Development of Selenium Nanoparticle Based Agriculture Sensor for Heavy Metal Toxicity Detection
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Growth
2.2. Biosynthesis of Elemental SeNPs
2.3. Characterization of SeNPs
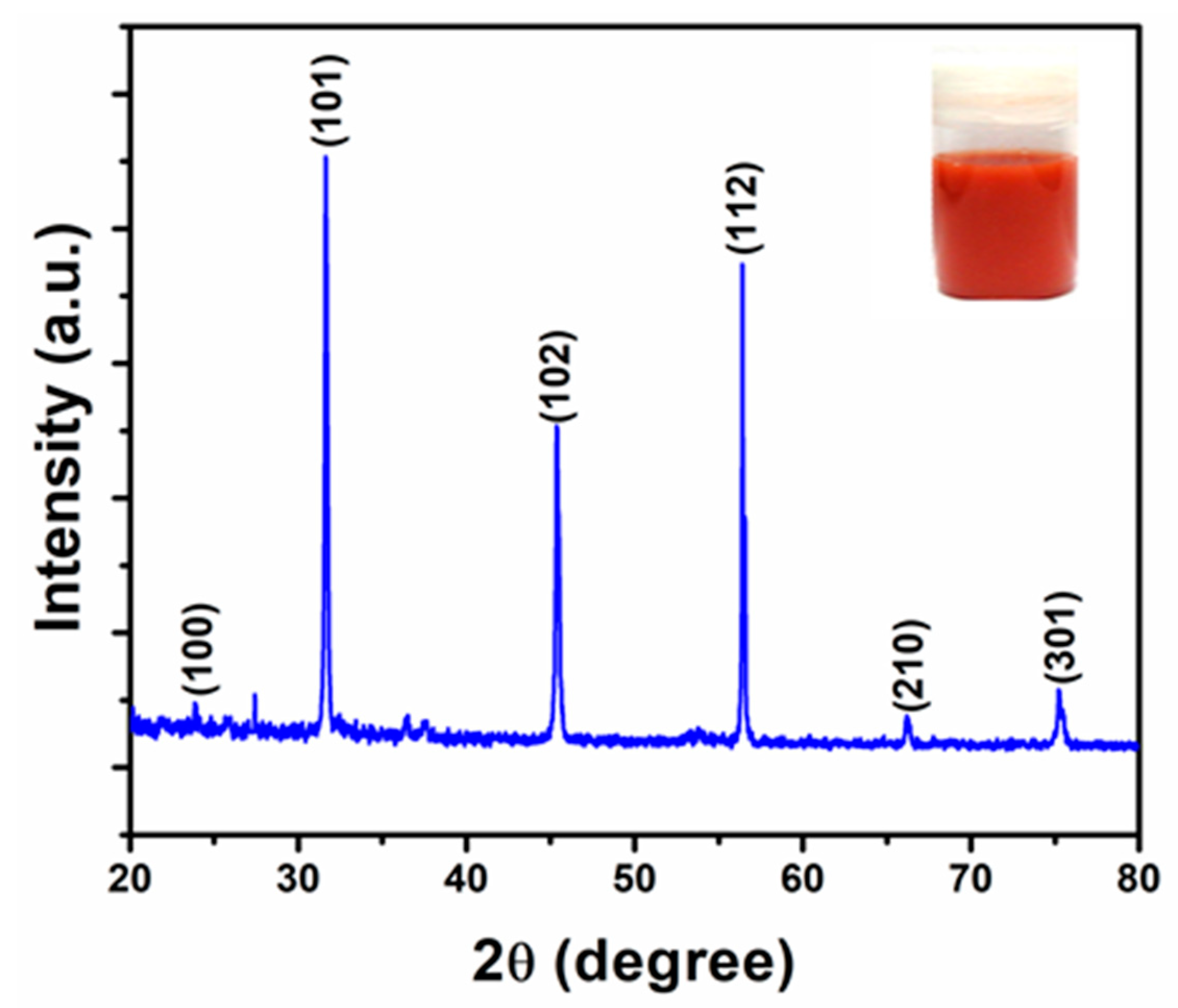
2.3.1. X-ray Diffraction Analysis
2.3.2. Optical Properties (UV–Vis)
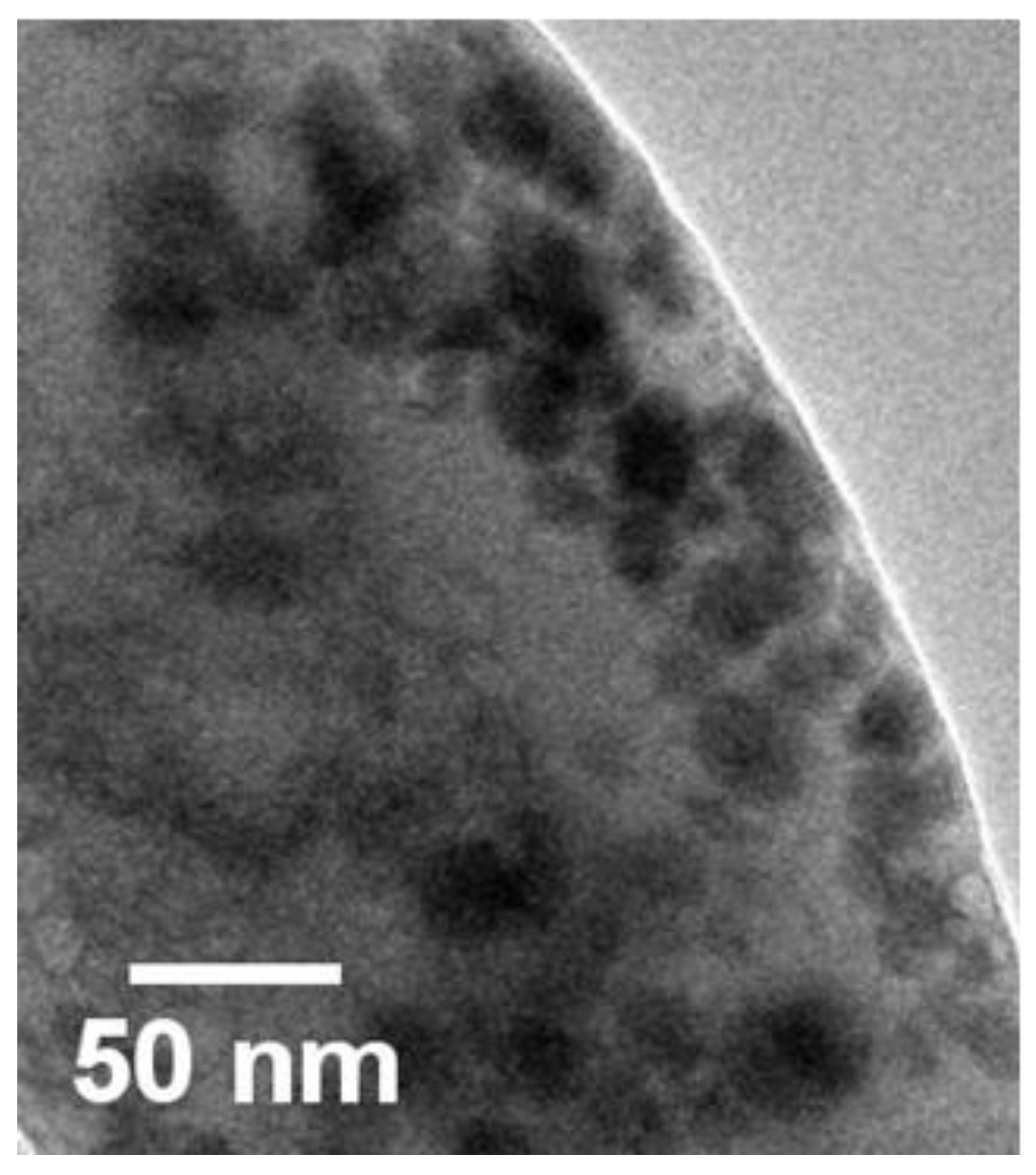
2.3.3. Transmission Electron Microscopic (TEM) Analysis
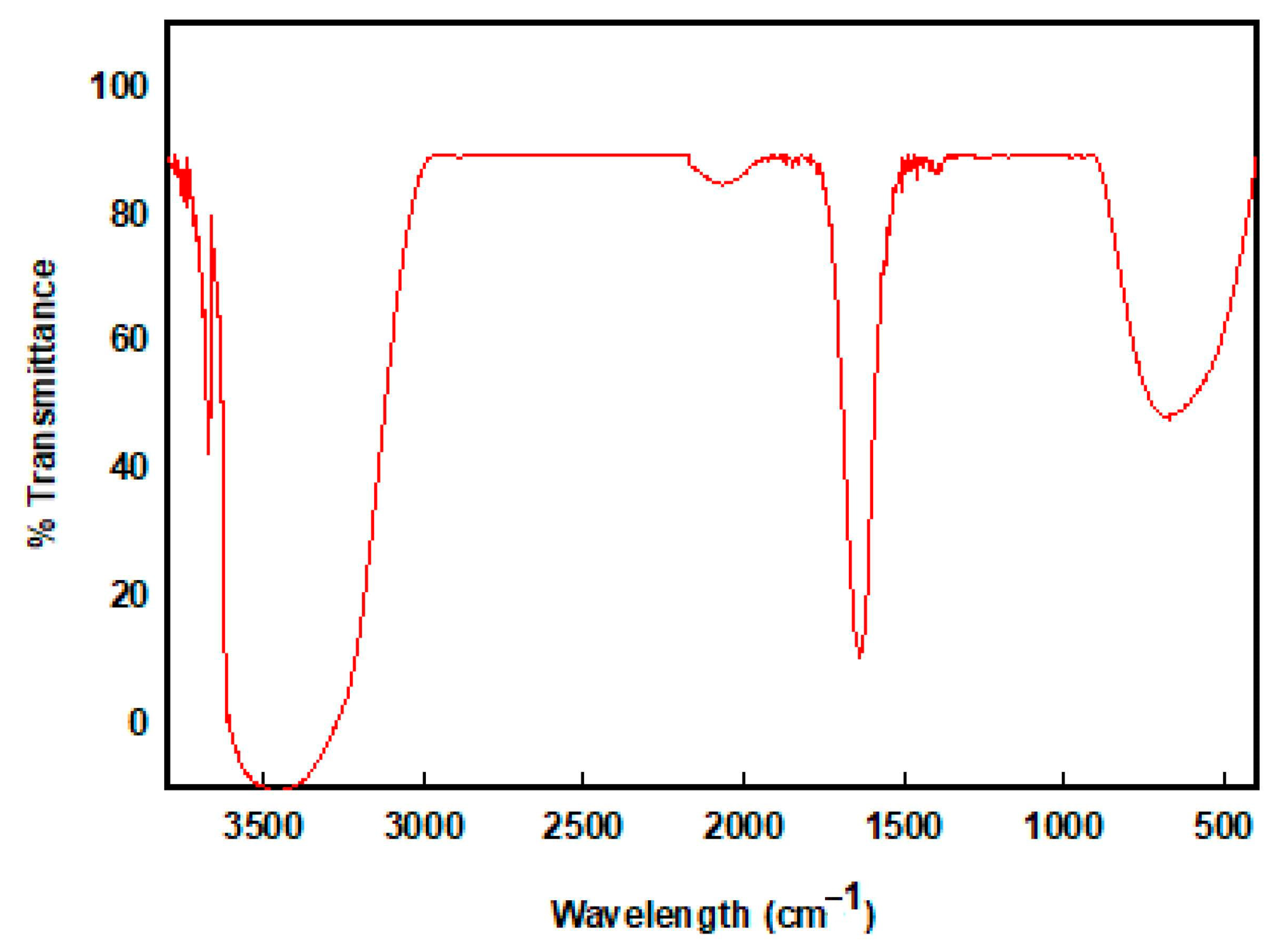
Fourier Transform Infrared (FTIR) Analysis
2.3.4. Fluorescence Measurements
2.3.5. Heavy Metal Toxicity Assessment
3. Results and Discussion
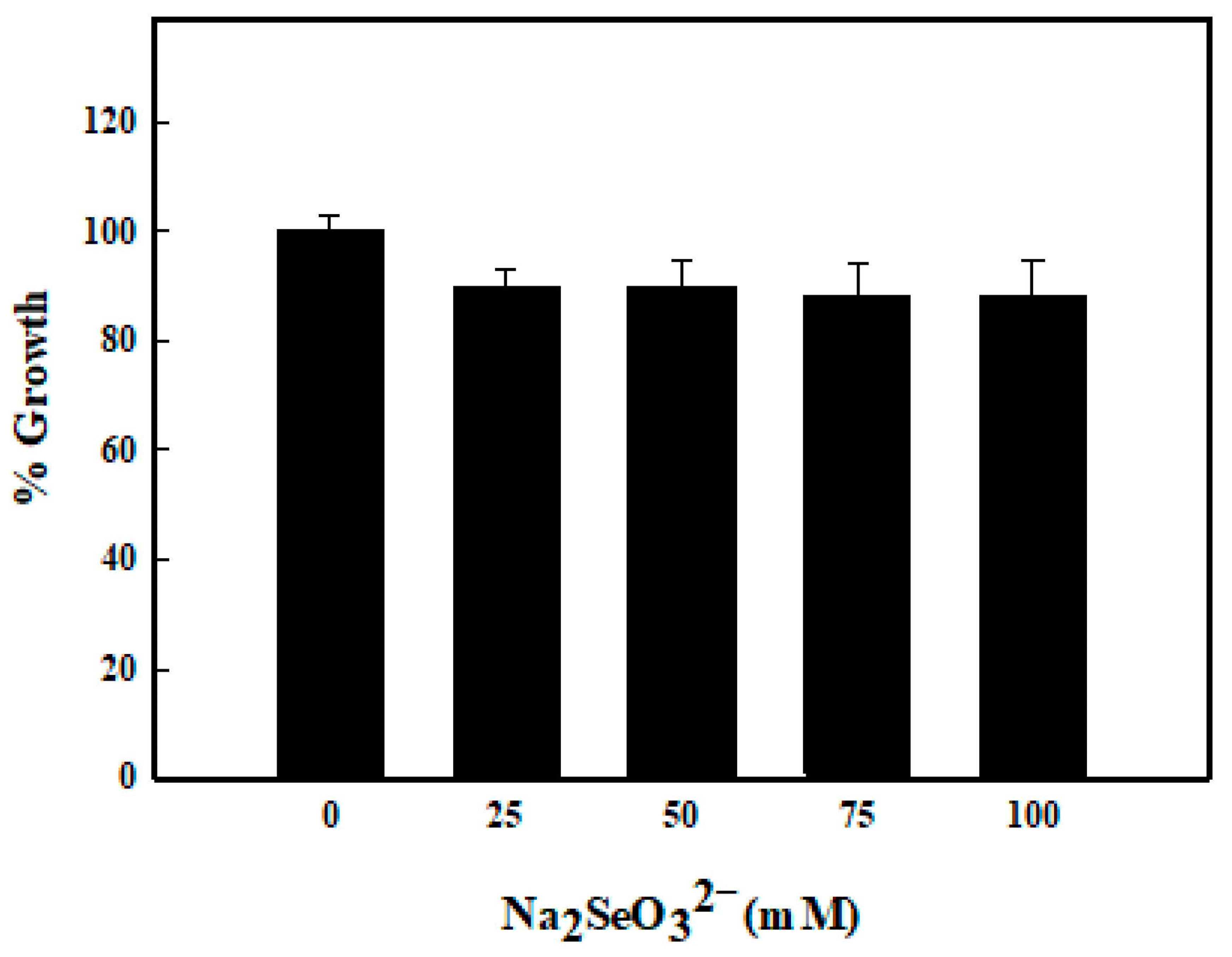
3.1. Selenite Tolerance
3.2. Biosynthesis of SeNPs Using Bacterial Supernatant
3.3. Characterization of SeNPs
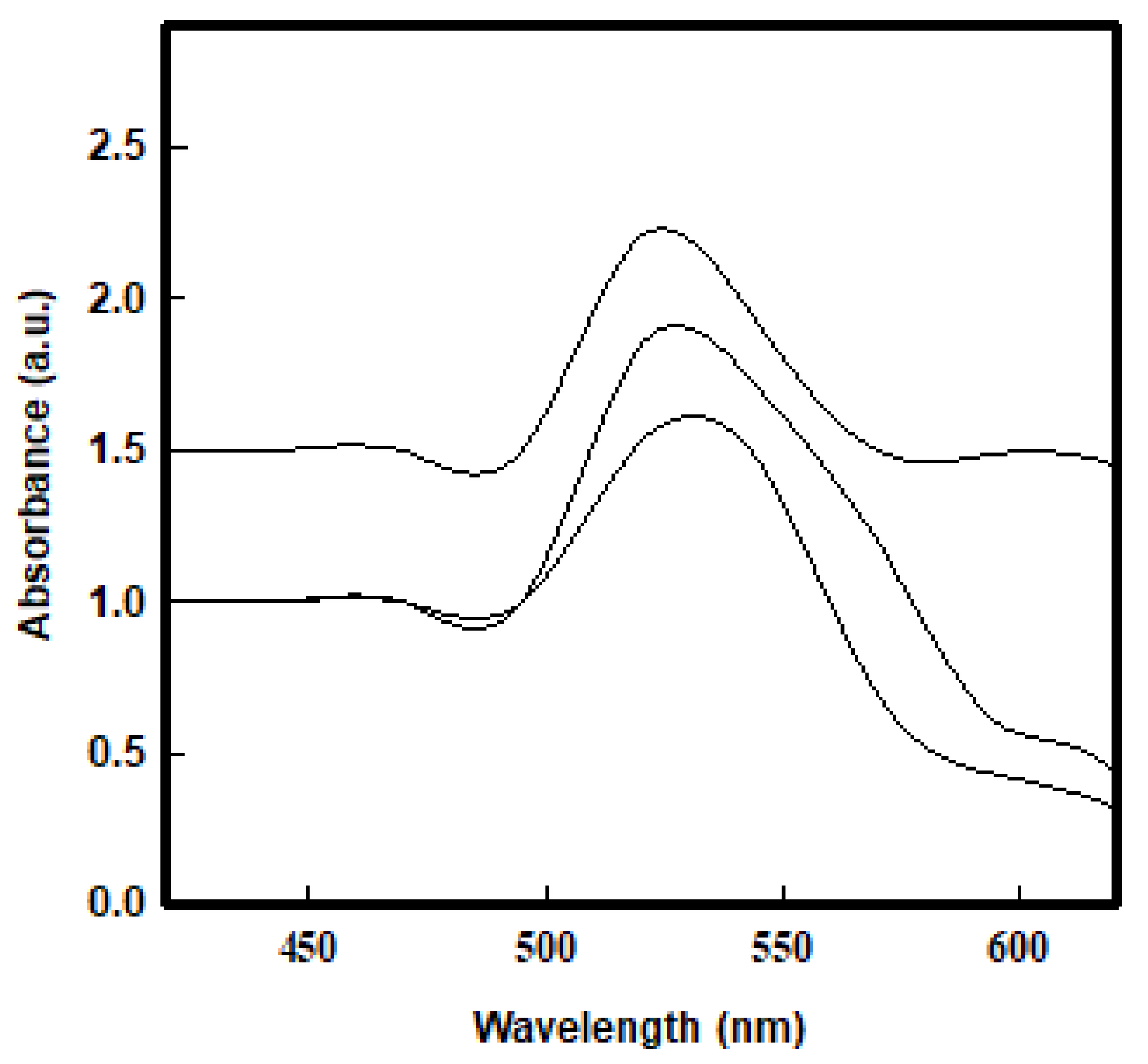
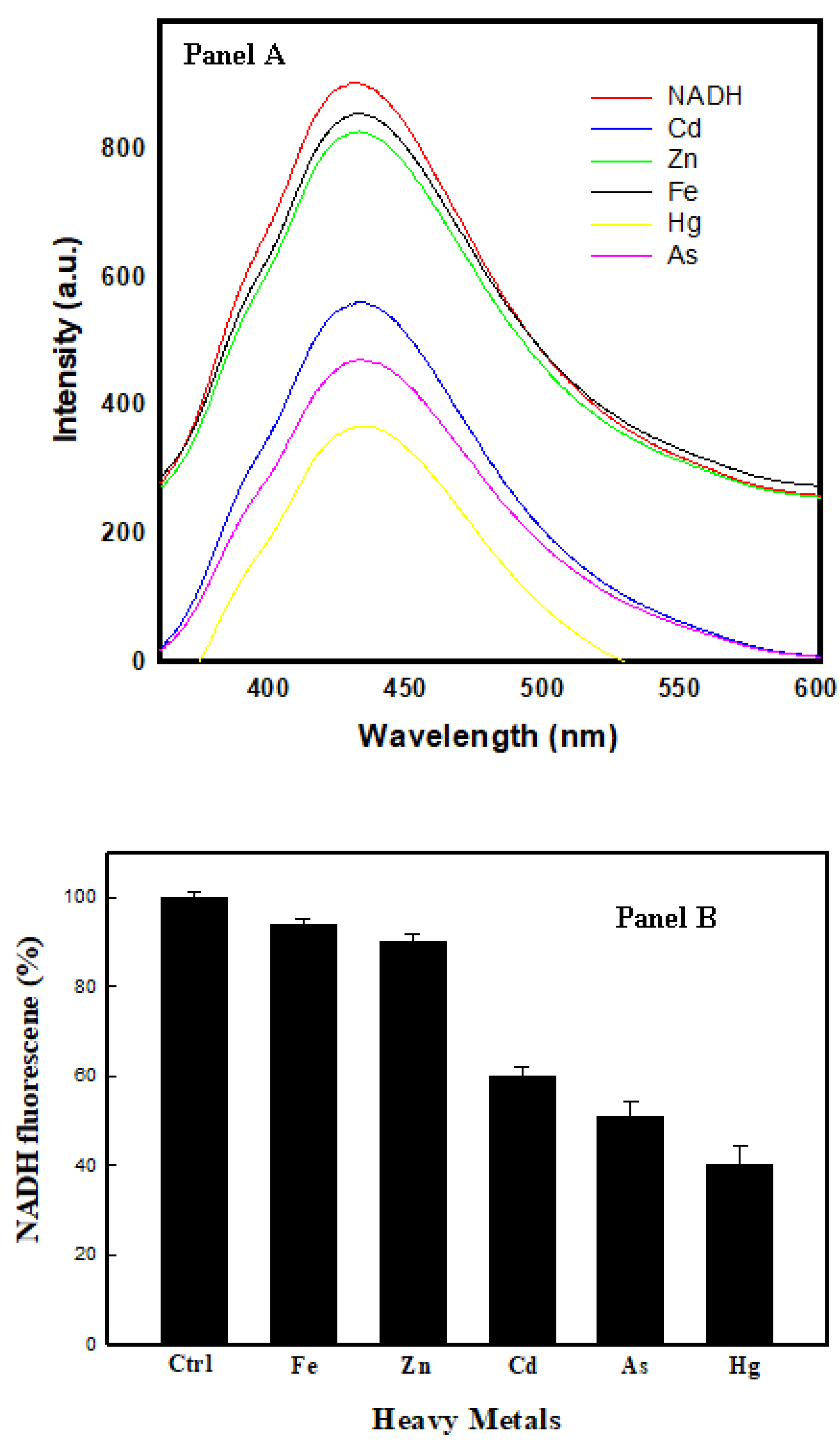
3.4. Fluorescence-Based Assay
3.5. Assessment of Toxicity Biosensor
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Srivastava, N.; Majumder, C. Novel biofiltration methods for the treatment of heavy metals from industrial wastewater. J. Hazard. Mater. 2008, 151, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Gou, H.; Al-Ogaidi, I.; Wu, N. Nanostructured Sensors for Detection of Heavy Metals: A Review. ACS Sustain. Chem. Eng. 2013, 1, 713–723. [Google Scholar] [CrossRef]
- Oehme, I.; Wolfbeis, O.S. Optical sensors for determination of heavy metal ions. Microchim. Acta 1997, 126, 177–192. [Google Scholar] [CrossRef]
- Verma, N.; Singh, M. A Bacillus sphaericus Based Biosensor for Monitoring Nickel Ions in Industrial Effluents and Foods. J. Autom. Methods Manag. Chem. 2006, 2006, 83427. [Google Scholar] [CrossRef]
- Han, S.; Zhu, M.; Yuan, Z.; Li, X. A methylene blue-mediated enzyme electrode for the determination of trace mercury(II), mercury(I), methylmercury, and mercury–glutathione complex. Biosens. Bioelectron. 2001, 16, 9–16. [Google Scholar] [CrossRef]
- Aruoja, V.; Dubourguier, H.-C.; Kasemets, K.; Kahru, A. Toxicity of nanoparticles of CuO, ZnO and TiO2 to microalgae Pseudokirchneriella subcapitata. Sci. Total. Environ. 2009, 407, 1461–1468. [Google Scholar] [CrossRef]
- Buledi, J.A.; Amin, S.; Haider, S.I.; Bhanger, M.I.; Solangi, A.R. A review on detection of heavy metals from aqueous media using nanomaterial-based sensors. Environ. Sci. Pollut. Res. 2020, 20, 1–9. [Google Scholar] [CrossRef]
- Borah, S.B.; Bora, T.; Baruah, S.; Dutta, J. Heavy metal ion sensing in water using surface plasmon resonance of metallic nanostructures. Groundw. Sustain. Dev. 2015, 1, 1–11. [Google Scholar] [CrossRef]
- Ravi, S.S.; Christena, L.R.; Saisubramanian, N.; Anthony, S.P. Green synthesized silver nanoparticles for selective colorimetric sensing of Hg2+ in aqueous solution at wide pH range. Analyst 2013, 138, 4370–4377. [Google Scholar] [CrossRef]
- Walters, F.; Ali, M.M.; Burwell, G.; Rozhko, S.; Tehrani, Z.; Ahmadi, E.D.; Evans, J.E.; Abbasi, H.Y.; Bigham, R.; Mitchell, J.J.; et al. A Facile Method for the Non-Covalent Amine Functionalization of Carbon-Based Surfaces for Use in Biosensor Development. Nanomaterials 2020, 10, 1808. [Google Scholar] [CrossRef]
- Devnani, H.; Satsangee, S.P. Green gold nanoparticle modified anthocyanin-based carbon paste electrode for voltammetric determination of heavy metals. Int. J. Environ. Sci. Technol. 2014, 12, 1269–1282. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, S.Y.; Xu, J.J.; Chen, H.Y. A new method for the synthesis of selenium nanoparticles and the application to construction of H2O2 biosensor. Chin. Chem. Lett. 2004, 15, 1345–1348. [Google Scholar]
- Eustice, D.C.; Kull, F.J.; Shrift, A. Selenium Toxicity: Aminoacylation and Peptide Bond Formation with Selenomethionine. Plant Physiol. 1981, 67, 1054–1058. [Google Scholar] [CrossRef] [PubMed]
- Lortie, L.; Gould, W.D.; Rajan, S.; Meeready, R.G.L.; Cheng, K.J. Reduction of elemental selenium by a Pseudomonas stutzeri isolate. Appl. Environ. Microbiol. 1992, 58, 4042–4044. [Google Scholar] [CrossRef] [PubMed]
- Sabaty, M.; Avazeri, C.; Pignol, D.; Vermeglio, A. Characterization of the Reduction of Selenate and Tellurite by Nitrate Reductases. Appl. Environ. Microbiol. 2001, 67, 5122–5126. [Google Scholar] [CrossRef]
- Dong, Y.; Zhang, H.; Hawthorn, L.; Ganther, H.E.; Ip, C. Delineation of the molecular basis for selenium-induced growth arrest in human prostate cancer cells by oligonucleotide array. Cancer Res. 2003, 63, 52–59. [Google Scholar]
- Turner, R.J.; Weiner, J.H.; Taylor, D.E. Selenium metabolism in Escherichia coli. BioMetals 1998, 11, 223–227. [Google Scholar] [CrossRef]
- Dwivedi, S.; Al-Khedhairy, A.A.; Ahamed, M.; Musarrat, J. Biomimetic Synthesis of Selenium Nanospheres by Bacterial Strain JS-11 and Its Role as a Biosensor for Nanotoxicity Assessment: A Novel Se-Bioassay. PLoS ONE 2013, 8, e57404. [Google Scholar] [CrossRef]
- Dwivedi, S.; Singh, B.R.; Al-Khedhairy, A.A.; Alarifi, S.; Musarrat, J. Isolation and characterization of butachlor-catabolizing bacterial strain Stenotrophomonasacidaminiphila JS-1 from soil and assessment of its biodegradation. Lett. Appl. Microbiol. 2010, 51, 54–60. [Google Scholar]
- Dwivedi, S.; Saquib, Q.; Al-Khedhairy, A.A.; Ahmad, J.; Siddiqui, M.A.; Musarrat, J. Rhamnolipids functionalized AgNPs-induced oxidative stress and modulation of toxicity pathway genes in cultured MCF-7 cells. Colloids Surf. B Biointerfaces 2015, 132, 290–298. [Google Scholar] [CrossRef]
- Musarrat, J.; Dwivedi, S.; Singh, B.R.; Al-Khedhairy, A.A.; Azam, A.; Naqvi, A. Production of antimicrobial silver nanoparticles in water extracts of the fungus Amylomycesrouxii strain KSU-09. Bioresour. Technol. 2010, 101, 8772–8776. [Google Scholar] [CrossRef] [PubMed]
- Patterson, A.L. The Scherrer Formula for X-Ray Particle Size Determination. Phys. Rev. 1939, 56, 978–982. [Google Scholar] [CrossRef]
- Wahab, R.; Dwivedi, S.; Khan, M.S.; Al-Senaidy, A.M.; Shin, H.-S.; Musarrat, J.; Al-Khedhairy, A.A.A. Optical Analysis of Zinc Oxide Quantum Dots with Bovine Serum Albumin and Bovine Hemoglobin. J. Pharm. Innov. 2014, 9, 48–52. [Google Scholar] [CrossRef]
- Hunter, W.J.; Kuykendall, L.D. Reduction of Selenite to Elemental Red Selenium by Rhizobium sp. Strain B1. Curr. Microbiol. 2007, 55, 344–349. [Google Scholar] [CrossRef] [PubMed]
- Hunter, W.J.; Kuykendall, L.D.; Manter, D.K. Rhizobium selenireducens sp. nov.: A Selenite-Reducing α-Proteobacteria Isolated from a Bioreactor. Curr. Microbiol. 2007, 55, 455–460. [Google Scholar] [CrossRef] [PubMed]
- Lloyd-Jones, G.; Williamson, W.M.; Slootweg, T. The Te-Assay: A black and white method for environmental sample pre-screening exploiting tellurite reduction. J. Microbiol. Methods 2006, 67, 549–556. [Google Scholar] [CrossRef]
- Binaeian, E.; Rashidi, A.M.; Attar, H. Toxicity study of two different synthesized silver nanoparticles on bacteria Vibrio Fischeri. World Acad. Sci. Eng. Technol. 2012, 67, 1219–1225. [Google Scholar]
- Tomei, F.A.; Barton, L.L.; Lemanski, C.L.; Zocco, T.G. Reduction of selenate and selenite to elemental selenium by Wolinella succinogenes. Can. J. Microbiol. 1992, 38, 1328–1333. [Google Scholar] [CrossRef]
- Tong, X.; Shi, S.; Tong, C.; Ali, I.; Long, R.; Zhu, Y. Quantum/carbon dots-based fluorescent assays for enzyme activity. TrAC Trends Anal. Chem. 2020, 131, 116008. [Google Scholar] [CrossRef]
- Sauer, M. Single-Molecule-Sensitive Fluorescent Sensors Based on Photoinduced Intramolecular Charge Transfer. Angew. Chem. Int. Ed. 2003, 42, 1790–1793. [Google Scholar] [CrossRef]
- Ono, A.; Togashi, H. Highly Selective Oligonucleotide-Based Sensor for Mercury(II) in Aqueous Solutions. Angew. Chem. Int. Ed. 2004, 43, 4300–4302. [Google Scholar] [CrossRef] [PubMed]
- Sulistiawaty, L.; Sugiarti, S.; Darmawan, N. Detection of Hg2+ Metal Ions Using Silver Nanoparticles Stabilized by Gelatin and Tween-20. Indones. J. Chem. 2015, 15, 1–8. [Google Scholar] [CrossRef]
- Bobik, M.; Korus, I. Controlled synthesis of iron oxide nanoparticles used as an efficient heavy metal ions adsorbent. In Proceedings of the ECOpole 9, Jarnoltowek, Poland, 14–16 October 2015; Volume 9. [Google Scholar]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahmed, F.; Dwivedi, S.; Shaalan, N.M.; Kumar, S.; Arshi, N.; Alshoaibi, A.; Husain, F.M. Development of Selenium Nanoparticle Based Agriculture Sensor for Heavy Metal Toxicity Detection. Agriculture 2020, 10, 610. https://doi.org/10.3390/agriculture10120610
Ahmed F, Dwivedi S, Shaalan NM, Kumar S, Arshi N, Alshoaibi A, Husain FM. Development of Selenium Nanoparticle Based Agriculture Sensor for Heavy Metal Toxicity Detection. Agriculture. 2020; 10(12):610. https://doi.org/10.3390/agriculture10120610
Chicago/Turabian StyleAhmed, Faheem, Sourabh Dwivedi, Nagih M. Shaalan, Shalendra Kumar, Nishat Arshi, Adil Alshoaibi, and Fohad Mabood Husain. 2020. "Development of Selenium Nanoparticle Based Agriculture Sensor for Heavy Metal Toxicity Detection" Agriculture 10, no. 12: 610. https://doi.org/10.3390/agriculture10120610
APA StyleAhmed, F., Dwivedi, S., Shaalan, N. M., Kumar, S., Arshi, N., Alshoaibi, A., & Husain, F. M. (2020). Development of Selenium Nanoparticle Based Agriculture Sensor for Heavy Metal Toxicity Detection. Agriculture, 10(12), 610. https://doi.org/10.3390/agriculture10120610









