Comparison of 2-Year Outcomes between Intravitreal Ranibizumab and Intravitreal Aflibercept for Diabetic Macular Edema with “Treat-and-Extend” Regimen—Its Usefulness and Problems
Abstract
:1. Introduction
2. Experimental Section
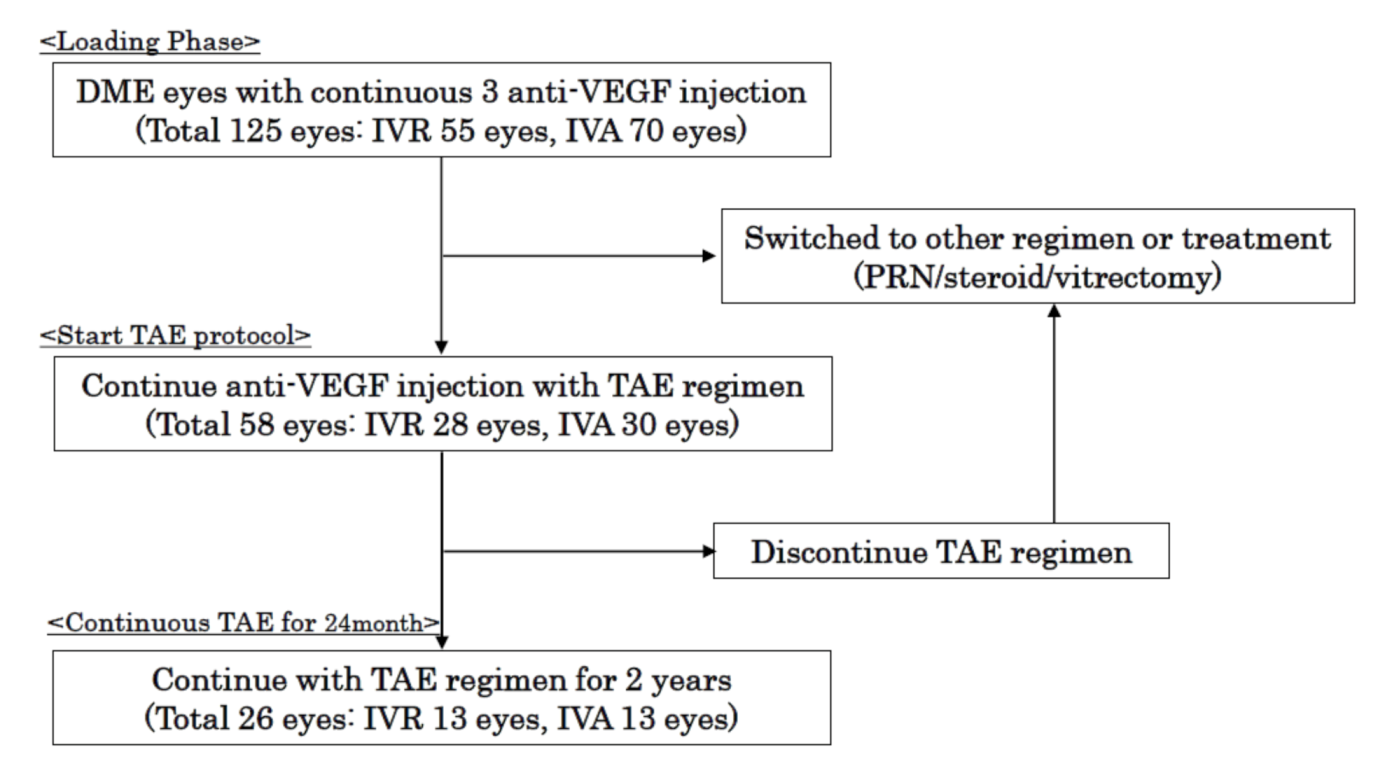
2.1. Patients and Methods
2.2. Intravitreal Injection of Anti-VEGF Agents
2.3. Modified Treat and Extend (TAE) Regimen for DME
2.4. Measurements of Best-Corrected Visual Acuity (BCVA)
2.5. Optical Coherence Tomography (OCT)
2.6. Statistical Analyses
3. Results
3.1. Clinical Characteristics of Patients at Baseline
3.2. Changes in DRSS during TAE Treatment Regimen
3.3. Changes in BCVA and CRT Following Treat-and-Extend Regimen
3.4. Number of Injections with TAE Regimen
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Data Availability
References
- McMeel, J.W.; Trempe, C.L.; Franks, E.B. Diabetic maculopathy. Trans. Sect. Ophthalmol. Am. Acad. Ophthalmol. Otolaryngol. 1977, 83, 476–487. [Google Scholar]
- Moss, S.E.; Klein, R.; Klein, B.E.K. The incidence of visual loss in a diabetic population. Ophthalmology 1988, 95, 1340–1348. [Google Scholar] [CrossRef]
- Joussen, A.M. Therapeutic approach to (Diabetic) macular edema. In Retinal Vascular Disease; Joussen, A.M., Gardner, T.W., Kirchhof, B., Ryan, S.J., Eds.; Springer: Heidelberg, Germany, 2007; pp. 353–376. [Google Scholar]
- Haritoglou, C.; Kook, D.; Neubauer, A.; Wolf, A.; Priglinger, S.; Strauss, R.; Gandorfer, A.; Ulbig, M.; Kampik, A. Intravitreal bevacizumab (Avastin) therapy for persistent diffuse diabetic macular edema. Retina 2006, 26, 999–1005. [Google Scholar] [CrossRef] [PubMed]
- Arevalo, J.F.; Fromow-Guerra, J.; Quiroz-Mercado, H.; Sanchez, J.G.; Wu, L.; Maia, M.; Berrocal, M.H.; Solis-Vivanco, A.; Farah, M.E. Pan-American Collaborative Retina Study Group. Primary intravitreal bevacizumab (Avastin) for diabetic macular edema. Ophthalmology 2007, 114, 743–750. [Google Scholar] [CrossRef]
- Elman, M.J.; Aiello, L.P.; Beck, R.W.; Bressler, N.M.; Bressler, S.B.; Edwards, A.R.; Ferris, F.L., III; Friedman, S.M.; Glassman, A.R.; Miller, K.M.; et al. Randomized trial evaluating ranibizumab plus prompt or deferred laser or triamcinolone plus prompt laser for diabetic macular edema. Ophthalmology 2010, 117, 1064–1077. [Google Scholar] [CrossRef] [Green Version]
- Elman, M.J.; Ayala, A.; Bressler, N.M.; Browning, D.; Flaxel, C.J.; Glassman, A.R.; Jampol, L.M.; Stone, T.W. Diabetic Retinopathy Clinical Research Network. Intravitreal ranibizumab for diabetic macular edema with prompt versus deferred laser treatment: 5-year randomized trial results. Ophthalmology 2015, 122, 375–381. [Google Scholar] [CrossRef] [Green Version]
- Brown, D.M.; Schmidt-Erfurth, U.; Do, D.V.; Holz, F.G.; Boyer, D.S.; Midena, E.; Heier, J.S.; Terasaki, H.; Kaiser, P.K.; Marcus, D.M.; et al. Aflibercept for Diabetic Macular Edema: 100-Week Results from the VISTA and VIVID studies. Ophthalmology 2015, 122, 2044–2052. [Google Scholar] [CrossRef]
- Sugimoto, M.; Tsukitome, H.; Okamoto, F.; Oshika, T.; Ueda, T.; Niki, M.; Mitamura, Y.; Ishikawa, H.; Gomi, F.; Kitano, S.; et al. Clinical preferences and trends of anti-vascular endothelial growth factor treatments for diabetic macular edema in Japan. J. Diabetes Investig. 2019, 10, 475–483. [Google Scholar] [CrossRef] [Green Version]
- Singer, M.A.; Awh, C.C.; Sadda, S.; Freeman, W.R.; Antoszyk, A.N.; Wong, P.; Tuomi, L. HORIZON: An open label extension trial of ranibizumab for choroidal neovascularization secondary to age-related macular degeneration. Ophthalmology 2012, 119, 1175–1183. [Google Scholar] [CrossRef]
- Engelbert, M.; Zweifel, S.A.; Freund, K.B. Long-term follow-up for type 1 (subretinal pigment epithelium) neovascularization using a modified “treat and extend” dosing regimen of intravitreal antivascular endothelial growth factor therapy. Retina 2010, 30, 1368–1375. [Google Scholar] [CrossRef]
- Gupta, O.P.; Shienbaum, G.; Patel, A.H.; Fecarotta, C.; Kaiser, R.S.; Regillo, C.D. A treat and extend regimen using ranibizumab for neovascular age-related macular degeneration clinical and economic impact. Ophthalmology 2010, 117, 2134–2140. [Google Scholar] [CrossRef] [PubMed]
- Shienbaum, G.; Gupta, O.P.; Fecarotta, C.; Patel, A.H.; Kaiser, R.S.; Regillo, C.D. Bevacizumab for neovascular age-related macular degeneration using a treatand-extend regimen: Clinical and economic impact. Am. J. Ophthalmol. 2012, 153, 468–473. [Google Scholar] [CrossRef] [PubMed]
- Oubraham, H.; Cohen, S.Y.; Samimi, S.; Marotte, D.; Bouzaher, I.; Bonicel, P.; Fajnkuchen, F.; Tadayoni, R. Inject and extend dosing versus dosing as needed: A comparative retrospective study of ranibizumab in exudative age-related macular degeneration. Retina 2011, 31, 26–30. [Google Scholar] [CrossRef] [PubMed]
- Toalster, N.; Russell, M.; Ng, P. A 12-month prospective trial of inject and extend regimen for ranibizumab treatment of age-related macular degeneration. Retina 2013, 33, 1351–1358. [Google Scholar] [CrossRef] [PubMed]
- Kim, L.N.; Mehta, H.; Barthelmes, D.; Nguyen, V.; Gillies, M.C. Meta-analysis of real-world outcomes of intravitreal ranibizumab for the treatment of neovascular age related macular degeneration. Retina 2016, 36, 1418–1431. [Google Scholar] [CrossRef] [Green Version]
- Prünte, C.; Fajnkuchen, F.; Mahmood, S.; Ricci, F.; Hatz, K.; Studnička, J.; Bezlyak, V.; Parikh, S.; Stubbings, W.J.; Wenzel, A.; et al. Ranibizumab 0.5 mg treat-and-extend regimen for diabetic macular oedema: The RETAIN study. Br. J. Ophthalmol 2016, 100, 787–795. [Google Scholar]
- Sugimoto, M.; Ichio, A.; Nunome, T.; Kondo, M. Two year result of intravitreal bevacizumab for diabetic macular edema using treat and extend protocol. Medicine (Baltim.) 2017, 96, e6406. [Google Scholar] [CrossRef]
- Payne, J.F.; Wykoff, C.C.; Clark, W.L.; Bruce, B.B.; Boyer, D.S.; Brown, D.M.; Wells, J.A.; Johnson, D.L.; Benz, M.; Chen, E.; et al. Randomized trial of treat and extend ranibizumab with and without navigated laser versus monthly dosing for diabetic macular edema: TREX-DME 2-year outcomes. Am. J. Ophthalmol. 2019, 202, 91–99. [Google Scholar] [CrossRef]
- Ebneter, A.; Waldmeier, D.; Zysset-Burri, D.C.; Wolf, S.; Zinkernagel, M.S. Comparison of two individualized treatment regimens with ranibizumab for diabetic macular edema. Graefes Arch. Clin. Exp. Ophthalmol. 2017, 255, 549–555. [Google Scholar] [CrossRef]
- Diabetic Retinopathy Clinical Research Network; A Wells, J.; Glassman, A.R.; Ayala, A.R.; Jampol, L.M.; Aiello, L.P.; Antoszyk, A.N.; Arnold-Bush, B.; Baker, C.W.; Bressler, N.M.; et al. Aflibercept, bevacizumab, or ranibizumab for diabetic macular edema. N. Engl. J. Med. 2015, 372, 1193–1203. [Google Scholar] [CrossRef] [Green Version]
- Wilkinson, C.; Ferris, F.L.; Klein, R.; Lee, P.P.; Agardh, C.D.; Davis, M.; Dills, D.; Kampik, A.; Pararajasegaram, R.; Verdaguer, J.T. Global Diabetic Retinopathy Project Group. Proposed international clinical diabetic retinopathy and diabetic macular edema disease severity scales. Ophthalmology 2003, 110, 1677–1682. [Google Scholar] [CrossRef]
- Mitchell, P.; McAllister, I.; Larsen, M.; Staurenghi, G.; Korobelnik, J.-F.; Boyer, D.S.; Do, D.V.; Brown, D.M.; Katz, T.A.; Berliner, A.; et al. Evaluating the impact of intravitreal aflibercept on diabetic retinopathy progression in the VIVID-DME and VISTA-DME studies. Ophthalmol. Retin. 2018, 2, 988–996. [Google Scholar] [CrossRef] [PubMed]
- Writing Committee for the Diabetic Retinopathy Clinical Research Network; Gross, J.G.; Glassman, A.R.; Jampol, L.M.; Inusah, S.; Aiello, L.P.; Antoszyk, A.N.; Baker, C.W.; Berger, B.B.; Bressler, N.M.; et al. Panretinal photocoagulation vs intravitreous ranibizumab for proliferative diabetic retinopathy: A randomized clinical trial. JAMA 2015, 314, 2137–2146. [Google Scholar]
- Wykoff, C.C.; Eichenbaum, D.; Roth, D.B.; Hill, L.; Fung, A.E.; Haskova, Z. Ranibizumab induces regression of diabetic retinopathy in most patients at high risk of progression to proliferative diabetic retinopathy. Ophthalmol. Retin. 2018, 2, 997–1009. [Google Scholar] [CrossRef] [PubMed]
- Sugimoto, M.; Ichio, A.; Mochida, D.; Tenma, Y.; Miyata, R.; Matsubara, H.; Kondo, M. Multiple effects of intravitreal aflibercept on microvascular regression in eyes with diabetic macular edema. Ophthalmol. Retin. 2019, 3, 1067–1075. [Google Scholar] [CrossRef]
- Flynn, H.W.; Smiddy, W.E. Diabetes and Ocular Disease; The Foundation of the American Academy of Ophthalmology: San Francisco, CA, USA, 2000. [Google Scholar]
- Diabetic Retinopathy Clinical Research Network; Browning, D.J.; Glassman, A.R.; Aiello, L.P.; Beck, R.W.; Brown, D.M.; Fong, N.S.; Bressler, N.M.; Danis, R.P.; Kinyoun, J.L.; et al. Relationship between optical coherence tomography-measured central retinal thickness and visual acuity in diabetic macular edema. Ophthalmology 2007, 114, 525–536. [Google Scholar]
- Ehlers, J.P.; Wang, K.; Singh, R.P.; Babiuch, A.S.; Schachat, A.P.; Yuan, A.; Reese, J.L.; Stiegel, L.; Srivastava, S.K. A prospective randomized comparative dosing trial of ranibizumab in bevacizumab-resistant diabetic macular edema: The REACT Study. Ophthalmol. Retin. 2018, 2, 217–224. [Google Scholar] [CrossRef]
- Julien, S.; Biesemeier, A.; Taubitz, T.; Schraermeyer, U. Different effects of intravitreally injected ranibizumab and aflibercept on retinal and choroidal tissues of monkey eyes. Br. J. Ophthalmol. 2014, 98, 813–825. [Google Scholar] [CrossRef]
- Niwa, Y.; Kakinoki, M.; Sawada, T.; Wang, X.; Ohji, M. Ranibizumab and aflibercept: Intraocular Pharmacokinetics and Their Effects on Aqueous VEGF Level in Vitrectomized and Nonvitrectomized Macaque Eyes. Invest. Ophthalmol. Vis. Sci. 2015, 56, 6501–6505. [Google Scholar] [CrossRef] [Green Version]
- Kanda, A.; Noda, K.; Saito, W.; Ishida, S. Aflibercept Traps Galectin-1, an angiogenic factor associated with diabetic retinopathy. Sci. Rep. 2015, 5, 17946. [Google Scholar] [CrossRef]
- Schwarzer, P.; Ebneter, A.; Munk, M.; Wolf, S.; Zinkernagel, M.S. One-year results of using a treat-and-extend regimen without a loading phase with anti-VEGF agents in patients with treatment-naive diabetic macular edema. Ophthalmologica 2019, 241, 220–225. [Google Scholar] [CrossRef]
- Campochiaro, P.A.; Brown, D.M.; Pearson, A.; Chen, S.; Boyer, D.; Ruiz-Moreno, J.; Garretson, B.; Gupta, A.; Hariprasad, S.M.; Bailey, C.; et al. FAME Study Group. Sustained delivery fluocinolone acetonide vitreous inserts provide benefit for at least 3 years in patients with diabetic macular edema. Ophthalmology 2012, 119, 2125–2132. [Google Scholar] [CrossRef] [PubMed]



| Agent | Age (Years) | HbA1c (%) | eGFR (ml/min/1.73m2) | BCVA (logMAR) | CRT (μm) |
|---|---|---|---|---|---|
| Ranibuzumab | 70.9 ± 6.0 | 7.6 ± 1.6 | 87.1 ± 16.1 | 0.31 ± 0.19 | 440.9 ± 69.3 |
| Aflibercept | 65.9 ± 8.6 | 7.4 ± 1.8 | 71.0 ± 21.7 | 0.41 ± 0.19 | 473.9 ± 71.5 |
| p-values | 0.08 | 0.33 | 0.31 | 0.12 | 0.33 |
| DR Stage | Ranibuzumab | Aflibercept | ||
|---|---|---|---|---|
| Baseline 24 months | ||||
| Minimum NPDR | 2 | 12 | 5 | 12 |
| Moderate NPDR | 8 | 1 | 2 | 1 |
| Severe NPDR | 3 | 3 | 0 | 6 |
| p-value | 0.0004 ** | 0.009 ** | ||
| Period | Ranibuzumab | Aflibercept | p-Value |
|---|---|---|---|
| 12 months | 7.1 ± 0.3 | 6.5 ± 0.5 | 0.13 |
| 18 months | 9.5 ± 0.8 | 8.8 ± 0.9 | 0.13 |
| 24 months | 12.0 ± 1.0 | 11.0 ± 1.2 | 0.29 |
| Study | Regimen | VA Change (Letters) | CRT Change (μm) | Injection Numbers | Injection Interval (Patients Visit) |
|---|---|---|---|---|---|
| Protocol I # | R PRN | +9.8 | w.o laser −157 | 12 | 23 times/2 years ### |
| Protocol T (IVR) | R PRN | +12.3 | −149 | 16 | 21.3 times/2 years ### |
| RETAIN (PRN) | R PRN | +8.1 | −108 ## (24.97%) | 10.7 | 2.8 months (11.2 weeks) |
| T-REX (monthly) | R Fix | +7.5 | −139 | 24.7 | 4.3 weeks |
| Protocol T (IVA) | A PRN | +12.8 | −171 | 15 | 21.4 times/2 years ### |
| RETAIN (TAE) | R TAE | +6.5 | w. o laser −113 ## | w. o laser (24.98%) 12.8 w. o laser | 2.3 months (9.2 weeks) |
| T-REX (TAE) | R TAE | +9.6 | w. o laser −140 | w. o laser 18.9 w. o laser | 6.2 weeks |
| This study (IVR) | R TAE | 0.21 | Log MAR −133.4 | 12.0 | 12.0 weeks |
| This study (IVA) | A TAE | 0.23 | Log MAR −140.5 | 11.0 | 12.2 weeks |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chujo, S.; Sugimoto, M.; Sasaki, T.; Matsui, Y.; Kato, K.; Ichio, A.; Miyata, R.; Matsubara, H.; Kondo, M. Comparison of 2-Year Outcomes between Intravitreal Ranibizumab and Intravitreal Aflibercept for Diabetic Macular Edema with “Treat-and-Extend” Regimen—Its Usefulness and Problems. J. Clin. Med. 2020, 9, 2848. https://doi.org/10.3390/jcm9092848
Chujo S, Sugimoto M, Sasaki T, Matsui Y, Kato K, Ichio A, Miyata R, Matsubara H, Kondo M. Comparison of 2-Year Outcomes between Intravitreal Ranibizumab and Intravitreal Aflibercept for Diabetic Macular Edema with “Treat-and-Extend” Regimen—Its Usefulness and Problems. Journal of Clinical Medicine. 2020; 9(9):2848. https://doi.org/10.3390/jcm9092848
Chicago/Turabian StyleChujo, Shinichiro, Masahiko Sugimoto, Taku Sasaki, Yoshitsugu Matsui, Kumiko Kato, Atsushi Ichio, Ryohei Miyata, Hisashi Matsubara, and Mineo Kondo. 2020. "Comparison of 2-Year Outcomes between Intravitreal Ranibizumab and Intravitreal Aflibercept for Diabetic Macular Edema with “Treat-and-Extend” Regimen—Its Usefulness and Problems" Journal of Clinical Medicine 9, no. 9: 2848. https://doi.org/10.3390/jcm9092848
APA StyleChujo, S., Sugimoto, M., Sasaki, T., Matsui, Y., Kato, K., Ichio, A., Miyata, R., Matsubara, H., & Kondo, M. (2020). Comparison of 2-Year Outcomes between Intravitreal Ranibizumab and Intravitreal Aflibercept for Diabetic Macular Edema with “Treat-and-Extend” Regimen—Its Usefulness and Problems. Journal of Clinical Medicine, 9(9), 2848. https://doi.org/10.3390/jcm9092848





