Dysregulations of MicroRNA and Gene Expression in Chronic Venous Disease
Abstract
1. Introduction
2. Experimental Section
2.1. Study Participants Characteristics
2.2. Study Material Preparation
2.3. Libraries Preparation and Sequencing
2.4. Statistical Analysis
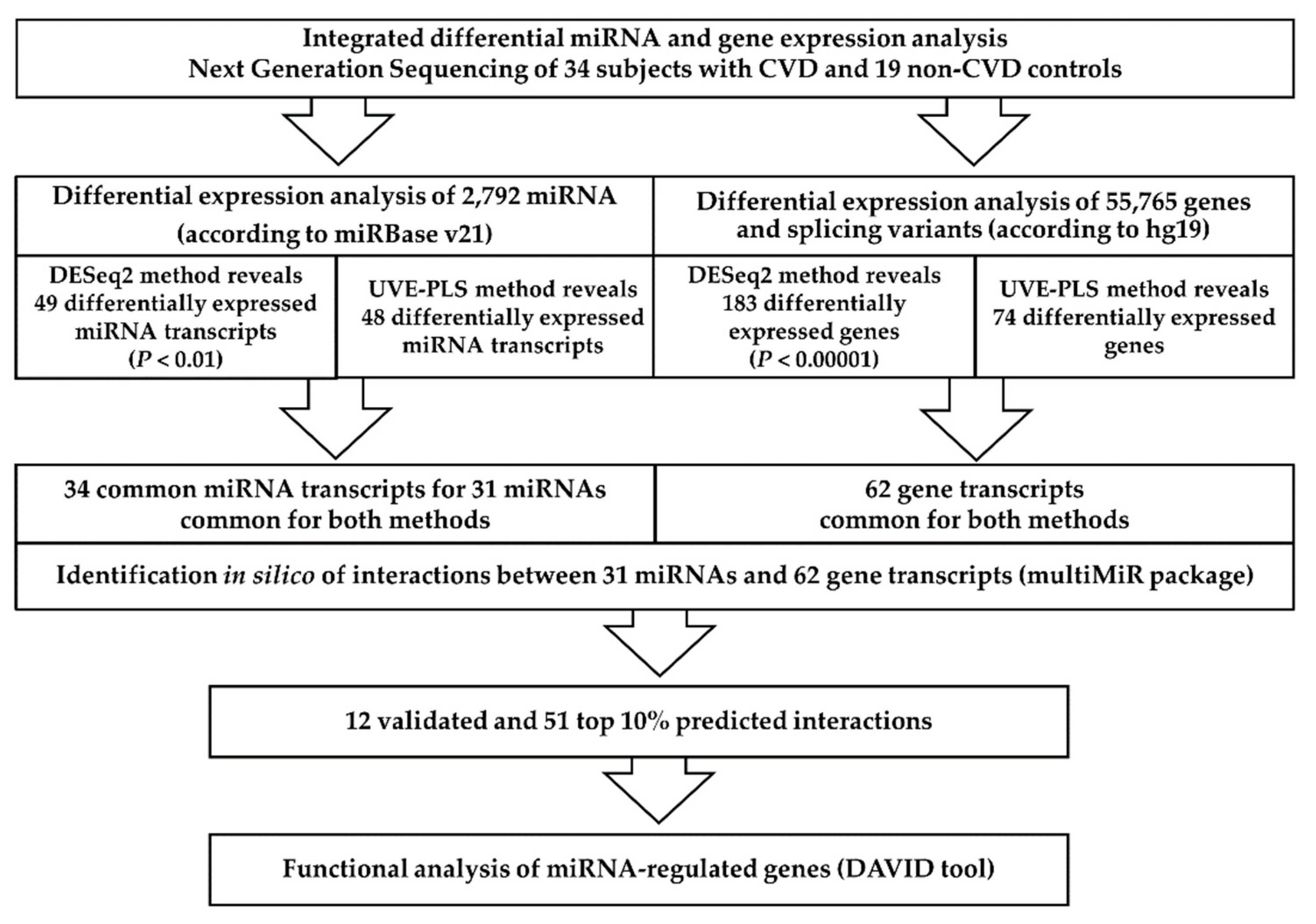
3. Results
3.1. Study Population Analysis
3.2. Primary Results
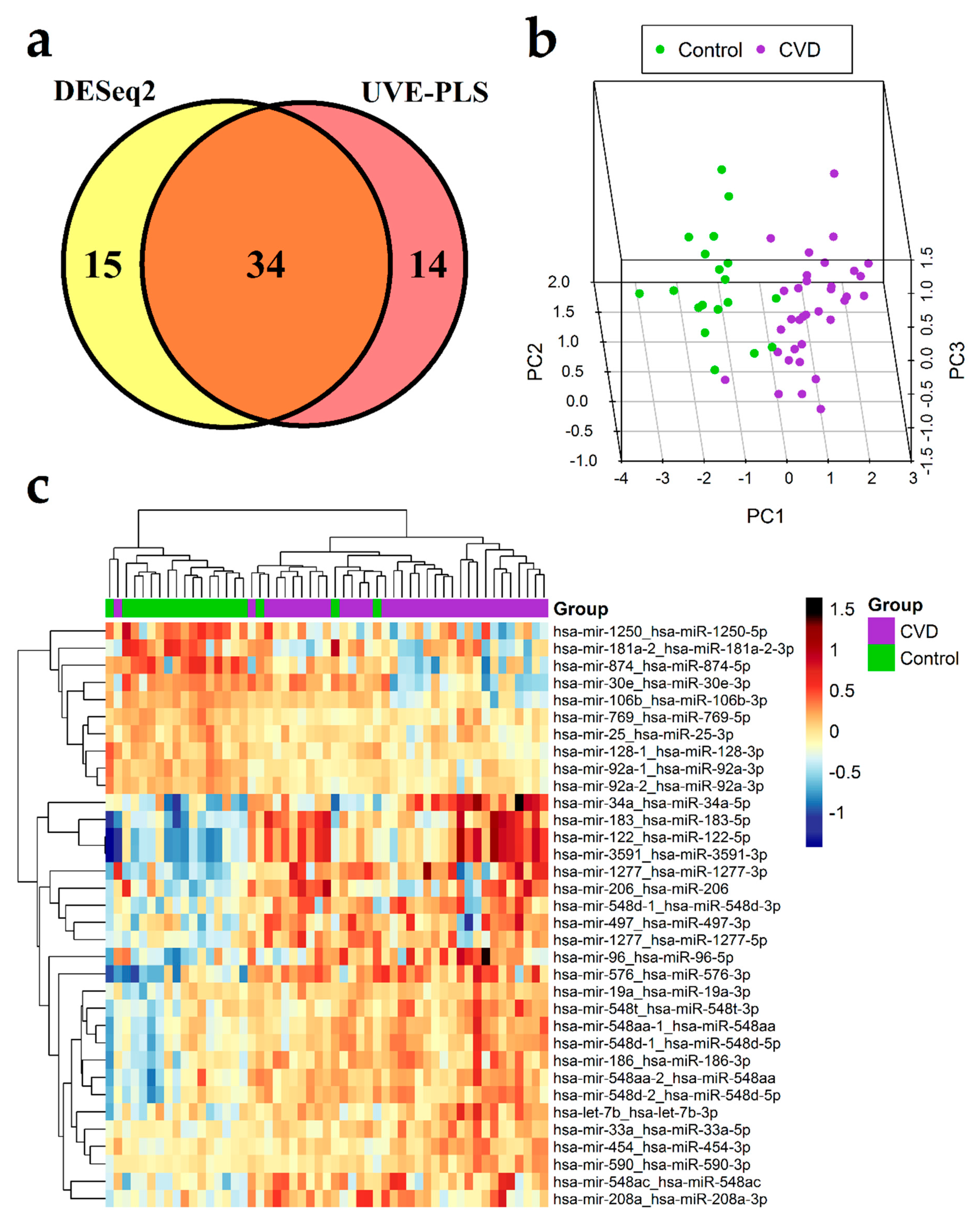
3.3. Differential Expression Analysis of miRNA
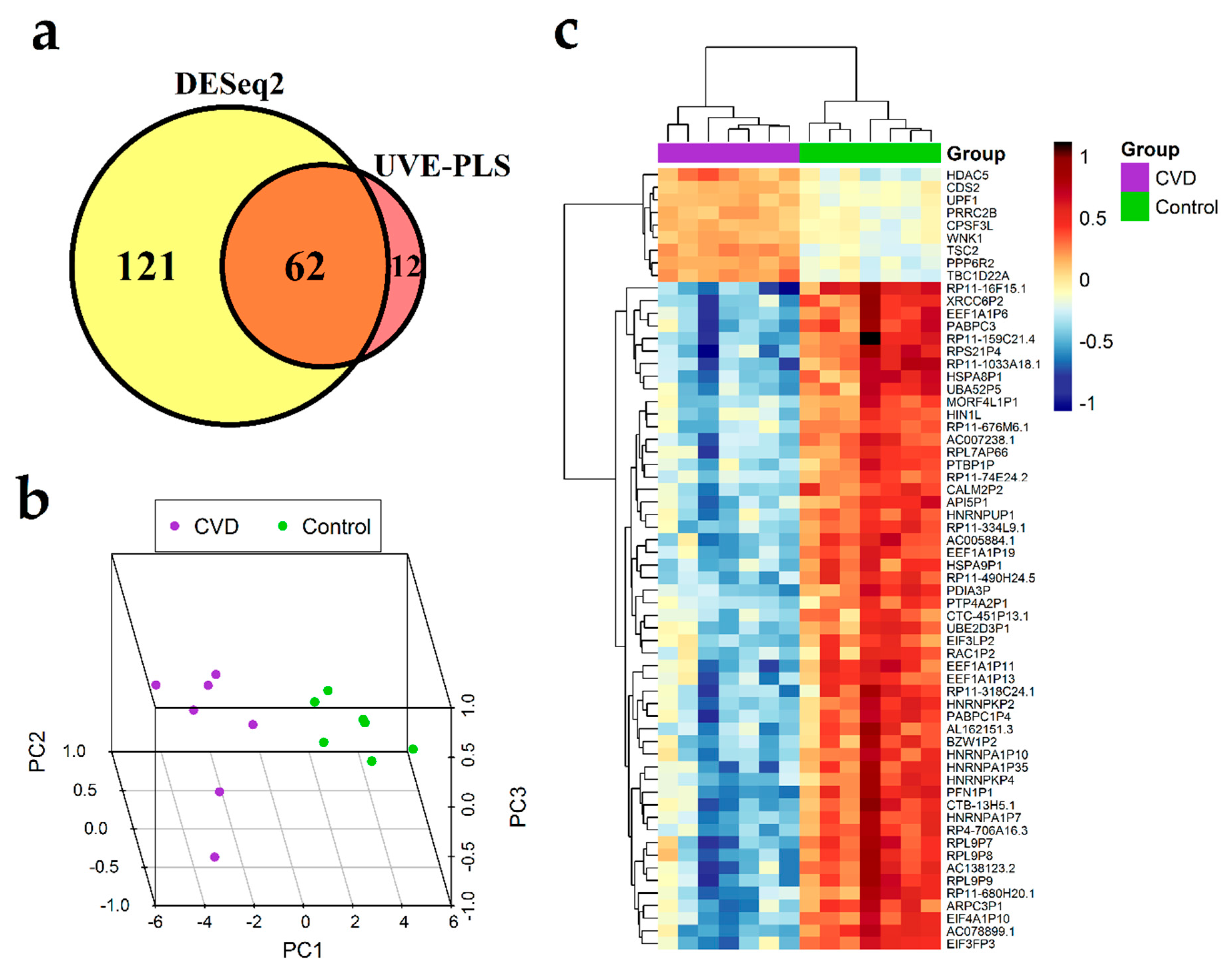
3.4. Differential Expression Analysis of Genes
3.5. In Silico Identification of miRNA:Gene Interactions
3.6. Functional Analysis of miRNA Targets
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Data Availability
References
- Rabe, E.; Guex, J.J.; Puskas, A.; Scuderi, A.; Fernandez Quesada, F. VCP Coordinators. Epidemiology of chronic venous disorders in geographically diverse populations: Results from the Vein Consult Program. Int. Angiol. 2012, 31, 105–115. [Google Scholar] [PubMed]
- Vuylsteke, M.E.; Colman, R.; Thomis, S.; Guillaume, G.; Van Quickenborne, D.; Staelens, I. An epidemiological survey of venous disease among general practitioner attendees in different geographical regions on the globe: The final results of the Vein Consult Program. Angiology 2018, 69, 779–785. [Google Scholar] [CrossRef] [PubMed]
- Eklof, B.; Perrin, M.; Delis, K.; Rutherford, R. Updated terminology of chronic venous disorders: The vein term transatlantic interdisciplinary consensus document. J. Vasc. Surg. 2009, 49, 498–501. [Google Scholar] [CrossRef] [PubMed]
- Rabe, E.; Pannier, F. Clinical, aetiological, anatomical and pathological classification (CEAP): Gold standard and limits. Phlebology 2012, 27 (Suppl. 1), 114–118. [Google Scholar] [CrossRef]
- Eberhardt, R.T.; Raffetto, J.D. Chronic venous insufficiency. Circulation 2014, 130, 333–346. [Google Scholar] [CrossRef]
- Lurie, F.; Passman, M.; Meisner, M.; Dalsing, M.; Masuda, E.; Welch, H.; Bush, R.L.; Blebea, J.; Carpentier, P.H.; De Maeseneer, M.; et al. CEAP classification system and reporting standard, revision 2020. J. Vasc. Surg. Venous Lymphat. Disord. 2020, 8, 342–352. [Google Scholar] [CrossRef]
- Bergan, J.J.; Schmid-Schönbein, G.W.; Smith, P.D.; Nicolaides, A.N.; Boisseau, M.R.; Eklof, B. Chronic venous disease. N. Engl. J. Med. 2006, 355, 488–498. [Google Scholar] [CrossRef]
- Meissner, M.H.; Moneta, G.; Burnand, K.; Gloviczki, P.; Lohr, J.M.; Lurie, F.; Mattos, M.A.; McLafferty, R.B.; Mozes, G.; Rutherford, R.B.; et al. The hemodynamics and diagnosis of venous disease. J. Vasc. Surg. 2007, 46 (Suppl. S), 4S–24S. [Google Scholar] [CrossRef]
- Ligi, D.; Croce, L.; Mannello, F. Chronic venous disorders: The dangerous, the good, and the diverse. Int. J. Mol. Sci. 2018, 19, 2544. [Google Scholar] [CrossRef]
- Bergan, J. Molecular mechanisms in chronic venous insufficiency. Ann. Vasc. Surg. 2007, 21, 260–266. [Google Scholar] [CrossRef]
- Pocock, E.S.; Alsaigh, T.; Mazor, R.; Schmid-Schönbein, G.W. Cellular and molecular basis of venous insufficiency. Vasc. Cell 2014, 6, 24. [Google Scholar] [CrossRef] [PubMed]
- Mansilha, A.; Sousa, J. Pathophysiological mechanisms of chronic venous disease and implications for venoactive drug therapy. Int. J. Mol. Sci. 2018, 19, 1669. [Google Scholar] [CrossRef] [PubMed]
- Serralheiro, P.; Novais, A.; Cairrão, E.; Maia, C.; Costa Almeida, C.M.; Verde, I. Variability of MMP/TIMP and TGF-β1 receptors throughout the clinical progression of chronic venous disease. Int. J. Mol. Sci. 2018, 19, 6. [Google Scholar] [CrossRef] [PubMed]
- Tracz, E.; Zamojska, E.; Modrzejewski, A.; Zaborski, D.; Grzesiak, W. Quality of life in patients with venous stasis ulcers and others with advanced venous insufficiency. Holist Nurs. Pract. 2015, 29, 96–102. [Google Scholar] [CrossRef]
- Soydan, E.; Yilmaz, E.; Baydur, H. Effect of socio-demographic characteristics and clinical findings on the quality of life of patients with chronic venous insufficiency. Vascular 2017, 25, 382–389. [Google Scholar] [CrossRef]
- Wittens, C.; Davies, A.H.; Bækgaard, N.; Broholm, R.; Cavezzi, A.; Chastanet, S.; de Wolf, M.; Eggen, C.; Giannoukas, A.; Gohel, M.; et al. Editor’s Choice—Management of chronic venous disease: Clinical practice guidelines of the European Society for Vascular Surgery (ESVS). Eur. J. Vasc. Endovasc. Surg. 2015, 49, 678–737. [Google Scholar] [CrossRef]
- Attaran, R.R. Latest innovations in the treatment of venous disease. J. Clin. Med. 2018, 7, 77. [Google Scholar] [CrossRef]
- Ghildiyal, M.; Zamore, P.D. Small silencing RNAs: An expanding universe. Nat. Rev. Genet. 2009, 10, 94–108. [Google Scholar] [CrossRef]
- Kim, D.; Sung, Y.M.; Park, J.; Kim, S.; Kim, J.; Park, J.; Ha, H.; Bae, J.Y.; Kim, S.; Baek, D. General rules for functional microRNA targeting. Nat. Genet. 2016, 48, 1517–1526. [Google Scholar] [CrossRef]
- Guo, H.; Ingolia, N.T.; Weissman, J.S.; Bartel, D.P. Mammalian microRNAs predominantly act to decrease target mRNA levels. Nature 2010, 466, 835–840. [Google Scholar] [CrossRef]
- Mukherji, S.; Ebert, M.S.; Zheng, G.X.; Tsang, J.S.; Sharp, P.A.; van Oudenaarden, A. MicroRNAs can generate thresholds in target gene expression. Nat. Genet. 2011, 43, 854–859. [Google Scholar] [CrossRef] [PubMed]
- Ohtsuka, M.; Ling, H.; Doki, Y.; Mori, M.; Calin, G.A. MicroRNA Processing and Human Cancer. J. Clin. Med. 2015, 4, 1651–1667. [Google Scholar] [CrossRef] [PubMed]
- Rupaimoole, R.; Slack, F.J. MicroRNA therapeutics: Towards a new era for the management of cancer and other diseases. Nat. Rev. Drug Discov. 2017, 16, 203–222. [Google Scholar] [CrossRef] [PubMed]
- Inamura, K.; Ishikawa, Y. MicroRNA in lung cancer: novel biomarkers and potential tools for treatment. J. Clin. Med. 2016, 5, 36. [Google Scholar] [CrossRef] [PubMed]
- Qin, S.; Zhang, C. MicroRNAs in vascular disease. J. Cardiovasc. Pharmacol. 2011, 57, 8–12. [Google Scholar] [CrossRef]
- Fernández-Hernando, C.; Suárez, Y. MicroRNAs in endothelial cell homeostasis and vascular disease. Curr. Opin. Hematol. 2018, 25, 227–236. [Google Scholar] [CrossRef]
- Schulte, C.; Karakas, M.; Zeller, T. MicroRNAs in cardiovascular disease—Clinical application. Clin. Chem. Lab. Med. 2017, 55, 687–704. [Google Scholar] [CrossRef]
- Feinberg, M.W.; Moore, K.J. MicroRNA regulation of atherosclerosis. Circ. Res. 2016, 118, 703–720. [Google Scholar] [CrossRef]
- Zhou, S.S.; Jin, J.P.; Wang, J.Q.; Zhang, Z.G.; Freedman, J.H.; Zheng, Y.; Cai, L. miRNAS in cardiovascular diseases: Potential biomarkers, therapeutic targets and challenges. Acta Pharmacol. Sin. 2018, 39, 1073–1084. [Google Scholar] [CrossRef]
- Bogucka-Kocka, A.; Zalewski, D.P.; Ruszel, K.P.; Stępniewski, A.; Gałkowski, D.; Bogucki, J.; Komsta, Ł.; Kołodziej, P.; Zubilewicz, T.; Feldo, M.; et al. Dysregulation of microRNA regulatory network in lower extremities arterial disease. Front. Genet. 2019, 10, 1200. [Google Scholar] [CrossRef]
- Morelli, V.M.; Brækkan, S.K.; Hansen, J.-B. Role of microRNAs in venous thromboembolism. Int. J. Mol. Sci. 2020, 21, 2602. [Google Scholar] [CrossRef] [PubMed]
- Markovic, J.N.; Shortell, C.K. Genomics of varicose veins and chronic venous insufficiency. Semin. Vasc. Surg. 2013, 26, 2–13. [Google Scholar] [CrossRef] [PubMed]
- Cui, C.; Liu, G.; Huang, Y.; Lu, X.; Lu, M.; Huang, X.; Li, W.; Jiang, M. MicroRNA profiling in great saphenous vein tissues of patients with chronic venous insufficiency. Tohoku J. Exp. Med. 2012, 228, 341–350. [Google Scholar] [CrossRef] [PubMed]
- Pastar, I.; Khan, A.A.; Stojadinovic, O.; Lebrun, E.A.; Medina, M.C.; Brem, H.; Kirsner, R.S.; Jimenez, J.J.; Leslie, C.; Tomic-Canic, M. Induction of specific microRNAs inhibits cutaneous wound healing. J. Biol. Chem. 2012, 287, 29324–29335. [Google Scholar] [CrossRef]
- Jin, Y.; Xu, G.; Huang, J.; Zhou, D.; Huang, X.; Shen, L. Analysis of the association between an insertion/deletion polymorphism within the 3′ untranslated region of COL1A2 and chronic venous insufficiency. Ann. Vasc. Surg. 2013, 27, 959–963. [Google Scholar] [CrossRef]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef]
- Centner, V.; Massart, D.L.; de Noord, O.E.; de Jong, S.; Vandeginste, B.M.; Sterna, C. Elimination of uninformative variables for multivariate calibration. Anal. Chem. 1996, 68, 3851–3858. [Google Scholar] [CrossRef]
- Mehmood, T.; Hovde, K.H.; Liland Snipen, L.; Sæbø, S. A review of variable selection methods in Partial Least Squares Regression. Chemometr. Intell. Lab. Syst. 2012, 118, 62–69. [Google Scholar] [CrossRef]
- Chen, H.; Boutros, P.C. VennDiagram: A package for the generation of highly-customizable Venn and Euler diagrams in R. BMC Bioinform. 2011, 12, 35. [Google Scholar] [CrossRef]
- Ligges, U.; Mächler, M. Scatterplot3d—An R package for visualizing multivariate data. J. Stat. Softw. 2003, 8, 1–20. [Google Scholar] [CrossRef]
- Robin, X.; Turck, N.; Hainard, A.; Tiberti, N.; Lisacek, F.; Sanchez, J.C.; Müller, M. pROC: An open-source package for R and S+ to analyze and compare ROC curves. BMC Bioinform. 2011, 12, 77. [Google Scholar] [CrossRef] [PubMed]
- Finotello, F.; Mayer, C.; Plattner, C.; Laschober, G.; Rieder, D.; Hackl, H.; Krogsdam, A.; Loncova, Z.; Posch, W.; Wilflingseder, D.; et al. Molecular and pharmacological modulators of the tumor immune contexture revealed by deconvolution of RNA-seq data. Genome Med. 2019, 11, 34. [Google Scholar] [CrossRef] [PubMed]
- Becht, E.; Giraldo, N.A.; Lacroix, L.; Buttard, B.; Elarouci, N.; Petitprez, F.; Selves, J.; Laurent-Puig, P.; Sautès-Fridman, C.; Fridman, W.H.; et al. Estimating the population abundance of tissue-infiltrating immune and stromal cell populations using gene expression. Genome Biol. 2016, 17, 218. [Google Scholar] [CrossRef]
- Sturm, G.; Finotello, F.; Petitprez, F.; Zhang, J.D.; Baumbach, J.; Fridman, W.H.; List, M.; Aneichyk, T. Comprehensive evaluation of transcriptome-based cell-type quantification methods for immuno-oncology. Bioinformatics 2019, 35, i436–i445. [Google Scholar] [CrossRef] [PubMed]
- Ru, Y.; Kechris, K.J.; Tabakoff, B.; Hoffman, P.; Radcliffe, R.A.; Bowler, R.; Mahaffey, S.; Rossi, S.; Calin, G.A.; Bemis, L.; et al. The multiMiR R package and database: Integration of microRNA–target interactions along with their disease and drug associations. Nucleic Acids Res. 2014, 42, e133. [Google Scholar] [CrossRef] [PubMed]
- Shannon, P.; Markiel, A.; Ozier, O.; Baliga, N.S.; Wang, J.T.; Ramage, D.; Amin, N.; Schwikowski, B.; Ideker, T. Cytoscape: A software environment for integrated models of biomolecular interaction networks. Genome Res. 2003, 13, 2498–2504. [Google Scholar] [CrossRef]
- Huang, D.W.; Sherman, B.T.; Lempicki, R.A. Systematic and integrative analysis of large gene lists using DAVID Bioinformatics Resources. Nat. Protoc. 2009, 4, 44–57. [Google Scholar] [CrossRef]
- Huang, D.W.; Sherman, B.T.; Lempicki, R.A. Bioinformatics enrichment tools: Paths toward the comprehensive functional analysis of large gene lists. Nucleic Acids Res. 2009, 37, 1–13. [Google Scholar] [CrossRef]
- Gloviczki, P.; Comerota, A.J.; Dalsing, M.C.; Eklof, B.G.; Gillespie, D.L.; Gloviczki, M.L.; Lohr, J.M.; McLafferty, R.B.; Meissner, M.H.; Murad, M.H.; et al. The care of patients with varicose veins and associated chronic venous diseases: Clinical practice guidelines of the Society for Vascular Surgery and the American Venous Forum. J. Vasc. Surg. 2011, 53 (Suppl. 5), 2S–48S. [Google Scholar] [CrossRef]
- Han, H.; Qu, G.; Han, C.; Wang, Y.; Sun, T.; Li, F.; Wang, J.; Luo, S. MiR-34a, miR-21 and miR-23a as potential biomarkers for coronary artery disease: A pilot microarray study and confirmation in a 32 patient cohort. Exp. Mol. Med. 2015, 47, e138. [Google Scholar] [CrossRef]
- Raitoharju, E.; Lyytikäinen, L.P.; Levula, M.; Oksala, N.; Mennander, A.; Tarkka, M.; Klopp, N.; Illig, T.; Kähönen, M.; Karhune, P.J.; et al. miR-21, miR-210, miR-34a, and miR-146a/b are up-regulated in human atherosclerotic plaques in the Tampere Vascular Study. Atherosclerosis 2011, 219, 211–217. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Walias, F.J.; Vázquez, E.; Pacheco, Y.; Rodríguez-Fernández, J.M.; Pérez-Elías, M.J.; Dronda, F.; Casado, J.L.; Moreno, A.; Hermida, J.M.; Quereda, C.; et al. Risk, diagnostic and predictor factors for classical hodgkin lymphoma in HIV-1-infected individuals: Role of plasma exosome-derived miR-20a and miR-21. J. Clin. Med. 2020, 9, 760. [Google Scholar] [CrossRef]
- Irimie-Aghiorghiesei, A.I.; Pop-Bica, C.; Pintea, S.; Braicu, C.; Cojocneanu, R.; Zimța, A.-A.; Gulei, D.; Slabý, O.; Berindan-Neagoe, I. Prognostic Value of MiR-21: An updated meta-analysis in Head and Neck Squamous Cell Carcinoma (HNSCC). J. Clin. Med. 2019, 8, 2041. [Google Scholar] [CrossRef] [PubMed]
- Shi, J. Considering exosomal miR-21 as a biomarker for cancer. J. Clin. Med. 2016, 5, 42. [Google Scholar] [CrossRef] [PubMed]
- Ulivi, P.; Petracci, E.; Marisi, G.; Baglivo, S.; Chiari, R.; Billi, M.; Canale, M.; Pasini, L.; Racanicchi, S.; Vagheggini, A.; et al. Prognostic role of circulating miRNAs in early-stage non-small cell lung cancer. J. Clin. Med. 2019, 8, 131. [Google Scholar] [CrossRef]
- Saito, Y.; Nakaoka, T.; Saito, H. microRNA-34a as a therapeutic agent against human cancer. J. Clin. Med. 2015, 4, 1951–1959. [Google Scholar] [CrossRef]
- Cario-Toumaniantz, C.; Boularan, C.; Schurgers, L.J.; Heymann, M.F.; Le Cunff, M.; Léger, J.; Loirand, G.; Pacaud, P. Identification of differentially expressed genes in human varicose veins: Involvement of matrix gla protein in extracellular matrix remodeling. J. Vasc. Res. 2007, 44, 444–459. [Google Scholar] [CrossRef]
- Charles, C.A.; Tomic-Canic, M.; Vincek, V.; Nassiri, M.; Stojadinovic, O.; Eaglstein, W.H.; Kirsner, R.S. A gene signature of nonhealing venous ulcers: Potential diagnostic markers. J. Am. Acad Dermatol. 2008, 59, 758–771. [Google Scholar] [CrossRef]
- Chang, M.Y.; Chiang, P.T.; Chung, Y.C.; Ho, S.Y.; Lin, S.D.; Lin, S.R.; Neoh, C.A. Apoptosis and angiogenesis in varicose veins using gene expression profiling. Fooyin J. Health Sci. 2009, 1, 85–91. [Google Scholar] [CrossRef][Green Version]
- Hsieh, C.S.; Tsai, C.T.; Chen, Y.H.; Chang, S.N.; Hwang, J.J.; Chuang, E.Y.; Wu, I.H. Global expression profiling identifies a novel hyaluronan synthases 2 gene in the pathogenesis of lower extremity varicose veins. J. Clin. Med. 2018, 7, 537. [Google Scholar] [CrossRef]
- Dbouk, H.A.; Weil, L.M.; Perera, G.K.; Dellinger, M.T.; Pearson, G.; Brekken, R.A.; Cobb, M.H. Actions of the protein kinase WNK1 on endothelial cells are differentially mediated by its substrate kinases OSR1 and SPAK. Proc. Natl. Acad. Sci. USA 2014, 111, 15999–16004. [Google Scholar] [CrossRef]
- Sun, X.; Sit, A.; Feinberg, M.W. Role of miR-181 family in regulating vascular inflammation and immunity. Trends Cardiovasc. Med. 2014, 24, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Xie, W.; Li, M.; Xu, N.; Lv, Q.; Huang, N.; He, J.; Zhang, Y. MiR-181a regulates inflammation responses in monocytes and macrophages. PLoS ONE 2013, 8, e58639. [Google Scholar] [CrossRef] [PubMed]
- Du, X.J.; Lu, J.M.; Sha, Y. MiR-181a inhibits vascular inflammation induced by ox-LDL via targeting TLR4 in human macrophages. J. Cell Physiol. 2018, 233, 6996–7003. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Yao, K.; Wang, Q.; Guo, J.; Shi, H.; Ma, L.; Liu, H.; Gao, W.; Zou, Y.; Ge, J. Circulating miR-181a as a potential novel biomarker for diagnosis of acute myocardial infarction. Cell Physiol. Biochem. 2016, 40, 1591–1602. [Google Scholar] [CrossRef]
- Yentrapalli, R.; Azimzadeh, O.; Kraemer, A.; Malinowsky, K.; Sarioglu, H.; Becker, K.F.; Atkinson, M.J.; Moertl, S.; Tapio, S. Quantitative and integrated proteome and microRNA analysis of endothelial replicative senescence. J. Proteomics. 2015, 126, 12–23. [Google Scholar] [CrossRef]
- Ayaz, L.; Dinç, E. Evaluation of microRNA responses in ARPE-19 cells against the oxidative stress. Cutan. Ocul. Toxicol. 2018, 37, 121–126. [Google Scholar] [CrossRef]
- Zhou, J.; He, Z.; Guo, L.; Zeng, J.; Liang, P.; Ren, L.; Zhang, M.; Zhang, P.; Huang, X. MiR-128-3p directly targets VEGFC/VEGFR3 to modulate the proliferation of lymphatic endothelial cells through Ca(2+) signaling. Int. J. Biochem. Cell Biol. 2018, 102, 51–58. [Google Scholar] [CrossRef]
- Xia, Z.; Meng, F.; Liu, Y.; Fang, Y.; Wu, X.; Zhang, C.; Liu, D.; Li, G. Decreased MiR-128-3p alleviates the progression of rheumatoid arthritis by up-regulating the expression of TNFAIP3. Biosci. Rep. 2018, 38, BSR20180540. [Google Scholar] [CrossRef]
- Liu, S.; Zhu, J.; Jiang, T.; Zhong, Y.; Tie, Y.; Wu, Y.; Zheng, X.; Jin, Y.; Fu, H. Identification of lncRNA MEG3 binding protein using MS2-Tagged RNA affinity purification and mass spectrometry. Appl. Biochem. Biotechnol. 2015, 176, 1834–1845. [Google Scholar] [CrossRef]
- Zhang, X.; Zhou, Y.; Mehta, K.R.; Danila, D.C.; Scolavino, S.; Johnson, S.R.; Klibanski, A. A pituitary-derived MEG3 isoform functions as a growth suppressor in tumor cells. J. Clin. Endocrinol. Metab. 2003, 88, 5119–5126. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhong, Y.; Wang, Y.; Zhang, X.; Batista, D.L.; Gejman, R.; Ansell, P.J.; Zhao, J.; Weng, C.; Klibanski, A. Activation of p53 by MEG3 non-coding RNA. J. Biol. Chem. 2007, 282, 24731–24742. [Google Scholar] [CrossRef]
- Gong, L.; Xu, H.; Chang, H.; Tong, Y.; Zhang, T.; Guo, G. Knockdown of long non-coding RNA MEG3 protects H9c2 cells from hypoxia-induced injury by targeting microRNA-183. J. Cell Biochem. 2018, 119, 1429–1440. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.J.; Zheng, H.Q.; Chen, B.Y.; Sun, L.; Ma, M.M.; Wang, G.L.; Guan, Y.Y. WNK1 is required for proliferation induced by hypotonic challenge in rat vascular smooth muscle cells. Acta Pharmacol. Sin. 2018, 39, 35–47. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Xu, M.; Lu, P.; Zhou, F. MicroRNA-769 is downregulated in colorectal cancer and inhibits cancer progression by directly targeting cyclin-dependent kinase 1. Onco Targets Ther. 2018, 11, 9013–9025. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; He, J.; Gao, P.; Niu, Y.; Zhang, J.; Wang, L.; Liu, M.; Wei, X.; Liu, C.; Zhang, C.; et al. MiR-769-5p suppressed cell proliferation, migration and invasion by targeting TGFBR1 in non-small cell lung carcinoma. Oncotarget 2017, 8, 113558–113570. [Google Scholar] [CrossRef]
- Busch, A.; Busch, M.; Scholz, C.J.; Kellersmann, R.; Otto, C.; Chernogubova, E.; Maegdefessel, L.; Zernecke, A.; Lorenz, U. Aneurysm miRNA Signature Differs, Depending on Disease Localization and Morphology. Int. J. Mol. Sci. 2016, 17, 81. [Google Scholar] [CrossRef]
- Xu, X.; Ha, C.H.; Wong, C.; Wang, W.; Hausser, A.; Pfizenmaier, K.; Olson, E.N.; McKinsey, T.A.; Jin, Z.G. Angiotensin II stimulates protein kinase D-dependent histone deacetylase 5 phosphorylation and nuclear export leading to vascular smooth muscle cell hypertrophy. Arterioscler. Thromb. Vasc. Biol. 2007, 27, 2355–2362. [Google Scholar] [CrossRef]
- Wang, M.; Ji, Y.; Cai, S.; Ding, W. MiR-206 suppresses the progression of coronary artery disease by modulating Vascular Endothelial Growth Factor (VEGF) Expression. Med. Sci. Monit. 2016, 22, 5011–5020. [Google Scholar] [CrossRef]
- Stahlhut, C.; Suárez, Y.; Lu, J.; Mishima, Y.; Giraldez, A.J. MiR-1 and miR-206 regulate angiogenesis by modulating VegfA expression in zebrafish. Development 2012, 139, 4356–4364. [Google Scholar] [CrossRef]
- Lai, J.G.; Tsai, S.M.; Tu, H.C.; Chen, W.C.; Kou, F.J.; Lu, J.W.; Wang, H.D.; Huang, C.L.; Yuh, C.H. Zebrafish WNK lysine deficient protein kinase 1 (wnk1) affects angiogenesis associated with VEGF signaling. PLoS ONE 2014, 9, e106129. [Google Scholar] [CrossRef] [PubMed]
- Pan, W.; Pham, V.N.; Stratman, A.N.; Castranova, D.; Kamei, M.; Kidd, K.R.; Lo, B.D.; Shaw, K.M.; Torres-Vazquez, J.; Mikelis, C.M.; et al. CDP-diacylglycerol synthetase-controlled phosphoinositide availability limits VEGFA signaling and vascular morphogenesis. Blood 2012, 120, 489–498. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Brugarolas, J.B.; Vazquez, F.; Reddy, A.; Sellers, W.R.; Kaelin, W.G., Jr. TSC2 regulates VEGF through mTOR-dependent and -independent pathways. Cancer Cell 2003, 4, 147–158. [Google Scholar] [CrossRef]
- Deroanne, C.F.; Bonjean, K.; Servotte, S.; Devy, L.; Colige, A.; Clausse, N.; Blacher, S.; Verdin, E.; Foidart, J.M.; Nusgens, B.V.; et al. Histone deacetylases inhibitors as anti-angiogenic agents altering vascular endothelial growth factor signaling. Oncogene 2002, 21, 427–436. [Google Scholar] [CrossRef]
- Urbich, C.; Rössig, L.; Kaluza, D.; Potente, M.; Boeckel, J.N.; Knau, A.; Diehl, F.; Geng, J.G.; Hofmann, W.K.; Zeiher, A.M.; et al. HDAC5 is a repressor of angiogenesis and determines the angiogenic gene expression pattern of endothelial cells. Blood 2009, 113, 5669–5679. [Google Scholar] [CrossRef]
- Wang, X.T.; Wu, X.D.; Lu, Y.X.; Sun, Y.H.; Zhu, H.H.; Liang, J.B.; He, W.K.; Zeng, Z.Y.; Li, L. Potential involvement of mir-30e-3p in myocardial injury induced by coronary microembolization via autophagy activation. Cell Physiol. Biochem. 2017, 44, 1995–2004. [Google Scholar] [CrossRef]
- Kim, S.H.; Kim, G.J.; Umemura, T.; Lee, S.G.; Cho, K.J. Aberrant expression of plasma microRNA-33a in an atherosclerosis-risk group. Mol. Biol. Rep. 2017, 44, 79–88. [Google Scholar] [CrossRef]
- Dong, J.; Liang, Y.Z.; Zhang, J.; Wu, L.J.; Wang, S.; Hua, Q.; Yan, Y.X. Potential role of lipometabolism-related micrornas in peripheral blood mononuclear cells as biomarkers for coronary artery disease. J. Atheroscler. Thromb. 2017, 24, 430–441. [Google Scholar] [CrossRef]
- Deng, X.; Qin, S.; Chen, Y.; Liu, H.Y.; Yuan, E.; Deng, H.; Liu, S.M. B-RCA revealed circulating miR-33a/b associates with serum cholesterol in type 2 diabetes patients at high risk of ASCVD. Diabetes Res. Clin. Pract. 2018, 140, 191–199. [Google Scholar] [CrossRef]
- Siedlecki-Wullich, D.; Català-Solsona, J.; Fábregas, C.; Hernández, I.; Clarimon, J.; Lleó, A.; Boada, M.; Saura, C.A.; Rodríguez-Álvarez, J.; Miñano-Molina, A.J. Altered microRNAs related to synaptic function as potential plasma biomarkers for Alzheimer’s disease. Alzheimers Res. Ther. 2019, 11, 46. [Google Scholar] [CrossRef]
- Ma, J.; Shang, S.; Wang, J.; Zhang, T.; Nie, F.; Song, X.; Zhao, H.; Zhu, C.; Zhang, R.; Hao, D. Identification of miR-22-3p, miR-92a-3p, and miR-137 in peripheral blood as biomarker for schizophrenia. Psychiatry Res. 2018, 265, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Mei, Q.; Liu, J.; Liu, Y.; Li, C.; Wang, H.; Li, H.; Chen, X.; Lan, X. Expression of proline-rich coiled-coil 2B protein in developing rat brains. Neurosci. Lett. 2013, 557 Pt B, 171–176. [Google Scholar] [CrossRef]
- Fujii, T.; Shimada, K.; Nakai, T.; Ohbayashi, C. MicroRNAs in smoking-related carcinogenesis: Biomarkers, functions, and therapy. J. Clin. Med. 2018, 7, 98. [Google Scholar] [CrossRef] [PubMed]

| Characteristic | CVD Population (n = 34) | Control Population (n = 19) | P |
|---|---|---|---|
| Age | 44.12 ± 10.07 1 | 36.58 ± 9.97 1 | 8.387 × 10−3 |
| 27–78 2 | 24–55 2 | ||
| Body Mass Index | 23.85 ± 2.35 1 | 23.12 ± 3.93 1 | 0.117 |
| 20.13–28.76 2 | 19.33–32.6 2 | ||
| Smoking: Current | 5 (14.7%) | 0 (0%) | 1.296 × 10−4 |
| Smoking: Former | 13 (38%) | 0 (0%) | |
| Smoking: Never | 16 (47%) | 19 (100%) | |
| Sex: Male | 17 (50%) | 9 (47%) | 1 |
| Sex: Female | 17 (50%) | 10 (53%) | |
| Signs and symptoms | |||
| Pain | 7 (20.6%) | NA | |
| Ankle-brachial index | 0.96 ± 0.048 1 | NA | |
| 0.71–0.99 2 | |||
| Extended anatomical classification | |||
| Great saphenous vein (above knee) | 23 (67.7%) | NA | |
| Great saphenous vein (below knee) | 7 (20.6%) | NA | |
| Small saphenous vein | 3 (8.8%) | NA | |
| Great and small saphenous vein | 1 (2.9%) | NA | |
| Medication | |||
| Micronized diosmin | 19 (55.9%) | NA | |
| Preparation with vitaminum C, hesperidin and Ruscus aculeatus extract | 10 (29.4%) | NA | |
| Both medications | 5 (14.7%) | NA | |
| No. | miRNA Transcript | miRNA ID 1 | P | Fold Change | PLS Coefficient | ROC-AUC |
|---|---|---|---|---|---|---|
| Upregulated miRNA Transcripts | ||||||
| 1. | hsa-mir-122_hsa-miR-122-5p | hsa-miR-122-5p | 1.06 × 10−9 | 2.2135 | 4.71 × 10−2 | 0.930 |
| 2. | hsa-mir-3591_hsa-miR-3591-3p | hsa-miR-3591-3p | 1.06 × 10−9 | 2.2127 | 4.71 × 10−2 | 0.930 |
| 3. | hsa-mir-183_hsa-miR-183-5p | hsa-miR-183-5p | 2.05 × 10−6 | 1.9316 | 3.83 × 10−2 | 0.855 |
| 4. | hsa-mir-1277_hsa-miR-1277-3p | hsa-miR-1277-3p | 2.13 × 10−5 | 1.7727 | 4.04 × 10−2 | 0.850 |
| 5. | hsa-mir-548d-1_hsa-miR-548d-3p | hsa-miR-548d-3p | 2.13 × 10−5 | 1.6170 | 2.09 × 10−2 | 0.859 |
| 6. | hsa-mir-34a_hsa-miR-34a-5p | hsa-miR-34a-5p | 3.81 × 10−5 | 1.9308 | 3.45 × 10−2 | 0.847 |
| 7. | hsa-mir-576_hsa-miR-576-3p | hsa-miR-576-3p | 3.04 × 10−4 | 2.0430 | 3.21 × 10−2 | 0.842 |
| 8. | hsa-mir-454_hsa-miR-454-3p | hsa-miR-454-3p | 3.04 × 10−4 | 1.2133 | 1.05 × 10−2 | 0.833 |
| 9. | hsa-mir-548d-1_hsa-miR-548d-5p | hsa-miR-548d-5p | 3.44 × 10−4 | 1.3487 | 1.47 × 10−2 | 0.836 |
| 10. | hsa-mir-186_hsa-miR-186-3p | hsa-miR-186-3p | 3.61 × 10−4 | 1.3568 | 1.65 × 10−2 | 0.814 |
| 11. | hsa-mir-548d-2_hsa-miR-548d-5p | hsa-miR-548d-5p | 3.61 × 10−4 | 1.3498 | 1.47 × 10−2 | 0.811 |
| 12. | hsa-mir-548aa-1_hsa-miR-548aa | hsa-miR-548aa | 5.13 × 10−4 | 1.3248 | 1.46 × 10−2 | 0.819 |
| 13. | hsa-mir-548aa-2_hsa-miR-548aa | hsa-miR-548aa | 1.02 × 10−3 | 1.3381 | 1.46 × 10−2 | 0.797 |
| 14. | hsa-mir-33a_hsa-miR-33a-5p | hsa-miR-33a-5p | 1.02 × 10−3 | 1.2067 | 1.13 × 10−2 | 0.816 |
| 15. | hsa-mir-590_hsa-miR-590-3p | hsa-miR-590-3p | 1.02 × 10−3 | 1.1660 | 6.74 × 10−3 | 0.816 |
| 16. | hsa-mir-548t_hsa-miR-548t-3p | hsa-miR-548t-3p | 1.81 × 10−3 | 1.3233 | 8.10 × 10−3 | 0.796 |
| 17. | hsa-mir-1277_hsa-miR-1277-5p | hsa-miR-1277-5p | 1.84 × 10−3 | 1.3291 | 2.13 × 10−2 | 0.811 |
| 18. | hsa-let-7b_hsa-let-7b-3p | hsa-let-7b-3p | 2.06 × 10−3 | 1.3223 | 1.09 × 10−2 | 0.791 |
| 19. | hsa-mir-96_hsa-miR-96-5p | hsa-miR-96-5p | 3.73 × 10−3 | 2.2914 | 2.64 × 10−2 | 0.786 |
| 20. | hsa-mir-548ac_hsa-miR-548ac | hsa-miR-548ac | 5.53 × 10−3 | 1.7613 | 2.87 × 10−2 | 0.807 |
| 21. | hsa-mir-19a_hsa-miR-19a-3p | hsa-miR-19a-3p | 5.82 × 10−3 | 1.1944 | 8.38 × 10−3 | 0.757 |
| 22. | hsa-mir-206_hsa-miR-206 | hsa-miR-206 | 8.00 × 10−3 | 2.0356 | 2.76 × 10−2 | 0.759 |
| 23. | hsa-mir-497_hsa-miR-497-3p | hsa-miR-497-3p | 9.31 × 10−3 | 1.4368 | 1.63 × 10−2 | 0.782 |
| 24. | hsa-mir-208a_hsa-miR-208a-3p | hsa-miR-208a-3p | 9.81 × 10−3 | 3.2080 | 2.77 × 10−2 | 0.789 |
| Downregulated miRNA transcripts | ||||||
| 25. | hsa-mir-92a-1_hsa-miR-92a-3p | hsa-miR-92a-3p | 7.89 × 10−5 | 0.8323 | −1.40 × 10−2 | 0.856 |
| 26. | hsa-mir-874_hsa-miR-874-5p | hsa-miR-874-5p | 1.29 × 10−4 | 0.5428 | −3.43 × 10−2 | 0.916 |
| 27. | hsa-mir-106b_hsa-miR-106b-3p | hsa-miR-106b-3p | 2.47 × 10−4 | 0.7964 | −1.15 × 10−2 | 0.902 |
| 28. | hsa-mir-92a-2_hsa-miR-92a-3p | hsa-miR-92a-3p | 3.04 × 10−4 | 0.8414 | −1.43 × 10−2 | 0.842 |
| 29. | hsa-mir-181a-2_hsa-miR-181a-2-3p | hsa-miR-181a-2-3p | 1.02 × 10−3 | 0.6772 | −3.24 × 10−2 | 0.793 |
| 30. | hsa-mir-128-1_hsa-miR-128-3p | hsa-miR-128-3p | 2.67 × 10−3 | 0.8504 | −7.84 × 10−3 | 0.777 |
| 31. | hsa-mir-769_hsa-miR-769-5p | hsa-miR-769-5p | 5.53 × 10−3 | 0.8706 | −1.15 × 10−2 | 0.794 |
| 32. | hsa-mir-30e_hsa-miR-30e-3p | hsa-miR-30e-3p | 5.53 × 10−3 | 0.7400 | −1.51 × 10−2 | 0.805 |
| 33. | hsa-mir-1250_hsa-miR-1250-5p | hsa-miR-1250-5p | 8.56 × 10−3 | 0.6186 | −3.32 × 10−2 | 0.803 |
| 34. | hsa-mir-25_hsa-miR-25-3p | hsa-miR-25-3p | 8.94 × 10−3 | 0.8603 | −9.00 × 10−3 | 0.766 |
| No. | Gene Symbol | Gene Name | p Value | Fold Change | PLS Coefficient | ROC-AUC |
|---|---|---|---|---|---|---|
| Upregulated Genes | ||||||
| 1. | TSC2 | TSC complex subunit 2 | 4.87 × 10−17 | 1.437 | 8.197 × 10−4 | 1.000 |
| 2. | TBC1D22A | TBC1 domain family member 22A | 4.36 × 10−11 | 1.431 | 7.572 × 10−4 | 1.000 |
| 3. | PPP6R2 | protein phosphatase 6 regulatory subunit 2 | 9.52 × 10−9 | 1.361 | 6.225 × 10−4 | 1.000 |
| 4. | UPF1 | UPF1, RNA helicase and ATPase | 2.82 × 10−7 | 1.247 | 5.077 × 10−4 | 1.000 |
| 5. | WNK1 | WNK lysine deficient protein kinase 1 | 4.59 × 10−7 | 1.258 | 4.134 × 10−4 | 1.000 |
| 6. | CDS2 | CDP-diacylglycerol synthase 2 | 5.31 × 10−7 | 1.241 | 4.756 × 10−4 | 1.000 |
| 7. | PRRC2B | proline rich coiled-coil 2B | 1.56 × 10−6 | 1.273 | 4.693 × 10−4 | 1.000 |
| 8. | HDAC5 | histone deacetylase 5 | 4.89 × 10−6 | 1.432 | 5.694 × 10−4 | 1.000 |
| 9. | INTS11 (CPSF3L) | integrator complex subunit 11 | 5.95 × 10−6 | 1.246 | 4.683 × 10−4 | 1.000 |
| Downregulated genes | ||||||
| 10. | AC078899.1 | Unmatched | 1.18 × 10−13 | 0.393 | −1.586 × 10−3 | 1.000 |
| 11. | RP11-16F15.1 | Unmatched | 1.18 × 10−13 | 0.327 | −2.068 × 10−3 | 1.000 |
| 12. | EEF1A1P19 | eukaryotic translation elongation factor 1 alpha 1 pseudogene 19 | 8.40 × 10−13 | 0.500 | −1.305 × 10−3 | 1.000 |
| 13. | PFN1P1 | profilin 1 pseudogene 1 | 4.04 × 10−11 | 0.367 | −1.457 × 10−3 | 1.000 |
| 14. | RP4-706A16.3 | Unmatched | 4.36 × 10−11 | 0.455 | −1.394 × 10−3 | 1.000 |
| 15. | AC005884.1 | Unmatched | 4.36 × 10−11 | 0.401 | −1.488 × 10−3 | 1.000 |
| 16. | CALM2P2 | calmodulin 2 pseudogene 2 | 4.36 × 10−11 | 0.386 | −1.498 × 10−3 | 1.000 |
| 17. | HSPA8P1 | heat shock protein family A (Hsp70) member 8 pseudogene 1 | 4.36 × 10−11 | 0.379 | −1.575 × 10−3 | 1.000 |
| 18. | RP11-490H24.5 | Unmatched | 4.36 × 10−11 | 0.312 | −1.508 × 10−3 | 1.000 |
| 19. | EIF4A1P10 | eukaryotic translation initiation factor 4A1 pseudogene 10 | 4.66 × 10−11 | 0.461 | −1.286 × 10−3 | 1.000 |
| 20. | RP11-1033A18.1 | Unmatched | 7.00 × 10−11 | 0.381 | −1.495 × 10−3 | 1.000 |
| 21. | EIF3FP3 | eukaryotic translation initiation factor 3 subunit F pseudogene 3 | 1.35 × 10−10 | 0.443 | −1.423 × 10−3 | 1.000 |
| 22. | PDIA3P1 (PDIA3P) | protein disulfide isomerase family A member 3 pseudogene 1 | 2.38 × 10−10 | 0.465 | −1.240 × 10−3 | 1.000 |
| 23. | HSPA9P1 | heat shock protein family A (Hsp70) member 9 pseudogene 1 | 2.76 × 10−10 | 0.420 | −1.414 × 10−3 | 1.000 |
| 24. | AC007238.1 | Unmatched | 3.62 × 10−10 | 0.422 | −1.398 × 10−3 | 1.000 |
| 25. | HNRNPA1P7 | heterogeneous nuclear ribonucleoprotein A1 pseudogene 7 | 3.72 × 10−10 | 0.462 | −1.232 × 10−3 | 1.000 |
| 26. | RP11-159C21.4 | Unmatched | 4.81 × 10−10 | 0.390 | −1.552 × 10−3 | 1.000 |
| 27. | PABPC3 | poly(A) binding protein cytoplasmic 3 | 1.70 × 10−9 | 0.414 | −1.468 × 10−3 | 1.000 |
| 28. | RP11-74E24.2 | Unmatched | 1.94 × 10−9 | 0.537 | −1.067 × 10−3 | 1.000 |
| 29. | EEF1A1P6 | eukaryotic translation elongation factor 1 alpha 1 pseudogene 6 | 1.94 × 10−9 | 0.441 | −1.375 × 10−3 | 1.000 |
| 30. | XRCC6P2 | X-ray repair cross complementing 6 pseudogene 2 | 2.89 × 10−9 | 0.373 | −1.535 × 10−3 | 1.000 |
| 31. | HNRNPKP2 | heterogeneous nuclear ribonucleoprotein K pseudogene 2 | 3.13 × 10−9 | 0.424 | −1.163 × 10 ^-3 | 1.000 |
| 32. | EEF1A1P11 | eukaryotic translation elongation factor 1 alpha 1 pseudogene 11 | 8.40 × 10−9 | 0.448 | −1.369 × 10−3 | 1.000 |
| 33. | UBA52P5 | ubiquitin A-52 residue ribosomal protein fusion product 1 pseudogene 5 | 8.40 × 10−9 | 0.397 | −1.306 × 10−3 | 1.000 |
| 34. | RPL9P7 | ribosomal protein L9 pseudogene 7 | 9.10 × 10−9 | 0.414 | −1.417 × 10−3 | 1.000 |
| 35. | RPS21P4 | ribosomal protein S21 pseudogene 4 | 1.37 × 10−8 | 0.376 | −1.531 × 10−3 | 1.000 |
| 36. | RP11-334L9.1 | Unmatched | 1.37 × 10−8 | 0.333 | −1.206 × 10−3 | 1.000 |
| 37. | HNRNPKP4 | heterogeneous nuclear ribonucleoprotein K pseudogene 4 | 1.38 × 10−8 | 0.462 | −1.120 × 10−3 | 1.000 |
| 38. | RPL9P9 | ribosomal protein L9 pseudogene 9 | 1.38 × 10−8 | 0.418 | −1.302 × 10−3 | 1.000 |
| 39. | AC138123.2 | Unmatched | 1.38 × 10−8 | 0.407 | −1.422 × 10−3 | 1.000 |
| 40. | HNRNPA1P10 | heterogeneous nuclear ribonucleoprotein A1 pseudogene 10 | 1.39 × 10−8 | 0.475 | −1.227 × 10−3 | 1.000 |
| 41. | MORF4L1P1 | mortality factor 4 like 1 pseudogene 1 | 3.98 × 10−8 | 0.535 | −1.045 × 10−3 | 1.000 |
| 42. | RP11-676M6.1 | Unmatched | 8.19 × 10−8 | 0.498 | −1.208 × 10−3 | 1.000 |
| 43, | RPL7AP66 | ribosomal protein L7a pseudogene 66 | 9.71 × 10−8 | 0.485 | −1.089 × 10−3 | 1.000 |
| 44. | RP11-680H20.1 | Unmatched | 9.99 × 10−8 | 0.411 | −1.155 × 10−3 | 1.000 |
| 45. | CTB-13H5.1 | Unmatched | 1.41 × 10−7 | 0.418 | −1.175 × 10−3 | 1.000 |
| 46. | HNRNPA1P35 | heterogeneous nuclear ribonucleoprotein A1 pseudogene 35 | 1.49 × 10−7 | 0.350 | −1.223 × 10−3 | 1.000 |
| 47. | PTBP1P | polypyrimidine tract binding protein 1 pseudogene | 1.53 × 10−7 | 0.443 | −1.095 × 10−3 | 1.000 |
| 48. | API5P1 | apoptosis inhibitor 5 pseudogene 1 | 1.57 × 10−7 | 0.347 | −1.204 × 10−3 | 1.000 |
| 49. | UBE2D3P1 | ubiquitin conjugating enzyme E2 D3 pseudogene 1 | 1.69 × 10−7 | 0.485 | −8.801 × 10−4 | 1.000 |
| 50. | AL162151.3 | Unmatched | 1.94 × 10−7 | 0.431 | −1.258 × 10−3 | 1.000 |
| 51. | RPL9P8 | ribosomal protein L9 pseudogene 8 | 2.34 × 10−7 | 0.446 | −1.253 × 10−3 | 1.000 |
| 52. | EEF1A1P13 | eukaryotic translation elongation factor 1 alpha 1 pseudogene 13 | 2.51 × 10−7 | 0.521 | −1.211 × 10−3 | 1.000 |
| 53. | PABPC1P4 | poly(A) binding protein cytoplasmic 1 pseudogene 4 | 2.60 × 10−7 | 0.465 | −1.031 × 10−3 | 1.000 |
| 54. | HNRNPUP1 | heterogeneous nuclear ribonucleoprotein U pseudogene 1 | 2.73 × 10−7 | 0.441 | −1.106 × 10−3 | 1.000 |
| 55. | ARPC3P1 | actin related protein 2/3 complex subunit 3 pseudogene 1 | 3.72 × 10−7 | 0.331 | −1.272 × 10−3 | 1.000 |
| 56. | PTP4A2P1 | protein tyrosine phosphatase type IVA, member 2 pseudogene 1 | 4.59 × 10−7 | 0.500 | −9.014 × 10−4 | 1.000 |
| 57. | CTC-451P13.1 | Unmatched | 4.77 × 10−7 | 0.513 | −9.263 × 10−4 | 1.000 |
| 58. | BZW1P2 | basic leucine zipper and W2 domains 1 pseudogene 2 | 7.97 × 10−7 | 0.445 | −9.598 × 10−4 | 1.000 |
| 59. | RP11-318C24.1 | Unmatched | 1.94 × 10−6 | 0.314 | −1.062 × 10−3 | 0.980 |
| 60. | OTUD4P1 (HIN1L) | OTUD4 pseudogene 1 | 2.08 × 10−6 | 0.480 | −9.962 × 10−4 | 1.000 |
| 61. | EIF3LP2 | eukaryotic translation initiation factor 3 subunit L pseudogene 2 | 2.33 × 10−6 | 0.457 | −9.995 × 10−4 | 1.000 |
| 62. | RAC1P2 | Rac family small GTPase 1 pseudogene 2 | 3.29 × 10−6 | 0.489 | −8.192 × 10−4 | 0.980 |
| Functional Analysis of Upregulated Genes (CDS2, HDAC5, PPP6R2, PRRC2B, TBC1D22A, WNK1) | |
|---|---|
| KEGG, Reactome, GAD and GAD Class | |
| CDS2 | KEGG: Glycerophospholipid metabolism, Phosphatidylinositol signaling system, Metabolic pathways |
| Reactome: Synthesis of PG (Phosphatidylglycerol) | |
| GAD: Type 2 Diabetes|edema|rosiglitazone, Tobacco Use Disorder | |
| GAD Class: pharmacogenomic, chemdependency | |
| HDAC5 | KEGG: Alcoholism, Viral carcinogenesis, |
| Reactome: NOTCH1 Intracellular Domain Regulates Transcription, Constitutive Signaling by NOTCH1 PEST Domain Mutants, Constitutive Signaling by NOTCH1 HD + PEST Domain Mutants | |
| GAD: antidepressant response, Bone Density, Bone mineral density (hip), Bone mineral density (spine), bronchodilator response, Fractures, Bone, Type 2 Diabetes| edema | rosiglitazone | |
| GAD Class: immune, metabolic, pharmacogenomic | |
| PPP6R2 | No information |
| PRRC2B | No information |
| TBC1D22A | GAD: Albumins, Arteries, Attention Deficit Disorder with Hyperactivity, Blood Pressure, Body Mass Index, Body Weight, Breath Tests, Cardiomegaly, Cholesterol, Erythrocyte Count, Fibrinogen, Heart Failure, Heart Rate, Leukocyte Count, longevity, Metabolism, Myocardial Infarction, Parkinson Disease, Resistin, Stroke, Thyrotropin, Tobacco Use Disorder, Waist Circumference, Waist-Hip Ratio |
| GAD Class: aging, cardiovascular, chemdependency, hematological, immune, metabolic, neurological, other, psych | |
| WNK1 | Reactome: Stimuli-sensing channels |
| GAD: Apoplexy|Brain Ischemia|Stroke, blood pressure, arterial, Chronic renal failure|Kidney Failure, Chronic, Essential Hypertension, Hereditary Sensory and Autonomic Neuropathies, HIV Infections|[X]Human immunodeficiency virus disease, hypertension, null, Tobacco Use Disorder, Type 2 Diabetes| edema | rosiglitazone | |
| GAD Class: cardiovascular, chemdependency, infection, neurological, pharmacogenomic, renal, unknown | |
| Gene Ontology terms associated with EASE score <0.1 | |
| GO Biological Process | cellular developmental process, positive regulation of molecular function |
| GO Molecular Function | enzyme binding |
| Functional analysis of downregulated gene (PABPC3) | |
| KEGG, Reactome, GAD and GAD Class | |
| PABPC3 | KEGG: RNA transport, mRNA surveillance pathway, RNA degradation |
| GAD: Body Mass Index, Body Weight, Body Weight Changes, Glomerular Filtration Rate | |
| GAD Class: metabolic, renal | |
| Gene Ontology terms associated with PABPC3 | |
| GO Biological Process | nucleobase-containing compound metabolic process, cellular aromatic compound metabolic process, nitrogen compound metabolic process, metabolic process, cellular process, RNA metabolic process, mRNA metabolic process, cellular nitrogen compound metabolic process, macromolecule metabolic process, cellular metabolic process, primary metabolic process, cellular macromolecule metabolic process, heterocycle metabolic process, organic substance metabolic process, nucleic acid metabolic process, organic cyclic compound metabolic process |
| GO Molecular Function | nucleotide binding, nucleic acid binding, RNA binding, single-stranded RNA binding, binding, poly(A) binding, small molecule binding, poly-purine tract binding, organic cyclic compound binding, nucleoside phosphate binding, heterocyclic compound binding |
| GO Cellular Component | extracellular region, intracellular, cell, cytoplasm, vesicle, membrane-bounded vesicle, organelle, membrane-bounded organelle, extracellular organelle, extracellular region part, intracellular part, cell part, extracellular exosome, extracellular vesicle |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zalewski, D.P.; Ruszel, K.P.; Stępniewski, A.; Gałkowski, D.; Bogucki, J.; Komsta, Ł.; Kołodziej, P.; Chmiel, P.; Zubilewicz, T.; Feldo, M.; et al. Dysregulations of MicroRNA and Gene Expression in Chronic Venous Disease. J. Clin. Med. 2020, 9, 1251. https://doi.org/10.3390/jcm9051251
Zalewski DP, Ruszel KP, Stępniewski A, Gałkowski D, Bogucki J, Komsta Ł, Kołodziej P, Chmiel P, Zubilewicz T, Feldo M, et al. Dysregulations of MicroRNA and Gene Expression in Chronic Venous Disease. Journal of Clinical Medicine. 2020; 9(5):1251. https://doi.org/10.3390/jcm9051251
Chicago/Turabian StyleZalewski, Daniel P., Karol P. Ruszel, Andrzej Stępniewski, Dariusz Gałkowski, Jacek Bogucki, Łukasz Komsta, Przemysław Kołodziej, Paulina Chmiel, Tomasz Zubilewicz, Marcin Feldo, and et al. 2020. "Dysregulations of MicroRNA and Gene Expression in Chronic Venous Disease" Journal of Clinical Medicine 9, no. 5: 1251. https://doi.org/10.3390/jcm9051251
APA StyleZalewski, D. P., Ruszel, K. P., Stępniewski, A., Gałkowski, D., Bogucki, J., Komsta, Ł., Kołodziej, P., Chmiel, P., Zubilewicz, T., Feldo, M., Kocki, J., & Bogucka-Kocka, A. (2020). Dysregulations of MicroRNA and Gene Expression in Chronic Venous Disease. Journal of Clinical Medicine, 9(5), 1251. https://doi.org/10.3390/jcm9051251







