Venous Thromboembolism among Hospitalized Patients with COVID-19 Undergoing Thromboprophylaxis: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Methods
2.1. Search Strategies and Selection Criteria
2.2. Data Extraction
2.3. Quality Assessment
2.4. Study Endpoints
2.5. Statistical Analysis
3. Results
3.1. Incidence of VTE
3.2. Incidence of PE
3.3. Incidence of DVT
3.4. Mortality
3.5. Difference in D-dimer between Cases and Controls
4. Discussion
Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| COVID-19 | coronavirus disease 2019 |
| DVT | deep vein thrombosis |
| LMWH | low-molecular-weight heparin |
| PE | pulmonary embolism |
| PPS | Padua prediction score |
| UFH | unfractionated heparin |
| VTE | venous thromboembolism |
References
- Li, L.Q.; Huang, T.; Wang, Y.Q.; Wang, Z.P.; Liang, Y.; Huang, T.B.; Zhang, H.Y.; Sun, W.; Wang, Y. COVID-19 patients’ clinical characteristics, discharge rate, and fatality rate of meta-analysis. J. Med. Virol. 2020, 92, 577–583. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Zheng, Y.; Gou, X.; Pu, K.; Chen, Z.; Guo, Q.; Ji, R.; Wang, H.; Wang, Y.; Zhou, Y. Prevalence of comorbidities and its effects in patients infected with SARS-CoV-2: A systematic review and meta-analysis. Int. J. Infect. Dis. 2020, 94, 91–95. [Google Scholar] [CrossRef] [PubMed]
- Connors, J.M.; Levy, J.H. COVID-19 and its implications for thrombosis and anticoagulation. Blood 2020, 135, 2033–2040. [Google Scholar] [CrossRef] [PubMed]
- Tang, N.; Li, D.; Wang, X.; Sun, Z. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J. Thromb. Haemost. 2020, 18, 844–847. [Google Scholar] [CrossRef] [PubMed]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- Guan, W.J.; Ni, Z.Y.; Hu, Y.; Liang, W.H.; Ou, C.Q.; He, J.X.; Liu, L.; Shan, H.; Lei, C.L.; Hui, D.S.C.; et al. Clinical Characteristics of Coronavirus Disease 2019 in China. N. Engl. J. Med. 2020, 382, 1708–1720. [Google Scholar] [CrossRef]
- Wang, D.; Hu, B.; Hu, C.; Zhu, F.; Liu, X.; Zhang, J.; Wang, B.; Xiang, H.; Cheng, Z.; Xiong, Y.; et al. Clinical Characteristics of 138 Hospitalized Patients with 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, China. JAMA 2020, 323, 1061–1069. [Google Scholar] [CrossRef]
- Thachil, J.; Tang, N.; Gando, S.; Falanga, A.; Cattaneo, M.; Levi, M.; Clark, C.; Iba, T. ISTH interim guidance on recognition and management of coagulopathy in COVID-19. J. Thromb. Haemost. 2020, 18, 1023–1026. [Google Scholar] [CrossRef]
- Tapson, V.F.; Decousus, H.; Pini, M.; Chong, B.H.; Froehlich, J.B.; Monreal, M.; Spyropoulos, A.C.; Merli, G.J.; Zotz, R.B.; Bergmann, J.F.; et al. Venous thromboembolism prophylaxis in acutely ill hospitalized medical patients: Findings from the International Medical Prevention Registry on Venous Thromboembolism. Chest 2007, 132, 936–945. [Google Scholar] [CrossRef]
- Geerts, W.; Cook, D.; Selby, R.; Etchells, E. Venous thromboembolism and its prevention in critical care. J. Crit. Care 2002, 17, 95–104. [Google Scholar] [CrossRef]
- Cade, J.F. High risk of the critically ill for venous thromboembolism. Crit. Care Med. 1982, 10, 448–450. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, M.; Kupfer, Y.Y.; Tessler, S. Subcutaneous heparin prophylaxis significantly reduces the incidence of venous thromboembolic events in the critically ill. Crit. Care Med. 1999, 27, A69. [Google Scholar] [CrossRef]
- Fraisse, F.; Holzapfel, L.; Couland, J.M.; Simonneau, G.; Bedock, B.; Feissel, M.; Herbecq, P.; Pordes, R.; Poussel, J.F.; Roux, L. Nadroparin in the prevention of deep vein thrombosis in acute decompensated COPD. The Association of Non-University Affiliated Intensive Care Specialist Physicians of France. Am. J. Respir. Crit. Care Med. 2000, 161, 1109–1114. [Google Scholar] [CrossRef] [PubMed]
- Chi, G.; Gibson, C.M.; Kalayci, A.; Cohen, A.T.; Hernandez, A.F.; Hull, R.D.; Kahe, F.; Jafarizade, M.; Sharfaei, S.; Liu, Y.; et al. Extended-duration betrixaban versus shorter-duration enoxaparin for venous thromboembolism prophylaxis in critically ill medical patients: An APEX trial substudy. Intensive Care Med. 2019, 45, 477–487. [Google Scholar] [CrossRef]
- Bikdeli, B.; Madhavan, M.V.; Jimenez, D.; Chuich, T.; Dreyfus, I.; Driggin, E.; Nigoghossian, C.; Ageno, W.; Madjid, M.; Guo, Y.; et al. COVID-19 and Thrombotic or Thromboembolic Disease: Implications for Prevention, Antithrombotic Therapy, and Follow-Up: JACC State-of-the-Art Review. J. Am. Coll. Cardiol. 2020, 75, 2950–2973. [Google Scholar] [CrossRef]
- Driggin, E.; Madhavan, M.V.; Bikdeli, B.; Chuich, T.; Laracy, J.; Biondi-Zoccai, G.; Brown, T.S.; Der Nigoghossian, C.; Zidar, D.A.; Haythe, J.; et al. Cardiovascular Considerations for Patients, Health Care Workers, and Health Systems During the COVID-19 Pandemic. J. Am. Coll. Cardiol. 2020, 75, 2352–2371. [Google Scholar] [CrossRef]
- Hunt, B.; Retter, A.; McClintock, C. Practical Guidance for the Prevention of Thrombosis and Management of Coagulopathy and Disseminated Intravascular Coagulation of Patients Infected with COVID-19; British Society for Haematology: London, England, 2020. [Google Scholar]
- World Health Organization. Clinical Management of Severe Acute Respiratory Infection When Novel Coronavirus (2019-nCoV) Infection Is Suspected: Interim Guidance; WHO/nCoV/Clinical/2020.3; World Health Organization: Geneva, Switzerland, 2020. [Google Scholar]
- Helms, J.; Tacquard, C.; Severac, F.; Leonard-Lorant, I.; Ohana, M.; Delabranche, X.; Merdji, H.; Clere-Jehl, R.; Schenck, M.; Fagot Gandet, F.; et al. High risk of thrombosis in patients with severe SARS-CoV-2 infection: A multicenter prospective cohort study. Intensive Care Med. 2020, 46, 1089–1098. [Google Scholar] [CrossRef]
- Middeldorp, S.; Coppens, M.; van Haaps, T.F.; Foppen, M.; Vlaar, A.P.; Muller, M.C.A.; Bouman, C.C.S.; Beenen, L.F.M.; Kootte, R.S.; Heijmans, J.; et al. Incidence of venous thromboembolism in hospitalized patients with COVID-19. J. Thromb. Haemost. 2020. [Google Scholar] [CrossRef]
- Lipsey, M.W.; Wilson, D.B. Practical Meta-Analysis; SAGE Publications, Inc.: Thousand Oaks, CA, USA, 2001. [Google Scholar]
- Wan, X.; Wang, W.; Liu, J.; Tong, T. Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med. Res. Methodol. 2014, 14, 35. [Google Scholar] [CrossRef]
- Klok, F.A.; Kruip, M.; van der Meer, N.J.M.; Arbous, M.S.; Gommers, D.; Kant, K.M.; Kaptein, F.H.J.; van Paassen, J.; Stals, M.A.M.; Huisman, M.V.; et al. Confirmation of the high cumulative incidence of thrombotic complications in critically ill ICU patients with COVID-19: An updated analysis. Thromb. Res. 2020, 191, 48–150. [Google Scholar] [CrossRef]
- Llitjos, J.F.; Leclerc, M.; Chochois, C.; Monsallier, J.M.; Ramakers, M.; Auvray, M.; Merouani, K. High incidence of venous thromboembolic events in anticoagulated severe COVID-19 patients. J. Thromb. Haemost. 2020, 18, 1743–1746. [Google Scholar] [CrossRef]
- Maatman, T.K.; Jalali, F.; Feizpour, C.; Douglas, A., 2nd; McGuire, S.P.; Kinnaman, G.; Hartwell, J.L.; Maatman, B.T.; Kreutz, R.P.; Kapoor, R.; et al. Routine Venous Thromboembolism Prophylaxis May Be Inadequate in the Hypercoagulable State of Severe Coronavirus Disease 2019. Crit. Care Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- Poissy, J.; Goutay, J.; Caplan, M.; Parmentier, E.; Duburcq, T.; Lassalle, F.; Jeanpierre, E.; Rauch, A.; Labreuche, J.; Susen, S.; et al. Pulmonary Embolism in COVID-19 Patients: Awareness of an Increased Prevalence. Circulation 2020. [Google Scholar] [CrossRef] [PubMed]
- Artifoni, M.; Danic, G.; Gautier, G.; Gicquel, P.; Boutoille, D.; Raffi, F.; Neel, A.; Lecomte, R. Systematic assessment of venous thromboembolism in COVID-19 patients receiving thromboprophylaxis: Incidence and role of D-dimer as predictive factors. J. Thromb. Thrombolysis 2020, 50, 211–216. [Google Scholar] [CrossRef]
- Demelo-Rodriguez, P.; Cervilla-Munoz, E.; Ordieres-Ortega, L.; Parra-Virto, A.; Toledano-Macias, M.; Toledo-Samaniego, N.; Garcia-Garcia, A.; Garcia-Fernandez-Bravo, I.; Ji, Z.; de-Miguel-Diez, J.; et al. Incidence of asymptomatic deep vein thrombosis in patients with COVID-19 pneumonia and elevated D-dimer levels. Thromb. Res. 2020, 192, 3–26. [Google Scholar] [CrossRef]
- Lodigiani, C.; Iapichino, G.; Carenzo, L.; Cecconi, M.; Ferrazzi, P.; Sebastian, T.; Kucher, N.; Studt, J.D.; Sacco, C.; Alexia, B.; et al. Venous and arterial thromboembolic complications in COVID-19 patients admitted to an academic hospital in Milan, Italy. Thromb. Res. 2020, 191, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Tang, N.; Bai, H.; Chen, X.; Gong, J.; Li, D.; Sun, Z. Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy. J. Thromb. Haemost. 2020, 18, 1094–1099. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Feng, X.; Zhang, D.; Jiang, C.; Mei, H.; Wang, J.; Zhang, C.; Li, H.; Xia, X.; Kong, S.; et al. Deep Vein Thrombosis in Hospitalized Patients with Coronavirus Disease 2019 (COVID-19) in Wuhan, China: Prevalence, Risk Factors, and Outcome. Circulation 2020. [Google Scholar] [CrossRef]
- Cohoon, K.P.; Mahe, G.; Tafur, A.J.; Spyropoulos, A.C. Emergence of institutional antithrombotic protocols for coronavirus 2019. Res. Pract. Thromb. Haemost. 2020, 4, 510–517. [Google Scholar] [CrossRef]
- Chi, G.; Goldhaber, S.Z.; Kittelson, J.M.; Turpie, A.G.G.; Hernandez, A.F.; Hull, R.D.; Gold, A.; Curnutte, J.T.; Cohen, A.T.; Harrington, R.A.; et al. Effect of extended-duration thromboprophylaxis on venous thromboembolism and major bleeding among acutely ill hospitalized medical patients: A bivariate analysis. J. Thromb. Haemost. 2017, 15, 1913–1922. [Google Scholar] [CrossRef]
- Cattaneo, M.; Bertinato, E.M.; Birocchi, S.; Brizio, C.; Malavolta, D.; Manzoni, M.; Muscarella, G.; Orlandi, M. Pulmonary Embolism or Pulmonary Thrombosis in COVID-19? Is the Recommendation to Use High-Dose Heparin for Thromboprophylaxis Justified? Thromb. Haemost. 2020. [Google Scholar] [CrossRef] [PubMed]
- Wichmann, D.; Sperhake, J.P.; Lutgehetmann, M.; Steurer, S.; Edler, C.; Heinemann, A.; Heinrich, F.; Mushumba, H.; Kniep, I.; Schroder, A.S.; et al. Autopsy Findings and Venous Thromboembolism in Patients With COVID-19. Ann. Intern. Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- Lax, S.F.; Skok, K.; Zechner, P.; Kessler, H.H.; Kaufmann, N.; Koelblinger, C.; Vander, K.; Bargfrieder, U.; Trauner, M. Pulmonary Arterial Thrombosis in COVID-19 With Fatal Outcome: Results from a Prospective, Single-Center, Clinicopathologic Case Series. Ann. Intern. Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- Varga, Z.; Flammer, A.J.; Steiger, P.; Haberecker, M.; Andermatt, R.; Zinkernagel, A.S.; Mehra, M.R.; Schuepbach, R.A.; Ruschitzka, F.; Moch, H. Endothelial cell infection and endotheliitis in COVID-19. Lancet 2020, 395, 1417–1418. [Google Scholar] [CrossRef]
- Joly, B.S.; Siguret, V.; Veyradier, A. Understanding pathophysiology of hemostasis disorders in critically ill patients with COVID-19. Intensive Care Med. 2020. [Google Scholar] [CrossRef]
- Appel, G.B. Thrombotic microangiopathies: Similar presentations, different therapies. Cleve Clin. J. Med. 2017, 84, 114–130. [Google Scholar] [CrossRef]
- Wang, T.; Chen, R.; Liu, C.; Liang, W.; Guan, W.; Tang, R.; Tang, C.; Zhang, N.; Zhong, N.; Li, S. Attention should be paid to venous thromboembolism prophylaxis in the management of COVID-19. Lancet Haematol. 2020, 7, e362–e363. [Google Scholar] [CrossRef]
- Spyropoulos, A.C.; Lipardi, C.; Xu, J.; Peluso, C.; Spiro, T.E.; De Sanctis, Y.; Barnathan, E.S.; Raskob, G.E. Modified IMPROVE VTE Risk Score and Elevated D-Dimer Identify a High Venous Thromboembolism Risk in Acutely Ill Medical Population for Extended Thromboprophylaxis. Open 2020, 4, e59–e65. [Google Scholar] [CrossRef]
- Gibson, C.M.; Spyropoulos, A.C.; Cohen, A.T.; Hull, R.D.; Goldhaber, S.Z.; Yusen, R.D.; Hernandez, A.F.; Korjian, S.; Daaboul, Y.; Gold, A.; et al. The IMPROVEDD VTE Risk Score: Incorporation of D-Dimer into the IMPROVE Score to Improve Venous Thromboembolism Risk Stratification. Open 2017, 1, e56–e65. [Google Scholar] [CrossRef]
- Chen, N.; Zhou, M.; Dong, X.; Qu, J.; Gong, F.; Han, Y.; Qiu, Y.; Wang, J.; Liu, Y.; Wei, Y.; et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: A descriptive study. Lancet 2020, 395, 507–513. [Google Scholar] [CrossRef]
- Henry, B.M.; de Oliveira, M.H.S.; Benoit, S.; Plebani, M.; Lippi, G. Hematologic, biochemical and immune biomarker abnormalities associated with severe illness and mortality in coronavirus disease 2019 (COVID-19): A meta-analysis. Clin. Chem. Lab. Med. 2020, 58, 1021–1028. [Google Scholar] [CrossRef] [PubMed]
- Chi, G.; Gibson, C.M.; Liu, Y.; Hernandez, A.F.; Hull, R.D.; Cohen, A.T.; Harrington, R.A.; Goldhaber, S.Z. Inverse relationship of serum albumin to the risk of venous thromboembolism among acutely ill hospitalized patients: Analysis from the APEX trial. Am. J. Hematol. 2019, 94, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Chi, G.; Gibson, C.M.; Hernandez, A.F.; Hull, R.D.; Kazmi, S.H.A.; Younes, A.; Walia, S.S.; Pitliya, A.; Singh, A.; Kahe, F.; et al. Association of Anemia with Venous Thromboembolism in Acutely Ill Hospitalized Patients: An APEX Trial Substudy. Am. J. Med. 2018, 131, 972.e971–972.e977. [Google Scholar] [CrossRef] [PubMed]
- Chi, G.; Goldhaber, S.Z.; Hull, R.D.; Hernandez, A.F.; Kerneis, M.; Al Khalfan, F.; Cohen, A.T.; Harrington, R.A.; Gibson, C.M. Thrombus Burden of Deep Vein Thrombosis and Its Association with Thromboprophylaxis and D-Dimer Measurement: Insights from the APEX Trial. Thromb. Haemost. 2017, 117, 2389–2395. [Google Scholar] [CrossRef] [PubMed]
- Amin, A.N.; Varker, H.; Princic, N.; Lin, J.; Thompson, S.; Johnston, S. Duration of venous thromboembolism risk across a continuum in medically ill hospitalized patients. J. Hosp. Med. 2012, 7, 231–238. [Google Scholar] [CrossRef]
- Heit, J.A.; Crusan, D.J.; Ashrani, A.A.; Petterson, T.M.; Bailey, K.R. Effect of a near-universal hospitalization-based prophylaxis regimen on annual number of venous thromboembolism events in the US. Blood 2017, 130, 109–114. [Google Scholar] [CrossRef]
- Chi, G.; Yee, M.K.; Amin, A.N.; Goldhaber, S.Z.; Hernandez, A.F.; Hull, R.D.; Cohen, A.T.; Harrington, R.A.; Gibson, C.M. Extended-Duration Betrixaban Reduces the Risk of Rehospitalization Associated with Venous Thromboembolism among Acutely Ill Hospitalized Medical Patients: Findings from the APEX Trial (Acute Medically Ill Venous Thromboembolism Prevention with Extended Duration Betrixaban Trial). Circulation 2018, 137, 91–94. [Google Scholar]
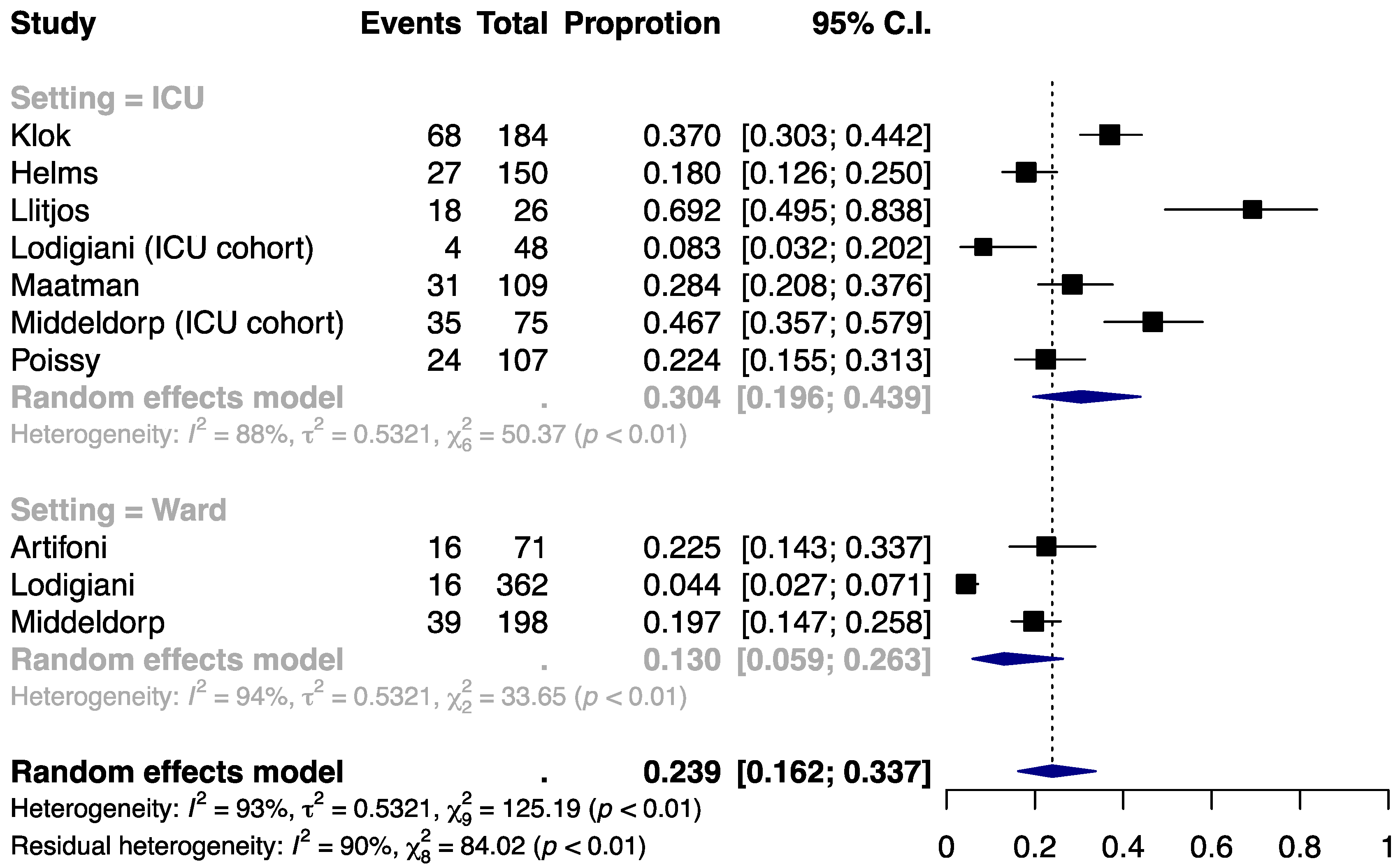
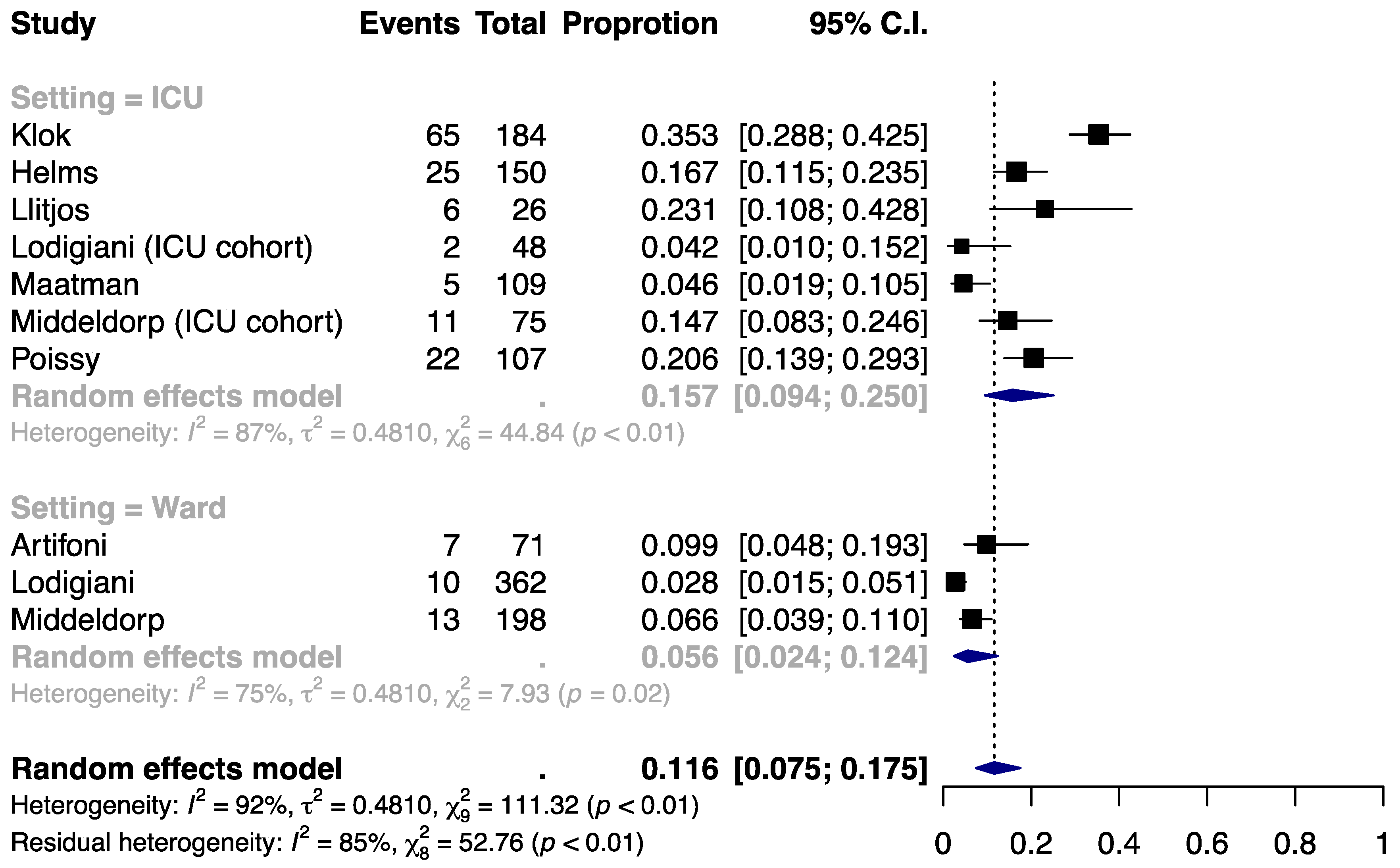
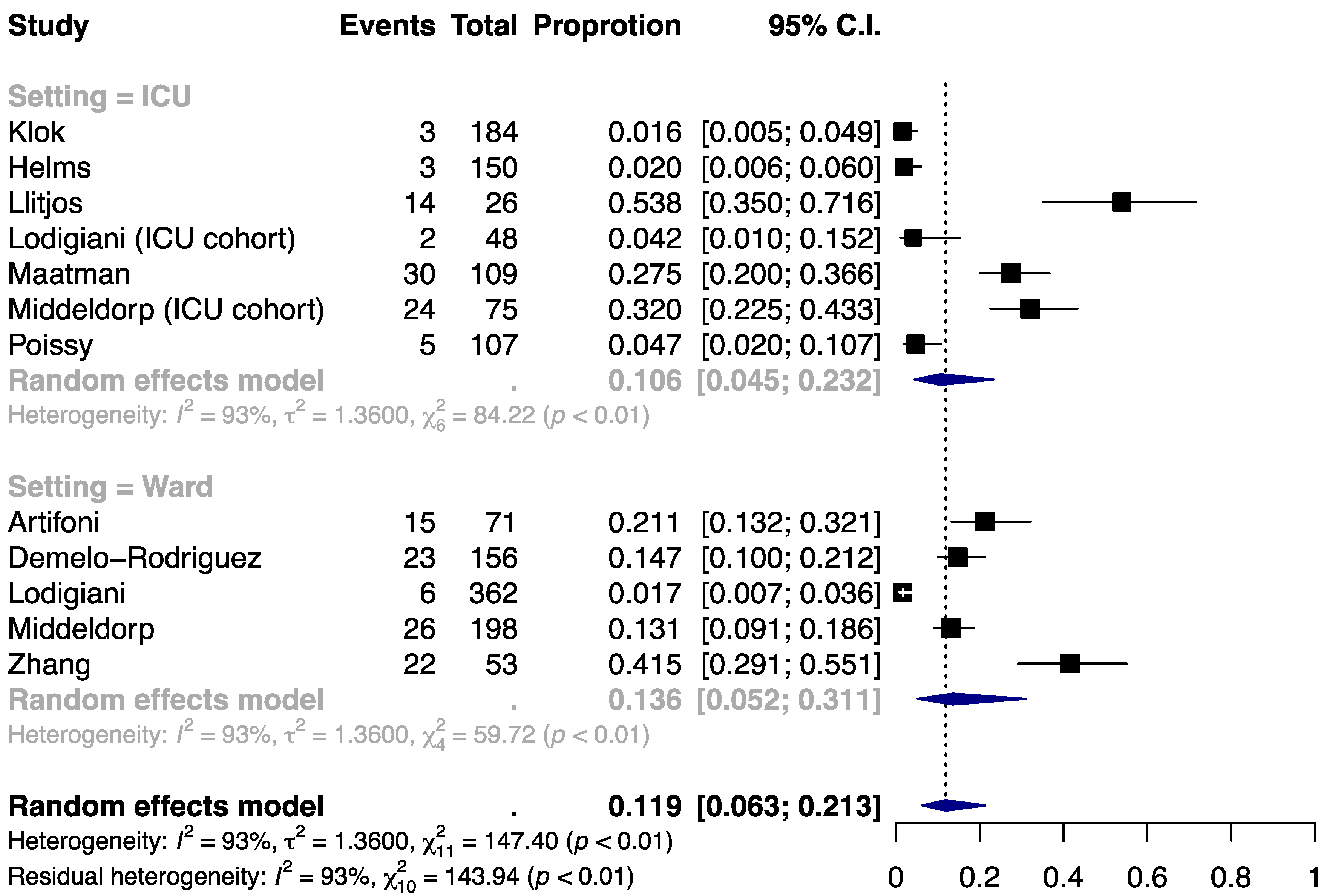
| Author | Population | Setting | Thromboprophylaxis | Indication for VTE Detection | N | Age, year | Male, % | BMI, kg/m2 | HTN, % | DM, % | Cancer, % | Smoking, % | History of VTE, % | ICU Admission, % | D-Dimer, μg/mL | Follow-up |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Klok | COVID-19 patients admitted to the ICU | ICU | All patients received at least standard doses thromboprophylaxis (nadroparin 2850–5700 IU once daily to twice daily), with 9.2% receiving therapeutic anticoagulation at admission | CTPA/ultrasonography for PE/DVT if thrombotic complications were clinically suspected | 184 | 64 ± 12 | 76 | NR | NR | NR | 2.7 | NR | NR | 100 | NR | 14 days |
| Helms | COVID-19 patients with acute respiratory distress syndrome admitted to the ICU | ICU | 70% received prophylactic dosing (4000 UI/day for LMWH or if contraindicated, UFH at 5–8 U/kg/h) and 30% received therapeutic dosing | CTPA, either at the admission in ICU or during the stay, if PE was suspected based on clinical or laboratory parameters evolution | 150 | 63 (53–71) | 81.3 | NR | NR | 20 | 6 | NR | 5.3 | 100 | 2.27 (1.16–20) | 7–30 days |
| Llitjos | Severe ICU COVID-19 patients | ICU | All patients were anticoagulated from admission: 31% (n = 8) with prophylactic anticoagulation and 69% (n = 18) with therapeutic anticoagulation | CTPA was performed systematically in patients with persistent hypoxemia or secondary deterioration. CDU was performed between day 1 and day 3 after admission. In patients without VTE during the first CDU, a second CDU was performed at day 7 | 26 | 68 (51.5–74.5) | 77 | NR | 85 | NR | 0 | 27 | 4 | 100 | 1.750 (1.130–2.850) | NR |
| Maatman | Patients with laboratory-confirmed SARS-CoV-2 infection requiring intensive care | ICU | All patients admitted with COVID-19 receive VTE chemoprophylaxis including either 5000 U subcutaneous heparin every 8 h, 40 mg enoxaparin daily, or 30 mg enoxaparin bid | Extremity DVT was diagnosed on four-extremity duplex ultrasound performed for clinical suspicion for DVT. PE was diagnosed on contrast-enhanced cross-sectional imaging | 109 | 61 ± 16 | 57 | NR | 68 | 39 | NR | 30 | NR | 100 | 0.506 (0.321–0.973) | 36–55 days |
| Poissy | COVID-19 patients admitted to the ICU for pneumonia | ICU | All patients received thromboprophylaxis (UFH or LMWH) | CTPA was performed on suspicion of PE upon admission and/or acute degradation of hemodynamic or respiratory status | 107 | NR | NR | NR | NR | NR | NR | NR | NR | 100 | NR | 6 days |
| Artifoni | Hospitalized patients with COVID-19 | Ward | Daily administration of weight-appropriate enoxaparin following institutional recommendations (40 mg/day for BMI < 30 kg/m2, 60 mg/day for BMI 30 to 40 kg/m2 and 40 mg twice daily for BMI > 40 kg/m2) and covering the whole hospital stay | All patients were systematically examined for DVT by low limb venous duplex ultrasonography at hospital discharge or earlier if thrombosis was clinically suspected. Chest angio-CT scan was performed in case of suspicion of PE | 71 | 64 (46.0–75) | 60.6 | 27.3 (25.0–31.2) | 41 | 20 | 6 | 9 | 7 | 18 | 0.79 (0.48–1.61) | 13 days |
| Demelo-Rodríguez | Hospitalized in non-ICU with diagnosis of COVID-19 pneumonia and D-dimer > 1000 ng/ml | Ward | All but three patients received standard doses of thromboprophylaxis: enoxaparin 40 mg per day or bemiparin 3500 UI per day | Complete CUS was performed to screen for asymptomatic DVT | 156 | 68.1 ± 14.5 | 65.4 | 26.9 ± 4.2 | NR | NR | 10.3 | NR | 1.3 | 10.3 | 2.148 (1.532–4.002) | 9 days |
| Lodigiani | Adult symptomatic patients with laboratory-proven COVID-19 | Ward/ICU | ICU cohort (n = 61): LMWH: The dosage was weight-adjusted in 17 patients and therapeutic in two patients on ambulatory treatment with direct oral anticoagulants General ward cohort (n = 327): 75% received initial in-hospital thromboprophylaxis: A prophylactic dosage was used in 41% of patients, 21% were treated with inter-mediate-dosage thromboprophylaxis, and 23% received therapeutic-dose anticoagulation | CTPA/ultrasonography were performed in subjects with signs or symptoms of DVT or with an unexplained clinical worsening of the respiratory function, primarily assessed using the PaO2/FIO2 ratio, or a rapid increase of D-dimer levels. Two-point CUS was used in the ICU; whole-leg ultrasound was performed in symptomatic patients in the general ward | 388 | 66 (55–75) | 68 | NR | 47.2 | 22.7 | 6.4 | 11.6 | 3.1 | 16 | NR | 18 days |
| Middeldorp | Hospitalized patients with COVID-19 | Ward/ICU | Ward patients received prophylaxis with nadroparin 2850 IU once daily or 5700 IU for patients with a body weight of ≥100 kg. From April 3 onwards, patients in ICU received a double dose of nadroparin as compared to patients on the wards, which was nadroparin 2850 IU twice daily (bid) for patients with a body weight <100 kg and 5700 IU bid for those ≥100 kg | Based on concerns of a high risk of fatal VTE following early observations, during the follow-up period CUS screening was performed in the ICU every 5 days, while also performing a single cross-sectional round of CUS at the ward in the 10 days prior to data collection | 198 | 61 ± 14 | 66 | 27 (24–31) | NR | NR | 3.5 | NR | 5.6 | 38 | 1.1 (0.7–2.3) | 7 days |
| Tang | Hospitalized patients with severe COVID-19. | Ward | Heparins (n = 99; 94 received 40–60 mg enoxaparin/d and 5 received UFH 10,000–15,000 U/d) vs. control (n = 350) | NR | 449 | 65.1 ± 12.0 | 60 | NR | NR | NR | NR | NR | NR | NR | 1.94 (0.90–9.44) | 28 days |
| Zhang | Critically ill adult patients with COVID-19 | Ward | 53 (37.1%) patients were given DVT prophylaxis with LMWH vs. control | CTPA was performed on suspicion of PE; ultrasound was performed to screen for DVT | 143 | 63 ± 14 | 51.7 | 23.6 ± 3.0 | 39.2 | 18.2 | 4.9 | 6.3 | 0.7 | 10.5 | 2.7 (0.6–8.0) | 24–55 days |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chi, G.; Lee, J.J.; Jamil, A.; Gunnam, V.; Najafi, H.; Memar Montazerin, S.; Shojaei, F.; Marszalek, J. Venous Thromboembolism among Hospitalized Patients with COVID-19 Undergoing Thromboprophylaxis: A Systematic Review and Meta-Analysis. J. Clin. Med. 2020, 9, 2489. https://doi.org/10.3390/jcm9082489
Chi G, Lee JJ, Jamil A, Gunnam V, Najafi H, Memar Montazerin S, Shojaei F, Marszalek J. Venous Thromboembolism among Hospitalized Patients with COVID-19 Undergoing Thromboprophylaxis: A Systematic Review and Meta-Analysis. Journal of Clinical Medicine. 2020; 9(8):2489. https://doi.org/10.3390/jcm9082489
Chicago/Turabian StyleChi, Gerald, Jane J. Lee, Adeel Jamil, Vamsikrishna Gunnam, Homa Najafi, Sahar Memar Montazerin, Fahimehalsadat Shojaei, and Jolanta Marszalek. 2020. "Venous Thromboembolism among Hospitalized Patients with COVID-19 Undergoing Thromboprophylaxis: A Systematic Review and Meta-Analysis" Journal of Clinical Medicine 9, no. 8: 2489. https://doi.org/10.3390/jcm9082489
APA StyleChi, G., Lee, J. J., Jamil, A., Gunnam, V., Najafi, H., Memar Montazerin, S., Shojaei, F., & Marszalek, J. (2020). Venous Thromboembolism among Hospitalized Patients with COVID-19 Undergoing Thromboprophylaxis: A Systematic Review and Meta-Analysis. Journal of Clinical Medicine, 9(8), 2489. https://doi.org/10.3390/jcm9082489





