Autism Spectrum Disorders in Greece: Nationwide Prevalence in 10–11 Year-Old Children and Regional Disparities
Abstract
1. Introduction
2. Population and Methods
2.1. Population
2.2. Source of the Data
2.3. Data Collection
2.4. Data Collection Tool
2.5. Case Definition
2.6. Age at Diagnosis
2.7. Statistical Analysis
2.8. Ethics
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fombonne, E. Editorial: The Rising Prevalence of Autism. J. Child Psychol. Psychiatry 2018, 59, 717–720. [Google Scholar] [CrossRef]
- American Psychological Association (APA). Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Publishing, Inc.: Arlington, VA, USA, 2013. [Google Scholar] [CrossRef]
- Fombonne, E. Epidemiological Surveys of Autism and Other Pervasive Developmental Disorders: An Update. J. Autism Dev. Disord. 2003, 33, 365–382. [Google Scholar] [CrossRef]
- Farley, M.A.; McMahon, W.M.; Fombonne, E.; Jenson, W.R.; Miller, J.; Gardner, M.; Block, H.; Pingree, C.B.; Ritvo, E.R.; Ritvo, R.A.; et al. Twenty-Year Outcome for Individuals with Autism and Average or near-Average Cognitive Abilities. Autism Res. 2009, 2, 109–118. [Google Scholar] [CrossRef] [PubMed]
- Hervás Zúñiga, A. One Autism, Several Autisms. Phenotypical Variability in Autism Spectrum Disorders. Rev. Neurol. 2016, 62 (Suppl. 1), 9. [Google Scholar] [CrossRef]
- World Health Organization. International Statistical Classification of Diseases and Related Health Problems, 10th ed.; World Health Organization: Geneva, Switzerland, 2010. [Google Scholar]
- Lord, C.; Brugha, T.S.; Charman, T.; Cusack, J.; Dumas, G.; Frazier, T.; Jones, E.J.H.; Jones, R.M.; Pickles, A.; State, M.W.; et al. Autism Spectrum Disorder. Nat. Rev. Dis. Prim. 2020, 6, 5. [Google Scholar] [CrossRef] [PubMed]
- Huguet, G.; Ey, E.; Bourgeron, T. The Genetic Landscapes of Autism Spectrum Disorders. Annu. Rev. Genomics Hum. Genet. 2013, 14, 191–213. [Google Scholar] [CrossRef]
- Modabbernia, A.; Velthorst, E.; Reichenberg, A. Environmental Risk Factors for Autism: An Evidence-Based Review of Systematic Reviews and Meta-Analyses. Mol. Autism 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Taylor, M.J.; Rosenqvist, M.A.; Larsson, H.; Gillberg, C.; D’Onofrio, B.M.; Lichtenstein, P.; Lundström, S. Etiology of Autism Spectrum Disorders and Autistic Traits Over Time. JAMA Psychiatry 2020. [Google Scholar] [CrossRef]
- Fombonne, E. The Epidemiology of Autism: A Review. Psychol. Med. 1999, 29, 769–786. [Google Scholar] [CrossRef]
- Pérez-Crespo, L.; Prats-Uribe, A.; Tobias, A.; Duran-Tauleria, E.; Coronado, R.; Hervás, A.; Guxens, M. Temporal and Geographical Variability of Prevalence and Incidence of Autism Spectrum Disorder Diagnoses in Children in Catalonia, Spain. Autism Res. 2019, 12, 1693–1705. [Google Scholar] [CrossRef]
- Elsabbagh, M.; Divan, G.; Koh, Y.J.; Kim, Y.S.; Kauchali, S.; Marcín, C.; Montiel-Nava, C.; Patel, V.; Paula, C.S.; Wang, C.; et al. Global Prevalence of Autism and Other Pervasive Developmental Disorders. Autism Res. 2012, 5, 160–179. [Google Scholar] [CrossRef]
- Baxter, A.J.; Brugha, T.S.; Erskine, H.E.; Scheurer, R.W.; Vos, T.; Scott, J.G. The Epidemiology and Global Burden of Autism Spectrum Disorders. Psychol. Med. 2015, 45, 601–613. [Google Scholar] [CrossRef]
- Lyall, K.; Croen, L.; Daniels, J.; Fallin, M.D.; Ladd-Acosta, C.; Lee, B.K.; Park, B.Y.; Snyder, N.W.; Schendel, D.; Volk, H.; et al. The Changing Epidemiology of Autism Spectrum Disorders. Annu. Rev. Public Health 2017, 38, 81–102. [Google Scholar] [CrossRef]
- Onaolapo, Y.A.; Onaolapo, J.O. Global Data on Autism Spectrum Disorders Prevalence: A Review of Facts, Fallacies and Limitations. Univers. J. Clin. Med. 2017, 5, 14–23. [Google Scholar] [CrossRef]
- Loomes, R.; Hull, L.; Mandy, W.P.L. What Is the Male-to-Female Ratio in Autism Spectrum Disorder? A Systematic Review and Meta-Analysis. J. Am. Acad. Child Adolesc. Psychiatry 2017, 56, 466–474. [Google Scholar] [CrossRef] [PubMed]
- Maenner, M.J.; Shaw, K.A.; Baio, J.; Washington, A.; Patrick, M.; DiRienzo, M.; Christensen, D.L.; Wiggins, L.D.; Pettygrove, S.; Andrews, J.G.; et al. Prevalence of Autism Spectrum Disorder Among Children Aged 8 Years—Autism and Developmental Disabilities Monitoring Network, 11 Sites, United States, 2016. MMWR. Surveill. Summ. 2020, 69, 1–12. [Google Scholar] [CrossRef]
- Zablotsky, B.; Colpe, L.J.; Pringle, B.A.; Kogan, M.D.; Rice, C.; Blumberg, S.J. Age of Parental Concern, Diagnosis, and Service Initiation among Children with Autism Spectrum Disorder. Am. J. Intellect. Dev. Disabil. 2017, 122, 49–61. [Google Scholar] [CrossRef]
- Vivanti, G.; Prior, M.; Williams, K.; Dissanayake, C. Predictors of Outcomes in Autism Early Intervention: Why Don’t We Know More? Front. Pediatr. 2014, 2, 58. [Google Scholar] [CrossRef]
- Wetherby, A.M.; Woods, J.; Guthrie, W.; Delehanty, A.; Brown, J.A.; Morgan, L.; Holland, R.D.; Schatschneider, C.; Lord, C. Changing Developmental Trajectories of Toddlers With Autism Spectrum Disorder: Strategies for Bridging Research to Community Practice. J. Speech Lang. Hear. Res. 2018, 61, 2615–2628. [Google Scholar] [CrossRef]
- Guthrie, W.; Wallis, K.; Bennett, A.; Brooks, E.; Dudley, J.; Gerdes, M.; Pandey, J.; Levy, S.E.; Schultz, R.T.; Miller, J.S. Accuracy of Autism Screening in a Large Pediatric Network. Pediatrics 2019, 144, e20183963. [Google Scholar] [CrossRef]
- Pierce, K.; Gazestani, V.H.; Bacon, E.; Barnes, C.C.; Cha, D.; Nalabolu, S.; Lopez, L.; Moore, A.; Pence-Stophaeros, S.; Courchesne, E. Evaluation of the Diagnostic Stability of the Early Autism Spectrum Disorder Phenotype in the General Population Starting at 12 Months. JAMA Pediatr. 2019, 173, 578–587. [Google Scholar] [CrossRef] [PubMed]
- Daniels, A.M.; Mandell, D.S. Explaining Differences in Age at Autism Spectrum Disorder Diagnosis: A Critical Review. Autism 2014, 18, 583–597. [Google Scholar] [CrossRef]
- Jo, H.; Schieve, L.A.; Rice, C.E.; Yeargin-Allsopp, M.; Tian, L.H.; Blumberg, S.J.; Kogan, M.D.; Boyle, C.A. Age at Autism Spectrum Disorder (ASD) Diagnosis by Race, Ethnicity, and Primary Household Language Among Children with Special Health Care Needs, United States, 2009–2010. Matern. Child Health J. 2015, 19, 1687–1697. [Google Scholar] [CrossRef]
- Hansen, S.N.; Schendel, D.E.; Parner, E.T. Explaining the Increase in the Prevalence of Autism Spectrum Disorders: The Proportion Attributable to Changes in Reporting Practices. JAMA Pediatr. 2015, 169, 56–62. [Google Scholar] [CrossRef]
- Wang, F.; Lu, L.; Wang, S.B.; Zhang, L.; Ng, C.H.; Ungvari, G.S.; Cao, X.L.; Lu, J.P.; Hou, C.L.; Jia, F.J.; et al. The Prevalence of Autism Spectrum Disorders in China: A Comprehensive Meta-Analysis. Int. J. Biol. Sci. 2018, 14, 717–725. [Google Scholar] [CrossRef] [PubMed]
- India State-Level Disease Burden Initiative Mental Disorders Collaborators. The Burden of Mental Disorders across the States of India: The Global Burden of Disease Study 1990–2017. Lancet Psychiatry 2019, 7, 148–161. [Google Scholar] [CrossRef]
- Al-Mamri, W.; Idris, A.B.; Dakak, S.; Al-Shekaili, M.; Al-Harthi, Z.; Alnaamani, A.M.; Alhinai, F.I.; Jalees, S.; Al Hatmi, M.; El-Naggari, M.A.; et al. Revisiting the Prevalence of Autism Spectrum Disorder among Omani Children: A Multicentre Study. Sultan Qaboos Univ. Med. J. 2019, 19, e305–e309. [Google Scholar] [CrossRef] [PubMed]
- Surén, P.; Havdahl, A.; Øyen, A.-S.; Schjølberg, S.; Reichborn-Kjennerud, T.; Magnus, P.; Bakken, I.J.L.; Stoltenberg, C. Diagnosing Autism Spectrum Disorder among Children in Norway. Tidsskr. Den Nor. Legeforening 2019, 139. [Google Scholar] [CrossRef]
- Oliveira, G.; Ataíde, A.; Marques, C.; Miguel, T.S.; Coutinho, A.M.; Mota-vieira, L.; Gonçalves, E.; Lopes, N.M.; Rodrigues, V.; Carmona Da Mota, H.; et al. Epidemiology of Autism Spectrum Disorder in Portugal: Prevalence, Clinical Characterization, and Medical Conditions. Dev. Med. Child Neurol. 2007, 49, 726–733. [Google Scholar] [CrossRef]
- Siddiqua, A.; Duku, E.; Georgiades, K.; Mesterman, R.; Janus, M. Neighbourhood-Level Prevalence of Teacher-Reported Autism Spectrum Disorder among Kindergarten Children in Canada: A Population Level Study. SSM-Popul. Health 2020, 10, 100520. [Google Scholar] [CrossRef]
- Baio, J.; Wiggins, L.; Christensen, D.L.; Maenner, M.J.; Daniels, J.; Warren, Z.; Kurzius-Spencer, M.; Zahorodny, W.; Rosenberg, C.R.; White, T.; et al. Prevalence of Autism Spectrum Disorder among Children Aged 8 Years—Autism and Developmental Disabilities Monitoring Network, 11 Sites, United States, 2014. MMWR Surveill. Summ. 2018, 67, 1. [Google Scholar] [CrossRef] [PubMed]
- Ofner, M.; Coles, A.; Decou, M.L.; Do, M.T.; Bienek, A.; Snider, J.; Ugnat, A.-M. Autism Spectrum Disorder Among Children and Youth in Canada 2018. Available online: https://www.canada.ca/en/public-health/services/publications/diseases-conditions/autism-spectrum-disorder-children-youth-canada-2018.html (accessed on 14 May 2020).
- Lundström, S.; Reichenberg, A.; Anckarsäter, H.; Lichtenstein, P.; Gillberg, C. Autism Phenotype versus Registered Diagnosis in Swedish Children: Prevalence Trends over 10 Years in General Population Samples. BMJ 2015, 350, h1961. [Google Scholar] [CrossRef] [PubMed]
- Delobel-Ayoub, M.; Saemundsen, E.; Gissler, M.; Ego, A.; Moilanen, I.; Ebeling, H.; Rafnsson, V.; Klapouszczak, D.; Thorsteinsson, E.; Arnaldsdóttir, K.M.; et al. Prevalence of Autism Spectrum Disorder in 7-9-Year-Old Children in Denmark, Finland, France and Iceland: A Population-Based Registries Approach Within the ASDEU Project. J. Autism Dev. Disord. 2019, 50, 949–959. [Google Scholar] [CrossRef] [PubMed]
- Baron-Cohen, S.; Scott, F.J.; Allison, C.; Williams, J.; Bolton, P.; Matthews, F.E.; Brayne, C. Prevalence of Autism-Spectrum Conditions: UK School-Based Population Study. Br. J. Psychiatry 2009, 194, 500–509. [Google Scholar] [CrossRef] [PubMed]
- Boilson, A.M.; Staines, A.; Ramirez, A.; Posada, M.; Sweeney, M.R. Operationalisation of the European Protocol for Autism Prevalence (EPAP) for Autism Spectrum Disorder Prevalence Measurement in Ireland. J. Autism Dev. Disord. 2016, 46, 3054–3067. [Google Scholar] [CrossRef]
- Fortea Sevilla, M.S.; Escandell Bermúdez, M.O.; Castro Sánchez, J.J. Estimating the Prevalence of Autism Spectrum Disorders in CanaryEstimated Prevalence of Autism Spectrum Disorders in the Canary Islands. An. Pediatr. 2013, 79, 352–359. [Google Scholar] [CrossRef] [PubMed]
- Pantelis, P.C.; Kennedy, D.P. Estimation of the Prevalence of Autism Spectrum Disorder in South Korea, Revisited. Autism 2016, 20, 517–527. [Google Scholar] [CrossRef]
- Chan, H.-L.; Liu, W.-S.; Hsieh, Y.-H.; Lin, C.-F.; Ling, T.-S.; Huang, Y. Screening for Attention Deficit and Hyperactivity Disorder, Autism Spectrum Disorder, and Developmental Delay in Taiwanese Aboriginal Preschool Children. Neuropsychiatr. Dis. Treat. 2016, 12, 2521–2526. [Google Scholar] [CrossRef] [PubMed]
- Fombonne, E.; Marcin, C.; Manero, A.C.; Bruno, R.; Diaz, C.; Villalobos, M.; Ramsay, K.; Nealy, B. Prevalence of Autism Spectrum Disorders in Guanajuato, Mexico: The Leon Survey. J. Autism Dev. Disord. 2016, 46, 1669–1685. [Google Scholar] [CrossRef]
- Zablotsky, B.; Black, L.I.; Maenner, M.J.; Schieve, L.A.; Blumberg, S.J. Estimated Prevalence of Autism and Other Developmental Disabilities Following Questionnaire Changes in the 2014 National Health Interview Survey. Natl. Health Stat. Rep. 2015, 1–20. [Google Scholar]
- Kogan, M.D.; Blumberg, S.J.; Schieve, L.A.; Boyle, C.A.; Perrin, J.M.; Ghandour, R.M.; Singh, G.K.; Strickland, B.B.; Trevathan, E.; Van Dyck, P.C. Prevalence of Parent-Reported Diagnosis of Autism Spectrum Disorder among Children in the US, 2007. Pediatrics 2009, 124, 1395–1403. [Google Scholar] [CrossRef]
- Catalá-López, F.; Ridao, M.; Hurtado, I.; Núñez-Beltrán, A.; Gènova-Maleras, R.; Alonso-Arroyo, A.; Tobías, A.; Aleixandre-Benavent, R.; Catalá, M.A.; Tabarés-Seisdedos, R. Prevalence and Comorbidity of Autism Spectrum Disorder in Spain: Study Protocol for a Systematic Review and Meta-Analysis of Observational Studies. Syst. Rev. 2019, 8, 141. [Google Scholar] [CrossRef]
- Rydzewska, E.; Hughes-McCormack, L.A.; Gillberg, C.; Henderson, A.; MacIntyre, C.; Rintoul, J.; Cooper, S.-A.A. Age at Identification, Prevalence and General Health of Children with Autism: Observational Study of a Whole Country Population. BMJ Open 2019, 9, e025904. [Google Scholar] [CrossRef] [PubMed]
- Christensen, D.L.; Bilder, D.A.; Zahorodny, W.; Pettygrove, S.; Durkin, M.S.; Fitzgerald, R.T.; Rice, C.; Kurzius-Spencer, M.; Baio, J.; Yeargin-Allsopp, M. Prevalence and Characteristics of Autism Spectrum Disorder among 4-Year-Old Children in the Autism and Developmental Disabilities Monitoring Network. J. Dev. Behav. Pediatr. 2016, 37, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Málaga, I.; Lago, R.B.; Hedrera-Fernández, A.; Álvarez-álvarez, N.; Oreña-Ansonera, V.A.; Baeza-Velasco, M. Prevalence of Autism Spectrum Disorders in USA, Europe and Spain: Coincidences and Discrepancies. Medicina 2019, 79, 4–9. [Google Scholar]
- Ferrante, M.; Barone, R.; Fazio, A.; Zerbo, S.; Margherita, V.; Rizzo, R.; Fiore, M. Prevalence and Age at Diagnosis of Autism Spectrum Disorder in South Italy, 2004–2014. Eur. J. Public Health 2015, 25 (Suppl. 3), ckv175.099. [Google Scholar] [CrossRef]
- Matson, J.L.; Matheis, M.; Burns, C.O.; Esposito, G.; Venuti, P.; Pisula, E.; Misiak, A.; Kalyva, E.; Tsakiris, V.; Kamio, Y.; et al. Examining Cross-Cultural Differences in Autism Spectrum Disorder: A Multinational Comparison from Greece, Italy, Japan, Poland, and the United States. Eur. Psychiatry 2017, 42, 70–76. [Google Scholar] [CrossRef]
- Narzisi, A.; Posada, M.; Barbieri, F.; Chericoni, N.; Ciuffolini, D.; Pinzino, M.; Romano, R.; Scattoni, M.L.; Tancredi, R.; Calderoni, S.; et al. Prevalence of Autism Spectrum Disorder in a Large Italian Catchment Area: A School-Based Population Study within the ASDEU Project. Epidemiol. Psychiatr. Sci. 2018, 29, e5. [Google Scholar] [CrossRef]
- Fombonne, E. Is There an Epidemic of Autism? Pediatrics 2001, 107, 411–413. [Google Scholar] [CrossRef]
- Nassar, N.; Dixon, G.; Bourke, J.; Bower, C.; Glasson, E.; De Klerk, N.; Leonard, H. Autism Spectrum Disorders in Young Children: Effect of Changes in Diagnostic Practices. Int. J. Epidemiol. 2009, 38, 1245–1254. [Google Scholar] [CrossRef]
- Efthimiou, A.; Skounti, M.; Philalithis, A. Prevalence and Comorbidity of the Autistic Disorder in School Age Children in Crete-Greece. Eur. Psychiatry 2013, 28, 1. [Google Scholar] [CrossRef]
- Manuel Posada de la Paz. Autism Spectrum Disorders in the European Union (ASDEU). Available online: http://asdeu.eu/wp-content/uploads/2016/12/ASDEUExecSummary27September2018.pdf (accessed on 6 June 2020).
- Surén, P.; Bakken, I.J.; Aase, H.; Chin, R.; Gunnes, N.; Lie, K.K.; Magnus, P.; Reichborn-Kjennerud, T.; Schjølberg, S.; Øyen, A.S.; et al. Autism Spectrum Disorder, ADHD, Epilepsy, and Cerebral Palsy in Norwegian Children. Pediatrics 2012, 130, e152–e158. [Google Scholar] [CrossRef] [PubMed]
- Saemundsen, E.; Magnússon, P.; Georgsdóttir, I.; Egilsson, E.; Rafnsson, V. Prevalence of Autism Spectrum Disorders in an Icelandic Birth Cohort. BMJ Open 2013, 3, e002748. [Google Scholar] [CrossRef]
- Idring, S.; Lundberg, M.; Sturm, H.; Dalman, C.; Gumpert, C.; Rai, D.; Lee, B.K.; Magnusson, C. Changes in Prevalence of Autism Spectrum Disorders in 2001–2011: Findings from the Stockholm Youth Cohort. J. Autism Dev. Disord. 2015, 45, 1766–1773. [Google Scholar] [CrossRef]
- Valenti, M.; Vagnetti, R.; Masedu, F.; Pino, M.C.; Rossi, A.; Scattoni, M.L.; Mazza, M.; Di Giovanni, C.; Attanasio, M.; Filocamo, A.; et al. Register-Based Cumulative Prevalence of Autism Spectrum Disorders during Childhood and Adolescence in Central Italy. Epidemiol. Biostat. Public Health 2019, 16, e13226-1–e13226-7. [Google Scholar] [CrossRef]
- Skonieczna-Żydecka, K.; Gorzkowska, I.; Pierzak-Sominka, J.; Adler, G. The Prevalence of Autism Spectrum Disorders in West Pomeranian and Pomeranian Regions of Poland. J. Appl. Res. Intellect. Disabil. 2017, 30, 283–289. [Google Scholar] [CrossRef]
- Morales-Hidalgo, P.; Roigé-Castellví, J.; Hernández-Martínez, C.; Voltas, N.; Canals, J. Prevalence and Characteristics of Autism Spectrum Disorder Among Spanish School-Age Children. J. Autism Dev. Disord. 2018, 48, 3176–3190. [Google Scholar] [CrossRef]
- Kim, S.K. Recent Update of Autism Spectrum Disorders. Korean, J. Pediatr. 2015, 58, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Allison, C.; Matthews, F.E.; Zhang, Z.; Auyeung, B.; Baron-Cohen, S.; Brayne, C. Exploring the Underdiagnosis and Prevalence of Autism Spectrum Conditions in Beijing. Autism Res. 2015, 8, 250–260. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Allison, C.; Wei, L.; Matthews, F.E.; Auyeung, B.; Wu, Y.Y.; Griffiths, S.; Zhang, J.; Baron-Cohen, S.; Brayne, C. Autism Prevalence in China Is Comparable to Western Prevalence. Mol. Autism 2019, 10, 7. [Google Scholar] [CrossRef] [PubMed]
- Xu, G.; Strathearn, L.; Liu, B.; Bao, W. Prevalence of Autism Spectrum Disorder among US Children and Adolescents, 2014-2016. JAMA 2018, 319, 81–82. [Google Scholar] [CrossRef] [PubMed]
- May, T.; Sciberras, E.; Brignell, A.; Williams, K. Autism Spectrum Disorder: Updated Prevalence and Comparison of Two Birth Cohorts in a Nationally Representative Australian Sample. BMJ Open 2017, 7, e015549. [Google Scholar] [CrossRef] [PubMed]
- Chiarotti, F.; Venerosi, A. Epidemiology of Autism Spectrum Disorders: A Review of Worldwide Prevalence Estimates since 2014. Brain Sci. 2020, 10, 274. [Google Scholar] [CrossRef]
- Werling, D.M. The Role of Sex-Differential Biology in Risk for Autism Spectrum Disorder. Biol. Sex Differ. 2016, 7, 58. [Google Scholar] [CrossRef] [PubMed]
- Baron-Cohen, S.; Lombardo, M.V.; Auyeung, B.; Ashwin, E.; Chakrabarti, B.; Knickmeyer, R. Why Are Autism Spectrum Conditions More Prevalent in Males? PLoS Biol. 2011, 9, e1001081. [Google Scholar] [CrossRef]
- Cauvet, É.; Van’T Westeinde, A.; Toro, R.; Kuja-Halkola, R.; Neufeld, J.; Mevel, K.; Bölte, S. Sex Differences along the Autism Continuum: A Twin Study of Brain Structure. Cereb. Cortex 2019, 29, 1342–1350. [Google Scholar] [CrossRef]
- Dean, M.; Harwood, R.; Kasari, C. The Art of Camouflage: Gender Differences in the Social Behaviors of Girls and Boys with Autism Spectrum Disorder. Autism 2017, 21, 678–689. [Google Scholar] [CrossRef]
- Lai, M.C.; Lombardo, M.V.; Auyeung, B.; Chakrabarti, B.; Baron-Cohen, S. Sex/Gender Differences and Autism: Setting the Scene for Future Research. J. Am. Acad. Child Adolesc. Psychiatry 2015, 54, 11–24. [Google Scholar] [CrossRef]
- Hiller, R.M.; Young, R.L.; Weber, N. Sex Differences in Autism Spectrum Disorder Based on DSM-5 Criteria: Evidence from Clinician and Teacher Reporting. J. Abnorm. Child Psychol. 2014, 42, 1381–1393. [Google Scholar] [CrossRef]
- Ozonoff, S.; Young, G.S.; Brian, J.; Charman, T.; Shephard, E.; Solish, A.; Zwaigenbaum, L. Diagnosis of Autism Spectrum Disorder After Age 5 in Children Evaluated Longitudinally Since Infancy HHS Public Access. J. Am. Acad. Child Adolesc. Psychiatry 2018, 57, 849–857. [Google Scholar] [CrossRef]
- Mandell, D.S.; Morales, K.H.; Xie, M.; Lawer, L.J.; Stahmer, A.C.; Marcus, S.C. Age of Diagnosis among Medicaid-Enrolled Children with Autism, 2001-2004. Psychiatr. Serv. 2010, 61, 822–829. [Google Scholar] [CrossRef]
- Brett, D.; Warnell, F.; McConachie, H.; Parr, J.R. Factors Affecting Age at ASD Diagnosis in UK: No Evidence That Diagnosis Age Has Decreased Between 2004 and 2014. J. Autism Dev. Disord. 2016, 46, 1974–1984. [Google Scholar] [CrossRef]
- Belinchón, M.; Hernández, J.M.; Sotillo, M. Personas Con Síndrome de As-Perger: Funcionamiento, Detección y Necesidades. [People with Asperger Syndrome: Functioning, Detection and Needs]; Centro de Psicología Aplicada de la Universidad Autónoma de Madrid (CPA-UAM): Madrid, Spain, 2008. [Google Scholar]
- Valicenti-Mcdermott, M.; Hottinger, K.; Seijo, R.; Shulman, L. Age at Diagnosis of Autism Spectrum Disorders. J. Pediatr. 2012, 161, 554–556. [Google Scholar] [CrossRef]
- Maenner, M.J.; Schieve, L.A.; Rice, C.E.; Cunniff, C.; Giarelli, E.; Kirby, R.S.; Lee, L.C.; Nicholas, J.S.; Wingate, M.S.; Durkin, M.S. Frequency and Pattern of Documented Diagnostic Features and the Age of Autism Identification. J. Am. Acad. Child Adolesc. Psychiatry 2013, 52, 401–413. [Google Scholar] [CrossRef]
- Olsson, I.; Roll-Pettersson, L. “No No, You Cannot Say That!” Perceptions and Experiences of Parents of Preschool Children with Intellectual Disabilities in Sweden. Eur. J. Spec. Needs Educ. 2012, 27, 69–80. [Google Scholar] [CrossRef][Green Version]
- Sheldrick, R.C.; Maye, M.P.; Carter, A.S. Age at First Identification of Autism Spectrum Disorder: An Analysis of Two US Surveys. J. Am. Acad. Child Adolesc. Psychiatry 2017, 56, 313–320. [Google Scholar] [CrossRef] [PubMed]
- Kurasawa, S.; Tateyama, K.; Iwanaga, R.; Ohtoshi, T.; Nakatani, K.; Yokoi, K. The Age at Diagnosis of Autism Spectrum Disorder in Children in Japan. Int. J. Pediatr. 2018, 2018, 5374725. [Google Scholar] [CrossRef]
- Spikol, A.; McAteer, D.; Murphy, J. Recognising Autism: A Latent Transition Analysis of Parental Reports of Child Autistic Spectrum Disorder ‘Red Flag’ Traits before and after Age 3. Soc. Psychiatry Psychiatr. Epidemiol. 2019, 54, 703–713. [Google Scholar] [CrossRef] [PubMed]
- Nygren, G.; Cederlund, M.; Sandberg, E.; Gillstedt, F.; Arvidsson, T.; Carina Gillberg, I.; Westman Andersson, G.; Gillberg, C. The Prevalence of Autism Spectrum Disorders in Toddlers: A Population Study of 2-Year-Old Swedish Children. J. Autism Dev. Disord. 2012, 42, 1491–1497. [Google Scholar] [CrossRef] [PubMed]
- Fountain, C.; King, M.D.; Bearman, P.S. Age of Diagnosis for Autism: Individual and Community Factors across 10 Birth Cohorts. J. Epidemiol. Community Health 2011, 65, 503–510. [Google Scholar] [CrossRef] [PubMed]
- Delobel-Ayoub, M.; Ehlinger, V.; Klapouszczak, D.; Maffre, T.; Raynaud, J.P.; Delpierre, C.; Arnaud, C. Socioeconomic Disparities and Prevalence of Autism Spectrum Disorders and Intellectual Disability. PLoS ONE 2015, 10, e0141964. [Google Scholar] [CrossRef] [PubMed]
- Tountas, Y.; Oikonomou, N.; Pallikarona, G.; Dimitrakaki, C.; Tzavara, C.; Souliotis, K.; Mariolis, A.; Pappa, E.; Kontodimopoulos, N.; Niakas, D. Sociodemographic and Socioeconomic Determinants of Health Services Utilization in Greece: The Hellas Health I Study. Health Serv. Manag. Res. 2011, 24, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Filippidis, F.T.; Gerovasili, V.; Millett, C.; Tountas, Y. Medium-Term Impact of the Economic Crisis on Mortality, Health-Related Behaviours and Access to Healthcare in Greece. Sci. Rep. 2017, 7, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Robins, D.L.; Fein, D.; Barton, M.L.; Green, J.A. The Modified Checklist for Autism in Toddlers: An Initial Study Investigating the Early Detection of Autism and Pervasive Developmental Disorders. J. Autism Dev. Disord. 2001, 31, 131–144. [Google Scholar] [CrossRef]
- Thomaidis, L.; Choleva, A.; Kyprianou, M. Age Adjustment of the Psychological Development Questionnaire-1. J. Dev. Behav. Pediatr. 2018, 39, 766. [Google Scholar] [CrossRef]
- Thomaidis, L.; Choleva, A.; Kyprianou, M. Age-Related Issues of Instruments Screening for Autism in Young Children. Neuropsychiatr. Dis. Treat. 2016, 12, 3093–3095. [Google Scholar] [CrossRef]
- Thomaidis, L.; Kyprianou, M.; Choleva, A. Early Screening of Autism: Is Age a Confounding Factor When Screening for Autism? J. Paediatr. Child Health 2015, 51, 1046–1047. [Google Scholar] [CrossRef]
- Davidovitch, M.; Levit-Binnun, N.; Golan, D.; Manning-Courtney, P. Late Diagnosis of Autism Spectrum Disorder after Initial Negative Assessment by a Multidisciplinary Team. J. Dev. Behav. Pediatr. 2015, 36, 227–234. [Google Scholar] [CrossRef]
- Berger, N.I.; Ingersoll, B. An Exploration of Imitation Recognition in Young Children with Autism Spectrum Disorders. Autism Res. 2013, 6, 411–416. [Google Scholar] [CrossRef]
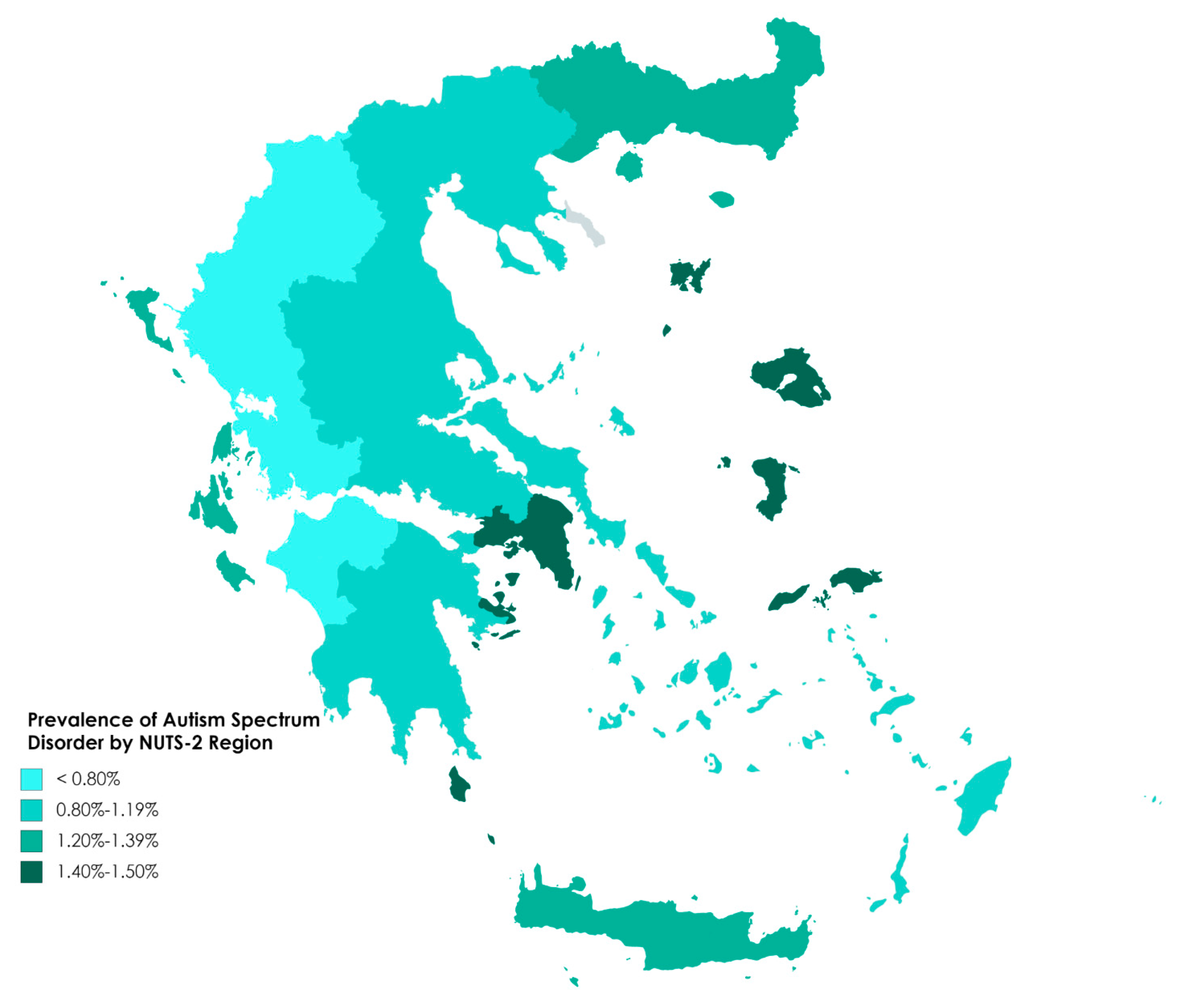

| Administrative Region | N * | ASD ** | |
|---|---|---|---|
| n | % | ||
| North Aegean | 3070 | 46 | 1.50 |
| Attica | 57,186 | 837 | 1.46 |
| Ionian Islands | 4606 | 64 | 1.39 |
| Crete | 15,052 | 203 | 1.35 |
| Eastern Macedonia and Thrace | 9920 | 121 | 1.22 |
| Central Macedonia | 31,072 | 317 | 1.02 |
| Central Greece | 9636 | 97 | 1.01 |
| Thessaly | 14,372 | 133 | 0.93 |
| Peloponnese | 9160 | 83 | 0.91 |
| South Aegean | 6835 | 58 | 0.85 |
| Epirus | 6107 | 48 | 0.79 |
| Western Macedonia | 5149 | 38 | 0.74 |
| Western Greece | 10,714 | 63 | 0.59 |
| Total Greece | 18,2879 | 2108 | 1.15 |
| Administrative Region | Males | Females | M/F Ratio | ||||
|---|---|---|---|---|---|---|---|
| N * | ASD ** | N * | ASD | ||||
| n | % | n | % | ||||
| North Aegean | 1567 | 42 | 2.68 | 1503 | 4 | 0.27 | 10.1 |
| Attica | 29,349 | 672 | 2.29 | 27,837 | 165 | 0.59 | 3.9 |
| Ionian Islands | 2418 | 52 | 2.15 | 2188 | 12 | 0.55 | 3.9 |
| Crete | 7651 | 169 | 2.21 | 7401 | 34 | 0.46 | 4.8 |
| Eastern Macedonia & Thrace | 5074 | 91 | 1.79 | 4846 | 30 | 0.62 | 2.9 |
| Central Macedonia | 15,843 | 257 | 1.62 | 15,229 | 60 | 0.39 | 4.1 |
| Central Greece | 5024 | 78 | 1.55 | 4612 | 19 | 0.41 | 3.8 |
| Thessaly | 7371 | 115 | 1.56 | 7001 | 18 | 0.26 | 6.1 |
| Peloponnese | 4703 | 62 | 1.32 | 4457 | 21 | 0.47 | 2.8 |
| South Aegean | 3464 | 48 | 1.39 | 3371 | 10 | 0.30 | 4.6 |
| Epirus | 3210 | 41 | 1.28 | 2897 | 7 | 0.24 | 5.3 |
| Western Macedonia | 2663 | 32 | 1.20 | 2486 | 6 | 0.24 | 5.0 |
| Western Greece | 5560 | 56 | 1.01 | 5154 | 7 | 0.14 | 7.4 |
| Total Greece | 93,897 | 1715 | 1.83 | 88,982 | 393 | 0.44 | 4.14 |
| Year (Mean Age) * | School Grade | Males % | Females % | Total% | Cumulative | ||
|---|---|---|---|---|---|---|---|
| Males % | Females % | Total % | |||||
| 3 | - | 3.7 | 4.1 | 3.8 | 3.7 | 4.1 | 3.8 |
| 4 | Pre-school | 13.2 | 14.6 | 13.5 | 16.9 | 18.7 | 17.3 |
| 5 | Kindergarten | 24.4 | 30.3 | 25.5 | 41.3 | 49.0 | 42.7 |
| 6 | 1st | 21.1 | 20.3 | 20.9 | 62.3 | 69.2 | 63.6 |
| 7 | 2nd | 16.6 | 15.9 | 16.5 | 78.9 | 85.1 | 80.1 |
| 8 | 3rd | 9.3 | 6.2 | 8.7 | 88.2 | 91.3 | 88.8 |
| 9 | 4th | 7.7 | 5.4 | 7.3 | 95.9 | 96.7 | 96.1 |
| 10 | 5th | 4.1 | 3.3 | 3.9 | 100 | 100 | 100 |
| Total | 100 | 100 | 100 | ||||
| Year (Mean Age) * | School Grade | Males % | Females % | Total% | |
|---|---|---|---|---|---|
| Male-to-Female Ratio | |||||
| 3 | - | 0.07 | 0.02 | 0.04 | 3.73 |
| 4 | Pre-school | 0.30 | 0.08 | 0.20 | 3.71 |
| 5 | Kindergarten | 0.74 | 0.21 | 0.49 | 3.46 |
| 6 | 1st | 1.12 | 0.30 | 0.72 | 3.70 |
| 7 | 2nd | 1.42 | 0.37 | 0.91 | 3.81 |
| 8 | 3rd | 1.59 | 0.40 | 1.01 | 3.97 |
| 9 | 4th | 1.73 | 0.42 | 1.09 | 4.07 |
| 10 | 5th | 1.80 | 0.44 | 1.14 | 4.11 |
| Region | Number Diagnosed | Mean Age at Diagnosis | Modal Year |
|---|---|---|---|
| Epirus | 48 | 5y2m | 5 |
| Western Macedonia | 38 | 5y3m | 4 |
| North Aegean | 46 | 5y4m | 5 |
| Attica | 837 | 5y7m | 5 |
| Peloponnese | 83 | 5y11m | 5 |
| Crete | 203 | 6y2m | 6 |
| Thessaly | 133 | 6y2m | 5 |
| E Macedonia & Thrace | 121 | 6y3m | 5 |
| Central Greece | 97 | 6y7m | 7 |
| South Aegean | 58 | 6y9m | 6 |
| Ionian Islands | 64 | 6y10m | 7 |
| Central Macedonia | 317 | 7y0m | 6 |
| Western Greece | 63 | 7y0m | 7 |
| Total Greece | 2108 | 6y1m | 5 |
| Region | Cumulative Frequency (%) by Year of ASD Diagnosis | |||||||
|---|---|---|---|---|---|---|---|---|
| 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | |
| Epirus | 10.4 | 31.3 | 66.7 | 87.5 | 91.7 | 100 | 100 | 100 |
| Western Macedonia | 7.9 | 44.7 | 65.8 | 79.0 | 86.8 | 97.4 | 97.4 | 100 |
| North Aegean | 20.0 | 33.3 | 55.6 | 75.6 | 91.1 | 93.3 | 100 | 100 |
| Attica | 4.1 | 21.3 | 53.8 | 76.4 | 90.2 | 95.2 | 99.2 | 100 |
| Peloponnese | 6.0 | 16.9 | 53.0 | 71.1 | 83.1 | 86.8 | 94.0 | 100 |
| Crete | 3.1 | 20.9 | 38.3 | 60.2 | 79.6 | 86.2 | 95.4 | 100 |
| Thessaly | 2.3 | 14.1 | 41.4 | 60.2 | 81.3 | 91.4 | 93.8 | 100 |
| E Macedonia & Thrace | 2.5 | 17.8 | 41.5 | 58.5 | 74.6 | 83.1 | 94.1 | 100 |
| Central Greece | 3.1 | 8.3 | 19.8 | 43.8 | 76.0 | 90.6 | 97.9 | 100 |
| South Aegean | 0 | 3.5 | 25.9 | 53.5 | 67.2 | 84.5 | 94.8 | 100 |
| Ionian Islands | 3.2 | 6.5 | 24.0 | 42.9 | 59.0 | 74.5 | 91.2 | 100 |
| Central Macedonia | 1.0 | 6.6 | 24.0 | 42.9 | 59.0 | 74.5 | 91.2 | 100 |
| Western Greece | 5.5 | 9.1 | 20.0 | 30.9 | 61.8 | 83.6 | 90.9 | 100 |
| Total Greece | 3.8 | 17.3 | 42.7 | 63.6 | 80.1 | 88.8 | 96.1 | 100 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thomaidis, L.; Mavroeidi, N.; Richardson, C.; Choleva, A.; Damianos, G.; Bolias, K.; Tsolia, M. Autism Spectrum Disorders in Greece: Nationwide Prevalence in 10–11 Year-Old Children and Regional Disparities. J. Clin. Med. 2020, 9, 2163. https://doi.org/10.3390/jcm9072163
Thomaidis L, Mavroeidi N, Richardson C, Choleva A, Damianos G, Bolias K, Tsolia M. Autism Spectrum Disorders in Greece: Nationwide Prevalence in 10–11 Year-Old Children and Regional Disparities. Journal of Clinical Medicine. 2020; 9(7):2163. https://doi.org/10.3390/jcm9072163
Chicago/Turabian StyleThomaidis, Loretta, Nikoletta Mavroeidi, Clive Richardson, Antigoni Choleva, George Damianos, Konstantinos Bolias, and Maria Tsolia. 2020. "Autism Spectrum Disorders in Greece: Nationwide Prevalence in 10–11 Year-Old Children and Regional Disparities" Journal of Clinical Medicine 9, no. 7: 2163. https://doi.org/10.3390/jcm9072163
APA StyleThomaidis, L., Mavroeidi, N., Richardson, C., Choleva, A., Damianos, G., Bolias, K., & Tsolia, M. (2020). Autism Spectrum Disorders in Greece: Nationwide Prevalence in 10–11 Year-Old Children and Regional Disparities. Journal of Clinical Medicine, 9(7), 2163. https://doi.org/10.3390/jcm9072163





