Using Machine Learning to Predict ICU Transfer in Hospitalized COVID-19 Patients
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Cohort and Features
2.2. Sampling Strategy
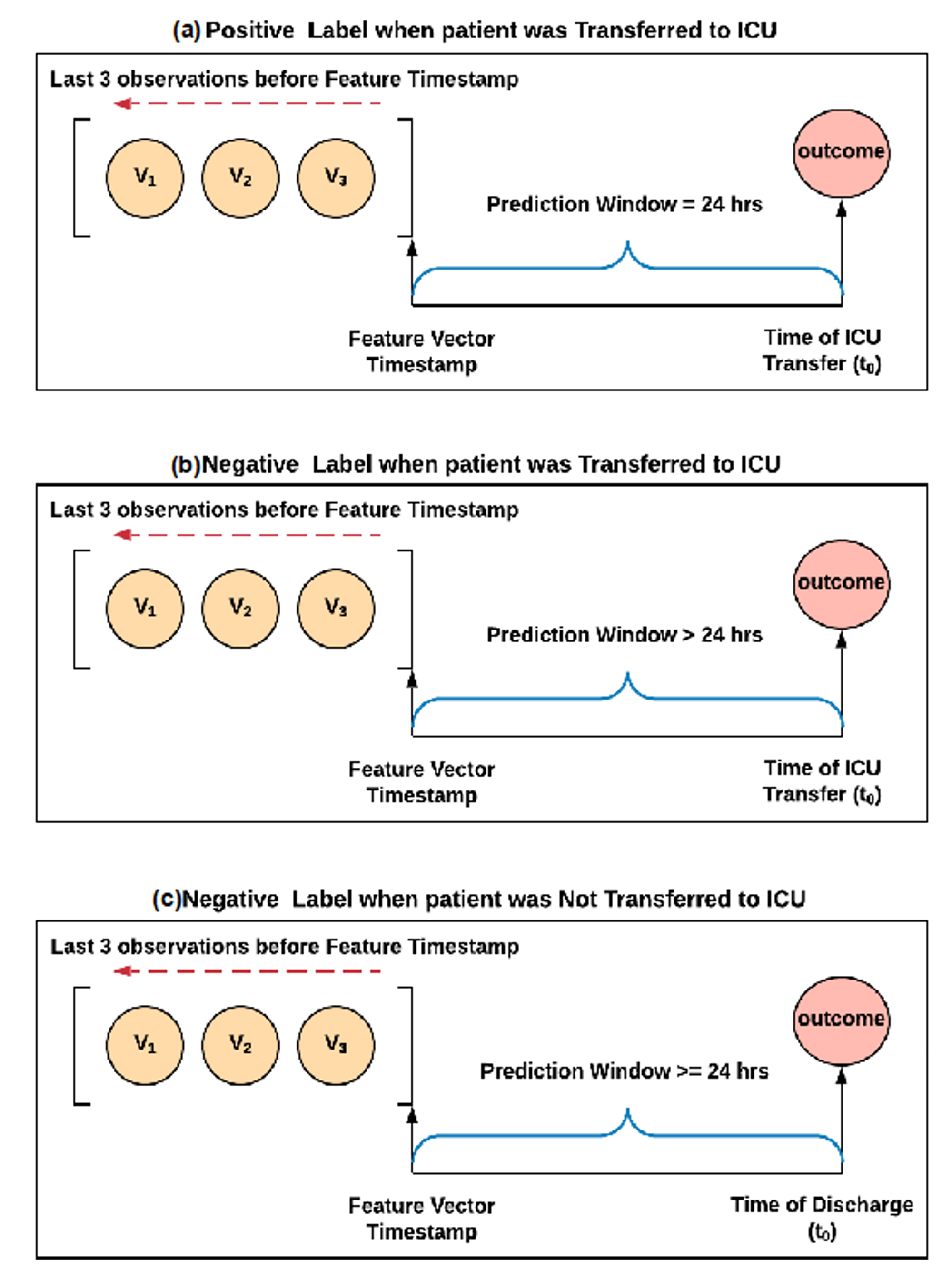
2.3. Labeling
2.4. Training, Testing, and Cross-Validation
2.5. Feature Selection
2.6. Model Testing
3. Results
3.1. Cohort Characteristics
3.2. Features and Model Hyperparameters
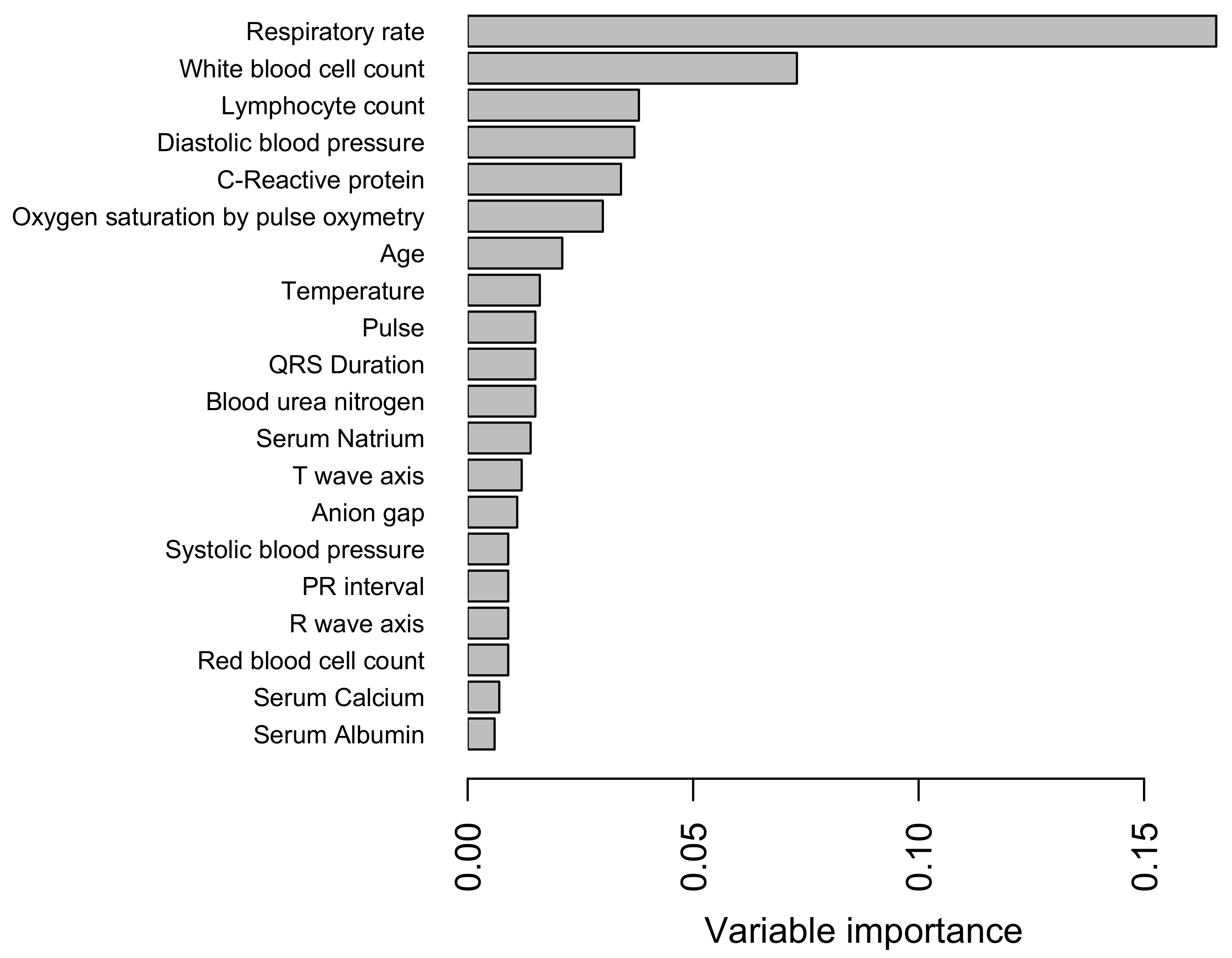
3.3. Predictors and Their Importance
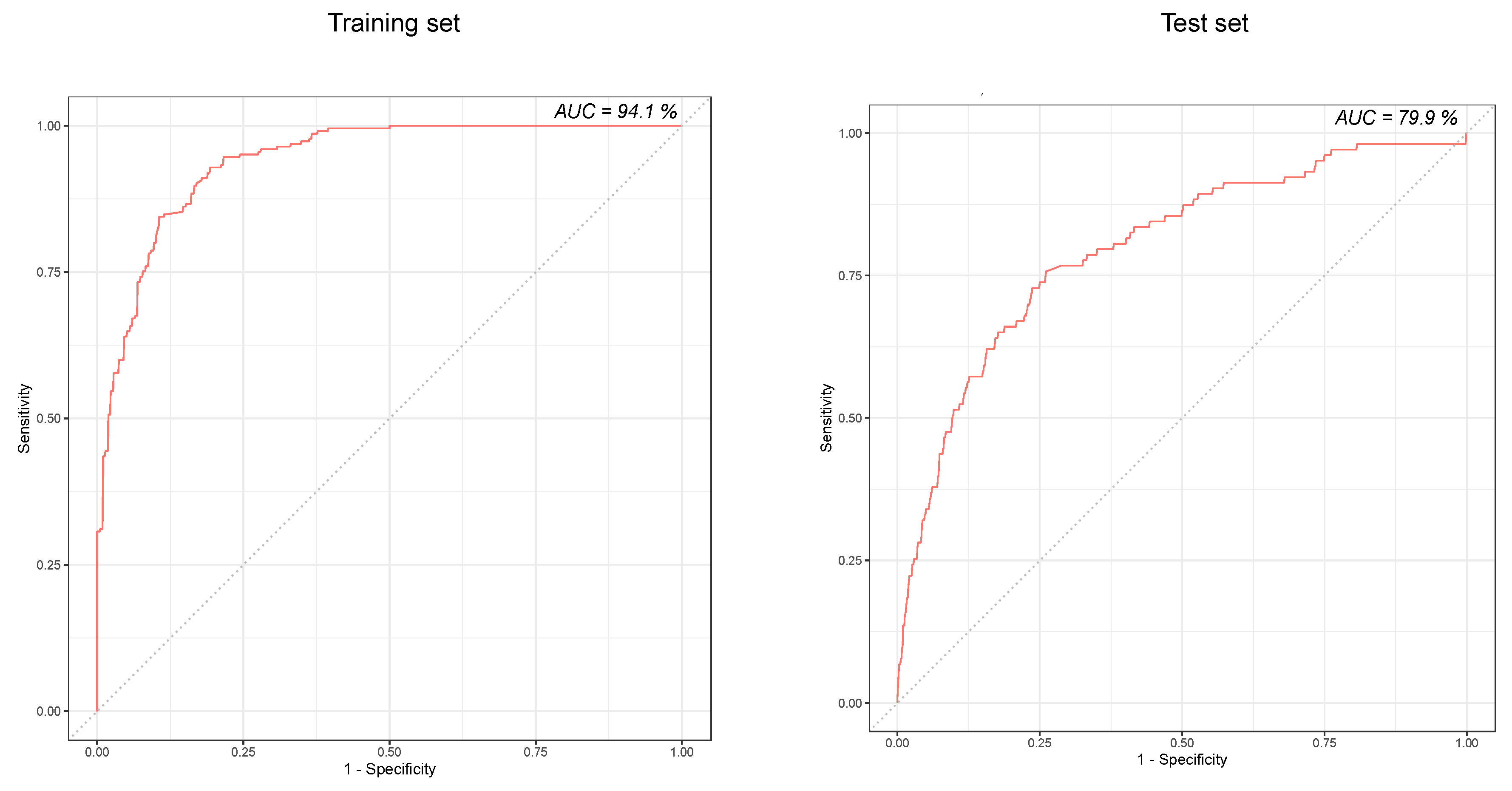
3.4. Predictive Performance of the Model
4. Discussion
4.1. Model Variables of Interest
4.2. Model Strengths
4.3. Model Limitations
4.4. Practice Implications
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization. WHO COVID-19 Dashboard. Available online: https://who.sprinklr.com/ (accessed on 9 April 2020).
- Fauci, A.S.; Lane, H.C.; Redfield, R.R. Covid-19—Navigating the Uncharted. N. Engl. J. Med. 2020, 382, 1268–1269. [Google Scholar] [CrossRef]
- CDC COVID-19 Response Team. Severe Outcomes Among Patients with Coronavirus Disease 2019 (COVID-19)—United States, February 12–March 16, 2020. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 343–346. [CrossRef]
- Myers, L.C.; Parodi, S.M.; Escobar, G.J.; Liu, V.X. Characteristics of Hospitalized Adults With COVID-19 in an Integrated Health Care System in California. JAMA 2020. [Google Scholar] [CrossRef]
- Richardson, S.; Hirsch, J.S.; Narasimhan, M.; Crawford, J.M.; McGinn, T.; Davidson, K.W.; Cookingham, J. Presenting Characteristics, Comorbidities, and Outcomes Among 5700 Patients Hospitalized With COVID-19 in the New York City Area. JAMA 2020. [Google Scholar] [CrossRef] [PubMed]
- Howell, E. Active Bed Management by Hospitalists and Emergency Department Throughput. Ann. Intern. Med. 2008, 149, 804. [Google Scholar] [CrossRef] [PubMed]
- Moriarty, J.P.; Schiebel, N.E.; Johnson, M.G.; Jensen, J.B.; Caples, S.M.; Morlan, B.W.; Huddleston, J.M.; Heubner, M.; Naessens, J.M. Evaluating implementation of a rapid response team: Considering alternative outcome measures. Int. J. Qual. Health Care 2014, 26, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Verity, R.; Okell, L.C.; Dorigatti, I.; Winskill, P.; Whittaker, C.; Imai, N.; Cori, A.; Fu, H.; Baguelin, M.; Dighe, A.; et al. Estimates of the severity of coronavirus disease 2019: A model-based analysis. Lancet Infect. Dis. 2020, 20, 669–677. [Google Scholar] [CrossRef]
- CDC COVID-19 Response Team. Preliminary Estimates of the Prevalence of Selected Underlying Health Conditions Among Patients with Coronavirus Disease 2019—United States, February 12–March 28, 2020. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 382–386. [Google Scholar]
- American Hospital Association. Fast Facts on U.S. Hospitals, 2020|AHA. 2018 AHA Annual Survey. 2020. Available online: https://www.aha.org/statistics/fast-facts-us-hospitals (accessed on 9 April 2020).
- Halpern, N.A.; Goldman, D.A.; Tan, K.S.; Pastores, S.M. Trends in Critical Care Beds and Use Among Population Groups and Medicare and Medicaid Beneficiaries in the United States. Crit. Care Med. 2016, 44, 1490–1499. [Google Scholar] [CrossRef]
- Halpern, N.A.; Tan, K.S. United States Resource Availability for COVID-19. 2020. Available online: https://sccm.org/getattachment/Blog/March-2020/United-States-Resource-Availability-for-COVID-19/United-States-Resource-Availability-for-COVID-19.pdf (accessed on 9 April 2020).
- National Center for Immunization and Respiratory Diseases (NCIRD). Interim Clinical Guidance for Management of Patients with Confirmed Coronavirus Disease (COVID-19). 2020. Available online: https://www.cdc.gov/coronavirus/2019-ncov/hcp/clinical-guidance-management-patients.html (accessed on 9 April 2020).
- Tang, N.; Li, D.; Wang, X.; Sun, Z. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J. Thromb. Haemost. 2020, 18, 844–847. [Google Scholar] [CrossRef]
- Breiman, L. Random Forrests. Mach. Learn. 2001, 45, 5–32. [Google Scholar] [CrossRef]
- Basu, S.; Kumbier, K.; Brown, J.B.; Yu, B. Iterative random forests to discover predictive and stable high-order interactions. Proc. Natl. Acad. Sci. USA 2018, 115, 1943–1948. [Google Scholar] [CrossRef] [PubMed]
- Batista, G.E.A.P.A.; Monard, M.C. An analysis of four missing data treatment methods for supervised learning. Appl. Artif. Intell. 2003, 17, 519–533. [Google Scholar] [CrossRef]
- Japkowicz, N. The Class Imbalance Problem: Significance and Strategies. In Proceedings of the 2000 International Conference on Artificial Intelligence, Las Vegas, NV, USA, 26 June 2000. [Google Scholar]
- Kia, A.; Timsina, P.; Joshi, H.N.; Klang, E.; Gupta, R.R.; Freeman, R.M.; Reich, D.L.; Tomlinson, M.S.; Dudley, J.T.; Mazumdar, M.; et al. MEWS++: Enhancing the Prediction of Clinical Deterioration in Admitted Patients through a Machine Learning Model. J. Clin. Med. 2020, 9, 343. [Google Scholar] [CrossRef] [PubMed]
- The Apache Software Foundation. MLlib: Main Guide—Spark 2.3.0 Documentation. 2018. Available online: https://spark.apache.org/docs/2.3.0/mllib-ensembles.html (accessed on 1 June 2020).
- Nicastri, E.; Petrosillo, N.; Bartoli, T.A.; Lepore, L.; Mondi, A.; Palmieri, F.; Antinori, A. National Institute for the Infectious Diseases “L. Spallanzani” IRCCS. Recommendations for COVID-19 Clinical Management. Infect. Dis. Rep. 2020, 12, 8543. [Google Scholar] [CrossRef] [PubMed]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Guan, L.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- Guo, T.; Fan, Y.; Chen, M.; Wu, X.; Zhang, L.; He, T.; Wang, H.; Wan, J.; Wang, X.; Lu, Z. Cardiovascular Implications of Fatal Outcomes of Patients With Coronavirus Disease 2019 (COVID-19). JAMA Cardiol. 2020. [Google Scholar] [CrossRef]
- Maxim, L.D.; Niebo, R.; Utell, M.J. Screening tests: A review with examples. Inhal. Toxicol. 2014, 26, 811–828. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Core Team: Vienna, Austria, 2019; Available online: https://www.R-project.org/ (accessed on 1 June 2020).
- Grau, J.; Grosse, I.; Keilwagen, J. PRROC: Computing and visualizing precision-recall and receiver operating characteristic curves in R. Bioinformatics 2015, 31, 2595–2597. [Google Scholar] [CrossRef]
- Robin, X.; Turck, N.; Hainard, A.; Tiberti, N.; Lisacek, F.; Sanchez, J.C.; Müller, M. pROC: An open-source package for R and S+ to analyze and compare ROC curves. BMC Bioinform. 2011, 12, 77. [Google Scholar] [CrossRef]
- Stevenson, M.; Nunes, T.; Sanchez, J.; Thornton, R.; Reiczigel, J.; Robison-Cox, J.; Sebastiani, P. EpiR: An R Package for the Analysis of Epidemiological Data. 2013. Available online: http://www2.uaem.mx/r-mirror/web/packages/epiR/epiR.pdf (accessed on 1 June 2020).
- Cascella, M.; Rajnik, M.; Cuomo, A.; Dulebohn, S.C.; Di Napoli, R. Features, Evaluation and Treatment Coronavirus (COVID-19); StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Qin, C.; Zhou, L.; Hu, Z.; Zhang, S.; Yang, S.; Tao, Y.; Xie, C.; Ma, K.; Shang, K.; Tian, D.S.; et al. Dysregulation of Immune Response in Patients With Coronavirus 2019 (COVID-19) in Wuhan, China. Clin. Infect. Dis. 2020. [Google Scholar] [CrossRef] [PubMed]
- Bhatraju, P.K.; Ghassemieh, B.J.; Nichols, M.; Kim, R.; Jerome, K.R.; Nalla, A.K.; Greninger, A.L.; Pipavath, S.; Wurfel, M.M.; Kritek, P.A.; et al. Covid-19 in Critically Ill Patients in the Seattle Region—Case Series. N. Engl. J. Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- Matthay, M.A.; Aldrich, J.M.; Gotts, J.E. Treatment for severe acute respiratory distress syndrome from COVID-19. Lancet Respir. Med. 2020. [Google Scholar] [CrossRef]
- Wang, L. C-reactive protein levels in the early stage of COVID-19. Méd. Mal. Infect. 2020. [Google Scholar] [CrossRef]
- Kyriacos, U.; Jelsma, J.; Jordan, S. Monitoring vital signs using early warning scoring systems: A review of the literature. J. Nurs. Manag. 2011, 19, 311–330. [Google Scholar] [CrossRef]
- Aird, W.C. The Hematologic System as a Marker of Organ Dysfunction in Sepsis. Mayo Clin. Proc. 2003, 78, 869–881. [Google Scholar] [CrossRef]
- Lippi, G.; South, A.M.; Henry, B.M. Electrolyte Imbalances in Patients with Severe Coronavirus Disease 2019 (COVID-19). Ann. Clin. Biochem. 2020, 57, 262–265. [Google Scholar] [CrossRef]
- Goyal, P.; Choi, J.J.; Pinheiro, L.C.; Schenck, E.J.; Chen, R.; Jabri, A.; Satlin, M.J.; Nahid, M.; Nahid, J.B.; Hoffman, K.L.; et al. Clinical Characteristics of Covid-19 in New York City. N. Engl. J. Med. 2020. [Google Scholar] [CrossRef]
- Lippi, G.; Plebani, M. Laboratory abnormalities in patients with COVID-2019 infection. Clin. Chem. Lab. Med. 2020. [Google Scholar] [CrossRef]
- Sun, Q.; Qiu, H.; Huang, M.; Yang, Y. Lower mortality of COVID-19 by early recognition and intervention: Experience from Jiangsu Province. Ann. Intensive Care 2020, 10, 33. [Google Scholar] [CrossRef]
- Churpek, M.M.; Wendlandt, B.; Zadravecz, F.J.; Adhikari, R.; Winslow, C.; Edelson, D.P. Association between intensive care unit transfer delay and hospital mortality: A multicenter investigation. J. Hosp. Med. 2016. [Google Scholar] [CrossRef] [PubMed]
- Hu, W.; Chan, C.W.; Zubizarreta, J.R.; Escobar, G.J. An examination of early transfers to the ICU based on a physiologic risk score. Manuf. Serv. Oper. Manag. 2018. [Google Scholar] [CrossRef]
- Medrzycka-Dabrowska, W.; Lewandowska, K.; Kwiecień-Jagus, K.; Czyz-Szypenbajl, K. Sleep deprivation in Intensive Care Unit-systematic review. Open Med. 2018. [Google Scholar] [CrossRef] [PubMed]
- Kotfis, K.; Williams Roberson, S.; Wilson, J.E.; Dabrowski, W.; Pun, B.T.; Ely, E.W. COVID-19: ICU delirium management during SARS-CoV-2 pandemic. Crit. Care 2020. [Google Scholar] [CrossRef]
| Overall (n = 1987) | Test (n = 612) | |
|---|---|---|
| Age Groups | ||
| 18–45 | 643 (32.4%) | 202 (33.0%) |
| 45–65 | 638 (32.1%) | 190 (31.0%) |
| 65–80 | 491 (24.7%) | 154 (25.2%) |
| >80 | 215 (10.8%) | 66 (10.8%) |
| Gender | ||
| Male | 904 (45.5%) | 283 (46.2%) |
| Female | 1083 (54.5%) | 329 (53.8%) |
| Length of Stay | ||
| 0–3 days | 692 (34.8%) | 216 (35.3%) |
| 3–10 days | 830 (41.8%) | 256 (41.8%) |
| >10 days | 332 (16.7%) | 94 (15.4%) |
| Not discharged | 133 (6.7%) | 46 (7.5%) |
| ICU care received | ||
| Yes | 330 (16.6%) | 103 (16.8%) |
| No | 1657 (83.4%) | 509 (83.2%) |
| Geographic origin | ||
| Bronx | 226 (11.4%) | 68 (11.1%) |
| Brooklyn | 330 (16.6%) | 111 (18.1%) |
| Manhattan | 833 (41.9%) | 256 (41.8%) |
| Queens | 339 (17.1%) | 103 (16.8%) |
| Staten Island | 27 (1.4%) | 8 (1.3%) |
| Out of New York City | 174 (8.8%) | 48 (7.8%) |
| Out of NY State | 57 (2.9%) | 17 (2.8%) |
| Missing | 1 (0.1%) | 1 (0.2%) |
| Race and Ethnicity | ||
| Non-Hispanic Black | 331 (16.7%) | 103 (16.8%) |
| Non-Hispanic White | 546 (27.5%) | 168 (27.5%) |
| Hispanic Black | 45 (2.3%) | 12 (2.0%) |
| Hispanic White | 75 (3.8%) | 23 (3.8%) |
| Asian | 115 (5.8%) | 35 (5.7%) |
| Others | 739 (37.2%) | 227 (37.1%) |
| Unspecified | 136 (6.8%) | 44 (7.2%) |
| Smoking history | ||
| Current Smoker | 105 (5.3%) | 34 (5.6%) |
| Past smoker | 431 (21.7%) | 135 (22.1%) |
| Never smoked | 1120 (56.4%) | 339 (55.4%) |
| Unknown | 324 (16.3%) | 104 (17.0%) |
| Missing | 7 (0.4%) | 0 (0%) |
| Hypertension | ||
| Yes | 566 (28.5%) | 162 (26.5%) |
| No | 1414 (71.2%) | 450 (73.5%) |
| Missing | 7 (0.4%) | 0 (0%) |
| Diabetes | ||
| Yes | 431 (21.7%) | 140 (22.9%) |
| No | 1549 (78.0%) | 472 (77.1%) |
| Missing | 7 (0.4%) | 0 (0%) |
| COPD and Asthma | ||
| Yes | 170 (8.6%) | 49 (8.0%) |
| No | 1810 (91.1%) | 563 (92.0%) |
| Missing | 7 (0.4%) | 0 (0%) |
| Obesity | ||
| Yes | 176 (8.9%) | 53 (8.7%) |
| No | 1804 (90.8%) | 559 (91.3%) |
| Missing | 7 (0.4%) | 0 (0%) |
| Model | Total Feature Vectors in Test Cohort | % Prevalence of Positive Label * | Sensitivity (%) | Specificity (%) | Accuracy (%) | Precision (%) | Negative Predictive Value (%) | AUC-ROC (%) |
|---|---|---|---|---|---|---|---|---|
| Random Forest classifier | 2812 | 3.7 | 72.8 (63.2–81.1) | 76.3 (74.7–77.9) | 76.2 (74.6–77.7) | 10.5 (8.3–12.9) | 98.7 (98.1–99.1) | 79.9 (75.2–84.6) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cheng, F.-Y.; Joshi, H.; Tandon, P.; Freeman, R.; Reich, D.L.; Mazumdar, M.; Kohli-Seth, R.; Levin, M.A.; Timsina, P.; Kia, A. Using Machine Learning to Predict ICU Transfer in Hospitalized COVID-19 Patients. J. Clin. Med. 2020, 9, 1668. https://doi.org/10.3390/jcm9061668
Cheng F-Y, Joshi H, Tandon P, Freeman R, Reich DL, Mazumdar M, Kohli-Seth R, Levin MA, Timsina P, Kia A. Using Machine Learning to Predict ICU Transfer in Hospitalized COVID-19 Patients. Journal of Clinical Medicine. 2020; 9(6):1668. https://doi.org/10.3390/jcm9061668
Chicago/Turabian StyleCheng, Fu-Yuan, Himanshu Joshi, Pranai Tandon, Robert Freeman, David L Reich, Madhu Mazumdar, Roopa Kohli-Seth, Matthew A. Levin, Prem Timsina, and Arash Kia. 2020. "Using Machine Learning to Predict ICU Transfer in Hospitalized COVID-19 Patients" Journal of Clinical Medicine 9, no. 6: 1668. https://doi.org/10.3390/jcm9061668
APA StyleCheng, F.-Y., Joshi, H., Tandon, P., Freeman, R., Reich, D. L., Mazumdar, M., Kohli-Seth, R., Levin, M. A., Timsina, P., & Kia, A. (2020). Using Machine Learning to Predict ICU Transfer in Hospitalized COVID-19 Patients. Journal of Clinical Medicine, 9(6), 1668. https://doi.org/10.3390/jcm9061668





