google Scholar] [CrossRef]
- Hernández-Herrera, R.M.; Santacruz-Ruvalcaba, F.; Ruiz-López, M.A.; Norrie, J.; Hernández-Carmona, G. Effect of liquid seaweed extracts on growth of tomato seedlings (Solanum lycopersicum L.). J. Appl. Phycol. 2014, 26, 619–628. [Google Scholar] [CrossRef]
- Drobek, M.; Frąc, M.; Cybulska, J. Plant Biostimulants: Importance of the Quality and Yield of Horticultural Crops and the Improvement of Plant Tolerance to Abiotic Stress—A Review. Agronomy 2019, 9, 335. [Google Scholar] [CrossRef]
- Rouphael, Y.; de Micco, V.; Arena, C.; Raimondi, G.; Colla, G.; de Pascale, S. Effect of Ecklonia maxima seaweed extract on yield, mineral composition, gas exchange, and leaf anatomy of Zucchini squash grown under saline conditions. J. Appl. Phycol. 2017, 29, 459–470. [Google Scholar] [CrossRef]
- Slavik, M. Production of Norway spruce (Picea abies [L.] Karst.) seedlings on substrate mixes using growth stimulants. J. For. Sci. 2005, 51, 15–23. [Google Scholar] [CrossRef]
- Ashour, M. Current and future perspectives of microalgae-aquaculture in Egypt, case study: SIMAF-prototype-project. Egypt. J. Anim. Prod. 2020, 57, 163–170. [Google Scholar] [CrossRef]
- Murugalakshmikumari, R.; Ramasubramanian, V.; Muthuchezhian, K. Studies on the utilization of seaweed as an organic fertilizer on the growth and some biochemical characteristics of black gram and cucumber. Seaweed Res. Util. 2002, 24, 125–128. [Google Scholar]
- Ahmed, Y.M.; Shalaby, E.A. Effect of different seaweed extracts and compost on vegetative growth, yield and fruit quality of cucumber. J. Hortic. Sci. Ornam. Plants 2012, 4, 235–240. [Google Scholar]
- Valencia, R.T.; Acosta, L.S.; Hernández, M.F.; Rangel, P.P.; Robles, M.; Ángel, G.; Cruz, R.D.C.A.; Vázquez, C.V. Effect of seaweed aqueous extracts and compost on vegetative growth, yield, and nutraceutical quality of cucumber (Cucumis sativus L.) Fruit. Agronomy 2018, 8, 264. [Google Scholar] [CrossRef]
- Nelson, W.R.; van Staden, J. The effect of seaweed concentrate on wheat culms. J. Plant Physiol. 1984, 115, 433–437. [Google Scholar] [CrossRef]
- Elshobary, M.; El-Shenody, R.; Ashour, M.; Zabed, H.M.; Qi, X. Antimicrobial and antioxidant characterization of bioactive components from Chlorococcum minutum, a newly isolated green microalga. Food Biosci. J. 2020, 35, 100567. [Google Scholar] [CrossRef]
- Page, A.L.; Miller, R.H.; Keeney, D.R. Methods of Soil Analysis. Part 2. Chemical and Microbiological Properties; American Society of Agronomy; Soil Science Society of America: Madison, WI, USA, 1982; Volume 2. [Google Scholar]
- Laane, H.M. The effects of foliar sprays with different silicon compounds. Plants 2018, 7, 45. [Google Scholar] [CrossRef] [PubMed]
- Mekki, B.E.D. Effect of boron foliar application on yield and quality of some sunflower (Helianthus annuus L.) cultivars. J. Agric. Sci. Technol. B 2015, 5, 309–3016. [Google Scholar] [CrossRef][Green Version]
- Timotiwu, P.B.; Amalia, S. The Effects of Foliar Boron and Silica Through the Leaves on Soybean Growth and Yield. J. Agric. Stud. 2018, 6, 34–48. [Google Scholar] [CrossRef]
- Shobier, A.H.; Ghani, S.A.A.; Barakat, K.M. GC/MS spectroscopic approach and antifungal potential of bioactive extracts produced by marine macroalgae. Egypt. J. Aquat. Res. 2016, 42, 289–299. [Google Scholar] [CrossRef]
- Ibrahim, E.A.; Aly, H.F.; Baker, D.H.A.; Mahmoud, K.H.A.; El-Baz, F.K. Marine algal sterol hydrocarbon with anti-inflammatory, anticancer and anti-oxidant properties. Int. J. Pharma Bio Sci. 2016, 7, 392–398. [Google Scholar]
- El-Din, S.M.M.; Mohyeldin, M.M. Component analysis and antifungal activity of the compounds extracted from four brown seaweeds with different solvents at different seasons. J. Ocean Univ. China 2018, 17, 1178–1188. [Google Scholar] [CrossRef]
- Hassan, W.M.S.; Shobier, H.A. GC/MS identification and applications of bioactive seaweed extracts from Mediterranean coast of Egypt. Egypt. J. Aquat. Biol. Fish. 2018, 22, 1–21. [Google Scholar] [CrossRef]
- Abdel-Latif, H.H.; Shams El-Din, N.G.; Ibrahim, H.A.H. Antimicrobial activity of the newly recorded red alga Grateloupia doryphora collected from the Eastern Harbor, Alexandria, Egypt. J. Appl. Microbiol. 2018, 125, 1321–1332. [Google Scholar] [CrossRef] [PubMed]
- Hirayama, O.; Nakamura, K.; Hamada, S.; Kobayasi, Y. Singlet oxygen quenching ability of naturally occurring carotenoids. Lipids 1994, 29, 149–150. [Google Scholar] [CrossRef]
- El-Din, S.M.M.; El-Ahwany, A.M. Bioactivity and phytochemical constituents of marine red seaweeds (Jania rubens, Corallina mediterranea, and Pterocladia capillacea). J. Taibah Univ. Sci. 2016, 10, 471–484. [Google Scholar] [CrossRef]
- Prichard, R.; Ménez, C.; Lespine, A. Moxidectin and the avermectins: Consanguinity but not identity. Int. J. Parasitol. Drugs Drug Resist. 2012, 2, 134–153. [Google Scholar] [CrossRef]
- Kumar, K.; Banerjee, D.; Das, D. Carbon dioxide sequestration from industrial flue gas by Chlorella sorokiniana. Bioresour. Technol. 2014, 152, 225–233. [Google Scholar] [CrossRef]
- Takiguchi, Y.; Mishima, H.; Okuda, M.; Terao, M.; Aoki, A.; Fukuda, R. Milbemycins, a new family of macrolide antibiotics: Fermentation, isolation and physico-chemical properties. J. Antibiot. 1980, 33, 1120–1127. [Google Scholar] [CrossRef]
- Azam, M.M.; Waris, A.; Nahar, N.M. Prospects and potential of fatty acid methyl esters of some non-traditional seed oils for use as biodiesel in India. Biomass Bioenergy 2005, 29, 293–302. [Google Scholar] [CrossRef]
- Agoramoorthy, G.; Chandrasekaran, M.; Venkatesalu, V.; Hsu, M.J. Antibacterial and antifungal activities of fatty acid methyl esters of the blind-your-eye mangrove from India. Braz. J. Microbiol. 2007, 38, 739–742. [Google Scholar] [CrossRef]
- Synowiec, A.; Halecki, W.; Wielgusz, K.; Byczynska, M.; Czaplicki, S. Effect of fatty acid methyl esters on the herbicidal effect of essential oils on corn and weeds. Weed Technol. 2017, 31, 301–309. [Google Scholar] [CrossRef]
- Matanjun, P.; Mohamed, S.; Mustapha, N.M.; Muhammad, K. Nutrient content of tropical edible seaweeds, Eucheuma cottonii, Caulerpa lentillifera and Sargassum polycystum. J. Appl. Phycol. 2009, 21, 75–80. [Google Scholar] [CrossRef]
- Vassiliou, E.K.; Gonzalez, A.; Garcia, C.; Tadros, J.H.; Chakraborty, G.; Toney, J.H. Oleic acid and peanut oil high in oleic acid reverse the inhibitory effect of insulin production of the inflammatory cytokine TNF-α both in vitro and in vivo systems. Lipids Health Dis. 2009, 8, 25. [Google Scholar] [CrossRef] [PubMed]
- Carrillo, P.C.; Camarero, C.; del Mar, M.; de La Torre, S.A. Role of oleic acid in immune system; mechanism of action; A review. Nutr. Hosp. 2012, 27, 978–990. [Google Scholar] [CrossRef]
- Cardoso, C.; Ripol, A.; Afonso, C.; Freire, M.; Varela, J.; Quental-Ferreira, H.; Pousão-Ferreira, P.; Bandarra, N. Fatty acid profiles of the main lipid classes of green seaweeds from fish pond aquaculture. Food Sci. Nutr. 2017, 5, 1186–1194. [Google Scholar] [CrossRef] [PubMed]
- Santos, C.C.D.; Salvadori, M.S.; Mota, V.G.; Costa, L.M.; de Almeida, A.A.C.; de Oliveira, G.A.L.; Costa, J.P.; de Sousa, D.P.; de Freitas, R.M.; de Almeida, R.N. Antinociceptive and antioxidant activities of phytol in vivo and in vitro models. Neurosci. J. 2013, 2013, 949452. [Google Scholar] [CrossRef]
- Gutbrod, K.; Romer, J.; Dormann, P. Phytol metabolism in plants. Prog. Lipid Res. 2019, 74, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Hassan, S.M.; Ghoneim, I.M.; El-Araby, S.M.; Elsharkawy, A. Effect of Different Plastic Covers on Vegetative Growth and Yield Quality of Cucumber Plants (Cucumis sativus L.). World Appl. Sci. J. 2014, 32, 217–225. [Google Scholar] [CrossRef]
- Hassan, S.M.; El-Bebany, A.F.; Salem, M.Z.M.; Komeil, D.A. Productivity and Post-Harvest Fungal Resistance of Hot Pepper as Affected by Potassium Silicate, Clove Extract Foliar Spray and Nitrogen Application. Plants 2021, 10, 662. [Google Scholar] [CrossRef]
- Evenhuis, B. Simplified Methods for Foliar Analysis Parts. I. VII. International Report; Royal Tropical Institute: Amsterdam, The Netherlands, 1976. [Google Scholar]
- Cottenie, A.; Verloo, M.; Kiekens, L.; Velghe, G.; Camerlynck, R. Chemical analysis of plants and soils. IWONL Bruss. 1982, 63. [Google Scholar] [CrossRef]
- Hamed, S.M.; Abd El-Rhman, A.A.; Abdel-Raouf, N.; Ibraheem, I.B.M. Role of marine macroalgae in plant protection & improvement for sustainable agriculture technology. Beni Suef Univ. J. Basic Appl. Sci. 2018, 7, 104–110. [Google Scholar] [CrossRef]
- Vijayanand, N.; Sivasangari, R.S.; Rathinavel, S. Potential of liquid extracts of Sargassum wightii on growth, biochemical and yield parameters of cluster bean plant. Asian Pac. J. Reprod. 2014, 3, 150–155. [Google Scholar] [CrossRef]
- Metwally, A.S.; El-Naggar, H.A.; El-Damhougy, K.A.; Bashar, M.A.E.; Ashour, M.; Abo-Taleb, H.A.H. GC-MS analysis of bioactive components in six different crude extracts from the Soft Coral (Sinularia maxim) collected from Ras Mohamed, Aqaba Gulf, Red Sea, Egypt. Egypt. J. Aquat. Biol. Fish. 2020, 24, 425–434. [Google Scholar] [CrossRef]
- Ashour, M.; Abo-Taleb, H.A.; Abou-Mahmoud, M.M.; El-Feky, M.M.M. Effect of the integration between plankton natural productivity and environmental assessment of irrigation water, El-Mahmoudia Canal, on aquaculture potential of Oreochromis niloticus. Turk. J. Fish. Aquat. Sci. 2018, 18, 1163–1175. [Google Scholar] [CrossRef]
- Hassan, S.M.; Ashour, M.; Soliman, A.A.F.; Elshobary, M.E. Evaluation of a New Commercial Seaweeds Extract (TAM®) as Biofertilizer for Improving Morpho-Agronomic and Bioactive Traits of Eruca sativa Mill. Sustinability 2021. (Accepted Paper). [Google Scholar]
- Ashour, M.; Mabrouk, M.M.; Abo-Taleb, H.A.; Sharawy, Z.Z.; Ayoub, F.H.; El-Haroun, E.; Goda, A.M.S.-A. Efficacy of a Liquid Seaweed Extract (TAM®) in Improving Culture Waters, Diversity, Abundance of the Zooplankton Community, and Enhancing Growth and Immune Stimulation of Nile Tilapia, Oreochromis niloticus, Challenged by Aeromonas hydrophila. Aquaculture 2021. (Accepted Paper). [Google Scholar]
- Sheela, D.; Uthayakumari, F. GC-MS analysis of bioactive constituents from coastal sand dune taxon-Sesuvium portulacastrum (L.). Biosci. Discov. 2013, 4, 47–53. [Google Scholar]
- Upchurch, R.G. Fatty acid unsaturation, mobilization, and regulation in the response of plants to stress. Biotechnol. Lett. 2008, 30, 967–977. [Google Scholar] [CrossRef]
- Rathore, S.S.; Chaudhary, D.R.; Boricha, G.N.; Ghosh, A.; Bhatt, B.P.; Zodape, S.T.; Patolia, J.S. Effect of seaweed extract on the growth, yield and nutrient uptake of soybean (Glycine max) under rainfed conditions. S. Afr. J. Bot. 2009, 75, 351–355. [Google Scholar] [CrossRef]
- Shah, M.T.; Zodape, S.T.; Chaudhary, D.R.; Eswaran, K.; Chikara, J. Seaweed sap as an alternative liquid fertilizer for yield and quality improvement of wheat. J. Plant Nutr. 2013, 36, 192–200. [Google Scholar] [CrossRef]
- Layek, J.; Das, A.; Ghosh, A.; Sarkar, D.; Idapuganti, R.G.; Boragohain, J.; Lal, R. Foliar application of seaweed sap enhances growth, yield and quality of maize in eastern himalayas. Proc. Natl. Acad. Sci. USA India Sect. B Biol. Sci. 2019, 89, 221–229. [Google Scholar] [CrossRef]
- Hidangmayum, A.; Sharma, R. Effect of different concentrations of commercial seaweed liquid extract of Ascophyllum nodosum as a plant bio stimulant on growth, yield and biochemical constituents of onion (Allium cepa L.). J. Pharmacogn. Phytochem. 2017, 6, 658–663. [Google Scholar]
- Vijayakumar, S.; Durgadevi, S.; Arulmozhi, P.; Rajalakshmi, S.; Gopalakrishnan, T.; Parameswari, N. Effect of seaweed liquid fertilizer on yield and quality of Capsicum annum L. Acta Ecol. Sin. 2018, 39, 406–410. [Google Scholar] [CrossRef]
- Jayasinghe, P.S.; Pahalawattaarachchi, V.; Ranaweera, K.K. Effect of seaweed liquid fertilizer on plant growth of Capsicum annum. Discovery 2016, 52, 723–734. [Google Scholar]
- DeGannes, A.; Heru, K.R.; Mohammed, A.; Paul, C.; Rowe, J.; Sealy, L.; Seepersad, G. Tropical Greenhouse Manual for the Caribbean; The University of the West Indies, St. Augustine, The Caribbean Agricultural Research and Development Institute (CARDI): Port of Spain, Trinidad and Tobago, 2014. [Google Scholar]
- Farrag, D.K.H.; Omara, A.A.; El-Said, M.N. Significance of foliar spray with some growth promoting rhizobacteria and some natural biostimulants on yield and quality of cucumber plant. Egypt. J. Hortic. 2015, 42, 321–332. [Google Scholar] [CrossRef]
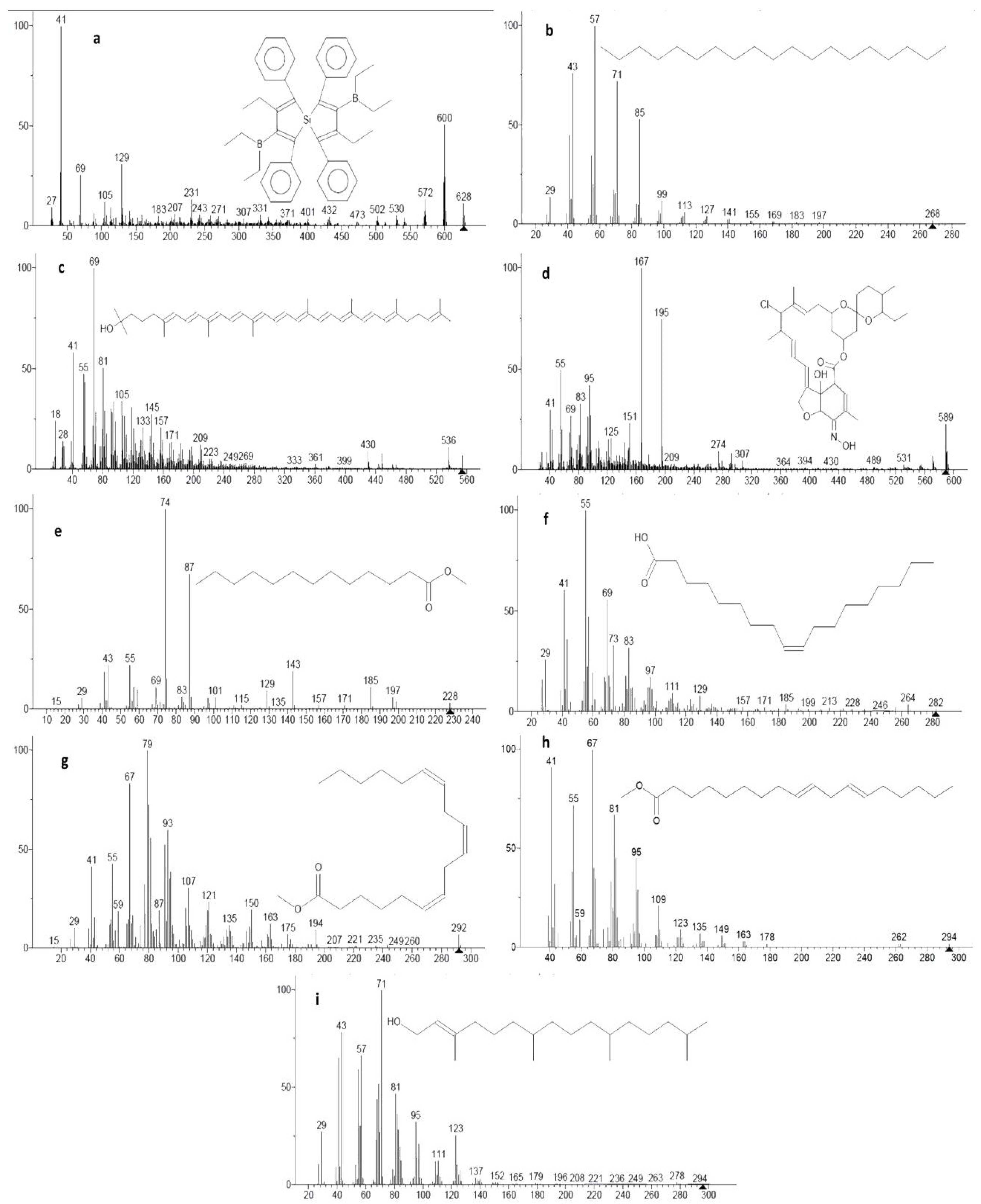
| RT (min) | Compound Name | Formula | Molecular Weight | Nature | Biological Properties | Literatures |
|---|---|---|---|---|---|---|
| 8.99 | 5-Silaspiro[4.4]nona-1,3,6,8-tetraene,3,8-bis (diethylboryl)-2,7-diethyl-1,4,6,9-tetraphenyl- | C44H50B2Si | 628.39 | Silicon–boron compound | Growth and immunity enhancer; antifungal for plant pathogen | [13,29,30,31] |
| 16.31 | Nonadecane | C19H40 | 268.31 | Alkane | Antioxidant; anticancer; antimicrobial; anti-inflammatory | [32,33,34,35,36] |
| 19.45 | Rhodopin | C40H58O | 554.45 | Carotenoid | Growth enhancer; antioxidant | [13,37,38] |
| 20.07 | Milbemycin B, 5-demethoxy-5-one-6,28-anhydro-25-ethyl-4-methyl-13-chloro-oxime | C32H44ClNO7 | 589.28 | Macrocyclic lactones | Immunity enhancer; antiparasitic; antihelminthic; insecticidal | [13,39,40,41] |
| 20.90 | Tridecanoic acid, methyl ester | C14H28O2 | 228.21 | Fatty acid methyl esters (FAMEs) | Antioxidant; surfactants; herbicidal; antimicrobial | [13,42,43,44] |
| 21.63 | Oleic acid | C18H34O2 | 282.26 | Fatty acid | Immunity enhancer; enhancing insulin production; anti-inflammatory | [13,45,46,47,48] |
| 23.74 | γ-Linolenic acid, methyl ester | C19H32O2 | 292.24 | FAMEs | Antioxidant; surfactants; herbicidal; antimicrobial | [13,42,43,44] |
| 24.02 | 9,12-Octadecadienoic acid, methyl ester, (E,E)- | C19H34O2 | 294.26 | FAMEs | Antioxidant; surfactants; herbicidal; antimicrobial | [13,42,43,44] |
| 24.37 | Phytol | C20H40O | 296.31 | Diterpene alcohol | Antioxidant; antinociceptive | [13,49,50] |
| Item | Value |
|---|---|
| Physical analyses | |
| Color | Dark brown |
| Odor | Seaweed |
| Density | 1.20 |
| pH | 9–9.5 |
| Biochemical analyses (% DM) | |
| Total polysaccharides | 15 |
| Total organic matter | 8.2 |
| Total dissolved solids | 2.6 |
| Chemical analyses | |
| Major elements (%) | |
| Potassium | 15 |
| Phosphorus | 2.4 |
| Total nitrogen | 0.14 |
| Minor elements (ppm) | |
| Copper | 0.39 |
| Iron | 16.18 |
| Magnesium | 19.72 |
| Zinc | 1.19 |
| Manganese | 3.72 |
| Heavy metals (ppm) | |
| Cadmium | 0.00 |
| Chromium | 0.00 |
| Lead | 0.00 |
| Nickel | 0.00 |
| Arsenic | 0.55 |
| Properties | Seasons | |
|---|---|---|
| Winter 2017 | Winter 2018 | |
| Physical properties | ||
| Sand% | 43.3 | 42.8 |
| Silt% | 25.5 | 23.5 |
| Clay% | 31.2 | 33.7 |
| Soil texture | Loamy | Loamy |
| Chemical properties | ||
| pH | 8.45 | 8.88 |
| E.C. (dS m−1) | 3.01 | 3.00 |
| Soluble cations (m.eq/L) | ||
| Ca+ | 2.13 | 2.02 |
| Mg++ | 2.03 | 1.93 |
| Na+ | 2.51 | 2.43 |
| K+ | 0.41 | 0.38 |
| Soluble anions (m.eq/L) | ||
| CO3 | Zero | Zero |
| HCO3 | 1.35 | 1.20 |
| Cl− | 2.00 | 1.90 |
| SO− | 3.20 | 3.11 |
| Total nitrogen (%) | 0.19 | 0.15 |
| Phosphorus (ppm) | 0.41 | 0.44 |
| NPK Rate | Week after Transplanting | Ammonium Nitrate (g 100 m−2) | Phosphoric Acid (cm3 100 m−2) | Potassium Sulfate (g 100 m−2) |
|---|---|---|---|---|
| 2% | 2 | 200 | 37 | 160 |
| 4% | 3 | 400 | 74 | 320 |
| 6% | 4 | 600 | 111 | 480 |
| 8% | 5 | 800 | 148 | 640 |
| 12% | 6 | 1200 | 222 | 960 |
| 12% | 7 | 1200 | 222 | 960 |
| 12% | 8 | 1200 | 222 | 960 |
| 12% | 9 | 1200 | 222 | 960 |
| 8% | 10 | 800 | 148 | 640 |
| 8% | 11 | 800 | 148 | 640 |
| 8% | 12 | 800 | 148 | 640 |
| 8% | 13 | 800 | 148 | 640 |
| Treatments * | C0 | C25 | C50 | C75 | C100 | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Parameters | 2017 | 2018 | 2017 | 2018 | 2017 | 2018 | 2017 | 2018 | 2017 | 2018 |
| Plant height (cm) | 193.7 ± 2.3 b | 195.3 ± 2.5 b | 199.0 ± 1.0 a,b | 197.3 ± 4.0 b | 200.0 ± 3.5 a,b | 200.3 ± 3.2 b | 200.7 ± 3.8 a,b | 201 ± 2.6 b | 203.3 ± 5.9 a | 209.7 ± 1.5 a |
| Leaf number | 27.7 ± 1.2 d | 28.3 ± 0.6 c | 29.7 ± 0.6 c | 30.7 ± 0.6b c | 30.7 ± 0.6 b,c | 32.3 ± 0.6 a | 32.3 ± 0.6 a | 32.6 ± 0.6 a | 31.3 ± 1.2 a,b | 31.6 ± 1.1 a,b |
| Leaf area (plant/cm2) | 275 ± 5.0 c | 288 ± 3.0 b | 299 ± 1.5 b | 299 ± 2.3 a | 301 ± 0.6 a,b | 304 ± 4.5 a | 306 ± 4.2 a | 306 ± 1.5 a | 305 ± 2.9 a,b | 306 ± 10.4 a |
| Chlorophyll (mg.100g f.w.) | 37.9 ± 0.7 a | 38.7 ± 0.5 a | 38.9 ± 0.4 a | 39.0 ± 0.4 a | 39.3 ± 0.1 a | 39.0 ± 1.5 a | 38.8 ± 1.6 a | 38.7 ± 0.5 a | 38.7 ± 1.6 a | 39.4 ± 0.9 a |
| Dry matter (%) | 14.8 ± 0.9 b | 14.6 ± 1.0 a | 15.1 ± 0.2 a b | 15.4 ± 0.6 a b | 15.7 ± 0.4 a,b | 15.9 ± 0.5 a,b | 16.4 ± 0.5 a | 16.6 ± 0.7 a | 15.8 ± 0.9 a,b | 16.6 ± 0.5 a |
| Leaf nitrogen (%) | 3.25 ± 0.14 a | 3.48 ± 0.55 a | 2.96 ± 0.28 a | 2.99 ± 0.13 a,b | 3.21 ± 0.17 a | 3.27 ± 0.14 a b | 3.01 ± 0.20 a | 2.98 ± 0.15 a,b | 3.03 ± 0.06 a | 2.91 ± 0.17 b |
| Leaf phosphorus (%) | 0.51 ± 0.02 a | 0.55 ± 0.05 a | 0.48 ± 0.07 a | 0.47 ± 0.06 a | 0.52 ± 0.02 a | 0.52 ± 0.03 a | 0.48 ± 0.04 a | 0.51 ± 0.01 a | 0.54 ± 0.01 a | 0.50 ± 0.03 a |
| Leaf potassium (%) | 2.97 ± 0.09 a | 3.02 ± 0.42 a | 2.92 ± 0.07 a | 2.87 ± 0.12 a | 2.97 ± 0.09 a | 2.96 ± 0.17 a | 2.80 ± 0.20 a | 2.96 ± 0.03 a | 2.83 ± 0.16 a | 2.96 ± 0.09 a |
| Treatments * | C0 | C25 | C50 | C75 | C100 | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Parameters | 2017 | 2018 | 2017 | 2018 | 2017 | 2018 | 2017 | 2018 | 2017 | 2018 |
| Total yield (kg/m2) | 2.47 ± 0.42 c | 2.80 ± 0.10 c | 3.40 ± 0.20 b | 3.47 ± 0.25 b | 4.07 ± 0.12 a | 3.93 ± 0.23 a | 4.07 ± 0.15 a | 4.01 ± 0.02 a | 3.93 ± 0.23 a | 4.00 ± 0.00 a |
| Dry matter (%) | 4.84 ± 0.46 a | 4.85 ± 0.25 b | 5.11 ± 0.19 a | 5.29 ± 0.15 a | 5.20 ± 0.18 a | 5.15 ± 0.12 a,b | 5.17 ± 0.17 a | 5.19 ± 0.19 a | 5.15 ± 0.03 a | 5.29 ± 0.07 a |
| Length (Cm) | 14.3 ± 0.86 a | 13.3 ± 0.16 b | 14.7 ± 0.45 a | 14.4 ± 0.69 a | 14.9 ± 0.47 a | 14.9 ± 0.53 a | 14.7 ± 0.53 a | 15.1 ± 0.14 a | 15.2 ± 0.20 a | 15.1 ± 0.21 a |
| Diameter (Cm) | 3.06 ± 0.06 b | 3.08 ± 0.06 a | 3.09 ± 0.01 b | 3.09 ± 0.02 a | 3.15 ± 0.06 b | 3.09 ± 0.13 a | 3.26 ± 0.25 b | 3.13 ± 0.11 a | 3.57 ± 0.16 a | 3.11 ± 0.04 a |
| Fruit nitrogen (%) | 2.24 ± 0.28 a | 2.09 ± 0.08 a | 2.00 ± 0.01 a b | 1.97 ± 0.07 a | 2.05 ± 0.04 a,b | 2.06 ± 0.05 a | 1.97 ± 0.05 b | 2.04 ± 0.05 a | 2.03 ± 0.04 a b | 2.00 ± 0.08 a |
| Fruit phosphorus (%) | 0.41 ± 0.02 a | 0.47 ± 0.03 a | 0.42 ± 0.03 a | 0.43 ± 0.06 a | 0.40 ± 0.02 a | 0.47 ± 0.06 a | 0.43 ± 0.06 a | 0.40 ± 0.02 a | 0.41 ± 0.02 a | 0.42 ± 0.04 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hassan, S.M.; Ashour, M.; Sakai, N.; Zhang, L.; Hassanien, H.A.; Gaber, A.; Ammar, G. Impact of Seaweed Liquid Extract Biostimulant on Growth, Yield, and Chemical Composition of Cucumber (Cucumis sativus). Agriculture 2021, 11, 320. https://doi.org/10.3390/agriculture11040320
Hassan SM, Ashour M, Sakai N, Zhang L, Hassanien HA, Gaber A, Ammar G. Impact of Seaweed Liquid Extract Biostimulant on Growth, Yield, and Chemical Composition of Cucumber (Cucumis sativus). Agriculture. 2021; 11(4):320. https://doi.org/10.3390/agriculture11040320
Chicago/Turabian StyleHassan, Shimaa M., Mohamed Ashour, Nobumitsu Sakai, Lixin Zhang, Hesham A. Hassanien, Ahmed Gaber, and Gamal Ammar. 2021. "Impact of Seaweed Liquid Extract Biostimulant on Growth, Yield, and Chemical Composition of Cucumber (Cucumis sativus)" Agriculture 11, no. 4: 320. https://doi.org/10.3390/agriculture11040320
APA StyleHassan, S. M., Ashour, M., Sakai, N., Zhang, L., Hassanien, H. A., Gaber, A., & Ammar, G. (2021). Impact of Seaweed Liquid Extract Biostimulant on Growth, Yield, and Chemical Composition of Cucumber (Cucumis sativus). Agriculture, 11(4), 320. https://doi.org/10.3390/agriculture11040320











