ADMA: A Key Player in the Relationship between Vascular Dysfunction and Inflammation in Atherosclerosis
Abstract
:1. Introduction and Overview
2. Clinical Association of ADMA with Atherosclerosis
3. Underlying Mechanisms of Atherosclerosis
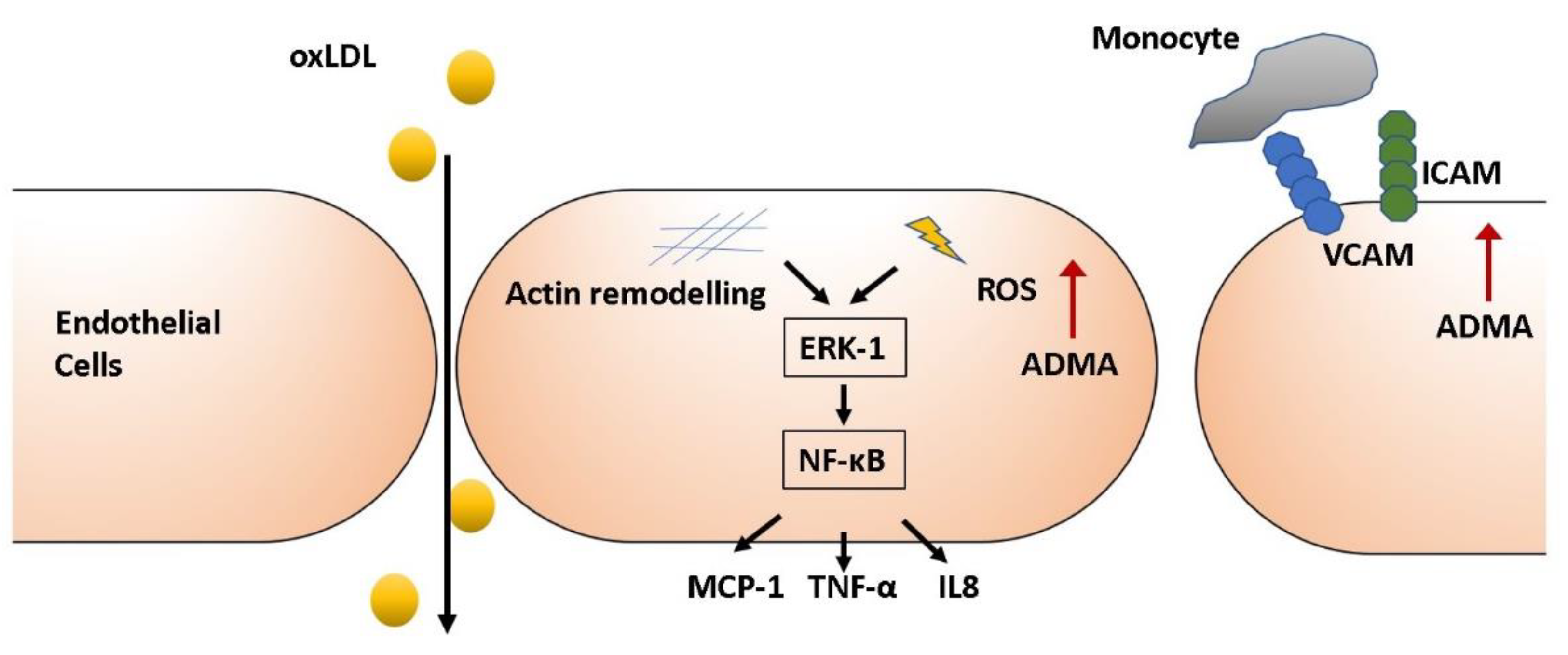
3.1. Endothelial Activation
3.2. Oxidative Stress in Endothelial Cells
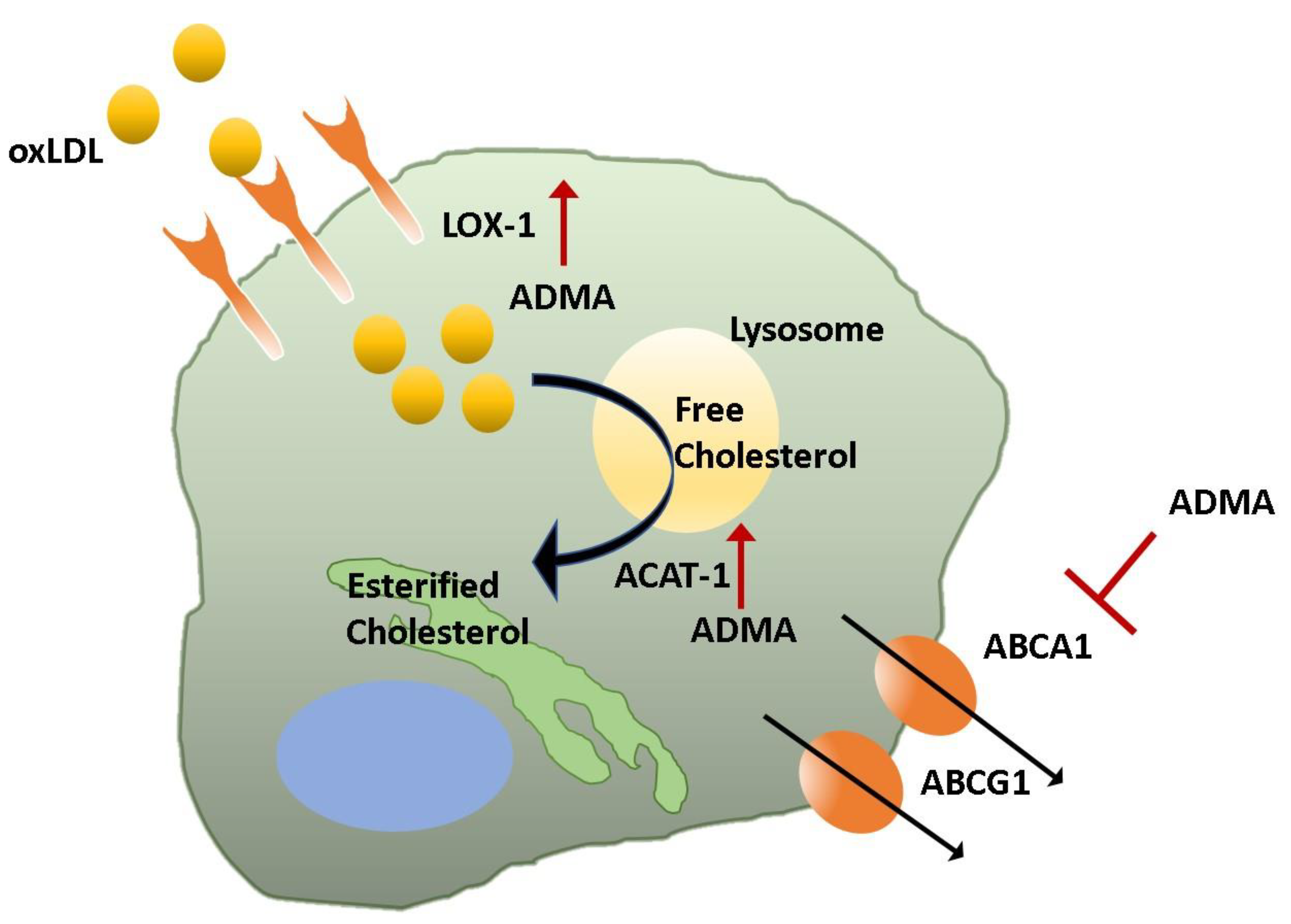
3.3. Plaque Formation
3.4. Inflammation
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Vallance, P.; Leone, A.; Calver, A.; Collier, J.; Moncada, S. Accumulation of an endogenous inhibitor of nitric oxide synthesis in chronic renal failure. Lancet 1992, 339, 572–575. [Google Scholar] [CrossRef] [PubMed]
- Böger, R.H.; Bode-Böger, S.M.; Szuba, A.; Tsao, P.S.; Chan, J.R.; Tangphao, O.; Blaschke, T.F.; Cooke, J.P. Asymmetric dimethylarginine (ADMA): A novel risk factor for endothelial dysfunction: Its role in hypercholesterolemia. Circulation 1998, 98, 1842–1847. [Google Scholar] [CrossRef] [Green Version]
- Surdacki, A.; Nowicki, M.; Sandmann, J.; Tsikas, D.; Boeger, R.H.; Bode-Boeger, S.M.; Kruszelnicka-Kwiatkowska, O.; Kokot, F.; Dubiel, J.S.; Froelich, J.C. Reduced urinary excretion of nitric oxide metabolites and increased plasma levels of asymmetric dimethylarginine in men with essential hypertension. J. Cardiovasc. Pharmacol. 1999, 33, 652–658. [Google Scholar] [CrossRef]
- Eid, H.M.; Arnesen, H.; Hjerkinn, E.M.; Lyberg, T.; Seljeflot, I. Relationship between obesity, smoking, and the endogenous nitric oxide synthase inhibitor, asymmetric dimethylarginine. Metabolism 2004, 53, 1574–1579. [Google Scholar] [CrossRef] [PubMed]
- Arlouskaya, Y.; Sawicka, A.; Głowala, M.; Giebułtowicz, J.; Korytowska, N.; Tałałaj, M.; Nowicka, G.; Wrzosek, M. Asymmetric Dimethylarginine (ADMA) and Symmetric Dimethylarginine (SDMA) concentrations in patients with obesity and the risk of Obstructive Sleep Apnea (OSA). J. Clin. Med. 2019, 8, 897. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lundman, P.; Eriksson, M.J.; Stühlinger, M.; Cooke, J.P.; Hamsten, A.; Tornvall, P. Mild-to-moderate hypertriglyceridemia in young men is associated with endothelial dysfunction and increased plasma concentrations of asymmetric dimethylarginine. J. Am. Coll. Cardiol. 2001, 38, 111–116. [Google Scholar] [CrossRef] [Green Version]
- Altinova, A.E.; Arslan, M.; Sepici-Dincel, A.; Akturk, M.; Altan, N.; Toruner, F.B. Uncomplicated type 1 diabetes is associated with increased asymmetric dimethylarginine concentrations. J. Clin. Endocrinol. Metab. 2007, 92, 1881–1885. [Google Scholar] [CrossRef] [Green Version]
- Abbasi, F.; Asagmi, T.; Cooke, J.P.; Lamendola, C.; McLaughlin, T.; Reaven, G.M.; Stuehlinger, M.; Tsao, P.S. Plasma concentrations of asymmetric dimethylarginine are increased in patients with type 2 diabetes mellitus. Am. J. Cardiol. 2001, 88, 1201–1203. [Google Scholar] [CrossRef]
- Leong, T.; Zylberstein, D.; Graham, I.; Lissner, L.; Ward, D.; Fogarty, J.; Bengtsson, C.; Björkelund, C.; Thelle, D.; Collaboration, S.-I.-N. Asymmetric dimethylarginine independently predicts fatal and nonfatal myocardial infarction and stroke in women: 24-year follow-up of the population study of women in Gothenburg. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 961–967. [Google Scholar] [CrossRef] [Green Version]
- Yoo, J.H.; Lee, S.C. Elevated levels of plasma homocyst(e)ine and asymmetric dimethylarginine in elderly patients with stroke. Atherosclerosis 2001, 158, 425–430. [Google Scholar] [CrossRef]
- Bedford, M.T.; Clarke, S.G. Protein arginine methylation in mammals: Who, what, and why. Mol. Cell 2009, 33, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Strobel, J.; Müller, F.; Zolk, O.; Endreß, B.; König, J.; Fromm, M.F.; Maas, R. Transport of asymmetric dimethylarginine (ADMA) by cationic amino acid transporter 2 (CAT2), organic cation transporter 2 (OCT2) and multidrug and toxin extrusion protein 1 (MATE1). Amino Acids 2013, 45, 989–1002. [Google Scholar] [CrossRef] [PubMed]
- Hibbs, J.B.; Vavrin, Z.; Taintor, R.R. L-arginine is required for expression of the activated macrophage effector mechanism causing selective metabolic inhibition in target cells. J. Immunol. 1987, 138, 550–565. [Google Scholar]
- Horowitz, J.D.; Heresztyn, T. An overview of plasma concentrations of asymmetric dimethylarginine (ADMA) in health and disease and in clinical studies: Methodological considerations. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2007, 851, 42–50. [Google Scholar] [CrossRef] [PubMed]
- Jarzebska, N.; Mangoni, A.A.; Martens-Lobenhoffer, J.; Bode-Böger, S.M.; Rodionov, R.N. The second life of methylarginines as cardiovascular targets. Int. J. Mol. Sci. 2019, 20, 4592. [Google Scholar] [CrossRef] [Green Version]
- Achan, V.; Broadhead, M.; Malaki, M.; Whitley, G.; Leiper, J.; MacAllister, R.; Vallance, P. Asymmetric dimethylarginine causes hypertension and cardiac dysfunction in humans and is actively metabolized by dimethylarginine dimethylaminohydrolase. Arterioscler. Thromb. Vasc. Biol. 2003, 23, 1455–1459. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Calver, A.; Collier, J.; Leone, A.; Moncada, S.; Vallance, P. Effect of local intra-arterial asymmetric dimethylarginine (ADMA) on the forearm arteriolar bed of healthy volunteers. J. Hum. Hypertens. 1993, 7, 193–194. [Google Scholar]
- Wahbi, N.; Dalton, R.N.; Turner, C.; Denton, M.; Abbs, I.; Swaminathan, R. Dimethylarginines in chronic renal failure. J. Clin. Pathol. 2001, 54, 470–473. [Google Scholar] [CrossRef] [Green Version]
- Zoccali, C.; Bode-Böger, S.; Mallamaci, F.; Benedetto, F.; Tripepi, G.; Malatino, L.; Cataliotti, A.; Bellanuova, I.; Fermo, I.; Frölich, J.; et al. Plasma concentration of asymmetrical dimethylarginine and mortality in patients with end-stage renal disease: A prospective study. Lancet 2001, 358, 2113–2117. [Google Scholar] [CrossRef]
- Leiper, J.; Nandi, M.; Torondel, B.; Murray-Rust, J.; Malaki, M.; O’Hara, B.; Rossiter, S.; Anthony, S.; Madhani, M.; Selwood, D.; et al. Disruption of methylarginine metabolism impairs vascular homeostasis. Nat. Med. 2007, 13, 198–203. [Google Scholar] [CrossRef]
- Hu, X.; Atzler, D.; Xu, X.; Zhang, P.; Guo, H.; Lu, Z.; Fassett, J.; Schwedhelm, E.; Böger, R.H.; Bache, R.J.; et al. Dimethylarginine dimethylaminohydrolase-1 is the critical enzyme for degrading the cardiovascular risk factor asymmetrical dimethylarginine. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 1540–1546. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dowsett, L.; Piper, S.; Slaviero, A.; Dufton, N.; Wang, Z.; Boruc, O.; Delahaye, M.; Colman, L.; Kalk, E.; Tomlinson, J.; et al. Endothelial dimethylarginine dimethylaminohydrolase 1 is an important regulator of angiogenesis but does not regulate vascular reactivity or hemodynamic homeostasis. Circulation 2015, 131, 2217–2225. [Google Scholar] [CrossRef] [PubMed]
- Matsuguma, K.; Ueda, S.; Yamagishi, S.; Matsumoto, Y.; Kaneyuki, U.; Shibata, R.; Fujimura, T.; Matsuoka, H.; Kimoto, M.; Kato, S.; et al. Molecular mechanism for elevation of asymmetric dimethylarginine and its role for hypertension in chronic kidney disease. J. Am. Soc. Nephrol. 2006, 17, 2176–2183. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dayoub, H.; Achan, V.; Adimoolam, S.; Jacobi, J.; Stuehlinger, M.C.; Wang, B.Y.; Tsao, P.S.; Kimoto, M.; Vallance, P.; Patterson, A.J.; et al. Dimethylarginine dimethylaminohydrolase regulates nitric oxide synthesis: Genetic and physiological evidence. Circulation 2003, 108, 3042–3047. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lambden, S.; Kelly, P.; Ahmetaj-Shala, B.; Wang, Z.; Lee, B.; Nandi, M.; Torondel, B.; Delahaye, M.; Dowsett, L.; Piper, S.; et al. Dimethylarginine dimethylaminohydrolase 2 regulates nitric oxide synthesis and hemodynamics and determines outcome in polymicrobial sepsis. Arterioscler. Thromb. Vasc. Biol. 2015, 35, 1382–1392. [Google Scholar] [CrossRef] [Green Version]
- Hasegawa, K.; Wakino, S.; Tatematsu, S.; Yoshioka, K.; Homma, K.; Sugano, N.; Kimoto, M.; Hayashi, K.; Itoh, H. Role of asymmetric dimethylarginine in vascular injury in transgenic mice overexpressing dimethylarginie dimethylaminohydrolase 2. Circ. Res. 2007, 101, e2–e10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lange, C.; Mowat, F.; Sayed, H.; Mehad, M.; Duluc, L.; Piper, S.; Luhmann, U.; Nandi, M.; Kelly, P.; Smith, A.; et al. Dimethylarginine dimethylaminohydrolase-2 deficiency promotes vascular regeneration and attenuates pathological angiogenesis. Exp. Eye Res. 2016, 147, 148–155. [Google Scholar] [CrossRef] [Green Version]
- Caplin, B.; Wang, Z.; Slaviero, A.; Tomlinson, J.; Dowsett, L.; Delahaye, M.; Salama, A.; Wheeler, D.C.; Leiper, J.; International Consortium for Blood Pressure Genome-Wide Association Studies. Alanine-glyoxylate aminotransferase-2 metabolizes endogenous methylarginines, regulates NO, and controls blood pressure. Arterioscler. Thromb. Vasc. Biol. 2012, 32, 2892–2900. [Google Scholar] [CrossRef] [Green Version]
- Di Pietro, N.; Formoso, G.; Pandolfi, A. Physiology and pathophysiology of oxLDL uptake by vascular wall cells in atherosclerosis. Vasc. Pharmacol. 2016, 84, 1–7. [Google Scholar] [CrossRef]
- Chan, J.R.; Böger, R.H.; Bode-Böger, S.M.; Tangphao, O.; Tsao, P.S.; Blaschke, T.F.; Cooke, J.P. Asymmetric dimethylarginine increases mononuclear cell adhesiveness in hypercholesterolemic humans. Arterioscler. Thromb. Vasc. Biol. 2000, 20, 1040–1046. [Google Scholar] [CrossRef] [Green Version]
- Eid, H.; Eritsland, J.; Larsen, J.; Arnesen, H.; Seljeflot, I. Increased levels of asymmetric dimethylarginine in populations at risk for atherosclerotic disease. Effects of pravastatin. Atherosclerosis 2003, 166, 279–284. [Google Scholar] [CrossRef]
- Miyazaki, H.; Matsuoka, H.; Cooke, J.P.; Usui, M.; Ueda, S.; Okuda, S.; Imaizumi, T. Endogenous nitric oxide synthase inhibitor: A novel marker of atherosclerosis. Circulation 1999, 99, 1141–1146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Furuki, K.; Adachi, H.; Matsuoka, H.; Enomoto, M.; Satoh, A.; Hino, A.; Hirai, Y.; Imaizumi, T. Plasma levels of asymmetric dimethylarginine (ADMA) are related to intima-media thickness of the carotid artery: An epidemiological study. Atherosclerosis 2007, 191, 206–210. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, K.; Akishita, M.; Yu, W.; Hashimoto, M.; Ohni, M.; Toba, K. Interrelationship between non-invasive measurements of atherosclerosis: Flow-Mediated dilation of brachial artery, carotid intima-media thickness and pulse wave velocity. Atherosclerosis 2004, 173, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Lind, L.; Larsson, A.; Teerlink, T. L-Arginine is related to endothelium-dependent vasodilation in resistance and conduit arteries in divergent ways-The Prospective Investigation of the Vasculature in Uppsala Seniors (PIVUS) study. Atherosclerosis 2009, 203, 544–549. [Google Scholar] [CrossRef]
- Juonala, M.; Viikari, J.S.; Alfthan, G.; Marniemi, J.; Kähönen, M.; Taittonen, L.; Laitinen, T.; Raitakari, O.T. Brachial artery flow-mediated dilation and asymmetrical dimethylarginine in the cardiovascular risk in young Finns study. Circulation 2007, 116, 1367–1373. [Google Scholar] [CrossRef] [Green Version]
- Furuki, K.; Adachi, H.; Enomoto, M.; Otsuka, M.; Fukami, A.; Kumagae, S.; Matsuoka, H.; Nanjo, Y.; Kakuma, T.; Imaizumi, T. Plasma level of asymmetric dimethylarginine (ADMA) as a predictor of carotid intima-media thickness progression: Six-Year prospective study using carotid ultrasonography. Hypertens. Res. 2008, 31, 1185–1189. [Google Scholar] [CrossRef] [Green Version]
- Maas, R.; Xanthakis, V.; Polak, J.F.; Schwedhelm, E.; Sullivan, L.M.; Benndorf, R.; Schulze, F.; Vasan, R.S.; Wolf, P.A.; Böger, R.H.; et al. Association of the endogenous nitric oxide synthase inhibitor ADMA with carotid artery intimal media thickness in the Framingham Heart Study offspring cohort. Stroke 2009, 40, 2715–2719. [Google Scholar] [CrossRef] [Green Version]
- Notsu, Y.; Yano, S.; Shibata, H.; Nagai, A.; Nabika, T. Plasma arginine/ADMA ratio as a sensitive risk marker for atherosclerosis: Shimane CoHRE study. Atherosclerosis 2015, 239, 61–66. [Google Scholar] [CrossRef]
- Abhary, S.; Burdon, K.P.; Kuot, A.; Javadiyan, S.; Whiting, M.J.; Kasmeridis, N.; Petrovsky, N.; Craig, J.E. Sequence variation in DDAH1 and DDAH2 genes is strongly and additively associated with serum ADMA concentrations in individuals with type 2 diabetes. PLoS ONE 2010, 5, e9462. [Google Scholar] [CrossRef] [Green Version]
- Ding, H.; Wu, B.; Wang, H.; Lu, Z.; Yan, J.; Wang, X.; Shaffer, J.R.; Hui, R.; Wang, D.W. A novel loss-of-function DDAH1 promoter polymorphism is associated with increased susceptibility to thrombosis stroke and coronary heart disease. Circ. Res. 2010, 106, 1145–1152. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Amir, M.; Hassanein, S.I.; Abdel Rahman, M.F.; Gad, M.Z. AGXT2 and DDAH-1 genetic variants are highly correlated with serum ADMA and SDMA levels and with incidence of coronary artery disease in Egyptians. Mol. Biol. Rep. 2018, 45, 2411–2419. [Google Scholar] [CrossRef] [PubMed]
- Xuan, C.; Xu, L.Q.; Tian, Q.W.; Li, H.; Wang, Q.; He, G.W.; Lun, L.M. Dimethylarginine Dimethylaminohydrolase 2 (DDAH 2) Gene Polymorphism, Asymmetric Dimethylarginine (ADMA) concentrations, and risk of coronary artery disease: A case-control study. Sci. Rep. 2016, 6, 33934. [Google Scholar] [CrossRef] [PubMed]
- Valkonen, V.P.; Tuomainen, T.P.; Laaksonen, R. DDAH gene and cardiovascular risk. Vasc. Med. 2005, 10 (Suppl. S1), S45–S48. [Google Scholar] [CrossRef]
- Zsuga, J.; Torok, J.; Magyar, M.T.; Valikovics, A.; Gesztelyi, R.; Kéki, S.; Csiba, L.; Zsuga, M.; Bereczki, D. Serum asymmetric dimethylarginine negatively correlates with intima-media thickness in early-onset atherosclerosis. Cerebrovasc. Dis. 2007, 23, 388–394. [Google Scholar] [CrossRef]
- Xuan, C.; Tian, Q.; Li, H.; Bei-Bei, Z.; He, G.; Lun, L. Levels of asymmetric dimethylarginine (ADMA), an endogenous nitric oxide synthase inhibitor, and risk of coronary artery disease: A meta-analysis based on 4713 participants. Eur. J. Prev. Cardiol. 2015, 23, 502–510. [Google Scholar] [CrossRef]
- Bai, Y.; Sun, L.; Du, L.; Zhang, T.; Xin, W.; Lan, X.; Du, G. Association of circulating levels of asymmetric dimethylarginine (ADMA) with carotid intima-media thickness: Evidence from 6168 participants. Ageing Res. Rev. 2013, 12, 699–707. [Google Scholar] [CrossRef]
- Zeller, M.; Korandji, C.; Guilland, J.; Sicard, P.; Vergely, C.; Lorgis, L.; Beer, J.; Duvillard, L.; Lagrost, A.; Moreau, D.; et al. Impact of asymmetric dimethylarginine on mortality after acute myocardial infarction. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 954–960. [Google Scholar] [CrossRef] [Green Version]
- Schnabel, R.; Blankenberg, S.; Lubos, E.; Lackner, K.; Rupprecht, H.; Espinola-Klein, C.; Jachmann, N.; Post, F.; Peetz, D.; Bickel, C.; et al. Asymmetric Dimethylarginine and the risk of cardiovascular events and death in patients with coronary artery disease. Circ. Res. 2005, 97, e53–e59. [Google Scholar] [CrossRef] [Green Version]
- Willeit, P.; Freitag, D.; Laukkanen, J.; Chowdhury, S.; Gobin, R.; Mayr, M.; Angelantonio, E.; Chowdhury, R. Asymmetric Dimethylarginine and cardiovascular risk: Systematic review and meta-analysis of 22 prospective studies. J. Am. Heart Assoc. 2015, 4. [Google Scholar] [CrossRef] [Green Version]
- Moore, K.J.; Sheedy, F.J.; Fisher, E.A. Macrophages in atherosclerosis: A dynamic balance. Nat. Rev. Immunol. 2013, 13, 709–721. [Google Scholar] [CrossRef] [PubMed]
- Gimbrone, M.A.; García-Cardeña, G. Endothelial cell dysfunction and the pathobiology of atherosclerosis. Circ. Res. 2016, 118, 620–636. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Warboys, C.M.; Amini, N.; de Luca, A.; Evans, P.C. The role of blood flow in determining the sites of atherosclerotic plaques. F1000 Med. Rep. 2011, 3, 5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bennett, M.R.; Sinha, S.; Owens, G.K. Vascular smooth muscle cells in atherosclerosis. Circ. Res. 2016, 118, 692–702. [Google Scholar] [CrossRef]
- Yu, X.J.; Li, Y.J.; Xiong, Y. Increase of an endogenous inhibitor of nitric oxide synthesis in serum of high cholesterol fed rabbits. Life Sci. 1994, 54, 753–758. [Google Scholar] [CrossRef]
- Böger, R.H.; Bode-Böger, S.M.; Sydow, K.; Heistad, D.D.; Lentz, S.R. Plasma concentration of asymmetric dimethylarginine, an endogenous inhibitor of nitric oxide synthase, is elevated in monkeys with hyperhomocyst(e)inemia or hypercholesterolemia. Arterioscler. Thromb. Vasc. Biol. 2000, 20, 1557–1564. [Google Scholar] [CrossRef] [Green Version]
- Jacobi, J.; Maas, R.; Cardounel, A.J.; Arend, M.; Pope, A.J.; Cordasic, N.; Heusinger-Ribeiro, J.; Atzler, D.; Strobel, J.; Schwedhelm, E.; et al. Dimethylarginine dimethylaminohydrolase overexpression ameliorates atherosclerosis in apolipoprotein E-deficient mice by lowering asymmetric dimethylarginine. Am. J. Pathol. 2010, 176, 2559–2570. [Google Scholar] [CrossRef]
- Xiao, H.B.; Yang, Z.C.; Jia, S.J.; Li, N.S.; Jiang, D.J.; Zhang, X.H.; Guo, R.; Zhou, Z.; Deng, H.W.; Li, Y.J. Effect of asymmetric dimethylarginine on atherogenesis and erythrocyte deformability in apolipoprotein E deficient mice. Life Sci. 2007, 81, 1–7. [Google Scholar] [CrossRef]
- Suda, O.; Tsutsui, M.; Morishita, T.; Tasaki, H.; Ueno, S.; Nakata, S.; Tsujimoto, T.; Toyohira, Y.; Hayashida, Y.; Sasaguri, Y.; et al. Asymmetric dimethylarginine produces vascular lesions in endothelial nitric oxide synthase-deficient mice: Involvement of renin-angiotensin system and oxidative stress. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 1682–1688. [Google Scholar] [CrossRef] [Green Version]
- Konishi, H.; Sydow, K.; Cooke, J.P. Dimethylarginine dimethylaminohydrolase promotes endothelial repair after vascular injury. J. Am. Coll. Cardiol. 2007, 49, 1099–1105. [Google Scholar] [CrossRef] [Green Version]
- Sitia, S.; Tomasoni, L.; Atzeni, F.; Ambrosio, G.; Cordiano, C.; Catapano, A.; Tramontana, S.; Perticone, F.; Naccarato, P.; Camici, P.; et al. From endothelial dysfunction to atherosclerosis. Autoimmun. Rev. 2010, 9, 830–834. [Google Scholar] [CrossRef] [PubMed]
- Wojciak-Stothard, B.; Torondel, B.; Zhao, L.; Renné, T.; Leiper, J.M. Modulation of Rac1 activity by ADMA/DDAH regulates pulmonary endothelial barrier function. Mol. Biol. Cell 2009, 20, 33–42. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, Y.H.; Xu, X.; Sheng, M.J.; Zheng, Z.; Gu, Q. Effects of asymmetric dimethylarginine on bovine retinal capillary endothelial cell proliferation, reactive oxygen species production, permeability, intercellular adhesion molecule-1, and occludin expression. Mol. Vis. 2011, 17, 332–340. [Google Scholar] [PubMed]
- Wang, L.Y.; Zhang, D.L.; Zheng, J.F.; Zhang, Y.; Zhang, Q.D.; Liu, W.H. Apelin-13 passes through the ADMA-damaged endothelial barrier and acts on vascular smooth muscle cells. Peptides 2011, 32, 2436–2443. [Google Scholar] [CrossRef] [PubMed]
- Watson, C.P.; Pazarentzos, E.; Fidanboylu, M.; Padilla, B.; Brown, R.; Thomas, S.A. The transporter and permeability interactions of asymmetric dimethylarginine (ADMA) and L-arginine with the human blood-brain barrier in vitro. Brain Res. 2016, 1648, 232–242. [Google Scholar] [CrossRef] [Green Version]
- Wojciak-Stothard, B.; Torondel, B.; Tsang, L.Y.; Fleming, I.; Fisslthaler, B.; Leiper, J.M.; Vallance, P. The ADMA/DDAH pathway is a critical regulator of endothelial cell motility. J. Cell Sci. 2007, 120, 929–942. [Google Scholar] [CrossRef] [Green Version]
- Krieglstein, C.F.; Granger, D.N. Adhesion molecules and their role in vascular disease. Am. J. Hypertens. 2001, 14, 44S–54S. [Google Scholar] [CrossRef] [Green Version]
- Konukoglu, D.; Firtina, S.; Serin, O. The relationship between plasma asymmetrical dimethyl-L-arginine and inflammation and adhesion molecule levels in subjects with normal, impaired, and diabetic glucose tolerance. Metabolism 2008, 57, 110–115. [Google Scholar] [CrossRef]
- Nanayakkara, P.W.; Teerlink, T.; Stehouwer, C.D.; Allajar, D.; Spijkerman, A.; Schalkwijk, C.; ter Wee, P.M.; van Guldener, C. Plasma asymmetric dimethylarginine (ADMA) concentration is independently associated with carotid intima-media thickness and plasma soluble vascular cell adhesion molecule-1 (sVCAM-1) concentration in patients with mild-to-moderate renal failure. Kidney Int. 2005, 68, 2230–2236. [Google Scholar] [CrossRef] [Green Version]
- Konukoglu, D.; Uzun, H.; Firtina, S.; Cigdem Arica, P.; Kocael, A.; Taskin, M. Plasma adhesion and inflammation markers: Asymmetrical dimethyl-L-arginine and secretory phospholipase A2 concentrations before and after laparoscopic gastric banding in morbidly obese patients. Obes. Surg. 2007, 17, 672–678. [Google Scholar] [CrossRef]
- Guo, W.-K.; Zhang, D.-L.; Wang, X.; Kong, W.; Zhang, Y.; Zhang, Q.-D.; Liu, W. Actin cytoskeleton modulates ADMA-induced NF-kappaB nuclear translocation and ICAM-1 expression in endothelial cells. Med. Sci. Monit. 2011, 17, BR242–BR247. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guo, W.; Zhang, D.; Wang, L.; Zhang, Y.; Liu, W. Disruption of asymmetric dimethylarginine-induced RelA/P65 association with actin in endothelial cells. Acta Biochim. Biophys. Sin. 2013, 45, 229–235. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smirnova, I.V.; Sawamura, T.; Goligorsky, M.S. Upregulation of lectin-like oxidized low-density lipoprotein receptor-1 (LOX-1) in endothelial cells by nitric oxide deficiency. Am. J. Physiol. Ren. Physiol. 2004, 287, F25–F32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Böger, R.H.; Bode-Böger, S.M.; Tsao, P.S.; Lin, P.S.; Chan, J.R.; Cooke, J.P. An endogenous inhibitor of nitric oxide synthase regulates endothelial adhesiveness for monocytes. J. Am. Coll. Cardiol. 2000, 36, 2287–2295. [Google Scholar] [CrossRef] [Green Version]
- Jiang, J.L.; Wang, S.; Li, N.S.; Zhang, X.H.; Deng, H.W.; Li, Y.J. The inhibitory effect of simvastatin on the ADMA-induced inflammatory reaction is mediated by MAPK pathways in endothelial cells. Biochem. Cell Biol. 2007, 85, 66–77. [Google Scholar] [CrossRef]
- Gareus, R.; Kotsaki, E.; Xanthoulea, S.; van der Made, I.; Gijbels, M.J.; Kardakaris, R.; Polykratis, A.; Kollias, G.; de Winther, M.P.; Pasparakis, M. Endothelial cell-specific NF-kappaB inhibition protects mice from atherosclerosis. Cell Metab. 2008, 8, 372–383. [Google Scholar] [CrossRef] [Green Version]
- Scalera, F.; Borlak, J.; Beckmann, B.; Martens-Lobenhoffer, J.; Thum, T.; Täger, M.; Bode-Böger, S.M. Endogenous nitric oxide synthesis inhibitor asymmetric dimethyl L-arginine accelerates endothelial cell senescence. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 1816–1822. [Google Scholar] [CrossRef] [Green Version]
- Minamino, T.; Miyauchi, H.; Yoshida, T.; Ishida, Y.; Yoshida, H.; Komuro, I. Endothelial cell senescence in human atherosclerosis: Role of telomere in endothelial dysfunction. Circulation 2002, 105, 1541–1544. [Google Scholar] [CrossRef] [Green Version]
- Azumi, H.; Inoue, N.; Ohashi, Y.; Terashima, M.; Mori, T.; Fujita, H.; Awano, K.; Kobayashi, K.; Maeda, K.; Hata, K.; et al. Superoxide generation in directional coronary atherectomy specimens of patients with angina pectoris: Important role of NAD(P)H oxidase. Arterioscler. Thromb. Vasc. Biol. 2002, 22, 1838–1844. [Google Scholar] [CrossRef]
- Vassalle, C.; Bianchi, S.; Bianchi, F.; Landi, P.; Battaglia, D.; Carpeggiani, C. Oxidative stress as a predictor of cardiovascular events in coronary artery disease patients. Clin. Chem. Lab. Med. 2012, 50, 1463–1468. [Google Scholar] [CrossRef] [Green Version]
- Sydow, K.; Münzel, T. ADMA and oxidative stress. Atheroscler. Suppl. 2003, 4, 41–51. [Google Scholar] [CrossRef]
- Wilcox, C.S. Asymmetric dimethylarginine and reactive oxygen species: Unwelcome twin visitors to the cardiovascular and kidney disease tables. Hypertension 2012, 59, 375–381. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Antoniades, C.; Shirodaria, C.; Leeson, P.; Antonopoulos, A.; Warrick, N.; Van-Assche, T.; Cunnington, C.; Tousoulis, D.; Pillai, R.; Ratnatunga, C.; et al. Association of plasma asymmetrical dimethylarginine (ADMA) with elevated vascular superoxide production and endothelial nitric oxide synthase uncoupling: Implications for endothelial function in human atherosclerosis. Eur. Heart J. 2009, 30, 1142–1150. [Google Scholar] [CrossRef] [Green Version]
- Patel, R.P.; Levonen, A.; Crawford, J.H.; Darley-Usmar, V.M. Mechanisms of the pro- and anti-oxidant actions of nitric oxide in atherosclerosis. Cardiovasc. Res. 2000, 47, 465–474. [Google Scholar] [CrossRef] [Green Version]
- Alacam, H.; Avci, B.; Şaliş, O.; Dilek, A.; Kozan, A.; Mertoğlu, C.; Şahin, M.; Okuyucu, A. Does ADMA affect the oxidant/antioxidant balance in rats? Turk. J. Med. Sci. 2013, 43, 405–410. [Google Scholar] [CrossRef]
- Toth, J.; Racz, A.; Kaminski, P.M.; Wolin, M.S.; Bagi, Z.; Koller, A. Asymmetrical dimethylarginine inhibits shear stress-induced nitric oxide release and dilation and elicits superoxide-mediated increase in arteriolar tone. Hypertension 2007, 49, 563–568. [Google Scholar] [CrossRef] [Green Version]
- Veresh, Z.; Racz, A.; Lotz, G.; Koller, A. ADMA impairs nitric oxide-mediated arteriolar function due to increased superoxide production by angiotensin II-NAD(P)H oxidase pathway. Hypertension 2008, 52, 960–966. [Google Scholar] [CrossRef] [Green Version]
- Leiper, J.; Murray-Rust, J.; McDonald, N.; Vallance, P. S-nitrosylation of dimethylarginine dimethylaminohydrolase regulates enzyme activity: Further interactions between nitric oxide synthase and dimethylarginine dimethylaminohydrolase. Proc. Natl. Acad. Sci. USA 2002, 99, 13527–13532. [Google Scholar] [CrossRef] [Green Version]
- Stühlinger, M.C.; Tsao, P.S.; Her, J.H.; Kimoto, M.; Balint, R.F.; Cooke, J.P. Homocysteine impairs the nitric oxide synthase pathway: Role of asymmetric dimethylarginine. Circulation 2001, 104, 2569–2575. [Google Scholar] [CrossRef]
- Ito, A.; Tsao, P.S.; Adimoolam, S.; Kimoto, M.; Ogawa, T.; Cooke, J.P. Novel mechanism for endothelial dysfunction: Dysregulation of dimethylarginine dimethylaminohydrolase. Circulation 1999, 99, 3092–3095. [Google Scholar] [CrossRef] [Green Version]
- Böger, R.H.; Sydow, K.; Borlak, J.; Thum, T.; Lenzen, H.; Schubert, B.; Tsikas, D.; Bode-Böger, S.M. LDL cholesterol upregulates synthesis of asymmetrical dimethylarginine in human endothelial cells: Involvement of S-adenosylmethionine-dependent methyltransferases. Circ. Res. 2000, 87, 99–105. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Poston, R.N. Atherosclerosis: Integration of its pathogenesis as a self-perpetuating propagating inflammation: A review. Cardiovasc. Endocrinol. Metab. 2019, 8, 51–61. [Google Scholar] [CrossRef] [PubMed]
- Ueda, S.; Kato, S.; Matsuoka, H.; Kimoto, M.; Okuda, S.; Morimatsu, M.; Imaizumi, T. Regulation of cytokine-induced nitric oxide synthesis by asymmetric dimethylarginine: Role of dimethylarginine dimethylaminohydrolase. Circ. Res. 2003, 92, 226–233. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, H.Y.; Kim, J.H.; Kim, H.S. Effect of CCL5 on dimethylarginine dimethylaminohydrolase-1 production in vascular smooth muscle cells from spontaneously hypertensive rats. Cytokine 2013, 64, 227–233. [Google Scholar] [CrossRef]
- Wang, D.; Gill, P.S.; Chabrashvili, T.; Onozato, M.L.; Raggio, J.; Mendonca, M.; Dennehy, K.; Li, M.; Modlinger, P.; Leiper, J.; et al. Isoform-Specific regulation by N(G),N(G)-dimethylarginine dimethylaminohydrolase of rat serum asymmetric dimethylarginine and vascular endothelium-derived relaxing factor/NO. Circ. Res. 2007, 101, 627–635. [Google Scholar] [CrossRef] [Green Version]
- Sun, L.; Zhang, T.; Yu, X.; Xin, W.; Lan, X.; Zhang, D.; Huang, C.; Du, G. Asymmetric dimethylarginine confers the communication between endothelial and smooth muscle cells and leads to VSMC migration through p38 and ERK1/2 signaling cascade. FEBS Lett. 2011, 585, 2727–2734. [Google Scholar] [CrossRef] [Green Version]
- Li, X.H.; Peng, J.; Tan, N.; Wu, W.H.; Li, T.T.; Shi, R.Z.; Li, Y.J. Involvement of asymmetric dimethylarginine and Rho kinase in the vascular remodeling in monocrotaline-induced pulmonary hypertension. Vasc. Pharmacol. 2010, 53, 223–229. [Google Scholar] [CrossRef]
- Zhou, Y.M.; Lan, X.; Guo, H.B.; Zhang, Y.; Ma, L.; Cao, J.B. Rho/ROCK signal cascade mediates asymmetric dimethylarginine-induced vascular smooth muscle cells migration and phenotype change. Biomed. Res. Int. 2014, 2014, 683707. [Google Scholar] [CrossRef] [Green Version]
- Ajtay, Z.; Scalera, F.; Cziráki, A.; Horváth, I.; Papp, L.; Sulyok, E.; Szabo, C.; Martens-Lobenhoffer, J.; Awiszus, F.; Bode-Böger, S.M. Stent placement in patients with coronary heart disease decreases plasma levels of the endogenous nitric oxide synthase inhibitor ADMA. Int. J. Mol. Med. 2009, 23, 651–657. [Google Scholar] [CrossRef] [Green Version]
- Huang, W.C.; Teng, H.I.; Chen, H.Y.; Wu, C.J.; Tsai, C.T.; Hsueh, C.H.; Chen, Y.Y.; Hau, W.K.; Lu, T.M. Association between asymmetric dimethylarginine and in-stent restenosis tissue characteristics assessed by optical coherence tomography. Int. J. Cardiol. 2019, 289, 131–137. [Google Scholar] [CrossRef]
- MacAllister, R.J.; Whitley, G.S.; Vallance, P. Effects of guanidino and uremic compounds on nitric oxide pathways. Kidney Int. 1994, 45, 737–742. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pekarova, M.; Kubala, L.; Martiskova, H.; Bino, L.; Twarogova, M.; Klinke, A.; Rudolph, T.K.; Kuchtova, Z.; Kolarova, H.; Ambrozova, G.; et al. Asymmetric dimethylarginine regulates the lipopolysaccharide-induced nitric oxide production in macrophages by suppressing the activation of NF-kappaB and iNOS expression. Eur. J. Pharmacol. 2013, 713, 68–77. [Google Scholar] [CrossRef] [PubMed]
- Tran, C.T.; Fox, M.F.; Vallance, P.; Leiper, J.M. Chromosomal localization, gene structure, and expression pattern of DDAH1: Comparison with DDAH2 and implications for evolutionary origins. Genomics 2000, 68, 101–105. [Google Scholar] [CrossRef] [PubMed]
- Smirnova, I.V.; Kajstura, M.; Sawamura, T.; Goligorsky, M.S. Asymmetric dimethylarginine upregulates LOX-1 in activated macrophages: Role in foam cell formation. Am. J. Physiol. Heart Circ. Physiol. 2004, 287, H782–H790. [Google Scholar] [CrossRef] [Green Version]
- Zhu, Z.D.; Jia, J.Q.; Zhang, X.; Wang, Y.J.; Wang, D.H. Asymmetric dimethylarginine upregulates the expression of ACAT-1 in THP-1 macrophage-derived foam cells. Nan Fang Yi Ke Da Xue Xue Bao 2010, 30, 2613–2618. [Google Scholar]
- Chen, C.H.; Zhao, J.F.; Hsu, C.P.; Kou, Y.R.; Lu, T.M.; Lee, T.S. The detrimental effect of asymmetric dimethylarginine on cholesterol efflux of macrophage foam cells: Role of the NOX/ROS signaling. Free Radic. Biol. Med. 2019, 143, 354–365. [Google Scholar] [CrossRef]
- Hong, D.; Gao, H.; Wang, X.; Li, L.; Li, C.; Luo, Y.; Wang, K.; Bai, Y.; Zhang, G. Asymmetric dimethylarginine triggers macrophage apoptosis via the endoplaspic reticulum stress pathway. Mol. Cell. Biochem. 2015, 398, 31–38. [Google Scholar] [CrossRef]
- Ketelhuth, D.F.; Hansson, G.K. Adaptive response of T and B cells in atherosclerosis. Circ. Res. 2016, 118, 668–678. [Google Scholar] [CrossRef] [Green Version]
- Koenig, W. Inflammation revisited: Atherosclerosis in the post-CANTOS era. Eur. Cardiol. 2017, 12, 89–91. [Google Scholar] [CrossRef]
- Nidorf, S.; Thompson, P. Why Colchicine should be considered for secondary prevention of atherosclerosis: An overview. Clin. Ther. 2019, 41, 41–48. [Google Scholar] [CrossRef] [Green Version]
- Antoniades, C.; Demosthenous, M.; Tousoulis, D.; Antonopoulos, A.S.; Vlachopoulos, C.; Toutouza, M.; Marinou, K.; Bakogiannis, C.; Mavragani, K.; Lazaros, G.; et al. Role of asymmetrical dimethylarginine in inflammation-induced endothelial dysfunction in human atherosclerosis. Hypertension 2011, 58, 93–98. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gremmel, T.; Perkmann, T.; Kopp, C.W.; Seidinger, D.; Eichelberger, B.; Koppensteiner, R.; Steiner, S.; Panzer, S. Interleukin-6 and asymmetric dimethylarginine are associated with platelet activation after percutaneous angioplasty with stent implantation. PLoS ONE 2015, 10, e0122586. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, T.; Sato, E.; Fujiwara, N.; Kawagoe, Y.; Suzuki, T.; Ueda, Y.; Yamada, S.; Shoji, H.; Takeuchi, M.; Ueda, S.; et al. Circulating levels of advanced glycation end products (AGE) and interleukin-6 (IL-6) are independent determinants of serum asymmetric dimethylarginine (ADMA) levels in patients with septic shock. Pharmacol. Res. 2009, 60, 515–518. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dowsett, L.; Higgins, E.; Alanazi, S.; Alshuwayer, N.A.; Leiper, F.C.; Leiper, J. ADMA: A Key Player in the Relationship between Vascular Dysfunction and Inflammation in Atherosclerosis. J. Clin. Med. 2020, 9, 3026. https://doi.org/10.3390/jcm9093026
Dowsett L, Higgins E, Alanazi S, Alshuwayer NA, Leiper FC, Leiper J. ADMA: A Key Player in the Relationship between Vascular Dysfunction and Inflammation in Atherosclerosis. Journal of Clinical Medicine. 2020; 9(9):3026. https://doi.org/10.3390/jcm9093026
Chicago/Turabian StyleDowsett, Laura, Erin Higgins, Sarah Alanazi, Noha A. Alshuwayer, Fiona C. Leiper, and James Leiper. 2020. "ADMA: A Key Player in the Relationship between Vascular Dysfunction and Inflammation in Atherosclerosis" Journal of Clinical Medicine 9, no. 9: 3026. https://doi.org/10.3390/jcm9093026
APA StyleDowsett, L., Higgins, E., Alanazi, S., Alshuwayer, N. A., Leiper, F. C., & Leiper, J. (2020). ADMA: A Key Player in the Relationship between Vascular Dysfunction and Inflammation in Atherosclerosis. Journal of Clinical Medicine, 9(9), 3026. https://doi.org/10.3390/jcm9093026




