Automatic Detection and Monitoring of Insect Pests—A Review
Abstract
1. Introduction
2. Automatic Monitoring of Lepidoptera Pest Species
3. Automatic Monitoring of Sucking Insects
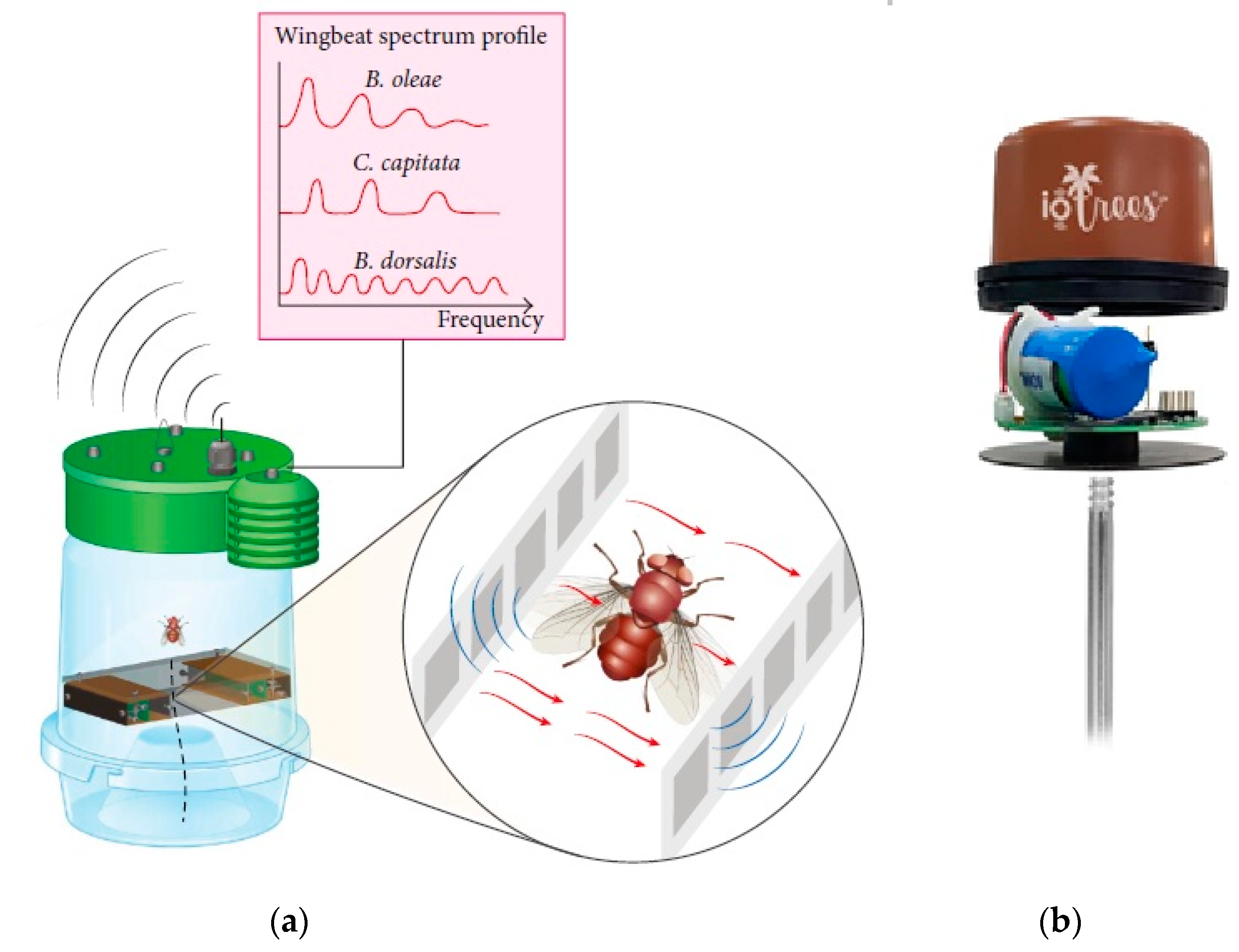
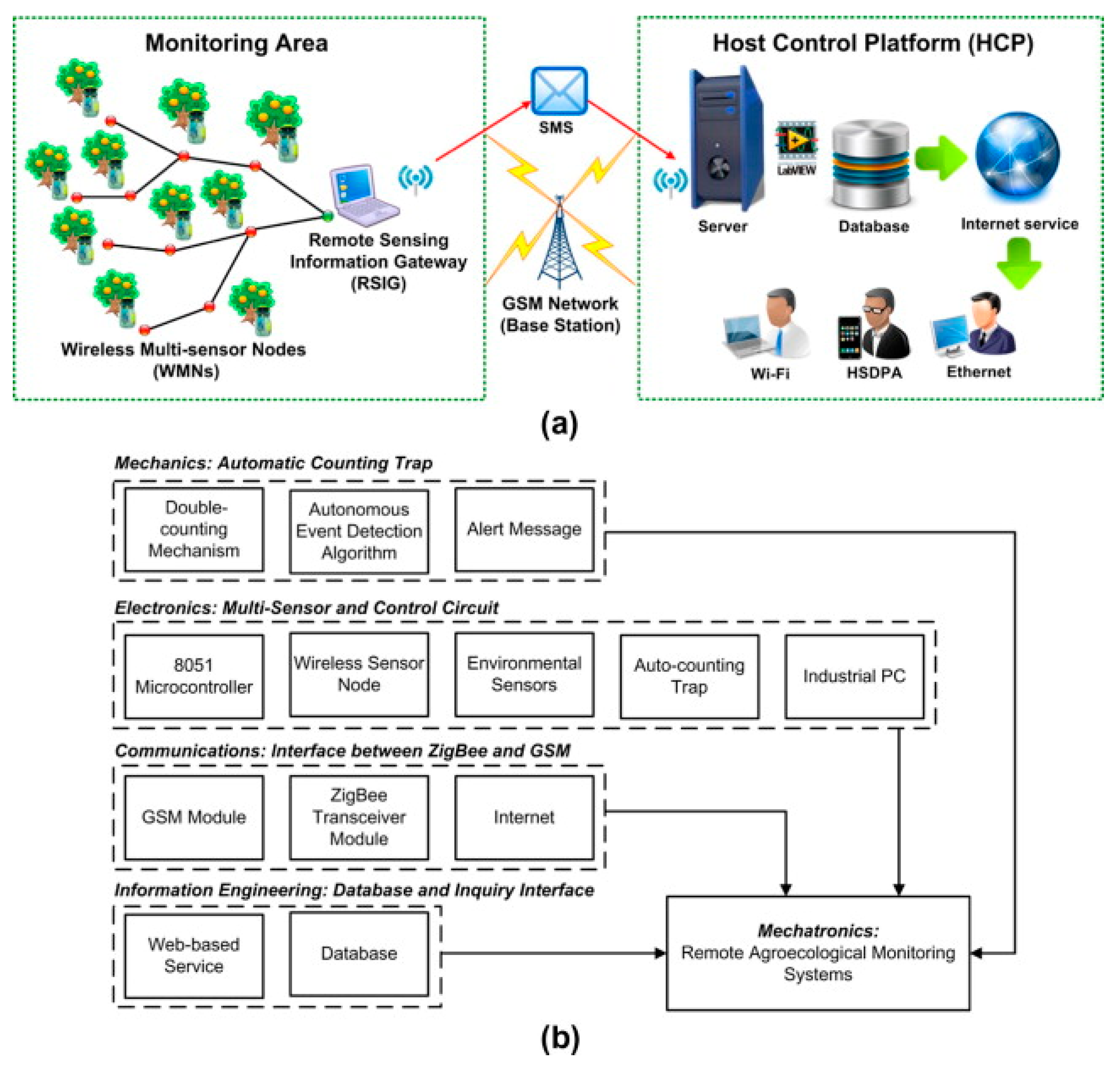
4. Automatic Identification and Monitoring of Fruit Flies
5. Automatic Monitoring of Weevil in Palm Trees and Other Borer Insects
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Group of Insect | Species | Sensors | Automatic Detection Technique | Efficacy | Authors |
|---|---|---|---|---|---|
| Aphids | Aphis glycines | SONY camera, images of leaves | Hue, Intensity components and similarity algorithm | >90% | Maharlooei et al., 2017 |
| Aphids | Myzus persicae | Digital camera, images of Pakchoi leaves | Convolutional neural networks | >80% | Chen et al., 2018 |
| Aphids | Not specified | Yellow sticky traps and smartphone camera | GrabCut method, OTSU algorithm and boundary extraction | 92.5–95% | Xuesong et al., 2017 |
| Aphids | Wheat aphids - Not specified species | Digital camera, images at field level | Maximally stable extremal region descriptor to simplify the background of field images containing aphids, and then used histograms of oriented gradient features and a support vector machine. | 86.81% | Liu et al., 2016 |
| Borer Insects | Anoplophora glabripennis | Screw with piezoelectric sensor (Oyster 723), amplifier (model ENC1485) and stored on an audio recorder (model TCD-D10 Pro II) | Custom-written insect signal analysis program: “Digitize, Analyze, View, Insect Sounds” (DAVIS). | 79–84% | Mankin et al., 2008b |
| Coleoptera, Hemiptera, Hymenoptera, Lepidoptera, Megaloptera, Neuroptera, Odonata and Orthoptera | 64 families, 221 species | Series of morphological features, Artificial neural networks (ANNs) and a support vector machine (SVM) | 93% | Wang et al., 2012 | |
| Fruit Flies | Anastrepha fraterculus, Anastrepha obliqua, Anastrepha sororcula | Nikon DS-Fi1 camera attached to a Nikon SMZ 1500 stereomicroscope | K-nearest neighbors classifier, Naive Bayes, Naive Bayes Tree, support vector machine and multimodal classifiers | 88–96% | Faria et al., 2014 |
| Fruit Flies | Bactrocera dorsalis | Sensors for measuring wind speed, temperature, and humidity, microcontroller (TI MSP430F449 chip), GSM module, GPS receiver, PC and Infrared counting device. | Complex system based on a remote monitoring platform (RMP) and a host control platform (HCP). | 72–92% | Jiang et al., 2008 |
| Fruit Flies | Bactrocera dorsalis | Sensors for measuring wind speed, temperature, and humidity, microcontroller (TI MSP430F449 chip), GSM module, GPS receiver, PC, Infrared counting device and wireless sensor network | Complex system based on a remote monitoring platform (RMP) and a host control platform (HCP). | - | Okuyama et al., 2011 |
| Fruit Flies | Bactrocera dorsalis | GSM and ZigBee, with three major components: remote sensing information gateway (RSIG), a host control platform (HCP) and a wireless monitoring nodes (WMNs). The WMNs transmits the collected data (relative humidity, illumination, temperature and the number of Oriental fruit flies captured) to the RSIG, the RSIG deliver the data to the database server (HCP) for storage and analysis. | Cloud computing image processing and environmental data | 98–100% | Liao et al., 2012 |
| Fruit Flies | Bactrocera oleae | Several webcams associated in a modified McPhail trap connected to internet | Gaussian blur filter, OTSU algorithm threshold and counting | 75% | Doitsidis et al., 2017 |
| Fruit Flies | Bactrocera oleae, Ceratitis capitata | Modified traps with Fresnel lenses and associated wingbeat stereo-recording device | Linear support vector classifier, radial basis function support vector machine, random forests, adaboost metaclassifier, extra randomized trees, gradient boosting classifier, convolutional neural network. | 98–99% | Potamitis et al., 2018 |
| Fruit Flies | Bactrocera oleae, Ceratitis capitata, Bactrocera Dorsalis | Modified traps with optoelectronic sensor | Linear support vector classifier, radial basis function support vector machine, random forests, adaboost metaclassifier, extra randomized trees, gradient boosting classifier, convolutional neural network. | 81–90% | Potamitis et al., 2017 |
| Fruit Flies | Dacus ciliatus, Rhagoletis cerasi, Bactrocera oleae | Digital camera inserted in a modified trap | Remote visual inspection | >88% | Shaked et al., 2018 |
| Lepidoptera | 19 species of the family Pieridae | Image database | Texture and color filter with gray level co-occurrence matrix (GLCM) | 92.85% | Kaya and Kayci 2014 |
| Lepidoptera Coleoptera Orthoptera | 40 species | Image database | Deep convolutional neural networks and transfer learning | 95–97% | Thenmozhi and Reddy, 2019 |
| Lepidoptera | 5 species of the family Papilinidae | Image database | Artificial neural network with binary patterns | 70–98% | Kaya et al., 2015 |
| Lepidoptera | 5 species of the family Papilinidae | Nikon Professional camera | Texture gabor filter-based, and extreme machine learning | 97% | Kaya et al., 2013 |
| Lepidoptera | 50 species | Digital camera | Histograms of multi-scale curvature (HoMSC), gray-level co-occurrence matrix of image blocks (GLCMoIB) and weight-based k-nearest neighbor classifier | 98% | Li and Xiong 2017 |
| Lepidoptera | Bicyclus anynana | Nikon SMZ1500 dissecting microscope at 3.8× magnification and a Digital Camera | Machine learning algorithm with features based on circularity and symmetry (1D Hough Transform which corresponds to histogramming) | 96% | Silveira and Monteiro 2009 |
| Lepidoptera | Celastrina argiolus, Cynthia cardui, Dilipa fenestra, Favonius orientalis, Graphium sarpedon, Libythea celtis, Luehdorfia puziloi, Lycaena dispar, Lycaena phlaeas, Ochlodes subhyalina, Papilio maackii, Papilio xuthus, Parantica sita, Parnassius bremeri, Sasakia charonda | Image database | BLS entropy profile and artificial neural network | 89–100% | Kang et al., 2014 |
| Lepidoptera | Cydia pomonella | Modified commercial trap with mobile camera with different resolutions | Remote visual inspection | Up to 100% | Guarnieri et al., 2011 |
| Lepidoptera | Cydia pomonella, Choristoneura rosaceana, Argyrotaenia velutinana, Grapholita molesta, Platynota idaeusalis, Spilonota ocellana Rhagoletis pomonella, Rhagoletis cingulata, Grapholita prunivora | Yellow sticky traps and webcam camera (Creative Inc., USA) | Combination of shape, color, texture and numerical features. Then, a pyramidal stacked de-noising auto-encoder (IpSDAE) was proposed to generate a deep neural network | 98.13% | Wen et al., 2015 |
| Lepidoptera | Helicoverpa armigera, Autographa gamma and Spodoptera spp. | Bucket/funnel trap with Camera | Cloud computing image processing | - | EFOS, Slovenia |
| Lepidoptera | Pyralidae family(do not specify species) | Mobile robot car with camera | Gaussian Mixture Model (GMM), Aggregation Dispersion Variance (ADV) and Distance Regularization Level Set Evolution (DRLSE) | 95% | Zhao et al., 2019 |
| Lepidoptera | Pyralidae family(do not specify species) | Mobile robot car with camera | Conversion into HSV space, extraction of the H spatial matrix, normalization of histogram, Otsu segmentation and object contour recognition based on Hu moments. | 94.3% | Liu et al., 2019 |
| Palm Weevil | Rhynchophorus cruentatus | Sensor-preamplifier module (model SP-1L Acoustic Emission Consulting AEC) and an amplifier AED-2000 connected to a digital audio recorder (model HD-P2, Tascam) | Custom-written insect signal analysis program: “Digitize, Analyze, View, Insect Sounds” (DAVIS). A bird-noise profile and a Traffic profile was created, after that a 512-point Fourier transformation was performed | 48.6–93.7% | Dosunmu et al., 2014 |
| Palm Weevil | Rhynchophorus ferrugineus | Piezoelectric microphone and 12v amplifier | Vector quantization, Gaussian mixture modeling and nearest neighborhood classifier | 92–98.1% | Pinhas et al., 2008 |
| Palm Weevil | Rhynchophorus ferrugineus | Digital voice recorder and soundproof chamber | Analysis of frequency and decibels | - | Martin et al., 2015 |
| Palm Weevil | Rhynchophorus ferrugineus | Digital Voice Recorder | Mel frequency coefficient characteristics and vector quantization | - | Martin and Juliet, 2010 |
| Palm Weevil | Rhynchophorus ferrugineus | An MCE-100 microphone, low-power processor, wireless communication interface and power supply unit | Hanning filter, decibel threshold, wavelet packet transform and vector quantization | >90% | Rach et al., 2013 |
| Palm Weevil | Rhynchophorus ferrugineus | SP-1 probe with an AED-2000 amplifier and also a Sony model TCD-D10 Pro II recorder device | Time-frequency distribution (TFD) based on spectrogram | - | Al-Manie and Alkanhal 2007 |
| Palm Weevil | Rhynchophorus ferrugineus | Laar ultrasound gate hard disk recording System (frequency range 50 Hz–250 kHz) and Laar WD 60 detector with amplifying system and insertion sensors of different types (contact microphone, airborne ultrasound microphone, contact acceleration sensor and a combined contact/airborne probe sensor) | High pass filter, several time domain features and several frequency domain features | >94% | Hussein et al., 2010 |
| Palm Weevil | Rhynchophorus ferrugineus | POM-3542P-R acoustic microphone coupled a preamplifier stage based on OP37 operational amplifier and a 4th order pass-band, continuous-time active filter (MAX274) applied to the signal in order to select the range of frequencies of interest. | Active filter, frequency spectra analysis | 70% | Gutiérrez et al., 2010 |
| Palm Weevil | Rhynchophorus ferrugineus | Magnetic cartridge head | Time-frequency wavelet analysis to relate the spectral frequencies available in the unique acoustic signature of red palm weevil larvae to its time of occurrence. | 92–97% | Siriwardena et al., 2010 |
| Palm Weevil | Rhynchophorus ferrugineus | Microphones, radio amplifier, repeater and transmitter connected to a wireless sensor network | Low-pass anti-aliasing filter and Down sampling; Butterworth IIR filter; Threshold filter | Srinivas et al., 2013 | |
| Palm Weevil | Rhynchophorus ferrugineus | Piezo-electric microphone with 50 mm (dia.) membrane was connected to a 20 mm (dia.) magnet via a hollow metal cone. Signal was preamplifier using model MP13. Recordings were performed using Raven Pro 64 | ‘Learning data set’ based on the multivariate distribution of nine pre-selected frequencies. threshold criterion (dynamically set) and multivariate classifier | 75–95% | Hetzroni et al., 2016 |
| Palm Weevil | Rhynchophorus ferrugineus | AED-2000, digital recorder, and sensor-preamplifier module magnetically attached to a screw and inserted into the base of a pruned palm frond | Custom-written insect signal analysis program: “Digitize, Analyze, View, Insect Sounds” (DAVIS). | >80% | Herrick and Mankin 2012 |
| Palm Weevil | Rhynchophorus ferrugineus | 1.59-mm titanium drill bit was inserted into the palm tree trunk and a sensor-preamplifier module (model SP-1) was attached magnetically, AED2000 amplifier and a digital recorder (HD-P2) | Oscillogram and spectrogram analysis | - | Fiaboe et al., 2011 |
| Palm Weevil | Rhynchophorus ferrugineus | Larva Lausher sensor | Sampling frequences to create digital signature | - | Soroker et al., 2004 |
| Palm Weevil | Rhynchophorus ferrugineus, Orycites rhinoceros | Digital recorder device | Analysis of frequency and decibels, Los Mel Frequency Cepstral Coefficients and Euclidean distance | - | Martin and Juliet, 2013 |
| Palm Weevil | Rhynchophorus ferrugineus, Rhynchophorus cruentatus | Microphone and amplifier | Custom-written insect signal analysis program: “Digitize, Analyze, View, Insect Sounds” (DAVIS). | 90% | Mankin et al., 2008a |
| Sucking pests | Bemisia tabaci, Aphis gossypii Thrips tabaci | Scanned sticky traps | Convert image to YCbCr, Segmentation by watershed, colour features and Melanobis distance | >80% relating to human counting | Xia et al., 2015 |
| Sucking pests | Bemisia tabaci, Myzus persicae subsp. nicotianae, Frankliniella fusca | Scanned sticky traps | Colour transformation YUV, fixed threshold and Prewittedge detection method | 66–100% | Cho et al., 2008 |
| Sucking pests | Bemisia tabaco, Frankliniella occidentalis | Digital camera (Nikon Coolpix S9200) and yellow and blue sticky traps | Two-dimensional Fourier transformation spectrum | 96% | Sun et al., 2017 |
| Sucking pests | Bemisia tabaco, Frankliniella occidentalis | Digitalization of sticky traps | Image-processing algorithm and artificial neural networks | 92–96% | Espinoza et al., 2016 |
| Sucking pests | Whiteflies and thrips (do not specify species) | Yellow sticky traps, Raspberry Pi v2 cameras | Convolutional neural network (CNN) classifier model through a generative adversarial network (GAN) image augmentation | 85–95% | Lu et al., 2019 |
| Sucking pests | Whiteflies, aphids and thrips (Do not Specify Species) | Scanned yellow sticky traps | Colour transformation, fixed threshold, morphological analysis | 66–100% | Cho et al., 2007 |
| Sucking pests | Whiteflies, thrips and aphids (do not specify species) | Sticky traps, camera, humidity and temperature sensor (AM2301 (Guangzhou Aosong Electronics Co., Guangzhou, China). Ambient light sensor (BH1750 (ROHM Semiconductor, Kyoto, Japan) | RGB-to-LUV color model conversion, Extraction of the V-channel color component. Static thresholding for image segmentation. Selective blob filtering. | 90–96% | Rustia et al., 2020 |
| Psyllids | (Not Specify) | Camera Raspberry Pi V2 (3280 × 2464pixels) | Insects trap and automatic image collection and storage in a server. | - | Blasco, et al., 2019 |
| Thrips | Do not Specify | Digital camera (Canon EOS M, 18 MP, CMOS, Japan) mounted in robot arm to capture the flower images | Support Vector Machine classification method with region index and intensifyas color index | >97% | Ebrahimi et al., 2017 |
| Wheat mites | Not specified | Image database | Convolutional neural network (CNN) of ZF (Zeiler and Fergus model) and a region proposal network (RPN) with Non-Maximum Suppression (NMS). | 88.5% | Li et al., 2019 |
| Wheat mites Rice planthopper | Not specified | Sony CX-10 GCD camera | Deep convolutional neural networks | Wang et al., 2020 | |
| Whiteflies | Bemisia tabaci | Camera coupled with a tube of 10 cm in diameter | Binary masks using colour transformation and fixed threshold | 83–95% | Barbedo (2013) |
| Whiteflies | Bemisia tabaci | Scanned yellow sticky traps | Image transformation 8-bit grayscale, binarization, boundary tracking and counting | 94.1–98.1% | Qiao et al., 2008 |
| Whiteflies | Bemisia tabaci | Digital camera, sticky traps and plants | Features corresponding to the eccentricity, area and machine learning | 97% | Soliz-Sánchez et al., 2009 |
| Whiteflies | Do not Specify | Image database | Shape, Gray Scale Intensity and Texture analysis | 74–85% | Ghods and Shojaeddini 2015 |
| Whiteflies | Do not Specify | Image database | Gray level run length matrix (GLRLM) and gray level co-occurrence matrix (GLCM). Various classifiers like support vector machine, artificial neural network, Bayesian classifier, binary decision tree classifier and k-nearest neighbor classifier | 90–98% | Dey et al., 2016 |
| Whiteflies | Do not specify | Low resolution digital camera | Algorithm based on relative difference in pixel intensities (RDI) using image processing | 96% | Huddar et al., 2012 |
| Whiteflies | Trialeurodes vaporariorum | Scanned leaves | Object extraction (Gaussian blur and Laplacian filter) and feature extraction | - | Bhadane et al., 2013 |
| Whiteflies | Trialeurodes vaporariorum | Scanned leaves | Cognitive vision with knowledge-based systems | >85% | Boissard et al., 2008 |
| Whiteflies | Trialeurodes vaporariorum and Bemisia Tabaci | Mobile aspiration mechanism and CCD Camera | Shape and colour properties | 85% | Bauch and Rath (2005) |
| Whitefly | Trialeurodes vaporariorum Bemisia tabaci | Automatic dataset generator, which is composed by two microcontrollers, two cameras, two tripods, two USB flash drives, two artificial illumination systems, one IP65 box and a portable Wi-Fi 4G router | K-nearest neighbor (KNN) and multilayer perceptron (MLP) | 66–81% | Gutierrez et al., 2019 |
Appendix B
| Supplier | Trap Type | Pest | Website |
|---|---|---|---|
| Agrint, USA | Seismic Sensor | Rhynchophorus ferrugineus in palm trees | https://www.agrint.net/ |
| IOTrees, Spain | Seismic Sensor | Rhynchophorus ferrugineus in palm trees | https://www.iotrees.es/ |
| TrapView, Slovenia | Delta trap (Standard) | Cydia pomonella in apples and pears | https://www.trapview.com |
| Grapholita funebrana in plums | |||
| Tuta absoluta in tomatoes | |||
| Lobesia botrana and Eupoecilia ambiguella in wine grapes | |||
| Cydia molesta in peaches | |||
| Plutella Xylostella in plants from Cruciferae family/Brassica | |||
| TrapView, Slovenia | Bucket/funnel (Self Cleaning) | Helicoverpa armigera | https://www.trapview.com |
| Autographa gamma | |||
| Spodoptera spp. | |||
| TrapView, Slovenia | Polarized UV light (Aura) | Ostrinia nubilalis in corn | https://www.trapview.com |
| TrapView, Slovenia | McPhail (Fly) | Ceratitis capitata in citrus and peaches | https://www.trapview.com |
| Drosophila suzukii in grapes and other fruits | |||
| TrapView, Slovenia | Support for Camera and Sticky Trap (Vertical) | Flying and Sucking Pests (not selective for species) | https://www.trapview.com |
References
- Fraser, A. Land grab/data grab: Precision agriculture and its new horizons. J. Peasant Stud. 2019, 46, 893–912. [Google Scholar] [CrossRef]
- Pedersen, S.M.; Lind, K.M. Precision Agriculture—From Mapping to Site-Specific Application. In Precision Agriculture: Technology and Economic Perspectives, 1st ed.; Springer Nature: Basel, Switzerland, 2017; pp. 1–20. [Google Scholar]
- Jiang, J.A.; Tseng, C.L.; Lu, F.M.; Yang, E.C.; Wu, Z.S.; Chen, C.P.; Lin, S.H.; Lin, K.C.; Liao, C.S. A GSM-based remote wireless automatic monitoring system for field information: A case study for ecological monitoring of the oriental fruit fly, Bactrocera dorsalis (Hendel). Comput. Electron. Agric. 2008, 62, 243–259. [Google Scholar] [CrossRef]
- Bradshaw, C.J.; Leroy, B.; Bellard, C.; Roiz, D.; Albert, C.; Fournier, A.; Barbet-Massin, M.; Salles, J.M.; Simard, F.; Courchamp, F.; et al. Massive yet grossly underestimated global costs of invasive insects. Nat. Commun. 2016, 7, 12986. [Google Scholar] [CrossRef]
- Gautam, M.P.; Singh, H.; Kumar, S.; Kumar, V.; Singh, G.; Singh, S.N. Diamondback moth, Plutella xylostella (Linnaeus) (Insecta: Lepidoptera: Plutellidae) a major insect of cabbage in India: A review. J. Entomol. Zool. Stud. 2018, 6, 1394–1399. [Google Scholar]
- Wen, C.; Wu, D.; Hu, H.; Pan, W. Pose estimation-dependent identification method for field moth images using deep learning architecture. Biosyst. Eng. 2015, 136, 117–128. [Google Scholar] [CrossRef]
- Kang, S.H.; Cho, J.H.; Lee, S.H. Identification of butterfly based on their shapes when viewed from different angles using an artificial neural network. J. Asia-Pac. Entomol. 2014, 17, 143–149. [Google Scholar] [CrossRef]
- Silveira, M.; Monteiro, A. Automatic recognition and measurement of butterfly eyespot patterns. Biosystems 2009, 95, 130–136. [Google Scholar] [CrossRef] [PubMed]
- Guarnieri, A.; Maini, S.; Molari, G.; Rondelli, V. Automatic trap for moth detection in integrated pest management. Bull. Insectol. 2011, 64, 247–251. [Google Scholar]
- Kaya, Y.; Kayci, L.; Uyar, M. Automatic identification of butterfly species based on local binary patterns and artificial neural network. Appl. Soft Comput. 2015, 28, 132–137. [Google Scholar] [CrossRef]
- Wang, J.; Lin, C.; Ji, L.; Liang, A. A new automatic identification system of insect images at the order level. Knowl. Based Syst. 2012, 33, 102–110. [Google Scholar] [CrossRef]
- Kaya, Y.; Kayci, L. Application of artificial neural network for automatic detection of butterfly species using color and texture features. Vis. Comput. 2014, 30, 71–79. [Google Scholar] [CrossRef]
- Kaya, Y.; Kayci, L.; Tekin, R. A computer vision system for the automatic identification of butterfly species via gabor-filter-based texture features and extreme learning machine: GF+ELM. TEM J. 2013, 2, 13–20. [Google Scholar]
- Thenmozhi, K.; Reddy, U.S. Crop pest classification based on deep convolutional neural network and transfer learning. Comput. Electron. Agric. 2019, 164, 104906. [Google Scholar] [CrossRef]
- Li, F.; Yin, X. Automatic identification of butterfly species based on HoMSC and GLCMoIB. Vis. Comput. 2018, 34, 1525–1533. [Google Scholar] [CrossRef]
- Liu, B.; Hu, Z.; Zhao, Y.; Bai, Y.; Wang, Y. Recognition of Pyralidae Insects Using Intelligent Monitoring Autonomous Robot Vehicle in Natural Farm Scene. arXiv 2019, arXiv:1903.10827. [Google Scholar]
- Zhao, Y.; Wang, Y.; Wang, J.; Hu, Z.; Lin, F.; Xu, M. GMM and DRLSE Based Detection and Segmentation of Pests: A Case Study. In Proceedings of the 2019 4th International Conference on Multimedia Systems and Signal Processing, Guangzhou China, 10–12 May 2019; pp. 62–66. [Google Scholar]
- Xia, C.; Chon, T.S.; Ren, Z.; Lee, J.M. Automatic identification and counting of small size pests in greenhouse conditions with low computational cost. Ecol. Inform. 2015, 29, 139–146. [Google Scholar] [CrossRef]
- Bhadane, G.; Sharma, S.; Nerkar, V.B. Early pest identification in agricultural crops using image processing techniques. Int. J. Electr. Electron. Comput. Eng. 2013, 2, 77–82. [Google Scholar]
- Boissard, P.; Martin, V.; Moisan, S. A cognitive vision approach to early pest detection in greenhouse crops. Comput. Electron. Agric. 2008, 62, 81–93. [Google Scholar] [CrossRef]
- Bodhe, T.S.; Mukherji, P. Selection of color space for image segmentation in pest detection. In Proceedings of the International Conference on Advances in Technology and Engineering, Mumbai, India, 23–25 January 2015. [Google Scholar]
- Ghods, S.; Shojaeddini, V. A novel automated image analysis method for counting the population of whiteflies on leaves of crops. J. Crop Prot. 2015, 5, 59–73. [Google Scholar] [CrossRef]
- Blasco, J.; Sanjuan, S.; Chueca, P.; Fereres, A.; Cubero, S.; Lopez, S.; Alegre, V. Dispositivo de Captura y Envio de Imagenes a un Servidor Remoto para Monitorizar Trampas para Insectos en el Campo; No. COMPON-2019-agri-3469; X Congresso Ibérico de Agroengenharia: Huesca, Spain, 2019. [Google Scholar] [CrossRef]
- Dey, A.; Bhoumik, D.; Dey, K.N. Automatic Detection of Whitefly Pest using Statistical Feature Extraction and Image Classification Methods. Int. Res. J. Eng. Technol. 2016, 3, 950–959. [Google Scholar]
- Ebrahimi, M.A.; Khoshtaghaza, M.H.; Minaei, S.; Jamshidi, B. Vision-based pest detection based on SVM classification method. Comput. Electron. Agric. 2017, 137, 52–58. [Google Scholar] [CrossRef]
- Barbedo, J.G.A. Automatic method for counting and measuring whiteflies in soybean leaves using digital image processing. In Proceedings of the IX Brazilian Congress of Agro-Informatics, Cuiaba, Brazil, 21–25 October 2013. [Google Scholar]
- Huddar, S.R.; Gowri, S.; Keerthana, K.; Vasanthi, S.; Rupanagudi, S.R. Novel algorithm for segmentation and automatic identification of pests on plants using image processing. In Proceedings of the Third International Conference on Computing Communication and Networking Technologies, Karur, India, 26–28 July 2012. [Google Scholar]
- Cho, J.; Choi, J.; Qiao, M.; Ji, C.W.; Kim, H.Y.; Uhm, K.B.; Chon, T.S. Automatic identification of whiteflies, aphids and thrips in greenhouse based on image analysis. Int. J. Math. Comput. Simul. 2007, 1, 46–53. [Google Scholar]
- Cho, J.; Choi, J.; Qiao, M.; Ji, C.; Kim, H.; Uhm, K.; Chon, T. Automatic identification of tobacco whiteflies, aphids and thrips in greenhouse using image processing techniques. In Proceedings of the 4th WSEAS International Conference on Mathematical Biology and Ecology, Acapulco, Mexico, 25–27 January 2008. [Google Scholar]
- Qiao, M.; Lim, J.; Ji, C.W.; Chung, B.K.; Kim, H.Y.; Uhm, K.B.; Myung, C.S.; Cho, J.; Chon, T.S. Density estimation of Bemisia tabaci (Hemiptera: Aleyrodidae) in a greenhouse using sticky traps in conjunction with an image processing system. J. Asia-Pac. Entomol. 2008, 11, 25–29. [Google Scholar] [CrossRef]
- Sun, Y.; Cheng, H.; Cheng, Q.; Zhou, H.; Li, M.; Fan, Y.; Shan, G.; Damerow, L.; Lammers, P.S.; Jones, S.B. A smart-vision algorithm for counting whiteflies and thrips on sticky traps using two-dimensional Fourier transform spectrum. Biosyst. Eng. 2017, 153, 82–88. [Google Scholar] [CrossRef]
- Solis-Sánchez, J.J.; García-Escalante, R.; Castañeda-Miranda, I.; Torres-Pacheco, R.G. González. Machine vision algorithm for whiteflies (Bemisia tabaci Genn.) scouting under greenhouse environment. J. Appl. Entomol. 2009, 133, 546–552. [Google Scholar] [CrossRef]
- Espinoza, K.; Valera, D.L.; Torres, J.A.; López, A.; Molina-Aiz, F.D. Combination of image processing and artificial neural networks as a novel approach for the identification of Bemisia tabaci and Frankliniella occidentalis on sticky traps in greenhouse agriculture. Comput. Electron. Agric. 2016, 127, 495–505. [Google Scholar] [CrossRef]
- Bauch, C.; Rath, T. Prototype of a vision based system for measurements of white fly infestation. In Proceedings of the International Conference on Sustainable Greenhouse Systems (Greensys 2004), Leuven, Belgium, 12–16 September 2004. [Google Scholar]
- Lu, C.Y.; Rustia, D.J.A.; Lin, T.T. Generative Adversarial Network Based Image Augmentation for Insect Pest Classification Enhancement. IFAC-PapersOnLine 2019, 52, 1–5. [Google Scholar] [CrossRef]
- Suo, X.; Liu, Z.; Sun, L.; Wang, J.; Zhao, Y. Aphid Identification and Counting Based on Smartphone and Machine Vision. J. Sens. 2017, 1–7. [Google Scholar] [CrossRef]
- Liu, T.; Chen, W.; Wu, W. Detection of aphids in wheat fields using a computer vision technique. Biosyst. Eng. 2016, 141, 82–93. [Google Scholar] [CrossRef]
- Li, W.; Chen, P.; Wang, B.; Xie, C. Automatic localization and count of agricultural crop pests based on an improved deep learning pipeline. Sci. Rep. 2019, 9, 1–11. [Google Scholar]
- Maharlooei, M.; Sivarajan, S.; Bajwa, S.G.; Harmon, J.P.; Nowatzki, J. Detection of soybean aphids in a greenhouse using an image processing technique. Comput. Electron. Agric. 2017, 132, 63–70. [Google Scholar] [CrossRef]
- Chen, J.; Fan, Y.; Wang, T.; Zhang, C.; Qiu, Z.; He, Y. Automatic Segmentation and Counting of Aphid Nymphs on Leaves Using Convolutional Neural Networks. Agronomy 2018, 8, 129. [Google Scholar] [CrossRef]
- Rustia, D.J.A.; Lin, C.E.; Chung, J.Y.; Zhuang, Y.J.; Hsu, J.C.; Lin, T.T. Application of an image and environmental sensor network for automated greenhouse insect pest monitoring. J. Asia-Pac. Entomol. 2020, 23, 17–28. [Google Scholar] [CrossRef]
- Gutierrez, A.; Ansuategi, A.; Susperregi, L.; Tubío, C.; Rankić, I.; Lenža, L. A Benchmarking of Learning Strategies for Pest Detection and Identification on Tomato Plants for Autonomous Scouting Robots Using Internal Databases. J. Sens. 2019, 2019, 5219471. [Google Scholar] [CrossRef]
- Hendrichs, J.; Vera, M.T.; De Meyer, M.; Clarke, A.R. Resolving cryptic species complexes of major tephritid pests. Zookeys 2015, 540, 5–39. [Google Scholar] [CrossRef]
- Oliveira, C.M.; Auad, A.M.; Mendes, S.M.; Frizzas, M.R. Economic impact of insect pests in Brazilian agriculture. J. Appl. Entomol. 2012, 137, 1–15. [Google Scholar] [CrossRef]
- White, I.M.; Elson-Harris, M.M. Fruit Flies of Economic Significance: Their Identification and Bionomics, 1st ed.; CABI: Wallingford, UK, 1992; p. 70. [Google Scholar]
- Downweerd, C.; Leblanc, L.; Norrbom, A.L.; Jose, M.S.; Rubinoff, D. A global checklist of the 932 fruit fly species in the tribe Dacini (Diptera, Tephritidae). Zookeys 2018, 730, 19. [Google Scholar] [CrossRef]
- Potamitis, I.; Rigakis, I.; Tatlas, N.A. Automated surveillance of fruit flies. Sensors 2017, 17, 110. [Google Scholar] [CrossRef]
- Duyck, P.; David, P.; Quilici, S. A review of relationships between interspecific competition and invasions of fruit flies (Diptera: Tephritidae). Ecol. Enthomol. 2004, 29, 511–520. [Google Scholar] [CrossRef]
- Duyck, P.; David, P.; Junod, G.; Brunel, C.; Dupont, R.; Quilici, S. Importance of competition mechanisms in successive invasions by polyphagous tephritis in La Reunion. Ecology 2006, 87, 1770–1780. [Google Scholar] [CrossRef]
- Villalobos, J.; Flores, S.; Liedo, P.; Malo, E. Mass trapping is as effective as ground bait sprays for the control of Anastrepha (Diptera: Tephritidae) fruit flies in mango orchards. Pest Manag. Sci. 2017, 73, 2105–2110. [Google Scholar] [CrossRef] [PubMed]
- Hendrichs, J.; Franz, G.; Rendon, P. Increased effectiveness and applicability of the sterile insect technique through male-only releases for control of Mediterranean fruit flies during fruiting seasons. J. Appl. Entomol. 1995, 119, 371–377. [Google Scholar] [CrossRef]
- Potamitis, I.; Rigakis, I.; Vidakis, N.; Petousis, M.; Weber, M. Affordable Bimodal Optical Sensors to Spread the Use of Automated Insect Monitoring. J. Sens. 2018, 1–25. [Google Scholar] [CrossRef]
- Faria, F.A.; Perre, P.; Zucchi, R.A.; Jorge, L.R.; Lewinsohn, T.M.; Rocha, A.; Torres, R.D.S. Automatic identification of fruit flies (Diptera: Tephritidae). J. Vis. Commun. Image Represent. 2014, 25, 1516–1527. [Google Scholar] [CrossRef]
- Doitsidis, L.; Fouskitakis, G.N.; Varikou, K.N.; Rigakis, I.I.; Chatzichristofis, S.A.; Papafilippaki, A.K.; Birouraki, A.E. Remote monitoring of the Bactrocera oleae (Gmelin)(Diptera: Tephritidae) population using an automated McPhail trap. Comput. Electron. Agric. 2017, 137, 69–78. [Google Scholar] [CrossRef]
- Okuyama, T.; Yang, E.C.; Chen, C.P.; Lin, T.S.; Chuang, C.L.; Jiang, J.A. Using automated monitoring systems to uncover pest population dynamics in agricultural fields. Agric. Syst. 2011, 104, 666–670. [Google Scholar] [CrossRef]
- Liao, M.S.; Chuang, C.L.; Lin, T.S.; Chen, C.P.; Zheng, X.Y.; Chen, P.T.; Liao, K.C.; Jiang, J.A. Development of an autonomous early warning system for Bactrocera dorsalis (Hendel) outbreaks in remote fruit orchards. Comput. Electron. Agric. 2012, 88, 1–12. [Google Scholar] [CrossRef]
- Shaked, B.; Amore, A.; Ioannou, C.; Valdés, F.; Alorda, B.; Papanastasiou, S.; Goldshtein, E.; Shenderey, C.; Leza, M.; Pontikakos, C.; et al. Electronic traps for detection and population monitoring of adult fruit flies (Diptera: Tephritidae). J. Appl. Entomol. 2018, 142, 43–51. [Google Scholar] [CrossRef]
- Haff, R.P.; Saranwong, S.; Thanapase, W.; Janhiran, A.; Kasemsumran, S.; Kawano, S. Automatic image analysis and spot classification for detection of fruit fly infestation in hyperspectral images of mangoes. Postharvest Biol. Technol. 2013, 86, 23–28. [Google Scholar] [CrossRef]
- FAO. Current Situation of Red Palm Weevil in the NENA Region. In Current Situation of Management Practices, Challenges/Weaknesses and Available Research and Technologies for Its Improvement, Proceedings of Scientific Consultation and High-Level Meeting on Red Palm Weevil Management, Rome, Italy, 29–31 March 2017; FAO: Rome, Italy, 2017. [Google Scholar]
- Pinhas, J.; Soroker, V.; Hetzroni, A.; Mizrach, A.; Teicher, M.; Goldberger, J. Automatic acoustic detection of the red palm weevil. Comput. Electron. Agric. 2008, 63, 131–139. [Google Scholar] [CrossRef]
- Martin, B.; Shaby, S.M.; Premi, M.G. Studies on acoustic activity of red palm weevil the deadly pest on coconut crops. Procedia Mater. Sci. 2015, 10, 455–466. [Google Scholar] [CrossRef]
- Nangai, V.L. Interpreting the Acoustic Characteristics of Rpw Towards Its Detection-A Review. IOP Conf. Ser. Mater. Sci. Eng. 2017, 225, 1–9. [Google Scholar] [CrossRef]
- Martin, B.; Juliet, V. A novel approach to identify red palm weevil on palms. Adv. Mater. Res. 2013, 634, 3853–3857. [Google Scholar] [CrossRef]
- Dosunmu, O.G.; Herrick, N.J.; Haseeb, M.; Hix, R.L.; Mankin, R.W. Acoustic detectability of Rhynchophorus cruentatus (Coleoptera: Dryophthoridae). Fla. Entomol. 2014, 97, 431–438. [Google Scholar] [CrossRef]
- Martin, B.; Juliet, V. Discriminating human whispers from pest sound during recordings in coconut palm grooves using MFCC and vector quantization. Int. J. Appl. Bioeng. 2010, 4, 29–33. [Google Scholar] [CrossRef][Green Version]
- Rach, M.M.; Gomis, H.M.; Granado, O.L.; Malumbres, M.P.; Campoy, A.M.; Martín, J.J.S. On the design of a bioacoustic sensor for the early detection of the red palm weevil. Sensors 2013, 13, 1706–1729. [Google Scholar] [CrossRef]
- Al-Manie, M.A.; Alkanhalm, M.I. Acoustic detection of the red date palm weevil. Int. J. Electr. Comput. Energ. Electron. Commun. Eng. 2007, 1, 345–348. [Google Scholar]
- Hussein, W.; Hussein, M.; Becker, T. Detection of the red palm weevil Rhynchophorus Ferrugineus using its bioacoustics features. Bioacoustics 2010, 19, 177–194. [Google Scholar] [CrossRef]
- Gutiérrez, A.; Ruiz, V.; Moltó, E.; Tapia, G.; Téllez, M. Development of a bioacoustic sensor for the early detection of Red Palm Weevil (Rhynchophorus ferrugineus Olivier). Crop Prot. 2010, 29, 671–676. [Google Scholar] [CrossRef]
- Siriwardena, K.A.P.; Fernando, L.C.P.; Nanayakkara, N.; Perera, K.F.G.; Kumara, A.D.N.T.; Nanayakkara, T. Portable acoustic device for detection of coconut palms infested by Rhynchophorus ferrugineus (Coleoptera: Curculionidae). Crop Prot. 2010, 29, 25–29. [Google Scholar] [CrossRef]
- Srinivas, S.; Harsha, K.S.; Sujatha, A.; Kumar, N.G. Efficient protection of palms from RPW larvae using wireless sensor networks. Int. J. Comput. Sci. Issues 2013, 10, 192–200. [Google Scholar]
- Hetzroni, A.; Soroker, V.; Cohen, Y. Toward practical acoustic red palm weevil detection. Comput. Electron. Agric. 2016, 124, 100–106. [Google Scholar] [CrossRef]
- Mankin, R.W.; Mizrach, A.; Hetzroni, A.; Levsky, S.; Nakache, Y.; Soroker, V. Temporal and spectral features of sounds of wood-boring beetle larvae: Identifiable patterns of activity enable improved discrimination from background noise. Fla. Entomol. 2008, 91, 241–248. [Google Scholar] [CrossRef]
- Herrick, N.J.; Mankin, R.W. Acoustical detection of early instar Rhynchophorus ferrugineus (Coleoptera: Curculionidae) in Canary Island date palm, Phoenix canariensis (Arecales: Arecaceae). Fla. Entomol. 2012, 95, 983–990. [Google Scholar] [CrossRef]
- Mankin, R.W.; Smith, M.T.; Tropp, J.M.; Atkinson, E.B.; Jong, D.Y. Detection of Anoplophora glabripennis (Coleoptera: Cerambycidae) larvae in different host trees and tissues by automated analyses of sound-impulse frequency and temporal patterns. J. Econ. Entomol. 2008, 101, 838–849. [Google Scholar] [CrossRef]
- Soroker, V.; Nakache, Y.; Landau, U.; Mizrach, A.; Hetzroni, A.; Gerling, D. Note: Utilization of sounding methodology to detect infestation by Rhynchophorus ferrugineus on palm offshoots. Phytoparasitica 2004, 32, 6–8. [Google Scholar]
- Fiaboe, K.K.M.; Mankin, R.W.; Roda, A.L.; Kairo, M.T.K.; Johanns, C. Pheromone-Food-Bait Trap and Acoustic Surveys of Rhynchophorus ferrugineus (Coleoptera: Curculionidae) in Curacao. Fla. Entomol. 2011, 94, 766–773. [Google Scholar] [CrossRef]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cardim Ferreira Lima, M.; Damascena de Almeida Leandro, M.E.; Valero, C.; Pereira Coronel, L.C.; Gonçalves Bazzo, C.O. Automatic Detection and Monitoring of Insect Pests—A Review. Agriculture 2020, 10, 161. https://doi.org/10.3390/agriculture10050161
Cardim Ferreira Lima M, Damascena de Almeida Leandro ME, Valero C, Pereira Coronel LC, Gonçalves Bazzo CO. Automatic Detection and Monitoring of Insect Pests—A Review. Agriculture. 2020; 10(5):161. https://doi.org/10.3390/agriculture10050161
Chicago/Turabian StyleCardim Ferreira Lima, Matheus, Maria Elisa Damascena de Almeida Leandro, Constantino Valero, Luis Carlos Pereira Coronel, and Clara Oliva Gonçalves Bazzo. 2020. "Automatic Detection and Monitoring of Insect Pests—A Review" Agriculture 10, no. 5: 161. https://doi.org/10.3390/agriculture10050161
APA StyleCardim Ferreira Lima, M., Damascena de Almeida Leandro, M. E., Valero, C., Pereira Coronel, L. C., & Gonçalves Bazzo, C. O. (2020). Automatic Detection and Monitoring of Insect Pests—A Review. Agriculture, 10(5), 161. https://doi.org/10.3390/agriculture10050161





