Prevention and Management of Hormonal Crisis during Theragnosis with LU-DOTA-TATE in Neuroendocrine Tumors. A Systematic Review and Approach Proposal
Abstract
1. Introduction
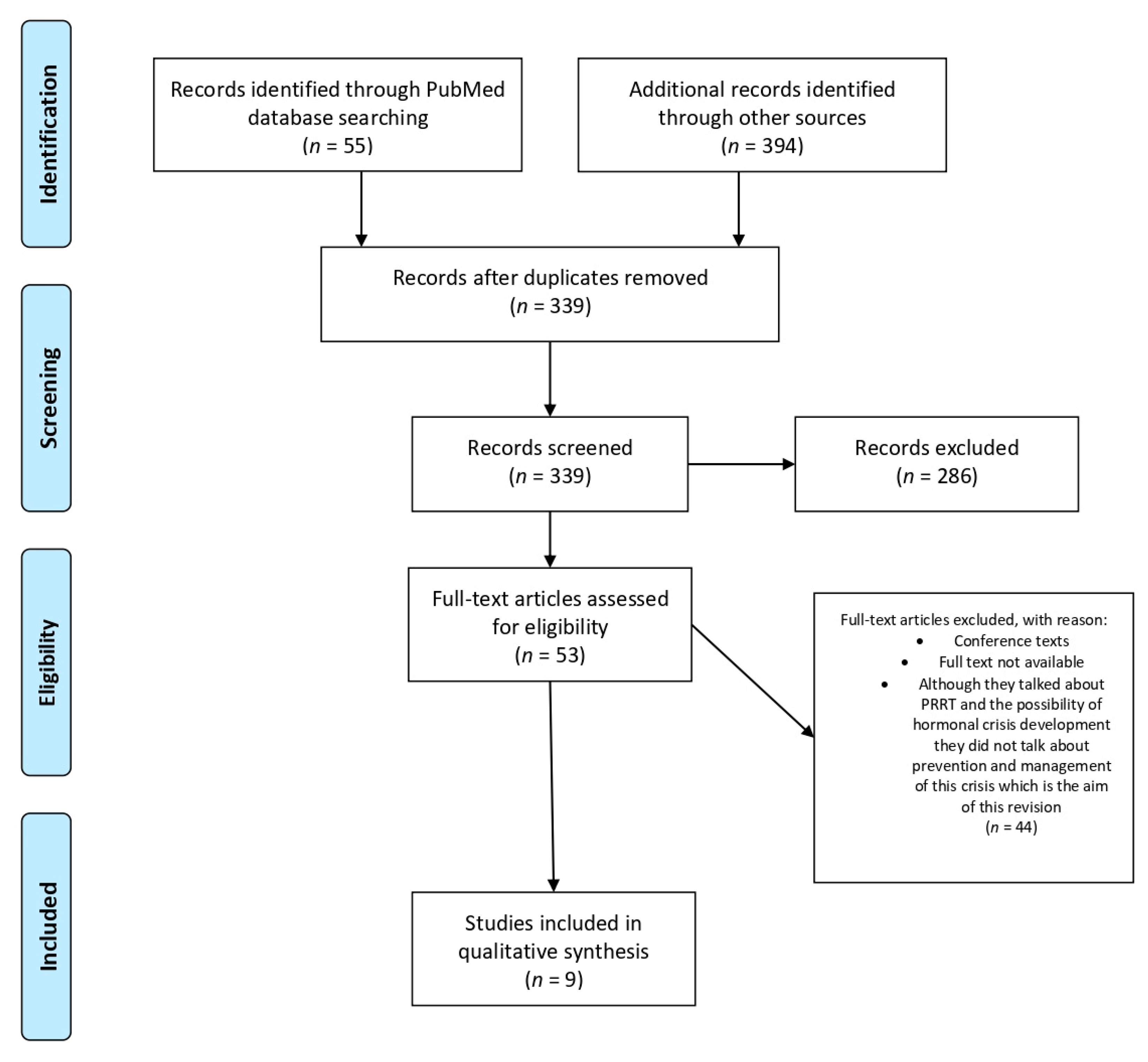
2. Methods
3. Results
4. Discussion
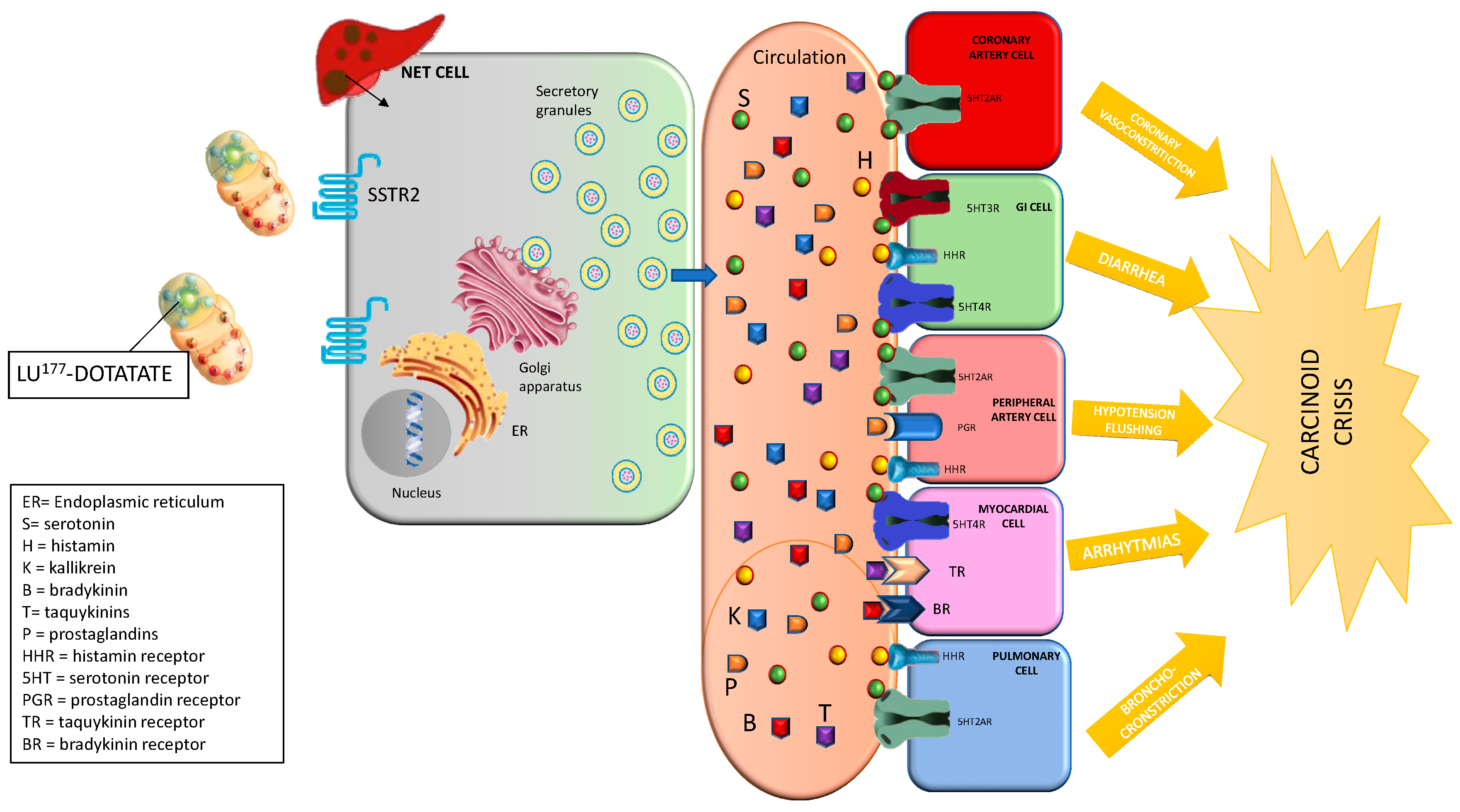
4.1. Carcinoid Crisis
4.1.1. Prevention
4.1.2. Management and Treatment
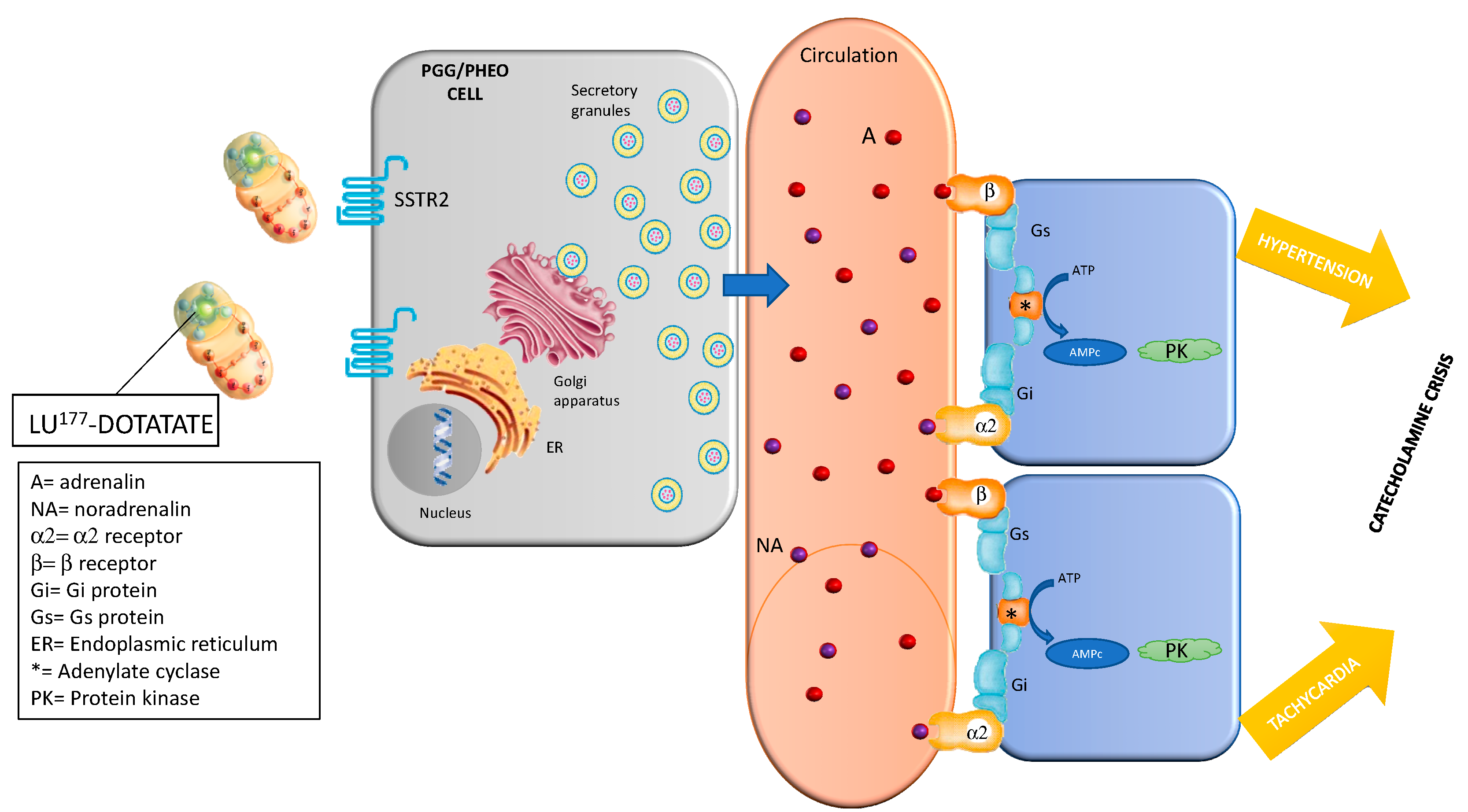
4.2. Catecholaminergic Crisis
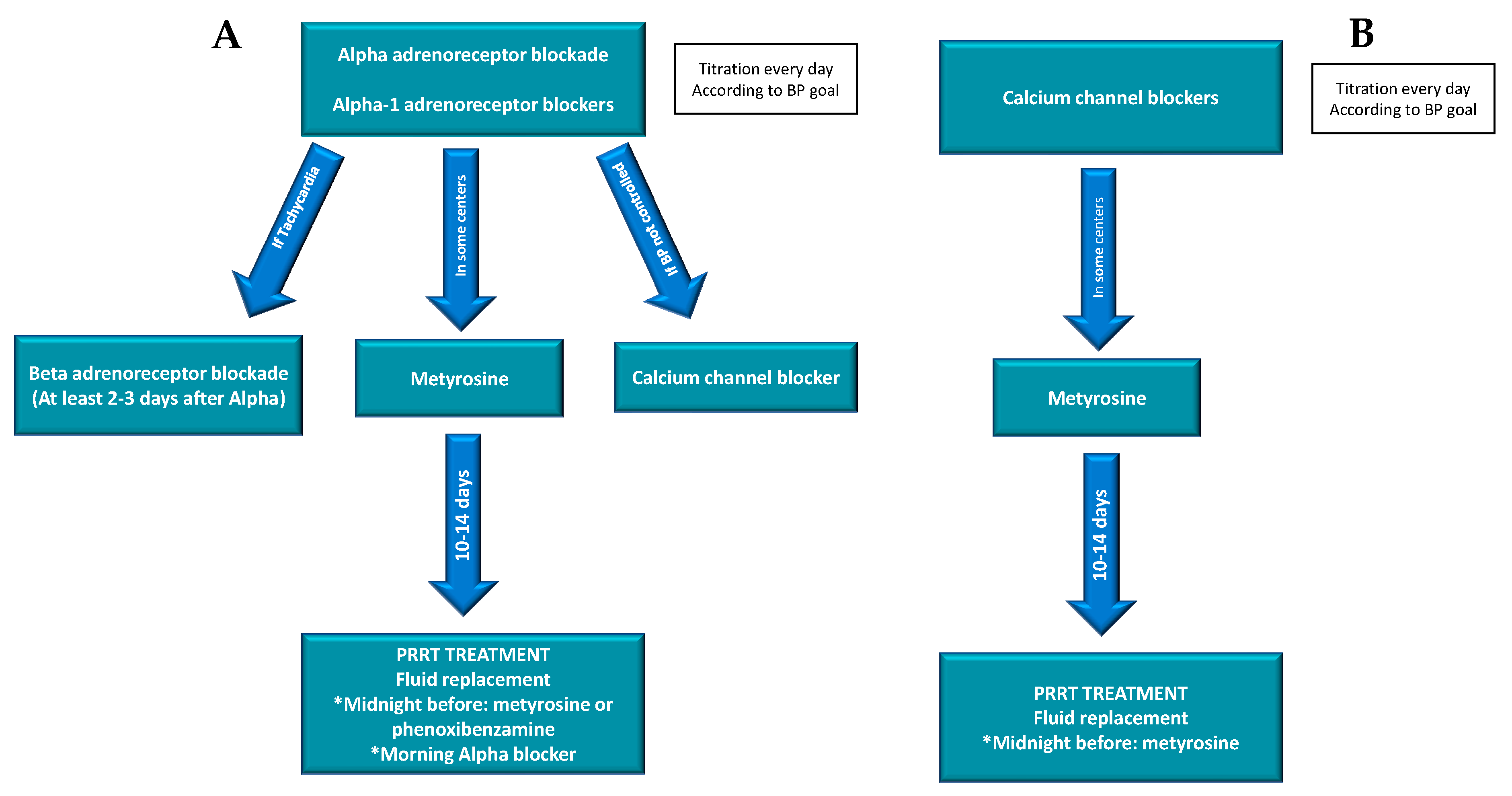
4.2.1. Prevention
4.2.2. Management and Treatment
4.3. Other Complications
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Dasari, A.; Shen, C.; Halperin, D.; Zhao, B.; Zhou, S.; Xu, Y.; Shih, T.; Yao, J.C. Trends in the Incidence, Prevalence, and Survival Outcomes in Patients With Neuroendocrine Tumors in the United States. JAMA Oncol. 2017, 3, 1335–1342. [Google Scholar] [CrossRef] [PubMed]
- WHO Classification of Tumours Editorial Board. Digestive system tumours. Lyon (France): International agency for research on cancer. In WHO Classification of Tumours Series, 5th ed.; WHO: Geneva, Switzerland, 2019; Volume 1. [Google Scholar]
- Zandee, W.T.; Brabander, T.; Blazevic, A.; Kam, B.L.R.; Teunissen, J.J.M.; Feelders, R.A.; Hofland, J.; de Herder, W.W. Symtomatic and Radiological Response to 177Lu-DOTATATE for the Treatment of Functioning Pancreatic Neuroendocrine Tumors. J. Clin. Endocrinol. Metab. 2019, 104, 1336–1344. [Google Scholar] [CrossRef] [PubMed]
- Hartmut, P.H.; Neumann, H.P.H.; Young, W.F.; Eng, C. Pheochromocytoma and Paraganglioma. N. Engl. J. Med. 2019, 381, 552–565. [Google Scholar] [CrossRef]
- Jerzy, H.; Roman, T. Neuroendocrine neoplasms and somatostatin receptor subtypes expression. Nucl. Med. Rev. 2016, 19, 111–117. [Google Scholar] [CrossRef]
- Remes, S.T.; Leijon, L.L.; Vesterinen, T.J.; Arola, J.J.; Haglund, C.H. Immunohistochemical Expression of Somatostatin Receptor Subtypes in a Panel of Neuroendocrine Neoplasias. J. Histochem. Cytochem. 2019, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Baumann, T.; Rottenburger, C.; Nicolas, G.; Wild, D. Gastroenteropancreatic neuroendocrine tumours (GEP-NET)- Imaging and staging. Best Pract. Res. Clin. Endocrinol. Metab. 2016, 30, 45–57. [Google Scholar] [CrossRef] [PubMed]
- Ito, T.; Jensen, R.T. Molecular imaging in neuroendocrine tumors: Recent advances, controversies, unresolved issues, and roles in management. Curr. Opin. Endocrinol. Diabetes Obes. 2017, 24, 15–24. [Google Scholar] [CrossRef]
- Lee, L.; Ito, T.; Jensen, R.T. Imaging of pancreatic neuroendocrine tumors: Recent advances, current status, and controversies. Expert Rev. Anticancer Ther. 2018, 18, 837–860. [Google Scholar] [CrossRef]
- Haider, M.; Al-Toubah, T.; El-Haddad, G.; Strosberg, J. Molecular imaging and radionuclide therapy of neuroendocrine tumors. Curr. Opin. Endocrinol. Diabetes Obes. 2020, 27, 16–21. [Google Scholar] [CrossRef]
- Hope, T.A.; Bergsland, E.K.; Bozkurt, M.F.; Graham, M.; Heaney, A.P.; Herrmann, K. Appropriate Use Criteria for Somatostatin Receptor PET Imaging in Neuroendocrine Tumors. J. Nucl. Med. 2018, 59, 66–74. [Google Scholar] [CrossRef]
- Deppen, S.A.; Blume, J.; Bobbey, A.J.; Shah, C.; Graham, M.M.; Lee, P.; Delbeke, D.; Walker, R.C. 68Ga-DOTATATE Compared with 111In-DTPA-Octreotide and Conventional Imaging for Pulmonary and Gastroenteropancreatic Neuroendocrine Tumors: A Systematic Review and Meta-Analysis. J. Nucl. Med. 2016, 57, 872–878. [Google Scholar] [CrossRef] [PubMed]
- Desai, H.; Borges-Neto, S.; Wong, T.Z. Molecular Imaging and Therapy for Neuroendocrine Tumors. Curr. Treat Opt. Oncol. 2019, 20, 78. [Google Scholar] [CrossRef] [PubMed]
- Crona, J.; Taïeb, D.; Pacak, K. New Perspectives on Pheochromocytoma and Paraganglioma: Toward a Molecular Classification. Endocr. Rev. 2017, 38, 489–515. [Google Scholar] [CrossRef] [PubMed]
- Brabander, T.; van der Zwan, W.A.; Teunissen, J.J.M.; Kam, B.L.R.; Feelders, R.A.; de Herder, W.W.; van Eijck, C.H.J.; Franssen, G.J.H.; Krenning, E.P.; Kwekkeboom, D.J. Long-Term Efficacy, Survival, and Safety of [177 Lu-DOTA 0,Tyr 3]octreotate in Patients With Gastroenteropancreatic and Bronchial Neuroendocrine Tumors. Clin. Cancer Res. 2017, 23, 4617–4624. [Google Scholar] [CrossRef]
- Kong, G.; Grozinsky-Glasberg, S.; Hofman, M.S.; Callahan, J.; Meirovitz, A.; Maimon, O.; Pattison, D.A.; Gross, D.J.; Hicks, R.J. Efficacy of Peptide Receptor Radionuclide Therapy for Functional Metastatic Paraganglioma and Pheochromocytoma. J. Clin. Endocrinol. Metab. 2017, 102, 3278–3287. [Google Scholar] [CrossRef] [PubMed]
- De Keizer, B.; van Aken, M.O.; Feelders, R.A.; de Herder, W.W.; Kam, B.L.; van Essen, M.; Krenning, E.P.; Kwekkeboom, D.J. Hormonal crisis following receptor radionuclide therapy with the radiolabeled somatostatin analogue [177Lu-DOTA0,Tyr3]octreotate. Eur. J. Nucl. Med. Mol. Imaging 2008, 35, 749–755. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Group, P. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. Ann. Intern. Med. 2009, 151, 65–94. [Google Scholar] [CrossRef]
- National Heart, Lung and Blood Institute. Quality Assessment Tool for Case Series Studies. Available online: https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools (accessed on 6 May 2020).
- Pryma, D.A.; Chin, B.B.; Noto, R.B.; Dillon, J.S.; Perkins, S.; Solnes, L.; Kostakoglu, L.; Serafini, A.N.; Pampaloni, M.H.; Jensen, J.; et al. Efficacy and Safety of High-Specific-Activity 131I-MIBG Therapy in Patients with Advanced Pheochromocytoma or Paraganglioma. J. Nucl. Med. 2019, 60, 623–630. [Google Scholar] [CrossRef]
- Makis, W.; McCann, K.; Mc Ewan, A. The Challenges of Treating Paraganglioma Patients With (177)-Lu-DOTATATE PRRT: Catecholamine Crisis, Tumor Lysis Syndrome and the Need of Modification of Treatment Protocols. Nucl. Med. Mol. Imaging 2015, 49, 223–230. [Google Scholar] [CrossRef]
- Yadav, S.K.; Jha, C.K.; Patil, S.; Datta, D.; Mishra, A.; Pradhan, P.K. Lutetium therapy-induced carcinoid crisis: A case report and review of literature. J. Can. Res. Ther. 2020. [Google Scholar] [CrossRef]
- Stentzel, J.; Noe, S.; Holzapfel, K.; Erlmeier, F.; Eyer, F. Fatal Systemic Vasoconstriction in a Case of Metastatic Small-Intestinal NET. Case Rep. Gastrointest. Med. 2017, 2017, 9810194. [Google Scholar] [CrossRef]
- Kolasinska -C´wikła, A.; Peczkowska, M.; Cwikła, J.B.; Michałowska, I.; Pałucki, J.M.; Bodei, L.; Lewczuk-My´slicka, A.; Januszewicz, A. A Clinical Efficacy of PRRT in Patients with Advanced, Nonresectable, Paraganglioma-Pheochromocytoma, Related to SDHx Mutation. J. Clin. Med. 2019, 8, 952. [Google Scholar] [CrossRef]
- Kwekkeboom, D.J.; de Herder, W.W.; Kam, B.L.; van Eijck, C.H.; van Essen, M.; Kooij, P.P.; Feelders, R.A.; van Aken, M.O.; Krenning, E.P. Treatment With the Radiolabeled Somatostatin Analog [177Lu-DOTA0,Tyr3]Octreotate: Toxicity, Efficacy, and Survival. J. Clin. Oncol. 2008, 26, 2124–2130. [Google Scholar] [CrossRef] [PubMed]
- Tapia Rico, G.; Li, M.; Pavlakis, N.; Cehic, G.; Price, T.J. Prevention and management of carcinoid crises in patients with high-risk neuroendocrine tumours undergoing peptide receptor radionuclide therapy (PRRT): Literature review and case series from two Australian tertiary medical institutions. Cancer Treat. Rev. 2018, 66, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Aluri, V.; Dillon, J.S. Biochemical Testing in Neuroendocrine Tumors. Endocrinol. Metab. Clin. N. Am. 2017, 46, 669–677. [Google Scholar] [CrossRef]
- Keskin, O.; Yalcin, S. Carcinoid Crisis in the Intensive Care Unit; Nates, J.L., Price, K.J., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 995–1001. [Google Scholar]
- Raedler, L. Lutathera (Lutetium Lu 177 Dotatate) First Radioactive Drug Approved for Gastroenteropancreatic Neuroendocrine Tumors. Oncol. Guide FDA Approv. Spec. Feature 2019, 37–39. [Google Scholar]
- Mittra, E. Neuroendocrine Tumor Therapy: 177Lu-DOTATATE. AJR 2018, 211, 278–285. [Google Scholar] [CrossRef]
- Hope, T.; Abbott, A.; Colucci, K.; Bushnell, D.L.; Gardner, L.; Graham, W.S.; Lindsay, S.; MetzDC, P.D.; Stabin, M.G.; Strosberg, J.R. NANETS/SNMMI Procedure Standard for Somatostatin Receptor–Based Peptide Receptor Radionuclide Therapy with 177Lu-DOTATATE. J. Nucl. Med. 2019, 60, 937–943. [Google Scholar] [CrossRef]
- Ito, T.; Lee, L.; Jensen, R. Carcinoid-syndrome: Recent advances, current status and controversies. Curr. Opin. Endocrinol. Diabetes Obes. 2018, 25, 22–35. [Google Scholar] [CrossRef]
- Gut, P.; Waligórska-Stachura, J.; Czarnywojtek, A.; Sawicka-Gutaj, N.; Bączyk, M.; Ziemnicka, K.; Fischbach, J.; Wolinski, K.; Kaznowski, J.; Wrotkowska, E. Management of the hormonal syndrome of neuroendocrine tumors. Arch. Med. Sci. 2017, 3, 515–524. [Google Scholar] [CrossRef] [PubMed]
- Hofland, J.; Zandee, W.T.; de Herder, W.W. Role of biomarker tests for diagnosis of neuroendocrine tumours. Nat. Rev. Endocrinol. 2018, 29, 656–669. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.-J.; Tang, C.-W. Somatostatin analogues do not prevent carcinoid crisis. Asian Pac. J. Cancer Prev. 2014, 15, 6679–6683. [Google Scholar] [CrossRef] [PubMed]
- Hofland, J.; Herrera-Martinez, A.D.; Zandee, W.T.; de Herder, W.W. Management of carcinoid syndrome: A systematic review and meta-analysis. Endocr. Relat. Cancer 2019, 26, R145–R156. [Google Scholar] [CrossRef] [PubMed]
- Naraev, B.G.; Halland, M.; Halperin, D.M.; Purvis, A.J.; O’Dorisio, T.M.; Halfdanarson, T.R. Management of Diarrhea in Patients With Carcinoid Syndrome. Pancreas 2019, 48, 961–972. [Google Scholar] [CrossRef] [PubMed]
- Safford, S.D.; Coleman, R.E.; Gockerman, J.P.; Moore, J.; Feldman, J.M.; Leight, G.S., Jr.; Tyler, D.S.; Olson, J.A., Jr. Iodine-131 metaiodobenzylguanidine is an effective treatment for malignant pheochromocytoma and paraganglioma. Surgery 2003, 134, 956–962. [Google Scholar] [CrossRef]
- Rosas, A.L.; Kasperlik-Zaluska, A.A.; Papierska, L.; Bass, B.L.; Pacak, K.; Eisenhofer, G. Pheochromocytoma crisis induced by glucocorticoids: A report of four cases and review of the literature. Eur. J. Endocrinol. 2008, 158, 423–429. [Google Scholar] [CrossRef]
- Mak, I.; Hayes, A.; Khoo, B.; Grossman, A. Peptide Receptor Radionuclide Therapy as a Novel Treatment for Metastatic and Invasive Phaeochromocytoma and Paraganglioma. Neuroendocrinology 2019, 109, 287–298. [Google Scholar] [CrossRef]
- Martucci, V.; Pacak, K. Pheochromocytoma and Paraganglioma: Diagnosis, Genetics, Management, and Treatment. Curr. Probl. Cancer 2014, 38, 7–41. [Google Scholar] [CrossRef]
- Del Olmo-García, M.I.; Palasí, R.; Camara, R.; Ponce, J.L.; Merino-Torres, J.F. Surgical and pharmacological management of functioning pheochromocytoma and paraganglioma. In On Paraganglioma: A Multidisciplinary Approach; Mariani-Costantini, R., Ed.; Codon Publications: Brisbane, Australia, 2019. [Google Scholar]
- Ramachabdran, R.; Rewari, V. Current perioperative management of pheochromoytomas. Indian J. Urol. 2017, 33, 19–25. [Google Scholar] [CrossRef]
- Fishbein, L.; Orlowski, R.; Cohen, D. Pheochromocytoma/paraganglioma: Review of perioperative management of blood pressure and update on genetic mutations associated with pheochromocytoma. J. Clin. Hypertens. 2013, 15, 428–434. [Google Scholar] [CrossRef] [PubMed]
- Cohen, D.; Fishbein, L. Hypertension: A companion to Braunwald’s Heart Disease Secondary Hypertension, 1st ed.; Elsevier: Chicago, IL, USA, 2018; pp. 136–143. [Google Scholar]
- Pacak, K. Perioperative management of the pheochromocytoma patient. J. Clin. Endocrinol. Metab. 2007, 92, 4069–4079. [Google Scholar] [CrossRef] [PubMed]
- Zaknun, J.; Bodei, L.; Mueller-Brand, J.; Pavel, M.; Baum, R.P.; Hörsch, D.; O’Dorisio, M.S.; O’Dorisiol, T.M.; Howe, J.R.; Cremonesi, M.; et al. The joint IAEA, EANM, and SNMMI practical guidance on peptide receptor radionuclide therapy (PRRNT) in neuroendocrine tumours. Eur. J. Nucl. Med. Mol. Imaging 2013, 40, 800–816. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association. Diabetes Care in the Hospital: Standards of Medical Care in Diabetes 2020. Diabetes Care 2020, 43, S193–S202. [Google Scholar] [CrossRef]
- American Diabetes Association. Glycemic Targets: Standards of Medical Care in Diabetes 2020. Diabetes Care 2020, 43, S66–S76. [Google Scholar] [CrossRef]
- International Hypoglycaemia Study Group. Minimizing Hypoglycemia in Diabetes. Diabetes Care 2015, 38, 1583–1591.
- Huang, K.; Brenner, W.; Prasad, V. Tumor Lysis Syndrome: A Rare but Serious Complication of Radioligand Therapies. J. Nucl. Med. 2019, 60, 752–755. [Google Scholar] [CrossRef]
- Study to Evaluate the Efficacy and Safety of Lutathera in Patients With Grade 2 and Grade 3 Advanced GEP-NET (NETTER-2). Available online: https://clinicaltrials.gov/ct2/show/NCT03972488 (accessed on 6 May 2020).
| WHO Classification | SSTR1 | SSTR2 | SSTR3 | SSTR4 | SSTR5 |
| G1 | + + + | + + + | + + | − | + + + |
| G2 | + + + | + + + | + + | − | + + |
| G3 | + + + | + + + | + + | − | − |
| Primary Site | SSTR1 | SSTR2 | SSTR3 | SSTR4 | SSTR5 |
| Pancreas | − | + + + | − | − | + + |
| Insulinoma | + | + + | + + | − | + + |
| Gastrinoma | + | + + + | + + | − | + + |
| Gastric | − | + + + | − | − | + + |
| Intestinal | − | + + | − | − | + + |
| Pulmonary | |||||
| Typical carcinoma | + + | + + + | + + | − | − |
| Atypical carcinoma | + + | + + | + | − | − |
| Large cell | + + | + + | + + | − | + |
| Small cell | + | + + | + | − | − |
| Pheochromocytoma | + + + | + + + | + + | − | + + |
| Paraganglioma | + + + | + + + | + + | − | + + |
| Type | Hormonal Crisis Described | Prevention Recommendations | Management Recommendations | Ref. |
|---|---|---|---|---|
| Retrospective review of efficacy of PRRT on metastatic PHEO and PGG in 20 patients | 1 catecholamine crisis (n = 20) | Adequate preparation with alpha- and beta-blockade is mandatory, with inpatient monitoring and access to intensive care support if required. Withholding dexamethasone as a premedication should also be considered because this may exacerbate hypertension in patients with PGG/PHEO | nr | [16] |
| Original article of a retrospective series of 479 patients treated with Lu-DOTATATE during 2000–2007 | 7 patients/479 1% incidence Carcinoid crisis VIPoma crisis catecholaminergic crisis (n = 479) Proposes physio-pathological explanation of these crisis | Alpha and beta blockade in metastatic pheochromocytoma Continuation of SSAs in risk patients Consider withholding of corticosteroids in PHEO/PGG | Fluids Octreotide Loperamide Metoclopramide Corticosteroids Potassium Bisoprolol (in PHEO) | [17] |
| Multicenter open-label single-arm trial phase II trial to establish efficacy and safety of MIBG in pheochromocytoma and paraganglioma | 0 hormonal crisis (n = 74) | Strict blood pressure control before treatment. No changes on antihypertensive drugs 30 days before treatment | nr | [21] |
| Case report and review of 3 paraganglioma and pheochromocytoma patients treated with LU-DOTA-TATE | 2/3 hormonal crisis Catecholaminergic crisis 1 required intensive care unit stay | Avoid corticosteroids during infusion Recommendation on lengthening or reduction of dose infusion of Lu-DOTA-TATE | Review on basic treatment of tumoral lysis syndrome and other electrolytic disturbances | [22] |
| Case report and review of the literature | Case report of carcinoid crisis after Lu-DOTATATE | nr | Treatment of a carcinoid crisis aims at preventing the release of the mediators from tumor tissue and/or blocking their effects on target organs. Intravenous administration of octreotide, corticosteroids, and correction of fluids, and electrolyte disturbances is the backbone of therapy | [23] |
| Case report | Case report of a carcinoid crisis triggered by PRRT. Death of the patient is reported. | (Pre)treatment with octreotide is recommended for therapeutic interventions in functional midgut NET. Other drugs have been successfully used, cyproheptadine, ketanserin, 5-HT receptor antagonists, corticosteroids, and H1-and H2-receptor antagonist. Somatostatin analogues are considered most effective and are recommended as first-line therapy | Treatment with nimodipine applied in the early phase of the ICU course seemed to be more effective compared to phentolamine. Intravenous octreotide applied in a dose of 500 μg/24 h continuously iv was also ineffective (recommended dose for carcinoid crisis 50–600 μg/day iv). | [24] |
| Phase II trial of efficacy of PRRT with Y90-DOTATATE in Patients with Advanced, Nonresectable Paraganglioma-Pheochromocytoma, Related to SDHx Gene Mutation 13 patients | No reported hormonal crisis | One of the mainstays to increase the safety of PRRT is that all patients with a hormonally functional PPGL should undergo a pretreatment blockade to prevent cardiovascular complications with alpha-adrenergic receptor blockers as the first choice. Clinicians should avoid medications that can trigger hemodynamic instability and cardiovascular events (for example, steroids, dopamine D2 receptor antagonists, sympathomimetics, selective serotonin reuptake inhibitors, opioid analgesics, tricyclic antidepressants, and others). | nr | [25] |
| Retrospective study on 504 patients treated with Lu-DOTA-TATE | 6 patients with hormonal crisis which required hospitalization | With adequate clinical scrutiny, patients who have an increased risk to develop hormone related crises can be identified and adequate measures to contain such events can be taken. Does not specify which. | nr | [26] |
| Literature review and cases series from two tertiary hospitals of carcinoid crisis after LU-DOTA-TATE | Seven cases of carcinoid crisis after PRRT | Identification of high-risk cases Correction of electrolyte disturbance, dehydration and hypoproteinemia before PRRT PRRT pre-medication | In the event of carcinoid crisis: octreotide in bolus or continuous infusion. H1 receptor blockers, H2 blockers, and occasionally, steroids. | [27] |
| BEFORE LU-DOTATE INFUSION | ||
|---|---|---|
| Identify Risk Factors for Carcinoid Crisis | Previous CS, Elevated 5HIAA, Chromogranin A, High Tumor Burden, Metastatic Disease (Mainly Hepatic), Carcinoid Heart Disease, Advanced Age, Histamine Secretion. | |
| Nutritional assessment [27] | Diagnose and correct hydro-electrolytic disorders | ACTION |
Check sodium, potassium, magnesium, phosphorus levels | ||
| Diagnose and correct malnutrition | Add vitamins and/or supplements | |
| Diagnose and correct malabsorption | Add pancreatic enzymes | |
| Avoid food triggers Avoid high intensity exercise the previous days | Recommend diet free of alcohol, spices, or foods rich in tryptophan | |
| Carcinoid tumor [23,26,27] | Tumor debulking | ACTION |
| Consider surgery, ablation, radiotherapy, or embolization | ||
Somatostatin analogs | Octreotide LAR 10–30 mg/28 days Lanreotide autogel 60–120 mg/28 days | |
| Other antitumoral treatments | ||
| Diarrhea [23,26,27] | Antidiarrheal drugs | ACTION |
| Loperamide 4–16 mg/day/oral Codeine 10–90 mg/day/oral | ||
| Anti-serotoninergic drugs Serotonin inhibitors | Cyproheptadine 4 mg/8 h Telotristat ethyl 250 mg/8 h/oral | |
| Etiopathogenic | Bile acid binders Antibiotics Pancreatic enzymes Niacin supplementation | |
| DURING LUDOTATATE INFUSION | ||
| Premedication [21,22,25,30,33,36] | Corticoid treatment | ACTION |
Dexametasone 4–8 mg if high risk patient | ||
| Antiemetic | Ondansetron 4 mg oral | |
| Somatostatin analogue | Octreotide 100 mcg sc or 50 mcg/iv if high risk patient | |
| Antihistaminic H1 | Dexchlorpheniramine 5 mg iv in slow infusion if high risk patient | |
| Antihistaminic H2 | Ranitidine 50 mg iv in slow infusion if high risk patient | |
| Carcinoid crisis [17,21,26,27,28,30,34,36,37] | Symptomatology control Monitor BP, HR, EKG Maintain volemia | ACTION |
| Stop Lu-DOTATATE infusion Octreotide 100–500 mcg sc or iv in saline, maintaining 50–100 mcg/h infusion Consider corticoid treatment (100 mg hydrocortisone or metilprednisolone1–2 mg/kg/iv slow infusion) Consider ICU If hypotension: phenylephrine or vasopresin (in ICU) If hypertension: α, β blockers Saline 0.9% infusion | ||
| Flushing, pruritus, uvula or facial edema [25,28,36] | Antihistaminic H1 | ACTION |
Dexchlorpheniramine 5–10 mg iv slow infusion, maintain 5 mg/6 h | ||
| Antihistaminic H2 | Ranitidine 50 mg iv slow infusion, maintain 50 mg/6–8 h slow infusion | |
| Diarrhea [17,28,30,37] | Monitor BP, HR, EKG | |
| Monitor electrolytes | Correct hydro-electrolytic disorders | |
| Monitor kidney function | Creatinine levels | |
| Monitor liver function | Hemostasia and transaminases | |
| Bronchospasm [37] | Avoid beta adrenergic stimuli | ACTION |
Avoid terbutalin, salbutamol, salmeterol, bambuterol, indacaterol, olodaterol, formoterol, salmeterol | ||
| Corticoid treatment | Hydrocortisone 100 mg iv slow infusion Metilrednisolone 1–2 mg/kg/iv slow infusion Beclomethasone 100–500 mcg inh Budesonide 200–400 mcg inh Fluticasone 100–250 mcg inh | |
| Anticholinergic | Bromure ipratropium 50–60 mg inh | |
| BEFORE LU-DOTATE INFUSION | |||
|---|---|---|---|
| Identify Risk Factors for Catecholamine Crisis | Tumors larger than 3–4 cm, uncontrolled blood pressure, high catecholamine levels, or pretreatment orthostatic hypotension | ||
| Nutritional assessment [27,42] | Diagnose and correct hydro-electrolytic disorders Diagnose and correct malnutrition Diagnose and correct constipation Avoid high intensity exercise the previous days | ACTION | |
| Check sodium, potassium, magnesium, phosphorus levels Add vitamins and/or supplements Specific diet for constipation | |||
| Catecholamine producing tumor [32,45] | Tumor debulking | ACTION | |
| Consider surgery, ablation, radiotherapy, or embolization | |||
Alpha adrenergic blockade | Phenoxybenzamine | Initial dose: 10 mg 1–2 times day Titration: 10–20 mg every 2–3 days Average daily dose: 20–100 mg/day Maximum dose: 240 mg/day | |
| Prazosin | Initial dose: 0.5–1 mg per dose every 4–6 h Average daily dose: 2–5 mg two or three times a day Maximum dose: 20–24 mg/day | ||
| Doxazosin | Initial dose: 1–2 mg/day TitrationMaximum dose: 16 mg/day | ||
| Terazosin | Initial dose: 1 mg/day Titration Average dose: 2–5 mg/day Maximum dose: 20 mg/day | ||
| Beta adrenergic blockade | Metoprolol | 25–50 mg three to four times a day | |
| Atenolol | 12.5–25 mg two to three times a day | ||
| Propranolol | 20–80 mg one to three times a day | ||
| Metyrosine | Initial dose: 250 mg orally every 8–12 h Titration Average dose: 1.5–2 gr per day High fluid intake to avoid crystalluria is suggested for patients taking more than 2 gr/day | ||
| Calcium channel blockers | Amlodipine | 10–20 mg/day | |
| Nicardipine | 60–90 mg/day | ||
| Verapamil | 180–540 mg/day | ||
| DURING LUDOTATATE INFUSION | |||
| Premedication [16,17,24,29] | Corticoid treatment | ACTION | |
AVOID | |||
| Antiemetic | Ondansetron 4 mg oral | ||
| Catecholamine crisis [24,29,45,46,47,48,49] | Symptomatology control | Stop Lu-DOTATATE infusion Consider slowing infusion rate over 2 h at least and preferably during 4 h | |
| Monitor BP, HR, EKG Maintain volemia | Saline 0.9% infusion | ||
| If hypertension | Captopril 50 mg oral | ||
| If severe hypertension | Sodium nitroprusside: 0.5–5.0 mcg/kg /min, Maximum dose 3 mcg/kg/min Phentolamine: initial dose of 1 mg, if necessary, repeat 5 mg boluses or continuous infusion Nicardipine started at 5 mg/h and titrated for blood pressure control (may be increased by 2.5 mg/h every 15 min up to a maximum of 15 mg/h). | ||
| If hypertension + tachycardia | Labetalol infusion 20 mg/iv in slow boluses every 5–10 min until a maximum dose of 300 mg. If continuous infusion is needed 250 mg in 250 mL of glucose 5% at a rhythm of 2–10 mg/min. | ||
| If cardiac arrhythmias | Lidocaine 50–100 mg intravenously Esmolol (50–200 mcg/kg/min intravenously) | ||
| If other complications or not control | Always consider ICU | ||
| General Recommendations | Before Infusion | Known Diabetes | Unknown Diabetes | After Infusion |
|---|---|---|---|---|
| Digital blood glucose [49] | Glucose test | Every h | Every two h | Before meals or every 6 h if fasting |
| Treated with oral drugs [49] | Do not administer oral DM drugs that morning | Correction with rapid insulin if indicated | Correction with rapid insulin if indicated | Restart before dinner if indicated |
| If Metformin, withdrawal 48 h before. DPP4 inhibitor, can be used | ||||
| Treated with insulin [49] | Administer Basal dose | Adjust with rapid insulin every 6 h | Restart before lunch if indicated | |
| Fluid therapy [49] | Start glucose 5% 500 mL/6 h or 10% 500/12 h | Stop glucose when oral tolerance | ||
| If Hypoglycemia [50] | Treatment according general recommendations | |||
| General Recommendations | Glucose 56–70 mg/dL | Glucose < 55 mg/dL or Neurologic Symptoms |
|---|---|---|
| Glucose test [50,51] | Every 15 min up to glucose > 80 mg/dL | Every 5 min up to glucose > 80 mg/dL |
| Glucose 10% [50,51] | 100 mL (glucose 10 g) in 5–10 min. Repeat if necessary. | |
| Glucose 50% [50,51] | 30 mL (Glucose 15 g) bolus. Repeat if necessary. | |
| Fluid [50,51] | Glucose 5–10% continuous, 500 mL/4–12 h Minimum 100 g of glucose in 24 h | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
del Olmo-García, M.I.; Muros, M.A.; López-de-la-Torre, M.; Agudelo, M.; Bello, P.; Soriano, J.M.; Merino-Torres, J.-F. Prevention and Management of Hormonal Crisis during Theragnosis with LU-DOTA-TATE in Neuroendocrine Tumors. A Systematic Review and Approach Proposal. J. Clin. Med. 2020, 9, 2203. https://doi.org/10.3390/jcm9072203
del Olmo-García MI, Muros MA, López-de-la-Torre M, Agudelo M, Bello P, Soriano JM, Merino-Torres J-F. Prevention and Management of Hormonal Crisis during Theragnosis with LU-DOTA-TATE in Neuroendocrine Tumors. A Systematic Review and Approach Proposal. Journal of Clinical Medicine. 2020; 9(7):2203. https://doi.org/10.3390/jcm9072203
Chicago/Turabian Styledel Olmo-García, Maria Isabel, Maria Angustias Muros, Martín López-de-la-Torre, Marc Agudelo, Pilar Bello, Jose M. Soriano, and Juan-Francisco Merino-Torres. 2020. "Prevention and Management of Hormonal Crisis during Theragnosis with LU-DOTA-TATE in Neuroendocrine Tumors. A Systematic Review and Approach Proposal" Journal of Clinical Medicine 9, no. 7: 2203. https://doi.org/10.3390/jcm9072203
APA Styledel Olmo-García, M. I., Muros, M. A., López-de-la-Torre, M., Agudelo, M., Bello, P., Soriano, J. M., & Merino-Torres, J.-F. (2020). Prevention and Management of Hormonal Crisis during Theragnosis with LU-DOTA-TATE in Neuroendocrine Tumors. A Systematic Review and Approach Proposal. Journal of Clinical Medicine, 9(7), 2203. https://doi.org/10.3390/jcm9072203






