Polyphenols and Resveratrol from Discarded Leaf Biomass of Grapevine (Vitis sp.): Effect of Cultivar and Viticultural Practices in Estonia
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Sites and Plant Material
2.2. Analyses of Total Phenolic Content (TPC) and Resveratrol
2.3. Statistical Analysis
3. Results and Discussion
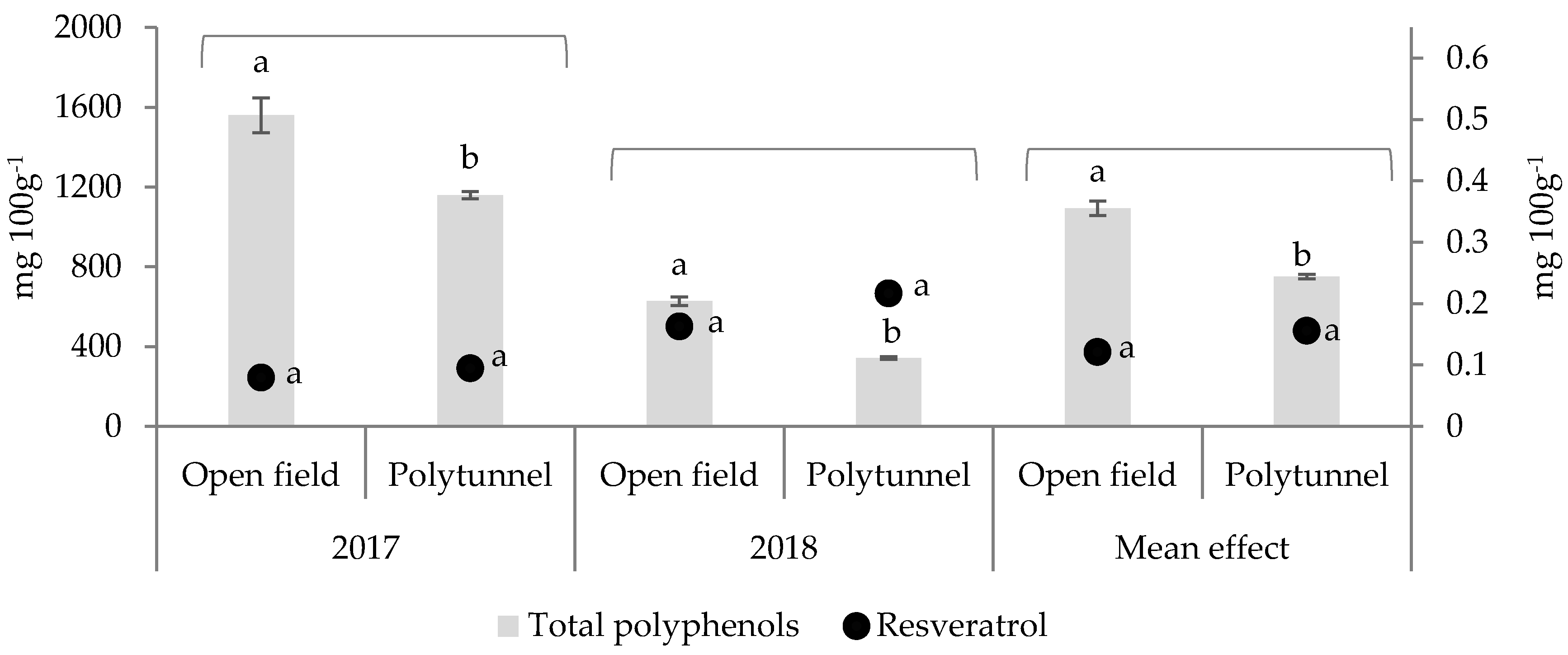
3.1. Effect of Cultivation under Polytunnel Conditions on TPC and Resveratrol
3.2. Effect of Harvest Time under Open Field Conditions
3.3. Effect of the Cultivation Site on the Content of Polyphenols and Resveratrol in ‘Rondo’
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lorenz, D.H.; Eichhorn, K.W.; Bleiholder, H.; Klose, R.; Meier, U.; Weber, E. Phänologische entwicklungsstadien der weinrebe (Vitis vinifera L. ssp. vinifera). Wein Wiss. 1994, 49, 66–70. [Google Scholar] [CrossRef]
- Lima, A.; Bento, A.; Baraldi, I.; Malheiro, R. Selection of grapevine leaf varieties for culinary process based on phytochemical composition and antioxidant properties. Food Chem. 2016, 212, 291–295. [Google Scholar] [CrossRef] [PubMed]
- Doshi, P.; Adsule, P.; Banerjee, K. Phenolic composition and antioxidant activity in grapevine parts and berries (Vitis vinifera L.) cv. kishmish chornyi (shared seedless) during maturation. Int. J. Food Sci. Technol. 2006, 41, 1–9. [Google Scholar] [CrossRef]
- Zwingelstein, M.; Draye, M.; Besombes, J.L.; Piot, C.; Chatel, G. Viticultural wood waste as a source of polyphenols of interest: Opportunities and perspectives through conventional and emerging extraction methods. Waste Manag. 2020, 102, 782–794. [Google Scholar] [CrossRef] [PubMed]
- Pachón, E.R.; Mandade, P.; Gnansounou, E. Conversion of vine shoots into bioethanol and chemicals: Prospective LCA of biorefinery concept. Bioresour. Technol. 2020, 303, 122946. [Google Scholar] [CrossRef]
- Jeandet, P.; Clément, C.; Cordelier, S. Regulation of resveratrol biosynthesis in grapevine: New approaches for disease resistance? J. Exp. Bot. 2019, 70, 375–378. [Google Scholar] [CrossRef]
- Kocsis, M.; Abrankó, L.; Ayaydin, F.; Csepregi, K.; Papp, N.; Teszlák, P.; Jakab, G. Main leaf polyphenolic components of berry color variant grapevines and their acclimative responses to sunlight exposure. Appl. Sci. 2015, 5, 1955–1969. [Google Scholar] [CrossRef]
- Yin, X.; Singer, S.D.; Qiao, H.; Liu, Y.; Jiao, C.; Wang, H.; Li, Z.; Fei, Z.; Wang, Y.; Fan, C.; et al. Insights into the mechanisms underlying ultraviolet-c induced resveratrol metabolism in grapevine (V. amurensis rupr.) cv. “Tonghua-3”. Front. Plant Sci. 2016, 7, 1–16. [Google Scholar] [CrossRef]
- De Souza, C.R.; Da Mota, R.V.; Dias, F.A.N.; De Melo, E.T.; Pimentel, R.M.D.A.; De Souza, L.C.; Regina, M.D.A. Physiological and agronomical responses of Syrah grapevine under protected cultivation. Bragantia 2015, 74, 270–278. [Google Scholar] [CrossRef]
- Katalinić, V.; Generalić, I.; Skroza, D.; Ljubenkov, I.; Teskera, A.; Konta, I.; Boban, M. Insight in the phenolic composition and antioxidative properties of Vitis vinifera leaves extracts. Croat. J. Food Sci. Technol. 2009, 1, 7–15. [Google Scholar] [CrossRef]
- Pantelić, M.M.; Zagorac, D.Č.D.; Ćirić, I.; Pergal, M.V.; Relić, D.J.; Todić, S.R.; Natić, M.M. Phenolic profiles, antioxidant activity and minerals in leaves of different grapevine varieties grown in Serbia. J. Food Compos. Anal. 2017, 62, 76–83. [Google Scholar] [CrossRef]
- Acquadro, S.; Appleton, S.; Marengo, A.; Bicchi, C.; Sgorbini, B.; Mandrone, M.; Gai, F.; Peiretti, P.G.; Cagliero, C.; Rubiolo, P. Grapevine green pruning residues as a promising and sustainable source of bioactive phenolic compounds. Molecules 2020, 25, 464. [Google Scholar] [CrossRef] [PubMed]
- Eftekhari, M.; Yadollahi, A.; Ford, C.M.; Shojaeiyan, A.; Ayyari, M.; Hokmabadi, H. Chemodiversity evaluation of grape (Vitis vinifera) vegetative parts during summer and early fall. Ind. Crop. Prod. 2017, 108, 267–277. [Google Scholar] [CrossRef]
- Kedrina-Okutan, O.; Novello, V.; Hoffmann, T.; Hadersdorfer, J.; Schneider, A.; Schwab, W.; Ferrandino, A. Polyphenolic diversity in Vitis sp. leaves. Sci. Hortic. 2019, 256, 108569. [Google Scholar] [CrossRef]
- Martín-Tornero, E.; de Jorge Páscoa, R.N.M.; Espinosa-Mansilla, A.; Martín-Merás, I.D.; Lopes, J.A. Comparative quantification of chlorophyll and polyphenol levels in grapevine leaves sampled from different geographical locations. Sci. Rep. 2020, 10, 1–13. [Google Scholar] [CrossRef]
- Wang, L.; Xu, M.; Liu, C.; Wang, J.; Xi, H.; Wu, B.; Loescher, W.; Duan, W.; Fan, P.; Li, S. Resveratrols in grape berry skins and leaves in vitis germplasm. PLoS ONE 2013, 8, e61642. [Google Scholar] [CrossRef]
- Bábíková, P.; Vrchotová, N.; Tříska, J.; Kyseláková, M. Content of trans-resveratrol in leaves and berries of interspecific grapevine (Vitis sp.) varieties. Czech J. Food Sci. 2009, 26, S13–S17. [Google Scholar] [CrossRef]
- Lambert, M.; Meudec, E.; Verbaere, A.; Mazerolles, G.; Wirth, J.; Masson, G.; Cheynier, V.; Sommerer, N. A high-throughput UHPLC-QqQ-MS method for polyphenol profiling in rosé wines. Molecules 2015, 20, 7890–7914. [Google Scholar] [CrossRef]
- Fernandes, F.; Ramalhosa, E.; Pires, P.; Verdial, J.; Valentão, P.; Andrade, P.; Bento, A.; Pereira, J.A. Vitis vinifera leaves towards bioactivity. Ind. Crop. Prod. 2013, 43, 434–440. [Google Scholar] [CrossRef]
- Maante-Kuljus, M.; Vool, E.; Mainla, L.; Starast, M.; Karp, K. Berry quality of hybrid grapevine (vitis) cultivars grown in the field and in a polytunnel. Agric. Food Sci. 2019, 28, 137–144. [Google Scholar] [CrossRef]
- Fernandes De Oliveira, A.; Mercenaro, L.; Nieddu, G. Assessing thermal efficiency for berry anthocyanin accumulation in four different sites and field-growing conditions. Acta Hortic. 2017, 1188, 181–188. [Google Scholar] [CrossRef]
- Cohen, S.D.; Tarara, J.M.; Gambetta, G.A.; Matthews, M.A.; Kennedy, J.A. Impact of diurnal temperature variation on grape berry development, proanthocyanidin accumulation, and the expression of flavonoid pathway genes. J. Exp. Bot. 2012, 63, 2655–2665. [Google Scholar] [CrossRef] [PubMed]
- Wojdyło, A.; Samoticha, J.; Nowicka, P.; Chmielewska, J. Characterisation of (poly)phenolic constituents of two interspecific red hybrids of rondo and regent (Vitis vinifera) by LC–PDA–ESI-MS QTof. Food Chem. 2018, 239, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Kriedemann, P.E.; Kuewer, W.M.; Harris, J.M. Leaf age and photosynthesis in Vitis vinifera L. Vitis 1970, 9, 97–104. [Google Scholar]
- Ferrandino, A.; Lovisolo, C. Abiotic stress effects on grapevine (Vitis vinifera L.): Focus on abscisic acid-mediated consequences on secondary metabolism and berry quality. Environ. Exp. Bot. 2014, 103, 138–147. [Google Scholar] [CrossRef]
- Kolb, C.A.; Pfündel, E.E. Origins of non-linear and dissimilar relationships between epidermal UV absorbance and UV absorbance of extracted phenolics in leaves of grapevine and barley. Plant Cell Environ. 2005, 28, 580–590. [Google Scholar] [CrossRef]
| Mean Temperatures, °C | Precipitation, mm | |||||||
|---|---|---|---|---|---|---|---|---|
| Open Field | Polytunnel | Open Field | ||||||
| Month(s)/Year(s) | 2017 | 2018 | 1981–2010 | 2017 | 2018 | 2017 | 2018 | 1981–2010 |
| April | 3.4 | 7.2 | 5.3 | 4.6 | 10.2 | 68 | 43 | 36 |
| May | 10.2 | 15.2 | 11.3 | 16.1 | 20.1 | 28 | 10 | 48 |
| June | 13.8 | 15.5 | 14.9 | 17.1 | 20.4 | 65 | 66 | 87 |
| July | 15.7 | 20.2 | 17.5 | 20.6 | 23.6 | 57 | 23 | 83 |
| August | 16.5 | 18.5 | 16.1 | 18.8 | 20.8 | 112 | 80 | 91 |
| September | 12.1 | 14.0 | 11.0 | 12.7 | 15.6 | 119 | 99 | 68 |
| October | 5.2 | 7.2 | 6.0 | 6.0 | 8.6 | 86 | 78 | 81 |
| 2017 | 2018 | |||||
|---|---|---|---|---|---|---|
| Cultivar | TPC, mg 100 g−1 fw | Resveratrol, mg 100 g−1 fw | TPC, mg 100 g−1 fw | Resveratrol, mg 100 g−1 fw | ||
| ‘Rondo’ | 1159 b | 0.095 b | 342 b | 0.216 a | ||
| ‘Regent’ | 1944 a | 0.272 a | 427 b | 0.235 a | ||
| ‘Boskoop’s Glory’ | 1237 b | 0.063 b | 328 b | 0.282 a | ||
| ‘Solaris’ | 1139 b | 0.116 b | 553 a | 0.178 a | ||
| TPC, mg 100 g−1 fw | Resveratrol, mg 100 g−1 fw | |||||
| Mean effect of year | *** | * | ||||
| Mean effect of cultivar | *** | * | ||||
| Leaf-Harvest Time | 2017 | 2018 | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Cultivar | TPC, mg 100 g−1 fw | Resveratrol, mg 100 g−1 fw | TPC, mg 100 g−1 fw | Resveratrol, mg 100 g−1 fw | |||||
| Stage 85 | ‘Zilga’ | 1264 b | 0.167 b | 351 c | 0.206 c | ||||
| Stage 89 | ‘Zilga’ | 1841 a | 1.061 a | 464 a | 1.971 a | ||||
| Stage 85 | ‘Hasansky Sladky’ | 1163 b | 0.222 b | 395 abc | 0.274 c | ||||
| Stage 89 | ‘Hasansky Sladky’ | 1213 b | 0.882 a | 444 ab | 0.678 b | ||||
| Mean effect of leaf harvest time | *** | ** | * | *** | |||||
| Mean effect of cultivar | *** | - | - | *** | |||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rätsep, R.; Karp, K.; Maante-Kuljus, M.; Aluvee, A.; Bhat, R. Polyphenols and Resveratrol from Discarded Leaf Biomass of Grapevine (Vitis sp.): Effect of Cultivar and Viticultural Practices in Estonia. Agriculture 2020, 10, 393. https://doi.org/10.3390/agriculture10090393
Rätsep R, Karp K, Maante-Kuljus M, Aluvee A, Bhat R. Polyphenols and Resveratrol from Discarded Leaf Biomass of Grapevine (Vitis sp.): Effect of Cultivar and Viticultural Practices in Estonia. Agriculture. 2020; 10(9):393. https://doi.org/10.3390/agriculture10090393
Chicago/Turabian StyleRätsep, Reelika, Kadri Karp, Mariana Maante-Kuljus, Alar Aluvee, and Rajeev Bhat. 2020. "Polyphenols and Resveratrol from Discarded Leaf Biomass of Grapevine (Vitis sp.): Effect of Cultivar and Viticultural Practices in Estonia" Agriculture 10, no. 9: 393. https://doi.org/10.3390/agriculture10090393
APA StyleRätsep, R., Karp, K., Maante-Kuljus, M., Aluvee, A., & Bhat, R. (2020). Polyphenols and Resveratrol from Discarded Leaf Biomass of Grapevine (Vitis sp.): Effect of Cultivar and Viticultural Practices in Estonia. Agriculture, 10(9), 393. https://doi.org/10.3390/agriculture10090393






