Antiarthritic Potential of Calotropis procera Leaf Fractions in FCA-Induced Arthritic Rats: Involvement of Cellular Inflammatory Mediators and Other Biomarkers
Abstract
1. Introduction
2. Materials and Methods
2.1. Procurement and Authentication of the Plant
2.2. Materials
2.3. Plant Extraction and Characterization
2.3.1. Extraction Method
2.3.2. Qualitative Phytochemical Screening
2.4. Biological Evaluation
2.4.1. Prediction of Biological Activity
2.4.2. Experimental Animals
2.4.3. Acute Toxicity Studies
2.4.4. FCA-Induced Arthritis
- Group I—Vehicle control, 1% w/v suspension of sodium carboxymethyl cellulose (SCMC) was administered orally;
- Group II—Arthritic control, 1% w/v suspension of SCMC was administered orally;
- Group III—Arthritic animals treated with oral administration of standard indomethacin at 10 mg/kg dose;
- Group IV—Arthritic animals treated with oral administration of standard ibuprofen at 15 mg/kg dose;
- Group V—Arthritic animals treated with oral administration of methanolic fraction of Swallow wort (MFCP) at a 300 mg/kg dose;
- Group VI—Arthritic animals treated with oral administration of ethyl acetate fraction of Swallow wort (EAFCP) at a 300 mg/kg dose.
2.4.5. Paw Volume Evaluation
2.4.6. Visual Arthritis Scoring System
2.4.7. Evaluation of Thermal Hyperalgesia
2.4.8. Body Weight Recording
2.4.9. Determination of the Weight of Spleen and Thymus
2.4.10. Biochemical Estimation
2.4.11. Estimation of Serum Parameters
2.4.12. Proinflammatory Biomarkers (TNF-α and IL-6)
2.4.13. Radiological Analysis of Ankle Joints
2.5. Data and Statistical Analysis
3. Results
3.1. Plant Extraction and Characterization
3.1.1. Extractive Value of Fraction
3.1.2. Phytochemical Screening
3.2. Biological Evaluation
3.2.1. PASS Prediction
3.2.2. FCA-Induced Arthritis
3.2.3. Effect of Swallow Wort Fractions on Paw Volume
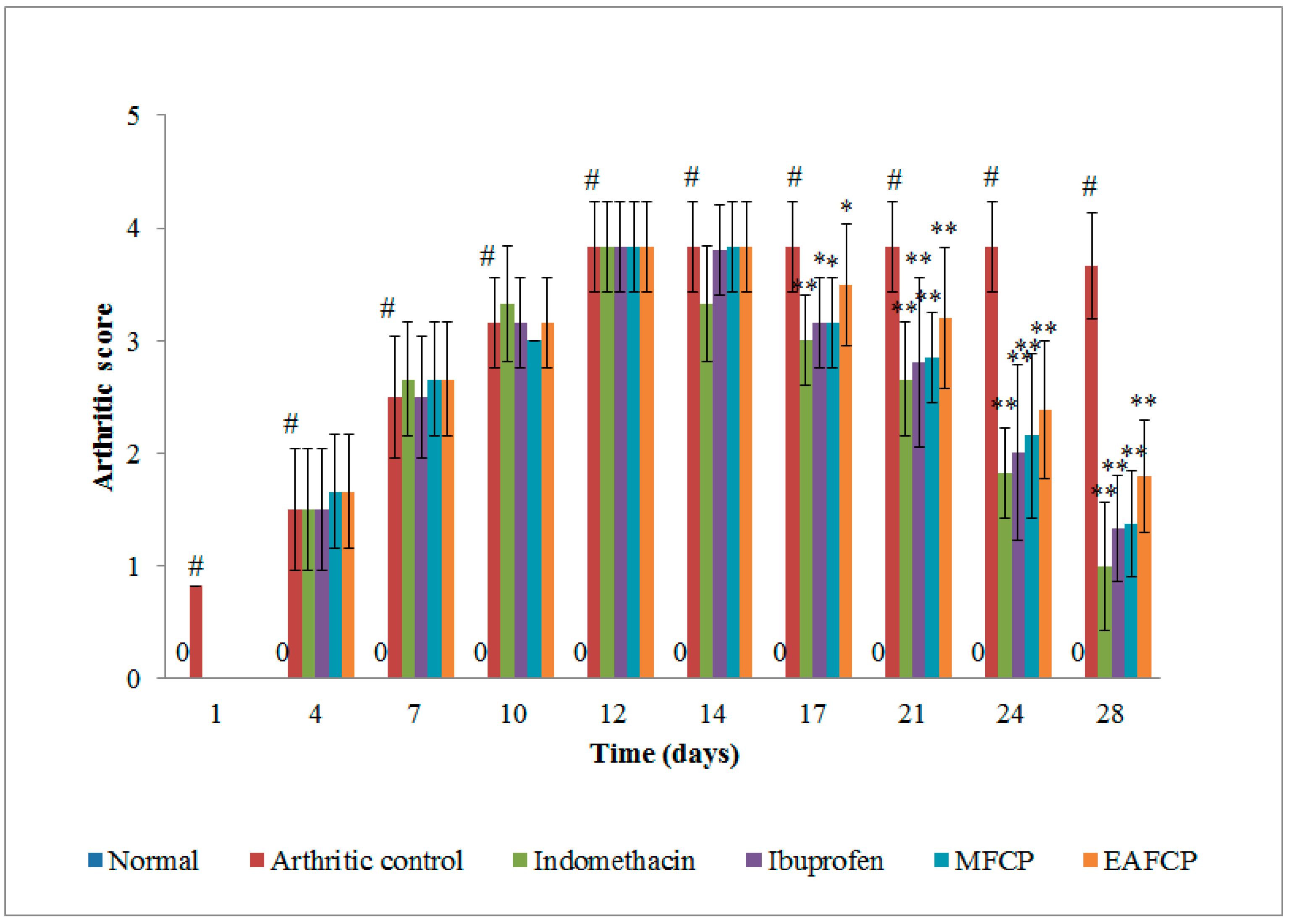
3.2.4. Arthritic Scoring System
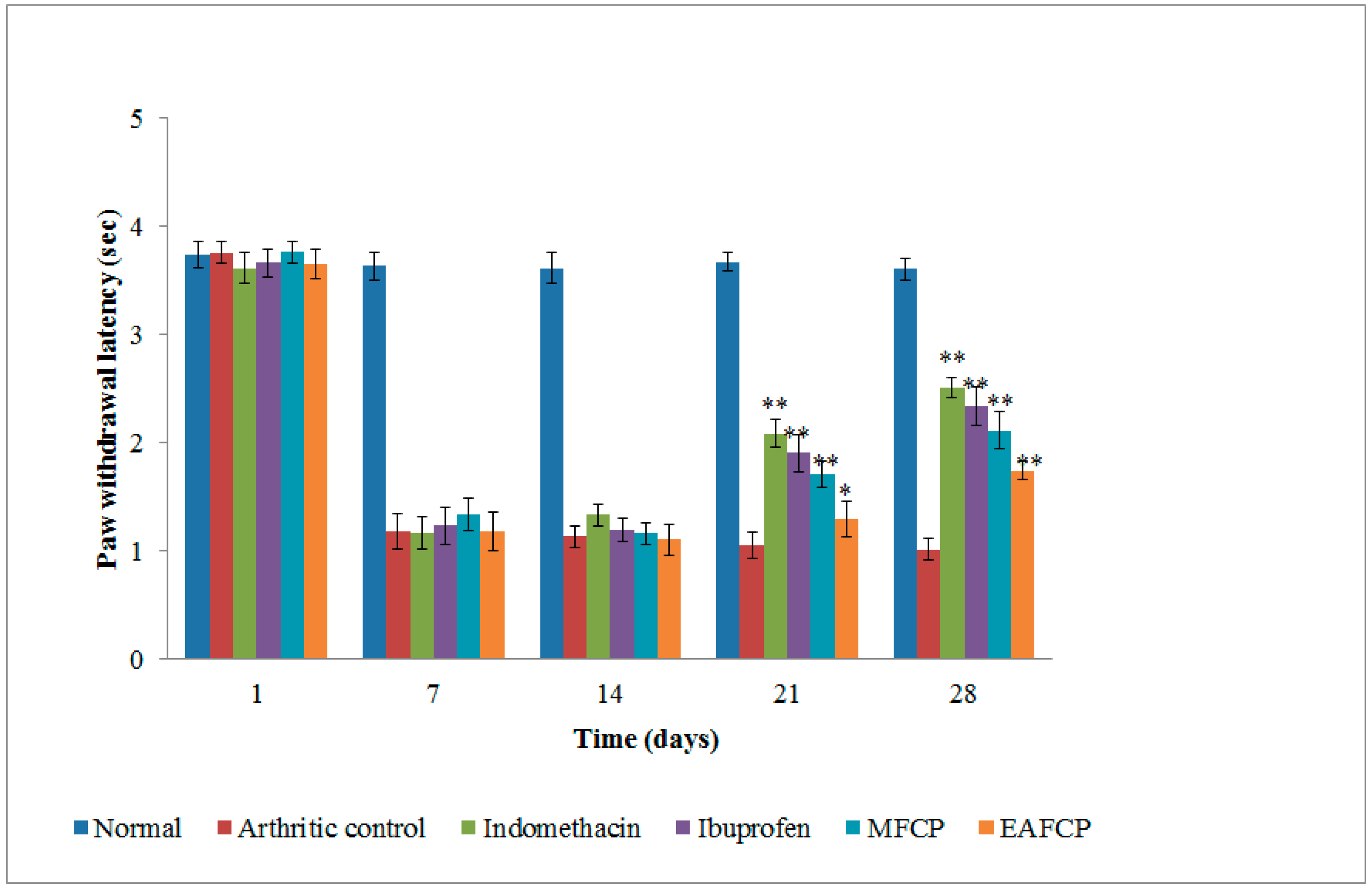
3.2.5. Measurement of Paw Withdrawal Latency
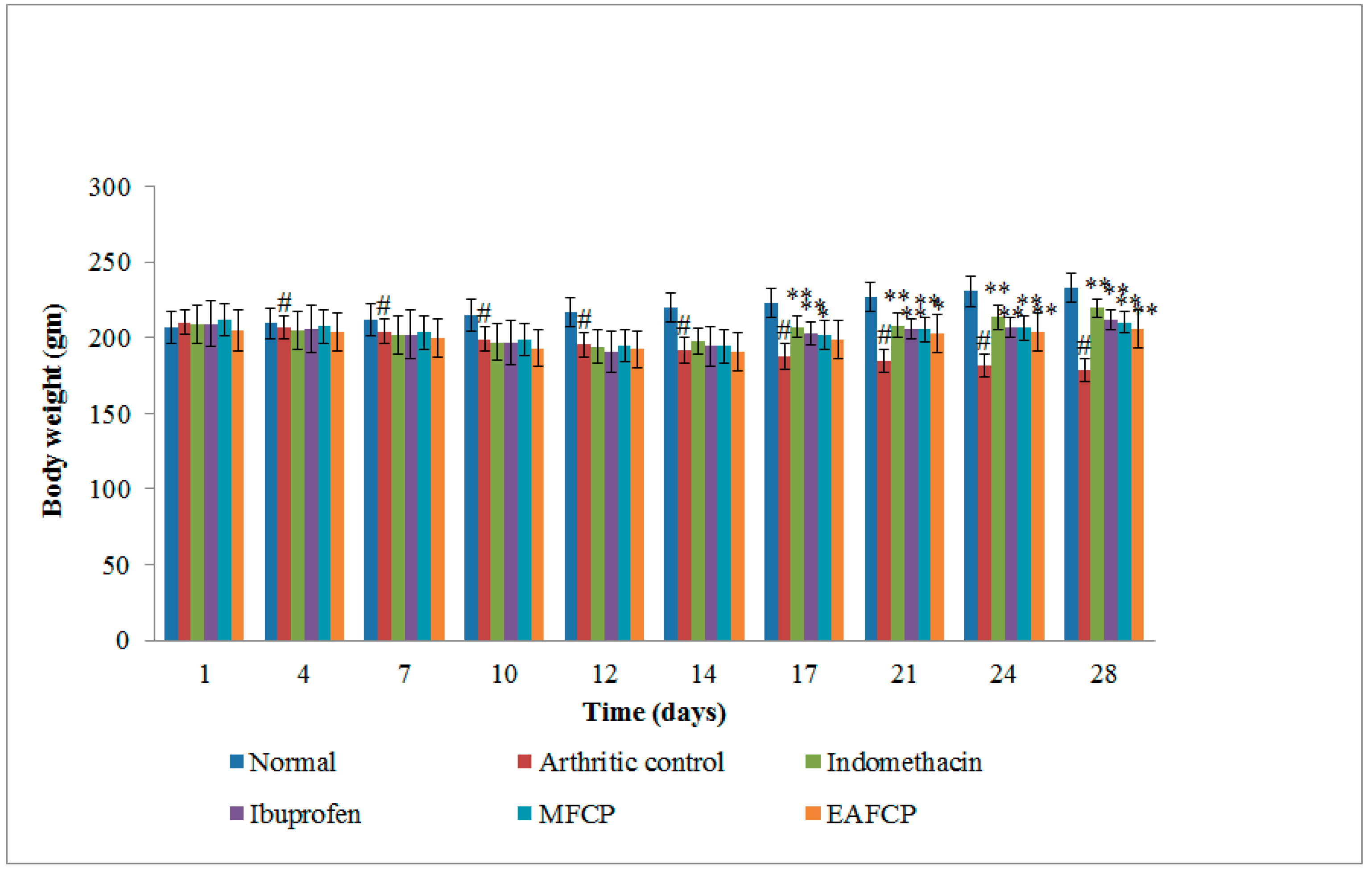
3.2.6. Body Weight
3.2.7. Measurement of Spleen and Thymus Weights
3.2.8. Serum Lysosomal Enzymes in FCA-Induced Arthritis
3.2.9. Alterations in CRP and RF in FCA-Induced Arthritis in Rats
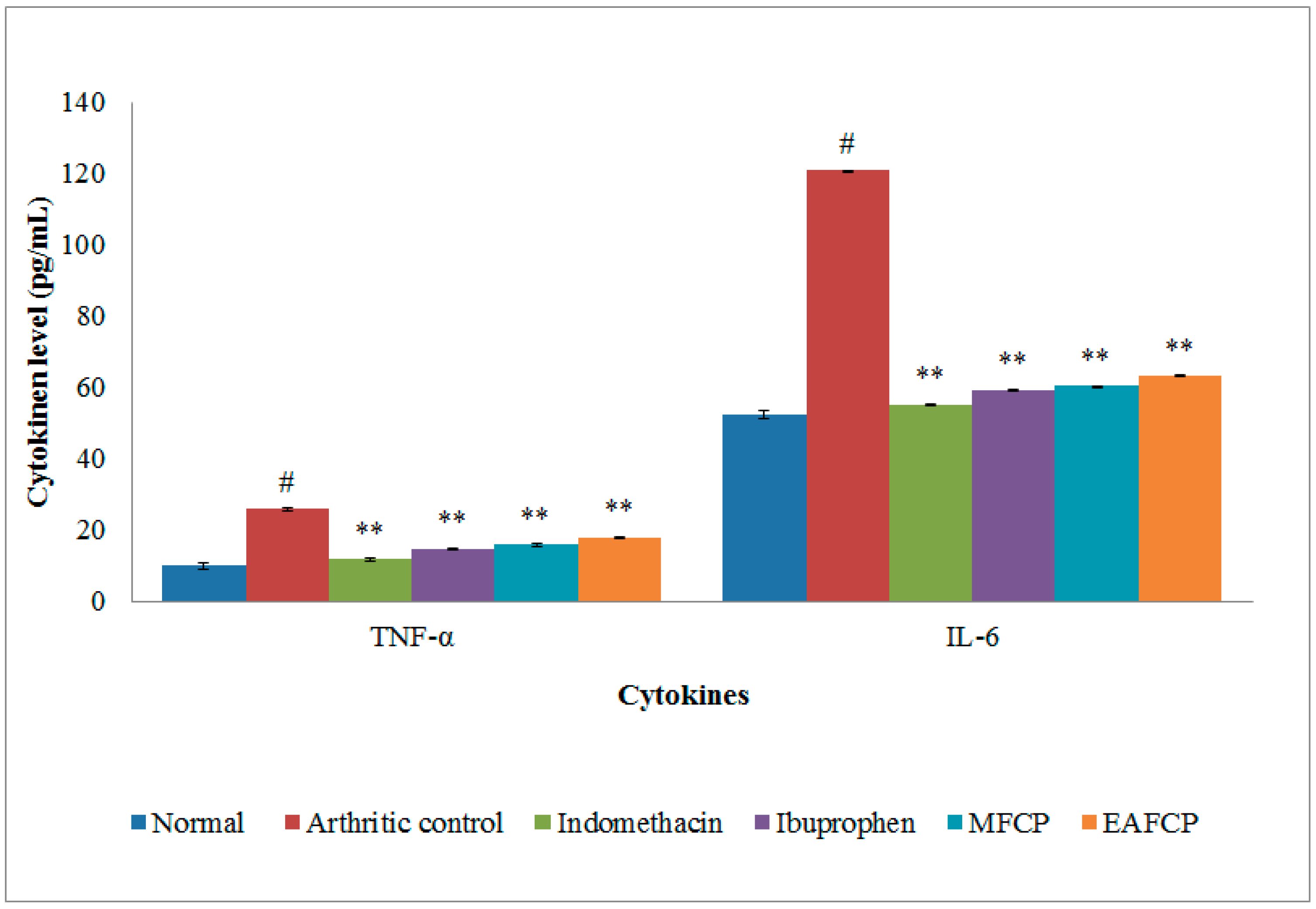
3.2.10. TNF-α and IL-6
3.2.11. Radiological Analysis of Ankle Joints
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Putterman, C. Introduction: New treatment paradigms in rheumatoid arthritis. Am. J. Orthopaed. 2006, 36, S2–S3. [Google Scholar]
- Tanaka, T. Introduction for inflammation and cancer. Sem. Immunopathol. 2013, 35, 121–122. [Google Scholar] [CrossRef] [PubMed]
- Bihania, G.V.; Rojatkar, S.R.; Bodhankar, S.L. Anti-arthritic activity of methanol extract of Cyathocline purpurea (whole plant) in Freund’s complete adjuvant-induced arthritis in rats. Biomed. Aging Pathol. 2014, 4, 197–206. [Google Scholar] [CrossRef]
- Reddy, D.; Trost, L.W.; Lee, T.; Baluch, A.R.; Kaye, A.D. Rheumatoid arthritis: Current pharmacologic treatment and anesthetic considerations. Middle East J. Anesthesiol. 2007, 19, 311–335. [Google Scholar]
- McInnes, I.B.; Schett, G. The pathogenesis of rheumatoid arthritis. N. Eng. J. Med. 2011, 365, 2205–2219. [Google Scholar] [CrossRef]
- Bhardwaj, L.K.; Chandrul, K.K.; Sharma, U.S. Evaluation of anti-arthritic activity of Ficus benghalensis Linn. root extracts on Freund’s adjuvant induced arthritis. J. Phytopharmacol. 2006, 5, 10–14. [Google Scholar]
- Fan, A.; Lao, L.; Zhang, R.; Zhou, A.; Wang, L.; Moudgil, K. Effects of an acetone extract of Boswellia carterii Birdw (Burseraceae) gum resin on adjuvant-induced arthritis in lewis rats. J. Ethnopharmacol. 2005, 101, 104–109. [Google Scholar] [CrossRef]
- Ofman, J.J.; Badamgarav, E.; Henning, J.M.; Knight, K.; Laine, L. Utilization of nonsteroidal anti-inflammatory drugs and anti secretory agents: A managed care claims analysis. Am. J. Med. 2004, 116, 835–842. [Google Scholar] [CrossRef]
- American college of rheumatology ad hoc Committee on clinical guidelines. Guidelines for the management of rheumatoid arthritis. Arth. Rheum. 1996, 39, 713–722. [Google Scholar] [CrossRef]
- Choudhary, M.; Kumar, V.; Gupta, P.; Singh, S. Investigation of antiarthritic potential of Plumeria alba L. leaves in acute and chronic models of arthritis. BioMed. Res. Int. 2014, 2014, 474616. [Google Scholar] [CrossRef]
- Choudhary, M.; Kumar, V.; Gupta, P.K.; Singh, S. Anti-arthritic activity of Barleria prionitis Linn. leaves in acute and chronic models in Sprague Dawley rats. Bull. Facul. Pharm. Cairo Univ. 2014, 52, 199–209. [Google Scholar] [CrossRef]
- Vedpal; Gupta, S.K.; Gupta, A.K.; Pakash, D.; Gupta, A. Anti-arthritic activity of Desmodium gangeticum root. Int. Res. J. Pharm. 2013, 4, 100–102. [Google Scholar]
- Gupta, R.K.; Hussain, T.; Panigrahi, G.; Das, A.; Singh, G.N.; Sweety, K.; Faiyazuddin, M.; Rao, C.V. Hepatoprotective effect of Solanum xanthocarpum fruit extract against CCl4 induced acute liver toxicity in experimental animals. Asian Pac. J. Trop. Med. 2011, 4, 964–968. [Google Scholar] [CrossRef]
- Perera, H.D.S.M.; Samarasekera, J.K.R.R.; Handunnetti, S.M.; Weerasena, O.V.D.S.J. In vitro anti-inflammatory and anti-oxidant activities of Sri Lankan medicinal plants. Ind. Crops Prod. 2016, 94, 610–620. [Google Scholar] [CrossRef]
- Mahboubi, M. Elaeagnus angustifolia and its therapeutic applications in osteoarthritis. Ind. Crops Prod. 2018, 121, 36–45. [Google Scholar] [CrossRef]
- Ali, B.; Mujeeb, M.; Aeri, V.; Mir, S.R.; Faiyazuddin, M.; Shakeel, F. Anti-inflammatory and antioxidant activity of Ficus carica Linn. leaves. Nat. Prod. Res. 2012, 26, 460–465. [Google Scholar] [CrossRef]
- Azhar, M.F.; Siddiqui, M.T.; Ishaque, M.; Tanveer, A. Study of ethnobotany and indegenous use of Calotropis procera (Ait.) in Cholistan desert, Punjab, Pakistan. J. Agric. Res. 2014, 52, 117–126. [Google Scholar]
- Meena, A.K.; Yadav, A.; Rao, M.M. Ayurvedic uses and pharmacological activities of Calotropis procera Linn. Asian J. Trad. Med. 2011, 6, 45–53. [Google Scholar]
- Panda, P.; Das, B.; Sahu, D.S.; Meher, S.K.; Das, B.K.; Rao, M.M.; Lakshmi, G.C.H.D.N. Important uses of arka (Calotropis procera Linn) in Indian system of medicine with pharmacological evidence. Res. J. Pharmacol. Pharmacodyn. 2015, 7, 46–49. [Google Scholar] [CrossRef]
- Biswasroy, P.; Panda, S.; Das, D.; Kar, D.M.; Ghosh, D. Pharmacological investigation of Calotropis gigantea: A benevolent herb of nature. Res. J. Pharm. Technol. 2020, 13, 461–467. [Google Scholar] [CrossRef]
- Ramachandran Setty, S.; Quereshi, A.A.; Viswanath Swamy, A.H.; Patil, T.; Prakash, T.; Prabhu, K.; Veeran Gouda, A. Hepatoprotective activity of Calatropis procera flowers against paracetamol-induced hepatic injury in rats. Fitoterapia 2007, 78, 451–454. [Google Scholar] [CrossRef] [PubMed]
- Moustafa, A.M.; Ahmed, S.H.; Nabil, Z.I.; Hussein, A.A.; Omran, M.A. Extraction and phytochemical investigation of Calotropis procera: Effect of plant extracts on the activity of diverse muscles. Pharm. Biol. 2010, 48, 1080–1190. [Google Scholar] [CrossRef] [PubMed]
- Nenaah, G.E. Potential of using flavonoids, latex and extracts from Calotropis procera (Ait.) as grain protectants against two coleopteran pests of stored rice. Ind. Crops Prod. 2013, 45, 327–334. [Google Scholar] [CrossRef]
- Kumar, V.L.; Basu, N. Anti-inflammatory activity of the latex Calotropis procera. J. Ethnopharmacol. 1994, 44, 123–125. [Google Scholar] [CrossRef]
- Iqbal, Z.; Lateef, M.; Jabbar, A.; Muhammad, G.; Khan, M.N. Anthelmintic activity of Calotropis procera (Ait.) Ait. F. flowers in sheep. J. Ethanopharmacol. 2005, 102, 256–261. [Google Scholar] [CrossRef] [PubMed]
- Kamath, J.V.; Rana, A.C. Pharmacological activities of ethanolic extract of Calotropis procera roots. Indian Drugs 2003, 40, 292–295. [Google Scholar]
- Kareem, S.O.; Akpan, I.; Ojo, O.P. Antimicrobial activities of Calotropis procera on selected pathogenic microorganisms. Afr. J. Biomed. Res. 2008, 11, 105–110. [Google Scholar] [CrossRef]
- Butler, S.H.; Godefroy, F.; Besson, J.M.; Weil-Fugazza, J. A limited arthritic model for chronic pain studies in the rat. Pain 1992, 48, 73–78. [Google Scholar] [CrossRef]
- Kumar, V.L.; Roy, S. Calotropis procera Latex extract affords protection against inflammation and oxidative stress in Freund’s complete adjuvant-induced monoarthritis in rats. Med. Inflamm. 2007, 2007, 47523. [Google Scholar] [CrossRef]
- Kumar, V.L.; Roy, S. Protective effect of latex of Calotropis procera in Freund’s complete adjuvant induced monoarthritis. Phytother. Res. 2009, 23, 1–5. [Google Scholar] [CrossRef]
- Sangraula, H.; Dewan, S.; Kumar, V.L. Evaluation of anti-inflammatory activity of latex of Calotropis procera in different models of inflammation. Inflammopharmacology 2002, 9, 257–264. [Google Scholar] [CrossRef]
- Kumar, V.L.; Chaudhary, P.; Ramos, M.V.; Moahn, M.; Matos, M.P.V. Protective effects of proteins derived latex of Calotropis procera against inflammatory hyperalgesia in monoarthritic rats. Phytother. Res. 2011, 25, 1336–1341. [Google Scholar] [CrossRef] [PubMed]
- Chaudhary, P.; Ramos, M.V.; Vasconcelos, M.D.S.; Kumar, V.L. Protective effects of high molecular weight protein sub-fraction of Calotropis procera latex in monoarthritic rats. Pharmacog. Mag. 2016, 112, S147–S151. [Google Scholar]
- Chandrasekar, R.; Chandrasekar, S. Natural herbal treatment for rheumatoid arthritis-a review. Int. J. Pharm. Sci. Res. 2017, 8, 368–384. [Google Scholar]
- Khandelwal, K.R. Practical Pharmacognosy; Nirali Prakashan: Pune, India, 2004; pp. 149–155. [Google Scholar]
- Parasuraman, S. Computer-aided prediction of biological activity spectra, pharmacological and toxicological properties of cleistanthin A and B. Int. J. Res. Pharm. Sci. 2010, 1, 333–337. [Google Scholar]
- Filimonov, D.A.; Lagunin, A.A.; Gloriozova, T.A.; Rudik, A.V.; Druzhilovskii, D.S.; Pogodin, P.V.; Poroikov, V.V. Prediction of the biological activity spectra of organic compounds using the pass online web resource. Chem. Het. Comp. 2014, 50, 444–457. [Google Scholar] [CrossRef]
- Jamkhande, P.G.; Barde, S.R. Evaluation of anthelmintic activity and in silico PASS assisted prediction of Cordia dichotoma (Forst.) root extract. Anc. Sci. Life 2014, 34, 39–43. [Google Scholar] [CrossRef]
- Sambavekar, P.P.; Aitawade, M.M.; Patil, D.R.; Kolekar, G.B.; Deshmukh, M.B.; Anbhule, P.V. In-silico, in-vitro antibacterial activity and toxicity profile of new quinoline derivatives. Indian J. Chem. 2013, 52, 1521–1526. [Google Scholar]
- Thite, A.T.; Patil, R.R.; Naik, S.R. Anti-arthritic activity profile of methanolic extract of Ficus bengalensis: Comparison with some clinically effective drugs. Biomed. Aging Pathol. 2014, 4, 207–217. [Google Scholar] [CrossRef]
- Kshirsagar, A.D.; Panchal, P.V.; Harle, U.N.; Nanda, R.K.; Shaikh, H.M. Anti-Inflammatory and antiarthritic activity of anthraquinone derivatives in rodents. Int. J. Inflamm. 2014, 2014, 690596. [Google Scholar] [CrossRef]
- Mali, S.M.; Sinnathambi, A.; Kapase, C.U.; Bodhankar, S.L.; Mahadik, K.R. Anti-arthritic activity of standardised extract of Phyllanthus amarus in Freund’s complete adjuvant induced arthritis. Biomed. Aging Pathol. 2011, 1, 185–190. [Google Scholar] [CrossRef]
- Arya, S.; Kumar, V.L. Anti-inflammatory efficacy of extracts of latex of Calotropis procera against different mediators of inflammation. Med. Inflamm. 2005, 4, 228–232. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.L.; Roy, S.; Sehgal, R.; Padhy, B.M. A comparative study on the efficacy of rofecoxib in monoarticular arthritis induced by latex of calotropis procera and freund’s complete adjuvant. Inflammopharmacology 2006, 14, 17–21. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.Q.; Huang, X.D.; Wang, H.; Leung, A.K.; Chan, C.L.; Fong, D.W.; Yu, Z.L. Anti-inflammatory and analgesic effects of the ethanol extract of Rosa multiflora Thunb. hips. J. Ethnopharmacol. 2008, 118, 290–294. [Google Scholar] [CrossRef]
- Asquith, D.L.; Miller, A.M.; McInnes, I.B.; Liew, F.Y. Animal models of rheumatoid arthritis. Eur. J. Immunol. 2009, 39, 2040–2044. [Google Scholar] [CrossRef]
- Mythilypriya, R.; Shanthi, P.; Sachdanandam, P. Salubrious effect of Kalpaamruthaa, a modified indigenous preparation in adjuvant-induced arthritis in rats–a biochemical approach. Chem. Biol. Inter. 2008, 173, 148–158. [Google Scholar] [CrossRef]
- Pawar, S.; Patil, D.A. Folk remedies against rheumatic disorders in Jalgaon district, Maharashtra. Indian J. Trad. Know. 2006, 5, 314–316. [Google Scholar]
- Singh, V.S.; Dhawale, S.C.; Shakeel, F.; Faiyazuddin, M.; Alshehri, S. In vitro and in vivo anti-inflammatory effects of Calotropis procera leave fractions. unpublished work, manuscript in preparation.
- Sameeh, M.Y.; Mohamed, A.A. Characterization of polyphenols, their antioxidant and GC-MS analysis of wild Calotropis procera leaves and fruit extracts. Int. J. Chem. Tech. Res. 2018, 11, 319–327. [Google Scholar]
- Wu, C.R.; Hseu, Y.C.; Lien, J.C.; Lin, L.W.; Lin, Y.T.; Ching, H. Triterpenoid contents and anti-inflammatory properties of the methanol extracts of Ligustrum species leaves. Molecules 2011, 16, 1–15. [Google Scholar] [CrossRef]
- Mallick, S.S.; Dighe, V.V. Detection and estimation of alpha-amyrin, beta-sitosterol, lupeol, and n-triacontane in two medicinal plants by high performance thin layer chromatography. Adv. Chem. 2014, 2014, 143948. [Google Scholar] [CrossRef]
- Tuncel, J.; Haag, S.; Hoffmann, M.H.; Yau, A.C.Y.; Hulqvist, M.; Olofsson, P.; Backlund, J.; Nandakumar, K.S.; Weidner, D.; Fischer, A.; et al. Animal models of rheumatoid arthritis (I): Pristine-induced arthritis in the rat. PLoS ONE 2016, 11, e0155936. [Google Scholar] [CrossRef] [PubMed]
- Patil, K.R.; Patil, C.R.; Jadhav, R.B.; Mahajan, V.K.; Raosaheb, P.; Gaikwad, P.S. Anti-arthritic activity of bartogenic acid isolated from fruits of Barringtonia racemosa Roxb. (Lecythidaceae). Evid.-Based Complement. Altern. Med. 2011, 2011, 785245. [Google Scholar] [CrossRef] [PubMed]
- Petchi, R.R.; Parasuraman, S.; Vijaya, C.; Gopala Krishna, S.V.; Kumar, M.K. Antiarthritic activity of a polyherbal formulation against Freund’s complete adjuvant induced arthritis in female Wistar rats. J. Basic Clin. Pharmacol. 2015, 6, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Jalalpure, S.S.; Mandavkar, Y.D.; Khalure, P.R.; Shinde, G.S.; Shelar, P.A.; Shah, A.S. Antiarthritic activity of various extracts of Mesua ferrea Linn. Seed. J. Ethnopharmacol. 2011, 138, 700–704. [Google Scholar] [CrossRef] [PubMed]
- Kubo, M.; Matsuda, H.; Tanaka, M.; Kimura, Y.; Okuda, H.; Higashino, M.; Tani, T.; Namba, K.; Aaichi, S. Studies on scutellariaradix.VII. anti-arthritic and anti-inflammatory action of methanolic extract and flavonoid components from scutellariae radix. Chem. Pharm. Bull. 1984, 32, 2724–2729. [Google Scholar] [CrossRef] [PubMed]
- Prasad, P.J. Conceptual Pharmacology; Universities Press: Hydrabad, India, 2010; pp. 254–268. [Google Scholar]
- Ekambaram, S.; Perumal, S.S.; Subramanian, V. Evaluation of anti-arthritic activity of Strychnos potatorum Linn seeds in Freund’s adjuvant induced arthritic rat model. BMC Complement. Altern. Med. 2010, 10, 56. [Google Scholar] [CrossRef]
- Zhang, X.; Dong, Y.; Li, F. Inevestigation of the effect of phlomisoside F on complete Freund’s adjuvant-inducced arthritis. Exp. Ther. Med. 2017, 12, 710–718. [Google Scholar] [CrossRef]
- Mbiantcha, M.; Almas, J.; Shabana, S.U.; Nida, D.; Aisha, F. Anti-arthritic property of crude extracts of Piptadeniastrum africanum (Mimosaceae) in complete Freund’s adjuvant-induced arthritis in rats. BMC Complment. Altern. Med. 2017, 17, E111. [Google Scholar] [CrossRef]
- Perumal, S.S.; Ekambaram, P.S.; Dhanam, T. In vivo antiarthritic activity of the ethanol extracts of stem bark and seeds of Calophyllum inophyllum in Freund’s complete adjuvant induced arthritis. Pharm. Biol. 2017, 55, 1330–1336. [Google Scholar] [CrossRef]
- Almarestani, L.; Fitzcharles, M.A.; Bennett, G.J.; Ribeiro-da-Silva, A. Imaging studies in Freund’s complete adjuvant model of regional polyarthritis, a model suitable for the study of pain mechanisms, in the rat. Arth. Rheum. 2011, 63, 1573–1581. [Google Scholar] [CrossRef] [PubMed]

| Phytoconstituent | Pa | Pi | Activity |
|---|---|---|---|
| α-amyrin | 0.889 | 0.004 | Anti-inflammatory |
| 0.835 | 0.002 | Nitric oxide antagonist | |
| 0.522 | 0.043 | Antiarthritic | |
| β-amyrin | 0.411 | 0.011 | Antioxidant |
| 0.843 | 0.005 | Anti-inflammatory | |
| 0.793 | 0.003 | Nitric oxide antagonist | |
| Calitropigenin | 0.405 | 0.012 | Antioxidant |
| 0.357 | 0.119 | Anti-inflammatory | |
| 0.490 | 0.060 | Anti-inflammatory | |
| Asclepin | 0.357 | 0.119 | Anti-inflammatory |
| 0.490 | 0.060 | Anti-inflammatory | |
| 0.608 | 0.005 | Calcium regulator | |
| β-sitosterol | 0.572 | 0.038 | Anti-inflammatory |
| 0.482 | 0.004 | Anti-inflammatory, Ophthalmic | |
| 0.740 | 0.011 | Anti-inflammatory | |
| Stigmasterol | 0.669 | 0.004 | Calcium regulator |
| 0.662 | 0.006 | Bone diseases treatment | |
| 0.373 | 0.017 | Anti-inflammatory, ophthalmic |
| Groups | Paw Volume on Different Days (mL) | Inhibition (%) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 4th | 7th | 10th | 12th | 14th | 17th | 21st | 24th | 28th | ||
| Normal | 0.18 ± 0.009 | 0.19 ± 0.012 | 0.2 ± 0.008 | 0.20 ± 0.008 | 0.2 ± 0.08 | 0.19 ± 0.01 | 0.2 ± 0.02 | 0.21 ± 0.01 | 0.21 ± 0.008 | _ |
| Arthritic control | 0.38 ± 0.011 # | 0.50 ± 0.012 # | 0.59 ± 0.008 # | 0.61 ± 0.01 # | 0.59 ± 0.008 # | 0.59 ± 0.008 # | 0.58 ± 0.005 # | 0.57 ± 0.005 # | 0.56 ± 0.004 # | _ |
| Indomethacin | 0.38 ± 0.009 | 0.51 ± 0.012 | 0.59 ± 0.014 | 0.61 ± 0.01 | 0.58 ± 0.004 | 0.42 ± 0.005 ** | 0.38 ± 0.005 ** | 0.34 ± 0.006 ** | 0.27 ± 0.01 ** | 51.78 |
| Ibuprofen | 0.39 ± 0.011 | 0.51 ± 0.012 | 0.59 ± 0.008 | 0.60 ± 0.012 | 0.58 ± 0.012 | 0.44 ± 0.01 ** | 0.42 ± 0.008 ** | 0.37 ± 0.008 ** | 0.3 ± 0.008 ** | 46.42 |
| MFCP | 0.38 ± 0.019 | 0.51 ± 0.012 | 0.58 ± 0.006 | 0.59 ± 0.012 | 0.58 ± 0.009 | 0.44 ± 0.005 ** | 0.42 ± 0.007 ** | 0.38 ± 0.01 ** | 0.30 ± 0.008 ** | 46.42 |
| EAFCP | 0.39 ± 0.009 | 0.51 ± 0.012 | 0.59 ± 0.012 | 0.60 ± 0.011 | 0.59 ± 0.008 | 0.58 ± 0.007 | 0.56 ± 0.008 * | 0.49 ± 0.007 ** | 0.41 ± 0.008 ** | 26.78 |
| Groups | Organs Weight (g) | |
|---|---|---|
| Spleen | Thymus | |
| Normal | 0.21 ± 0.01 | 0.11 ± 0.01 |
| Arthritic control | 0.44 ± 0.012 # | 0.2 ± 0.011 # |
| Indomethacin 10 mg/kg | 0.29 ± 0.012 ** | 0.13 ± 0.016 ** |
| Ibuprofen15 mg/kg | 0.37 ± 0.01 ** | 0.14 ± 0.017 ** |
| MFCP 300 mg/kg | 0.33 ± 0.014 ** | 0.13 ± 0.017 ** |
| EAFCP 300 mg/kg | 0.41 ± 0.024 ** | 0.18 ± 0.008 ** |
| Groups | Serum Enzymes on Different Days | ||
|---|---|---|---|
| AST (U/mL) | ALT (U/mL) | ALP (U/mL) | |
| Normal | 32.33 ± 0.81 | 26.16 ± 0.98 | 42.83 ± 1.72 |
| Arthritic control | 76.66 ± 1.63 # | 74 ± 1.6 # | 122.66 ± 1.63 # |
| Indomethacin 10 mg/kg | 38.16 ± 1.72 ** | 35.66 ± 1.3 ** | 42.33 ± 1.36 ** |
| Ibuprofen15 mg/kg | 42.33 ± 1.36 ** | 38.66 ± 1.03 ** | 55.83 ± 1.16 ** |
| MFCP 300 mg/kg | 45.16 ± 0.75 ** | 41.83 ± 0.75 ** | 56 ± 1.78 ** |
| EAFCP 300 mg/kg | 49.66 ± 1.5 ** | 47.5 ± 1.04 ** | 62.16 ± 1.47 ** |
| Groups | RF (IU/L) | CRP (mg/L) |
|---|---|---|
| Normal control | - | 1.19 ± 0.3 |
| Arthritic control | 59.71 ± 1.18 # | 7.1 ± 0.48 # |
| Indomethacin (10 mg/kg) | 34.55 ± 0.96 ** | 2.41 ± 0.51 ** |
| Ibuprofen (15 mg/kg) | 37.08 ± 0.91 ** | 2.93 ± 0.51 ** |
| MFCP 300 mg/kg | 46.03 ± 1.25 ** | 4.1 ± 0.49 ** |
| EAFCP 300 mg/kg | 49.34 ± 1.21 ** | 6.38 ± 0.34 ** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Singh, V.S.; Dhawale, S.C.; Shakeel, F.; Faiyazuddin, M.; Alshehri, S. Antiarthritic Potential of Calotropis procera Leaf Fractions in FCA-Induced Arthritic Rats: Involvement of Cellular Inflammatory Mediators and Other Biomarkers. Agriculture 2021, 11, 68. https://doi.org/10.3390/agriculture11010068
Singh VS, Dhawale SC, Shakeel F, Faiyazuddin M, Alshehri S. Antiarthritic Potential of Calotropis procera Leaf Fractions in FCA-Induced Arthritic Rats: Involvement of Cellular Inflammatory Mediators and Other Biomarkers. Agriculture. 2021; 11(1):68. https://doi.org/10.3390/agriculture11010068
Chicago/Turabian StyleSingh, Vandana S., Shashikant C. Dhawale, Faiyaz Shakeel, Md. Faiyazuddin, and Sultan Alshehri. 2021. "Antiarthritic Potential of Calotropis procera Leaf Fractions in FCA-Induced Arthritic Rats: Involvement of Cellular Inflammatory Mediators and Other Biomarkers" Agriculture 11, no. 1: 68. https://doi.org/10.3390/agriculture11010068
APA StyleSingh, V. S., Dhawale, S. C., Shakeel, F., Faiyazuddin, M., & Alshehri, S. (2021). Antiarthritic Potential of Calotropis procera Leaf Fractions in FCA-Induced Arthritic Rats: Involvement of Cellular Inflammatory Mediators and Other Biomarkers. Agriculture, 11(1), 68. https://doi.org/10.3390/agriculture11010068






