Pain Neuroscience Education and Physical Therapeutic Exercise for Patients with Chronic Spinal Pain in Spanish Physiotherapy Primary Care: A Pragmatic Randomized Controlled Trial
Abstract
1. Introduction
2. Experimental Section
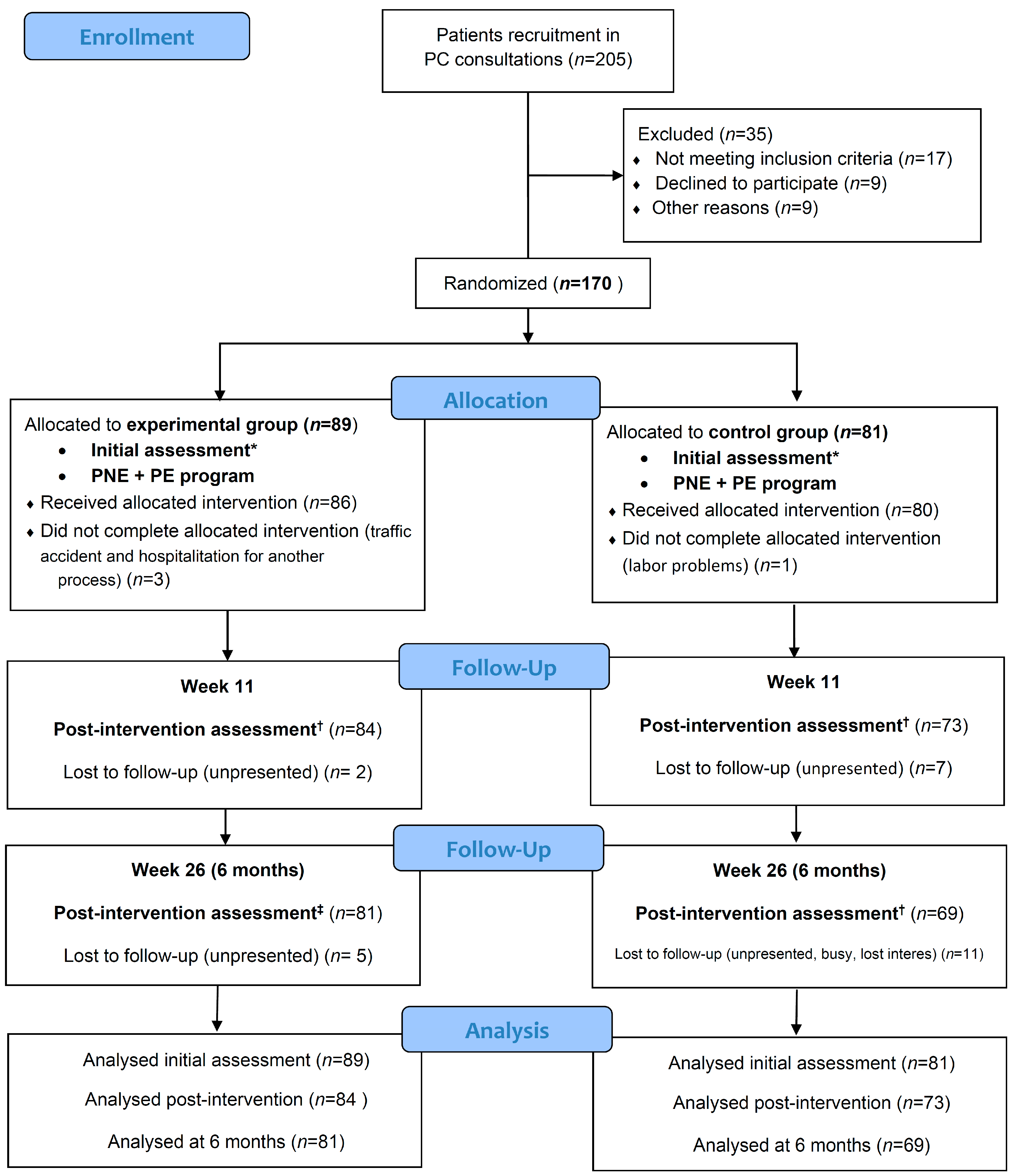
2.1. Patient Recruitment
2.2. Sample Size Calculation
2.3. Randomization
2.4. Blinding
2.5. Interventions
2.5.1. Experimental Group Intervention
2.5.2. Control Group Intervention
2.6. Outcome Variables
2.6.1. Personal and Sociodemographic Variables
2.6.2. Primary Outcome Variables
2.6.3. Secondary Outcome Variables
2.7. Statistical Analysis
3. Results
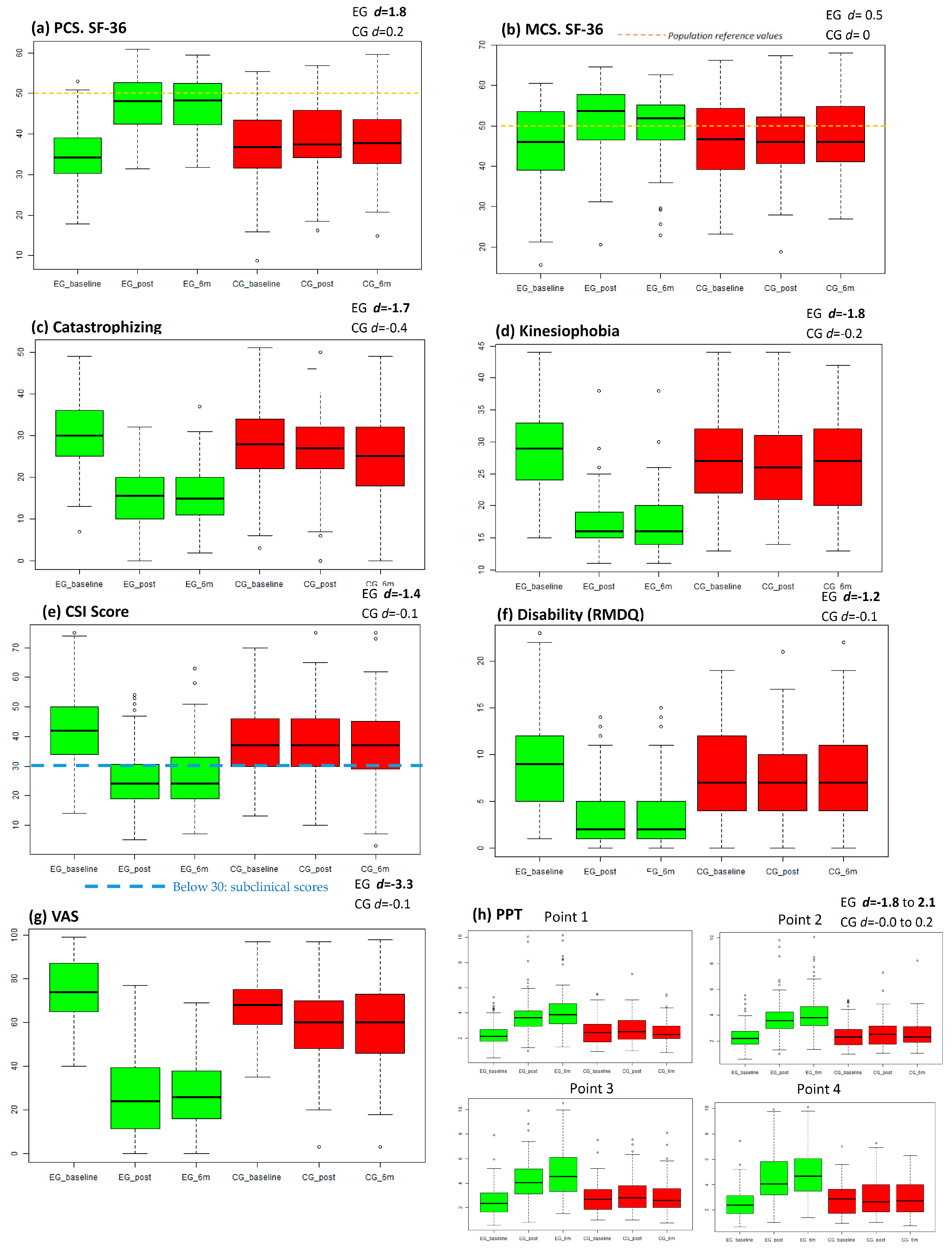
Primary and Secondary Outcomes
4. Discussion
4.1. Practical Implications and Recommendations for Research
4.2. Limitations and Strengths
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CMP | Chronic musculoskeletal pain |
| CNS | Central nervous system |
| CS | Central sensitization |
| CSI | Central sensitization inventory |
| CSP | Chronic spinal pain |
| CSQ-8 | Client satisfaction questionnaire |
| EG | Experimental group |
| HRQL | Health-related quality of life |
| PC | Primary care |
| PCS | Pain catastrophizing scale |
| PE | Physical exercise |
| PNE | Pain neuroscience education |
| PPTs | Pain pressure thresholds |
| RMDQ | Roland–Morris disability questionnaire |
| TSK-11 | Tampa Scale for Kinesiophobia-11 |
| VAS | Visual analogue scale |
Appendix A
References
- Langley, P.C. The prevalence, correlates and treatment of pain in the European Union. Curr. Med. Res. Opin. 2011, 27, 463–480. [Google Scholar] [CrossRef] [PubMed]
- Breivik, H.; Eisenberg, E.; O’Brien, T. The individual and societal burden of chronic pain in Europe: The case for strategic prioritisation and action to improve knowledge and availability of appropriate care. BMC Public Health 2013, 13, 1229. [Google Scholar] [CrossRef] [PubMed]
- James, S.L.; Abate, D.; Abate, K.H.; Abay, S.M.; Abbafati, C.; Abbasi, N.; Abbastabar, H.; Abd-Allah, F.; Abdela, J.; Abdelalim, A.; et al. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2018, 392, 1789–1858. [Google Scholar] [CrossRef]
- Langley, P.; Müller-Schwefe, G.; Nicolaou, A.; Liedgens, H.; Pergolizzi, J.; Varrassi, G. The societal impact of pain in the European Union: Health-related quality of life and healthcare resource utilization. J. Med. Econ. 2010, 13, 571–581. [Google Scholar] [CrossRef] [PubMed]
- Barbaglia, G.; Adroher, N.D.; Vilagut, G.; Bruffaerts, R.; Bunting, B.; Caldas de Almeida, J.M.; Florescu, S.; de Girolamo, G.; de Graaf, R.; Haro, J.M.; et al. Health conditions and role limitation in three European Regions: A public-health perspective. Gac. Sanit. 2017, 31, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Torralba, A.; Miquel, A.; Darba, J. Situación actual del dolor cronico en España: Iniciativa “Pain Proposal”. Revista de la Sociedad Española del Dolor 2014, 21, 16–22. [Google Scholar] [CrossRef]
- Loeser, J.D.; Schatman, M.E. Chronic pain management in medical education: A disastrous omission. Postgrad. Med. 2017, 129, 332–335. [Google Scholar] [CrossRef]
- Watt-Watson, J.; McGillion, M.; Hunter, J.; Choiniere, M.; Clark, A.J.; Dewar, A.; Johnston, C.; Lynch, M.; Morley-Forster, P.; Moulin, D.; et al. A survey of prelicensure pain curricula in health science faculties in Canadian universities. Pain Res. Manag. 2009, 14, 439–444. [Google Scholar] [CrossRef]
- Foster, N.E.; Anema, J.R.; Cherkin, D.; Chou, R.; Cohen, S.P.; Gross, D.P.; Ferreira, P.H.; Fritz, J.M.; Koes, B.W.; Peul, W.; et al. Prevention and treatment of low back pain: Evidence, challenges, and promising directions. Lancet 2018, 391, 2368–2383. [Google Scholar] [CrossRef]
- Wong, J.J.; Côté, P.; Sutton, D.A.; Randhawa, K.; Yu, H.; Varatharajan, S.; Goldgrub, R.; Nordin, M.; Gross, D.P.; Shearer, H.M.; et al. Clinical practice guidelines for the noninvasive management of low back pain: A systematic review by the Ontario Protocol for Traffic Injury Management (OPTIMa) Collaboration. Eur. J. Pain 2017, 21, 201–216. [Google Scholar] [CrossRef]
- Stochkendahl, M.J.; Kjaer, P.; Hartvigsen, J.; Kongsted, A.; Aaboe, J.; Andersen, M.Ø.M.M.; Andersen, M.Ø.M.M.; Fournier, G.; Højgaard, B.; Jensen, M.B.; et al. National Clinical Guidelines for non-surgical treatment of patients with recent onset low back pain or lumbar radiculopathy. Eur. Spine J. 2018, 27, 60–75. [Google Scholar] [CrossRef] [PubMed]
- Qaseem, A.; Wilt, T.J.; McLean, R.M.; Forciea, M.A. Noninvasive Treatments for Acute, Subacute, and Chronic Low Back Pain: A Clinical Practice Guideline from the American College of Physicians. Ann. Intern. Med. 2017, 166, 514–530. [Google Scholar] [CrossRef] [PubMed]
- Babatunde, O.O.; Jordan, J.L.; Van der Windt, D.A.; Hill, J.C.; Foster, N.E.; Protheroe, J. Effective treatment options for musculoskeletal pain in primary care: A systematic overview of current evidence. PLoS ONE 2017, 12, e0178621. [Google Scholar] [CrossRef] [PubMed]
- Lin, I.; Wiles, L.; Waller, R.; Goucke, R.; Nagree, Y.; Gibberd, M.; Straker, L.; Maher, C.G.; O’Sullivan, P.P.B. What does best practice care for musculoskeletal pain look like? Eleven consistent recommendations from high-quality clinical practice guidelines: Systematic review. Br. J. Sports Med. 2019. [Google Scholar] [CrossRef]
- Aguirrezabal-Bazterrica, I.; Galán-Martín, M.; Montero-Cuadrado, F. Abordaje no farmacológico del dolor. FMC-Formación Médica Continuada en Atención Primaria 2020, 27, 145–153. [Google Scholar] [CrossRef]
- Agencia Española de Medicamentos y Productos Sanitarios. Utilización de Medicamentos Opioides en España Durante el Periodo 2008–2015. Ministerio de Sanidad 2017. [Google Scholar]
- Calabozo-Freile, B. Opioides, del poco uso… al abuso. Portal del Medicamento El Ojo de Markov 2017, 59, 1–5. [Google Scholar]
- Sivasubramaniam, V.; Patel, H.C.; Ozdemir, B.A.; Papadopoulos, M.C. Trends in hospital admissions and surgical procedures for degenerative lumbar spine disease in England: A 15-year time-series study. BMJ Open 2015, 5, e009011. [Google Scholar] [CrossRef]
- Longo, D.L.; Volkow, N.D.; McLellan, A.T. Opioid Abuse in Chronic Pain—Misconceptions and Mitigation Strategies. N. Engl. J. Med. 2016, 374, 1253–1263. [Google Scholar]
- Stopka, T.J.; Amaravadi, H.; Kaplan, A.R.; Hoh, R.; Bernson, D.; Chui, K.K.H.; Land, T.; Walley, A.Y.; LaRochelle, M.R.; Rose, A.J. Opioid overdose deaths and potentially inappropriate opioid prescribing practices (PIP): A spatial epidemiological study. Int. J. Drug Policy 2019, 68, 37–45. [Google Scholar] [CrossRef]
- Compton, W.M.; Jones, C.M. Epidemiology of the U.S. opioid crisis: The importance of the vector. Ann. N. Y. Acad. Sci. 2019, 1451, 130–143. [Google Scholar] [CrossRef] [PubMed]
- Bell, A.; Bennett, A.S.; Jones, T.S.; Doe-Simkins, M.; Williams, L.D. Amount of naloxone used to reverse opioid overdoses outside of medical practice in a city with increasing illicitly manufactured fentanyl in illicit drug supply. Subst. Abus. 2019, 40, 52–55. [Google Scholar] [CrossRef] [PubMed]
- Harte, S.E.; Harris, R.E.; Clauw, D.J. The neurobiology of central sensitization. J. Appl. Biobehav. Res. 2018, 23, e12137. [Google Scholar] [CrossRef]
- Ji, R.-R.; Nackley, A.; Huh, Y.; Terrando, N.; Maixner, W. Neuroinflammation and Central Sensitization in Chronic and Widespread Pain. Anesthesiology 2018, 129, 343–366. [Google Scholar] [CrossRef]
- Woolf, C.J. Central sensitization: Implications for the diagnosis and treatment of pain. Pain 2011, 152, S2–S15. [Google Scholar] [CrossRef]
- Nijs, J.; Van Houdenhove, B.; Oostendorp, R.A.B. Recognition of central sensitization in patients with musculoskeletal pain: Application of pain neurophysiology in manual therapy practice. Man. Ther. 2010, 15, 135–141. [Google Scholar] [CrossRef]
- Woolf, C.J.; Bennett, G.J.; Doherty, M.; Dubner, R.; Kidd, B.; Koltzenburg, M.; Lipton, R.; Loeser, J.D.; Payne, R.; Torebjork, E. Towards a mechanism-based classification of pain? Pain 1998, 77, 227–229. [Google Scholar] [CrossRef]
- Nijs, J.; Torres-Cueco, R.; van Wilgen, C.P.; Girbes, E.L.; Struyf, F.; Roussel, N.; van Oosterwijck, J.; Daenen, L.; Kuppens, K.; Vanwerweeen, L.; et al. Applying modern pain neuroscience in clinical practice: Criteria for the classification of central sensitization pain. Pain Physician 2014, 17, 447–457. [Google Scholar]
- Cuesta-Vargas, A.I.; Neblett, R.; Chiarotto, A.; Kregel, J.; Nijs, J.; van Wilgen, C.P.; Pitance, L.; Knezevic, A.; Gatchel, R.J.; Mayer, T.G.; et al. Dimensionality and Reliability of the Central Sensitization Inventory in a Pooled Multicountry Sample. J. Pain 2018, 19, 317–329. [Google Scholar] [CrossRef]
- May, A. Chronic pain may change the structure of the brain. Pain 2008, 137, 7–15. [Google Scholar] [CrossRef]
- Pelletier, R.; Higgins, J.; Bourbonnais, D. Is neuroplasticity in the central nervous system the missing link to our understanding of chronic musculoskeletal disorders? BMC Musculoskelet. Disord. 2015, 16, 25. [Google Scholar] [CrossRef] [PubMed]
- Pace, M.C.; Passavanti, M.B.; De Nardis, L.; Bosco, F.; Sansone, P.; Pota, V.; Barbarisi, M.; Palagiano, A.; Iannotti, F.A.; Panza, E.; et al. Nociceptor plasticity: A closer look. J. Cell. Physiol. 2018, 233, 2824–2838. [Google Scholar] [CrossRef] [PubMed]
- Loeser, J.D.; Treede, R.-D. The Kyoto protocol of IASP basic pain terminology. Pain 2008, 137, 473–477. [Google Scholar] [CrossRef] [PubMed]
- Picavet, H.S.J.; Vlaeyen, J.W.S.; Schouten, J.S.A.G. Pain catastrophizing and kinesiophobia: Predictors of chronic low back pain. Am. J. Epidemiol. 2002, 156, 1028–1034. [Google Scholar] [CrossRef] [PubMed]
- Luque-Suarez, A.; Martinez-Calderon, J.; Falla, D. Role of kinesiophobia on pain, disability and quality of life in people suffering from chronic musculoskeletal pain: A systematic review. Br. J. Sports Med. 2018. [Google Scholar] [CrossRef] [PubMed]
- Meints, S.M.; Edwards, R.R. Evaluating psychosocial contributions to chronic pain outcomes. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2018, 87, 168–182. [Google Scholar] [CrossRef]
- Jensen, M.P.; Gianas, A.; Sherlin, L.H.; Howe, J.D. Pain Catastrophizing and EEG—A Asymmetry. Clin J Pain. 2015, 31, 852–858. [Google Scholar] [CrossRef]
- Bunzli, S.; Smith, A.; Schütze, R.; Lin, I.; O’Sullivan, P. Making Sense of Low Back Pain and Pain-Related Fear. J. Orthop. Sports Phys. Ther. 2017, 47, 628–636. [Google Scholar] [CrossRef]
- Leeuw, M.; Goossens, M.E.J.B.; Linton, S.J.; Crombez, G.; Boersma, K.; Vlaeyen, J.W.S. The Fear-Avoidance Model of Musculoskeletal Pain: Current State of Scientific Evidence. J. Behav. Med. 2007, 30, 77–94. [Google Scholar] [CrossRef]
- Vlaeyen, J.W.S.; Linton, S.J. Fear-avoidance and its consequences in chronic musculoskeletal pain: A state of the art. Pain 2000, 85, 317–332. [Google Scholar] [CrossRef]
- Crombez, G.; Eccleston, C.; Van Damme, S.; Vlaeyen, J.W.S.; Karoly, P. Fear-Avoidance Model of Chronic Pain. Clin. J. Pain 2012, 28, 475–483. [Google Scholar] [CrossRef] [PubMed]
- Neblett, R.; Mayer, T.G.; Williams, M.J.; Asih, S.; Cuesta-Vargas, A.I.; Hartzell, M.M.; Gatchel, R.J. The Fear-avoidance Components Scale (FACS): Responsiveness to Functional Restoration Treatment in a Chronic Musculoskeletal Pain Disorder (CMPD) Population. Clin. J. Pain 2017, 33, 1088–1099. [Google Scholar] [CrossRef] [PubMed]
- Aguirrezabal, I.; Pérez de San Román, M.S.; Cobos-Campos, R.; Orruño, E.; Goicoechea, A.; Martínez de la Eranueva, R.; Arroniz, M.; Uzquiza, E. Effectiveness of a primary care-based group educational intervention in the management of patients with migraine: A randomized controlled trial. Prim. Health Care Res. Dev. 2019, 20, e155. [Google Scholar] [CrossRef] [PubMed]
- Meeus, M.; Nijs, J.; Van Oosterwijck, J.; Van Alsenoy, V.; Truijen, S. Pain physiology education improves pain beliefs in patients with chronic fatigue syndrome compared with pacing and self-management education: A double-blind randomized controlled trial. Arch. Phys. Med. Rehabil. 2010, 91, 1153–1159. [Google Scholar] [CrossRef] [PubMed]
- Louw, A.; Diener, I.; Butler, D.S.; Puentedura, E.J. The Effect of Neuroscience Education on Pain, Disability, Anxiety, and Stress in Chronic Musculoskeletal Pain. Arch. Phys. Med. Rehabil. 2011, 92, 2041–2056. [Google Scholar] [CrossRef]
- Louw, A.; Zimney, K.; Puentedura, E.J.; Diener, I. The efficacy of pain neuroscience education on musculoskeletal pain: A systematic review of the literature. Physiother. Theory Pract. 2016, 32, 332–355. [Google Scholar] [CrossRef]
- Watson, J.A.; Ryan, C.G.; Cooper, L.; Ellington, D.; Whittle, R.; Lavender, M.; Dixon, J.; Atkinson, G.; Cooper, K.; Martin, D.J. Pain Neuroscience Education for Adults with Chronic Musculoskeletal Pain: A Mixed-Methods Systematic Review and Meta-Analysis. J. Pain 2019, 20, 1140.e1–1140.e22. [Google Scholar] [CrossRef]
- Marris, D.; Theophanous, K.; Cabezon, P.; Dunlap, Z.; Donaldson, M. The impact of combining pain education strategies with physical therapy interventions for patients with chronic pain: A systematic review and meta-analysis of randomized controlled trials. Physiother. Theory Pract. 2019, 1–12. [Google Scholar] [CrossRef]
- Maeda, Y.; Kan, S.; Fujino, Y.; Shibata, M. Verbal Instruction Can Induce Extinction of Fear of Movement-Related Pain. J. Pain 2018, 19, 1063–1073. [Google Scholar] [CrossRef]
- Nijs, J.; Leysen, L.; Vanlauwe, J.; Logghe, T.; Ickmans, K.; Polli, A.; Malfliet, A.; Coppieters, I.; Huysmans, E. Treatment of central sensitization in patients with chronic pain: Time for change? Expert Opin. Pharmacother. 2019, 20, 1961–1970. [Google Scholar] [CrossRef]
- Amer-Cuenca, J.J.; Pecos-Martín, D.; Martínez-Merinero, P.; Girbés, E.L.; Nijs, J.; Meeus, M.; Peña, R.F.; Fernández-Carnero, J. How Much Is Needed? Comparison of the Effectiveness of Different Pain Education Dosages in Patients with Fibromyalgia. Pain Med. 2020, 21, 782–793. [Google Scholar] [CrossRef] [PubMed]
- Geneen, L.J.; Martin, D.J.; Adams, N.; Clarke, C.; Dunbar, M.; Jones, D.; McNamee, P.; Schofield, P.; Smith, B.H. Effects of education to facilitate knowledge about chronic pain for adults: A systematic review with meta-analysis. Syst. Rev. 2015, 4, 132. [Google Scholar] [CrossRef] [PubMed]
- Gardner, T.; Refshauge, K.; McAuley, J.; Hübscher, M.; Goodall, S.; Smith, L. Combined education and patient-led goal setting intervention reduced chronic low back pain disability and intensity at 12 months: A randomised controlled trial. Br. J. Sports Med. 2019. [Google Scholar] [CrossRef] [PubMed]
- Snodgrass, S.J.; Heneghan, N.R.; Tsao, H.; Stanwell, P.T.; Rivett, D.A.; Van Vliet, P.M. Recognising neuroplasticity in musculoskeletal rehabilitation: A basis for greater collaboration between musculoskeletal and neurological physiotherapists. Man. Ther. 2014, 19, 614–617. [Google Scholar] [CrossRef] [PubMed]
- Rogge, A.-K.; Röder, B.; Zech, A.; Hötting, K. Exercise-induced neuroplasticity: Balance training increases cortical thickness in visual and vestibular cortical regions. Neuroimage 2018, 179, 471–479. [Google Scholar] [CrossRef] [PubMed]
- Amir, S.; Batouli, H.; Saba, V.; Amir, S. At least eighty percent of brain grey matter is modifiable by physical activity: A review study Medical Imaging View project Electromagnetic waves View project At least eighty percent of brain grey matter is modifiable by physical activity: A review study. Behav. Brain Res. 2017, 332, 204–217. [Google Scholar]
- Papale, A.E.; Hooks, B.M. Circuit Changes in Motor Cortex During Motor Skill Learning. Neuroscience 2018, 368, 283–297. [Google Scholar] [CrossRef]
- Dayan, E.; Cohen, L.G. Neuroplasticity Subserving Motor Skill Learning. Neuron 2011, 72, 443–454. [Google Scholar] [CrossRef]
- El-Sayes, J.; Harasym, D.; Turco, C.V.; Locke, M.B.; Nelson, A.J. Exercise-Induced Neuroplasticity: A Mechanistic Model and Prospects for Promoting Plasticity. Neurosci 2018, 25, 65–85. [Google Scholar] [CrossRef]
- Ray, C.A.; Carter, J.R. Central modulation of exercise-induced muscle pain in humans. J. Physiol. 2007, 585, 287–294. [Google Scholar] [CrossRef]
- Liu, J.; Chen, L.; Chen, X.; Hu, K.; Tu, Y.; Lin, M.; Huang, J.; Liu, W.; Wu, J.; Qiu, Z.; et al. Modulatory effects of different exercise modalities on the functional connectivity of the periaqueductal grey and ventral tegmental area in patients with knee osteoarthritis: A randomised multimodal magnetic resonance imaging study. Br. J. Anaesth. 2019, 123, 506–518. [Google Scholar] [CrossRef] [PubMed]
- Malfliet, A.; Kregel, J.; Coppieters, I.; De Pauw, R.; Meeus, M.; Roussel, N.; Cagnie, B.; Danneels, L.; Nijs, J. Effect of Pain Neuroscience Education Combined with Cognition-Targeted Motor Control Training on Chronic Spinal Pain: A Randomized Clinical Trial. JAMA Neurol. 2018, 75, 808–817. [Google Scholar] [CrossRef] [PubMed]
- Martín-Borràs, C.; Giné-Garriga, M.; Puig-Ribera, A.; Martín, C.; Solà, M.; Cuesta-Vargas, A.I. A new model of exercise referral scheme in primary care: Is the effect on adherence to physical activity sustainable in the long term? A 15-month randomised controlled trial. BMJ Open 2018, 8, e017211. [Google Scholar] [CrossRef]
- Cuesta-Vargas, A.I.; García-Romero, J.C.; Arroyo-Morales, M.; Diego-Acosta, Á.M.; Daly, D.J. Exercise, Manual Therapy, and Education with or Without High-Intensity Deep-Water Running for Nonspecific Chronic Low Back Pain. Am. J. Phys. Med. Rehabil. 2011, 90, 526–538. [Google Scholar] [CrossRef] [PubMed]
- Martín-Valero, R.; Cuesta-Vargas, A.I.; Labajos-Manzanares, M.T. Effectiveness of the physical activity promotion programme on the quality of life and the cardiopulmonary function for inactive people: Randomized controlled trial. BMC Public Health 2013, 13, 127. [Google Scholar] [CrossRef]
- Cuesta-Vargas, A.I.; Adams, N. A pragmatic community-based intervention of multimodal physiotherapy plus deep water running (DWR) for fibromyalgia syndrome: A pilot study. Clin. Rheumatol. 2011, 30, 1455–1462. [Google Scholar] [CrossRef] [PubMed]
- Galán-Martín, M.A.; Montero-Cuadrado, F.; Lluch-Girbes, E.; Coca-López, M.C.; Mayo-Iscar, A.; Cuesta-Vargas, A. Pain neuroscience education and physical exercise for patients with chronic spinal pain in primary healthcare: A randomised trial protocol. BMC Musculoskelet. Disord. 2019, 20, 505. [Google Scholar] [CrossRef]
- Burke, S.M.; Carron, A.V.; Eys, M.A.; Ntoumanis, N.; Estabrooks, P.A. Group versus individual approach? A meta-analysis of the effectiveness of interventions to promote physical activity. Sport Exerc. Psychol. Rev. 2006, 2, 19–35. [Google Scholar]
- Tsekoura, M.; Billis, E.; Tsepis, E.; Dimitriadis, Z.; Matzaroglou, C.; Tyllianakis, M.; Panagiotopoulos, E.; Gliatis, J.; Tsekoura, M.; Billis, E.; et al. The Effects of Group and Home-Based Exercise Programs in Elderly with Sarcopenia: A Randomized Controlled Trial. J. Clin. Med. 2018, 7, 480. [Google Scholar] [CrossRef]
- Yorks, D.M.; Frothingham, C.A.; Schuenke, M.D. Effects of Group Fitness Classes on Stress and Quality of Life of Medical Students. J. Am. Osteopath. Assoc. 2017, 117, e17. [Google Scholar] [CrossRef]
- Smallwood, R.F.; Laird, A.R.; Ramage, A.E.; Parkinson, A.L.; Lewis, J.; Clauw, D.J.; Williams, D.A.; Schmidt-Wilcke, T.; Farrell, M.J.; Eickhoff, S.B.; et al. Structural Brain Anomalies and Chronic Pain: A Quantitative Meta-Analysis of Gray Matter Volume. J. Pain 2013, 14, 663–675. [Google Scholar] [CrossRef] [PubMed]
- Apkarian, A.V. Chronic Back Pain Is Associated with Decreased Prefrontal and Thalamic Gray Matter Density. J. Neurosci. 2004, 24, 10410–10415. [Google Scholar] [CrossRef] [PubMed]
- Wood, P.B. Variations in Brain Gray Matter Associated with Chronic Pain. Curr. Rheumatol. Rep. 2010, 12, 462–469. [Google Scholar] [CrossRef] [PubMed]
- Hurwitz, E.L.; Randhawa, K.; Yu, H.; Côté, P.; Haldeman, S. The Global Spine Care Initiative: A summary of the global burden of low back and neck pain studies. Eur. Spine J. 2018, 27, 796–801. [Google Scholar] [CrossRef]
- Caneiro, J.P.; Roos, E.M.; Barton, C.J.; O’Sullivan, K.; Kent, P.; Lin, I.; Choong, P.; Crossley, K.M.; Hartvigsen, J.; Smith, A.J.; et al. It is time to move beyond ‘body region silos’ to manage musculoskeletal pain: Five actions to change clinical practice. Br. J. Sports Med. 2019. [Google Scholar] [CrossRef]
- Chan, A.-W.; Tetzlaff, J.M.; Altman, D.G.; Laupacis, A.; Gøtzsche, P.C.; Krleža-Jerić, K.; Hróbjartsson, A.; Mann, H.; Dickersin, K.; Berlin, J.A.; et al. SPIRIT 2013 Statement: Defining Standard Protocol Items for Clinical Trials. Ann. Intern. Med. 2013, 158, 200–207. [Google Scholar] [CrossRef]
- Moher, D.; Hopewell, S.; Schulz, K.F.; Montori, V.; Gøtzsche, P.C.; Devereaux, P.J.; Elbourne, D.; Egger, M.; Altman, D.G. CONSORT 2010 Explanation and Elaboration: Updated guidelines for reporting parallel group randomised trials. J. Clin. Epidemiol. 2010, 63, e1–e37. [Google Scholar] [CrossRef]
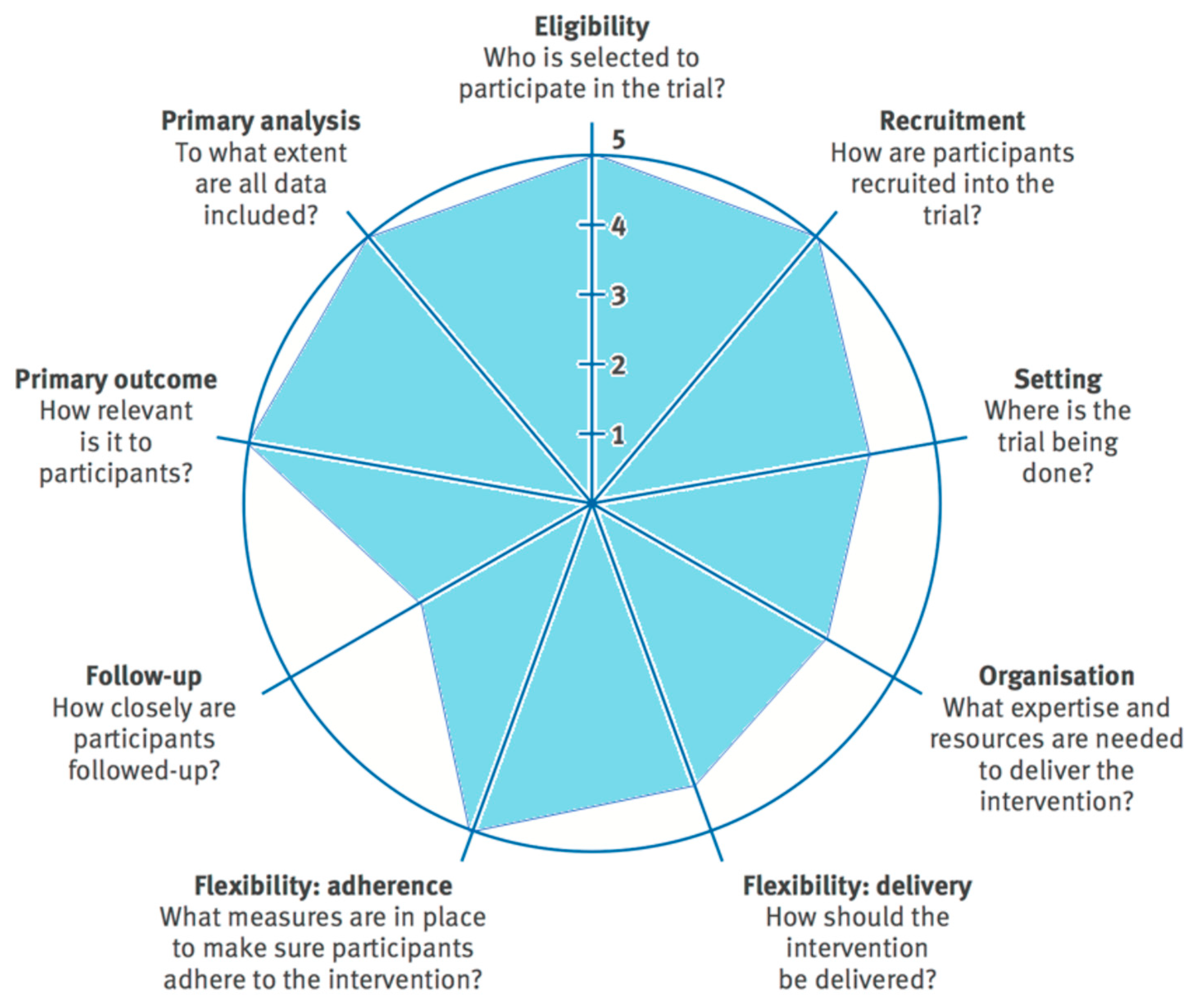
- Loudon, K.; Treweek, S.; Sullivan, F.; Donnan, P.; Thorpe, K.E.; Zwarenstein, M. The PRECIS-2 tool: Designing trials that are fit for purpose. BMJ 2015, 350, h2147. [Google Scholar] [CrossRef]
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. “Mini-mental state”: A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 1975, 12, 189–198. [Google Scholar] [CrossRef]
- Llamas Velasco, S.; Llorente Ayuso, L.; Contador, I.; Bermejo Pareja, F. Versiones en español del Minimental State Examination (MMSE). Cuestiones para su uso en la práctica clínica. Rev. Neurol. 2015, 61, 363–371. [Google Scholar] [CrossRef]
- Podsiadlo, D.; Richardson, S. The Timed “Up & Go”: A Test of Basic Functional Mobility for Frail Elderly Persons. J. Am. Geriatr. Soc. 1991, 39, 142–148. [Google Scholar] [PubMed]
- Cormier, S.; Lavigne, G.L.; Choinière, M.; Rainville, P. Expectations predict chronic pain treatment outcomes. Pain 2016, 157, 329–338. [Google Scholar] [CrossRef] [PubMed]
- Daenen, L.; Varkey, E.; Kellmann, M.; Nijs, J. Exercise, not to exercise, or how to exercise in patients with chronic pain? Applying science to practice. Clin. J. Pain 2015, 31, 108–114. [Google Scholar] [CrossRef] [PubMed]
- Nijs, J.; Kosek, E.; Van Oosterwijck, J.; Meeus, M. Dysfunctional endogenous analgesia during exercise in patients with chronic pain: To exercise or not to exercise? Pain Physician 2012, 15, 205–214. [Google Scholar]
- Smith, B.E.; Hendrick, P.; Smith, T.O.; Bateman, M.; Moffatt, F.; Rathleff, M.S.; Selfe, J.; Logan, P. Should exercises be painful in the management of chronic musculoskeletal pain? A systematic review and meta-analysis. Br. J. Sports Med. 2017, 51, 1679–1687. [Google Scholar] [CrossRef]
- Smith, B.E.; Hendrick, P.; Bateman, M.; Holden, S.; Littlewood, C.; Smith, T.O.; Logan, P. Musculoskeletal pain and exercise—Challenging existing paradigms and introducing new. Br. J. Sports Med. 2019, 53, 907–912. [Google Scholar] [CrossRef]
- Dirección General de Asistencia Sanitaria. Protocolos de Fisioterapia en Atención Primaria; Gerencia Regional de Salud: Valladolid, Spain, 2005. [Google Scholar]
- SERMEF (Sociedad Española de Medicina Física y Rehabilitación) WEB EJERCICIOS SERMEF—SERMEF. Available online: https://www.sermef.es/web-ejercicios-sermef/ (accessed on 6 April 2019).
- Vilagut, G.; Valderas, J.M.; Ferrer, M.; Garin, O.; López-García, E.; Alonso, J. Interpretación de los cuestionarios de salud SF-36 y SF-12 en España: Componentes físico y mental. Med. Clin. (Barc.) 2008, 130, 726–735. [Google Scholar] [CrossRef]
- Alonso, J.; Regidor, E.; Barrio, G.; Prieto, L.; Rodríguez, C.; De la Fuente, L. Valores poblacionales de referencia de la versión española del Cuestionario de Salud SF-36. Med. Clin. (Barc.) 1998, 111, 410–416. [Google Scholar]
- Vilagut, G.; Ferrer, M.; Rajmil, L.; Rebollo, P.; Permanyer-Miralda, G.; Quintana, J.M.; Santed, R.; Valderas, J.M.; Ribera, A.; Domingo-Salvany, A.; et al. El Cuestionario de Salud SF-36 español: Una década de experiencia y nuevos desarrollos por los investigadores de la Red-IRYSS*. Gac. Sanit. 2005, 19, 135–150. [Google Scholar] [CrossRef]
- García Campayo, J.; Rodero, B.; Alda, M.; Sobradiel, N.; Montero, J.; Moreno, S. Validación de la versión española de la escala de la catastrofización ante el dolor (Pain Catastrophizing Scale) en la fibromialgia. Med. Clin. 2008, 131, 487–492. [Google Scholar] [CrossRef]
- Sullivan, M.J.L.; Bishop, S.R.; Pivik, J. The Pain Catastrophizing Scale: Development and validation. Psychol. Assess. 1995, 7, 524–532. [Google Scholar] [CrossRef]
- Gómez-Pérez, L.; López-Martínez, A.E.; Ruiz-Párraga, G.T. Psychometric properties of the spanish version of the Tampa Scale for Kinesiophobia (TSK). J. Pain 2011, 12, 425–435. [Google Scholar] [CrossRef] [PubMed]
- Cuesta-Vargas, A.I.; Roldan-Jimenez, C.; Neblett, R.; Gatchel, R.J. Cross-cultural adaptation and validity of the Spanish central sensitization inventory. Springerplus 2016, 5, 1837. [Google Scholar] [CrossRef] [PubMed]
- Scerbo, T.; Colasurdo, J.; Dunn, S.; Unger, J.; Nijs, J.; Cook, C. Measurement Properties of the Central Sensitization Inventory: A Systematic Review. Pain Pract. 2018, 18, 544–554. [Google Scholar] [CrossRef]
- Neblett, R.; Hartzell, M.M.; Williams, M.; Bevers, K.R.; Mayer, T.G.; Gatchel, R.J. Use of the Central Sensitization Inventory (CSI) as a treatment outcome measure for patients with chronic spinal pain disorder in a functional restoration program. Spine J. 2017, 17, 1819–1829. [Google Scholar] [CrossRef]
- Neblett, R.; Hartzell, M.M.; Mayer, T.G.; Cohen, H.; Gatchel, R.J. Establishing Clinically Relevant Severity Levels for the Central Sensitization Inventory. Pain Pract. 2017, 17, 166–175. [Google Scholar] [CrossRef]
- Kovacs, F.M.; Llobera, J.; del Real, M.T.G.; Abraira, V.; Gestoso, M.; Fernández, C. Validation of the Spanish version of the Roland-Morris questionnaire. Spine 2002, 27, 538–542. [Google Scholar] [CrossRef]
- Melzack, R. The short-form McGill pain questionnaire. Pain 1987, 30, 191–197. [Google Scholar] [CrossRef]
- Chapman, C.R.; Casey, K.L.; Dubner, R.; Foley, K.M.; Gracely, R.H.; Reading, A.E. Pain measurement: An overview. Pain 1985, 22, 1–31. [Google Scholar] [CrossRef]
- Neziri, A.Y.; Scaramozzino, P.; Andersen, O.K.; Dickenson, A.H.; Arendt-Nielsen, L.; Curatolo, M. Reference values of mechanical and thermal pain tests in a pain-free population. Eur. J. Pain 2011, 15, 376–383. [Google Scholar] [CrossRef]
- Balaguier, R.; Madeleine, P.; Vuillerme, N. Is one trial sufficient to obtain excellent pressure pain threshold reliability in the low back of asymptomatic individuals? A test-retest study. PLoS ONE 2016, 11, e0160866. [Google Scholar] [CrossRef] [PubMed]
- Roberts, R.E.; Atrkisson, C.C.; Mendias, R.M. Assessing the Client Satisfaction Questionnaire in English and Spanish. Hisp. J. Behav. Sci. 1984, 6, 385–396. [Google Scholar] [CrossRef]
- Bodes Pardo, G.; Lluch Girbés, E.; Roussel, N.A.; Gallego Izquierdo, T.; Jiménez Penick, V.; Pecos Martín, D. Pain Neurophysiology Education and Therapeutic Exercise for Patients with Chronic Low Back Pain: A Single-Blind Randomized Controlled Trial. Arch. Phys. Med. Rehabil. 2018, 99, 338–347. [Google Scholar] [CrossRef] [PubMed]
- Moseley, L. Combined physiotherapy and education is efficacious for chronic low back pain. Aust. J. Physiother. 2002, 48, 297–302. [Google Scholar] [CrossRef]
- Nijs, J.; Polli, A.; Willaert, W.; Malfliet, A.; Huysmans, E.; Coppieters, I. Central sensitisation: Another label or useful diagnosis? DTB 2019, 57. [Google Scholar] [CrossRef] [PubMed]
- Malfliet, A.; Ickmans, K.; Huysmans, E.; Coppieters, I.; Willaert, W.; Van Bogaert, W.; Rheel, E.; Bilterys, T.; Van Wilgen, P.; Nijs, J.; et al. Best Evidence Rehabilitation for Chronic Pain Part 3: Low Back Pain. J. Clin. Med. 2019, 8, 1063. [Google Scholar] [CrossRef]
- Rice, D.; Nijs, J.; Kosek, E.; Wideman, T.; Hasenbring, M.I.; Koltyn, K.; Graven-Nielsen, T.; Polli, A. Exercise-Induced Hypoalgesia in Pain-Free and Chronic Pain Populations: State of the Art and Future Directions. J. Pain 2019, 20, 1249–1266. [Google Scholar] [CrossRef]
- Dysvik, E.; Furnes, B. Living a meaningful life with chronic pain—Further follow-up. Clin. Case Rep. 2018, 6, 896–900. [Google Scholar] [CrossRef]
- O’Leary, H.; Smart, K.M.; Moloney, N.A.; Doody, C.M. Nervous System Sensitization as a Predictor of Outcome in the Treatment of Peripheral Musculoskeletal Conditions: A Systematic Review. Pain Pract. 2017, 17, 249–266. [Google Scholar] [CrossRef]
- Main, C.J.; Foster, N. How important are back pain beliefs and expectations for satisfactory recovery from back pain? Best Pract. Res. Clin. Rheumatol. 2010, 24, 205–217. [Google Scholar] [CrossRef]
- Galán-Martín, M.; Montero-Cuadrado, F. Educación en Neurociencia del Dolor. Una Nueva Visión. Dossier para Pacientes, 1st ed.; Junta de Castilla y León: Valladolid, Spain, 2018; ISBN 978-84-9718-690-2. [Google Scholar]
- Butler, D.S.; David, S.; Moseley, G.L. Explain Pain, 2nd ed.; NOI Group Publications: Adelaide, Australia, 2013; ISBN 978-0-9873426-6-9. [Google Scholar]
- Goicoechea, A.; Echaávarri, R. MIGRAÑA: Una Pesadilla Cerebral, 8th ed.; Descleée de Brouwer: Bilbao, Spain, 2009; ISBN 9788433023605. [Google Scholar]
- McIsaac, T.L.; Lamberg, E.M.; Muratori, L.M. Building a framework for a dual task taxonomy. BioMed Res. Int. 2015, 2015, 591475. [Google Scholar] [CrossRef] [PubMed]
- Studer, M. Making balance automatic again: Using dual tasking as an intervention in balance rehabilitation for older adults. SM Gerontol. Geriatr. Res. 2018, 2, 1015. [Google Scholar] [CrossRef]
- Montero-Cuadrado, F.; Sánchez-Sánchez, J.; Calvo-Arenillas, J.; Sánchez-Domínguez, F.; Galán-Martín, M.; Coca-López, M. Ejercicio físico como estrategia terapéutica para la ansiedad y la depresión de cuidadoreas familiares de personas dependientes. Actas Coord. Socio Sanit. 2018, 22, 109–132. [Google Scholar]
- Lewis, J.S.; Cook, C.E.; Hoffmann, T.C.; O’Sullivan, P. The Elephant in the Room: Too Much Medicine in Musculoskeletal Practice. J. Orthop. Sports Phys. Ther. 2020, 50, 1–4. [Google Scholar] [CrossRef] [PubMed]

| EG (n = 89) | CG (n = 81) | |
|---|---|---|
| Age (mean ± SD) | 53.02 ± 10.7 | 49.14 ± 12.14 |
| Female (n) | 65 | 71 |
| Marital Status (%) | ||
| Married/in couple | 62 | 57 |
| Divorced | 5 | 5 |
| Single | 14 | 15 |
| Sidow | 8 | 4 |
| Education Level (%) | ||
| Incomplete Primary educat. | 3.7 | 6.2 |
| Primary education | 33.7 | 35.8 |
| Lower secondary education | 16.9 | 17.3 |
| Upper secondary education | 12.4 | 7.4 |
| Post-secondary non-tertiary | 14.6 | 17.3 |
| University degree | 19.1 | 16.0 |
| Employment Situation | ||
| Student | 0.0 | 2.5 |
| Active | 40.4 | 51.9 |
| Unemployed | 22.5 | 12.3 |
| Home chores | 15.7 | 19.8 |
| Pensioner | 21.3 | 13.6 |
| Anthropometrics | ||
| BMI (mean ± SD) | 27.6 ± 4.7 | 26.5 ± 5.0 |
| Pain | ||
| VAS (mm) (mean±SD) | 70.8 ± 14.8 | 67.2 ± 14.3 |
| Months with pain (mean±SD) | 93.13 ± 83.5 | 93.86 ± 84.91 |
| Mouths with pain (median) | 48 | 48 |
| CSP (%) | 100 | 100 |
| Cervical pain | 80.9 | 79.3 |
| Thoracic pain | 65.2 | 50.0 |
| Low back pain | 91.0 | 89.0 |
| Pain in Other Areas (%) | ||
| RUL | 42.7 | 35.4 |
| LUL | 40.4 | 31.7 |
| RLL | 37.1 | 34.1 |
| LLL | 25.8 | 23.2 |
| Group EG (n = 89) CG (n = 81) | Pre | Post | 6 mo | Intra. Dif (6 mo – Pre) 95% IC | Effect Size | Dif. Inter 6 mo (EG – CG) 95% IC | |
|---|---|---|---|---|---|---|---|
| HRQL (SF-36) | |||||||
| Physical Function (PF) | EG | 60 ± 18.2 | 80.2 ± 13.4 | 81.4 ± 13.8 | 22 (18.9, 25.2) ** | 1.2 | 18.7 (14.1, 23.4) † |
| CG | 63.1 ± 23.3 | 65.8 ± 22.3 | 65.8 ± 20.9 | 3.3 (−0.1, 6.7) | 0.1 | ||
| Role Physical (RP) | EG | 47.5 ± 26.3 | 77.8 ± 20.7 | 78.2 ± 20.4 | 32.3 (26.4, 38.2) ** | 1.2 | 30.7 (21.6, 39.8) † |
| CG | 56.2 ± 25.7 | 58.1 ± 25.4 | 59.1 ±24.7 | 1.6 (−5.2, 8.5) | 0.1 | ||
| Bodily Pain (BP) | EG | 32.7 ± 17.1 | 75.8 ± 16.5 | 73.1 ± 14.9 | 41 (36.9, 45) ** | 2.4 | 34.9 (28.3, 41.3) † |
| CG | 36.7 ± 19 | 44.2 ± 20.8 | 42.8 ± 23.2 | 6.1 (1, 11.2) * | 0.3 | ||
| Social Functioning (SF) | EG | 60.5 ± 24.1 | 88.1 ± 15.4 | 84.3 ± 18.5 | 24.1 (18.9, 29.3) ** | 1 | 22.7 (14.3, 30.9) † |
| CG | 68.7 ± 24.6 | 71.6 ± 25.3 | 70.7 ± 24.7 | 1.4 (−5.1, 8) | 0.1 | ||
| Mental Heath (MH) | EG | 61.4 ± 18.6 | 77.6 ± 15.7 | 74.1 ± 17.2 | 12.8 (9.3, 16.3) ** | 0.7 | 12.4 (7, 18) † |
| CG | 63.3 ± 19.5 | 63.4 ± 17.3 | 64.4 ± 19 | 0.4 (−3.9, 4.6) | 0 | ||
| Role Emotion (RE) | EG | 74.2 ± 24.7 | 89.2 ± 15.4 | 90.3 ± 15.1 | 16.9 (11.7, 22) ** | 0.7 | 16.7 (9.3, 23.9) † |
| CG | 80.2 ± 21.8 | 80.6 ± 21.4 | 82.6 ± 20.1 | 0.2 (−4.8, 5.3) | 0 | ||
| Vitality (VT) | EG | 40 ± 18.4 | 65.1 ± 17.8 | 59.8 ± 18.5 | 20.3 (16.3, 24.3) ** | 1.1 | 17.4 (10.7, 24.1) † |
| CG | 42.8 ± 20.1 | 44.1 ± 18.4 | 46.8 ± 19.6 | 2.9 (−2.5, 8.3) | 0.1 | ||
| General Health (GH) | EG | 42.4 ± 16.7 | 66.2 ± 16.8 | 65.4 ± 17.7 | 24 (20.4, 27.6) ** | 1.4 | 23.5 (18.1, 28.9) † |
| CG | 48.9 ± 19.9 | 50.7 ± 18.2 | 48.6 ± 20.4 | 0.5 (−3.5, 4.5) | 0 | ||
| Health Transition | EG | 30.1 ± 18.1 | 76.2 ± 15 | 75.9 ± 14.5 | 45.4 (40.5, 50.2) ** | 2.5 | 37.1 (29, 45.1) † |
| CG | 36.1 ± 19.4 | 41.4 ± 22.1 | 43.8 ± 23.6 | 8.3 (1.9, 14.8) | 0.4 | ||
| Physical component summary (PCS) | EG | 34.9 ± 7.3 | 47.4 ± 6.9 | 47.4 ± 6.7 | 12.9 (11.4, 14.3) ** | 1.8 | 11.4 (9.1, 13.6) † |
| CG | 37.1 ± 9.3 | 38.9 ± 9 | 38.3 ± 8.5 | 1.5 (−0.2, 3.3) | 0.2 | ||
| Mental component summary (MCS) | EG | 44.9 ± 10.1 | 51.7 ± 7.7 | 50 ± 7.9 | 5.1 (3.2, 7.1) ** | 0.5 | 5.2 (2.2, 8.1) † |
| CG | 46.6 ± 10.2 | 46.5 ± 8.8 | 47.3 ± 9.3 | −0.1 (−2.3, 2.1) | 0 | ||
| Catasthrophism (PCS) | EG | 30.3 ± 8.7 | 15.4 ± 7.3 | 15.5 ± 7.2 | −15 (−16.7, −13.4) ** | −1.7 | −11 (−13.6, −8.4) † |
| CG | 27.9 ± 9.1 | 26.6 ± 9.7 | 24.2 ± 10.3 | −4 (−6, −2.1) ** | −0.4 | ||
| Rumination | EG | 10.5 ± 2.8 | 5.6 ± 2.6 | 5.5 ± 2.6 | −5 (−5.6, −4.5) ** | −1.8 | −3.6 (−4.5, −2.7) † |
| CG | 9.6 ± 3.1 | 9.2 ± 3.2 | 8.2 ± 3.6 | −1.4 (−2.1, −0.7) ** | −0.5 | ||
| Magnification | EG | 6.3 ± 2.3 | 3.1 ± 2 | 3.2 ± 2 | −3.2 (−3.7, −2.7) ** | −1.4 | −2.5 (−3.3, −1.6) † |
| CG | 5.8 ± 2.7 | 5.5 ± 2.8 | 5.3 ± 2.6 | −0.7 (−1.4, −0.1) * | −0.3 | ||
| Helplessness | EG | 13.5 ± 4.6 | 6.7 ± 3.7 | 6.7 ± 3.8 | −6.8 (−7.8, −5.9) ** | −1.5 | −4.9 (−6.4, −3.6) † |
| CG | 12.5 ± 4.6 | 11.9 ± 4.8 | 10.7 ± 5 | −1.9 (−2.8, −0.9) ** | −0.4 | ||
| Kinesiophobia (TSK-11) | EG | 28.9 ± 6.6 | 17.1 ± 4 | 17.2 ± 4.7 | −12.2 (−13.5, −10.9) ** | −1.8 | −10.6 (−12.4, −8.7) † |
| CG | 27.5 ± 7.1 | 26.1 ± 6.3 | 26.3 ± 7.6 | −1.6 (−3, −0.3) * | −0.2 | ||
| Central Sensitization (CSI) | EG | 43.4 ± 12.5 | 25.7 ± 10.8 | 25.8 ± 10.5 | −17.7 (−19.3, −16) ** | −1.4 | −16.6 (−19, −14.1) † |
| CG | 38.6 ± 11.7 | 37.7±12.4 | 37.4 ± 13.5 | −1.1 (−2.9, 0.6) | −0.1 | ||
| Disability (RMDC) | EG | 9.2 ± 4.8 | 3.3 ± 3.5 | 3.3 ± 3.8 | −6 (−6.8, −5.2) ** | −1.2 | −5.6 (−6.7, −4.5) † |
| CG | 8 ± 4.7 | 7.6 ± 4.6 | 7.7 ± 4.8 | −0.4 (−1.1, 0.2) | −0.1 | ||
| Pain intensity (VAS) | EG | 74.1 ± 14.5 | 26.7 ± 18 | 27 ± 16.2 | −48.2 (−52.6, −43.8) ** | −3.3 | −40.9 (−46.7, −35.2) † |
| CG | 67.2 ± 14.3 | 58.4 ± 17.7 | 59.7 ± 19.8 | −7.3 (−10.6, −3.9) ** | −0.5 | ||
| Algometry (PPT). P1 | EG | 2.4 ± 1 | 3.8 ± 1.5 | 4.2 ± 1.7 | 1.8 (1.5, 2.2) ** | 1.8 | 1.8 (1.4, 2.2) † |
| CG | 2.5 ± 1 | 2.7 ± 1.1 | 2.5 ± 0.9 | 0 (−0.2, 0.3) | 0 | ||
| Algometry (PPT). P2 | EG | 2.3 ± 0.9 | 3.8 ± 1.5 | 4.1 ± 1.7 | 1.9 (1.6, 2.2) ** | 2.1 | 1.7 (1.3, 2.2) † |
| CG | 2.5 ± 1 | 2.6 ± 1.1 | 2.6 ± 1.1 | 0.2 (−0.1, 0.5) | 0.2 | ||
| Algometry (PPT). P3 | EG | 2.5 ± 1.2 | 4.3 ± 1.6 | 4.9 ± 2 | 2.4 (2, 2.8) ** | 2 | 2.2 (1.7, 2.8) † |
| CG | 2.8 ± 1.2 | 3.1 ± 1.5 | 3 ± 1.4 | 0.2 (−0.1, 0.5) | 0.2 | ||
| Algometry (PPT). P4 | EG | 2.6 ± 1.2 | 4.5 ± 1.8 | 5 ± 2.1 | 2.5 (2, 2.9) ** | 2.1 | 2.3 (1.7, 2.9) † |
| CG | 2.8 ± 1.3 | 3.1 ± 1.5 | 3 ± 1.4 | 0.2(-0.1,0.5) | 0.2 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Galan-Martin, M.A.; Montero-Cuadrado, F.; Lluch-Girbes, E.; Coca-López, M.C.; Mayo-Iscar, A.; Cuesta-Vargas, A. Pain Neuroscience Education and Physical Therapeutic Exercise for Patients with Chronic Spinal Pain in Spanish Physiotherapy Primary Care: A Pragmatic Randomized Controlled Trial. J. Clin. Med. 2020, 9, 1201. https://doi.org/10.3390/jcm9041201
Galan-Martin MA, Montero-Cuadrado F, Lluch-Girbes E, Coca-López MC, Mayo-Iscar A, Cuesta-Vargas A. Pain Neuroscience Education and Physical Therapeutic Exercise for Patients with Chronic Spinal Pain in Spanish Physiotherapy Primary Care: A Pragmatic Randomized Controlled Trial. Journal of Clinical Medicine. 2020; 9(4):1201. https://doi.org/10.3390/jcm9041201
Chicago/Turabian StyleGalan-Martin, Miguel Angel, Federico Montero-Cuadrado, Enrique Lluch-Girbes, María Carmen Coca-López, Agustín Mayo-Iscar, and Antonio Cuesta-Vargas. 2020. "Pain Neuroscience Education and Physical Therapeutic Exercise for Patients with Chronic Spinal Pain in Spanish Physiotherapy Primary Care: A Pragmatic Randomized Controlled Trial" Journal of Clinical Medicine 9, no. 4: 1201. https://doi.org/10.3390/jcm9041201
APA StyleGalan-Martin, M. A., Montero-Cuadrado, F., Lluch-Girbes, E., Coca-López, M. C., Mayo-Iscar, A., & Cuesta-Vargas, A. (2020). Pain Neuroscience Education and Physical Therapeutic Exercise for Patients with Chronic Spinal Pain in Spanish Physiotherapy Primary Care: A Pragmatic Randomized Controlled Trial. Journal of Clinical Medicine, 9(4), 1201. https://doi.org/10.3390/jcm9041201






