Dexmedetomidine: What’s New for Pediatrics? A Narrative Review
Abstract
1. Introduction
2. Updates on Pharmacology
3. Updates on End-Organ Effects
3.1. Respiration and Airway Collapsibility
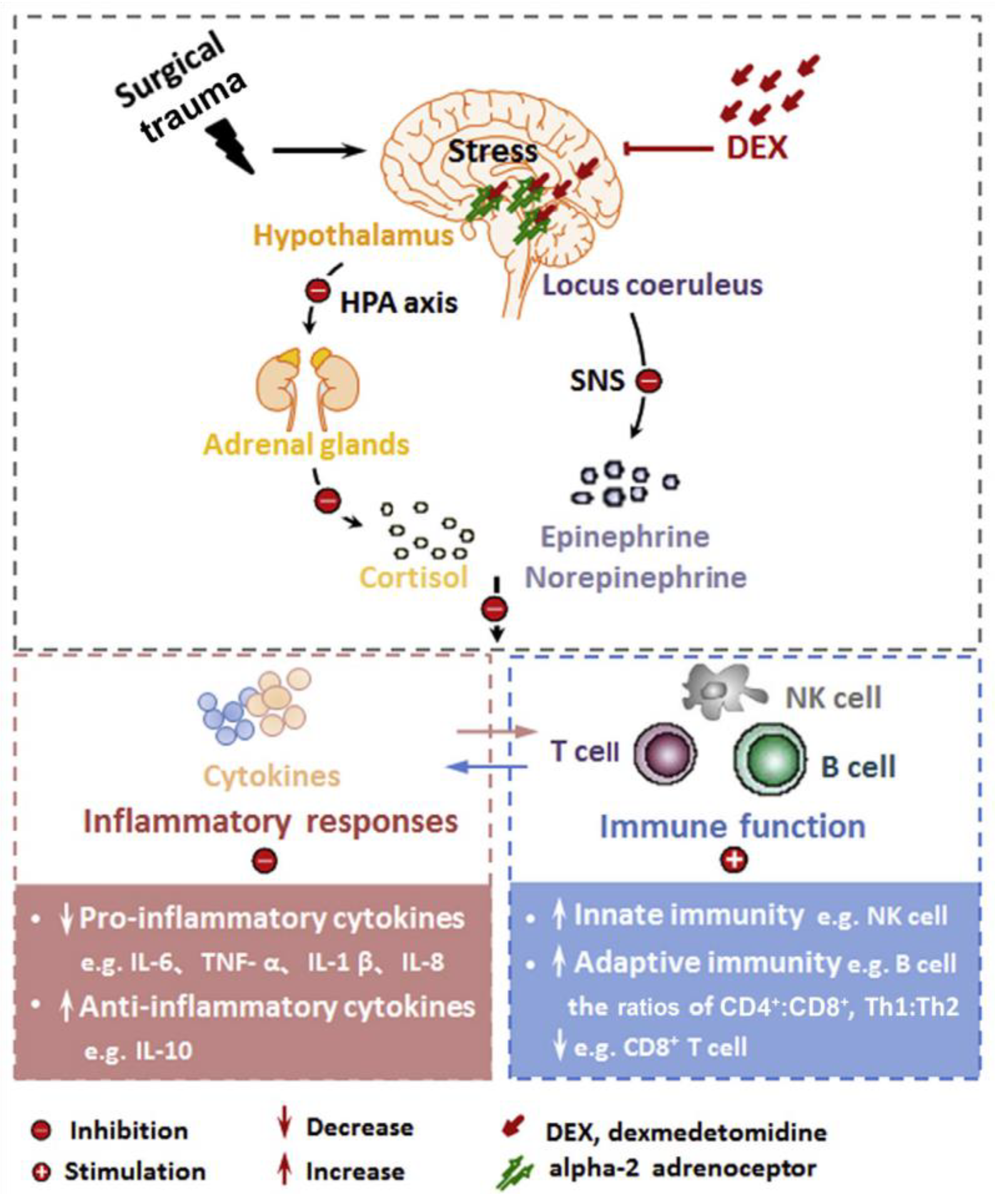
3.2. Inflammation and Immune System
3.3. Central Nervous System
3.4. Cardiovascular System
3.5. Thermal Regulation
4. Updates on Organ-Protective Effects
4.1. Neuroprotective Effects
4.2. Renoprotective Effects
4.3. Cardioprotective Effects
5. Updates on Alternative Routes of Administration
6. Updates of Synergistic Effects
6.1. Regional Anesthesia
6.2. Anesthesia
7. Post-Procedural Applications
7.1. Improving Post-Anesthesia Recovery
7.2. Neonatal Intensive Care
7.3. Pediatric Intensive Care Unit
7.4. Palliative Care
8. Limitations of the Literature and Future Directions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mahmoud, M.; Mason, K.P. Dexmedetomidine: Review, update, and future considerations of paediatric perioperative and periprocedural applications and limitations. Br. J. Anaesth. 2015, 115, 171–182. [Google Scholar] [CrossRef] [PubMed]
- Su, F.; Gastonguay, M.R.; Nicolson, S.C.; DiLiberto, M.; Ocampo-Pelland, A.; Zuppa, A.F. Dexmedetomidine Pharmacology in Neonates and Infants After Open Heart Surgery. Anesth. Analg. 2016, 122, 1556–1566. [Google Scholar] [CrossRef]
- Greenberg, R.G.; Wu, H.; Laughon, M.; Capparelli, E.; Rowe, S.; Zimmerman, K.O.; Smith, P.B.; Cohen-Wolkowiez, M. Population pharmacokinetics of dexmedetomidine in infants. J. Clin. Pharmacol. 2017, 57, 1174–1182. [Google Scholar] [CrossRef] [PubMed]
- van Dijkman, S.C.; De Cock, P.; Smets, K.; Decaluwe, W.; Smits, A.; Allegaert, K.; Vande Walle, J.; De Paepe, P.; Della Pasqua, O. Dose rationale and pharmacokinetics of dexmedetomidine in mechanically ventilated new-borns: Impact of design optimisation. Eur. J. Clin. Pharmacol. 2019, 75, 1393–1404. [Google Scholar] [CrossRef] [PubMed]
- Hannivoort, L.N.; Eleveld, D.J.; Proost, J.H.; Reyntjens, K.M.; Absalom, A.R.; Vereecke, H.E.; Struys, M.M. Development of an Optimized Pharmacokinetic Model of Dexmedetomidine Using Target-controlled Infusion in Healthy Volunteers. Anesthesiology 2015, 123, 357–367. [Google Scholar] [CrossRef]
- Miller, J.W.; Balyan, R.; Dong, M.; Mahmoud, M.; Lam, J.E.; Pratap, J.N.; Paquin, J.R.; Li, B.L.; Spaeth, J.P.; Vinks, A.; et al. Does intranasal dexmedetomidine provide adequate plasma concentrations for sedation in children: A pharmacokinetic study. Br. J. Anaesth. 2018, 120, 1056–1065. [Google Scholar] [CrossRef]
- Anttila, M.; Penttilä, J.; Helminen, A.; Vuorilehto, L.; Scheinin, H. Bioavailability of dexmedetomidine after extravascular doses in healthy subjects. Br. J. Clin. Pharmacol. 2003, 56, 691–693. [Google Scholar] [CrossRef]
- Iirola, T.; Vilo, S.; Manner, T.; Aantaa, R.; Lahtinen, M.; Scheinin, M.; Olkkola, K.T. Bioavailability of dexmedetomidine after intranasal administration. Eur. J. Clin. Pharmacol. 2011, 67, 825–831. [Google Scholar] [CrossRef]
- Uusalo, P.; Guillaume, S.; Siren, S.; Manner, T.; Vilo, S.; Scheinin, M.; Saari, T.I. Pharmacokinetics and Sedative Effects of Intranasal Dexmedetomidine in Ambulatory Pediatric Patients. Anesth. Analg. 2020, 130, 949–957. [Google Scholar] [CrossRef]
- Perez-Guille, M.G.; Toledo-Lopez, A.; Rivera-Espinosa, L.; Alemon-Medina, R.; Murata, C.; Lares-Asseff, I.; Chavez-Pacheco, J.L.; Gomez-Garduno, J.; Zamora Gutierrez, A.L.; Orozco-Galicia, C.; et al. Population Pharmacokinetics and Pharmacodynamics of Dexmedetomidine in Children Undergoing Ambulatory Surgery. Anesth. Analg. 2018, 127, 716–723. [Google Scholar] [CrossRef]
- Colin, P.J.; Hannivoort, L.N.; Eleveld, D.J.; Reyntjens, K.; Absalom, A.R.; Vereecke, H.E.M.; Struys, M. Dexmedetomidine pharmacodynamics in healthy volunteers: 2. Haemodynamic profile. Br. J. Anaesth. 2017, 119, 211–220. [Google Scholar] [CrossRef] [PubMed]
- Damian, M.A.; Hammer, G.B.; Elkomy, M.H.; Frymoyer, A.; Drover, D.R.; Su, F. Pharmacokinetics of Dexmedetomidine in Infants and Children After Orthotopic Liver Transplantation. Anesth. Analg. 2020, 130, 209–216. [Google Scholar] [CrossRef] [PubMed]
- Rolle, A.; Paredes, S.; Cortinez, L.I.; Anderson, B.J.; Quezada, N.; Solari, S.; Allende, F.; Torres, J.; Cabrera, D.; Contreras, V.; et al. Dexmedetomidine metabolic clearance is not affected by fat mass in obese patients. Br. J. Anaesth. 2018, 120, 969–977. [Google Scholar] [CrossRef] [PubMed]
- Akeju, O.; Kim, S.E.; Vazquez, R.; Rhee, J.; Pavone, K.J.; Hobbs, L.E.; Purdon, P.L.; Brown, E.N. Spatiotemporal Dynamics of Dexmedetomidine-Induced Electroencephalogram Oscillations. PLoS ONE 2016, 11, e0163431. [Google Scholar] [CrossRef] [PubMed]
- Guldenmund, P.; Vanhaudenhuyse, A.; Sanders, R.D.; Sleigh, J.; Bruno, M.A.; Demertzi, A.; Bahri, M.A.; Jaquet, O.; Sanfilippo, J.; Baquero, K.; et al. Brain functional connectivity differentiates dexmedetomidine from propofol and natural sleep. Br. J. Anaesth. 2017, 119, 674–684. [Google Scholar] [CrossRef]
- Nelson, L.E.; Lu, J.; Guo, T.; Saper, C.B.; Franks, N.P.; Maze, M. The alpha2-adrenoceptor agonist dexmedetomidine converges on an endogenous sleep-promoting pathway to exert its sedative effects. Anesthesiology 2003, 98, 428–436. [Google Scholar] [CrossRef]
- Doze, V.A.; Chen, B.X.; Maze, M. Dexmedetomidine produces a hypnotic-anesthetic action in rats via activation of central alpha-2 adrenoceptors. Anesthesiology 1989, 71, 75–79. [Google Scholar] [CrossRef]
- Hsu, Y.W.; Cortinez, L.I.; Robertson, K.M.; Keifer, J.C.; Sum-Ping, S.T.; Moretti, E.W.; Young, C.C.; Wright, D.R.; Macleod, D.B.; Somma, J. Dexmedetomidine pharmacodynamics: Part I: Crossover comparison of the respiratory effects of dexmedetomidine and remifentanil in healthy volunteers. Anesthesiology 2004, 101, 1066–1076. [Google Scholar] [CrossRef]
- Mahmoud, M.; Ishman, S.L.; McConnell, K.; Fleck, R.; Shott, S.; Mylavarapu, G.; Gutmark, E.; Zou, Y.; Szczesniak, R.; Amin, R.S. Upper Airway Reflexes are Preserved During Dexmedetomidine Sedation in Children With Down Syndrome and Obstructive Sleep Apnea. J. Clin. Sleep Med. 2017, 13, 721–727. [Google Scholar] [CrossRef]
- Chatterjee, D.; Friedman, N.; Shott, S.; Mahmoud, M. Anesthetic dilemmas for dynamic evaluation of the pediatric upper airway. Semin. Cardiothorac. Vasc. Anesth. 2014, 18, 371–378. [Google Scholar] [CrossRef]
- Mahmoud, M.; Gunter, J.; Donnelly, L.F.; Wang, Y.; Nick, T.G.; Sadhasivam, S. A comparison of dexmedetomidine with propofol for magnetic resonance imaging sleep studies in children. Anesth. Analg. 2009, 109, 745–753. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, M.; Jung, D.; Salisbury, S.; McAuliffe, J.; Gunter, J.; Patio, M.; Donnelly, L.F.; Fleck, R. Effect of increasing depth of dexmedetomidine and propofol anesthesia on upper airway morphology in children and adolescents with obstructive sleep apnea. J. Clin. Anesth. 2013, 25, 529–541. [Google Scholar] [CrossRef] [PubMed]
- Padiyara, T.V.; Bansal, S.; Jain, D.; Arora, S.; Gandhi, K. Dexmedetomidine versus propofol at different sedation depths during drug-induced sleep endoscopy: A randomized trial. Laryngoscope 2020, 130, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Chang, E.T.; Certal, V.; Song, S.A.; Zaghi, S.; Carrasco-Llatas, M.; Torre, C.; Capasso, R.; Camacho, M. Dexmedetomidine versus propofol during drug-induced sleep endoscopy and sedation: A systematic review. Sleep Breath. Schlaf Atm. 2017, 21, 727–735. [Google Scholar] [CrossRef]
- Di, M.; Han, Y.; Yang, Z.; Liu, H.; Ye, X.; Lai, H.; Li, J.; ShangGuan, W.; Lian, Q. Tracheal extubation in deeply anesthetized pediatric patients after tonsillectomy: A comparison of high-concentration sevoflurane alone and low-concentration sevoflurane in combination with dexmedetomidine pre-medication. BMC Anesthesiol. 2017, 17, 28. [Google Scholar] [CrossRef]
- Fan, Q.; Hu, C.; Ye, M.; Shen, X. Dexmedetomidine for tracheal extubation in deeply anesthetized adult patients after otologic surgery: A comparison with remifentanil. BMC Anesthesiol. 2015, 15, 106. [Google Scholar] [CrossRef]
- Yao, Y.; Qian, B.; Lin, Y.; Wu, W.; Ye, H.; Chen, Y. Intranasal dexmedetomidine premedication reduces minimum alveolar concentration of sevoflurane for laryngeal mask airway insertion and emergence delirium in children: A prospective, randomized, double-blind, placebo-controlled trial. Paediatr. Anaesth. 2015, 25, 492–498. [Google Scholar] [CrossRef]
- Wei, L.; Deng, X.; Sui, J.; Wang, L.; Liu, J. Dexmedetomidine Improves Intubating Conditions Without Muscle Relaxants in Children After Induction With Propofol and Remifentanil. Anesth. Analg. 2015, 121, 785–790. [Google Scholar] [CrossRef]
- He, L.; Wang, X.; Zheng, S. Effects of dexmedetomidine on sevoflurane requirement for 50% excellent tracheal intubation in children: A randomized, double-blind comparison. Paediatr. Anaesth. 2014, 24, 987–993. [Google Scholar] [CrossRef]
- Chen, K.Z.; Ye, M.; Hu, C.B.; Shen, X. Dexmedetomidine vs remifentanil intravenous anaesthesia and spontaneous ventilation for airway foreign body removal in children. Br. J. Anaesth. 2014, 112, 892–897. [Google Scholar] [CrossRef]
- Li, S.; Liu, H.; Zhang, J.; Liu, Y.; Yu, Q.; Sun, M.; Tian, Q.; Yang, F.; Lei, Y.; Liu, X.; et al. The 95% effective dose of intranasal dexmedetomidine sedation for pulmonary function testing in children aged 1–3 years: A biased coin design up-and-down sequential method. J. Clin. Anesth. 2020, 63, 109746. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Bao, H.; Si, Y.; Wang, X. Effects of dexmedetomidine on early and late cytokines during polymicrobial sepsis in mice. Inflamm. Res. 2013, 62, 507–514. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Miao, L.; Yao, Y.; Wu, W.; Wu, X.; Gong, C.; Qiu, L.; Chen, J. Dexmedetomidine Ameliorate CLP-Induced Rat Intestinal Injury via Inhibition of Inflammation. Mediat. Inflamm. 2015, 2015, 918361. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, J.A.; Bissell, B.D. Misdirected Sympathy: The Role of Sympatholysis in Sepsis and Septic Shock. J. Intensive Care Med. 2018, 33, 74–86. [Google Scholar] [CrossRef]
- Li, Y.; Wang, B.; Zhang, L.L.; He, S.F.; Hu, X.W.; Wong, G.T.; Zhang, Y. Dexmedetomidine Combined with General Anesthesia Provides Similar Intraoperative Stress Response Reduction When Compared with a Combined General and Epidural Anesthetic Technique. Anesth. Analg. 2016, 122, 1202–1210. [Google Scholar] [CrossRef]
- Wang, K.; Wu, M.; Xu, J.; Wu, C.; Zhang, B.; Wang, G.; Ma, D. Effects of dexmedetomidine on perioperative stress, inflammation, and immune function: Systematic review and meta-analysis. Br. J. Anaesth. 2019, 123, 777–794. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, S.; Fan, X.; Yuan, F.; Dai, J.; Hu, J. Dexmedetomidine Preconditioning Ameliorates Inflammation and Blood-Spinal Cord Barrier Damage After Spinal Cord Ischemia-Reperfusion Injury by Down-Regulation High Mobility Group Box 1-Toll-Like Receptor 4-Nuclear Factor kappaB Signaling Pathway. Spine 2019, 44, E74–E81. [Google Scholar] [CrossRef]
- Sun, Z.; Zhao, T.; Lv, S.; Gao, Y.; Masters, J.; Weng, H. Dexmedetomidine attenuates spinal cord ischemia-reperfusion injury through both anti-inflammation and anti-apoptosis mechanisms in rabbits. J. Transl. Med. 2018, 16, 209. [Google Scholar] [CrossRef]
- Huang, Y.; Lu, Y.; Zhang, L.; Yan, J.; Jiang, J.; Jiang, H. Perineural dexmedetomidine attenuates inflammation in rat sciatic nerve via the NF-kappaB pathway. Int. J. Mol. Sci. 2014, 15, 4049–4059. [Google Scholar] [CrossRef]
- Mason, K.P.; O’Mahony, E.; Zurakowski, D.; Libenson, M.H. Effects of dexmedetomidine sedation on the EEG in children. Paediatr. Anaesth. 2009, 19, 1175–1183. [Google Scholar] [CrossRef]
- Wu, X.H.; Cui, F.; Zhang, C.; Meng, Z.T.; Wang, D.X.; Ma, J.; Wang, G.F.; Zhu, S.N.; Ma, D. Low-dose Dexmedetomidine Improves Sleep Quality Pattern in Elderly Patients after Noncardiac Surgery in the Intensive Care Unit: A Pilot Randomized Controlled Trial. Anesthesiology 2016, 125, 979–991. [Google Scholar] [CrossRef] [PubMed]
- Hashmi, J.A.; Loggia, M.L.; Khan, S.; Gao, L.; Kim, J.; Napadow, V.; Brown, E.N.; Akeju, O. Dexmedetomidine Disrupts the Local and Global Efficiencies of Large-scale Brain Networks. Anesthesiology 2017, 126, 419–430. [Google Scholar] [CrossRef] [PubMed]
- Kallionpaa, R.E.; Scheinin, A.; Kallionpaa, R.A.; Sandman, N.; Kallioinen, M.; Laitio, R.; Laitio, T.; Kaskinoro, K.; Kuusela, T.; Revonsuo, A.; et al. Spoken words are processed during dexmedetomidine-induced unresponsiveness. Br. J. Anaesth. 2018, 121, 270–280. [Google Scholar] [CrossRef] [PubMed]
- Berkenbosch, J.W.; Tobias, J.D. Development of bradycardia during sedation with dexmedetomidine in an infant concurrently receiving digoxin. Pediatric Crit. Care Med. 2003, 4, 203–205. [Google Scholar] [CrossRef]
- Fritock, M.D.; Ing, R.J.; Twite, M.D. Cardiac Arrest in 2 Neonates Receiving Amiodarone and Dexmedetomidine. J. Cardiothorac. Vasc. Anesth. 2017, 31, 2135–2138. [Google Scholar] [CrossRef]
- Ohmori, T.; Shiota, N.; Haramo, A.; Masuda, T.; Maruyama, F.; Wakabayashi, K.; Adachi, Y.U.; Nakazawa, K. Post-operative cardiac arrest induced by co-administration of amiodarone and dexmedetomidine: A case report. J. Intensive Care 2015, 3, 43. [Google Scholar] [CrossRef]
- Mason, K.P.; Zgleszewski, S.; Forman, R.E.; Stark, C.; DiNardo, J.A. An exaggerated hypertensive response to glycopyrrolate therapy for bradycardia associated with high-dose dexmedetomidine. Anesth. Analg. 2009, 108, 906–908. [Google Scholar] [CrossRef]
- Subramanyam, R.; Cudilo, E.M.; Hossain, M.M.; McAuliffe, J.; Wu, J.; Patino, M.; Gunter, J.; Mahmoud, M. To Pretreat or Not to Pretreat: Prophylactic Anticholinergic Administration Before Dexmedetomidine in Pediatric Imaging. Anesth. Analg. 2015, 121, 479–485. [Google Scholar] [CrossRef]
- Kang, D.; Lim, C.; Shim, D.J.; Kim, H.; Kim, J.W.; Chung, H.J.; Shin, Y.; Kim, J.D.; Ryu, S.J. The correlation of heart rate between natural sleep and dexmedetomidine sedation. Korean J. Anesthesiol. 2019, 72, 164–168. [Google Scholar] [CrossRef]
- Slupe, A.M.; Minnier, J.; Raitt, M.H.; Zarraga, I.G.E.; MacMurdy, K.S.; Jessel, P.M. Dexmedetomidine Sedation for Paroxysmal Supraventricular Tachycardia Ablation Is Not Associated with Alteration of Arrhythmia Inducibility. Anesth. Analg. 2019, 129, 1529–1535. [Google Scholar] [CrossRef]
- Hultin, M.; Sundberg, E. Spontaneous Conversions of Supraventricular Tachycardia to Sinus Rhythm in Children After Premedication with Intranasal Dexmedetomidine: A Case Report. A A Pract. 2018, 11, 219–220. [Google Scholar] [CrossRef] [PubMed]
- Botros, J.M.; Mahmoud, A.M.S.; Ragab, S.G.; Ahmed, M.A.A.; Roushdy, H.M.S.; Yassin, H.M.; Bolus, M.L.; Goda, A.S. Comparative study between Dexmedetomidine and Ondansteron for prevention of post spinal shivering. A randomized controlled trial. BMC Anesthesiol. 2018, 18, 179. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.X.; Xu, F.Y.; Liang, X.; Zhou, M.; Wu, L.; Wu, J.R.; Xia, J.H.; Zou, Z. Efficacy of dexmedetomidine on postoperative shivering: A meta-analysis of clinical trials. Can. J. Anaesth. J. Can. D’anesthésie 2015, 62, 816–829. [Google Scholar] [CrossRef] [PubMed]
- Kruger, B.D.; Kurmann, J.; Corti, N.; Spahn, D.R.; Bettex, D.; Rudiger, A. Dexmedetomidine-Associated Hyperthermia: A Series of 9 Cases and a Review of the Literature. Anesth. Analg. 2017, 125, 1898–1906. [Google Scholar] [CrossRef]
- Pan, W.; Lin, L.; Zhang, N.; Yuan, F.; Hua, X.; Wang, Y.; Mo, L. Neuroprotective Effects of Dexmedetomidine Against Hypoxia-Induced Nervous System Injury are Related to Inhibition of NF-kappaB/COX-2 Pathways. Cell. Mol. Neurobiol. 2016, 36, 1179–1188. [Google Scholar] [CrossRef]
- Perez-Zoghbi, J.F.; Zhu, W.; Grafe, M.R.; Brambrink, A.M. Dexmedetomidine-mediated neuroprotection against sevoflurane-induced neurotoxicity extends to several brain regions in neonatal rats. Br. J. Anaesth. 2017, 119, 506–516. [Google Scholar] [CrossRef]
- Shan, Y.; Yang, F.; Tang, Z.; Bi, C.; Sun, S.; Zhang, Y.; Liu, H. Dexmedetomidine Ameliorates the Neurotoxicity of Sevoflurane on the Immature Brain Through the BMP/SMAD Signaling Pathway. Front. Neurosci. 2018, 12, 964. [Google Scholar] [CrossRef]
- Wang, X.; Shan, Y.; Tang, Z.; Gao, L.; Liu, H. Neuroprotective effects of dexmedetomidine against isoflurane-induced neuronal injury via glutamate regulation in neonatal rats. Drug Des. Dev. Ther. 2019, 13, 153–160. [Google Scholar] [CrossRef]
- Hu, J.; Vacas, S.; Feng, X.; Lutrin, D.; Uchida, Y.; Lai, I.K.; Maze, M. Dexmedetomidine Prevents Cognitive Decline by Enhancing Resolution of High Mobility Group Box 1 Protein-induced Inflammation through a Vagomimetic Action in Mice. Anesthesiology 2018, 128, 921–931. [Google Scholar] [CrossRef]
- Xu, H.; Zhao, B.; She, Y.; Song, X. Dexmedetomidine ameliorates lidocaine-induced spinal neurotoxicity via inhibiting glutamate release and the PKC pathway. Neurotoxicology 2018, 69, 77–83. [Google Scholar] [CrossRef]
- Wu, J.; Vogel, T.; Gao, X.; Lin, B.; Kulwin, C.; Chen, J. Neuroprotective effect of dexmedetomidine in a murine model of traumatic brain injury. Sci. Rep. 2018, 8, 4935. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.P.; Zhao, J.J.; Wang, W.X.; Liu, Y.; Wu, H.F.; Chen, C.; Yu, L.; Gui, J.B. Dexmedetomidine increases acetylation level of histone through ERK1/2 pathway in dopamine neuron. Hum. Exp. Toxicol. 2017, 36, 474–482. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Han, R.; Zuo, Z. Dexmedetomidine post-treatment induces neuroprotection via activation of extracellular signal-regulated kinase in rats with subarachnoid haemorrhage. Br. J. Anaesth. 2016, 116, 384–392. [Google Scholar] [CrossRef] [PubMed]
- Bell, M.T.; Puskas, F.; Bennett, D.T.; Herson, P.S.; Quillinan, N.; Fullerton, D.A.; Reece, T.B. Dexmedetomidine, an alpha-2a adrenergic agonist, promotes ischemic tolerance in a murine model of spinal cord ischemia-reperfusion. J. Thorac. Cardiovasc. Surg. 2014, 147, 500–506. [Google Scholar] [CrossRef]
- Huang, J.; Jiang, Q. Dexmedetomidine Protects Against Neurological Dysfunction in a Mouse Intracerebral Hemorrhage Model by Inhibiting Mitochondrial Dysfunction-Derived Oxidative Stress. J. Stroke Cerebrovasc. Dis. 2019, 28, 1281–1289. [Google Scholar] [CrossRef]
- Cai, Y.; Xu, H.; Yan, J.; Zhang, L.; Lu, Y. Molecular targets and mechanism of action of dexmedetomidine in treatment of ischemia/reperfusion injury. Mol. Med. Rep. 2014, 9, 1542–1550. [Google Scholar] [CrossRef]
- Sabir, H.; Bishop, S.; Cohen, N.; Maes, E.; Liu, X.; Dingley, J.; Thoresen, M. Neither xenon nor fentanyl induces neuroapoptosis in the newborn pig brain. Anesthesiology 2013, 119, 345–357. [Google Scholar] [CrossRef]
- McCann, M.E.; Soriano, S.G. Does general anesthesia affect neurodevelopment in infants and children? BMJ 2019, 367, l6459. [Google Scholar] [CrossRef]
- Andropoulos, D.B. Effect of Anesthesia on the Developing Brain: Infant and Fetus. Fetal Diagn. 2018, 43, 1–11. [Google Scholar] [CrossRef]
- Andropoulos, D.B.; Greene, M.F. Anesthesia and Developing Brains—Implications of the FDA Warning. N. Engl. J. Med. 2017, 376, 905–907. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration. FDA Drug Safety Communication: FDA Approves Label Changes for Use of General Anesthetic and Sedation Drugs in Young Children. Available online: https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-fda-approves-label-changes-use-general-anesthetic-and-sedation-drugs (accessed on 12 June 2020).
- Cho, J.S.; Shim, J.K.; Soh, S.; Kim, M.K.; Kwak, Y.L. Perioperative dexmedetomidine reduces the incidence and severity of acute kidney injury following valvular heart surgery. Kidney Int. 2016, 89, 693–700. [Google Scholar] [CrossRef]
- Jo, Y.Y.; Kim, J.Y.; Lee, J.Y.; Choi, C.H.; Chang, Y.J.; Kwak, H.J. The effect of intraoperative dexmedetomidine on acute kidney injury after pediatric congenital heart surgery: A prospective randomized trial. Medicine 2017, 96, e7480. [Google Scholar] [CrossRef]
- Kwiatkowski, D.M.; Axelrod, D.M.; Sutherland, S.M.; Tesoro, T.M.; Krawczeski, C.D. Dexmedetomidine Is Associated With Lower Incidence of Acute Kidney Injury After Congenital Heart Surgery. Pediatr. Crit. Care Med. 2016, 17, 128–134. [Google Scholar] [CrossRef] [PubMed]
- Shi, R.; Tie, H.T. Dexmedetomidine as a promising prevention strategy for cardiac surgery-associated acute kidney injury: A meta-analysis. Crit. Care (Lond. Engl.) 2017, 21, 198. [Google Scholar] [CrossRef] [PubMed]
- Bayram, A.; Ulgey, A.; Baykan, A.; Narin, N.; Narin, F.; Esmaoglu, A.; Boyaci, A. The effects of dexmedetomidine on early stage renal functions in pediatric patients undergoing cardiac angiography using non-ionic contrast media: A double-blind, randomized clinical trial. Paediatr. Anaesth. 2014, 24, 426–432. [Google Scholar] [CrossRef] [PubMed]
- Ji, F.; Li, Z.; Young, J.N.; Yeranossian, A.; Liu, H. Post-bypass dexmedetomidine use and postoperative acute kidney injury in patients undergoing cardiac surgery with cardiopulmonary bypass. PLoS ONE 2013, 8, e77446. [Google Scholar] [CrossRef]
- Ammar, A.S.; Mahmoud, K.M.; Kasemy, Z.A.; Helwa, M.A. Cardiac and renal protective effects of dexmedetomidine in cardiac surgeries: A randomized controlled trial. Saudi J. Anaesth. 2016, 10, 395–401. [Google Scholar] [CrossRef]
- Gao, J.M.; Meng, X.W.; Zhang, J.; Chen, W.R.; Xia, F.; Peng, K.; Ji, F.H. Dexmedetomidine Protects Cardiomyocytes against Hypoxia/Reoxygenation Injury by Suppressing TLR4-MyD88-NF-kappaB Signaling. BioMed Res. Int. 2017, 2017, 1674613. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.J.; Peng, K.; Zhang, J.; Meng, X.W.; Ji, F.H. Dexmedetomidine preconditioning may attenuate myocardial ischemia/reperfusion injury by down-regulating the HMGB1-TLR4-MyD88-NF-small ka, CyrillicB signaling pathway. PLoS ONE 2017, 12, e0172006. [Google Scholar] [CrossRef]
- Zhang, J.; Xia, F.; Zhao, H.; Peng, K.; Liu, H.; Meng, X.; Chen, C.; Ji, F. Dexmedetomidine-induced cardioprotection is mediated by inhibition of high mobility group box-1 and the cholinergic anti-inflammatory pathway in myocardial ischemia-reperfusion injury. PLoS ONE 2019, 14, e0218726. [Google Scholar] [CrossRef] [PubMed]
- Gong, Z.; Ma, L.; Zhong, Y.L.; Li, J.; Lv, J.; Xie, Y.B. Myocardial protective effects of dexmedetomidine in patients undergoing cardiac surgery: A meta-analysis and systematic review. Exp. Ther. Med. 2017, 13, 2355–2361. [Google Scholar] [CrossRef]
- Ríha, H.; Kotulák, T.; Březina, A.; Hess, L.; Kramář, P.; Szárszoi, O.; Netuka, I.; Pirk, J. Comparison of the effects of ketamine-dexmedetomidine and sevoflurane-sufentanil anesthesia on cardiac biomarkers after cardiac surgery: An observational study. Physiol. Res. 2012, 61, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Uusalo, P.; Al-Ramahi, D.; Tilli, I.; Aantaa, R.A.; Scheinin, M.; Saari, T.I. Subcutaneously administered dexmedetomidine is efficiently absorbed and is associated with attenuated cardiovascular effects in healthy volunteers. Eur. J. Clin. Pharmacol. 2018, 74, 1047–1054. [Google Scholar] [CrossRef] [PubMed]
- Jun, J.H.; Kim, K.N.; Kim, J.Y.; Song, S.M. The effects of intranasal dexmedetomidine premedication in children: A systematic review and meta-analysis. Can. J. Anaesth. J. Can. D’anesthésie 2017, 64, 947–961. [Google Scholar] [CrossRef] [PubMed]
- Tug, A.; Hanci, A.; Turk, H.S.; Aybey, F.; Isil, C.T.; Sayin, P.; Oba, S. Comparison of Two Different Intranasal Doses of Dexmedetomidine in Children for Magnetic Resonance Imaging Sedation. Paediatr. Drugs 2015, 17, 479–485. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.E.; Hui, T.; Wong, S.; Irwin, M.G.; Yuen, V.; Wong, G.L.S. Abstract PR251: A Comparison of Two Doses of Intranasal Dexmedetomidine for Sedative Premedication in Children. Anesth. Analg. 2016, 123, 327. [Google Scholar] [CrossRef]
- Yang, F.; Liu, Y.; Yu, Q.; Li, S.; Zhang, J.; Sun, M.; Liu, L.; Lei, Y.; Tian, Q.; Liu, H.; et al. Analysis of 17 948 pediatric patients undergoing procedural sedation with a combination of intranasal dexmedetomidine and ketamine. Paediatr. Anaesth. 2019, 29, 85–91. [Google Scholar] [CrossRef]
- Miller, J.W.; Divanovic, A.A.; Hossain, M.M.; Mahmoud, M.A.; Loepke, A.W. Dosing and efficacy of intranasal dexmedetomidine sedation for pediatric transthoracic echocardiography: A retrospective study. Can. J. Anaesth. J. Can. D’anesthésie 2016, 63, 834–841. [Google Scholar] [CrossRef]
- Tenney, J.R.; Miller, J.W.; Rose, D.F. Intranasal Dexmedetomidine for Sedation During Magnetoencephalography. J. Clin. Neurophysiol. 2019, 36, 371–374. [Google Scholar] [CrossRef]
- Behrle, N.; Birisci, E.; Anderson, J.; Schroeder, S.; Dalabih, A. Intranasal Dexmedetomidine as a Sedative for Pediatric Procedural Sedation. J. Pediatr. Pharmacol. Ther. 2017, 22, 4–8. [Google Scholar] [CrossRef]
- Li, L.; Zhou, J.; Yu, D.; Hao, X.; Xie, Y.; Zhu, T. Intranasal dexmedetomidine versus oral chloral hydrate for diagnostic procedures sedation in infants and toddlers: A systematic review and meta-analysis. Medicine 2020, 99, e19001. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Wang, Z.; Song, X.; Fan, Y.; Tian, H.; Li, B. Comparison of rescue techniques for failed chloral hydrate sedation for magnetic resonance imaging scans—Additional chloral hydrate vs. intranasal dexmedetomidine. Paediatr. Anaesth. 2016, 26, 273–279. [Google Scholar] [CrossRef] [PubMed]
- Ghai, B.; Jain, K.; Saxena, A.K.; Bhatia, N.; Sodhi, K.S. Comparison of oral midazolam with intranasal dexmedetomidine premedication for children undergoing CT imaging: A randomized, double-blind, and controlled study. Paediatr. Anaesth. 2017, 27, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, A.; Das, A.; Basunia, S.R.; Chattopadhyay, S.; Kundu, R.; Bhattacharyya, R. Emergence agitation prevention in paediatric ambulatory surgery: A comparison between intranasal Dexmedetomidine and Clonidine. J. Res. Pharm. Pract. 2015, 4, 24–30. [Google Scholar] [CrossRef]
- Cozzi, G.; Monasta, L.; Maximova, N.; Poropat, F.; Magnolato, A.; Sbisa, E.; Norbedo, S.; Sternissa, G.; Zanon, D.; Barbi, E. Combination of intranasal dexmedetomidine and oral midazolam as sedation for pediatric MRI. Paediatr. Anaesth. 2017, 27, 976–977. [Google Scholar] [CrossRef]
- Bua, J.; Massaro, M.; Cossovel, F.; Monasta, L.; Brovedani, P.; Cozzi, G.; Barbi, E.; Demarini, S.; Travan, L. Intranasal dexmedetomidine, as midazolam-sparing drug, for MRI in preterm neonates. Paediatr. Anaesth. 2018, 28, 747–748. [Google Scholar] [CrossRef]
- Patel, V.; Singh, N.; Saksena, A.K.; Singh, S.; Sonkar, S.K.; Jolly, S.M. A comparative assessment of intranasal and oral dexmedetomidine for procedural sedation in pediatric dental patients. J. Indian Soc. Pedod. Prev. Dent. 2018, 36, 370–375. [Google Scholar] [CrossRef]
- Boriosi, J.P.; Eickhoff, J.C.; Hollman, G.A. Safety and Efficacy of Buccal Dexmedetomidine for MRI Sedation in School-Aged Children. Hosp. Pediatrics 2019, 9, 348–354. [Google Scholar] [CrossRef]
- Xu, D.; Xiu, M.; Zhang, X.; Zhu, P.; Tian, L.; Feng, J.; Wu, Y.; Zhao, Z.; Luan, H. Effect of dexmedetomidine added to ropivicaine for caudal anesthesia in patients undergoing hemorrhoidectomy: A prospective randomized controlled trial. Medicine 2018, 97, e11731. [Google Scholar] [CrossRef]
- Tu, Z.; Tan, X.; Li, S.; Cui, J. The Efficacy and Safety of Dexmedetomidine Combined with Bupivacaine on Caudal Epidural Block in Children: A Meta-Analysis. Med. Sci. Monit. 2019, 25, 165–173. [Google Scholar] [CrossRef]
- Lundblad, M.; Marhofer, D.; Eksborg, S.; Lonnqvist, P.A. Dexmedetomidine as adjunct to ilioinguinal/iliohypogastric nerve blocks for pediatric inguinal hernia repair: An exploratory randomized controlled trial. Paediatr. Anaesth. 2015, 25, 897–905. [Google Scholar] [CrossRef]
- Abdulatif, M.; Fawzy, M.; Nassar, H.; Hasanin, A.; Ollaek, M.; Mohamed, H. The effects of perineural dexmedetomidine on the pharmacodynamic profile of femoral nerve block: A dose-finding randomised, controlled, double-blind study. Anaesthesia 2016, 71, 1177–1185. [Google Scholar] [CrossRef] [PubMed]
- Andersen, J.H.; Grevstad, U.; Siegel, H.; Dahl, J.B.; Mathiesen, O.; Jaeger, P. Does Dexmedetomidine Have a Perineural Mechanism of Action When Used as an Adjuvant to Ropivacaine?: A Paired, Blinded, Randomized Trial in Healthy Volunteers. Anesthesiology 2017, 126, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Vorobeichik, L.; Brull, R.; Abdallah, F.W. Evidence basis for using perineural dexmedetomidine to enhance the quality of brachial plexus nerve blocks: A systematic review and meta-analysis of randomized controlled trials. Br. J. Anaesth. 2017, 118, 167–181. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.S.; Seo, K.H.; Kang, J.H.; Jeong, J.Y.; Kim, Y.S.; Han, N.R. Optimal dose of perineural dexmedetomidine for interscalene brachial plexus block to control postoperative pain in patients undergoing arthroscopic shoulder surgery: A prospective, double-blind, randomized controlled study. Medicine 2018, 97, e0440. [Google Scholar] [CrossRef] [PubMed]
- Bharti, N.; Sardana, D.K.; Bala, I. The Analgesic Efficacy of Dexmedetomidine as an Adjunct to Local Anesthetics in Supraclavicular Brachial Plexus Block: A Randomized Controlled Trial. Anesth. Analg. 2015, 121, 1655–1660. [Google Scholar] [CrossRef] [PubMed]
- Dutta, A.; Sethi, N.; Sood, J.; Panday, B.C.; Gupta, M.; Choudhary, P.; Puri, G.D. The Effect of Dexmedetomidine on Propofol Requirements During Anesthesia Administered by Bispectral Index-Guided Closed-Loop Anesthesia Delivery System: A Randomized Controlled Study. Anesth. Analg. 2019, 129, 84–91. [Google Scholar] [CrossRef]
- Wu, X.; Hang, L.H.; Wang, H.; Shao, D.H.; Xu, Y.G.; Cui, W.; Chen, Z. Intranasally Administered Adjunctive Dexmedetomidine Reduces Perioperative Anesthetic Requirements in General Anesthesia. Yonsei Med. J. 2016, 57, 998–1005. [Google Scholar] [CrossRef]
- Nagoshi, M.; Reddy, S.; Bell, M.; Cresencia, A.; Margolis, R.; Wetzel, R.; Ross, P. Low-dose dexmedetomidine as an adjuvant to propofol infusion for children in MRI: A double-cohort study. Paediatr. Anaesth. 2018, 28, 639–646. [Google Scholar] [CrossRef]
- Di, M.; Yang, Z.; Qi, D.; Lai, H.; Wu, J.; Liu, H.; Ye, X.; ShangGuan, W.; Lian, Q.; Li, J. Intravenous dexmedetomidine pre-medication reduces the required minimum alveolar concentration of sevoflurane for smooth tracheal extubation in anesthetized children: A randomized clinical trial. BMC Anesthesiol. 2018, 18, 9. [Google Scholar] [CrossRef]
- Zhang, X.; Wu, J.; Wang, L.; Li, W. Dexmedetomidine facilitates extubation in children who require intubation and respiratory support after airway foreign body retrieval: A case-cohort analysis of 57 cases. J. Anesth. 2018, 32, 592–598. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Zhong, H.; Xie, X.; Sha, W.; Li, C.; Li, Z.; Huang, Z.; Chen, C. Effect of intratracheal dexmedetomidine administration on recovery from general anaesthesia after gynaecological laparoscopic surgery: A randomised double-blinded study. BMJ Open 2018, 8, e020614. [Google Scholar] [CrossRef] [PubMed]
- Tsiotou, A.G.; Malisiova, A.; Kouptsova, E.; Mavri, M.; Anagnostopoulou, M.; Kalliardou, E. Dexmedetomidine for the reduction of emergence delirium in children undergoing tonsillectomy with propofol anesthesia: A double-blind, randomized study. Paediatr. Anaesth. 2018, 28, 632–638. [Google Scholar] [CrossRef]
- Kim, H.S.; Byon, H.J.; Kim, J.E.; Park, Y.H.; Lee, J.H.; Kim, J.T. Appropriate dose of dexmedetomidine for the prevention of emergence agitation after desflurane anesthesia for tonsillectomy or adenoidectomy in children: Up and down sequential allocation. BMC Anesthesiol. 2015, 15, 79. [Google Scholar] [CrossRef] [PubMed]
- Hauber, J.A.; Davis, P.J.; Bendel, L.P.; Martyn, S.V.; McCarthy, D.L.; Evans, M.C.; Cladis, F.P.; Cunningham, S.; Lang, R.S.; Campbell, N.F.; et al. Dexmedetomidine as a Rapid Bolus for Treatment and Prophylactic Prevention of Emergence Agitation in Anesthetized Children. Anesth. Analg. 2015, 121, 1308–1315. [Google Scholar] [CrossRef]
- Keles, S.; Kocaturk, O. The Effect of Oral Dexmedetomidine Premedication on Preoperative Cooperation and Emergence Delirium in Children Undergoing Dental Procedures. BioMed Res. Int. 2017, 2017, 6742183. [Google Scholar] [CrossRef]
- Trivedi, S.; Kumar, R.; Tripathi, A.K.; Mehta, R.K. A Comparative Study of Dexmedetomidine and Midazolam in Reducing Delirium Caused by Ketamine. J. Clin. Diagn. Res. 2016, 10, Uc01–Uc04. [Google Scholar] [CrossRef]
- Song, Y.; Shim, J.K.; Song, J.W.; Kim, E.K.; Kwak, Y.L. Dexmedetomidine added to an opioid-based analgesic regimen for the prevention of postoperative nausea and vomiting in highly susceptible patients: A randomised controlled trial. Eur. J. Anaesthesiol. 2016, 33, 75–83. [Google Scholar] [CrossRef]
- Chrysostomou, C.; Schulman, S.R.; Herrera Castellanos, M.; Cofer, B.E.; Mitra, S.; da Rocha, M.G.; Wisemandle, W.A.; Gramlich, L. A phase II/III, multicenter, safety, efficacy, and pharmacokinetic study of dexmedetomidine in preterm and term neonates. J. Pediatrics 2014, 164, 276–282.e1-3. [Google Scholar] [CrossRef]
- Franciscovich, C.D.; Monk, H.M.; Brodecki, D.; Rogers, R.; Rintoul, N.E.; Hedrick, H.L.; Ely, E. Sedation Practices of Neonates Receiving Extracorporeal Membrane Oxygenation. ASAIO J. 2020, 66, 559–564. [Google Scholar] [CrossRef]
- Sperotto, F.; Mondardini, M.C.; Vitale, F.; Daverio, M.; Campagnano, E.; Ferrero, F.; Rossetti, E.; Vasile, B.; Dusio, M.P.; Ferrario, S.; et al. Prolonged sedation in critically ill children: Is dexmedetomidine a safe option for younger age? An off-label experience. Minerva Anestesiol. 2019, 85, 164–172. [Google Scholar] [CrossRef] [PubMed]
- Dersch-Mills, D.A.; Banasch, H.L.; Yusuf, K.; Howlett, A. Dexmedetomidine Use in a Tertiary Care NICU: A Descriptive Study. Ann. Pharmacother. 2019, 53, 464–470. [Google Scholar] [CrossRef] [PubMed]
- Puthoff, T.D.; Shah, H.; Slaughter, J.L.; Bapat, R. Reduction of Analgesia Duration after Tracheostomy during Neonatal Intensive Care: A Quality Initiative. Pediatr. Qual. Saf. 2018, 3, e106. [Google Scholar] [CrossRef] [PubMed]
- Weatherall, M.; Aantaa, R.; Conti, G.; Garratt, C.; Pohjanjousi, P.; Lewis, M.A.; Moore, N.; Perez-Gutthann, S. A multinational, drug utilization study to investigate the use of dexmedetomidine (Dexdor(R)) in clinical practice in the EU. Br. J. Clin. Pharmacol. 2017, 83, 2066–2076. [Google Scholar] [CrossRef]
- Grant, M.J.; Schneider, J.B.; Asaro, L.A.; Dodson, B.L.; Hall, B.A.; Simone, S.L.; Cowl, A.S.; Munkwitz, M.M.; Wypij, D.; Curley, M.A. Dexmedetomidine Use in Critically Ill Children With Acute Respiratory Failure. Pediatr. Crit. Care Med. 2016, 17, 1131–1141. [Google Scholar] [CrossRef]
- Piastra, M.; Pizza, A.; Gaddi, S.; Luca, E.; Genovese, O.; Picconi, E.; De Luca, D.; Conti, G. Dexmedetomidine is effective and safe during NIV in infants and young children with acute respiratory failure. BMC Pediatr. 2018, 18, 282. [Google Scholar] [CrossRef]
- Ghimire, L.V.; Chou, F.S. Efficacy of prophylactic dexmedetomidine in preventing postoperative junctional ectopic tachycardia in pediatric cardiac surgery patients: A systematic review and meta-analysis. Paediatr. Anaesth. 2018, 28, 597–606. [Google Scholar] [CrossRef]
- Flores-Gonzalez, J.C.; Estalella-Mendoza, A.; Lechuga-Sancho, A.M.; Hernandez-Gonzalez, A.; Rubio-Quinones, F.; Rodriguez-Campoy, P.; Saldana-Valderas, M. Supraventricular tachycardia after withdrawal of prolonged dexmedetomidine infusion in a paediatric patient without heart disease. J. Clin. Pharm. Ther. 2017, 42, 653–655. [Google Scholar] [CrossRef]
- Burns, J.; Jackson, K.; Sheehy, K.A.; Finkel, J.C.; Quezado, Z.M. The Use of Dexmedetomidine in Pediatric Palliative Care: A Preliminary Study. J. Palliat. Med. 2017, 20, 779–783. [Google Scholar] [CrossRef]
- De Zen, L.; Marchetti, F.; Barbi, E.; Benini, F. Off-label drugs use in pediatric palliative care. Ital. J. Pediatr. 2018, 44, 144. [Google Scholar] [CrossRef]
- De Zen, L.D.R., I; Robazza, M.; Barbieri, F.; Campagna, M.; Vaccher, S.; Barbi, E.; Dall′Amico, R. Home intranasal dexmedetomidine in a child with an intractable sleep disorder. J. Pediatr. Pharmacol. Ther. 2020, 25, 332–335. [Google Scholar] [CrossRef] [PubMed]
- De Zen, L.; Della Paolera, S.; Del Rizzo, I.; Taucar, V.; Skabar, A.; Barbi, E. Home Intranasal Dexmedetomidine for Refractory Dystonia in Pediatric Palliative Care. J. Pain Symptom Manag. 2020. [Google Scholar] [CrossRef] [PubMed]
- Li, B.L.; Yuen, V.M.; Zhang, N.; Zhang, H.H.; Huang, J.X.; Yang, S.Y.; Miller, J.W.; Song, X.R. A Comparison of Intranasal Dexmedetomidine and Dexmedetomidine Plus Buccal Midazolam for Non-painful Procedural Sedation in Children with Autism. J. Autism Dev. Disord. 2019, 49, 3798–3806. [Google Scholar] [CrossRef] [PubMed]
- Carlone, G.; Trombetta, A.; Amoroso, S.; Poropat, F.; Barbi, E.; Cozzi, G. Intramuscular Dexmedetomidine, a Feasible Option for Children With Autism Spectrum Disorders Needing Urgent Procedural Sedation. Pediatr. Emerg. Care 2019. [Google Scholar] [CrossRef]
- Lubisch, N.; Roskos, R.; Berkenbosch, J.W. Dexmedetomidine for procedural sedation in children with autism and other behavior disorders. Pediatr. Neurol. 2009, 41, 88–94. [Google Scholar] [CrossRef]
- Abulebda, K.; Louer, R.; Lutfi, R.; Ahmed, S.S. A Comparison of Safety and Efficacy of Dexmedetomidine and Propofol in Children with Autism and Autism Spectrum Disorders Undergoing Magnetic Resonance Imaging. J. Autism Dev. Disord. 2018, 48, 3127–3132. [Google Scholar] [CrossRef]
- Stuker, E.W.; Eskander, J.P.; Gennuso, S.A. Third time’s a charm: Oral midazolam vs intranasal dexmedetomidine for preoperative anxiolysis in an autistic pediatric patient. Paediatr. Anaesth. 2018, 28, 370–371. [Google Scholar] [CrossRef]
- Mason, K.P.; Lubisch, N.; Robinson, F.; Roskos, R.; Epstein, M.A. Intramuscular dexmedetomidine: An effective route of sedation preserves background activity for pediatric electroencephalograms. J. Pediatr. 2012, 161, 927–932. [Google Scholar] [CrossRef]
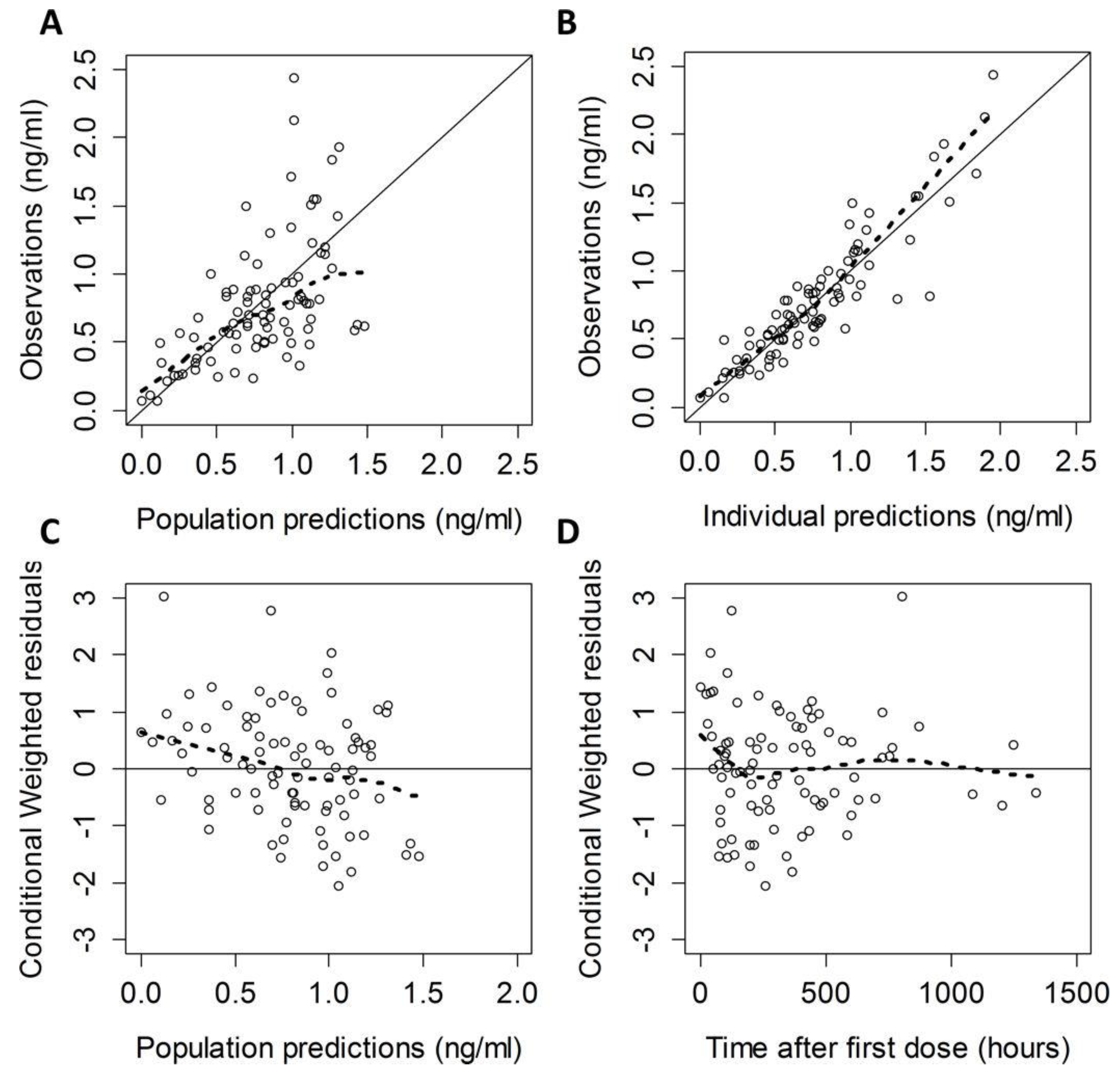
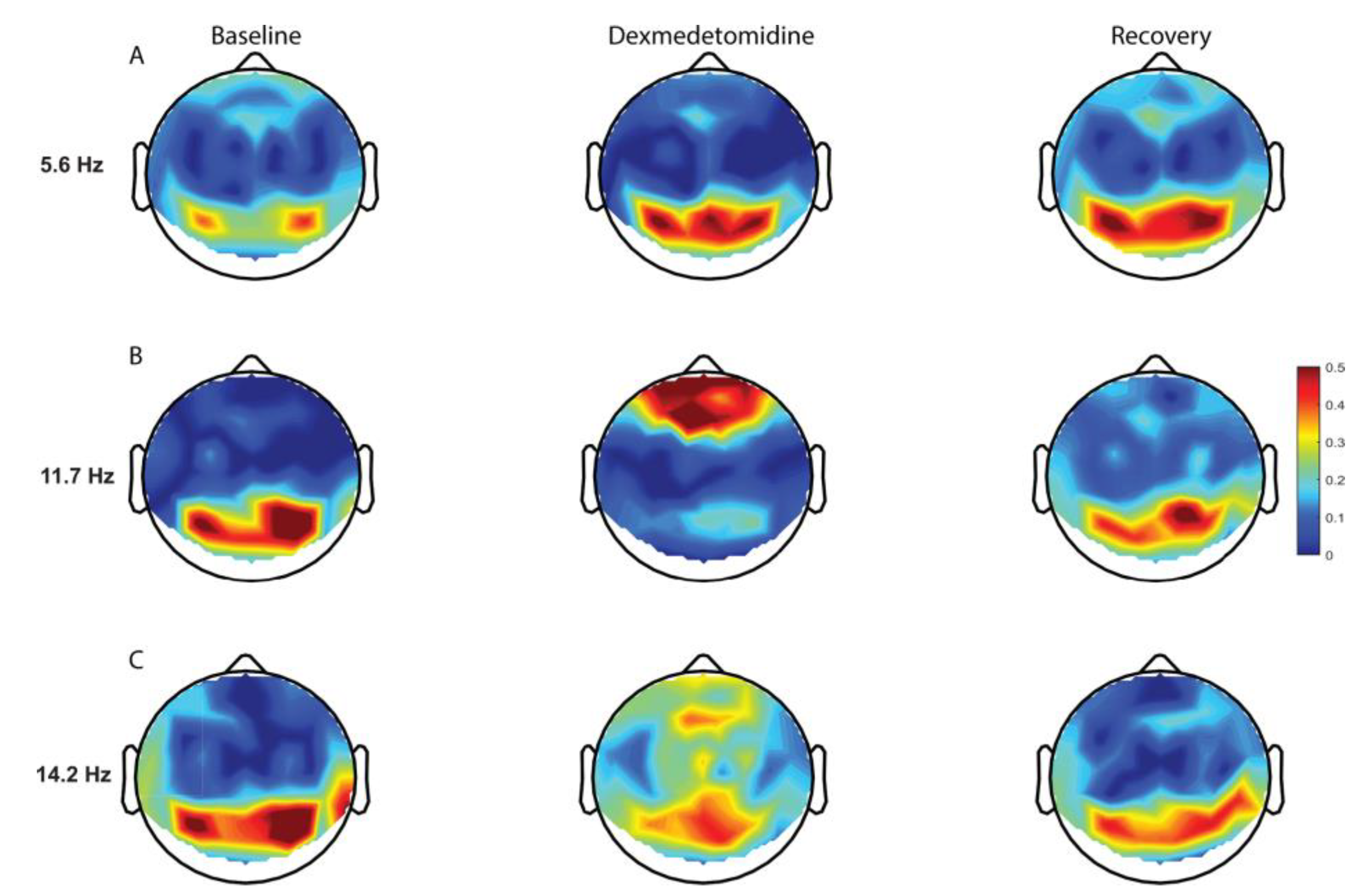

| Author | Year | Dose (ED = Effective Dose) | No. of Patients | Age | Procedure/Study | Onset Time (Median Time) | Sedation’s Success | Adverse Events | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Li et al. [31] | 2020 | 2.64 µg/kg (2.49–2.87 µg/kg ED 95%) | 68 | 1–3 years | Pulmonary function testing | 15.0 (12.3–19.0) min | 91% | None | ||||
| Tug et al. [86] | 2015 | 3 µg/kg | 4 µg/kg | 30 | 30 | 1–10 years | MRI I | 31 ± 13 min | 30 ± 9 min | 30% | 70% | None |
| Liu et al. [87] | 2016 | 2 µg/kg | 4 µg/kg | 121 | 6 months –5 years | Preoperative sedation | 15 min | 25 min | 85.5% | 77.6% | One child in the 2 µg/kg group had a reduction in systolic blood pressure to 29%. | |
| Yang et al. [88] | 2019 | 3.3 (2.48–3.53 ED50) µg/kg and 3.7 (3.44–3.73 ED 95) µg/kg in the cyanotic group and 1.7 (1.00–2.03 ED50) µg/kg and 2.2 (1.96–2.23 ED95) µg/kg in the acyanotic group. | 50 patients with congenital heart disease (23 in the cyanotic group and 27 in the acyanotic group) | ≤18 months | Transthoracic echocardiography in children with congenital heart disease | 13.9 min in the cyanotic group and 17.5 min in the acyanotic group | 50% | None | ||||
| Miller et al. [89] | 2016 | 2.5–3 µg/kg | 63 | 17.5 mean age | Transthoracic echocardiography | 28 min | 90% | Five patients with mild to moderate hypotension without need for intervention. | ||||
| Tenney et al. [90] | 2019 | 2 µg/kg | 26 | 5.5–20.5 years | Magnetoencephalography in patients with medically intractable epilepsy | 25.5 min | 100% | None | ||||
| Author | Year | Intervention | Dose | No. of Patients | Age | Procedure/ Intervention | Sedation’s Success | Adverse Events |
|---|---|---|---|---|---|---|---|---|
| Behrle et al. [91] | 2017 | IN Dex II vs. intravenous sedative medications | 3 µg/kg Dex vs. Midazolam, Propofol, Pentobarbital, Ketamine | 109 vs. 690 | 6 months–18 years | Non-invasive procedures | 92% (with 39% receiving in Midazolam for sedation ≥ 45 min) | Not significant |
| Li et al. [92] | 2020 | IN Dex II vs. Oral Chloral Hydrate | 1–3 µg/kg Dex | 720 | ≤4 years | Ophthalmic examination, transthoracic echocardiograph, auditory brainstem response testing, CT III and MRI IV imaging | Higher success rate in the first group (RR 1.12) | Lower incidence of nausea and vomiting in the IN Dex II group |
| 25 to 80 mg/kg Chloral Idrate | ||||||||
| Zhang et al. [93] | 2015 | Two rescue doses of IN Dex II vs. second oral dose of Chloral Hydrate after 50 mg of oral Chloral Hydrate | 1 and 2 µg/kg Dex | 150 | 1–6 months | MRI IV | 94% and 98% in the IN Dex groups | None |
| 25 mg/kg Chloral Idrate | ||||||||
| Ghai et al. [94] | 2017 | IN Dex II vs. Oral Midazolam | 2.5 µg/kg Dex | 59 | 6 years | IV cannulation CT III scan | 67% vs. 24% achieved a Ramsey Sedation Score higher than 4 | None |
| 0.5 mg/kg Midazolam | ||||||||
| Mukherjee et al. [95] | 2015 | IN Dex II vs. IN I Clonidine before Sevoflurane anesthesia | 1 µg/kg Dex | 80 | 3–7 years | Preoperative sedation | Emergence agitation was lower in the first group | Nausea and vomiting (no statistical differences between the 2 cohorts) |
| 4 µg/kg Clonidine |
| Author | Year | Intervention | Dose | No. of Patients | Age | Procedure/Intervention | Onset Time | Sedation’s Success | Adverse Events |
|---|---|---|---|---|---|---|---|---|---|
| Cozzi et al. [96] | 2017 | IN Dex II + Oral Midazolam | 3 µg/kg Dex | 108 | 4 months–17 years | MRI | 33 (10–65) min | 84% (90% < 2 years, 94% < 1 year) | Not significant i |
| 0.5 mg/kg Midazolam | |||||||||
| Bua et al. [97] | 2018 | IN Dex II + IN I Midazolam in case of failure | 3 µg/kg Dex | 52 | Ex pre-term babes at 40 weeks of gestational age | Brain MRI | 10 (IQR 8–12) min | 51% with Dex only, 100% with Dex+ Midazolam | 5% had apnea needing positive pressure ventilation 13% had brief self-resolving desaturation (SpO2 < 94%) |
| 0.2 mg/kg Midazolam | |||||||||
| Yang et al. [88] | 2019 | IN Dex II + IN I Ketamine | 2 µg/kg Dex | 17948 | 21 (IQR 10–34) months | Color doppler ultrasound, Pulmonary function, EEG III, MRI IV, ECGV, auditory brainstem response testing, Fundus examination, CT VI | 15 (15–20) min | 93% (1.8% required intranasal sedation rescue) ii | 0.02% emergent airway intervention 0.01% cardiac arrhythmias 0.59% minor adverse Events iii |
| 1 mg/kg Ketamine |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mahmoud, M.; Barbi, E.; Mason, K.P. Dexmedetomidine: What’s New for Pediatrics? A Narrative Review. J. Clin. Med. 2020, 9, 2724. https://doi.org/10.3390/jcm9092724
Mahmoud M, Barbi E, Mason KP. Dexmedetomidine: What’s New for Pediatrics? A Narrative Review. Journal of Clinical Medicine. 2020; 9(9):2724. https://doi.org/10.3390/jcm9092724
Chicago/Turabian StyleMahmoud, Mohamed, Egidio Barbi, and Keira P. Mason. 2020. "Dexmedetomidine: What’s New for Pediatrics? A Narrative Review" Journal of Clinical Medicine 9, no. 9: 2724. https://doi.org/10.3390/jcm9092724
APA StyleMahmoud, M., Barbi, E., & Mason, K. P. (2020). Dexmedetomidine: What’s New for Pediatrics? A Narrative Review. Journal of Clinical Medicine, 9(9), 2724. https://doi.org/10.3390/jcm9092724





