The Treatment of Cognitive, Behavioural and Motor Impairments from Brain Injury and Neurodegenerative Diseases through Cannabinoid System Modulation—Evidence from In Vivo Studies
Abstract
1. Introduction
1.1. Endocannabinoids
1.2. Phytocannabinoids
1.3. Synthetic Cannabinoids
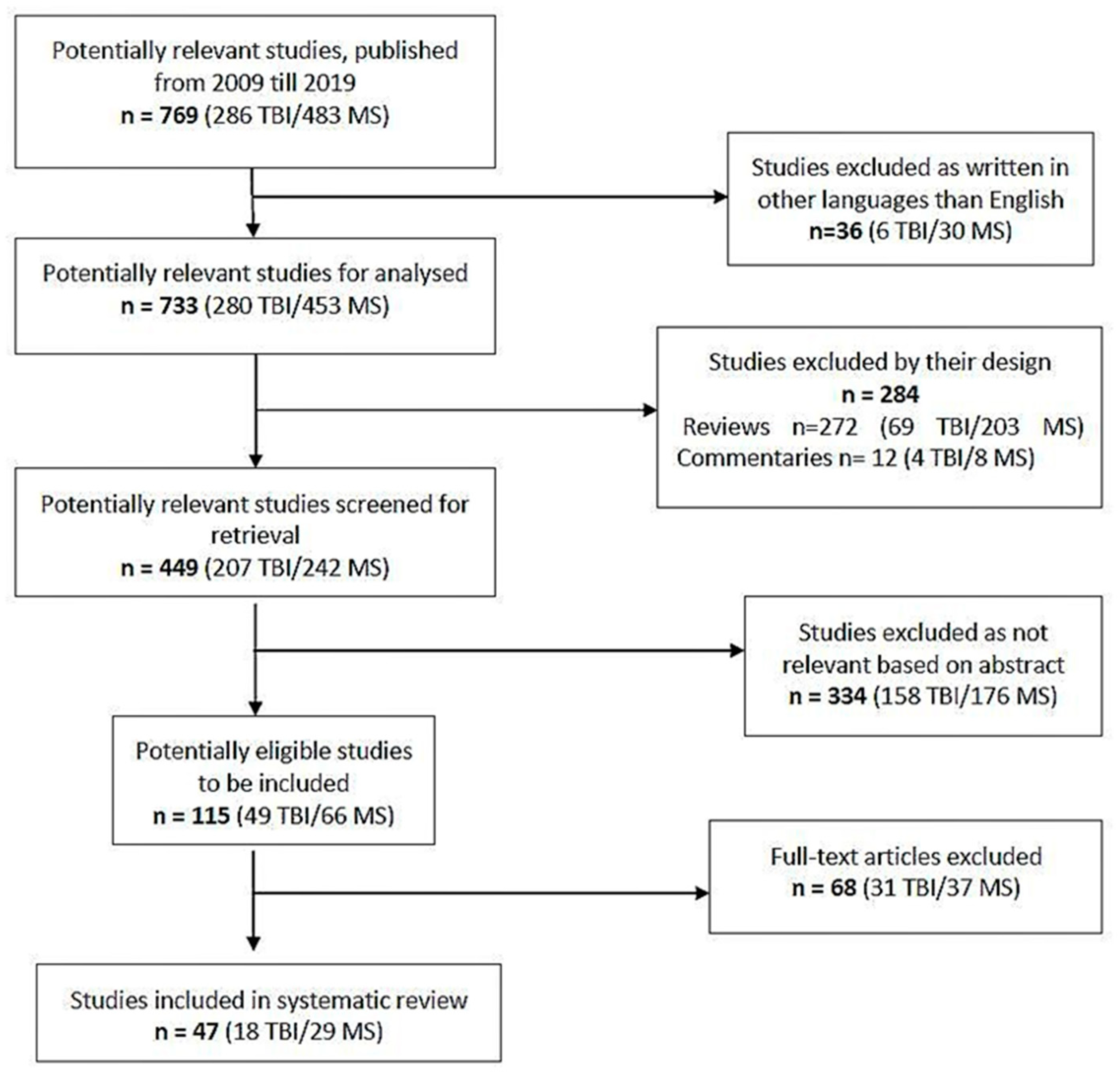
2. Methodology
3. Results
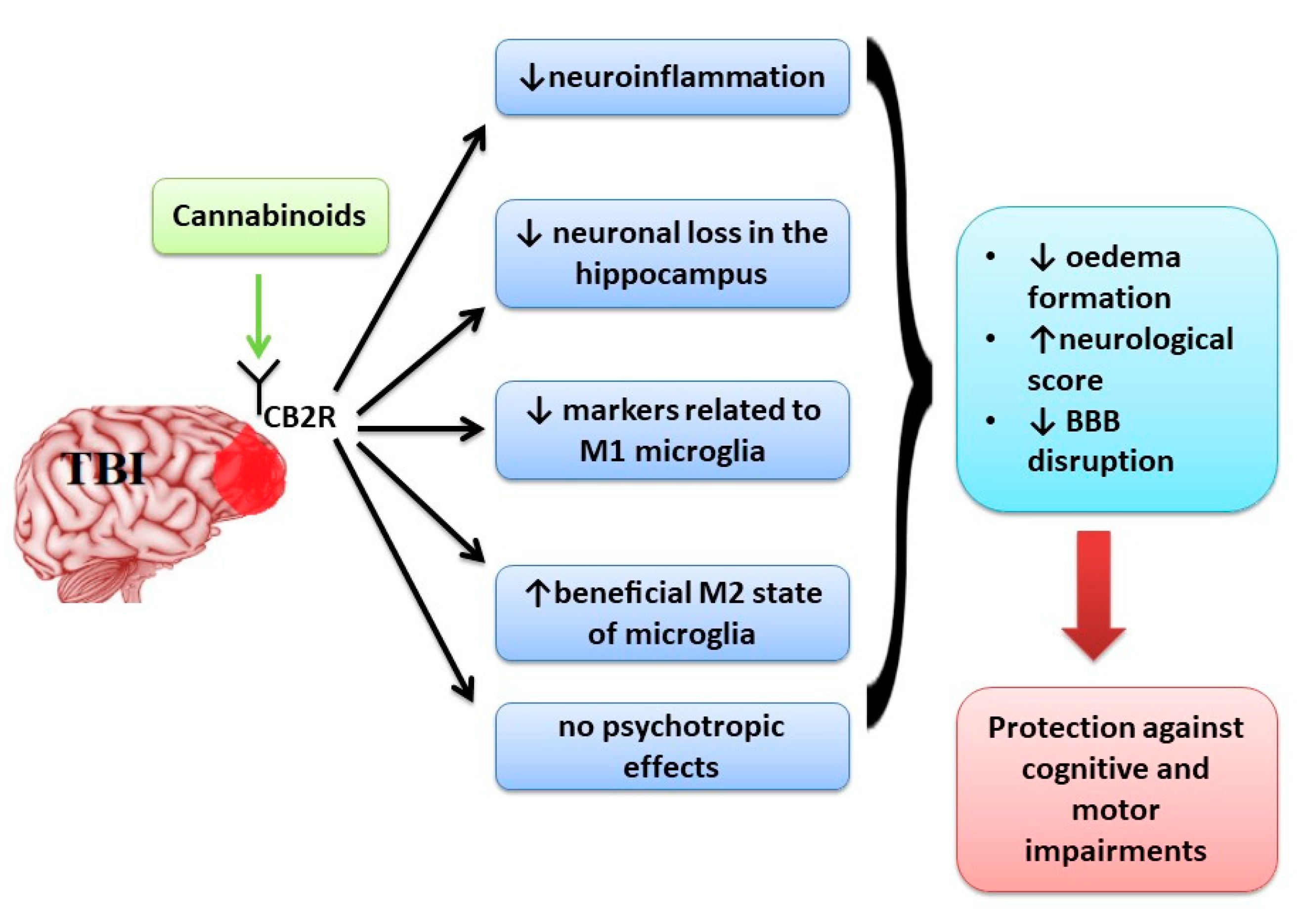
3.1. Cannabinoids Effects in Cognitive and Motor Impairment in Traumatic Brain Injury (TBI)
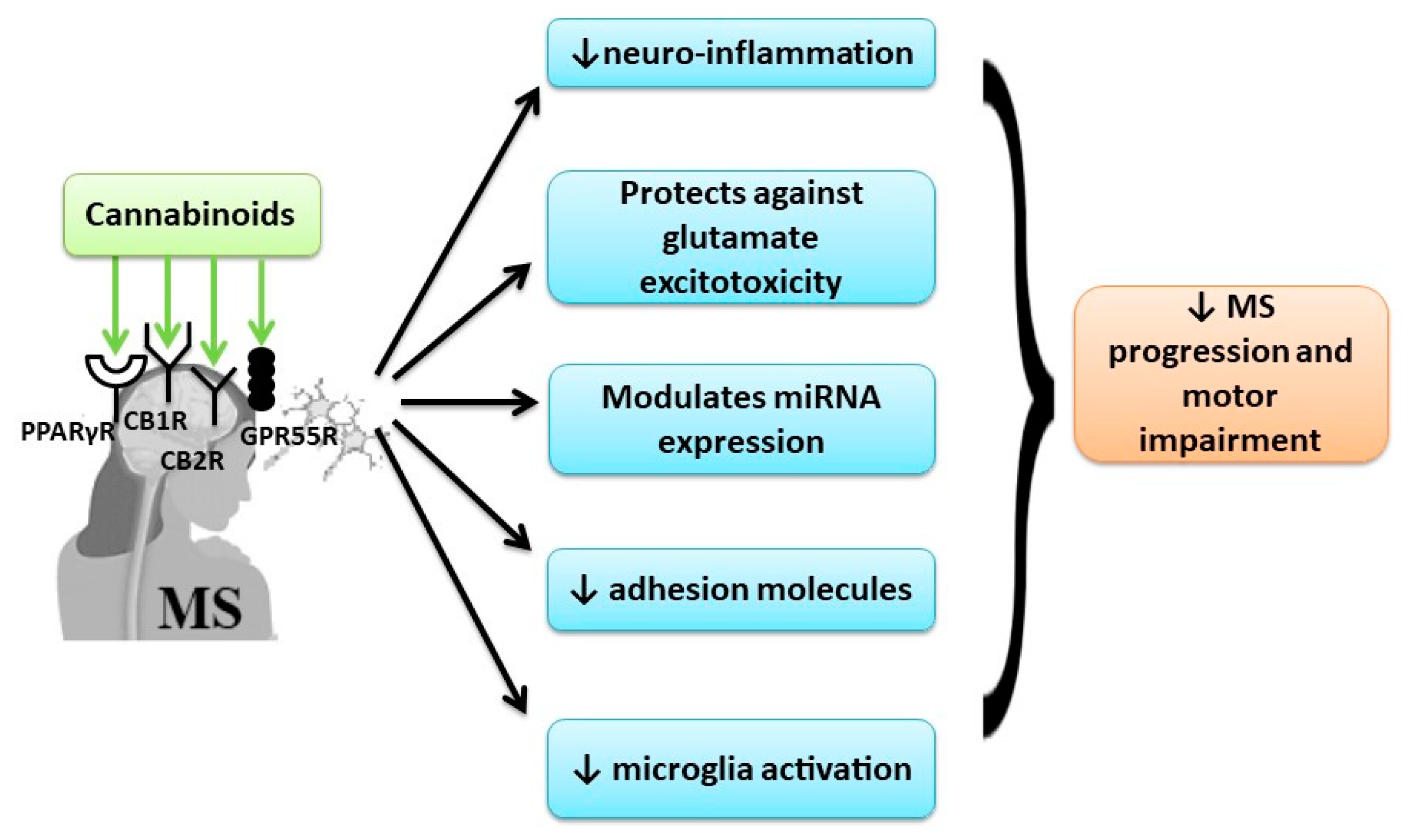
3.2. Cannabinoids Effects in Cognitive and Motor Impairment in Multiple Sclerosis (MS)
4. Discussion
4.1. Cannabinoids Effects in Cognitive and Motor Impairment in Traumatic Brain Injury (TBI)
4.2. Cannabinoids Effects in Cognitive and Motor Impairment in Multiple Sclerosis (MS)
4.3. Limitations and Strength of the Study
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Reiner, A.; Heldt, S.A.; Presley, C.S.; Guley, N.H.; Elberger, A.J.; Deng, Y.; D’Surney, L.; Rogers, J.T.; Ferrell, J.; Bu, W.; et al. Motor, visual and emotional deficits in mice after closed-head mild traumatic brain injury are alleviated by the novel CB2 inverse agonist SMM-189. Int. J. Mol. Sci. 2014, 16, 758–787. [Google Scholar] [CrossRef] [PubMed]
- Hallikainen, I.; Hongisto, K.; Välimäki, T.; Hänninen, T.; Martikainen, J.; Koivisto, A.M. The Progression of Neuropsychiatric Symptoms in Alzheimer’s Disease during a Five-Year Follow-Up: Kuopio ALSOVA Study. J. Alzheimer’s Dis. Jad 2018, 61, 1367–1376. [Google Scholar] [CrossRef] [PubMed]
- Hodges, J.R.; Piguet, O. Progress and Challenges in Frontotemporal Dementia Research: A 20-Year Review. J. Alzheimer’s Dis. Jad 2018, 62, 1467–1480. [Google Scholar] [CrossRef]
- Douven, E.; Aalten, P.; Staals, J.; Schievink, S.H.J.; van Oostenbrugge, R.J.; Verhey, F.R.J.; Köhler, S. Co-occurrence of depressive symptoms and executive dysfunction after stroke: Associations with brain pathology and prognosis. J. Neurol. Neurosurg. Psychiatry 2018, 89, 859–865. [Google Scholar] [CrossRef] [PubMed]
- Kokiko-Cochran, O.N.; Godbout, J.P. The Inflammatory Continuum of Traumatic Brain Injury and Alzheimer’s Disease. Front. Immunol. 2018, 9, 672. [Google Scholar] [CrossRef]
- Montero-Odasso, M.; Verghese, J.; Beauchet, O.; Hausdorff, J.M. Gait and cognition: A complementary approach to understanding brain function and the risk of falling. J Am Geriatr Soc 2012, 60, 2127–2136. [Google Scholar] [CrossRef]
- Montero-Odasso, M.; Bergman, H.; Phillips, N.A.; Wong, C.H.; Sourial, N.; Chertkow, H. Dual-tasking and gait in people with mild cognitive impairment. The effect of working memory. BMC Geriatr. 2009, 9, 41. [Google Scholar] [CrossRef]
- Ricciardi, L.; Bloem, B.R.; Snijders, A.H.; Daniele, A.; Quaranta, D.; Bentivoglio, A.R.; Fasano, A. Freezing of gait in Parkinson’s disease: The paradoxical interplay between gait and cognition. Parkinsonism Relat. Disord. 2014, 20, 824–829. [Google Scholar] [CrossRef]
- Aloizou, A.M.; Siokas, V.; Vogiatzi, C.; Peristeri, E.; Docea, A.O.; Petrakis, D.; Provatas, A.; Folia, V.; Chalkia, C.; Vinceti, M.; et al. Pesticides, cognitive functions and dementia: A review. Toxicol. Lett. 2020, 326, 31–51. [Google Scholar] [CrossRef]
- Sergievich, A.A.; Khoroshikh, P.P.; Artemenko, A.F.; Zakharenko, A.M.; Chaika, V.V.; Kodintsev, V.V.; Stroeva, O.A.; Lenda, E.G.; Tsatsakis, A.; Burykina, T.I.; et al. Behavioral impacts of a mixture of six pesticides on rats. Sci. Total Environ. 2020, 727, 138491. [Google Scholar] [CrossRef]
- Tsatsakis, A.; Docea, A.O.; Calina, D.; Tsarouhas, K.; Zamfira, L.M.; Mitrut, R.; Sharifi-Rad, J.; Kovatsi, L.; Siokas, V.; Dardiotis, E.; et al. A Mechanistic and Pathophysiological Approach for Stroke Associated with Drugs of Abuse. J. Clin. Med. 2019, 8, 1295. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Pan, W. The Treatment Strategies for Neurodegenerative Diseases by Integrative Medicine. Integr. Med. Int. 2014, 1, 223–225. [Google Scholar] [CrossRef]
- Sharifi-Rad, M.; Lankatillake, C.; Dias, D.A.; Docea, A.O.; Mahomoodally, M.F.; Lobine, D.; Chazot, P.L.; Kurt, B.; Tumer, T.B.; Moreira, A.C.; et al. Impact of Natural Compounds on Neurodegenerative Disorders: From Preclinical to Pharmacotherapeutics. J. Clin. Med. 2020, 9, 1061. [Google Scholar] [CrossRef] [PubMed]
- Salehi, B.; Calina, D.; Docea, A.O.; Koirala, N.; Aryal, S.; Lombardo, D.; Pasqua, L.; Taheri, Y.; Marina Salgado Castillo, C.; Martorell, M.; et al. Curcumin’s Nanomedicine Formulations for Therapeutic Application in Neurological Diseases. J. Clin. Med. 2020, 9, 430. [Google Scholar] [CrossRef]
- Lu, H.C.; Mackie, K. An Introduction to the Endogenous Cannabinoid System. Biol. Psychiatry 2016, 79, 516–525. [Google Scholar] [CrossRef]
- Galiègue, S.; Mary, S.; Marchand, J.; Dussossoy, D.; Carrière, D.; Carayon, P.; Bouaboula, M.; Shire, D.; LE Fur, G.; Casellas, P. Expression of Central and Peripheral Cannabinoid Receptors in Human Immune Tissues and Leukocyte Subpopulations. Eur. J. Biochem. 1995, 232, 54–61. [Google Scholar] [CrossRef]
- Lanciego, J.L.; Barroso-Chinea, P.; Rico, A.J.; Conte-Perales, L.; Callén, L.; Roda, E.; Gómez-Bautista, V.; López, I.P.; Lluis, C.; Labandeira-García, J.L.; et al. Expression of the mRNA coding the cannabinoid receptor 2 in the pallidal complex of Macaca fascicularis. J. Psychopharmacol. (Oxf. Engl.) 2011, 25, 97–104. [Google Scholar] [CrossRef]
- Li, Y.; Kim, J. Neuronal expression of CB2 cannabinoid receptor mRNAs in the mouse hippocampus. Neuroscience 2015, 311, 253–267. [Google Scholar] [CrossRef]
- García-Gutiérrez, M.S.; García-Bueno, B.; Zoppi, S.; Leza, J.C.; Manzanares, J. Chronic blockade of cannabinoid CB2 receptors induces anxiolytic-like actions associated with alterations in GABA(A) receptors. Br. J. Pharmacol. 2012, 165, 951–964. [Google Scholar] [CrossRef]
- Stempel, A.V.; Stumpf, A.; Zhang, H.Y.; Özdoğan, T.; Pannasch, U.; Theis, A.K.; Otte, D.M.; Wojtalla, A.; Rácz, I.; Ponomarenko, A.; et al. Cannabinoid Type 2 Receptors Mediate a Cell Type-Specific Plasticity in the Hippocampus. Neuron 2016, 90, 795–809. [Google Scholar] [CrossRef]
- Viscomi, M.T.; Oddi, S.; Latini, L.; Pasquariello, N.; Florenzano, F.; Bernardi, G.; Molinari, M.; Maccarrone, M. Selective CB2 receptor agonism protects central neurons from remote axotomy-induced apoptosis through the PI3K/Akt pathway. J. Neurosci. Off. J. Soc. Neurosci. 2009, 29, 4564–4570. [Google Scholar] [CrossRef] [PubMed]
- Cabral, G.A.; Griffin-Thomas, L. Emerging role of the cannabinoid receptor CB2 in immune regulation: Therapeutic prospects for neuroinflammation. Expert Rev. Mol. Med. 2009, 11, e3. [Google Scholar] [CrossRef] [PubMed]
- Sánchez, A.J.; García-Merino, A. Neuroprotective agents: Cannabinoids. Clin. Immunol. (Orlando Fla.) 2012, 142, 57–67. [Google Scholar] [CrossRef] [PubMed]
- Klein, T.W.; Newton, C.; Larsen, K.; Lu, L.; Perkins, I.; Nong, L.; Friedman, H. The cannabinoid system and immune modulation. J. Leukoc. Biol. 2003, 74, 486–496. [Google Scholar] [CrossRef]
- Zhang, H.Y.; Gao, M.; Liu, Q.R.; Bi, G.H.; Li, X.; Yang, H.J.; Gardner, E.L.; Wu, J.; Xi, Z.X. Cannabinoid CB2 receptors modulate midbrain dopamine neuronal activity and dopamine-related behavior in mice. Proc. Natl. Acad. Sci. USA 2014, 111, E5007–E5015. [Google Scholar] [CrossRef]
- Laezza, C.; Pagano, C.; Navarra, G.; Pastorino, O.; Proto, M.C.; Fiore, D.; Piscopo, C.; Gazzerro, P.; Bifulco, M. The Endocannabinoid System: A Target for Cancer Treatment. Int. J. Mol. Sci. 2020, 21, 747. [Google Scholar] [CrossRef]
- Scheau, C.; Badarau, I.A.; Mihai, L.-G.; Scheau, A.-E.; Costache, D.O.; Constantin, C.; Calina, D.; Caruntu, C.; Costache, R.S.; Caruntu, A. Cannabinoids in the Pathophysiology of Skin Inflammation. Molecules 2020, 25, 652. [Google Scholar] [CrossRef]
- Muller, C.; Morales, P.; Reggio, P.H. Cannabinoid Ligands Targeting TRP Channels. Front. Mol. Neurosci. 2018, 11, 487. [Google Scholar] [CrossRef]
- Pertwee, R.G.; Howlett, A.C.; Abood, M.E.; Alexander, S.P.H.; Di Marzo, V.; Elphick, M.R.; Greasley, P.J.; Hansen, H.S.; Kunos, G.; Mackie, K.; et al. International Union of Basic and Clinical Pharmacology. LXXIX. Cannabinoid receptors and their ligands: Beyond CB₁ and CB₂. Pharm. Rev. 2010, 62, 588–631. [Google Scholar] [CrossRef]
- Maurya, N.; Velmurugan, B.K. Therapeutic applications of cannabinoids. Chem. Biol. Interact. 2018, 293, 77–88. [Google Scholar] [CrossRef]
- Fezza, F.; Bisogno, T.; Minassi, A.; Appendino, G.; Mechoulam, R.; Di Marzo, V. Noladin ether, a putative novel endocannabinoid: Inactivation mechanisms and a sensitive method for its quantification in rat tissues. FEBS Lett. 2002, 513, 294–298. [Google Scholar] [CrossRef]
- Porter, A.C.; Sauer, J.M.; Knierman, M.D.; Becker, G.W.; Berna, M.J.; Bao, J.; Nomikos, G.G.; Carter, P.; Bymaster, F.P.; Leese, A.B.; et al. Characterization of a novel endocannabinoid, virodhamine, with antagonist activity at the CB1 receptor. J. Pharmacol. Exp. Ther. 2002, 301, 1020–1024. [Google Scholar] [CrossRef] [PubMed]
- Grabiec, U.; Dehghani, F. N-Arachidonoyl Dopamine: A Novel Endocannabinoid and Endovanilloid with Widespread Physiological and Pharmacological Activities. Cannabis Cannabinoid Res. 2017, 2, 183–196. [Google Scholar] [CrossRef]
- Zou, S.; Kumar, U. Cannabinoid Receptors and the Endocannabinoid System: Signaling and Function in the Central Nervous System. Int. J. Mol. Sci. 2018, 19, 833. [Google Scholar]
- Cifelli, P.; Ruffolo, G.; De Felice, E.; Alfano, V.; van Vliet, E.A.; Aronica, E.; Palma, E. Phytocannabinoids in Neurological Diseases: Could They Restore a Physiological GABAergic Transmission? Int. J. Mol. Sci. 2020, 21, 723. [Google Scholar] [CrossRef] [PubMed]
- Niesink, R.J.M.; van Laar, M.W. Does Cannabidiol Protect against Adverse Psychological Effects of THC? Front. Psychiatry 2013, 4, 130. [Google Scholar] [CrossRef]
- Bolognini, D.; Rock, E.M.; Cluny, N.L.; Cascio, M.G.; Limebeer, C.L.; Duncan, M.; Stott, C.G.; Javid, F.A.; Parker, L.A.; Pertwee, R.G. Cannabidiolic acid prevents vomiting in Suncus murinus and nausea-induced behaviour in rats by enhancing 5-HT1A receptor activation. Br. J. Pharmacol. 2013, 168, 1456–1470. [Google Scholar] [CrossRef]
- Morales, P.; Hurst, D.P.; Reggio, P.H. Molecular Targets of the Phytocannabinoids: A Complex Picture. Prog. Chem. Org. Nat. Prod. 2017, 103, 103–131. [Google Scholar]
- Izzo, A.A.; Capasso, R.; Aviello, G.; Borrelli, F.; Romano, B.; Piscitelli, F.; Gallo, L.; Capasso, F.; Orlando, P.; Di Marzo, V. Inhibitory effect of cannabichromene, a major non-psychotropic cannabinoid extracted from Cannabis sativa, on inflammation-induced hypermotility in mice. Br. J. Pharmacol. 2012, 166, 1444–1460. [Google Scholar] [CrossRef]
- Miliano, C.; Serpelloni, G.; Rimondo, C.; Mereu, M.; Marti, M.; De Luca, M.A. Neuropharmacology of New Psychoactive Substances (NPS): Focus on the Rewarding and Reinforcing Properties of Cannabimimetics and Amphetamine-Like Stimulants. Front. Neurosci. 2016, 10, 153. [Google Scholar]
- Bilici, R. Synthetic cannabinoids. North. Clin. Istanb. 2014, 1, 121–126. [Google Scholar] [CrossRef]
- Lassmann, H.; Bradl, M. Multiple sclerosis: Experimental models and reality. Acta Neuropathol. 2017, 133, 223–244. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
- Magid, L.; Heymann, S.; Elgali, M.; Avram, L.; Cohen, Y.; Liraz-Zaltsman, S.; Mechoulam, R.; Shohami, E. Role of CB(2) Receptor in the Recovery of Mice after Traumatic Brain Injury. J. Neurotrauma 2019, 36, 1836–1846. [Google Scholar] [CrossRef]
- Wang, Z.; Li, Y.; Cai, S.; Li, R.; Cao, G. Cannabinoid receptor 2 agonist attenuates blood-brain barrier damage in a rat model of intracerebral hemorrhage by activating the Rac1 pathway. Int. J. Mol. Med. 2018, 42, 2914–2922. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Yun, D.; Zhang, Y.; Tao, Y.; Tan, Q.; Qiao, F.; Luo, B.; Liu, Y.; Fan, R.; Xian, J.; et al. A cannabinoid receptor 2 agonist reduces blood-brain barrier damage via induction of MKP-1 after intracerebral hemorrhage in rats. Brain Res. 2018, 1697, 113–123. [Google Scholar] [CrossRef] [PubMed]
- Braun, M.; Khan, Z.T.; Khan, M.B.; Kumar, M.; Ward, A.; Achyut, B.R.; Arbab, A.S.; Hess, D.C.; Hoda, M.N.; Baban, B.; et al. Selective activation of cannabinoid receptor-2 reduces neuroinflammation after traumatic brain injury via alternative macrophage polarization. BrainBehav. Immun. 2018, 68, 224–237. [Google Scholar] [CrossRef]
- Mori, M.A.; Meyer, E.; Soares, L.M.; Milani, H.; Guimarães, F.S.; de Oliveira, R.M.W. Cannabidiol reduces neuroinflammation and promotes neuroplasticity and functional recovery after brain ischemia. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2017, 75, 94–105. [Google Scholar] [CrossRef]
- Liu, Y.; McAfee, S.S.; Guley, N.M.; Del Mar, N.; Bu, W.; Heldt, S.A.; Honig, M.G.; Moore II, B.M.; Reiner, A. Abnormalities in Dynamic Brain Activity Caused by Mild Traumatic Brain Injury Are Partially Rescued by the Cannabinoid Type-2 Receptor Inverse Agonist SMM-189. Eneuro 2017, 4. [Google Scholar] [CrossRef]
- Nissinen, J.; Andrade, P.; Natunen, T.; Hiltunen, M.; Malm, T.; Kanninen, K.; Soares, J.I.; Shatillo, O.; Sallinen, J.; Ndode-Ekane, X.E.; et al. Disease-modifying effect of atipamezole in a model of post-traumatic epilepsy. Epilepsy Res. 2017, 136, 18–34. [Google Scholar] [CrossRef]
- Bu, W.; Ren, H.; Deng, Y.; Del Mar, N.; Guley, N.M.; Moore, B.M.; Honig, M.G.; Reiner, A. Mild Traumatic Brain Injury Produces Neuron Loss That Can Be Rescued by Modulating Microglial Activation Using a CB2 Receptor Inverse Agonist. Front. Neurosci. 2016, 10, 449. [Google Scholar] [CrossRef] [PubMed]
- Arain, M.; Khan, M.; Craig, L.; Nakanishi, S.T. Cannabinoid agonist rescues learning and memory after a traumatic brain injury. Ann. Clin. Transl. Neurol. 2015, 2, 289–294. [Google Scholar] [CrossRef] [PubMed]
- Tchantchou, F.; Tucker, L.B.; Fu, A.H.; Bluett, R.J.; McCabe, J.T.; Patel, S.; Zhang, Y. The fatty acid amide hydrolase inhibitor PF-3845 promotes neuronal survival, attenuates inflammation and improves functional recovery in mice with traumatic brain injury. Neuropharmacology 2014, 85, 427–439. [Google Scholar] [CrossRef]
- Fujii, M.; Sherchan, P.; Krafft, P.R.; Rolland, W.B.; Soejima, Y.; Zhang, J.H. Cannabinoid type 2 receptor stimulation attenuates brain edema by reducing cerebral leukocyte infiltration following subarachnoid hemorrhage in rats. J. Neurol. Sci. 2014, 342, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Tchantchou, F.; Zhang, Y. Selective inhibition of alpha/beta-hydrolase domain 6 attenuates neurodegeneration, alleviates blood brain barrier breakdown, and improves functional recovery in a mouse model of traumatic brain injury. J. Neurotrauma 2013, 30, 565–579. [Google Scholar] [CrossRef] [PubMed]
- Cohen-Yeshurun, A.; Willner, D.; Trembovler, V.; Alexandrovich, A.; Mechoulam, R.; Shohami, E.; Leker, R.R. N-arachidonoyl-L-serine (AraS) possesses proneurogenic properties in vitro and in vivo after traumatic brain injury. J. Cereb. Blood Flow Metab. Off. J. Int. Soc. Cereb. Blood Flow Metab. 2013, 33, 1242–1250. [Google Scholar] [CrossRef]
- Amenta, P.S.; Jallo, J.I.; Tuma, R.F.; Elliott, M.B. A cannabinoid type 2 receptor agonist attenuates blood-brain barrier damage and neurodegeneration in a murine model of traumatic brain injury. J. Neurosci. Res. 2012, 90, 2293–2305. [Google Scholar] [CrossRef]
- Pazos, M.R.; Cinquina, V.; Gómez, A.; Layunta, R.; Santos, M.; Fernández-Ruiz, J.; Martínez-Orgado, J. Cannabidiol administration after hypoxia-ischemia to newborn rats reduces long-term brain injury and restores neurobehavioral function. Neuropharmacology 2012, 63, 776–783. [Google Scholar] [CrossRef]
- Schmidt, W.; Schäfer, F.; Striggow, V.; Fröhlich, K.; Striggow, F. Cannabinoid receptor subtypes 1 and 2 mediate long-lasting neuroprotection and improve motor behavior deficits after transient focal cerebral ischemia. Neuroscience 2012, 227, 313–326. [Google Scholar] [CrossRef]
- Elliott, M.B.; Tuma, R.F.; Amenta, P.S.; Barbe, M.F.; Jallo, J.I. Acute effects of a selective cannabinoid-2 receptor agonist on neuroinflammation in a model of traumatic brain injury. J. Neurotrauma 2011, 28, 973–981. [Google Scholar] [CrossRef] [PubMed]
- Al-Ghezi, Z.Z.; Miranda, K.; Nagarkatti, M.; Nagarkatti, P.S. Combination of Cannabinoids, Δ9- Tetrahydrocannabinol and Cannabidiol, Ameliorates Experimental Multiple Sclerosis by Suppressing Neuroinflammation Through Regulation of miRNA-Mediated Signaling Pathways. Front. Immunol. 2019, 10, 1921. [Google Scholar] [CrossRef] [PubMed]
- Zhou, T.; Ahmad, T.K.; Alrushaid, S.; Pozdirca, M.; Ethans, K.; Intrater, H.; Le, T.; Burczynski, F.; Kong, J.; Namaka, M. Therapeutic impact of orally administered cannabinoid oil extracts in an experimental autoimmune encephalomyelitis animal model of multiple sclerosis. Biochem. Biophys. Res. Commun. 2019, 516, 373–380. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Martet, M.; Feliú, A.; Espejo-Porras, F.; Mecha, M.; Carrillo-Salinas, F.J.; Fernández-Ruiz, J.; Guaza, C.; de Lago, E. The disease-modifying effects of a Sativex-like combination of phytocannabinoids in mice with experimental autoimmune encephalomyelitis are preferentially due to Δ9-tetrahydrocannabinol acting through CB1 receptors. Mult. Scler. Relat. Disord. 2015, 4, 505–511. [Google Scholar] [CrossRef] [PubMed]
- Feliú, A.; Moreno-Martet, M.; Mecha, M.; Carrillo-Salinas, F.J.; de Lago, E.; Fernández-Ruiz, J.; Guaza, C. A Sativex(®) -like combination of phytocannabinoids as a disease-modifying therapy in a viral model of multiple sclerosis. Br. J. Pharmacol. 2015, 172, 3579–3595. [Google Scholar] [CrossRef] [PubMed]
- Buccellato, E.; Carretta, D.; Utan, A.; Cavina, C.; Speroni, E.; Grassi, G.; Candeletti, S.; Romualdi, P. Acute and chronic cannabinoid extracts administration affects motor function in a CREAE model of multiple sclerosis. J. Ethnopharmacol. 2011, 133, 1033–1038. [Google Scholar] [CrossRef] [PubMed]
- Askari, V.R.; Baradaran Rahimi, V.; Tabatabaee, S.A.; Shafiee-Nick, R. Combination of Imipramine, a sphingomyelinase inhibitor, and β-caryophyllene improve their therapeutic effects on experimental autoimmune encephalomyelitis (EAE). Int. Immunopharmacol. 2019, 77, 105923. [Google Scholar] [CrossRef]
- Alberti, T.B.; Barbosa, W.L.; Vieira, J.L.; Raposo, N.R.; Dutra, R.C. (−)-β-Caryophyllene, a CB2 Receptor-Selective Phytocannabinoid, Suppresses Motor Paralysis and Neuroinflammation in a Murine Model of Multiple Sclerosis. Int. J. Mol. Sci. 2017, 18, 691. [Google Scholar] [CrossRef]
- Shi, Y.; Duan, Y.H.; Ji, Y.Y.; Wang, Z.L.; Wu, Y.R.; Gunosewoyo, H.; Xie, X.Y.; Chen, J.Z. Amidoalkylindoles as Potent and Selective Cannabinoid Type 2 Receptor Agonists with in Vivo Efficacy in a Mouse Model of Multiple Sclerosis. J. Med. Chem. 2017, 60, 7067–7083. [Google Scholar]
- Morales, P.; Gómez-Cañas, M.; Navarro, G.; Hurst, D.P.; Carrillo-Salinas, F.J.; Lagartera, L.; Pazos, R.; Goya, P.; Reggio, P.H.; Guaza, C.; et al. Chromenopyrazole, a Versatile Cannabinoid Scaffold with in Vivo Activity in a Model of Multiple Sclerosis. J. Med. Chem. 2016, 59, 6753–6771. [Google Scholar]
- Carrillo-Salinas, F.J.; Navarrete, C.; Mecha, M.; Feliú, A.; Collado, J.A.; Cantarero, I.; Bellido, M.L.; Muñoz, E.; Guaza, C. A cannabigerol derivative suppresses immune responses and protects mice from experimental autoimmune encephalomyelitis. PLoS ONE 2014, 9, e94733. [Google Scholar] [CrossRef]
- Granja, A.G.; Carrillo-Salinas, F.; Pagani, A.; Gómez-Cañas, M.; Negri, R.; Navarrete, C.; Mecha, M.; Mestre, L.; Fiebich, B.L.; Cantarero, I.; et al. A Cannabigerol Quinone Alleviates Neuroinflammation in a Chronic Model of Multiple Sclerosis. J. Neuroimmune Pharmacol. 2012, 7, 1002–1016. [Google Scholar] [CrossRef] [PubMed]
- Contarini, G.; Franceschini, D.; Facci, L.; Barbierato, M.; Giusti, P.; Zusso, M. A co-ultramicronized palmitoylethanolamide/luteolin composite mitigates clinical score and disease-relevant molecular markers in a mouse model of experimental autoimmune encephalomyelitis. J. Neuroinflamm. 2019, 16, 126. [Google Scholar] [CrossRef]
- Correa, F.; Hernangómez-Herrero, M.; Mestre, L.; Loría, F.; Docagne, F.; Guaza, C. The endocannabinoid anandamide downregulates IL-23 and IL-12 subunits in a viral model of multiple sclerosis: Evidence for a cross-talk between IL-12p70/IL-23 axis and IL-10 in microglial cells. Brain Behav. Immun. 2011, 25, 736–749. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, R.; Yu, F.; Wen, J.; Vana, A.; Zhang, Y. Therapeutic potential of a novel cannabinoid agent CB52 in the mouse model of experimental autoimmune encephalomyelitis. Neuroscience 2013, 254, 427–442. [Google Scholar] [CrossRef]
- Giacoppo, S.; Pollastro, F.; Grassi, G.; Bramanti, P.; Mazzon, E. Target regulation of PI3K/Akt/mTOR pathway by cannabidiol in treatment of experimental multiple sclerosis. Fitoterapia 2017, 116, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Giacoppo, S.; Soundara Rajan, T.; Galuppo, M.; Pollastro, F.; Grassi, G.; Bramanti, P.; Mazzon, E. Purified Cannabidiol, the main non-psychotropic component of Cannabis sativa, alone, counteracts neuronal apoptosis in experimental multiple sclerosis. Eur. Rev. Med. Pharmacol. Sci. 2015, 19, 4906–4919. [Google Scholar] [PubMed]
- Giacoppo, S.; Galuppo, M.; Pollastro, F.; Grassi, G.; Bramanti, P.; Mazzon, E. A new formulation of cannabidiol in cream shows therapeutic effects in a mouse model of experimental autoimmune encephalomyelitis. Daru J. Fac. Pharm. Tehran Univ. Med. Sci. 2015, 23, 48. [Google Scholar] [CrossRef]
- Rahimi, A.; Faizi, M.; Talebi, F.; Noorbakhsh, F.; Kahrizi, F.; Naderi, N. Interaction between the protective effects of cannabidiol and palmitoylethanolamide in experimental model of multiple sclerosis in C57BL/6 mice. Neuroscience 2015, 290, 279–287. [Google Scholar] [CrossRef]
- Mecha, M.; Feliú, A.; Iñigo, P.M.; Mestre, L.; Carrillo-Salinas, F.J.; Guaza, C. Cannabidiol provides long-lasting protection against the deleterious effects of inflammation in a viral model of multiple sclerosis: A role for A2A receptors. Neurobiol. Dis. 2013, 59, 141–150. [Google Scholar] [CrossRef]
- De Lago, E.; Moreno-Martet, M.; Cabranes, A.; Ramos, J.A.; Fernández-Ruiz, J. Cannabinoids ameliorate disease progression in a model of multiple sclerosis in mice, acting preferentially through CB1 receptor-mediated anti-inflammatory effects. Neuropharmacology 2012, 62, 2299–2308. [Google Scholar] [CrossRef] [PubMed]
- Kozela, E.; Lev, N.; Kaushansky, N.; Eilam, R.; Rimmerman, N.; Levy, R.; Ben-Nun, A.; Juknat, A.; Vogel, Z. Cannabidiol inhibits pathogenic T cells, decreases spinal microglial activation and ameliorates multiple sclerosis-like disease in C57BL/6 mice. Br. J. Pharmacol. 2011, 163, 1507–1519. [Google Scholar] [CrossRef] [PubMed]
- Hasseldam, H.; Johansen, F.F. Cannabinoid treatment renders neurons less vulnerable than oligodendrocytes in Experimental Autoimmune Encephalomyelitis. Int. J. Neurosci. 2011, 121, 510–520. [Google Scholar] [CrossRef]
- Mestre, L.; Docagne, F.; Correa, F.; Loría, F.; Hernangómez, M.; Borrell, J.; Guaza, C. A cannabinoid agonist interferes with the progression of a chronic model of multiple sclerosis by downregulating adhesion molecules. Mol. Cell. Neurosci. 2009, 40, 258–266. [Google Scholar] [CrossRef] [PubMed]
- Manterola, A.; Bernal-Chico, A.; Cipriani, R.; Ruiz, A.; Pérez-Samartín, A.; Moreno-Rodríguez, M.; Hsu, K.L.; Cravatt, B.F.; Brown, J.M.; Rodríguez-Puertas, R.; et al. Re-examining the potential of targeting ABHD6 in multiple sclerosis: Efficacy of systemic and peripherally restricted inhibitors in experimental autoimmune encephalomyelitis. Neuropharmacology 2018, 141, 181–191. [Google Scholar] [CrossRef] [PubMed]
- Wen, J.; Ribeiro, R.; Tanaka, M.; Zhang, Y. Activation of CB2 receptor is required for the therapeutic effect of ABHD6 inhibition in experimental autoimmune encephalomyelitis. Neuropharmacology 2015, 99, 196–209. [Google Scholar] [CrossRef]
- Feliú, A.; Bonilla Del Río, I.; Carrillo-Salinas, F.J.; Hernández-Torres, G.; Mestre, L.; Puente, N.; Ortega-Gutiérrez, S.; López-Rodríguez, M.L.; Grandes, P.; Mecha, M.; et al. 2-Arachidonoylglycerol Reduces Proteoglycans and Enhances Remyelination in a Progressive Model of Demyelination. J. Neurosci. Off. J. Soc. Neurosci. 2017, 37, 8385–8398. [Google Scholar]
- Brindisi, M.; Maramai, S.; Gemma, S.; Brogi, S.; Grillo, A.; Di Cesare Mannelli, L.; Gabellieri, E.; Lamponi, S.; Saponara, S.; Gorelli, B.; et al. Development and Pharmacological Characterization of Selective Blockers of 2-Arachidonoyl Glycerol Degradation with Efficacy in Rodent Models of Multiple Sclerosis and Pain. J. Med. Chem. 2016, 59, 2612–2632. [Google Scholar] [CrossRef]
- Hernández-Torres, G.; Cipriano, M.; Hedén, E.; Björklund, E.; Canales, Á.; Zian, D.; Feliú, A.; Mecha, M.; Guaza, C.; Fowler, C.J.; et al. A reversible and selective inhibitor of monoacylglycerol lipase ameliorates multiple sclerosis. Angew. Chem. (Int. Ed. Engl.) 2014, 53, 13765–13770. [Google Scholar] [CrossRef]
- Elliott, D.M.; Singh, N.; Nagarkatti, M.; Nagarkatti, P.S. Cannabidiol Attenuates Experimental Autoimmune Encephalomyelitis Model of Multiple Sclerosis through Induction of Myeloid-Derived Suppressor Cells. Front. Immunol. 2018, 9, 1782. [Google Scholar] [CrossRef]
- Dash, H.H.; Chavali, S. Management of traumatic brain injury patients. Korean J. Anesth. 2018, 71, 12–21. [Google Scholar] [CrossRef]
- Stocchetti, N.; Carbonara, M.; Citerio, G.; Ercole, A.; Skrifvars, M.B.; Smielewski, P.; Zoerle, T.; Menon, D.K. Severe traumatic brain injury: Targeted management in the intensive care unit. Lancet Neurol. 2017, 16, 452–464. [Google Scholar] [CrossRef]
- Kaur, P.; Sharma, S. Recent Advances in Pathophysiology of Traumatic Brain Injury. Curr. Neuropharmacol. 2018, 16, 1224–1238. [Google Scholar] [CrossRef]
- Savinainen, J.R.; Saario, S.M.; Laitinen, J.T. The serine hydrolases MAGL, ABHD6 and ABHD12 as guardians of 2-arachidonoylglycerol signalling through cannabinoid receptors. Acta Physiol. (Oxf. Engl.) 2012, 204, 267–276. [Google Scholar] [CrossRef]
- Blankman, J.L.; Simon, G.M.; Cravatt, B.F. A comprehensive profile of brain enzymes that hydrolyze the endocannabinoid 2-arachidonoylglycerol. Chem. Biol. 2007, 14, 1347–1356. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Rodriguez, A.B.; Siopi, E.; Finn, D.P.; Marchand-Leroux, C.; Garcia-Segura, L.M.; Jafarian-Tehrani, M.; Viveros, M.P. CB1 and CB2 cannabinoid receptor antagonists prevent minocycline-induced neuroprotection following traumatic brain injury in mice. Cereb. Cortex (N.Y. 1991) 2015, 25, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Baek, J.H.; Darlington, C.L.; Smith, P.F.; Ashton, J.C. Antibody testing for brain immunohistochemistry: Brain immunolabeling for the cannabinoid CB₂ receptor. J. Neurosci. Methods 2013, 216, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Cherry, J.D.; Olschowka, J.A.; O’Banion, M.K. Neuroinflammation and M2 microglia: The good, the bad, and the inflamed. J. Neuroinflamm. 2014, 11, 98. [Google Scholar] [CrossRef]
- Compston, A.; Coles, A. Multiple sclerosis. Lancet 2008, 372, 1502–1517. [Google Scholar] [CrossRef]
- Højsgaard Chow, H.; Schreiber, K.; Magyari, M.; Ammitzbøll, C.; Börnsen, L.; Romme Christensen, J.; Ratzer, R.; Soelberg Sørensen, P.; Sellebjerg, F. Progressive multiple sclerosis, cognitive function, and quality of life. Brain Behav. 2018, 8, e00875. [Google Scholar] [CrossRef]
- Handel, A.E.; Giovannoni, G.; Ebers, G.C.; Ramagopalan, S.V. Environmental factors and their timing in adult-onset multiple sclerosis. Nat. Rev. Neurol. 2010, 6, 156–166. [Google Scholar] [CrossRef]
- Global, regional, and national burden of motor neuron diseases 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2018, 17, 1083–1097. [CrossRef]
- Howard, J.; Trevick, S.; Younger, D.S. Epidemiology of Multiple Sclerosis. Neurol. Clin. 2016, 34, 919–939. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.J.; Chen, W.W.; Zhang, X. Multiple sclerosis: Pathology, diagnosis and treatments. Exp. Ther. Med. 2017, 13, 3163–3166. [Google Scholar] [CrossRef] [PubMed]
- Trapp, B.D.; Nave, K.A. Multiple sclerosis: An immune or neurodegenerative disorder? Annu. Rev. Neurosci. 2008, 31, 247–269. [Google Scholar] [CrossRef]
- Nussbaum, L.; Hogea, L.M.; Calina, D.; Andreescu, N.; Gradinaru, R.; Stefanescu, R.; Puiu, M. Modern treatment approaches in psychoses. Pharmacogenetic, neuroimagistic and clinical implications. Farmacia 2017, 65, 75–81. [Google Scholar]
- De Faria, O., Jr.; Moore, C.S.; Kennedy, T.E.; Antel, J.P.; Bar-Or, A.; Dhaunchak, A.S. MicroRNA dysregulation in multiple sclerosis. Front. Genet. 2012, 3, 311. [Google Scholar] [PubMed]
- Centonze, D.; Muzio, L.; Rossi, S.; Furlan, R.; Bernardi, G.; Martino, G. The link between inflammation, synaptic transmission and neurodegeneration in multiple sclerosis. Cell Death Differ. 2010, 17, 1083–1091. [Google Scholar] [CrossRef]
- Hampson, A.J.; Grimaldi, M.; Axelrod, J.; Wink, D. Cannabidiol and (-)Delta9-tetrahydrocannabinol are neuroprotective antioxidants. Proc. Natl. Acad. Sci. USA 1998, 95, 8268–8273. [Google Scholar] [CrossRef]
- Cabranes, A.; Venderova, K.; de Lago, E.; Fezza, F.; Sánchez, A.; Mestre, L.; Valenti, M.; García-Merino, A.; Ramos, J.A.; Di Marzo, V.; et al. Decreased endocannabinoid levels in the brain and beneficial effects of agents activating cannabinoid and/or vanilloid receptors in a rat model of multiple sclerosis. Neurobiol. Dis. 2005, 20, 207–217. [Google Scholar] [CrossRef]
- Loría, F.; Petrosino, S.; Mestre, L.; Spagnolo, A.; Correa, F.; Hernangómez, M.; Guaza, C.; Di Marzo, V.; Docagne, F. Study of the regulation of the endocannabinoid system in a virus model of multiple sclerosis reveals a therapeutic effect of palmitoylethanolamide. Eur. J. Neurosci. 2008, 28, 633–641. [Google Scholar] [CrossRef]
- Benito, C.; Romero, J.P.; Tolón, R.M.; Clemente, D.; Docagne, F.; Hillard, C.J.; Guaza, C.; Romero, J. Cannabinoid CB1 and CB2 receptors and fatty acid amide hydrolase are specific markers of plaque cell subtypes in human multiple sclerosis. J. Neurosci. Off. J. Soc. Neurosci. 2007, 27, 2396–2402. [Google Scholar] [CrossRef] [PubMed]
| Type of Tested Cannabinoid | Doses | Receptors/Effects | Experimental In Vivo Animal Model | Cognitive Effects (Used Test) | Motor Effects (Used Test) | Mechanisms of Action/Results | Ref. |
|---|---|---|---|---|---|---|---|
| HU-910 HU-914 (camphor -resorcinol derivatives) | 5–10 mg/kg (i.p.), 1 h after injury | CB2R agonists low CB1R affinity | C57Bl/6 WT mice CHI model | recovery of neurobehavioral deficits (NSS) | ↑sensor motor recovery | ↓inflammatory markers: ↓TNF-α, ↓IL-1α, ↓IL-1β, ↓IL-6 HU-914 showed the most important effects ↓TNF-α, ↓oedema ↓ BBB permeability ↓neuronal cell loss | [44] |
| JWH133 | 5.0 mg/kg (i.p.), 1 h after ICH | CB2R agonist | Sprague–Dawley rats CHI model | recovery of neurofunctional deficit (corner test) | ↑spontaneous activity (sensorimotor Garcia test) | ↓BBB breakdown ↓perihematoma ↓brain oedema ↑ adherens jonction proteins: ↑occludin, ↑ zo-1, ↑claudin-5 | [45] |
| JWH133 | 1.5 mg/kg (i.p.), 1 h after surgery | CB2R agonist | Sprague–Dawley adult male rats CHI model | ↑neurobehavioral outcomes (↓ NSS) | ↑ motor impairment (forelimb placing test, corner turn test) | ↓inflammatory markers: ↓IL-1β, ↓IL-6, ↓TNF-α, ↓ MMP2/9 ↑MKP-1→↓ MAPKs signalling pathway activation ↓neuroinflammation, ↑tight junction proteins: ↑zo-1, ↑cludin-5 ↓ BBB damage | [46] |
| GP1a | 3 mg/kg bw 10 min before TBI | CB2R agonist | C57BL/6 mice cortical impact model of TBI | ↓anxiety (OFT) | ↑motor coordination (stationary beam walk, rotarod test) | ↑anti-inflammatory markers ↑M2 macrophage polarization ↓cerebral oedema ↑ mean perfusion in the ipsilateral hemisphere | [47] |
| CBD | 10 mg/kg (i.p.), 30 min before and 3, 24 and 48 h after surgery | CB1R, CB2R agonist | C57BL/6 mice model of BCCAO | ↑cognitive performance (YM, OLT) ↓ anxiety (EZM) ↓ depression-like effects (FST) | ↑ motor activity (OF) | ↑nuclear receptors of the peroxisome proliferator-activated receptor family ↓adenosine uptake ↓reactive microglia and astrocytes, ↓ hippocampal neuroinflammation ↑BDNF protein, ↑DCX, ↑ MAP-2 | [48] |
| SMM-189 | 6 mg/kg (i.p.), 2 h after model and then daily for 2 weeks | CB2R inverse agonist | C57BL/6J male mice mTBI | working memory in a spontaneous cross-maze alternation task,↓depression, ↓fearfulness ↑ cognitive and emotional deficits | mitigate functional deficits | ↓ neuron loss preserve neuronal function and connectivity ↑beneficial M2 state of microglia | [49] |
| SR141716 | 10 mg/kg (i.p), 30 min after injury and then daily for 9 weeks | CB1R antagonist | Sprague–Dawley male rats TBI model | no improvement in spatial learning, memory (MWM) | no improvement (composite neuromotor score, beam-walking) | - | [50] |
| SMM-189 | 6 mg/, 2 h after injury and the daily for 14 days | CB2R inverse agonist | C57BL/6 male mice TBI model by single left-side blasts (50-60 psi) | - | ↓motor deficits (Rotarod test) | ↓ cortical and striatal neuron loss ↑ beneficial M2 state of microglia | [51] |
| ACEA | 1 mg/kg, daily (i.p.) first within 5–1 min after modelling and then 1 per day for 6 days | CB1R agonist | Sprague–Dawley male rats TBI model | ↑learning, ↑memory (NOR, MWT) No effects on anxiety (OFT, EPM) | no effects on locomotor coordination | ↓neuroinflammation, modulate metabolic processes → preserved neuronal tissues or functions | [52] |
| PF3845 (a selective FAAH inhibitor) | 5 mg/kg (i.p.) 30 min after TBI and then 1 per day for 14 days | CB1R, CB2R agonist | C57BL/6 male mice CCI model | ↓ anxiogenic behaviour (EZM) ↑ working memory (YM) | ↑ fine motor movements (BWBT) | ↓ degenerating neuronal cells (dentate gyri) (CB1R and CB2R) ↑BcL-2, ↑Hsp72, ↑Hsp 70 ↓ COX-2, ↓ iNOS ↑Arg-1 in the ipsilateral cortex ↑ERK1/2, ↑AKT phosphorylation | [53] |
| SMM-189 | 6 mg/kg (i.p) 2 h after model and then 1/day for 14 days | CB2R inverse agonist | C57BL/6 mice mTBI model | ↓depression (TST) ↓ fear response | ↓motor deficit (Rotarod test) | ↓ cytokines: IL-6, IFN –γ, IL-12p70, IL-10 ↓ Chemokines: IL-8, MIP-1β, TARC, MDC, eotaxin-3 ↓M1 microglial markers: CD11b, CD45, CD80 | [1] |
| JWH133 | 1.0mg/kg 1 h after surgery | CB2R (agonist) | Male Sprague–Dawley rats SAH model | ↓neurological deficits ↑neurological score ↑ neurobehavioral function (Garcia scoring system) | ↓neurological deficits (Garcia scoring system) | ↓brain oedema ↓BBB breakdown ↑ zo-1 ↓MPO ↑ TGF-beta1 | [54] |
| WWL70 (selective inhibitor of ABHD6) | 10 mg/kg (ip) 30 min after TBI and then 1 per day for 21 days | CB1R, CB2R agonist | C57BL/6 male mice CCI model | ↑working memory performance (YM) | ↑ fine motor movements (BWBT) ↑ motor coordination (Rotarod test) | ↓lesion volume in the cortex (CB1R) ↓neurodegeneration in the dentate gyrus (CB1R and CB2R) ↓BBB breakdown ↓iNOS, ↓COX-2 ↑Arg-1 in the ipsilateral cortex ↑ERK1/2, ↑AKT phosphorylation | [55] |
| AraS | 3 mg/kg (ip) 1 h after CHI model, after 7 days | CB1R, CB2R, TRPV1 channels | Sabra mice CHI model | ↑neurobehavioral function (alertness, NSS) | ↑neurobehavioral function (reflexes, coordination, motor abilities, balance) (NSS) | ↓infarct volume ↓terminal differentiation of NPC into astrocytes ↑ neuroblast differentiation, ↑ doublecortin- neuroblastic marker | [56] |
| 0-1966 | 5 mg/kg (ip) at 2, 24, 48 and 72 h after CCI | CB2R agonist | C57BL/6 mice CCI model | - | ↑postinjury motor performance (rota-rod test) Open-field (forced exploration) testing | ↓ BBB damage ↓ immune cell infiltration,↓ release of pro-inflammatory neurochemicals ↓ Iba-1-positive macrophages/microglia | [57] |
| CBD | 1 mg/kg single dose | CB1R, CB2R agonist | Newborn Wistar rats HI model | ↓ working memory impairment (NOR) | ↑ motor coordination (Rotarod test); ↑motor deficit in the contralateral (right) forepaw (CRT) | ↓ brain injury volume ↓ the extend of brain injury ↓ TNF-α ↓ oxidative stress | [58] |
| KN38-7271 | 0.1–10 mg/kg (i.p) 2 h before and 30 min, 4 h and on day 2 and 6 after MCAO | CB1R, CB2R agonist | Sprague–Dawley rat MCAO model | ↓ MCAO-motor impairment (ladder rung walking test) | ↓ cortical infarct size | [59] | |
| 0-1966 | 5 mg/kg (i.p) 1 h and 24 h after injury | CB2R agonist | C57BL/6 mice CCI model | ↑exploratory behaviour (OFT) | ↑Locomotor performance (rotarod, forelimb cylinder) | ↓cerebral oedema ↓ perivascular areas of substance P immunoreactivity ↓ activated macrophages/ microglial cells | [60] |
| Type of Tested Cannabinoid | Doses | Receptors/Effects | Experimental In Vivo Animal Model | Motor Effects | Mechanisms of Action/Results | Ref. |
|---|---|---|---|---|---|---|
| Δ9-THC +CBD | 10 mg/kg Δ9-THC and 10 mg/kg CBD in combination daily (i.p) from day ten until day 27 | CB1R, CB2R agonists | C57BL/6 female mice induced EAE model | ↓ clinical symptoms of EAE (several degrees of paralysis of hind limbs) | ↓neuro-inflammation ↓miR-21a-5p, miR-31-5p, miR-122-5p, miR-146a-5p, miR-150-5p, miR-155-5p, miR-27b-5p ↑miR-706-5p, miR-7116 | [61] |
| BCP alone or + IMP | 5 mg/kg/day (p.o) BCP; 5/2.5 mg/kg/day (p.o) BCP+10 mg/kg/day IMP from day 10 to 37 | CB2R selective agonist/sphingomyelinase inhibitor (IMP) | C57BL/6 mice induced EAE model | ↓ clinical symptoms of EAE (more pronounce in the BCP+IMP groups) | ↓pathological score (massive leukocyte infiltrations) (more pronounce in the BCP+IMP groups) ↓ inflammatory markers: TNF-α, IL-6, IL-17, IL-17/IL-10 ratio ↑ anti-inflammatory cytokines: IL-10 ↓ cell proliferation | [66] |
| PEALut (PEA + luteolin) | 5 mg/kg/day from day 11 after modelling until day 27 | PPAR-α and GPR55 receptor agonist | C57BL/6 mice induced EAE model | ↓ clinical symptoms of EAE ↓ motor disability | ↓TNF-α, IL-1β, IFN-γ, SAA1 mRNAs ↓ expression of receptors implicated in inflammation: CB2R, CD3-γ, Fpr2, TLR2, TCR-ζ chain | [72] |
| CanniMed oil Huile (10:10 and 1:20) | 215 mg/kg oil extract (p.o) daily from day 6 to 18 | CB1R, CB2R agonist | Lewis rats induced EAE model | ↓ motor disability | ↓TNF-α ↑BDNF protein expression | [62] |
| CBD | 20 mg/kg (i.p) daily from day 9 to day 25 | CB1R, CB2R agonist | C57BL/6 female mice induced EAE model | ↓clinical scores of paralysis | ↓ T cell infiltration in the CNS, ↓ IL-17, IFNγ | [62] |
| KT182 (pass BBB), KT203 (acts only in the periphery) (ABHD6 inhibitors) | 2 mg/kg (i.p.) from day 1 of modelling until day 10 | CB1R, CB2R agonist | C57BL/6 female mice induced EAE model | ↓ motor disability (KT182) No effect on corticospinal tract conduction latency | No effects on inflammatory molecules KT182 prevent calcium overload to the mitochondria | [84] |
| CBD | 10 mg/kg (i.p) daily from day 14 until day 28 | CB1R, CB2R agonist | C57BL/6 male mice induced EAE model | ↓clinical scores of paralysis | ↑ phosphorylation of PI3K, ↑Akt, ↑mTOR, ↑ BNDF, ↑PPARγ ↓ pro-inflammatory cytokines: ↓IFN-γ, ↓ IL-17 | [89] |
| UCM03025 (MAGL inhibitor) | 5 mg/kg/day (i.p) from day 75 until day 85 after modelling | CB1R, CB2R agonist | SJL/J mice infected with TMEV | ↓ motor disability | ↓ astrogliosis ↓ CSPGs level in spinal cord ↑OPCs differentiation→remyelination and axonal protection ↓neuroinflammation | [86] |
| BCP | 25/50 mg/kg twice/day (p.o) from day 0 until day 30 | CB2R (selective agonist) | C57BL/6 male mice induced EAE model | ↓clinical scores of paralysis ↓ progression of the disease | ↓Iba-1 and iNOS ↓pro-inflammatory cytokines: TNF-α, IL-6, IL-1β ↓ CD4+, ↓ CD8+ T cells | [67] |
| 2-amidoalkylindole derivatives | 10 mg/kg/day or 30 mg/kg/day (i.p) from day 3 until day 19 | CB2R selective agonist | C57BL/6 mice induced EAE model | ↓ motor disability ↓clinical scores of paralysis (dose-dependent manner) | ↓ leukocyte infiltration in the white matter region ↓ demyelination in the white matter | [68] |
| trans-(1-(1-(1H-1,2,4-Triazole-1-carbonyl)piperidin-4-yl)-4-benzo[d][1,3] dioxol-5-yl)-3-(4-fluorophenyl)azetidin-2-one (β-lactam-based MAGL inhibitor) | 3 mg/kg/day (i.p) from day 6 after modelling until day 2 | CB1R, CB2R agonist | C57BL/6 female mice induced EAE model | ↓clinical scores of paralysis ↓ progression of the disease | - | [87] |
| PM226 (isoxazole derivative) | 5 mg/kg/day (i.p) from day 1 until day 7 after modelling | CB2R selective agonist | SJL/J mice infected with TMEV | ↓clinical scores of paralysis | ↓ microglia activation (↓Iba-1+ cells) | [69] |
| CBD | 10 mg/kg (i.p) daily from day 14 until day 28 after modelling | CB1R, CB2R agonist | C57BL/6 male mice induced EAE model | ↓ motor disability | ↓ activation of MAPK signalling pathway ↓claved-caspase 3 (marker of apoptosis) ↑ Bcl-2, ↓Bax ↓mitochondrial alterations | [76] |
| CBD | 1% CBD-cream (CBD solubilized in propylene glycol and basis dense cream O/A daily for 28 days | CB1R, CB2R agonist | C57BL/6 mice induced EAE model | ↓ motor disability (↓ paralysis of hind limbs) | ↓ CD4 and CD8α T cells, ↓pro-inflammatory markers (Il-10, Foxp3, p-selectin, TGF-β, IFN-γ), ↓ cleaved caspase 3 (apoptosis) ↓iNOS, PARP, nitrotyrosine | [77] |
| CBD PEA | 5 mg/kg/day CBD/PEA or CBD+ PEA (i.p) for 3 consecutive days from the first sign of disease | CB1R, CB2R; (CBD); PPARα and GPR55 agonist (PEA) | C57BL/6 female mice induced EAE model | ↓ motor disability (CBD and PEA alone, no effect for CBD+PEA) | ↓inflammation ↓demyelination ↓axonal damage ↓inflammatory markers: IL-17, TNF-α, IFN- γ | [78] |
| WWL70 (ABHD6 inhibitor) | 10 mg/kg/day (i.p) from day 10 until day 21 or 28 after modelling | CB2R agonist (↑2-AG mainly in microglia/macrophage (activation of CB2R), not in T cell (activation of CB1R)) | C57BL/6 female mice induced EAE model | ↓ motor dissability ↓ disease progression | ↓microglia/macrophage activation ↓inflammatory mediators: ↓(TNF-α, ↓iNOS, ↓COX-2 ↓demyelination ↓axonal damage | [85] |
| Sativex® (Δ9-THC-BDS + CBD-BDS combination)/ Δ9-THC-BDS/ CBD-BDS | Sativex® (1:1; 10 mg/kg/day from each)/20 mg/kg/day (Δ9-THC-BDS/ CBD-BDS) from day 11 until day 31 after modelling | CB1R (Sativex® and Δ9-THC-BDS) agonist | C57BL/6 female mice induced EAE model | ↓ motor disability ↓ disease progression (Sativex® and Δ9-THC-BDS) | ↓ cell aggregates determined by microglia activation (Sativex® and Δ9-THC-BDS) | [63] |
| Sativex® (Δ9-THC-BDS+ CBD-BDS combination)/ Δ9-THC-BDS/ CBD-BDS | Sativex® (1:1; 5 mg/kg/day from each)/5 mg/kg/day (Δ9-THC-BDS/ CBD-BDS) (i.p) from day 70 until day 80 after modelling | PPARγ receptor agonist (CBD-BDS) CB1R (Δ9-THC-BDS) | SJL/J mice infected with TMEV | ↓ motor disability ↓ disease progression (Sativex® and CBD-BDS, less effects of Δ9-THC-BDS) | ↓ infiltrates ↓ VCAM-1, ICAM-1 ↓ microglial activity ↓ pro-inflammatory cytokines ↓ axonal damage ↓ astrocyte reactivity and accumulation of CSPGs in the spinal cord | [64] |
| VCE-003 | 5 mg/kg/day from day 6 until day 27 after modelling | CB2R PPARγ receptor agonist | C57BL/6 female mice induced EAE model | ↓ motor disability | ↓ infiltrates ↓ CD4+ T cells in the spinal cord ↓ microglia/macrophage activation ↓ demyelination ↓ axonal damage ↓ inflammatory markers: ICAM-1, TNFα, iNOS, IFNγ, IL-17 | [70] |
| 2-(4-benzylphenyl)- acetate and 6-(biphenyl-4-yl)hexanoate derivatives (MAGL inhibitor) | 5 mg/kg (i.p) from day 6 after modelling until day 27 | CB1R, CB2R agonist | C57BL/6 mice induced EAE model | ↓ motor disability | ↑ 2-AG levels (spinal cord) →↓leukocyte infiltration and microglial responses, ↓ axonal damage, restore myelin morphology | [88] |
| CBD | 5 mg/kg (i.p) daily from day 1 to 7 after infection | CB1R, CB2R, adenosine A2A receptors | SJL/J mice infected with TMEV | ↓ motor disability | ↓ VCAM-1, ↓chemokines: CCL2, CCL5 ↓ IL-1 ↓ microglia activation | [79] |
| CB52 (synthetic cannabinoid) | 2 mg/kg/day (i.p) from day 3 until day 30 after modelling | CB1R agonist | C57BL/6 female mice induced EAE model | ↓ motor disability | ↓ microglia activation, ↓ nitrotyrosine formation, ↓T cell infiltration, ↓TNF-a ↓oligodendrocyte toxicity, ↓axonal damage, ↓myelin loss | [74] |
| WIN55,212-2 | 5 mg/kg/day (i.p) from day 11 until day 17 | CB1R, CB2R agonist/ the effects are mediated more by CB1R | C57BL/6 mice induced EAE model | ↓ motor disability ↓ progression of the disease | ↓TNF-α, iNOS, COX-2 (spinal cord and brainstem) | [80] |
| VCE-003 | 5 mg/kg/day (i.p) for 14 days starting from day 60 after infection | PPARγ (partial agonist), CB2R (modest agonist) | SJL/J mice infected with TMEV | ↓ motor disability | ↓ microglial activation ↓ VCAM-1 ↓chemokines and chemokine receptors genes up-regulation | [71] |
| Δ9-THC -rich extract/ CBD – rich extract, Δ9-THC /CBD extract | 50 mg/kg (i.p) Δ9-THC -rich Extract/CBD—rich extract; 25 mg/kg (i.p) Δ9-THC/CBD extract (acute treatment 1 dose on day 32 after modelling and chronic treatment for 7 days from day 68 after modelling) | CB1R, CB2R agonist | Biozzi AB/H mice CREAE model | ↓ motor disability (chronic administration Δ9-THC-rich extract) | - | [65] |
| CBD | 5 mg/kg/day (i.p) on days 19, 20 and 21 after modelling | not mediated via CB1R or CB2R | C57BL/6 female mice EAE model | ↓ motor disability | ↓ axonal loss ↓infiltration of T cells, ↓microglial activation | [81] |
| AEA | 3.5 mg/kg/day (i.p) for 7 successive days from day 83 after modelling | CB1R, CB2R agonist | SJL/J mice infected with TMEV | ↓ motor disability | ↓ p35, p19 and p40 mRNAs ↓IL-17A | [73] |
| WIN55,212-2 | 10 mg/kg/day (i.p) for 15 days from the symptom’s debut | CB1R, CB2R agonist PPARγ receptor agonist | Dark Agouti female rat EAE model | ↓ motor disability No relapse | ↓ inflammatory load ↓ demyelination | [82] |
| WIN55,212-2 | 1.5 mg/kg (i.p) twice per day from day 1 for 3 successive days | CB1R, CB2R agonist PPARγ receptor agonist | SJL/J mice infected with TMEV | ↓ motor disability | ↓ adhesion molecules: ICAM-1, VCAM-1 | [83] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Calina, D.; Buga, A.M.; Mitroi, M.; Buha, A.; Caruntu, C.; Scheau, C.; Bouyahya, A.; El Omari, N.; El Menyiy, N.; Docea, A.O. The Treatment of Cognitive, Behavioural and Motor Impairments from Brain Injury and Neurodegenerative Diseases through Cannabinoid System Modulation—Evidence from In Vivo Studies. J. Clin. Med. 2020, 9, 2395. https://doi.org/10.3390/jcm9082395
Calina D, Buga AM, Mitroi M, Buha A, Caruntu C, Scheau C, Bouyahya A, El Omari N, El Menyiy N, Docea AO. The Treatment of Cognitive, Behavioural and Motor Impairments from Brain Injury and Neurodegenerative Diseases through Cannabinoid System Modulation—Evidence from In Vivo Studies. Journal of Clinical Medicine. 2020; 9(8):2395. https://doi.org/10.3390/jcm9082395
Chicago/Turabian StyleCalina, Daniela, Ana Maria Buga, Mihaela Mitroi, Aleksandra Buha, Constantin Caruntu, Cristian Scheau, Abdelhakim Bouyahya, Nasreddine El Omari, Naoual El Menyiy, and Anca Oana Docea. 2020. "The Treatment of Cognitive, Behavioural and Motor Impairments from Brain Injury and Neurodegenerative Diseases through Cannabinoid System Modulation—Evidence from In Vivo Studies" Journal of Clinical Medicine 9, no. 8: 2395. https://doi.org/10.3390/jcm9082395
APA StyleCalina, D., Buga, A. M., Mitroi, M., Buha, A., Caruntu, C., Scheau, C., Bouyahya, A., El Omari, N., El Menyiy, N., & Docea, A. O. (2020). The Treatment of Cognitive, Behavioural and Motor Impairments from Brain Injury and Neurodegenerative Diseases through Cannabinoid System Modulation—Evidence from In Vivo Studies. Journal of Clinical Medicine, 9(8), 2395. https://doi.org/10.3390/jcm9082395











