Assessment of Andean lupin (Lupinus mutabilis) Genotypes for Improved Frost Tolerance
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Experimental Design of Artificial-Frost Tolerance Investigations
2.3. Statistical Analysis
3. Results and Discussion
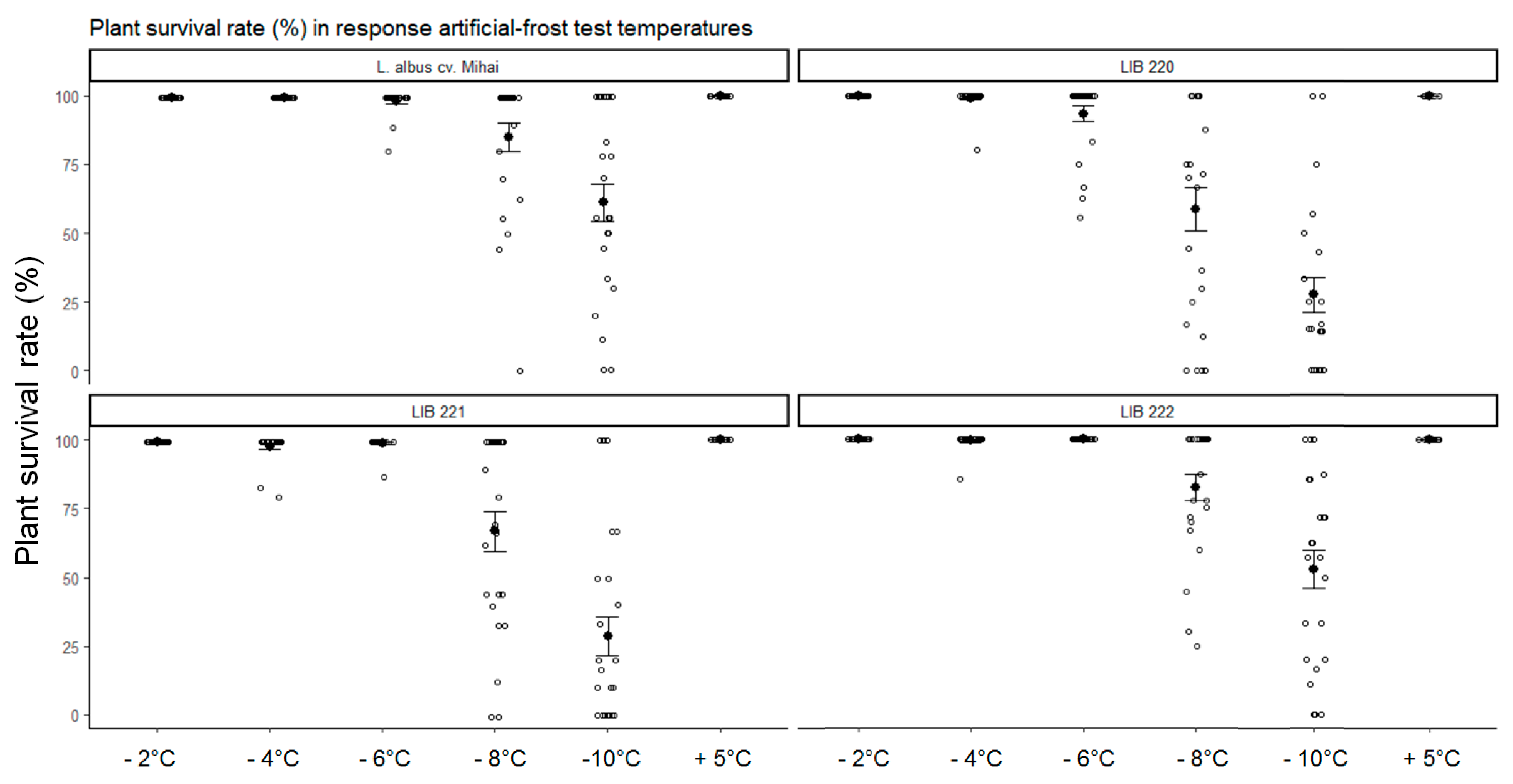
3.1. Artificial-Frost Tolerance Assessment of Andean lupin Genotypes
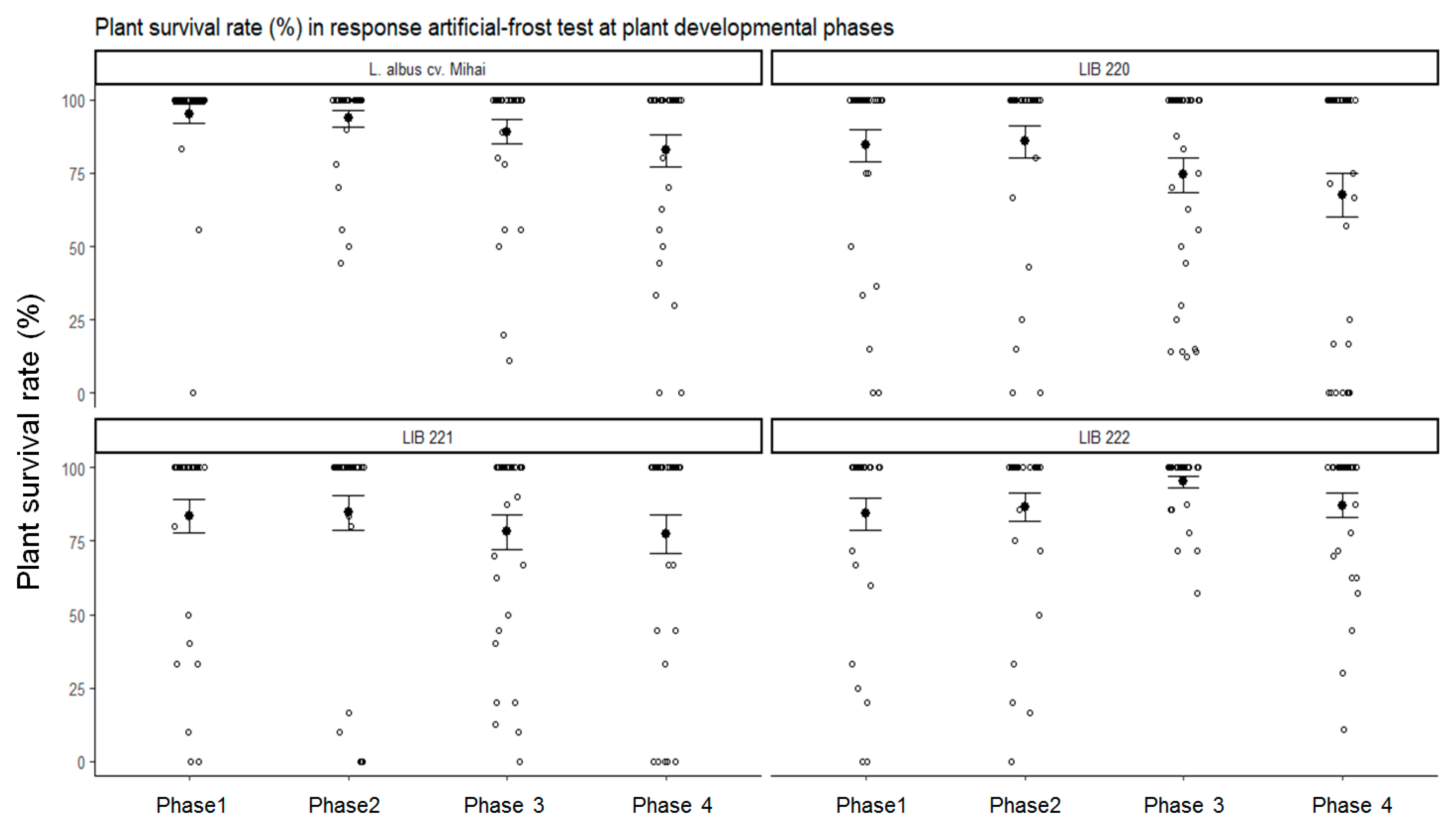
3.2. Plant Developmental Phases Influence Frost Tolerance of Andean lupin
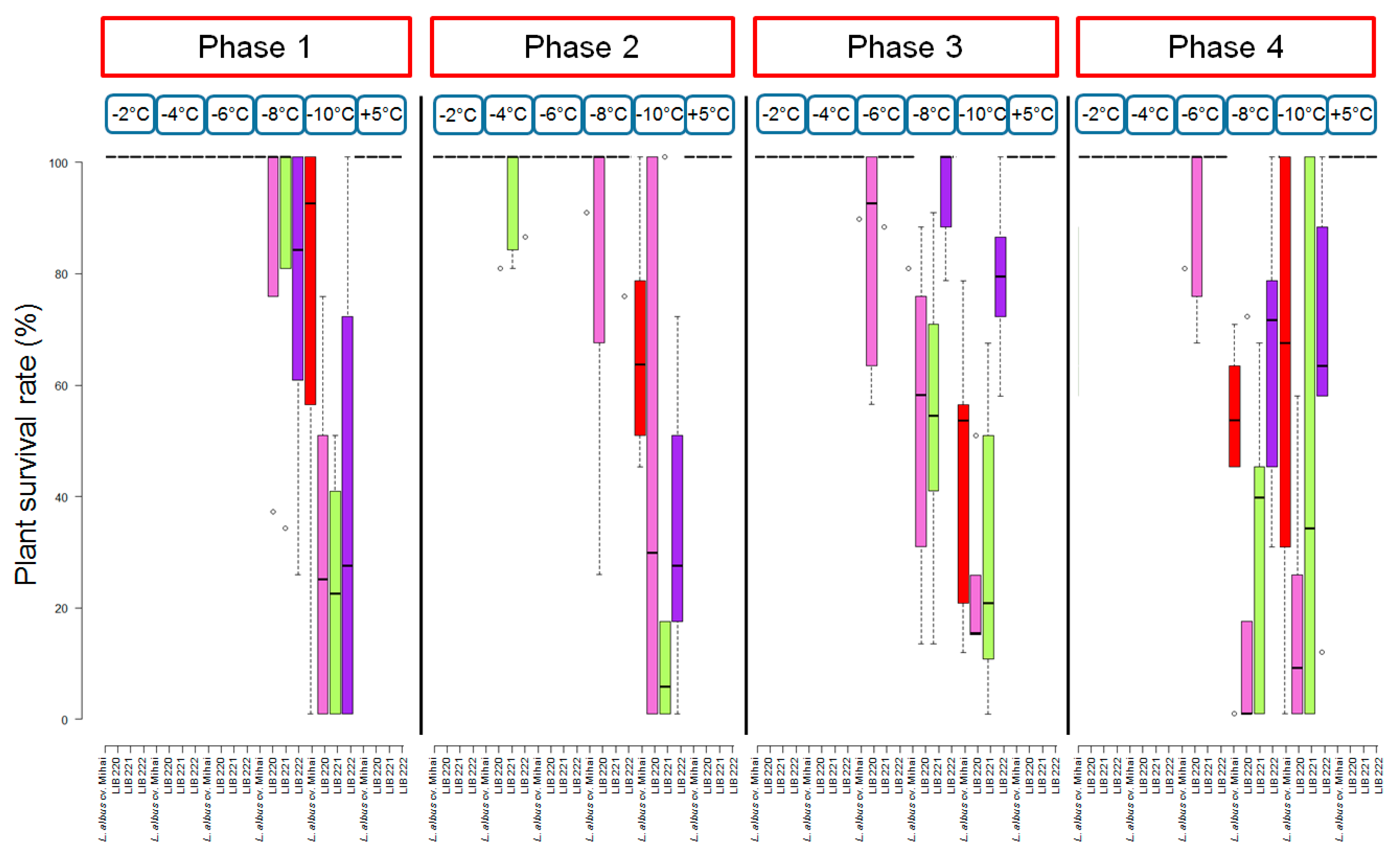
3.3. Investigation of Significance among Development Phases, Temperatures, and Accessions
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bebeli, P.J.; Lazaridi, E.; Chatzigeorgiou, T.; Suso, M.-J.; Hein, W.; Alexopoulos, A.A.; Canha, G.; van Haren, R.J.F.; Jóhannsson, M.H.; Mateos, C.; et al. State and Progress of Andean Lupin Cultivation in Europe: A Review. Agronomy 2020, 10, 1038. [Google Scholar] [CrossRef]
- De Ron, A.; Sparvoli, F.; Pueyo, J.; Bazile, D. Editorial: Protein Crops: Food and Feed for the Future. Front. Plant Sci. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Gresta, F.; Wink, M.; Prins, U.; Abberton, M.; Capraro, J.; Scarafoni, A.; Hill, G. Lupins in European cropping systems. Legumes Crop. Syst. 2017, CABI, 88–108. [Google Scholar] [CrossRef]
- Lucas, M.M.; Stoddard, F.; Annicchiarico, P.; Frias, J.; Martinez-Villaluenga, C.; Sussmann, D.; Duranti, M.; Seger, A.; Zander, P.M.; Pueyo, J.J. The future of lupin as a protein crop in Europe. Front. Plant Sci. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Gulisano, A.; Alves, S.; Neves Martins, J.; Trindade, L. Genetics and Breeding of Lupinus mutabilis: An Emerging Protein Crop. Front. Plant Sci. 2019, 10, 1385. [Google Scholar] [CrossRef] [PubMed]
- Galek, R.; Sawicka-Sienkiewicz, E.; Zalewski, D. Evaluation of interspecific hybrids of andean lupin and their parental forms with regard to some morphological and quantitative characters. Fragm. Agron. (Poland) 2007, 24, 81–87. [Google Scholar]
- Hardy, A.; Huyghe, C.; Papineau, J. Dry matter accumulation and partitioning, and seed yield in indeterminate Andean lupin (Lupinus mutabilis Sweet). Aust. J. Agric. Res. 1997, 48, 91–102. [Google Scholar] [CrossRef]
- Adhikari, K.N.; Buirchell, B.J.; Sweetingham, M.W. Length of vernalization period affects flowering time in three lupin species. Plant Breed. 2012, 131, 631–636. [Google Scholar] [CrossRef]
- Jacobsen, S.-E.; Mujica, A. Geographical distribution of the Andean lupin (Lupinus mutabilis Sweet). Plant Genet. Res. Newslett. 2008, 155, 1–8. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria. 2020. Available online: https://www.R-project.org/ (accessed on 10 July 2020).
- Chalker-Scott, L. Environmental Significance of Anthocyanins in Plant Stress Responses. Photochem. Photobiol. 1999, 70, 1–9. [Google Scholar] [CrossRef]
- Hardy, A.; Huyghe, C.; Rahirne, M.A.; Roemer, P.; Neves-Martins, J.M.; Sawieka-Sienkiewiez, E.; Caligarie, P.D.S. Effects of genotype and environment on architecture and flowering time of indeterminate Andean lupins (Lupinus mutabilis Sweet). Aust. J. Agric. Res. 1998, 49, 1241–1251. [Google Scholar] [CrossRef]
- Lazaridi, E.; Papadopoulos, G.K.; Bebeli, P.J. Andean Lupin Phenology and Agronomic Performance under Different Planting Dates in a Mediterranean Climate. Agronomy 2020, 10, 2020. [Google Scholar] [CrossRef]
- Neves Martins, J.M.; Talhinhas, P.; de Sousa, R.B. Yield and seed chemical composition of Lupinus mutabilis in Portugal. Rev. Ciências Agrárias 2016, 39, 518–525. [Google Scholar] [CrossRef]
- Falconi, C.E. Lupinus mutabilis in Ecuador with Special Emphasis on Anthracnose Resistance. Ph.D. Thesis, Wageningen Universi-ty, Wageningen, The Netherlands, 2012. [Google Scholar]
- Tang, C.; Robson, A.D.; Longnecker, N.E.; Buirchell, B.J. The Growth of Lupinus Species on Alkaline Soils. Aust. J. Agric. Res. 1995, 46, 255–268. [Google Scholar] [CrossRef]
- Kerley, S.J.; Huyghe, C. Comparison of acid and alkaline soil and liquid culture growth systems for studies of shoot and root characteristics of white lupin (Lupinus albus L.) genotypes. Plant Soil 2001, 236, 275–286. [Google Scholar] [CrossRef]
- Pszczółkowska, A.; Okorski, A.; Kotecki, A.; Gas, M.; Kullik, T.; Reczek, A. Incidence of seed-borne fungi on Lupinus mutabilis depending on a plant morphotype, sowing date and plant density. J. Elem. 2016, 21, 501–512. [Google Scholar] [CrossRef][Green Version]
- López-Bellido, L.; Fuentes, M.; Lhamby, J.C.B.; Castillo, J.E. Growth and yield of white lupin (Lupinus albus) under Mediterranean conditions: Effect of sowing date. Field Crop. Res. 1994, 36, 87–94. [Google Scholar] [CrossRef]
- Raabe, A.; von Sengbusch, R. Zuchterisch wichtige Beobachtungen an einigen Lupinenarten. Der Zuchter 1935, 7, 244–248. [Google Scholar]
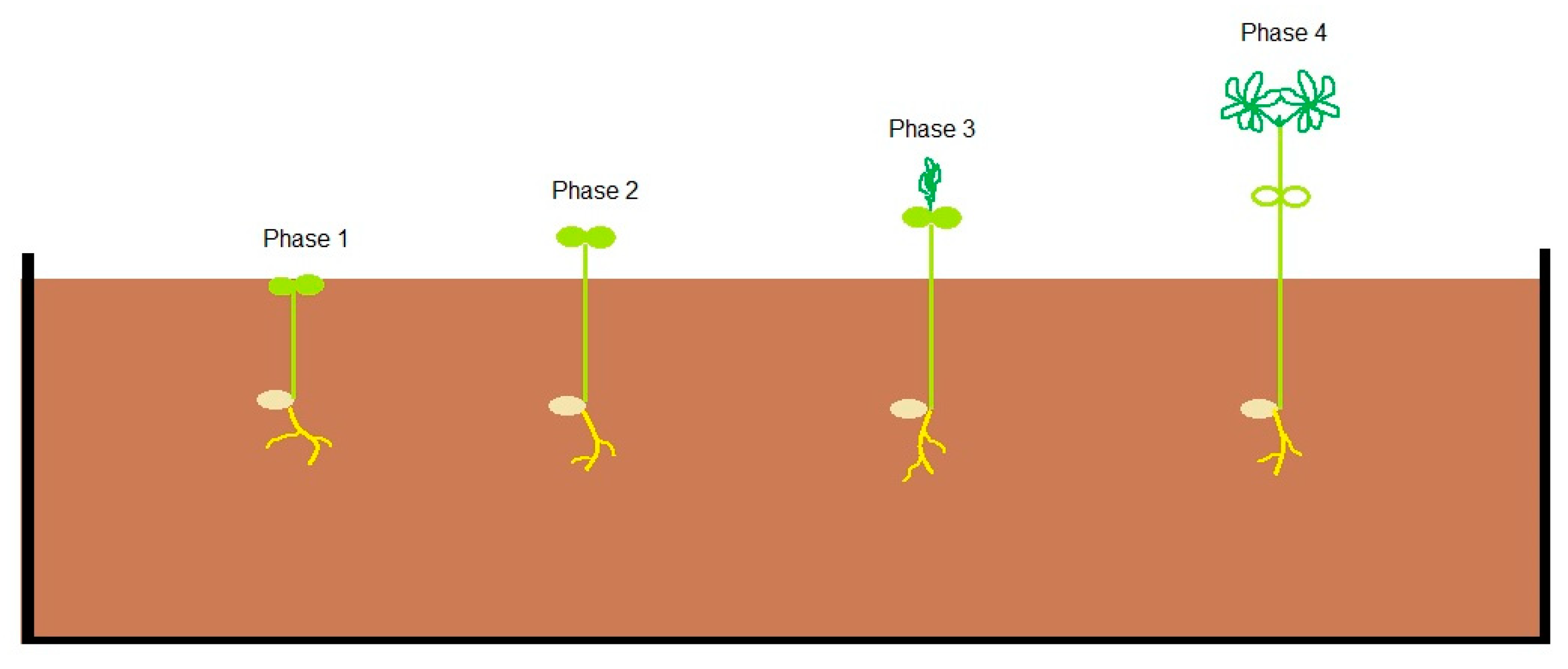

| Plant Developmental Phase | Test Temperature (Day/Night) |
|---|---|
| Phase 1—Cotyledons at breaking soil surface | −2 °C |
| −4 °C | |
| −6 °C | |
| −8 °C | |
| −10 °C | |
| 5 °C control | |
| Phase 2—Cotyledons fully above the soil | −2 °C |
| −4 °C | |
| −6 °C | |
| −8 °C | |
| −10 °C | |
| 5 °C control | |
| Phase 3—First leaves visible and erect but folded | −2 °C |
| −4 °C | |
| −6 °C | |
| −8 °C | |
| −10 °C | |
| 5 °C control | |
| Phase 4—First and second leaves have unfolded | −2 °C |
| −4 °C | |
| −6 °C | |
| −8 °C | |
| −10 °C | |
| 5 °C control |
| Df | Sum Sq | Mean Sq | F Value | p-Value (>F) | Significance | |
|---|---|---|---|---|---|---|
| Phase | 3 | 7024 | 2341 | 7229 | <0.0001 | *** |
| Temperature | 4 | 239224 | 59806 | 184,647 | <0.0001 | *** |
| Genotype | 3 | 14607 | 4869 | 15,033 | <0.0001 | *** |
| Phase × Temperature | 12 | 35840 | 2987 | 9221 | <0.0001 | *** |
| Phase × Genotype | 9 | 8418 | 935 | 2888 | 0.00256 | ** |
| Temperature × Genotype | 12 | 18455 | 1538 | 4748 | <0.0001 | *** |
| Phase x Temperature × Genotype | 36 | 18356 | 510 | 1574 | <0.0001 | * |
| Residuals | 400 | 129557 | 324 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Simioniuc, D.P.; Simioniuc, V.; Topa, D.; van den Berg, M.; Prins, U.; Bebeli, P.J.; Gabur, I. Assessment of Andean lupin (Lupinus mutabilis) Genotypes for Improved Frost Tolerance. Agriculture 2021, 11, 155. https://doi.org/10.3390/agriculture11020155
Simioniuc DP, Simioniuc V, Topa D, van den Berg M, Prins U, Bebeli PJ, Gabur I. Assessment of Andean lupin (Lupinus mutabilis) Genotypes for Improved Frost Tolerance. Agriculture. 2021; 11(2):155. https://doi.org/10.3390/agriculture11020155
Chicago/Turabian StyleSimioniuc, Danut Petru, Violeta Simioniuc, Denis Topa, Merlijn van den Berg, Udo Prins, Penelope J. Bebeli, and Iulian Gabur. 2021. "Assessment of Andean lupin (Lupinus mutabilis) Genotypes for Improved Frost Tolerance" Agriculture 11, no. 2: 155. https://doi.org/10.3390/agriculture11020155
APA StyleSimioniuc, D. P., Simioniuc, V., Topa, D., van den Berg, M., Prins, U., Bebeli, P. J., & Gabur, I. (2021). Assessment of Andean lupin (Lupinus mutabilis) Genotypes for Improved Frost Tolerance. Agriculture, 11(2), 155. https://doi.org/10.3390/agriculture11020155








