The Relevance of Dual Diagnoses among Drug-Dependent Patients with Sleep Disorders
Abstract
:1. Introduction
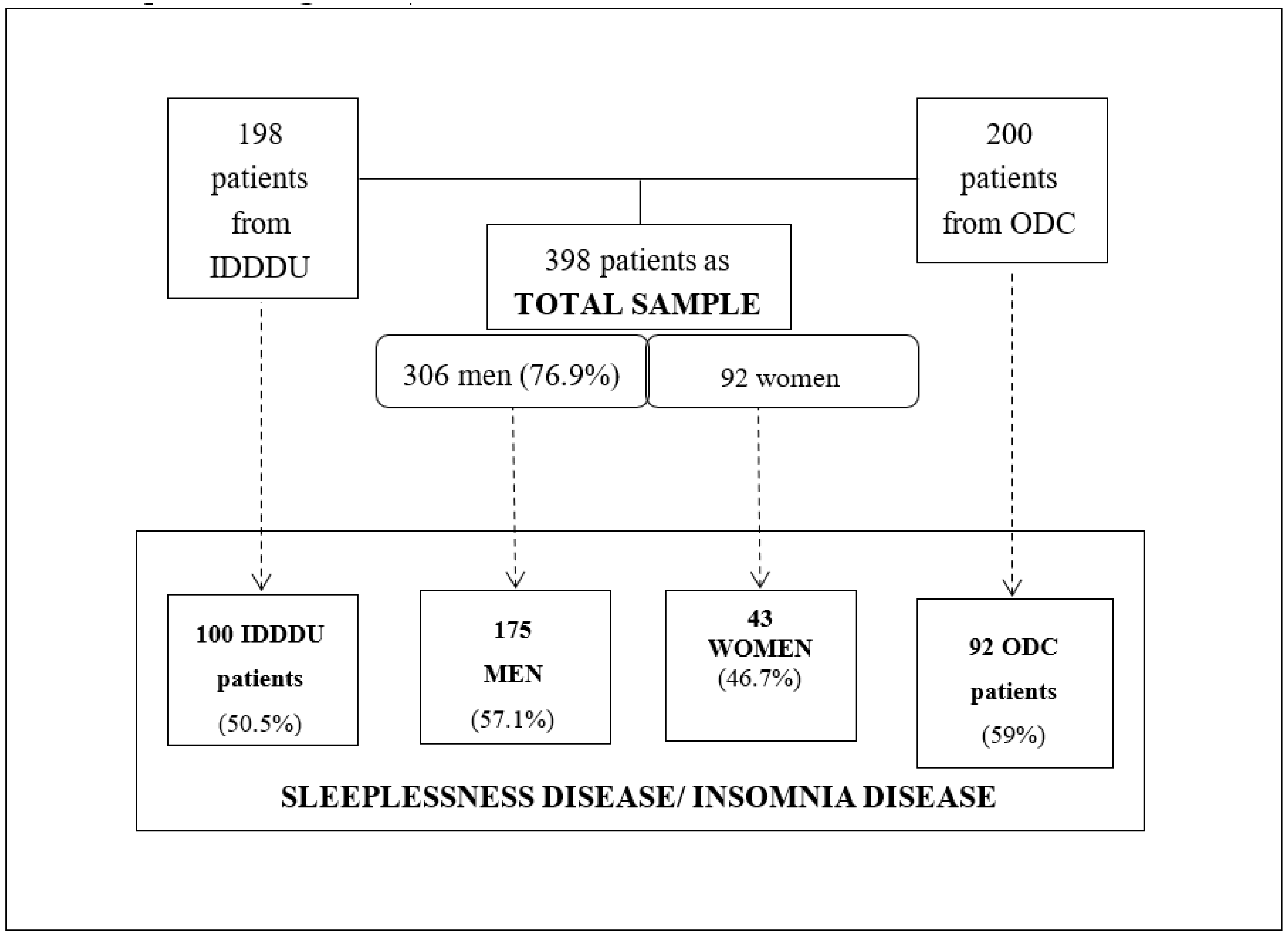
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Roncero, C.; Grau-López, L.; Díaz-Morán, S.; Miquel, L.; Martínez-Luna, N.; Casas, M. Evaluación de las alteraciones del sueño en pacientes drogodependientes hospitalizados (Evaluation of sleep disorders in hospitalized drug-dependent patients). Med. Clin. 2012, 138, 332–335. [Google Scholar] [CrossRef] [PubMed]
- Grau-López, L.; Daigre, C.; Grau-López, L.; Rodriguez-Cintas, L.; Egido, Á.; Casas, M.; Roncero, C. Administrative prevalence of insomnia and associated clinical features in patients with addiction during active substance use. Actas Esp. Psiquiatr. 2016, 44, 64–71. [Google Scholar] [PubMed]
- Johnson, E.O.; Breslau, N. Sleep problems and substance use in adolescence. Drug Alcohol Depend. 2001, 64, 1–7. [Google Scholar] [CrossRef]
- Haario, P.; Rahkonen, O.; Laaksonen, M.; Lahelma, E.; Lallukka, T. Bidirectional associations between insomnia symptoms and unhealthy behaviours. J. Sleep Res. 2013, 2289–2295. [Google Scholar] [CrossRef] [PubMed]
- Burke, C.K.; Peirce, J.M.; Kidorf, M.S.; Neubauer, D.; Punjabi, N.M.; Stoller, K.B.; Hursh, S.; Brooner, R.K. Sleep problems reported by patients entering opioid agonist treatment. J. Subst. Abuse Treat. 2008, 35, 328–333. [Google Scholar] [CrossRef] [Green Version]
- Brower, K.J.; Krentzman, A.; Robinson, E.A. Persistent insomnia, abstinence, and moderate drinking in alcohol-dependent individuals. Am. J. Addict. 2011, 20, 435–440. [Google Scholar] [CrossRef] [Green Version]
- Brower, K.J.; Aldrich, M.S.; Robinson, E.A.; Zucker, R.A.; Greden, J.F. Insomnia, self-medication, and relapse to alcoholism. Am. J. Psychiatry 2001, 158, 399–404. [Google Scholar] [CrossRef] [Green Version]
- Brower, K.J.; Perron, B.E. Sleep disturbance as a universal risk factor for relapse in addictions to psychoactive substances. Med. Hypotheses 2010, 74, 928–933. [Google Scholar] [CrossRef] [Green Version]
- Brower, K.J.; Aldrich, M.S.; Hall, J.M. Polysomnographic and subjective sleep predictors of alcoholic relapse. Alcoholism Clin. Exp. Res. 1998, 22, 1864–1871. [Google Scholar] [CrossRef]
- Morgan, P.T.; Angarita, G.A.; Canavan, S.; Pittman, B.; Oberleitner, L.; Malison, R.T.; Mohsenin, V.; Hodges, S.; Easton, C.; McKee, S.; et al. Modafinil and sleep architecture in an inpatient-outpatient treatment study of cocaine dependence. Drug Alcohol Depend. 2016, 160, 49–56. [Google Scholar] [CrossRef] [Green Version]
- Dijkstra, B.A.; De Jong, C.A.; Krabbe, P.F.; van der Staak, C.P. Prediction of abstinence in opioid-dependent patients. J. Addict. Med. 2008, 2, 194–201. [Google Scholar] [CrossRef] [PubMed]
- Vitiello, M. Sleep, alcohol and alcohol abuse. Addict. Biol. 1997, 2, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Brower, K.J. Alcohol’s effects on sleep in alcoholics. Alcohol Res. Health 2001, 25, 110–125. [Google Scholar] [PubMed]
- Arnedt, J.T.; Conroy, D.A.; Armitage, R.; Brower, K.J. Cognitive-behavioral therapy for insomnia in alcohol dependent patients: A randomized controlled pilot trial. Behav. Res. Ther. 2011, 49, 227–233. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meyrel, M.; Rolland, B.; Geoffroy, P.A. Alterations in circadian rhythms following alcohol use: A systematic review. Prog. Neuropsychopharmacol. Biol. Psychiatry 2020, 99, 109831. [Google Scholar] [CrossRef] [PubMed]
- Geoffroy, P.A.; Lejoyeuxa, M.; Rolland, B. Management of insomnia in alcohol use disorder. Expert. Opin. Pharmacother. 2020, 21, 297–306. [Google Scholar] [CrossRef]
- Conroy, D.A.; Arnedt, J.T. Sleep and substance use disorders: An update. Curr. Psychiatry Rep. 2014, 16, 487. [Google Scholar] [CrossRef]
- Haney, M.; Ward, A.S.; Comer, S.D.; Foltin, R.W.; Fischman, M.W. Abstinence symptoms following oral THC administration to humans. Psychopharmacology 1999, 141, 385–394. [Google Scholar] [CrossRef]
- Crowley, T.J.; Macdonald, M.J.; Whitmore, E.A.; Mikulich, S.K. Cannabis dependence, withdrawal, and reinforcing effects among adolescents with conduct symptoms and substance use disorders. Drug Alcohol Depend. 1998, 50, 27–37. [Google Scholar] [CrossRef]
- Brower, K.J.; Maddahian, E.; Blow, F.C.; Beresford, T.P. A comparison of self-reported symptoms and DSM-III-R criteria for cocaine withdrawal. Am. J. Drug Alcohol Abuse 1988, 14, 347–356. [Google Scholar] [CrossRef]
- Gawin, F.H.; Kleber, H.D. Abstinence symptomatology and psychiatric diagnosis in cocaine abusers. Clinical observations. Arch. Gen. Psychiatry 1986, 43, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Weddington, W.W.; Brown, B.S.; Haertzen, C.A.; Cone, E.J.; Dax, E.M.; Herning, R.I.; Michaelson, B.S. Changes in mood, craving, and sleep during short-term abstinence reported by male cocaine addicts. A controlled, residential study. Arch. Gen. Psychiatry 1990, 47, 861–868. [Google Scholar] [CrossRef] [PubMed]
- Asaad, T.A.; Ghanem, M.H.; Abdel Samee, A.M.; El–Habiby, M.M. Sleep Profile in Patients With Chronic Opioid Abuse: A Polysomnographic Evaluation in an Egyptian Sample. Addict. Disord. Treat. 2011, 10, 21–28. [Google Scholar] [CrossRef]
- Wang, D.; Teichtahl, H.; Drummer, O.; Goodman, C.; Cherry, G.; Cunnington, D.; Kronborg, I. Central sleep apnea in stable methadone maintenance treatment patients. Chest 2005, 128, 1348–1356. [Google Scholar] [CrossRef]
- Oyefeso, A.; Sedgwick, P.; Ghodse, H. Subjective sleep-wake parameters in treatment-seeking opiate addicts. Drug Alcohol Depend. 1997, 48, 9–16. [Google Scholar] [CrossRef]
- Kay, D.C. Human sleep and EEG through a cycle of methadone dependence. Electroencephalogr. Clin. Neurophysiol. 1975, 38, 35–43. [Google Scholar] [CrossRef]
- Peles, E.; Schreiber, S.; Hamburger, R.B.; Adelson, M. No change of sleep after 6 and 12 months of methadone maintenance treatment. J. Addict. Med. 2011, 5, 141–147. [Google Scholar] [CrossRef]
- Schmid, D.A.; Wichniak, A.; Uhr, M.; Ising, M.; Brunner, H.; Held, K.; Weikel, J.C.; Sonntag, A.; Steiger, A. Changes of sleep architecture, spectral composition of sleep EEG, the nocturnal secretion of cortisol, ACTH, GH, prolactin, melatonin, ghrelin, and leptin, and the DEX-CRH test in depressed patients during treatment with mirtazapine. Neuropsychopharmacology 2006, 31, 832–844. [Google Scholar] [CrossRef] [Green Version]
- Thompson, C. Onset of action of antidepressants: Results of different analyses. Hum. Psychopharmacol. 2002, 17, S27–S32. [Google Scholar] [CrossRef]
- Narowska, D.; Bozek, M.; Krysiak, K.; Antczak, J.; Holka-Pokorska, J.; Jernajczyk, W.; Wichniak, A. Frequent difficulties in the treatment of restless legs syndrome—Case report and literature review. Psychiatr. Pol. 2015, 49, 921–930. [Google Scholar] [CrossRef]
- Wichniak, A.; Wierzbicka, A.; Jernajczyk, W. Sleep and antidepressant treatment. Curr. Pharm. Des. 2012, 18, 5802–5817. [Google Scholar] [CrossRef] [PubMed]
- Wilson, S.; Argyropoulos, S. Antidepressants and sleep: A qualitative review of the literature. Drugs 2005, 65, 927–947. [Google Scholar] [CrossRef]
- Johnson, E.O.; Roth, T.; Breslau, N. The association of insomnia with anxiety disorders and depression: Exploration of the direction of risk. J. Psychiatr. Res. 2006, 40, 700–708. [Google Scholar] [CrossRef] [PubMed]
- Taylor, D.J.; Lichstein, K.L.; Durrence, H.H.; Reidel, B.W.; Bush, A.J. Epidemiology of insomnia, depression, and anxiety. Sleep 2005, 28, 1457–1464. [Google Scholar] [CrossRef]
- Tsuno, N.; Besset, A.; Ritchie, K. Sleep and Depression. J. Clin. Psychiatry 2005, 66, 1254–1269. [Google Scholar] [CrossRef] [PubMed]
- Winokur, G.; Clayton, P.J.; Reich, T. Manic Depressive Illness; Mosby: St. Louis, MO, USA, 1969; p. 165. [Google Scholar]
- Riemann, D.; Berger, M.; Voderholzer, U. Sleep and depression—Results from psychobiological studies: An overview. Biol. Psychol. 2001, 57, 67–103. [Google Scholar] [CrossRef]
- Torrens, M.; Mestre-Pintó, J.I.; Montanari, L.; Vicente, J.; Domingo-Salvany, A. Dual diagnosis: An European perspective. Adicciones 2017, 29, 3–5. [Google Scholar] [CrossRef] [Green Version]
- Stuppaeck, C.H.; Barnas, C.; Falk, M.; Guenther, V.; Hummer, M.; Oberbauer, H.; Pycha, R.; Whitworth, A.B.; Fleischhacker, W.W. Assessment of the alcohol withdrawal syndrome—Validity and reliability of the translated and modified Clinical Institute Withdrawal Assessment for Alcohol scale (CIWA-A). Addiction 1994, 89, 1287–1292. [Google Scholar] [CrossRef]
- International Classification of Sleep Disorders: Diagnostic and Coding Manual, 3rd ed.; American Academy of Sleep Medicine: Darien, IL, USA, 2014.
- Leger, D.; Guilleminault, C.; Dreyfus, J.P.; Delahaye, C.; Paillard, M. Prevalence of insomnia in a survey of 12,778 adults in France. J. Sleep Res. 2000, 9, 35–42. [Google Scholar] [CrossRef]
- Soldatos, C.R. Insomnia in relation to depression and anxiety: Epidemiologic considerations. J. Psychosom. Res. 1994, 38, 3–8. [Google Scholar] [CrossRef]
- Riemann, D. Insomnia and comorbid psychiatric disorders. Sleep Med. 2007, 8, 20. [Google Scholar] [CrossRef]
- Ohayon, M.M.; Caulet, M.; Lemoine, P. Comorbidity of mental and insomnia disorders in the general population. Compr. Psychiatry 1998, 39, 185–197. [Google Scholar] [CrossRef]
- Quera-Salva, M.A.; Orluc, A.; Goldenberg, F.; Guilleminault, C. Insomnia and use of hypnotics: Study of a French population. Sleep 1991, 14, 386–391. [Google Scholar] [CrossRef]
- Weissman, M.M.; Greenwald, S.; Niño-Murcia, G.; Dement, W.C. The morbidity of insomnia uncomplicated by psychiatric disorders. Gen. Hosp. Psychiatry 1997, 19, 245–250. [Google Scholar] [CrossRef]
- Staner, L. Comorbidity of insomnia and depression. Sleep Med. Rev. 2010, 14, 35–46. [Google Scholar] [CrossRef]
- Cernovsky, Z.Z. MMPI and nightmare reports in women addicted to alcohol and other drugs. Percept. Mot. Skills 1986, 62, 717–718. [Google Scholar] [CrossRef]
- Ford, D.E.; Kamerow, D.B. Epidemiologic study of sleep disturbances and psychiatric disorders. An opportunity for prevention? JAMA 1989, 262, 1479–1484. [Google Scholar] [CrossRef]
- Dijk, D.J. Slow-wave sleep deficiency and enhancement: Implications for insomnia and its management. World J. Biol. Psychiatry 2010, 11, 22–28. [Google Scholar] [CrossRef]
- Chait, L.D.; Zacny, J.P. Reinforcing and subjective effects of oral delta 9-THC and smoked marijuana in humans. Psychopharmacology 1992, 107, 255–262. [Google Scholar] [CrossRef]
- Budney, A.J.; Moore, B.A.; Vandrey, R.G.; Hughes, J.R. The time course and significance of cannabis withdrawal. J. Abnorm. Psychol. 2003, 112, 393–402. [Google Scholar] [CrossRef] [Green Version]
- Budney, A.J.; Hughes, J.R.; Moore, B.A.; Vandrey, R. Review of the validity and significance of cannabis withdrawal syndrome. Am. J. Psychiatry 2004, 161, 1967–1977. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vandrey, R.; Smith, M.T.; McCann, U.D.; Budney, A.J.; Curran, E.M. Sleep disturbance and the effects of extended-release zolpidem during cannabis withdrawal. Drug Alcohol Depend. 2011, 117, 38–44. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Copersino, M.L.; Boyd, S.J.; Tashkin, D.P.; Huestis, M.A.; Heishman, S.J.; Dermand, J.C.; Simmons, M.S.; Gorelick, D.A. Cannabis withdrawal among non-treatment-seeking adult cannabis users. Am. J. Addict. 2006, 15, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Schierenbeck, T.; Riemann, D.; Berger, M.; Hornyak, M. Effect of illicit recreational drugs upon sleep: Cocaine, ecstasy and marijuana. Sleep Med. Rev. 2008, 12, 381–389. [Google Scholar] [CrossRef] [PubMed]
- Garcia, A.N.; Salloum, I.M. Polysomnographic sleep disturbances in nicotine, caffeine, alcohol, cocaine, opioid, and cannabis use: A focused review. Am. J. Addict. 2015, 24, 590–598. [Google Scholar] [CrossRef]
- Angarita, G.A.; Emadi, N.; Hodges, S.; Morgan, P.T. Sleep abnormalities associated with alcohol, cannabis, cocaine, and opiate use: A comprehensive review. Addict. Sci. Clin. Pract. 2016, 11, 9. [Google Scholar] [CrossRef] [Green Version]
- Liao, Y.; Tang, J.; Liu, T.; Chen, X.; Luo, T.; Hao, W. Sleeping problems among Chinese heroin-dependent individuals. Am. J. Drug Alcohol Abuse 2011, 37, 179–183. [Google Scholar] [CrossRef]
- Kay, D.C.; Pickworth, W.B.; Neidert, G.L.; Falcone, D.; Fishman, P.M.; Othmer, E. Opioid effects on computer-derived sleep and EEG parameters in nondependent human addicts. Sleep 1979, 2, 175–191. [Google Scholar] [CrossRef] [Green Version]
- Lewis, S.A.; Oswald, I.; Evans, J.I.; Akindele, M.O.; Tompsett, S.L. Heroin and human sleep. Electroencephalogr. Clin. Neurophysiol. 1970, 28, 374–381. [Google Scholar] [CrossRef]
- Trksak, G.H.; Jensen, J.E.; Plante, D.T.; Penetar, D.M.; Tartarini, W.L.; Maywalt, M.A.; Brendel, M.; Dorsey, C.M.; Renshaw, P.F.; Lukas, S.E. Effects of sleep deprivation on sleep homeostasis and restoration during methadone-maintenance: A [31]P MRS brain imaging study. Drug Alcohol Depend. 2010, 106, 79–91. [Google Scholar] [CrossRef] [Green Version]
- de la Iglesia-Larrad, J.I.; Barral, C.; Casado-Espada, N.M.; de Alarcón, R.; Maciá-Casas, A.; Vicente Hernandez, B.; Roncero, C. Benzodiazepine abuse, misuse, dependence, and withdrawal among schizophrenic patients: A review of the literature. Psychiatry Res. 2020, 284, 112660. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, K.A.; McQuaid, J.; Batki, S.L.; Rosenlicht, N. Behavioral treatment of insomnia in early recovery. J. Addict. Med. 2014, 8, 395–398. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, C. Revisión de los diversos métodos de evaluación del trastorno de insomnio (Review of some methods of evaluation for insomnia disorder). Anales Psicol. 2007, 23, 109–117. [Google Scholar]
- Vorspan, F.; Barré, T.; Pariente, A.; Montastruc, F.; Tournier, M. Should the Duration of Treatment Be Limited Using Benzodiazepines? Presse Med. 2018, 47, 892–898. [Google Scholar] [CrossRef]
- Grau-López, L.; Grau-López, L.; Daigre, C.; Palma-Álvarez, R.F.; Rodriguez-Cintas, L.; Ros-Cucurull, E.; Roncero, C. Pharmacological Treatment of Insomnia Symptoms in Individuals with Substance Use Disorders in Spain: A Quasi-Experimental Study. Subst. Use Misuse 2018, 53, 1267–1274. [Google Scholar] [CrossRef]
| Characteristics | Sample | Insomnia/ Any Sleep Disorder | Delayed Sleep Induction | Sleep Fragmentation | Early Awakening | Nightmares | |
|---|---|---|---|---|---|---|---|
| N = 398 | N = 218 | N = 150 | N = 137 | N = 85 | N = 80 | ||
| Sex | Men | 306 (76.9%) | 175 (80.3%) | 121 (80.7%) | 107 (78.1%) | 65 (76.5%) | 53 (66.2%) |
| Women | 92 (23.1%) | 43 (19.7%) | 29 (19.3%) | 30 (21.9%) | 20 (23.5%) | 27 (33.8%) | |
| Age 18–27 years | 22 (5.5%) | 11 (5.0%) | 9 (6.0%) | 7 (5.1%) | 4 (4.7%) | 4 (5.0%) | |
| 28–37 years | 60 (15.0%) | 32 (14.7%) | 23 (15.4%) | 22 (16.1%) | 8 (9.4%) | 12 (15.0%) | |
| 38–47 years | 128 (32.2%) | 67 (30.7%) | 45 (30.0%) | 44 (32.1%) | 28 (32.9%) | 28 (35.0%) | |
| 48–57 years | 138 (34.7%) | 78 (35.8%) | 57 (38.0%) | 51 (37.2%) | 37 (43.5%) | 32 (40.0%) | |
| 58–67 years | 39 (9.8%) | 24 (11.0%) | 14 (9.3%) | 10 (7.3%) | 7 (8.3%) | 4 (5.0%) | |
| ≥68 years | 11 (2.8%) | 6 (2.8%) | 2 (1.3%) | 3 (2.2%) | 1 (1.2%) | 0 (0.0%) | |
| Origin | IDDDU | 198 (49.7%) | 100 (45.9%) | 58 (38.7%) | 58 (42.3%) | 32 (37.6%) | 31 (38.8%) |
| ODC | 200 (50.3%) | 118 (54.1%) | 92 (61.3%) | 79 (57.7%) | 53 (62.4%) | 49 (61.3%) | |
| Dual Disorder | 245 (61.6%) | 146 (67.0%) | 102 (68%) | 96 (70.1%) | 64 (75.3%) | 64 (80%) | |
| Affective disorder | 154 (40.8%) | 91 (42.7%) | 65 (43.3%) | 60 (44.1%) | 42 (50.6%) | 41 (51.9%) | |
| Psychotic disorder | 48 (12.7%) | 26 (12.2%) | 20 (13.3%) | 19 (14.0%) | 10 (12%) | 13 (16.5%) | |
| Personality disorder | 86 (22.3%) | 53 (24.3%) | 40 (26.7%) | 39 (28.5%) | 24 (28.2%) | 30 (37.5%) | |
| Previous treatment | 172 (43.2%) | 103 (49.5%) | 81 (55.9%) | 69 (53.1%) | 49 (61.3%) | 48 (62.3%) | |
| Multiple drug-users | 193 (48.5%) | 117 (53.7%) | 88 (58.7%) | 78 (57.0%) | 49 (57.6%) | 50 (62.5%) | |
| Occupational status | |||||||
| Working | 75 (18.8%) | 31 (14.2%) | 22 (14.7%) | 18 (13.1%) | 10 (11.8%) | 6 (7.5%) | |
| Unemployed | 151 (37.9%) | 96 (44.0%) | 68 (45.3%) | 66 (48.2%) | 45 (52.9%) | 46 (57.5%) | |
| On leave | 43 (10.8%) | 19 (8.7%) | 11 (7.3%) | 11 (8.0%) | 5 (5.9%) | 6 (7.5%) | |
| Retired | 108 (27.1%) | 61 (28.0%) | 40 (26.7%) | 24 (17.5%) | 19 (22.3%) | 20 (25.0%) | |
| Other conditions | 21 (5.4%) | 11 (5.1%) | 9 (6.0%) | 18 (13.1%) | 6 (7.1%) | 2 (2.5%) | |
| Pharmacological treatment on first interview | |||||||
| Alprazolam | 17 (4.3%) | 9 (4.1%) | 7 (4.7%) | 8 (5.8%) | 4 (4.7%) | 2 (2.5%) | |
| Disulfiram | 13 (3.3%) | 3 (1.4%) | 2 (1.3%) | 2 (1.5%) | 1 (1.2%) | 2 (3.4%) | |
| Clorazepate | 7 (1.8%) | 3 (1.4%) | 2 (1.3%) | 0 (0.0%) | 0 (0.0%) | 1 (1.3%) | |
| Diazepam | 13 (3.3%) | 3 (1.4%) | 1 (0.7%) | 1 (0.7%) | 0 (0.0%) | 1 (1.3%) | |
| Lorazepam | 24 (6.0%) | 15 (6.9%) | 10 (6.7%) | 8 (5.8%) | 8 (9.4%) | 6 (7.5%) | |
| Lormetazepam | 1 (0.3%) | 5 (2.3%) | 4 (2.7%) | 2 (1.5%) | 3 (3.5%) | 2 (2.5%) | |
| Methadone | 2 (0.5%) | 1 (0.6%) | 1 (0.7%) | 2 (1.5%) | 0 (0.0%) | 0 (0.0%) | |
| Mirtazapine | 8 (2.0%) | 6 (2.8%) | 6 (4.0%) | 3 (2.2%) | 3 (3.5%) | 3 (3.8%) | |
| Olanzapine | 9 (2.3%) | 7 (3.2%) | 4 (2.7%) | 5 (3.6%) | 2 (2.4%) | 3 (3.8%) | |
| Pregabalin | 4 (1.0%) | 4 (1.8%) | 3 (2.0%) | 3 (2.2%) | 0 (0.0%) | 2 (2.5%) | |
| Quetiapine | 12 (3.0%) | 4 (1.8%) | 2 (1.3%) | 1 (0.7%) | 2 (2.4%) | 2 (2.5%) | |
| Tiapride | 12 (3.0%) | 7 (3.2%) | 5 (3.3%) | 4 (2.9%) | 4 (4.7%) | 4 (5.0%) | |
| Trazodone | 10 (2.5%) | 7 (3.2%) | 5 (3.3%) | 5 (3.6%) | 4 (4.7%) | 3 (3.8%) | |
| Type of Consumers | Sample N = 398 | Insomnia/any Sleep Disorder N = 218 | Delayed Sleep Induction N = 150 | Sleep Fragmentation N = 137 | Early Awakening N = 85 | Nightmares N = 80 |
|---|---|---|---|---|---|---|
| N (%) | N (%) | N (%) | N (%) | N (%) | N (%) | |
| Cannabis Consumers | 115 (28.9%) | 65 (29.8%) | 50 (33.3%) | 43 (31.4%) | 23 (27.0%) | 25 (31.2%) |
| Quantity of Cannabis | ||||||
| 1–5 U | 95 (23.9%) | 55 (25.2%) | 42 (28.0%) | 36 (26.3%) | 20 (23.5%) | 22 (27.5%) |
| 6–10 U | 2 (0.5%) | 1 (0.5%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) |
| 11–15 U | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) |
| 16–30 U | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) |
| 31 U or more | 18 (4.5%) | 9 (4.1%) | 8 (5.3%) | 7 (5.1%) | 3 (3.5%) | 3 (3.8%) |
| Cannabis withdrawal | 38 (9.9%) | 22 (10.1%) | 17 (11.3%) | 18 (13.1%) | 7 (8.2%) | 9 (11.3%) |
| Cocaine consumers | 113 (28.4%) | 83 (38.1%) | 67 (44.7%) | 49 (64.2%) | 30 (35.3%) | 36 (45.0%) |
| Quantity of Cocaine | ||||||
| 1 g/week | 30 (7.5%) | 19 (9.5%) | 13 (9.6%) | 13 (10.1%) | 6 (7.6%) | 9 (12.3%) |
| 2 g/week | 21 (5.3%) | 13 (6.5%) | 11 (8.1%) | 9 (7.0%) | 3 (3.8%) | 3 (4.1%) |
| 3 g/week | 9 (2.3%) | 7 (3.5%) | 6 (4.4%) | 4 (3.1%) | 4 (5.1%) | 4 (5.5%) |
| 4 g/week | 11 (2.8%) | 6 (3.0%) | 5 (1.5%) | 1 (0.8%) | 1 (1.3%) | 3 (5.5%) |
| 5 g/week | 7 (1.0%) | 4 (2.0%) | 2 (1.5%) | 4 (3.1%) | 3 (3.8%) | 1 (1.4%) |
| 6 g/week or more | 35 (8.8%) | 17 (8.5%) | 15 (11.1%) | 10 (7.8%) | 7 (8.9%) | 9 (12.3%) |
| Alcohol consumers | 236 (66.1%) | 142 (65.1%) | 93 (62.0%) | 87 (63.5%) | 57 (67.1%) | 47 (58.7%) |
| Quantity of Alcohol | ||||||
| 1–20 units/day | 220 (55.3%) | 117 (53.7%) | 77(53.1%) | 71 (51.8%) | 47 (55.3%) | 38 (47.5%) |
| 21–40 units/day | 36 (9.0%) | 21 (9.6%) | 14 (9.3%) | 14 (10.2%) | 8 (9.4%) | 8 (10.0%) |
| 41 units/day or more | 7 (1.8%) | 3 (1.4%) | 2 (1.3%) | 2 (1.5%) | 2 (2.4%) | 1 (1.3%) |
| CIWA–Ar ≥ 10 | 169 (44.1%) | 94 (43.1%) | 58 (38.7%) | 55 (40.1%) | 30 (35.3%) | 25 (31.3%) |
| Opioids consumers | 72 (18.1%) | 48 (22.0%) | 40 (26.7%) | 32 (23.4%) | 21 (24.7%) | 17 (21.2%) |
| Quantity of Opioids | ||||||
| 1–10 g/week | 72 (18.1%) | 48 (22.0%) | 40 (26.7%) | 32 (23.4%) | 21 (24.7%) | 17 (21.2%) |
| Benzodiazepines consumers | 93 (23.7%) | 61 (28.0%) | 48 (32.0%) | 46 (33.6%) | 30 (35.3%) | 31 (38.7%) |
| 1–10 mg/day | 47 (11.8%) | 32 (14.7%) | 24 (16.0%) | 23 (16.8%) | 15 (17.6%) | 15 (12.4%) |
| 11–21 mg/day | 12 (3.0%) | 8 (3.7%) | 7 (4.7%) | 8 (5.8%) | 5 (5.9%) | 4 (5.0%) |
| 22–31 mg/day | 1 (0.3%) | 1 (0.5%) | 1 (0.7%) | 1 (0.7%) | 1 (1.2%) | 0 (0.0%) |
| 32 mg or more | 33 (8.3%) | 20 (9.2%) | 16 (10.7%) | 14 (10.2%) | 9 (10.6%) | 12 (15.0%) |
| Characteristics | Insomnia/Any Sleep Disorder (n = 218) | Delayed Sleep Induction (n = 150) | Sleep Fragmentation (n = 216) | Early Awakening (n = 85) | Nightmares (n = 80) | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| χ2 | p | χ2 | p | χ2 | p | χ2 | p | χ2 | p | |
| Sex | 3.023 | 0.082 | 1.781 | 0.182 | 0.121 | 0.728 | 0.016 | 0.898 | 7.069 | 0.008 * |
| Age | 2.585 | 0.764 | 2.758 | 0.737 | 1.322 | 0.93 | 5.313 | 0.379 | 5.346 | 0.375 |
| IDDDU/ODC | 2.092 | 0.148 | 10.48 | 0.010 * | 3.456 | 0.063 | 5.598 | 0.018 * | 3.898 | 0.048 * |
| Dual Disorder | 4.267 | 0.039 * | 2.877 | 0.090 | 4.862 | 0.027 * | 7.554 | 0.006 * | 12.988 | 0.000 * |
| Previous treatment | 2.442 | 0.118 | 9.291 | 0.002 * | 4.180 | 0.041 * | 9.913 | 0.002 * | 10.814 | 0.001 * |
| Affective disorder | 1.935 | 0.380 | 1.365 | 0.505 | 1.579 | 0.454 | 4.640 | 0.098 | 5.598 | 0.061 |
| Psychotic disorder | 0.122 | 0.727 | 0.056 | 0.813 | 247 | 0.619 | 0.033 | 0.855 | 1.353 | 0.254 |
| Personality disorder | 1.592 | 0.207 | 1.84 | 0.175 | 4.475 | 0.034 * | 2.183 | 0.140 | 13.596 | 0.000 * |
| Multiple drug- users | 4.254 | 0.039 * | 9.368 | 0.002 * | 5.469 | 0.019 * | 3.532 | 0.060 | 7.603 | 0.006 * |
| Unemployed | 13.557 | 0.019 * | 8.867 | 0.14 * | 12.438 | 0.029 * | 12.823 | 0.025 * | 25.732 | 0.000 * |
| Pharmacological treatment on first interview | 29.55 | 0.009 * | 24.06 | 0.064 | 28.737 | 0.017 * | 19.306 | 0.2 | 13.295 | 0.579 |
| Amount of cannabis consumed | 16.935 | 0.390 | 22.760 | 0.120 | 11.728 | 0.762 | 7.919 | 0.951 | 19.666 | 0.236 |
| Cannabis withdrawal | 0.016 | 0.898 | 0.490 | 0.484 | 2.345 | 0.126 | 0.401 | 0.527 | 0.255 | 0.614 |
| Amount of alcohol consumed | 1.447 | 0.695 | 2.018 | 0.569 | 1.491 | 0.684 | 0.589 | 0.899 | 2.827 | 0.419 |
| CIWA-Ar ≥ 10 | 0.208 | 0.649 | 2.805 | 0.094 | 1.257 | 0.262 | 3.511 | 0.061 | 6.488 | 0.011 * |
| Amount of cocaine consumed | 3.852 | 0.697 | 9.53 | 0.16 | 7.294 | 0.294 | 5.99 | 0.424 | 8.41 | 0.21 |
| Amount of opioids consumed | 0.122 | 0.727 | 0.056 | 0.813 | 0.247 | 0.619 | 0.33 | 0.855 | 1.353 | 0.245 |
| Amount of benzodiazepines consumed | 9.848 | 0.043 * | 15.21 | 0.04 * | 16.924 | 0.002 * | 13.316 | 0.010 * | 18.980 | 0.001 * |
| Dependent Variable | Wald | p | OR | IC 95% |
|---|---|---|---|---|
| Insomnia/any sleep disorder | ||||
| Benzodiazepine use disorder | 4.779 | 0.029 | 0.354 | 0.140–0.898 |
| Dual Disorder | 4.246 | 0.039 | 1.553 | 1.022–2.361 |
| Treatment with trazodone | 4.626 | 0.031 | 0.129 | 0.020–0.834 |
| Treatment with pregabalin | 4.626 | 0.031 | 0.129 | 0.020–0.834 |
| Delayed sleep induction | ||||
| Origin: ODC/IDDDU | 10.364 | 0.001 | 1.991 | 1.309–3.027 |
| Previous treatment | 9.198 | 0.002 | 0.519 | 0.34–0.793 |
| Benzodiazepine use disorder | 7.189 | 0.007 | 0.319 | 0.138–0.735 |
| Treatment with pregabalin | 3.857 | 0.05 | 0.091 | 0.008–0.995 |
| Sleep fragmentation | ||||
| Dual Disorder | 4.824 | 0.028 | 0.606 | 0.388–0.948 |
| Previous treatment | 4.158 | 0.041 | 0.639 | 0.515–0.983 |
| Benzodiazepine use disorder | 4.567 | 0.033 | 0.423 | 0.192–0.931 |
| Treatment with pregabalin | 3.857 | 0.05 | 0.091 | 0.008–0.995 |
| Personality disorder | 4.426 | 0.035 | 0.590 | 0.361–0.965 |
| Early awakening | ||||
| Dual Disorder | 7.353 | 0.007 | 0.470 | 0.273–0.811 |
| Origin: ODC/IDDDU | 5.519 | 0.019 | 1.810 | 1.103–2.970 |
| Previous treatment | 9.657 | 0.002 | 0.447 | 0.269–0.743 |
| Benzodiazepine use disorder | 3.900 | 0.048 | 0.433 | 0.189–0.994 |
| Nightmares | ||||
| Origin: ODC/IDDDU | 3.856 | 0.050 | 1.656 | 1.001–2.741 |
| Previous treatment | 10.490 | 0.001 | 0.425 | 0.254–0.714 |
| Dual Disorder | 12.256 | 0.000 | 0.346 | 0.191–0.627 |
| Benzodiazepine use disorder | 9.455 | 0.002 | 0.278 | 0.123–0.629 |
| Personality disorder | 4.426 | 0.035 | 0.590 | 0.361–0.965 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roncero, C.; García-Ullán, L.; Bullón, A.; Remón-Gallo, D.; Vicente-Hernández, B.; Álvarez, A.; Caldero, A.; Flores, A.; Aguilar, L. The Relevance of Dual Diagnoses among Drug-Dependent Patients with Sleep Disorders. J. Clin. Med. 2020, 9, 2862. https://doi.org/10.3390/jcm9092862
Roncero C, García-Ullán L, Bullón A, Remón-Gallo D, Vicente-Hernández B, Álvarez A, Caldero A, Flores A, Aguilar L. The Relevance of Dual Diagnoses among Drug-Dependent Patients with Sleep Disorders. Journal of Clinical Medicine. 2020; 9(9):2862. https://doi.org/10.3390/jcm9092862
Chicago/Turabian StyleRoncero, Carlos, Llanyra García-Ullán, Alberto Bullón, Diego Remón-Gallo, Begoña Vicente-Hernández, Ana Álvarez, Amaya Caldero, Andrea Flores, and Lourdes Aguilar. 2020. "The Relevance of Dual Diagnoses among Drug-Dependent Patients with Sleep Disorders" Journal of Clinical Medicine 9, no. 9: 2862. https://doi.org/10.3390/jcm9092862
APA StyleRoncero, C., García-Ullán, L., Bullón, A., Remón-Gallo, D., Vicente-Hernández, B., Álvarez, A., Caldero, A., Flores, A., & Aguilar, L. (2020). The Relevance of Dual Diagnoses among Drug-Dependent Patients with Sleep Disorders. Journal of Clinical Medicine, 9(9), 2862. https://doi.org/10.3390/jcm9092862






