Maternal Cardiovascular Dysfunction is Associated with Hypoxic Cerebral and Umbilical Doppler Changes
Abstract
:1. Introduction
2. Materials and Methods
2.1. Ultrasound Assessment
2.2. Cardiovascular Assessment
2.3. Statistical Analysis
3. Results
3.1. Cardiovascular Changes in Pathological Pregnancy Outcome
3.2. Association between Maternal Cardiovascular Parameters and Doppler Indices
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Mahendru, A.A.; Everett, T.; Wilkinson, I.B.; Lees, C.C.; McEniery, C.M. A longitudinal study of maternal cardiovascular function from preconception to the postpartum period. J. Hypertens. 2014, 32, 849–856. [Google Scholar] [CrossRef] [PubMed]
- Meah, V.L.; Cockcroft, J.R.; Backx, K.; Shave, R.; Stöhr, E.J. Cardiac output and related haemodynamics during pregnancy: A series of meta-analyses. Heart 2016, 102, 518–526. [Google Scholar] [CrossRef] [PubMed]
- Tay, J.; Foo, L.; Masini, G.; Bennett, P.R.; McEniery, C.M.; Wilkinson, I.B.; Lees, C.C. Early and late preeclampsia are characterized by high cardiac output, but in the presence of fetal growth restriction, cardiac output is low: Insights from a prospective study. Am. J. Obstet. Gynecol. 2018, 218, 517.e1–517.e12. [Google Scholar] [CrossRef]
- Tay, J.; Masini, G.; McEniery, C.M.; Giussani, D.A.; Shaw, C.J.; Wilkinson, I.B.; Bennett, P.R.; Lees, C.C. Uterine and fetal placental Doppler indices are associated with maternal cardiovascular function. Am. J. Obstet. Gynecol. 2019, 220, 96.e1–96.e8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Humphries, A.; Mirjalili, S.A.; Tarr, G.P.; Thompson, J.M.D.; Stone, P. The effect of supine positioning on maternal hemodynamics during late pregnancy. J. Matern. Neonatal Med. 2018, 32, 3923–3930. [Google Scholar] [CrossRef] [PubMed]
- Okwose, N.C.; Chowdhury, S.; Houghton, D.; Trenell, M.I.; Eggett, C.; Bates, M.; MacGowan, G.A.; Jakovljevic, D.G. Comparison of cardiac output estimates by bioreactance and inert gas rebreathing methods during cardiopulmonary exercise testing. Clin. Physiol. Funct. Imaging 2017, 38, 483–490. [Google Scholar] [CrossRef]
- Armstrong, S.; Fernando, R.; Columb, M.O. Minimally- and non-invasive assessment of maternal cardiac output: Go with the flow! Int. J. Obstet. Anesth. 2011, 20, 330–340. [Google Scholar] [CrossRef]
- Joosten, A.; Desebbe, O.; Suehiro, K.; Murphy, L.-L.; Essiet, M.; Alexander, B.; Fischer, M.-O.; Barvais, L.; Van Obbergh, L.; Maucort-Boulch, D.; et al. Accuracy and precision of non-invasive cardiac output monitoring devices in perioperative medicine: A systematic review and meta-analysis † †This Article is accompanied by Editorial Aew442. Br. J. Anaesth. 2017, 118, 298–310. [Google Scholar] [CrossRef] [Green Version]
- Masini, G.; Foo, L.F.; Cornette, J.; Tay, J.; Rizopoulos, D.; McEniery, C.M.; Wilkinson, I.B.; Lees, C.C. Cardiac output changes from prior to pregnancy to post partum using two non-invasive techniques. Heart 2018, 105, 715–720. [Google Scholar] [CrossRef]
- Peyton, P.J.; Thompson, B. Agreement of an Inert Gas Rebreathing Device with Thermodilution and the Direct Oxygen Fick Method in Measurement of Pulmonary Blood Flow. J. Clin. Monit. 2004, 18, 373–378. [Google Scholar] [CrossRef]
- Saur, J.; Fluechter, S.; Trinkmann, F.; Papavassiliu, T.; Schoenberg, S.; Weissmann, J.; Haghi, D.; Borggrefe, M.; Kaden, J.J. Noninvasive Determination of Cardiac Output by the Inert-Gas-Rebreathing Method—Comparison with Cardiovascular Magnetic Resonance Imaging. Cardiology 2009, 114, 247–254. [Google Scholar] [CrossRef] [PubMed]
- Vinayagam, D.; Patey, O.; Thilaganathan, B.; Khalil, A. Cardiac output assessment in pregnancy: Comparison of two automated monitors with echocardiography. Ultrasound Obstet. Gynecol. 2017, 49, 32–38. [Google Scholar] [CrossRef] [Green Version]
- Gagliardi, G.; Tiralongo, G.M.; Lopresti, D.; Pisani, I.; Farsetti, D.; Vasapollo, B.; Novelli, G.P.; Andreoli, A.; Valensise, H. Screening for pre-eclampsia in the first trimester: Role of maternal hemodynamics and bioimpedance in non-obese patients. Ultrasound Obstet. Gynecol. 2017, 50, 584–588. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Staelens, A.S.E.; Vonck, S.; Mesens, T.; Tomsin, K.; Molenberghs, G.; Gyselaers, W. Type-specific orthostatic hemodynamic response of hypertensive diseases in pregnancy. Clin. Exp. Pharmacol. Physiol. 2015, 42, 1036–1044. [Google Scholar] [CrossRef]
- Lees, C.; Marlow, N.; Arabin, B.; Bilardo, C.; Brezinka, C.; Derks, J.B.; Duvekot, J.; Frusca, T.; Diemert, A.; Ferrazzi, E.; et al. Perinatal morbidity and mortality in early-onset fetal growth restriction: Cohort outcomes of the trial of randomized umbilical and fetal flow in Europe (TRUFFLE). Ultrasound Obstet. Gynecol. 2013, 42, 400–408. [Google Scholar] [CrossRef] [PubMed]
- Gordijn, S.J.; Beune, I.M.; Thilaganathan, B.; Papageorghiou, A.; Baschat, A.A.; Baker, P.N.; Silver, R.M.; Wynia, K.; Ganzevoort, W. Consensus definition of fetal growth restriction: A Delphi procedure. Ultrasound Obstet. Gynecol. 2016, 48, 333–339. [Google Scholar] [CrossRef]
- Parra-Cordero, M.; Lees, C.; Missfelder-Lobos, H.; Seed, P.T.; Harris, C. Fetal arterial and venous Doppler pulsatility index and time averaged velocity ranges. Prenat. Diagn. 2007, 27, 1251–1257. [Google Scholar] [CrossRef]
- Foo, F.L.; Mahendru, A.A.; Masini, G.; Fraser, A.; Cacciatore, S.; MacIntyre, D.; McEniery, C.M.; Wilkinson, I.B.; Bennett, P.R.; Lees, C.C. Association Between Prepregnancy Cardiovascular Function and Subsequent Preeclampsia or Fetal Growth RestrictionNovelty and Significance. Hypertension 2018, 72, 442–450. [Google Scholar] [CrossRef]
- Tomsin, K.; Mesens, T.; Molenberghs, G.; Gyselaers, W. Impedance cardiography in uncomplicated pregnancy and pre-eclampsia: A reliability study. J. Obstet. Gynaecol. 2012, 32, 630–634. [Google Scholar] [CrossRef] [Green Version]
- Tomsin, K.; Mesens, T.; Molenberghs, G.; Peeters, L.; Gyselaers, W. Characteristics of heart, arteries, and veins in low and high cardiac output preeclampsia. Eur. J. Obstet. Gynecol. Reprod. Biol. 2013, 169, 218–222. [Google Scholar] [CrossRef]
- Tomsin, K.; Oben, J.; Staelens, A.; Molenberghs, G.; Mesens, T.; Peeters, L.; Gyselaers, W. PP078. The influence of maternal position on gestational hemodynamics. Pregnancy Hypertens. 2013, 3, 96. [Google Scholar] [CrossRef] [PubMed]
- Thomas, S.H. Impedance cardiography using the Sramek-Bernstein method: Accuracy and variability at rest and during exercise. Br. J. Clin. Pharmacol. 1992, 34, 467–476. [Google Scholar] [PubMed]
- Pisani, I.; Tiralongo, G.; Gagliardi, G.; Scala, R.; Todde, C.; Frigo, M.; Valensise, H. The maternal cardiovascular effect of carbetocin compared to oxytocin in women undergoing caesarean section. Pregnancy Hypertens. 2012, 2, 139–142. [Google Scholar] [CrossRef] [PubMed]
- Khong, T.Y.; Wolf, F.; Robertson, W.B.; Brosens, I. Inadequate maternal vascular response to placentation in pregnancies complicated by pre-eclampsia and by small-for-gestational age infants. BJOG Int. J. Obstet. Gynaecol. 1986, 93, 1049–1059. [Google Scholar] [CrossRef] [PubMed]
- Kingdom, J.C.; Audette, M.C.; Hobson, S.R.; Windrim, R.C.; Morgen, E. A placenta clinic approach to the diagnosis and management of fetal growth restriction. Am. J. Obstet. Gynecol. 2018, 218, S803–S817. [Google Scholar] [CrossRef] [Green Version]
- Pathak, S.; Lees, C.C.; Hackett, G.; Jessop, F.; Sebire, N.J. Frequency and clinical significance of placental histological lesions in an unselected population at or near term. Virchows Archiv 2011, 459, 565–572. [Google Scholar] [CrossRef]
- Browne, V.A.; Julian, C.G.; Toledo-Jaldin, L.; Cioffi-Ragan, D.; Vargas, E.; Moore, L.G. Uterine artery blood flow, fetal hypoxia and fetal growth. Philos. Trans. R. Soc. B Biol. Sci. 2015, 370, 20140068. [Google Scholar] [CrossRef] [Green Version]
- Buddeberg, B.S.; Sharma, R.; O’Driscoll, J.M.; Agten, A.K.; Khalil, A.; Thilaganathan, B. Cardiac maladaptation in term pregnancies with preeclampsia. Pregnancy Hypertens. 2018, 13, 198–203. [Google Scholar] [CrossRef]
- Gyselaers, W.; Tomsin, K.; Staelens, A.S.E.; Mesens, T.; Oben, J.; Molenberghs, G. Maternal venous hemodynamics in gestational hypertension and preeclampsia. BMC Pregnancy Childbirth 2014, 14, 212. [Google Scholar] [CrossRef]
- Ersbøll, A.; Hedegaard, M.; Søndergaard, L.; Ersbøll, M.; Johansen, M. Treatment with oral beta-blockers during pregnancy complicated by maternal heart disease increases the risk of fetal growth restriction. BJOG Int. J. Obstet. Gynaecol. 2014, 121, 618–626. [Google Scholar] [CrossRef] [Green Version]
- Tiralongo, G.M.; Pisani, I.; Vasapollo, B.; Khalil, A.; Thilaganathan, B.; Valensise, H. Effect of a nitric oxide donor on maternal hemodynamics in fetal growth restriction. Ultrasound Obstet. Gynecol. 2018, 51, 514–518. [Google Scholar] [CrossRef] [PubMed]
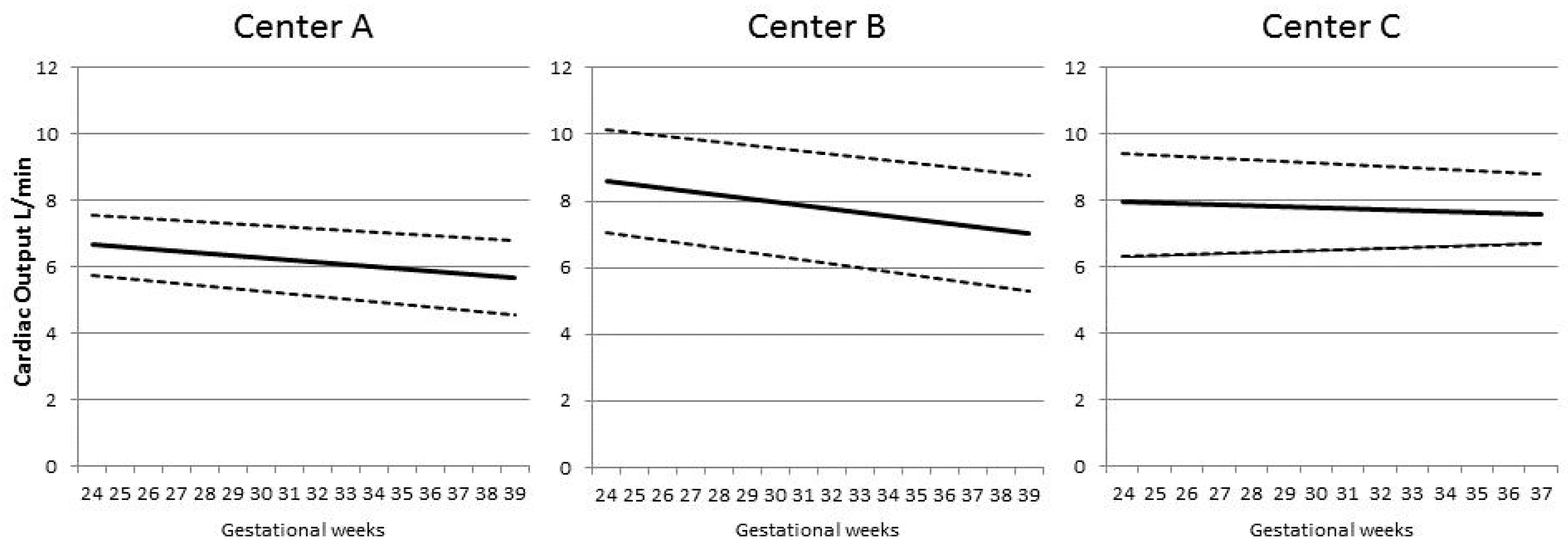
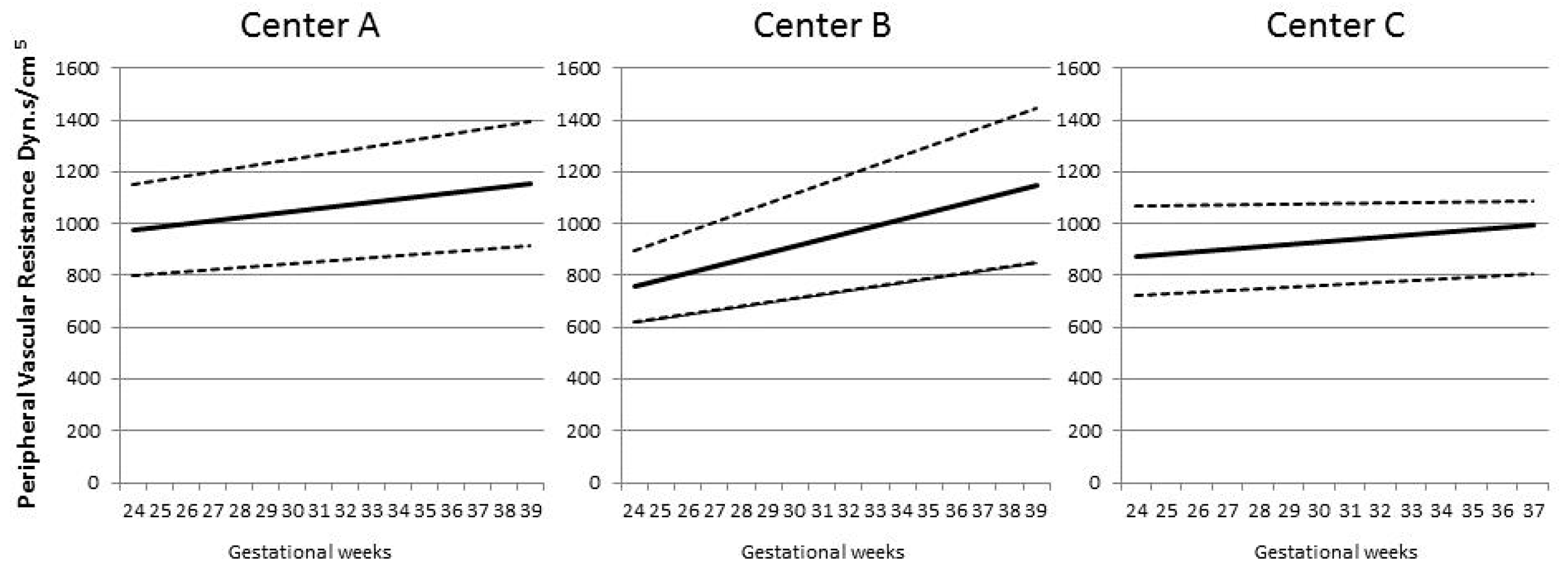

| Characteristic | Controls | Pre-Eclampsia | SGA/FGR | Pre-Eclampsia + SGA/FGR | Kruskal–Wallis p-Value |
|---|---|---|---|---|---|
| N. | 81 | 47 | 65 | 40 | - |
| Mean maternal age, years (SD) | 32.9 (4.5) | 31.6 (4.4) | 32.1 (5.2) | 31.9 (5.1) | 0.276 |
| Median BMI (range) | 23.9 (21.2–25.9) | 25.2 (22.7–29.1) | 24.1 (21.5–28.7) | 23.56 (22.5–27) | 0.082 |
| N. of nulliparous (%) | 38 (46.9) | 39 (83) | 38 (58.5) | 24 (60) | - |
| Median gestational age at CV test, weeks (IQR) | 33.6 (29.3–36.6) | 32.7 (28.1–35.9) | 32 (30.3–34.1) | 30.7 (26.6–33.5) | 0.026 |
| Median gestation at delivery, weeks (IQR) | 39 (38–40) | 34.3 (31.6–37) 1 | 36.6 (32.6–37.9) 1 | 33.1 (27.7–35.8) 1 | <0.001 |
| Median birthweight, g (IQR) | 3320 (3038.8–3552.5) | 1877.5 (1339.8–2685) 1 | 1990 (1280.5–2307.5) 1 | 1333 (704–1910) 1 | <0.001 |
| Variable | Controls | Pre-Eclampsia | SGA/FGR | Pre-Eclampsia + SGA/FGR | Kruskal–Wallis p-Value | ||||
|---|---|---|---|---|---|---|---|---|---|
| N. | Mean z-Score (SD) | N. | Mean z-Score (SD) | N. | Mean z-Score (SD) | N. | Mean z-Score (SD) | ||
| CO lying | 81 | −0.067 (0.96) | 45 | −0.009 (1.74) | 65 | −0.68 (1.34) | 40 | −0.51 (1.69) | 0.007 |
| CO standing | 81 | 0.02 (1.04) | 47 | 0.35 (2.006) | 44 | −0.48 (1.24) | 33 | −0.82 (1.24) 1 | 0.002 |
| PVR lying | 81 | 0.087 (0.92) | 45 | 1.696 (2.6) 2 | 64 | 1.52 (2.48) 2 | 39 | 2.56 (2.72) 2 | <0.001 |
| PVR standing | 81 | 0.037 (0.92) | 47 | 1.22 (2.26) | 44 | 0.83 (1.84) | 33 | 2.96 (3.27) 3 | <0.001 |
| Variable | Positive EDF | Absent/Negative EDF | p-Value (T-Test) | ||
|---|---|---|---|---|---|
| N. | Mean (SD) | N. | Mean (SD) | ||
| Lying CO | 192 | −0.22 (1.41) | 22 | −0.97 (1.57) | 0.019 |
| Lying PVR | 191 | 1.02 (2.12) | 21 | 2.88 (3.39) | 0.022 |
| Standing CO | 174 | −0.03 (1.49) | 14 | −0.91 (1.04) | 0.03 |
| Standing PVR | 174 | 0.69 (1.89) | 14 | 3.33 (4.01) | 0.029 |
| Doppler Variable | Cardiovascular Variable | r2 | p-Value | Regression Equation |
|---|---|---|---|---|
| Uterine artery PI | Lying CO z-score | 0.007 | 0.499 | y = −0.005x + 0.008x2 + 0.894 |
| Standing CO z-score | 0.024 | 0.135 | y = −0.037x + 0.015x2 + 0.868 | |
| Umbilical artery PI | Lying CO z-score | 0.008 | 0.407 | y = −0.14x + 0.013x2 + 1.466 |
| Standing CO z-score | 0.036 | 0.026 | y = −0.343x + 0.027x2 + 1.280 | |
| Middle cerebral artery PI | Lying CO z-score | 0.052 | 0.029 | y = 0.106x − 0.056x2 − 0.669 |
| Standing CO z-score | 0.018 | 0.389 | y = 0.135x − 0.016x2 − 0.826 |
| Doppler Variable | Cardiovascular Variable | r2 | p-Value | Regression Equation |
|---|---|---|---|---|
| Uterine artery PI | Lying PVR z-score | 0.049 | 0.009 | y = 0.031x + 0.001x2 + 0.870 |
| Standing PVR z-score | 0.054 | 0.011 | y = 0.060x − 0.004x2 + 0.867 | |
| Umbilical artery PI | Lying PVR z-score | 0.06 | 0.001 | y = 0.310x − 0.01x2 + 1.234 |
| Standing PVR z-score | 0.082 | <0.001 | y = 0.433x − 0.02x2 + 1.102 | |
| Middle cerebral artery PI | Lying PVR z-score | 0.062 | 0.016 | y = −0.156x + 0.003x2 − 0.613 |
| Standing PVR z-score | 0.045 | 0.088 | y = −0.167x + 0.007x2 − 0.722 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Masini, G.; Tay, J.; McEniery, C.M.; Wilkinson, I.B.; Valensise, H.; Tiralongo, G.M.; Farsetti, D.; Gyselaers, W.; Vonck, S.; Lees, C.C. Maternal Cardiovascular Dysfunction is Associated with Hypoxic Cerebral and Umbilical Doppler Changes. J. Clin. Med. 2020, 9, 2891. https://doi.org/10.3390/jcm9092891
Masini G, Tay J, McEniery CM, Wilkinson IB, Valensise H, Tiralongo GM, Farsetti D, Gyselaers W, Vonck S, Lees CC. Maternal Cardiovascular Dysfunction is Associated with Hypoxic Cerebral and Umbilical Doppler Changes. Journal of Clinical Medicine. 2020; 9(9):2891. https://doi.org/10.3390/jcm9092891
Chicago/Turabian StyleMasini, Giulia, Jasmine Tay, Carmel M McEniery, Ian B Wilkinson, Herbert Valensise, Grazia M Tiralongo, Daniele Farsetti, Wilfried Gyselaers, Sharona Vonck, and Christoph C. Lees. 2020. "Maternal Cardiovascular Dysfunction is Associated with Hypoxic Cerebral and Umbilical Doppler Changes" Journal of Clinical Medicine 9, no. 9: 2891. https://doi.org/10.3390/jcm9092891
APA StyleMasini, G., Tay, J., McEniery, C. M., Wilkinson, I. B., Valensise, H., Tiralongo, G. M., Farsetti, D., Gyselaers, W., Vonck, S., & Lees, C. C. (2020). Maternal Cardiovascular Dysfunction is Associated with Hypoxic Cerebral and Umbilical Doppler Changes. Journal of Clinical Medicine, 9(9), 2891. https://doi.org/10.3390/jcm9092891





