Neuroimaging-Based Scalp Acupuncture Locations for Dementia
Abstract
1. Introduction
2. Methods
2.1. Method 1: Identifying Dementia-Associated Surface Cortical Regions for Scalp Acupuncture Using Meta-Analysis
2.2. Method 2: Identifying Dementia-Associated Surface Regions from the Resting-State Functional Connectivity Analysis
2.2.1. Subjects and MRI Data Acquisition
2.2.2. fMRI Data Preprocessing
2.3. Method 3: Identifying Dementia-Associated Surface Regions from the DTI Analysis
2.3.1. Diffusion MRI Data Acquisition
2.3.2. dMRI Data Preprocessing and Tractography
2.4. Summarizing Results from Neuroimaging Analyses
3. Results
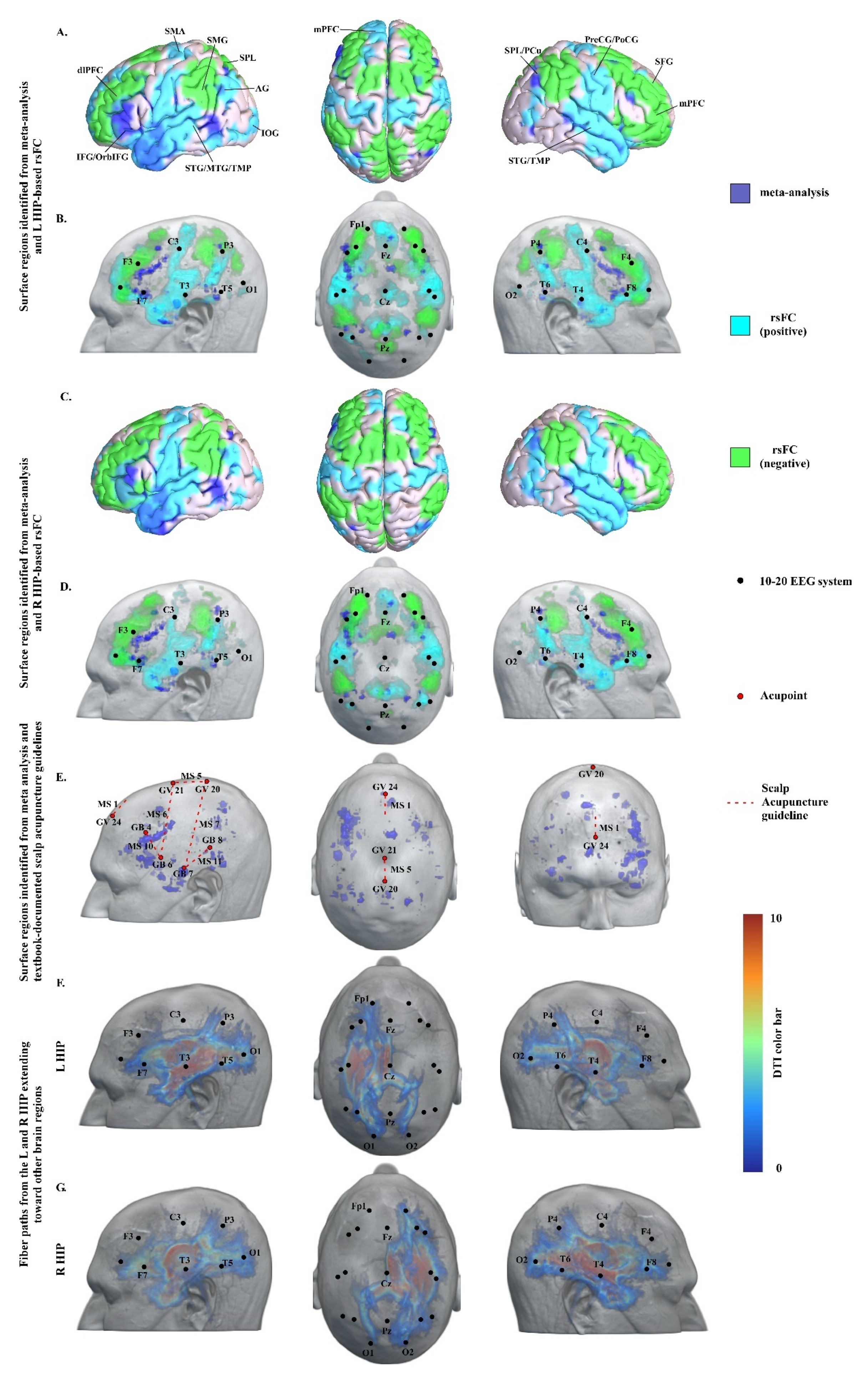
3.1. Meta-Analysis Results
3.2. Resting-State Functional Connectivity Analysis Results
3.3. DTI Data Analysis Results
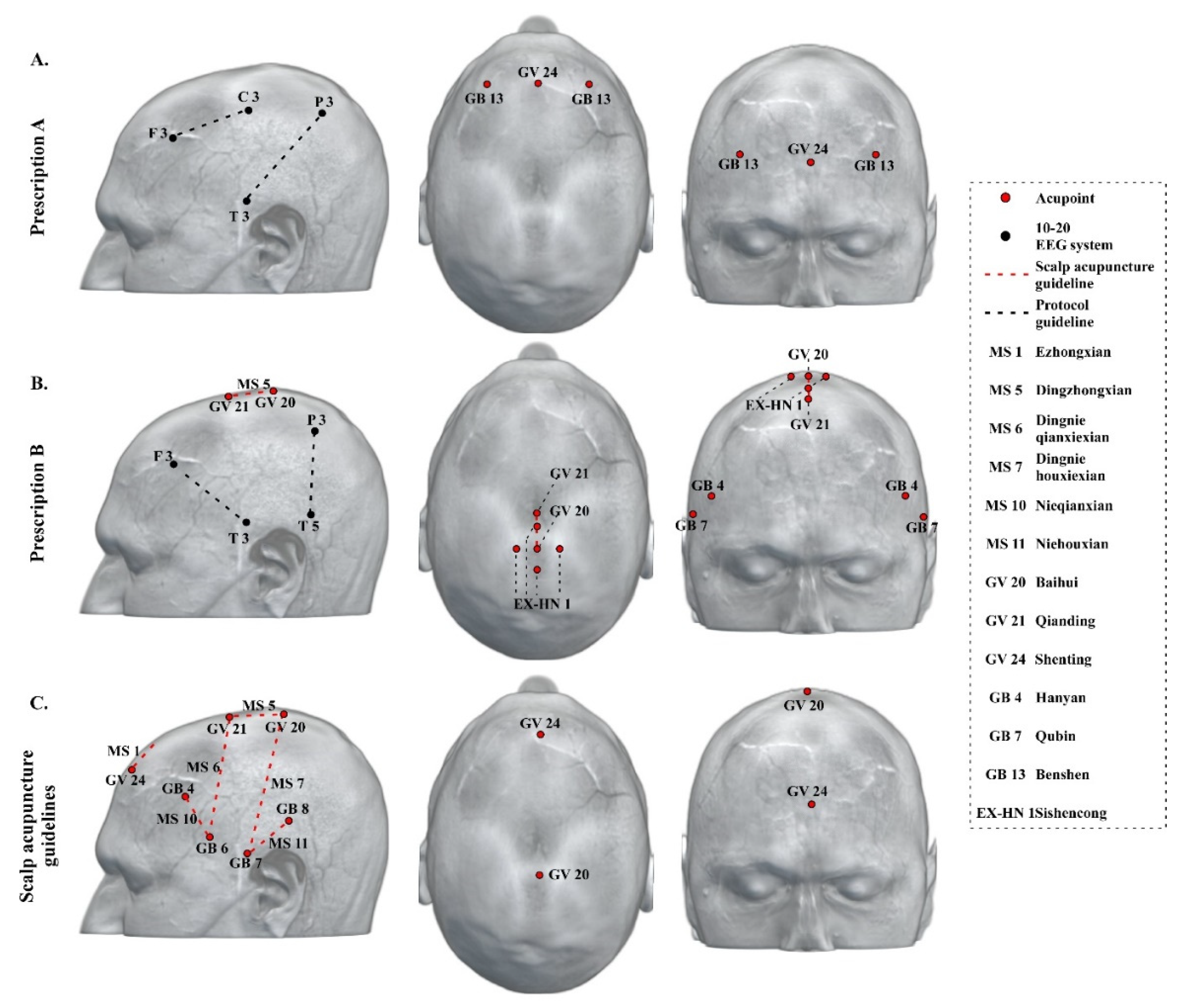
3.4. Neuroimaging-Based Scalp Acupuncture Protocol
4. Discussion
4.1. Key Regions/Locations in the Neuroimaging-Based Scalp Acupuncture Prescription
4.2. Current Scalp Acupuncture Protocols for Dementia and Differences Compared to the Neuroimaging-Based Scalp Acupuncture Protocol
4.3. Additional Application of the Neuroimaging-Based Scalp Acupuncture Prescription
4.4. Limitation and Future Directions
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Jiang, S.; Zhang, G.; Li, K.; Lang, Y.; Tan, Z.; Zhang, Y.; Zou, Y.; Wu, Y.; Cui, F. A resting-state functional magnetic resonance imaging study on the efficacy of brain function rehabilitation in post-stroke hemiplegia using scalp acupuncture. J. Med. Imaging Health Inform. 2015, 5, 1752–1758. [Google Scholar] [CrossRef]
- Park, S.U.; Shin, A.S.; Jahng, G.H.; Moon, S.K.; Park, J.M. Effects of scalp acupuncture versus upper and lower limb acupuncture on signal activation of blood oxygen level dependent (BOLD) fMRI of the brain and somatosensory cortex. J. Altern. Complement. Med. 2009, 15, 1193–1200. [Google Scholar] [CrossRef] [PubMed]
- Jing, L.; Jianhua, X.; Guirong, D. Clinical study on effect of scalp-acupuncture in treating acute cerebral hemorrhage. Chin. J. Integr. Tradit. West. Med. 1999, 5, 265–268. [Google Scholar] [CrossRef]
- Abou-Saleh, M.T. Neuroimaging in psychiatry: An update. J. Psychosom. Res. 2006, 61, 289–293. [Google Scholar] [CrossRef] [PubMed]
- Livingston, G.; Sommerlad, A.; Orgeta, V.; Costafreda, S.G.; Huntley, J.; Ames, D.; Ballard, C.; Banerjee, S.; Burns, A.; Cohen-Mansfield, J.; et al. Dementia prevention, intervention, and care. Lancet 2017, 390, 2673–2734. [Google Scholar] [CrossRef]
- Niu, W.M.; Liu, H.Y. Clinical study on treatment of vascular dementia with scalp acupuncture. J. Acupunct. Tuina Sci. 2008, 6, 24–26. [Google Scholar] [CrossRef]
- Liu, Y. Therapeutic effects of scalp-acupuncture in patients with vascular dementia induced by cerebral infarction: A randomized controlled trial. J. Chin. Integr. Med. 2008, 6, 806–809. [Google Scholar] [CrossRef]
- Teng, X.y.; Lai, Z.j. Clinical efficacy observation of scalp-acupuncture-based therapy for vascular dementia. J. Acupunct. Tuina Sci. 2012, 10, 34–37. [Google Scholar] [CrossRef]
- Yarkoni, T.; Poldrack, R.A.; Nichols, T.E.; Van Essen, D.C.; Wager, T.D. Large-scale automated synthesis of human functional neuroimaging data. Nat. Methods 2011, 8, 665–670. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, B.; Wilson, G.; Kong, J. New Perspective for Non-invasive Brain Stimulation Site Selection in Mild Cognitive Impairment: Based on Meta- and Functional Connectivity Analyses. Front. Aging Neurosci. 2019, 11, 228. [Google Scholar] [CrossRef]
- Zhang, B.; Liu, J.; Bao, T.; Wilson, G.; Park, J.; Zhao, B.; Kong, J. Locations for noninvasive brain stimulation in treating depressive disorders: A combination of meta-analysis and resting-state functional connectivity analysis. Aust. N. Z. J. Psychiatry 2020, 000486742092037. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Zhang, B.; Cao, J.; Yu, S.; Wilson, G.; Park, J.; Kong, J. Potential Locations for Noninvasive Brain Stimulation in Treating Autism Spectrum Disorders—A Functional Connectivity Study. Front. Psychiatry 2020, 11. [Google Scholar] [CrossRef]
- Fox, M.D.; Buckner, R.L.; White, M.P.; Greicius, M.D.; Pascual-Leone, A. Efficacy of transcranial magnetic stimulation targets for depression is related to intrinsic functional connectivity with the subgenual cingulate. Biol. Psychiatry 2012, 72, 595–603. [Google Scholar] [CrossRef] [PubMed]
- Tu, Y.; Zhang, B.; Cao, J.; Wilson, G.; Zhang, Z.; Kong, J. Identifying inter-individual differences in pain threshold using brain connectome: A test-retest reproducible study. Neuroimage 2019, 202, 116049. [Google Scholar] [CrossRef] [PubMed]
- Jack, C.R.; Dickson, D.W.; Parisi, J.E.; Xu, Y.C.; Cha, R.H.; O’Brien, P.C.; Edland, S.D.; Smith, G.E.; Boeve, B.F.; Tangalos, E.G.; et al. Antemortem MRI findings correlate with hippocampal neuropathology in typical aging and dementia. Neurology 2002, 58, 750–757. [Google Scholar] [CrossRef] [PubMed]
- Zarow, C.; Vinters, H.V.; Ellis, W.G.; Weiner, M.W.; Mungas, D.; White, L.; Chui, H.C. Correlates of hippocampal neuron number in Alzheimer’s disease and ischemic vascular dementia. Ann. Neurol. 2005, 57, 896–903. [Google Scholar] [CrossRef]
- Woolrich, M.W.; Jbabdi, S.; Patenaude, B.; Chappell, M.; Makni, S.; Behrens, T.; Beckmann, C.; Jenkinson, M.; Smith, S.M. Bayesian analysis of neuroimaging data in FSL. Neuroimage 2009, 45. [Google Scholar] [CrossRef]
- Andersson, J.L.R.; Sotiropoulos, S.N. An integrated approach to correction for off-resonance effects and subject movement in diffusion MR imaging. Neuroimage 2016, 125, 1063–1078. [Google Scholar] [CrossRef]
- Andersson, J.L.R.; Graham, M.S.; Zsoldos, E.; Sotiropoulos, S.N. Incorporating outlier detection and replacement into a non-parametric framework for movement and distortion correction of diffusion MR images. Neuroimage 2016, 141, 556–572. [Google Scholar] [CrossRef]
- Jbabdi, S.; Sotiropoulos, S.N.; Savio, A.M.; Graña, M.; Behrens, T.E.J. Model-based analysis of multishell diffusion MR data for tractography: How to get over fitting problems. Magn. Reson. Med. 2012, 68, 1846–1855. [Google Scholar] [CrossRef]
- Behrens, T.E.J.; Woolrich, M.W.; Jenkinson, M.; Johansen-Berg, H.; Nunes, R.G.; Clare, S.; Matthews, P.M.; Brady, J.M.; Smith, S.M. Characterization and Propagation of Uncertainty in Diffusion-Weighted MR Imaging. Magn. Reson. Med. 2003, 50, 1077–1088. [Google Scholar] [CrossRef] [PubMed]
- Behrens, T.E.J.; Berg, H.J.; Jbabdi, S.; Rushworth, M.F.S.; Woolrich, M.W. Probabilistic diffusion tractography with multiple fibre orientations: What can we gain? Neuroimage 2007, 34, 144–155. [Google Scholar] [CrossRef] [PubMed]
- Cutini, S.; Scatturin, P.; Zorzi, M. A new method based on ICBM152 head surface for probe placement in multichannel fNIRS. Neuroimage 2011, 54, 919–927. [Google Scholar] [CrossRef] [PubMed]
- Ciccarelli, O.; Behrens, T.E.; Altmann, D.R.; Orrell, R.W.; Howard, R.S.; Johansen-Berg, H.; Miller, D.H.; Matthews, P.M.; Thompson, A.J.; Ciccarelli, O. Probabilistic diffusion tractography: A potential tool to assess the rate of disease progression in amyotrophic lateral sclerosis. Brain 2006, 129, 1859–1871. [Google Scholar] [CrossRef] [PubMed]
- Burgmans, S.; van Boxtel, M.P.J.; Smeets, F.; Vuurman, E.F.P.M.; Gronenschild, E.H.B.M.; Verhey, F.R.J.; Uylings, H.B.M.; Jolles, J. Prefrontal cortex atrophy predicts dementia over a six-year period. Neurobiol. Aging 2009, 30, 1413–1419. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M.A.; Darwish, E.S.; Khedr, E.M.; El Serogy, Y.M.; Ali, A.M. Effects of low versus high frequencies of repetitive transcranial magnetic stimulation on cognitive function and cortical excitability in Alzheimer’s dementia. J. Neurol. 2012, 259, 83–92. [Google Scholar] [CrossRef]
- Cotelli, M.; Manenti, R.; Cappa, S.F.; Geroldi, C.; Zanetti, O.; Rossini, P.M.; Miniussi, C. Effect of transcranial magnetic stimulation on action naming in patients with Alzheimer disease. Arch. Neurol. 2006, 63, 1602–1604. [Google Scholar] [CrossRef]
- Cotelli, M.; Manenti, R.; Cappa, S.F.; Zanetti, O.; Miniussi, C. Transcranial magnetic stimulation improves naming in Alzheimer disease patients at different stages of cognitive decline. Eur. J. Neurol. 2008, 15, 1286–1292. [Google Scholar] [CrossRef]
- André, S.; Heinrich, S.; Kayser, F.; Menzler, K.; Kesselring, J.; Khader, P.H.; Lefaucheur, J.P.; Mylius, V. At-home tDCS of the left dorsolateral prefrontal cortex improves visual short-term memory in mild vascular dementia. J. Neurol. Sci. 2016, 369, 185–190. [Google Scholar] [CrossRef]
- Strafella, A.P.; Paus, T.; Barrett, J.; Dagher, A. Repetitive transcranial magnetic stimulation of the human prefrontal cortex induces dopamine release in the caudate nucleus. J. Neurosci. 2001, 21. [Google Scholar] [CrossRef]
- Sun, L.; Cao, Q.; Long, X.; Sui, M.; Cao, X.; Zhu, C.; Zuo, X.; An, L.; Song, Y.; Zang, Y.; et al. Abnormal functional connectivity between the anterior cingulate and the default mode network in drug-naïve boys with attention deficit hyperactivity disorder. Psychiatry Res. Neuroimaging 2012, 201, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Utevsky, A.V.; Smith, D.V.; Huettel, S.A. Precuneus is a functional core of the default-mode network. J. Neurosci. 2014, 34, 932–940. [Google Scholar] [CrossRef] [PubMed]
- Broyd, S.J.; Demanuele, C.; Debener, S.; Helps, S.K.; James, C.J.; Sonuga-Barke, E.J.S. Default-mode brain dysfunction in mental disorders: A systematic review. Neurosci. Biobehav. Rev. 2009, 33, 279–296. [Google Scholar] [CrossRef] [PubMed]
- Buckner, R.L.; Snyder, A.Z.; Shannon, B.J.; LaRossa, G.; Sachs, R.; Fotenos, A.F.; Sheline, Y.I.; Klunk, W.E.; Mathis, C.A.; Morris, J.C.; et al. Molecular, structural, and functional characterization of Alzheimer’s disease: Evidence for a relationship between default activity, amyloid, and memory. J. Neurosci. 2005, 25, 7709–7717. [Google Scholar] [CrossRef]
- Wang, L.; Zang, Y.; He, Y.; Liang, M.; Zhang, X.; Tian, L.; Wu, T.; Jiang, T.; Li, K. Changes in hippocampal connectivity in the early stages of Alzheimer’s disease: Evidence from resting state fMRI. Neuroimage 2006, 31, 496–504. [Google Scholar] [CrossRef]
- Adriaanse, S.M.; Binnewijzend, M.A.A.; Ossenkoppele, R.; Tijms, B.M.; Van Der Flier, W.M.; Koene, T.; Smits, L.L.; Wink, A.M.; Scheltens, P.; Van Berckel, B.N.M.; et al. Widespread disruption of functional brain organization in early-onset Alzheimer’s disease. PLoS ONE 2014, 9. [Google Scholar] [CrossRef]
- Song, J.; Qin, W.; Liu, Y.; Duan, Y.; Liu, J.; He, X.; Li, K.; Zhang, X.; Jiang, T.; Yu, C. Aberrant Functional Organization within and between Resting-State Networks in AD. PLoS ONE 2013, 8. [Google Scholar] [CrossRef]
- Rombouts, S.A.R.B.; Damoiseaux, J.S.; Goekoop, R.; Barkhof, F.; Scheltens, P.; Smith, S.M.; Beckmann, C.F. Model-free group analysis shows altered BOLD FMRI networks in dementia. Hum. Brain Mapp. 2009, 30, 256–266. [Google Scholar] [CrossRef]
- Galton, C.J.; Patterson, K.; Graham, K.; Lambon-Ralph, M.A.; Williams, G.; Antoun, N.; Sahakian, B.J.; Hodges, J.R. Differing patterns of temporal atrophy in Alzheimer’s disease and semantic dementia. Neurology 2001, 57, 216–225. [Google Scholar] [CrossRef]
- Liu, W.; Miller, B.L.; Kramer, J.H.; Rankin, K.; Wyss-Coray, C.; Gearhart, R.; Phengrasamy, L.; Weiner, M.; Rosen, H.J. Behavioral disorders in the frontal and temporal variants of frontotemporal dementia. Neurology 2004, 62, 742–748. [Google Scholar] [CrossRef]
- Landin-Romero, R.; Tan, R.; Hodges, J.R.; Kumfor, F. An update on semantic dementia: Genetics, imaging, and pathology. Alzheimer’s Res. Ther. 2016, 8, 52. [Google Scholar]
- Rottschy, C.; Langner, R.; Dogan, I.; Reetz, K.; Laird, A.R.; Schulz, J.B.; Fox, P.T.; Eickhoff, S.B. Modelling neural correlates of working memory: A coordinate-based meta-analysis. Neuroimage 2012, 60, 830–846. [Google Scholar] [CrossRef] [PubMed]
- Cañas, A.; Juncadella, M.; Lau, R.; Gabarrós, A.; Hernández, M. Working memory deficits after lesions involving the supplementary motor area. Front. Psychol. 2018, 9, 765. [Google Scholar] [CrossRef] [PubMed]
| Cluster ID | Cluster Size | Peak T | Peak Coordinates | Brain Regions | ||
|---|---|---|---|---|---|---|
| x | y | z | ||||
| 1 | 71 | 6.32 | −46 | 4 | −42 | L MTG/ITG |
| 2 | 30 | 4.79 | −46 | 10 | −26 | L STG/MTG/STP/MTP |
| 3 | 107 | 7.85 | −56 | −10 | −18 | L MTG |
| 4 | 47 | 7.09 | 46 | 10 | −24 | R STG/MTG/STP |
| 5 | 106 | 6.32 | −38 | 22 | −4 | L IFG/OrbIFG |
| 6 | 37 | 5.56 | −44 | −58 | −12 | L IOG |
| 7 | 33 | 5.56 | 34 | 24 | −10 | R IFG/OrbIFG |
| 8 | 44 | 5.56 | −50 | 34 | −4 | L IFG/OrbIFG |
| 9 | 49 | 6.32 | 4 | 50 | 0 | R rPFC/SupMFG |
| 10 | 292 | 7.09 | −46 | 32 | 16 | L dlPFC/MFG/IFG/TriIFG |
| 11 | 57 | 6.32 | 10 | 12 | 46 | R SMA |
| 12 | 35 | 6.32 | 26 | −64 | 50 | R SPL/PCu |
| rsFC | Brain Regions | Cluster | Peak | MNI Coordinates | ||
|---|---|---|---|---|---|---|
| Size | T | x | y | z | ||
| Positive | L STG/MTG/TMP | 8175 | 15.56 | −62 | −2 | −2 |
| R PCu | 2098 | 9.58 | 6 | −54 | 28 | |
| L SupMFG/dlPFC | 2744 | 14.66 | −2 | 48 | −14 | |
| R STG/TMP/operculum | 6228 | 11.86 | 56 | −2 | −12 | |
| R PreCG/SMA/PoCG | 1483 | 8.39 | 2 | −16 | 54 | |
| L SFG | 145 | 6.23 | −18 | 32 | 48 | |
| L AG | 282 | 5.34 | −46 | −70 | 30 | |
| R AG | 132 | 5.26 | 48 | −60 | 26 | |
| L IOG | 160 | 5.24 | −22 | −88 | −4 | |
| R IOG | 43 | 4.63 | 50 | −76 | 4 | |
| Negative | R SPL/AG | 1922 | 14.08 | 46 | −42 | 40 |
| R mPFC/dlPFC | 4098 | 12.70 | 38 | 34 | 38 | |
| L SPL/SMG | 1669 | 9.00 | −36 | −50 | 48 | |
| L mPFC/dlPFC | 2542 | 7.99 | −36 | 30 | 30 | |
| L SPL/cuneus | 1879 | 7.40 | −8 | −66 | 50 | |
| R SFG | 65 | 5.78 | 6 | 34 | 38 | |
| L SFG | 283 | 5.72 | −24 | 8 | 68 | |
| R operculum | 99 | 5.65 | 50 | 12 | 10 | |
| rsFC | Brain Regions | Cluster | Peak | MNI Coordinates | ||
|---|---|---|---|---|---|---|
| Size | T | x | y | z | ||
| Positive | R MTG/STG/TMP | 5785 | 12.22 | 56 | −4 | −26 |
| R PCu | 2182 | 11.90 | 14 | −36 | 4 | |
| L STG/TMP/MTG | 4615 | 10.63 | −60 | −2 | −4 | |
| R SFG/SupMFG/mPFC | 1503 | 9.88 | 4 | 34 | −10 | |
| L ITG | 177 | 7.39 | −34 | −36 | −14 | |
| R ITG/AG/MTG/MOG/IOG | 1100 | 7.03 | 50 | −46 | −28 | |
| L PreCG | 60 | 6.21 | −36 | −20 | 72 | |
| R SFG | 98 | 6.05 | 20 | 36 | 48 | |
| L AG/MOG | 133 | 5.57 | −40 | −66 | 22 | |
| L ITG | 53 | 4.99 | −48 | −58 | −14 | |
| R PreCG | 85 | 4.76 | 38 | −12 | 68 | |
| R SFG | 56 | 4.67 | 4 | 52 | 20 | |
| L SFG | 61 | 4.52 | −12 | 58 | 10 | |
| L SOG | 49 | 4.40 | −20 | −90 | 12 | |
| L PreCG | 70 | 4.39 | −6 | −26 | 62 | |
| R PreCG | 48 | 4.30 | 12 | −18 | 78 | |
| Negative | R MFG/mPFC/dlPFC | 2525 | 14.03 | 34 | 54 | 22 |
| L SMG/SPL/AG | 1759 | 12.14 | −48 | −46 | 40 | |
| L MFG | 3690 | 9.75 | −30 | 48 | −2 | |
| R SMG/AG | 1374 | 9.08 | 50 | −42 | 38 | |
| R SFG/SMA/MFG | 1263 | 8.60 | 16 | 20 | 60 | |
| L PCu | 190 | 6.11 | −2 | −52 | 70 | |
| R cuneus | 135 | 4.73 | 8 | −80 | 28 | |
| L SPL/PoCG | 42 | 4.33 | −20 | −56 | 72 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cao, J.; Huang, Y.; Meshberg, N.; Hodges, S.A.; Kong, J. Neuroimaging-Based Scalp Acupuncture Locations for Dementia. J. Clin. Med. 2020, 9, 2477. https://doi.org/10.3390/jcm9082477
Cao J, Huang Y, Meshberg N, Hodges SA, Kong J. Neuroimaging-Based Scalp Acupuncture Locations for Dementia. Journal of Clinical Medicine. 2020; 9(8):2477. https://doi.org/10.3390/jcm9082477
Chicago/Turabian StyleCao, Jin, Yiting Huang, Nathaniel Meshberg, Sierra A. Hodges, and Jian Kong. 2020. "Neuroimaging-Based Scalp Acupuncture Locations for Dementia" Journal of Clinical Medicine 9, no. 8: 2477. https://doi.org/10.3390/jcm9082477
APA StyleCao, J., Huang, Y., Meshberg, N., Hodges, S. A., & Kong, J. (2020). Neuroimaging-Based Scalp Acupuncture Locations for Dementia. Journal of Clinical Medicine, 9(8), 2477. https://doi.org/10.3390/jcm9082477





