Role of Vacha (Acorus calamus Linn.) in Neurological and Metabolic Disorders: Evidence from Ethnopharmacology, Phytochemistry, Pharmacology and Clinical Study
Abstract
1. Introduction
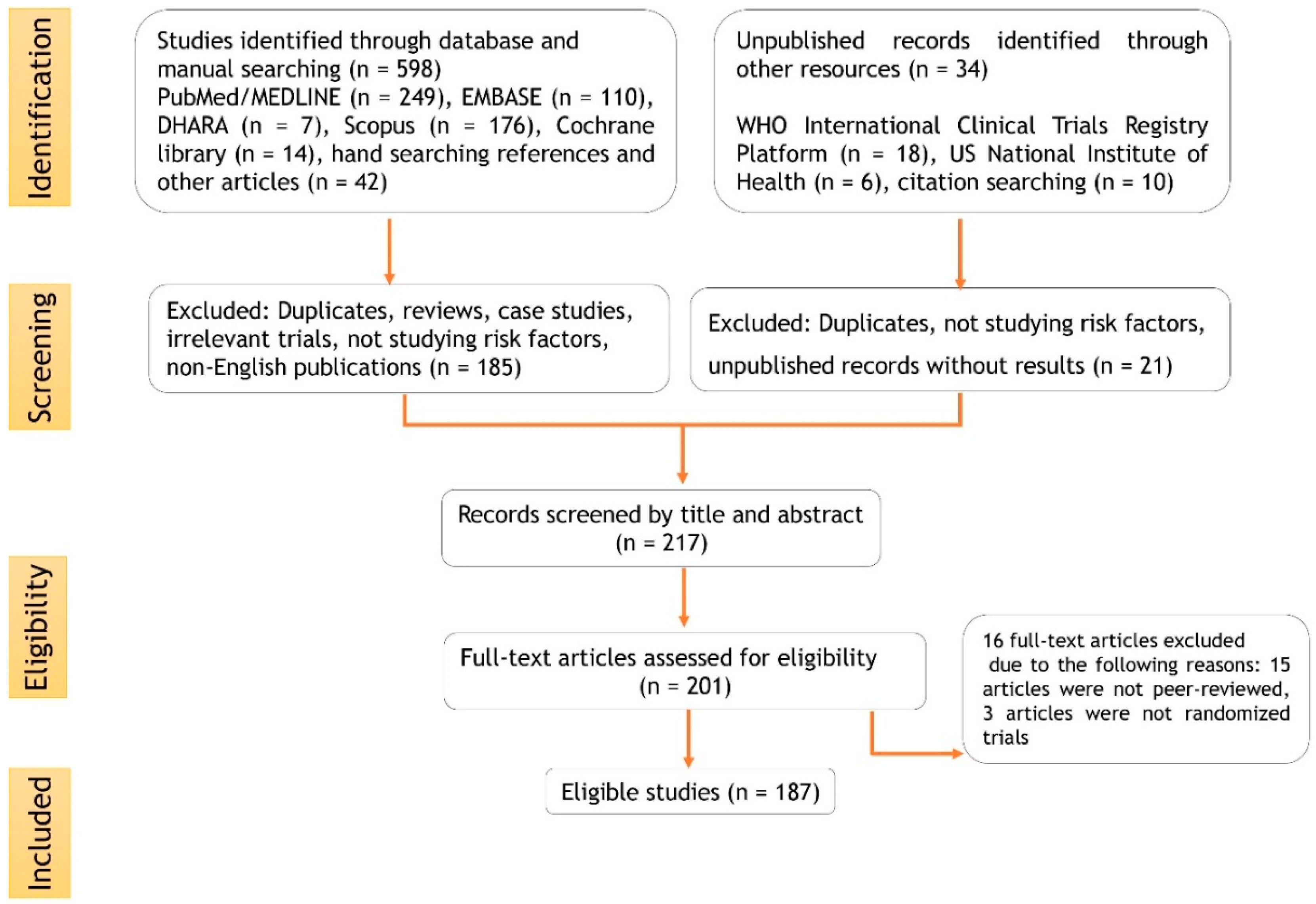
2. Methodology
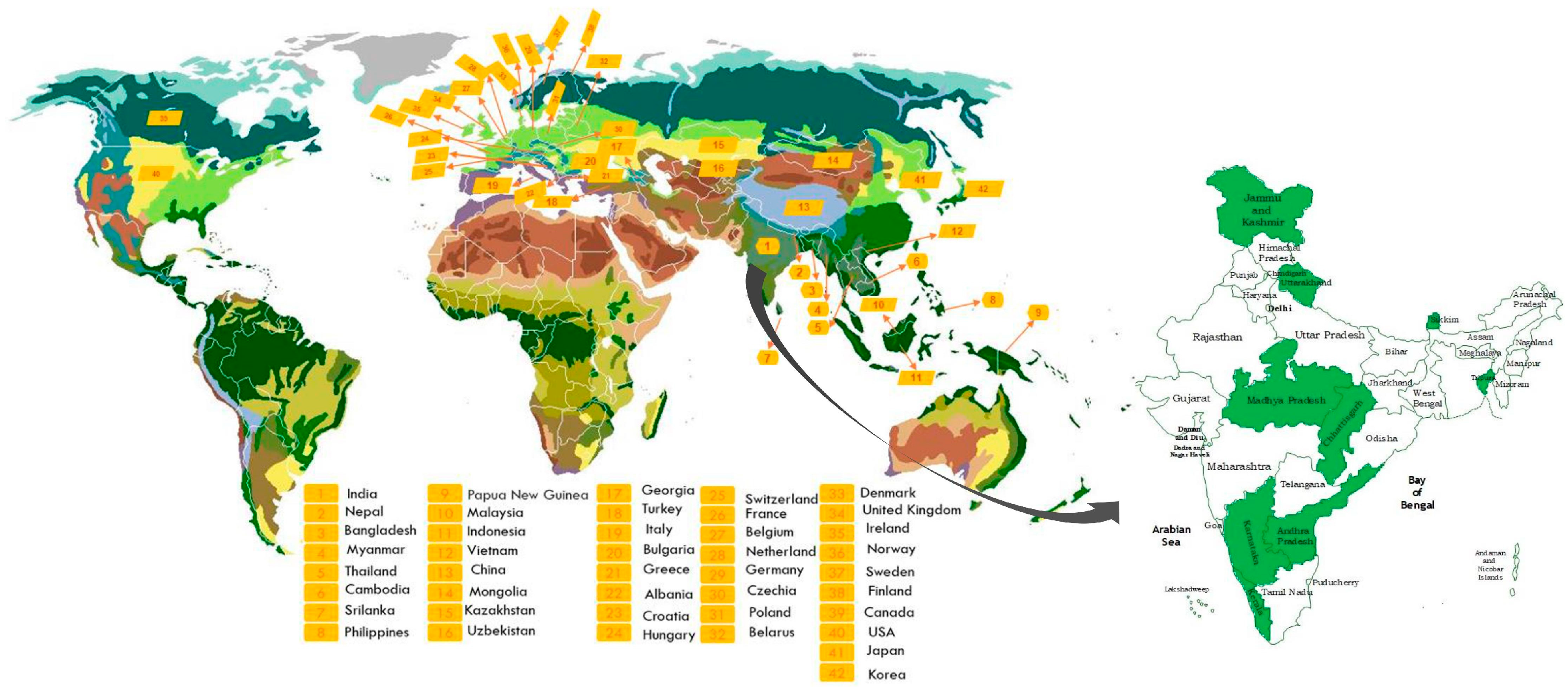
3. Geographical Distribution
4. Ethnomedicinal Use
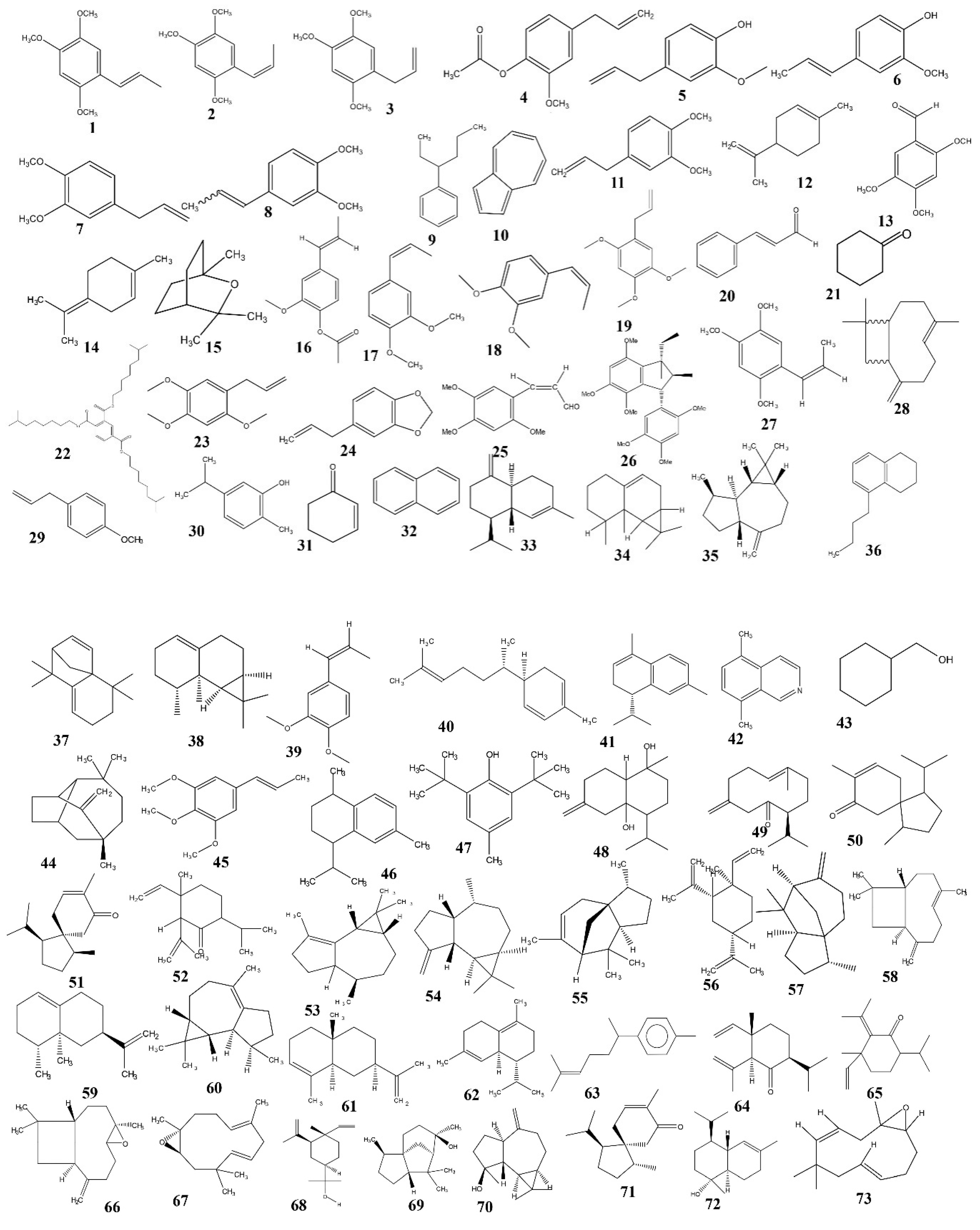
5. Phytochemistry
5.1. Phenylpropanoids
5.2. Sesquiterpenoids
5.3. Monoterpenes
5.4. Triterpenoid Saponins
5.5. Other Compounds
6. Pharmacological Properties
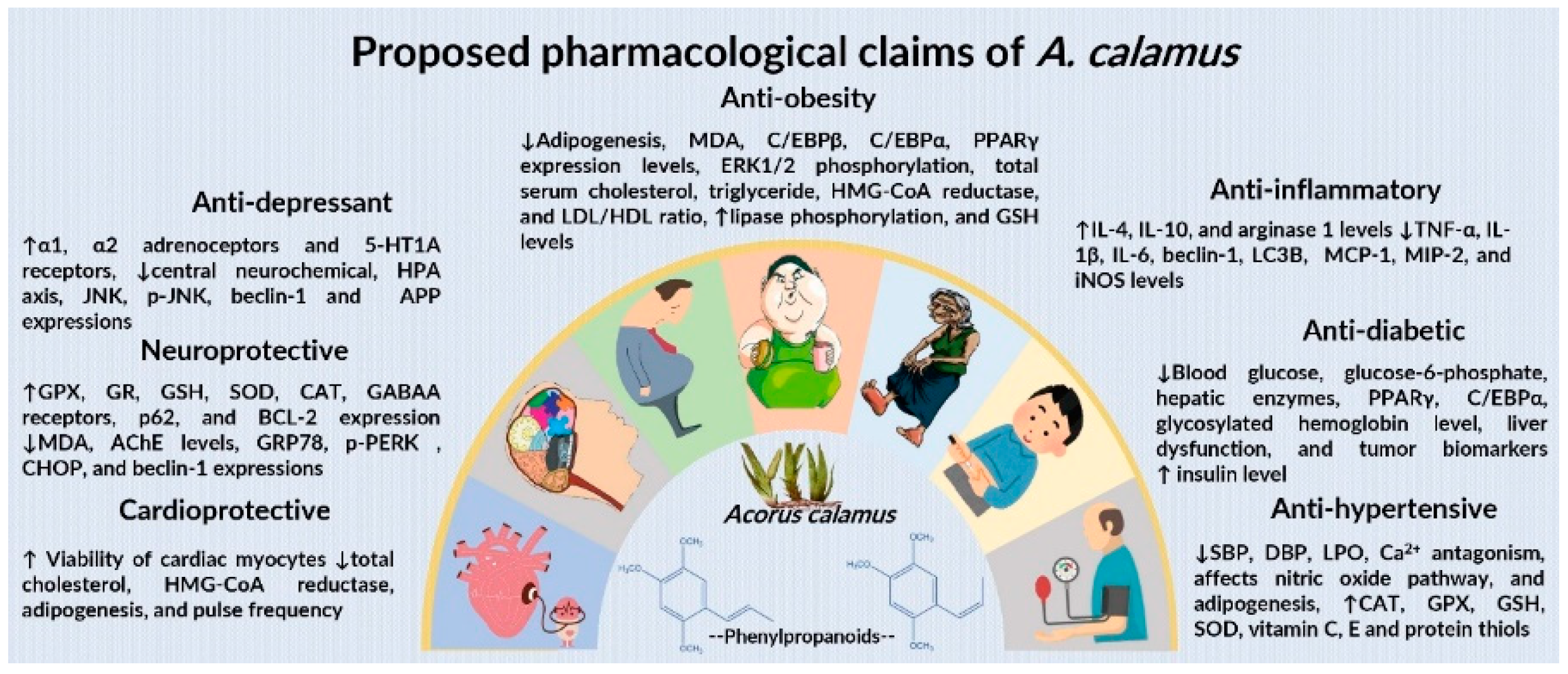
6.1. Antidiabetic Effect
6.2. Anti-Obesity Effect
6.3. Antihypertensive Effect
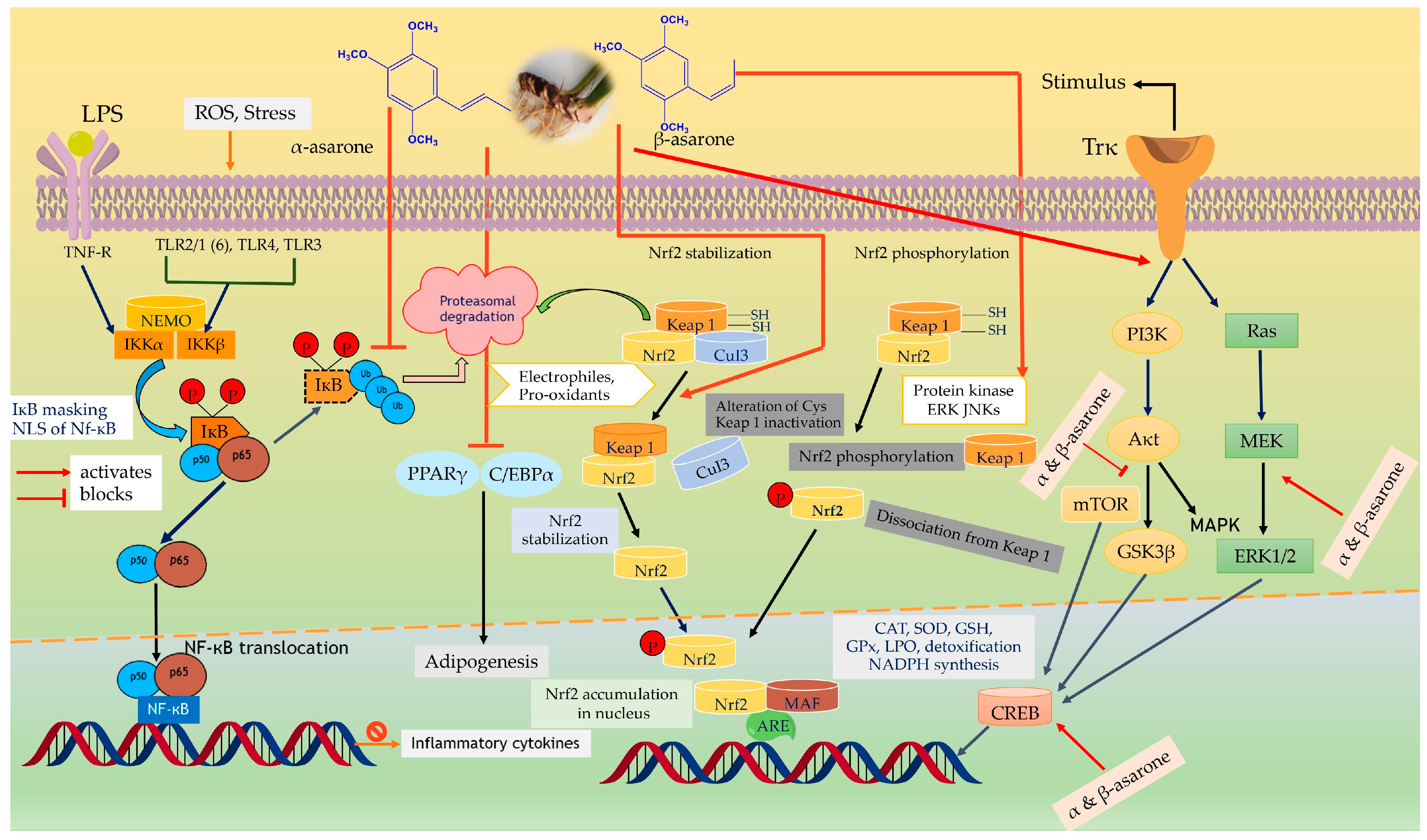
6.4. Anti-Inflammatory and Immunomodulatory Effect
6.5. Antioxidant Effect
6.6. Anticonvulsant Effect
6.7. Antidepressant Effect
6.8. Neuroprotective Effect
6.9. Cardioprotective Effect
6.10. Cytochrome Inhibitory Activities
6.11. Toxicity and Safety Concerns
7. Clinical Reports
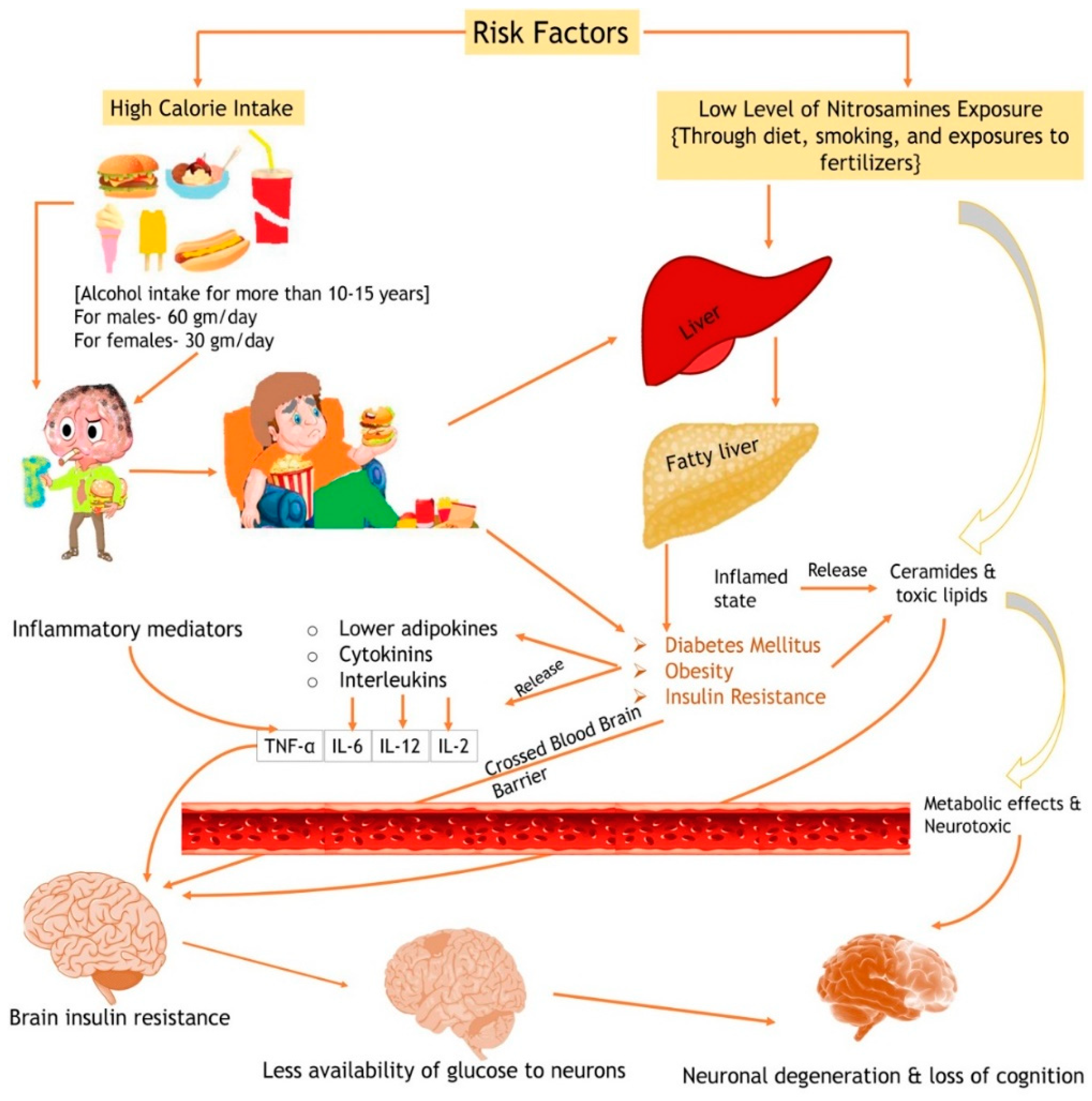
8. Mechanistic Role
9. Perspectives and Future Directions
10. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Report. Available online: https://www.who.int/whr/2001/media_centre/press_release/en/ (accessed on 4 October 2019).
- Toniolo, A.; Cassani, G.; Puggioni, A.; Rossi, A.; Colombo, A.; Onodera, T.; Ferrannini, E. The diabetes pandemic and associated infections: Suggestions for clinical microbiology. Rev. Med. Microbiol. 2019, 30, 1–17. [Google Scholar] [CrossRef]
- Younossi, Z.M. Non-alcoholic fatty liver disease-A global public health perspective. J. Hepatol. 2019, 70, 531–544. [Google Scholar] [CrossRef]
- Després, J.P. Is visceral obesity the cause of the metabolic syndrome. Ann. Med. 2006, 38, 52–63. [Google Scholar] [CrossRef]
- Farooqui, A.A.; Farooqui, T.; Panza, F.; Frisardi, V. Metabolic syndrome as a risk factor for neurological disorders. Cell. Mol. Life Sci. 2012, 69, 741–762. [Google Scholar] [CrossRef]
- Tilg, H.; Hotamisligil, G.S. Nonalcoholic fatty liver disease: Cytokine-adipokine interplay and regulation of insulin resistance. Gastroenterology 2006, 131, 934–945. [Google Scholar] [CrossRef] [PubMed]
- Suzanne, M.; Tong, M. Brain metabolic dysfunction at the core of Alzheimer’s disease. Biochem. Pharmacol. 2014, 88, 548–559. [Google Scholar]
- Quraishi, A.; Mehar, S.; Sahu, D.; Jadhav, S.K. In vitro mid-term conservation of Acorus calamus L. via cold storage of encapsulated microrhizome. Braz. Arch. Biol. Technol. 2017, 60, 1–9. [Google Scholar] [CrossRef]
- Balakumbahan, R.; Rajamani, K.; Kumanan, K. Acorus calamus: An overview. J. Med. Plant Res. 2010, 4, 2740–2745. [Google Scholar]
- Sharma, V.; Singh, I.; Chaudhary, P. Acorus calamus (The Healing Plant): A review on its medicinal potential, micropropagation and conservation. Nat. Prod. Res. 2014, 28, 1454–1466. [Google Scholar] [CrossRef]
- Singh, R.; Sharma, P.K.; Malviya, R. Pharmacological properties and ayurvedic value of Indian buch plant (Acorus calamus): A short review. Adv. Biol. Res. 2011, 5, 145–154. [Google Scholar]
- Global Biodiversity Information Facility. Available online: https://www.gbif.org/ (accessed on 10 February 2020).
- Kingston, C.; Jeeva, S.; Jeeva, G.M.; Kiruba, S.; Mishra, B.P.; Kannan, D. Indigenous knowledge of using medicinal plants in treating skin diseases in Kanyakumari district, Southern India. Indian J. Tradit. Knowl. 2009, 8, 196–200. [Google Scholar]
- Pradhan, B.K.; Badola, H.K. Ethnomedicinal plant use by Lepcha tribe of Dzongu valley, bordering Khangchendzonga Biosphere Reserve, in north Sikkim India. J. Ethnobiol. Ethnomed. 2008, 4, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.K.; Chauhan, N.S.; Lal, B. Observations on the traditional phytotherapy among the inhabitants of Parvati valley in western Himalaya, India. J. Ethnopharmacol. 2004, 92, 167–176. [Google Scholar] [CrossRef] [PubMed]
- Dwivedi, S.N.; Dwivedi, S.; Patel, P.C. Medicinal plants used by the tribal and rural people of Satna district, Madhya Pradesh for the treatment of gastrointestinal diseases and disorders. Nat. Prod. Rad. 2006, 5, 60–63. [Google Scholar]
- Usher, G. Spilanthes Acmella, a Dictionary of Plants Used by Man; CBS Publishers and Distributers: New Delhi, India, 1984; p. 38. [Google Scholar]
- Ghosh, A. Ethnomedicinal plants used in West Rarrh region of West Bengal. Nat. Prod. Rad. 2008, 7, 461–465. [Google Scholar]
- Natarajan, B.; Paulsen, B.S.; Korneliussen, V. An ethnopharmacological study from Kulu District, Himachal Pradesh, India: Traditional knowledge compared with modern biological science. Pharm. Biol. 2000, 38, 129–138. [Google Scholar] [CrossRef]
- Nisha, M.C.; Rajeshkumar, S. Survey of crude drugs from Coimbatore city. Indian J. Nat. Prod. Resour. 2010, 1, 376–383. [Google Scholar]
- Ragupathy, S.; Steven, N.G.; Maruthakkutti, M.; Velusamy, B.; Ul-Huda, M.M. Consensus of the ‘Malasars’ traditional aboriginal knowledge of medicinal plants in the Velliangiri holy hills, India. J. Ethnobiol. Ethnomed. 2008, 4, 8–16. [Google Scholar] [CrossRef]
- Tomar, A. Folk medicinal uses of plant roots from Meerut district, Uttar Pradesh. Indian J. Tradit. Knowl. 2009, 8, 298–301. [Google Scholar]
- Rajith, N.P.; Ramachandran, V.S. Ethnomedicines of Kurichyas, Kannur district, Western Ghats, Kerala. Indian J. Nat. Prod. Resour. 2010, 1, 249–253. [Google Scholar]
- Barbhuiya, A.R.; Sharma, G.D.; Arunachalam, A.; Deb, S. Diversity and conservation of medicinal plants in Barak valley, Northeast India. Indian J. Tradit. Knowl. 2009, 8, 169–175. [Google Scholar]
- Kadel, C.; Jain, A.K. Folklore claims on snakebite among some tribal communities of Central India. Indian J. Tradit. Knowl. 2008, 7, 296–299. [Google Scholar]
- Boktapa, N.R.; Sharma, A.K. Wild medicinal plants used by local communities of Manali, Himachal Pradesh, India. Ethnobot. Leafl. 2010, 3, 259–267. [Google Scholar]
- Kingston, C.; Nisha, B.S.; Kiruba, S.; Jeeva, S. Ethnomedicinal plants used by indigenous community in a traditional healthcare system. Ethnobot. Leafl. 2007, 11, 32–37. [Google Scholar]
- Jain, A.; Roshnibala, S.; Kanjilal, P.B.; Singh, R.S.; Singh, H.B. Aquatic /semi-aquatic plants used in herbal remedies in the wetlands of Manipur, Northeastern India. Indian J. Tradit. Knowl. 2007, 6, 346–351. [Google Scholar]
- Yabesh, J.M.; Prabhu, S.; Vijayakumar, S. An ethnobotanical study of medicinal plants used by traditional healers in silent valley of Kerala, India. J. Ethnopharmacol. 2014, 154, 774–789. [Google Scholar] [CrossRef]
- Sher, Z.; Khan, Z.; Hussain, F. Ethnobotanical studies of some plants of Chagharzai valley, district Buner, Pakistan. Pak. J. Bot. 2011, 43, 1445–1452. [Google Scholar]
- Poonam, K.; Singh, G.S. Ethnobotanical study of medicinal plants used by the Taungya community in Terai Arc Landscape, India. J. Ethnopharmacol. 2009, 123, 167–176. [Google Scholar] [CrossRef]
- Shrestha, P.M.; Dhillion, S.S. Medicinal plant diversity and use in the highlands of Dolakha district Nepal. J. Ethnopharmacol. 2003, 86, 81–96. [Google Scholar] [CrossRef]
- Khatun, M.A.; Harun-Or-Rashid, M.; Rahmatullah, M. Scientific validation of eight medicinal plants used in traditional medicinal systems of Malaysia: A review. Am. Eurasian J. Sustain. Agric. 2011, 5, 67–75. [Google Scholar]
- Dastur, J.F. Medicinal Plants of India and Pakistan; D. B. Taraporevala Sons and Co. Ltd: Bombay, India, 1951; p. 12. [Google Scholar]
- Satyavati, G.V.; Raina, M.K.; Sharmal, M. Medicinal Plants of India; Indian Council of Medical Research: New Delhi, India, 1976; Volume I, pp. 14–16. [Google Scholar]
- Jain, S.K. Medicinal Plants; National Book Trust: New Delhi, India, 1968.
- Malhi, B.S.; Trivedi, V.P. Vegetable antifertility drugs of India. Q. J. Crude Drug Res. 1972, 12, 19–22. [Google Scholar]
- Singh, M.P.; Malla, S.B.; Rajbhandari, S.B.; Manandhar, A. Medicinal plants of Nepal retrospect’s and prospects. Econ. Bot. 1979, 33, 185–198. [Google Scholar] [CrossRef]
- Kirtikar, K.R.; Basu, B.D. Indian Medicinal Plants; M/S. Bishen Singh Mahendra Pal Singh: Dehradun, India, 1975; Volume IV. [Google Scholar]
- Lama, S.; Santra, S.C. Development of Tibetan plant medicine. Sci. Cult. 1979, 45, 262–265. [Google Scholar] [PubMed]
- Burang, T. Cancer therapy of Tibetan healers. Comp. Med. East West 1979, 7, 294–296. [Google Scholar] [CrossRef] [PubMed]
- Wallnofer, H.; Rottauscher, A. Chinese Folk Medicine and Acupuncture; Bell Publishing Co, Inc: New York, NY, USA, 1965. [Google Scholar]
- Agarwal, S.L.; Dandiya, P.C.; Singh, K.P.; Arora, R.B. A note on the preliminary studies of certain pharmacological actions of Acorus calamus. J. Am. Pharm. Assoc. 1956, 45, 655–656. [Google Scholar] [CrossRef]
- Duke, J.A.; Ayensu, E.S. Medicinal Plants of China; Reference Publications, Inc: Algonac, MI, USA, 1985. [Google Scholar]
- Perry, L.M.; Metzger, J. Medicinal Plants of East and Southeast Asia; MIT Press: Cambridge, UK, 1980. [Google Scholar]
- Boissya, C.L.; Majumder, R. Some folklore claims from the Brahmaputra Valley (Assam). Ethnomedicine 1980, 6, 139–145. [Google Scholar]
- Dragendorff, G. Die Heilpflanzen der Verschie Denen Volker und Zeiten; F. Enke: Stuttgart, Germany, 1898. [Google Scholar]
- Li, H.L. Hallucinogenic plants in Chinese herbals. Harv. Univ. Bot. Mus. Leafl. 1977, 25, 161–177. [Google Scholar]
- Shih-Chen, L. Chinese Medicinal Herbs; Georgetown Press: San Francisco, CA, USA, 1973. [Google Scholar]
- Hirschhorn, H.H. Botanical remedies of the former Dutch East Indies (Indonesia) I: Eumycetes, Pteridophyta, Gymnospermae, Angiospermae (Monocotyledones only). J. Ethnopharmacol. 1983, 7, 123–156. [Google Scholar] [CrossRef]
- Wren, R.C. Potter’s New Cyclopaedia of Botanical Drugs and Preparations; Sir Isaac Pitman and Sons, Ltd: London, UK, 1956. [Google Scholar]
- Grieve, M. A Modern Herbal; Dover Publications, Inc: New York, NY, USA, 1971; Volume II. [Google Scholar]
- Wheelwright, E.G. Medicinal Plants and Their Stor; Dover Publications, Inc: New York, NY, USA, 1974. [Google Scholar]
- Moerman, D.E. Geraniums for the Iroquois; Reference Publications, Inc: Algonac, MI, USA, 1981. [Google Scholar]
- Jochle, W. Menses-inducing drugs: Their role in antique, medieval and renaissance gynecology and birth control. Contraception 1974, 10, 425–439. [Google Scholar] [CrossRef]
- Watt, J.M.; Breyer-Brandwijk, M.G. The Medicinal and Poisonous Plants of Southern and Eastern Africa; E. & S. Livingstone Ltd.: London, UK, 1962. [Google Scholar]
- Kantor, W. Quack abortifacients and declining birth rate. Therap. Monatsh. 1916, 30, 561–568. [Google Scholar]
- Herrmann, G. Therapy with medicinal plants in present medicine. Med. Monatsschr. Pharm. 1956, 10, 79. [Google Scholar]
- Burkill, I.H. Dictionary of the Economic Products of the Malay Peninsula; Ministry of Agriculture and Cooperatives: Kuala Lumpur, Malaysia, 1966; Volume 1.
- Motley, T.J. The Ethnobotany of Sweet Flag, Acorus calamus (Araceae). Econ. Bot. 1994, 48, 397–412. [Google Scholar] [CrossRef]
- Krochmal, A.; Krochmal, C. A Guide to the Medicinal Plants of the United States; Quadrangle/The New York Times Book Co: New York, NY, USA, 1975. [Google Scholar]
- El’Yashevych, O.H.; Cholii, R. Some means of treatment in the folk medicine of L’Vov. Farmatsevtychnyi Zhurnal 1972, 27, 78. [Google Scholar]
- Barton, B.H.; Castle, T. The British Flora Medica; Chatto and Windus: Piccadilly, London, UK, 1877. [Google Scholar]
- Mokkhasamit, M.; Ngarmwathana, W.; Sawasdimongkol, K.; Permphiphat, U. Pharmacological evaluation of Thai medicinal plants. (Continued). J. Med. Assoc. Thail. 1971, 54, 490–504. [Google Scholar]
- Harris, B.C. The Complete Herbal; Barre Publishers: Barre, MA, USA, 1972. [Google Scholar]
- Lindley, J. Flora Medica; Paternoster-Row: London, UK, 1838. [Google Scholar]
- Caius, J.F. The Medicinal and Poisonous Plants of India; Scientific Publishers: Jodhpur, India, 1986. [Google Scholar]
- Clymer, R.S. Nature’s Healing Agents; Dorrance and Company: Philadelphia, PA, USA, 1963. [Google Scholar]
- Manfred, L. Siete Mil Recetas Botanicas a Base de Mil Trescientas Plantas; Edit Kier: Buenos Aires, Argentina, 1947. [Google Scholar]
- Dobelis, I.N. Magic and Medicine of Plants; The Reader’s Digest Association, Inc.: Pleasantville, New York, NY, USA, 1986. [Google Scholar]
- Kumar, H.; Song, S.Y.; More, S.V.; Kang, S.M.; Kim, B.Y. Traditional Korean East Asian Medicines and Herbal Formulations for Cognitive Impairment. Molecules 2013, 18, 14670–14693. [Google Scholar] [CrossRef]
- Napagoda, M.T.; Sundarapperuma, T.; Fonseka, D.; Amarasiri, S.; Gunaratna, P. Traditional Uses of Medicinal Plants in Polonnaruwa District in North Central Province of Sri Lanka. Scientifica 2019, 2019, 1–12. [Google Scholar] [CrossRef]
- Chaudhury, S.S.; Gautam, S.K.; Handa, K.L. Composition of calamus oil from calamus roots growing in Jammu and Kashmir. Indian J. Pharm. Sci. 1957, 19, 183–186. [Google Scholar]
- Mukherjee, P.K. Quality Control of Herbal Drugs: An Approach to Evaluation of Botanicals; Business Horizons: New Delhi, India, 2002; pp. 692–694. [Google Scholar]
- Soledade, M.; Pedras, C.; Zheng, Q. The Chemistry of Arabidopsis thaliana. Comp. Nat. Prod. 2010, 3, 1297–1315. [Google Scholar]
- Sharma, J.D.; Dandiya, P.C.; Baxter, R.M.; Kandel, S.I. Pharmacodynamical effects of asarone and β-asarone. Nature 1961, 192, 1299–1300. [Google Scholar] [CrossRef]
- Sharma, P.K.; Dandiya, P.C. Synthesis and some pharmacological actions of asarone. Indian J. Appl. Chem. 1969, 32, 236–238. [Google Scholar]
- Nigam, M.C.; Ateeque, A.; Misra, L.N. GC-MS examination of essential oil of Acorus calamus. Indian Perfum. 1990, 34, 282–285. [Google Scholar]
- Matejić, J.; Šarac, Z.; Ranđelović, V. Pharmacological activity of sesquiterpene lactones. Biotech. Biotechnol. Equip. 2010, 24, S95–S100. [Google Scholar] [CrossRef]
- Benaiges, A.; Guillén, P. Botanical Extracts. Anal. Cosmet. Prod. 2007, 345–363. [Google Scholar] [CrossRef]
- Sparg, S.; Light, M.E.; Van Staden, J. Biological activities and distribution of plant saponins. J. Ethnopharmacol. 2007, 94, 219–243. [Google Scholar] [CrossRef]
- Rai, R.; Siddiqui, I.R.; Singh, J. Triterpenoid Saponins from Acorus calamus. ChemInform 1998, 29, 473–476. [Google Scholar]
- Rai, R.; Gupta, A.; Siddiqui, I.R.; Singh, J. Xanthone Glycoside from rhizome of Acorus calamus. Indian J. Chem. 1999, 38, 1143–1144. [Google Scholar]
- Kumar, S.S.; Akram, A.S.; Ahmed, T.F.; Jaabir, M.M. Phytochemical analysis and antimicrobial activity of the ethanolic extract of Acorus calamus rhizome. Orient. J. Chem. 2010, 26, 223–227. [Google Scholar]
- Wu, H.S.; Li, Y.Y.; Weng, L.J.; Zhou, C.X.; He, Q.J.; Lou, Y.J. A Fraction of Acorus calamus L. extract devoid of β-asarone Enhances adipocyte differentiation in 3T3-L1 cells. Phytother. Res. 2007, 21, 562–564. [Google Scholar] [CrossRef]
- Vashi, I.G.; Patel, H.C. Chemical constituents and antimicrobial activity of Acorus calamus Linn. Comp. Physiol. Ecol. 1987, 12, 49–51. [Google Scholar]
- Weber, M.; Brändle, R. Dynamics of nitrogen-rich compounds in roots, rhizomes, and leaves of the Sweet Flag (Acorus calamus L.) at its natural site. Flora 1994, 189, 63–68. [Google Scholar] [CrossRef]
- Asif, M.; Siddiqi, M.T.A.; Ahmad, M.U. Fatty acid and sugar composition of Acorus calamus Linn. Fette Seifen Anstrichm. 1984, 86, 24–25. [Google Scholar] [CrossRef]
- Lee, M.H.; Chen, Y.Y.; Tsai, J.W.; Wang, S.C.; Watanabe, T.; Tsai, Y.C. Inhibitory effect of β-asarone, a component of Acorus calamus essential oil, on inhibition of adipogenesis in 3T3-L1 cells. Food Chem. 2011, 126, 1–7. [Google Scholar] [CrossRef]
- Padalia, R.C.; Chauhan, A.; Verma, R.S.; Bisht, M.; Thul, S.; Sundaresan, V. Variability in rhizome volatile constituents of Acorus calamus L. from Western Himalaya. J. Essent. Oil Bear. Plants 2014, 17, 32–41. [Google Scholar] [CrossRef]
- Kumar, S.N.; Aravind, S.R.; Sreelekha, T.T.; Jacob, J.; Kumar, B.D. Asarones from Acorus calamus in combination with azoles and amphotericin b: A novel synergistic combination to compete against human pathogenic candida species In-vitro. Appl. Biochem. Biotech. 2015, 175, 3683–3695. [Google Scholar] [CrossRef]
- Srivastava, V.K.; Singh, B.M.; Negi, K.S.; Pant, K.C.; Suneja, P. Gas chromatographic examination of some aromatic plants of Uttar Pradesh hills. Indian Perfum. 1997, 41, 129–139. [Google Scholar]
- Özcan, M.; Akgül, A.; Chalchat, J.C. Volatile constituents of the essential oil of Acorus calamus L. grown in Konya province (Turkey). J. Essent. Oil Res. 2002, 14, 366–368. [Google Scholar] [CrossRef]
- Kim, W.J.; Hwang, K.H.; Park, D.G.; Kim, T.J.; Kim, D.W.; Choi, D.K.; Lee, K.H. Major constituents and antimicrobial activity of Korean herb Acorus calamus. Nat. Prod. Res. 2011, 25, 1278–1281. [Google Scholar] [CrossRef]
- Patra, A.; Mitra, A.K. Constituents of Acorus calamus: Structure of acoramone. Carbon-13 NMR spectra of cis-and trans-asarone. J. Nat. Prod. 1981, 44, 668–669. [Google Scholar] [CrossRef]
- Saxena, D.B. Phenyl indane from Acorus calamus. Phytochemistry 1986, 25, 553–555. [Google Scholar] [CrossRef]
- Radušienė, J.; Judžentienė, A.; Pečiulytė, D.; Janulis, V. Essential oil composition and antimicrobial assay of Acorus calamus leaves from different wild populations. Plant Genet. Resour. 2007, 5, 37–44. [Google Scholar] [CrossRef]
- Haghighi, S.R.; Asadi, M.H.; Akrami, H.; Baghizadeh, A. Anti-carcinogenic and anti-angiogenic properties of the extracts of Acorus calamus on gastric cancer cells. Avicenna J. Phytomed. 2017, 7, 145. [Google Scholar]
- Nawamaki, K.; Kuroyanagi, M. Sesquiterpenoids from Acorus calamus as germination inhibitors. Phytochemistry 1996, 43, 1175–1182. [Google Scholar] [CrossRef]
- Zaugg, J.; Eickmeier, E.; Ebrahimi, S.N.; Baburin, I.; Hering, S.; Hamburger, M. Positive GABAA receptor modulators from Acorus calamus and structural analysis of (+)-dioxosarcoguaiacol by 1D and 2D NMR and molecular modeling. J. Nat. Prod. 2011, 74, 1437–1443. [Google Scholar] [CrossRef] [PubMed]
- Yamamura, S.; Iguchi, M.; Nishiyama, A.; Niwa, M.; Koyama, H.; Hirata, Y. Sesquiterpenes from Acorus calamus L. Tetrahedron 1971, 27, 5419–5431. [Google Scholar] [CrossRef]
- Li, J.; Zhao, J.; Wang, W.; Li, L.; Zhang, L.; Zhao, X.F.; Li, S.X. New Acorane-Type Sesquiterpene from Acorus calamus L. Molecules 2017, 22, 529. [Google Scholar] [CrossRef]
- Zhou, C.X.; Qiao, D.; Yan, Y.Y.; Wu, H.S.; Mo, J.X.; Gan, L.S. A new anti-diabetic sesquiterpenoid from Acorus calamus. Chin. Chem. Lett. 2012, 23, 1165–1168. [Google Scholar] [CrossRef]
- Yao, X.; Ling, Y.; Guo, S.; Wu, W.; He, S.; Zhang, Q.; Zou, M.; Nandakumar, K.S.; Chen, X.; Liu, S. Tatanan A from the Acorus calamus L. root inhibited dengue virus proliferation and infections. Phytomedicine 2018, 42, 258–267. [Google Scholar] [CrossRef]
- Prisilla, D.H.; Balamurugan, R.; Shah, H.R. Antidiabetic activity of methanol extract of Acorus calamus in STZ induced diabetic rats. Asian Pac. J. Trop. Biomed. 2012, 2, S941–S946. [Google Scholar] [CrossRef]
- Prashanth, D.; Ahmed, F.Z. Evaluation of hypoglycemic activity of methanolic extract of Acorus calamus (linn). roots in alloxan induced diabetes rat model. Int. J. Basic Clin. Pharmacol. 2017, 6, 2665–2670. [Google Scholar]
- Wu, H.S.; Zhu, D.F.; Zhou, C.X.; Feng, C.R.; Lou, Y.J.; Yang, B.; He, Q.J. Insulin sensitizing activity of ethyl acetate fraction of Acorus calamus L. In-vitro and in-vivo. J. Ethnopharmacol. 2009, 123, 288–292. [Google Scholar] [CrossRef]
- Liu, Y.X.; Si, M.M.; Lu, W.; Zhang, L.X.; Zhou, C.X.; Deng, S.L.; Wu, H.S. Effects and molecular mechanisms of the antidiabetic fraction of Acorus calamus L. on GLP-1 expression and secretion in-vivo and In-vitro. J. Ethnopharmacol. 2015, 166, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Si, M.M.; Lou, J.S.; Zhou, C.X.; Shen, J.N.; Wu, H.H.; Yang, B.; Wu, H.S. Insulin releasing and alpha-glucosidase inhibitory activity of ethyl acetate fraction of Acorus calamus In-vitro and in-vivo. J. Ethnopharmacol. 2010, 128, 154–159. [Google Scholar] [CrossRef] [PubMed]
- Parab, R.S.; Mengi, S.A. Hypolipidemic activity of Acorus calamus L. in rats. Fitoterapia 2002, 73, 451–455. [Google Scholar] [CrossRef]
- D’Souza, T.; Mengi, S.A.; Hassarajani, S.; Chattopadhayay, S. Efficacy study of the bioactive fraction (F-3) of Acorus calamus in hyperlipidemia. Indian J. Pharmacol. 2007, 39, 196–200. [Google Scholar]
- Kumar, G.; Nagaraju, V.; Kulkarni, M.; Kumar, B.S.; Raju, S. Evaluation of Antihyperlipidemic Activity of Methanolic Extract of Acorus Calamus in fat diet Induced Rats. Asian J. Med. Pharm. Sci. 2016, 4, 71–76. [Google Scholar]
- Arun, K.S.; Augustine, A. Hypolipidemic Effect of Methanol Fraction of Acorus calamus Linn. in Diet-Induced Obese Rats. In Prospects in Bioscience: Addressing the Issues; Springer, Springer Science & Business Media: New Delhi, India, 2012; pp. 399–404. [Google Scholar]
- Athesh, K.; Jothi, G. Pharmacological screening of anti-obesity potential of Acorus calamus linn. In high fat cafeteria diet fed obese rats. Asian J. Pharm. Clin. Res. 2017, 10, 384–390. [Google Scholar]
- Patel, P.; Vaghasiya, J.; Thakor, A.; Jariwala, J. Antihypertensive effect of rhizome part of Acorus calamus on renal artery occlusion induced hypertension in rats. Asian Pac. J. Trop. Dis. 2012, 2, S6–S10. [Google Scholar] [CrossRef]
- Shah, A.J.; Gilani, A.H. Blood pressure-lowering and vascular modulator effects of Acorus calamus extract are mediated through multiple pathways. J. Cardiovasc. Pharmacol. 2009, 54, 38–46. [Google Scholar] [CrossRef]
- Sundaramahalingam, M.; Ramasundaram, S.; Rathinasamy, S.D.; Natarajan, R.P.; Somasundaram, T. Role of Acorus calamus and alpha-asarone on hippocampal dependent memory in noise stress exposed rats. Pak. J. Biol. Sci. 2013, 16, 770–778. [Google Scholar] [CrossRef]
- Jain, D.K.; Gupta, S.; Jain, R.; Jain, N. Anti-inflammatory Activity of 80% Ethanolic Extract of Acorus calamus Linn. Leaves in Albino Rats. Res. J. Pharm. Tech. 2010, 3, 882–884. [Google Scholar]
- Manikandan, S.; Devi, R.S. Antioxidant property of α-asarone against noise-stress-induced changes in different regions of rat brain. Pharmacol. Res. 2005, 52, 467–474. [Google Scholar] [CrossRef] [PubMed]
- Devi, S.A.; Ganjewala, D. Antioxidant activities of methanolic extracts of sweet-flag (Acorus calamus) leaves and rhizomes. J. Herbs Spices Med. Plants 2011, 1, 1–11. [Google Scholar] [CrossRef]
- Acuña, U.M.; Atha, D.E.; Ma, J.; Nee, M.H.; Kennelly, E.J. Antioxidant capacities of ten edible North American plants. Phytother. Res. 2002, 16, 63–65. [Google Scholar] [CrossRef] [PubMed]
- Palani, S.; Raja, S.; Kumar, R.P.; Parameswaran, P.; Kumar, B.S. Therapeutic efficacy of Acorus calamus on acetaminophen induced nephrotoxicity and oxidative stress in male albino rats. Acta Pharm. Sci. 2010, 52, 89–100. [Google Scholar]
- Venkatramaniah, C.; Praba, A.M.A. Effect of Beta Asarone–The Active Principle of Acorus Calamus in Neuroprotection and Nerve Cell Regeneration on the Pyramidal Region of Hippocampus in Mesial Temporal Lobe Epileptic Rat Models. J. Neurosci. 2019, 5, 19–24. [Google Scholar]
- Jayaraman, R.; Anitha, T.; Joshi, V.D. Analgesic and anticonvulsant effects of Acorus calamus roots in mice. Int. J. PharmTech Res. 2010, 2, 552–555. [Google Scholar]
- Kaushik, R.; Jain, J.; Yadav, R.; Singh, L.; Gupta, D.; Gupta, A. Isolation of β-Asarone from Acorus calamus Linn. and Evaluation of its Anticonvulsant Activity using MES and PTZ Models in Mice. Pharmacol. Toxicol. Biomed. Rep. 2017, 3, 21–26. [Google Scholar] [CrossRef]
- Chandrashekar, R.; Adake, P.; Rao, S.N. Anticonvulsant activity of ethanolic extract of Acorus calamus rhizome in swiss albino mice. J. Sci. Innov. Res. 2013, 2, 846–851. [Google Scholar]
- Yende, S.R.; Harle, U.N.; Bore, V.V.; Bajaj, A.O.; Shroff, K.K.; Vetal, Y.D. Reversal of neurotoxicity induced cognitive impairment associated with phenytoin and phenobarbital by Acorus calamus in mice. J. Herb. Med. Toxicol. 2009, 3, 111–115. [Google Scholar]
- Pawar, V.S.; Anup, A.; Shrikrishna, B.; Shivakumar, H. Antidepressant–like effects of Acorus calamus in forced swimming and tail suspension test in mice. Asian Pac. J. Trop. Biomed. 2011, 1, S17–S19. [Google Scholar] [CrossRef]
- Pushpa, V.H.; Padmaja, S.K.; Suresha, R.N.; Vaibhavi, P.S.; Kalabharathi, H.L.; Satish, A.M.; Naidu, S. Antidepressant Activity of Methanolic Extract of Acorus Calamus Leaves in Albino Mice. Int. J. Pharm. Tech. 2013, 5, 5458–5465. [Google Scholar]
- Shashikala, G.H.; Prashanth, D.; Jyothi, C.H.; Maniyar, I.; Manjunath, H. Evaluation of antidepressant activity of aqueous extract of roots of acorus calamus in albino mice. World J. Pharm. Res. 2015, 4, 1357–1365. [Google Scholar]
- De, A.; Singh, M.S. Acorus calamus linn. Rhizomes extract for antidepressant activity in mice model. Adv. Res. Pharm. Biol. 2013, 3, 520–525. [Google Scholar]
- Tripathi, A.K.; Singh, R.H. Experimental evaluation of antidepressant effect of Vacha (Acorus calamus) in animal models of depression. Ayu 2010, 31, 153–158. [Google Scholar] [CrossRef] [PubMed]
- Pandy, V.; Jose, N.; Subhash, H. CNS activity of methanol and acetone extracts of Acorus calamus leaves in mice. J. Pharmacol. Toxicol. 2009, 4, 79–86. [Google Scholar] [CrossRef]
- Tiwari, N.; Mishra, A.; Bhatt, G.; Chaudhary, A. Isolation of Principle Active Compound of Acorus Calamus. In-vivo assessment of pharmacological activity in the treatment of neurobiological disorder (stress). J. Med. Clin. Res. 2014, 2, 2201–2212. [Google Scholar]
- Muthuraman, A.; Singh, N. Neuroprotective effect of saponin rich extract of Acorus calamus L. in rat model of chronic constriction injury (CCI) of sciatic nerve-induced neuropathic pain. J. Ethnopharmacol. 2012, 142, 723–731. [Google Scholar] [CrossRef]
- Muthuraman, A.; Singh, N. Attenuating effect of Acorus calamus extract in chronic constriction injury induced neuropathic pain in rats: An evidence of anti-oxidative, anti-inflammatory, neuroprotective and calcium inhibitory effects. BMC Complement. Altern. Med. 2011, 11, 1–14. [Google Scholar] [CrossRef]
- Vengadesh Prabu, K.; George, T.; Vinoth Kumar, R.; Nancy, J.; Kalaivani, M.; Vijayapandi, P. Neuromodulatory effect of Acrous calamus leaves extract on dopaminergic system in mice. Int. J. PharmTech Res. 2009, 1, 1255–1259. [Google Scholar]
- Hazra, R.; Guha, D. Effect of chronic administration of Acorus calamus on electrical activity and regional monoamine levels in rat brain. Biog. Amines 2003, 17, 161–170. [Google Scholar] [CrossRef]
- Shukla, P.K.; Khanna, V.K.; Ali, M.M.; Maurya, R.; Khan, M.Y.; Srimal, R.C. Neuroprotective effect of Acorus calamus against middle cerebral artery occlusion–induced ischaemia in rat. Hum. Exp. Toxicol. 2006, 25, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Fathima, A.; Patil, H.V.; Kumar, S. Suppression of elevated reactive oxygen species by acorus calamus (vacha) a sweet flag in drosophila melanogaster under stress full conditions. Int. J. Pharm. Sci. Res. 2014, 5, 1431–1439. [Google Scholar]
- Kumar, M.S.; Hiremath, V.S.M.A. Cardioprotective effect of Acorus calamus against doxorubicin-induced myocardial toxicity in albino Wistar rats. Indian J. Health Sci. Biomed. Res. 2016, 9, 225–234. [Google Scholar]
- Shah, A.J.; Gilani, A.H. Bronchodilatory effect of Acorus calamus (Linn.) is mediated through multiple pathways. J. Ethnopharmacol. 2010, 131, 471–477. [Google Scholar] [CrossRef] [PubMed]
- Thakare, M.M.; Surana, S.J. β-Asarone modulate adipokines and attenuates high fat diet-induced metabolic abnormalities in Wistar rats. Pharmacol. Res. 2016, 103, 227–235. [Google Scholar] [CrossRef] [PubMed]
- Karthiga, T.; Venkatalakshmi, P.; Vadivel, V.; Brindha, P. In-vitro anti-obesity, antioxidant and anti-inflammatory studies on the selected medicinal plants. Int. J. Toxicol. Pharmacol. Res. 2016, 8, 332–340. [Google Scholar]
- Singh, D.K.; Kumar, N.; Sachan, A.; Lakhani, P.; Tutu, S.; Shankar, P.; Dixit, R.K. An experimental study to see the antihypertensive effects of gymnema sylvestre and acorus calamus in wistar rats and its comparison with amlodipine. Asian J. Med. Sci. 2017, 8, 11–15. [Google Scholar] [CrossRef]
- Tanaka, S.; Yoichi, S.; Ao, L.; Matumoto, M.; Morimoto, K.; Akimoto, N.; Zaini bin Asmawi, M. Potential immunosuppressive and anti-inflammatory activities of Malaysian medicinal plants characterized by reduced cell surface expression of cell adhesion molecules. Phytother. Res. 2001, 15, 681–686. [Google Scholar] [CrossRef]
- Kim, H.; Han, T.H.; Lee, S.G. Anti-inflammatory activity of a water extract of Acorus calamus L. leaves on keratinocyte HaCaT cells. J. Ethnopharmacol. 2009, 122, 149–156. [Google Scholar] [CrossRef]
- Ahmed, S.; Gul, S.; Zia-Ul-Haq, M.; Stanković, M.S. Pharmacological basis of the use of Acorus calamus L. in inflammatory diseases and underlying signal transduction pathways. Bol. Latinoam. Caribe Plantas Med. Aromát. 2014, 13, 38–46. [Google Scholar]
- Loying, R.; Gogoi, R.; Sarma, N.; Borah, A.; Munda, S.; Pandey, S.K.; Lal, M. Chemical Compositions, In-vitro Antioxidant, Anti-microbial, Anti-inflammatory and Cytotoxic Activities of Essential Oil of Acorus calamus L. Rhizome from North-East India. J. Essent. Oil Bear. Plants 2019, 22, 1299–1312. [Google Scholar] [CrossRef]
- Bahukhandi, A.; Rawat, S.; Bhatt, I.D.; Rawal, R.S. Influence of solvent types and source of collection on total phenolic content and antioxidant activities of Acorus calamus L. Natl. Acad. Sci. Lett. 2013, 36, 93–99. [Google Scholar] [CrossRef]
- Manju, S.; Chandran, R.P.; Shaji, P.K.; Nair, G.A. In-vitro free radical scavenging potential of Acorus Calamus L. rhizome from Kuttanad Wetlands, Kerala, India. Int. J. Pharm. Pharm. Sci. 2013, 5, 376–380. [Google Scholar]
- Barua, C.C.; Sen, S.; Das, A.S.; Talukdar, A.; Hazarika, N.J.; Barua, A.G.; Barua, I. A comparative study of the In-vitro antioxidant property of different extracts of Acorus calamus Linn. J. Nat. Prod. Plant Resour. 2014, 4, 8–18. [Google Scholar]
- Elayaraja, A.; Vijayalakshmi, M.; Devalarao, G. In-vitro free radical scavenging activity of various root and rhizome extracts of Acorus calamus Linn. Int. J. Pharm. Biol. Sci. 2010, 1, 301–304. [Google Scholar]
- Govindarajan, R.; Agnihotri, A.K.; Khatoon, S.; Rawat, A.K.S.; Mehrotra, S. Pharmacognostical evaluation of an antioxidant plant-Acorus calamus Linn. Nat. Prod. Sci. 2003, 9, 264–269. [Google Scholar]
- Sujitha, R.; Bhimba, B.V.; Sindhu, M.S.; Arumugham, P. Phytochemical Evaluation and Antioxidant Activity of Nelumbo nucifera, Acorus calamus and Piper longum. Int. J. Pharm. Chem. Sci. 2013, 2, 1573–1578. [Google Scholar]
- Shukla, R.; Singh, P.; Prakash, B.; Dubey, N.K. Efficacy of Acorus calamus L. essential oil as a safe plant-based antioxidant, A flatoxin B 1 suppressor and broad-spectrum antimicrobial against food-infesting fungi. Int. J. Food Sci. Tech. 2013, 48, 128–135. [Google Scholar] [CrossRef]
- Ahmeda, F.; Urooja, A.; KS, R. In-vitro antioxidant and anticholinesterase activity of Acorus calamus and Nardostachys jatamansi rhizomes. J. Pharm. Res. 2009, 2, 830–833. [Google Scholar]
- Bhat, S.D.; Ashok, B.K.; Acharya, R.N.; Ravishankar, B. Anticonvulsant activity of raw and classically processed Vacha (Acorus calamus Linn.) rhizomes. Ayu 2012, 33, 119–122. [Google Scholar] [CrossRef]
- Patel, S.; Rajshree, N.; Shah, P. Evaluation of antidepressant activity of herbomineral formulation. Int. J. Pharm. Pharm. Sci. 2016, 8, 145–147. [Google Scholar]
- Rauniar, G.P.; Deo, S.; Bhattacharya, S.K. Evaluation of anxiolytic activity of tensarin in mice. Kathman. Univ. Med. J. 2007, 5, 188–194. [Google Scholar]
- Naderi, G.A.; Khalili, M.; Karimi, M.; Soltani, M. The effect of oral and intraperitoneal administration of Acorus calamus L. extract on learning and memory in male rats. J. Med. Plant 2010, 2, 46–56. [Google Scholar]
- Vohora, S.B.; Shah, S.A.; Dandiya, P.C. Central nervous system studies on an ethanol extract of Acorus calamus rhizomes. J. Ethnopharmacol. 1990, 28, 53–62. [Google Scholar] [CrossRef]
- Singh, B.K.; Pillai, K.K.; Kohli, K.; Haque, S.E. Isoproterenol-Induced Cardiomyopathy in Rats: Influence of Acorus calamus Linn. Cardiovasc. Toxicol. 2011, 11, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Shah, A.J.; Gilani, A.H. Aqueous-methanolic extract of sweet flag (Acorus calamus) possesses cardiac depressant and endothelial-derived hyperpolarizing factor-mediated coronary vasodilator effects. J. Nat. Med. 2012, 66, 119–126. [Google Scholar] [CrossRef]
- Hasheminejad, G.; Caldwell, J. Genotoxicity of the alkenylbenzenes α− and β-asarone, myristicin and elemicin as determined by the UDS assay in cultured rat hepatocytes. Food Chem. Toxicol. 1994, 32, 223–231. [Google Scholar] [CrossRef]
- Cartus, A.T.; Schrenk, D. Metabolism of the carcinogen alpha-asarone in liver microsomes. Food Chem. Toxicol. 2016, 87, 103–112. [Google Scholar] [CrossRef]
- Cartus, A.T.; Stegmuller, S.; Simson, N.; Wahl, A.; Neef, S.; Kelm, H.; Schrenk, D. Hepatic metabolism of carcinogenic betaasarone. Chem. Res. Toxicol. 2015, 28, 1760–1773. [Google Scholar] [CrossRef]
- Cartus, A.T.; Schrenk, D. Metabolism of carcinogenic alpha-asarone by human cytochrome P450 enzymes. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2020, 393, 213–223. [Google Scholar] [CrossRef]
- Pandit, S.; Mukherjee, P.K.; Ponnusankar, S.; Venkatesh, M.; Srikanth, N. Metabolism mediated interaction of α-asarone and Acorus calamus with CYP3A4 and CYP2D6. Fitoterapia 2011, 82, 369–374. [Google Scholar] [CrossRef] [PubMed]
- Muthuraman, A.; Singh, N. Acute and sub-acute oral toxicity profile of Acorus calamus (Sweet flag) in rodents. Asian Pac. J. Trop Biomed. 2012, 2, S1017–S1023. [Google Scholar] [CrossRef]
- Areekul, S.; Sinchaisri, P.; Tigvatananon, S. Effects of Thai plant extracts on the oriental fruit fly III. Nat. Sci. 1988, 22, 160–164. [Google Scholar]
- Shah, P.D.; Ghag, M.; Deshmukh, P.B.; Kulkarni, Y.; Joshi, S.V.; Vyas, B.A.; Shah, D.R. Toxicity study of ethanolic extract of Acorus calamus rhizome. Int. J. Green Pharm. 2012, 6, 29–35. [Google Scholar] [CrossRef]
- Bhat, S.D.; Ashok, B.K.; Acharya, R.; Ravishankar, B. A comparative acute toxicity evaluation of raw and classically processed rhizomes of Vacha (Acorus calamus Linn.). Indian J. Nat. Prod. Resour. 2012, 3, 506–511. [Google Scholar]
- Keller, K.; Stahl, E. Composition of the essential oil from beta-Asarone free calamus. Planta Med. 1983, 47, 71–74. [Google Scholar] [CrossRef]
- JECFA (Joint FAO/WHO Expert Committee on Food Additives). Monograph on -asarone. In WHO Food Additive Series No. 16; WHO Food Additives Series; JECFA, WHO Press: Geneva, Switzerland, 1981. [Google Scholar]
- Opdyke, D.L.J. Monographs on fragrance raw materials. Food Cosmet. Toxicol. 1973, 11, 855–876. [Google Scholar] [CrossRef]
- Jenner, P.M.; Hagan, E.C.; Taylor, J.M.; Cook, E.L.; Fitzhugh, O.G. Food flavourings and compounds of related structure I. Acute oral toxicity. Food Cosmet. Toxicol. 1964, 2, 327–343. [Google Scholar] [CrossRef]
- Singh, A.K.; Ravishankar, B.; Sharma, P.P.; Pandaya, T. Clinical study of anti-hyperlipidaemic activity of vacha (Acorus calamus linn) w.s.r to sthaulya. Int. Ayurvedic Med. J. 2017, 5, 1–8. [Google Scholar]
- Tajadini, H.; Saifadini, R.; Choopani, R.; Mehrabani, M.; Kamalinejad, M.; Haghdoost, A.A. Herbal medicine Davaie Loban in mild to moderate Alzheimer’s disease: A 12-week randomized double-blind placebo-controlled clinical trial. Complement. Ther. Med. 2015, 23, 767–772. [Google Scholar] [CrossRef]
- Bhattacharyya, D.; Sur, T.K.; Lyle, N.; Jana, U.; Debnath, P.K. A clinical study on the management of generalized anxiety disorder with Vaca (Acorus calamus). Indian J. Tradit. Knowl. 2011, 10, 668–671. [Google Scholar]
- Soni1, P.; Sharma, C. A clinical study of Vachadi Churna in the management of obesity. Int. J. Ayurveda Allied Sci. 2012, 1, 179–186. [Google Scholar]
- Kulatunga, R.D.H.; Dave, A.R.; Baghel, M.S. Clinical efficacy of Guduchyadi Medhya Rasayana on senile memory impairment. Ayu 2012, 33, 202–208. [Google Scholar] [CrossRef] [PubMed]
- Pande, D.N.; Mishra, S.K. Vacha (Acorus Calamus) as an ayurvedic premedicant. Ayu 2009, 30, 279–283. [Google Scholar]
- Mishra, J.; Joshi, N.P.; Pandya, D.M. A comparative study of Shankhapushpyadi Ghana Vati and Sarpagandhadi Ghana Vati in the management of “Essential Hypertension”. Ayu 2012, 33, 54–61. [Google Scholar] [CrossRef]
- Ramu, M.G.; Senapati, H.M.; Janakiramaiah, N.; Shankara, M.R.; Chaturvedi, D.D.; Murthy, N.N. A pilot study of role of brahmyadiyoga in chronic unmada (schizophrenia). Anc. Sci. Life 1983, 2, 205–207. [Google Scholar]
- Appaji, R.R.; Sharma, R.D.; Katiyar, G.P.; Sai, P.A. Clinical study of the Immunoglobululin Enhancing Effect of “Bala compound” on Infants. Anc. Sci. Life 2009, 28, 18–22. [Google Scholar]
- Pawar, M.; Magdum, P. Clinical study of assessment of therapeutic potential of Vachadi ghrita, a medicated ghee formulation on healthy individual’s cognition. Int. J. Pharm. Sci. Res. 2018, 9, 3408–3413. [Google Scholar]
- Mishra, D.; Tubaki, B.R. Effect of Brahmi vati and Sarpagandha Ghana vati in management of essential hypertension–A randomized, double blind, clinical study. J. Ayurveda Integr. Med. 2019, 10, 269–276. [Google Scholar] [CrossRef]
- Sharma, Y.; Upadhyay, A.; Sharma, Y.K.; Chaudhary, V. A randomized clinical study to evaluate the effect of Tagaradi yoga in the management of insomnia. Indian J. Tradit. Knowl. 2017, 16, S75–S80. [Google Scholar]
- Paradkar, S.R.; Pardhi, S.N. Clinical evaluation of lekhaniya effect of vacha (acorus calamus) and musta (cyperus rotundus) in medoroga wsr to obesity: A comparative study. Res. Rev. J. Pharmacogn. 2019, 3, 1–8. [Google Scholar]
- Mamgain, P.; Singh, R.H. Control clinical trial of the lekhaniya drug vaca (Acorus calamus) in case of ischemic heart diseases. J. Res. Ayurveda Siddha 1994, 15, 35–51. [Google Scholar]
- Ning, B.; Zhang, Q.; Wang, N.; Deng, M.; Fang, Y. β-Asarone Regulates ER Stress and Autophagy Via Inhibition of the PERK/CHOP/Bcl-2/Beclin-1 Pathway in 6-OHDA-Induced Parkinsonian Rats. Neurochem. Res. 2019, 44, 1159–1166. [Google Scholar] [CrossRef]
- Ning, B.; Deng, M.; Zhang, Q.; Wang, N.; Fang, Y. β-Asarone inhibits IRE1/XBP1 endoplasmic reticulum stress pathway in 6-OHDA-induced parkinsonian rats. Neurochem. Res. 2016, 41, 2097–2101. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Deng, M.; Zhang, S.; Fang, Y.; Li, L. Coadministration of β-asarone and levodopa increases dopamine in rat brain by accelerating transformation of levodopa: A different mechanism from M adopar. Clin. Exp. Pharmacol. Physiol. 2014, 41, 685–690. [Google Scholar] [PubMed]
- Huang, L.; Deng, M.; He, Y.; Lu, S.; Ma, R.; Fang, Y. β-asarone and levodopa co-administration increase striatal dopamine level in 6-hydroxydopamine induced rats by modulating P-glycoprotein and tight junction proteins at the blood-brain barrier and promoting levodopa into the brain. Clin. Exp. Pharmacol. Physiol. 2016, 43, 634–643. [Google Scholar] [CrossRef]
- Chang, W.; Teng, J. β-asarone prevents Aβ25-35-induced inflammatory responses and autophagy in SH-SY5Y cells: Down expression Beclin-1, LC3B and up expression Bcl-2. Int. J. Clin. Exp. Med. 2015, 8, 20658. [Google Scholar]
- Liu, S.J.; Yang, C.; Zhang, Y.; Su, R.Y.; Chen, J.L.; Jiao, M.M.; Quan, S.J. Neuroprotective effect of β-asarone against Alzheimer’s disease: Regulation of synaptic plasticity by increased expression of SYP and GluR1. Drug Des. Dev. Ther. 2016, 10, 1461. [Google Scholar] [CrossRef]
- Li, C.; Xing, G.; Dong, M.; Zhou, L.; Li, J.; Wang, G.; Niu, Y. Beta-asarone protection against beta-amyloid-induced neurotoxicity in PC12 cells via JNK signaling and modulation of Bcl-2 family proteins. Eur. J. Pharmacol. 2010, 635, 96–102. [Google Scholar] [CrossRef]
- Xue, Z.; Guo, Y.; Zhang, S.; Huang, L.; He, Y.; Fang, R.; Fang, Y. Beta-asarone attenuates amyloid beta-induced autophagy via Akt/mTOR pathway in PC12 cells. Eur. J. Pharmacol. 2014, 741, 195–204. [Google Scholar] [CrossRef]
- Yang, Q.Q.; Xue, W.Z.; Zou, R.X.; Xu, Y.; Du, Y.; Wang, S.; Chen, X.T. β-Asarone rescues Pb-induced impairments of spatial memory and synaptogenesis in rats. PLoS ONE 2016, 11, e0167401. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.H.; Chen, Y.; Wei, G.; Nei, H.; Zhou, Y.; Cheng, S. Effects of active components of Rhizoma Acori Tatarinowii and their compatibility at different ratios on learning and memory abilities in dementia mice. Tradit. Chin. Drug Res. Clin. Pharmacol. 2012, 23, 144–147. [Google Scholar]
- Li, J.; Li, Z.X.; Zhao, J.P.; Wang, W.; Zhao, X.F.; Xu, B.; Li, S.X. A Novel Tropoloisoquinoline Alkaloid, Neotatarine, from Acorus calamus L. Chem. Biodivers. 2017, 14, e1700201. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Cai, Q.; Li, J.; Guo, W. Involvement of brain-gut axis in treatment of cerebral infarction by β-asaron and paeonol. Neurosci. Lett. 2018, 666, 78–84. [Google Scholar] [CrossRef]
- Gao, E.; Zhou, Z.Q.; Zou, J.; Yu, Y.; Feng, X.L.; Chen, G.D.; Gao, H. Bioactive Asarone-derived phenylpropanoids from the rhizome of Acorus tatarinowii Schott. J. Nat. Prod. 2017, 80, 2923–2929. [Google Scholar] [CrossRef]
- Zhang, S.; Gui, X.H.; Huang, L.P.; Deng, M.Z.; Fang, R.M.; Ke, X.H.; Fang, Y.Q. Neuroprotective effects of β-asarone against 6-hydroxy dopamine-induced parkinsonism via JNK/Bcl-2/Beclin-1 pathway. Mol. Neurobiol. 2016, 53, 83–94. [Google Scholar] [CrossRef]
- Liang, S.; Ying, S.S.; Wu, H.H.; Liu, Y.T.; Dong, P.Z.; Zhu, Y.; Xu, Y.T. A novel sesquiterpene and three new phenolic compounds from the rhizomes of Acorus tatarinowii Schott. Bioorg. Med. Chem. Lett. 2015, 25, 4214–4218. [Google Scholar] [CrossRef]
- Xu, F.; Wu, H.; Zhang, K.; Lv, P.; Zheng, L.; Zhao, J. Pro-neurogenic effect of β-asarone on RSC96 Schwann cells in vitro. In Vitro Cell. Dev. Biol. Anim. 2016, 52, 278–286. [Google Scholar] [CrossRef]
- Deng, M.; Huang, L.; Ning, B.; Wang, N.; Zhang, Q.; Zhu, C.; Fang, Y. β-asarone improves learning and memory and reduces Acetyl Cholinesterase and Beta-amyloid 42 levels in APP/PS1 transgenic mice by regulating Beclin-1-dependent autophagy. Brain Res. 2016, 1652, 188–194. [Google Scholar] [CrossRef]
- Yang, Y.; Xuan, L.; Chen, H.; Dai, S.; Ji, L.; Bao, Y.; Li, C. Neuroprotective Effects and Mechanism of β-Asarone against Aβ1–42-Induced Injury in Astrocytes. Evid.-Based Complement. Altern. Med. 2017, 2017, 8516518. [Google Scholar] [CrossRef]
- Dong, H.; Gao, Z.; Rong, H.; Jin, M.; Zhang, X. β-asarone reverses chronic unpredictable mild stress-induced depression-like behavior and promotes hippocampal neurogenesis in rats. Molecules 2014, 19, 5634–5649. [Google Scholar] [CrossRef] [PubMed]
- Chellian, R.; Pandy, V.; Mohamed, Z. Biphasic effects of α-asarone on immobility in the tail suspension test: Evidence for the involvement of the noradrenergic and serotonergic systems in its antidepressant-like activity. Front. Pharmacol. 2016, 7, 72. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Song, Z.; Liao, D.G.; Zhang, T.Y.; Liu, F.; Zhuang, K.; Lei, J.P. Anticonvulsant and sedative effects of eudesmin isolated from Acorus tatarinowii on mice and rats. Phytother. Res. 2015, 29, 996–1003. [Google Scholar] [CrossRef] [PubMed]
- Tian, J.; Tian, Z.; Qin, S.L.; Zhao, P.Y.; Jiang, X. Anxiolytic-like effects of α-asarone in a mouse model of chronic pain. Metab. Brain Dis. 2017, 32, 2119–2129. [Google Scholar] [CrossRef] [PubMed]
- Miao, J.K.; Chen, Q.X.; Li, C.; Li, X.W.; Wu, X.M.; Zhang, X.P. Modulation Effects of α-Asarone on the GABA homeostasis in the Lithium-Pilocarpine Model of Temporal Lobe Epilepsy. Pharmacology 2013, 9, 24–32. [Google Scholar]
- Wang, Z.J.; Levinson, S.R.; Sun, L.; Heinbockel, T. Identification of both GABAA receptors and voltage-activated Na+ channels as molecular targets of anticonvulsant α-asarone. Front. Pharmacol. 2014, 5, 40. [Google Scholar] [CrossRef]
- Chen, L.; Liao, W.P. Changes of amino acid content in hippocampus of epileptic rats treated with volatile oil of Acorus tatarinowii. Zhongguo ZhongYao ZaZhi 2004, 29, 670–673. [Google Scholar]
- Jo, M.J.; Kumar, H.; Joshi, H.P.; Choi, H.; Ko, W.K.; Kim, J.M.; Kim, K.T. Oral administration of α-Asarone promotes functional recovery in rats with spinal cord injury. Front. Pharmacol. 2018, 9, 445. [Google Scholar] [CrossRef]
- Lam, K.Y.; Yao, P.; Wang, H.; Duan, R.; Dong, T.T.; Tsim, K.W. Asarone from Acori Tatarinowii Rhizome prevents oxidative stress-induced cell injury in cultured astrocytes: A signaling triggered by Akt activation. PLoS ONE 2017, 12, e0179077. [Google Scholar] [CrossRef]
- Wu, Q.D.; Yuan, D.J.; Wang, Q.W.; Wu, X.R. Effects of volatile oil of Rhizoma Acori Tatarinowii on morphology and cell viability in cultured cardiac myocytes. Zhong Yao Cai 2009, 32, 242–245. [Google Scholar]
- Yong, H.Y.F.Y.J.; Shuying, L.Y.W. In-vitro Observation of β-asarone for Counteracting Arteriosclerosis. J. Guangzhou Univ. Tradit. Chin. Med. 2008, 3, 14. [Google Scholar]
- Yang, Y.X.; Chen, Y.T.; Zhou, X.J.; Hong, C.L.; Li, C.Y.; Guo, J.Y. Beta-asarone, a major component of Acorus tatarinowii Schott, attenuates focal cerebral ischemia induced by middle cerebral artery occlusion in rats. BMC Complement. Altern. Med. 2013, 13, 236. [Google Scholar] [CrossRef] [PubMed]
- Das, B.K.; Choukimath, S.M.; Gadad, P.C. Asarone and metformin delays experimentally induced hepatocellular carcinoma in diabetic milieu. Life Sci. 2019, 230, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Kim, K.Y.; Ryu, S.Y.; Yoon, Y.O.O.S.I.K.; Hahm, D.H.; Kang, S.A.; Lee, H.G. Asarone inhibits adipogenesis and stimulates lipolysis in 3T3-L1 adipocytes. Cell. Mol. Biol. 2010, 56, 1215–1222. [Google Scholar]

| Country | Ailment/Use | Part Used/Dosage Form | Route of Administration | References |
|---|---|---|---|---|
| India | Eczema | The paste of A. calamus rhizomes are given with the paste of Curcuma aromatica rhizomes and Azadirachta indica leaves | Oral | [13] |
| Skin diseases | Rhizomes paste A. calamus and C. aromatica are applied with the seed paste of Argemone Mexicana | |||
| Cough, stuttering, ulcer, fever, dermatitis, scab, sores | Rhizomes | [14] | ||
| Cold, cough, and fever | Rhizomes paste of A. calamus is given to children with mother’s milk, Myristica fragrance, and Calunarejan spinosa fruits | [15] | ||
| Two teaspoonfuls of herbal powder containing A. calamus rhizomes, Boerhaavia diffusa roots, Calonyction muricutum flower pedicles, Ipomoea muricate seeds, Senna leaves, Cassia fistula fruits pulp, Curcuma longa rhizomes, Helicteres isora fruits, and Mentha arvensis leaves, black pepper is taken with lukewarm water | [16] | |||
| Gastric disorders | A. calamus rhizomes paste is given with cow milk | [17] | ||
| Carminative, flavoring, tonic, and head lice infestation | Infusion of a dried rhizomes (collected and stored in the autumn season) | [17,18,19] | ||
| Epilepsy, dysentery, mental illnesses, diarrhea, kidney and liver disorders | A. calamus rhizomes paste is given with honey | [20] | ||
| Wounds, fever, body pain | Rhizomes | [21,22] | ||
| Dysentery | Fresh ground rhizomes is mixed with hot water and given for 3 days | [23] | ||
| Stimulant | Dry powder of A. calamus is given with honey | [24] | ||
| Injuries | External application of the A. calamus rhizomes paste | Dermal | [25] | |
| Stomachache | Ash of the A. calamus rhizomes paste | [26] | ||
| Otitis externa | A. calamus roots paste is given with coconut husk juice | [27] | ||
| Lotion | Fresh leaves of A. calamus | [28] | ||
| Cough, cancer, and fever | A. calamus roots juice is given with honey and MyristicaDactyloides | Oral | [29] | |
| Analgesic | A. calamus rhizomes are given with cinchona bark | [30] | ||
| Gastrointestinal, respiratory, emmenagogue, antihelmintic | Rhizomes | |||
| Prolonged labor | Rhizomes is applied with saffron and horse milk | |||
| Paralysis, arthritis | Rhizomes ash is applied with castor oil | |||
| Neurological disorder, gastrointestinal, respiratory, increases menstrual flow, analgesic, contraceptive | Rhizomes | Oral | [31,32,33] | |
| Herpangina, analgesic, neurological disorder, gastrointestinal, respiratory | [34] | |||
| Pakistan | Colic and diarrhea | Whole plant | [35] | |
| Nepal | Blood pressure | Roots infusion of A. calamus | [36] | |
| Cough, headache, snake bite, sore throat, and pain | Rhizomes | [37] | ||
| Dysentery | Rhizomes juice is given with hot water | |||
| Neurological, respiratory | Rhizomes | [38] | ||
| Malaysia | Rheumatism, diarrhea, dyspepsia, and hair loss | Whole plant | [39] | |
| Tibet | Fever, gastrointestinal | Dried rhizomes is given with Saussurea lappa, Ferula foetida, Terminalia chebula, Cuminum cyminum, Inula racemosa, and Zingiber officinale | [40] | |
| Cancer | Rhizomes | [41] | ||
| China | Gastrointestinal, respiratory, neuroprotective, analgesic, contraceptive, cancer | Rhizomes | [42,43,44] | |
| Antipyretic and ear-related disease | Rhizomes given with squeezed Coccinia cordifolia stems along with water | [45] | ||
| Detoxification | Rhizomes with vinegar, Alpinia galanga, Zingiber purpureum | |||
| Analgesic | Herbal baths of the rhizome | External | ||
| Hemorrhage | Rhizomes paste | [46] | ||
| Aphrodisiac | Rhizomes | Oral | [47] | |
| Hallucination | Rhizomesare mixed with Indian hemp and Podophyllum pleianthum | [48] | ||
| Fair skin | Leaves of A. calamus are given with Artemisia vulgaris | Dermal | [49] | |
| Indonesia | Gastrointestinal | Rhizomes | Oral | [50] |
| England | Rhizomes blended with chalk and magnesium oxide | [51] | ||
| Gastrointestinal, antibacterial, analgesic | Rhizomes | [52] | ||
| Neurological, dysentery, and chronic catarrh | Rhizomesare given with Gentiana campestris L. | |||
| Malaria | Rhizomes | [53] | ||
| Europe | Obesity, influenza, gastrointestinal, respiratory | [54,55] | ||
| Republic of South Africa | Tooth powder, gastrointestinal, tonic, aphrodisiac | [56] | ||
| Sweden | Liquor | [57] | ||
| Germany | Increases menstrual flow, gastrointestinal | [58,59] | ||
| Java | Lactation | [60] | ||
| Lithuania | Chest pain, diarrhea | Rhizomes and leaves are taken with sugar | [52] | |
| Relieves pain, gout, rheumatism | Leaves decoction | External | [61] | |
| New Guinea | Miscarriage | Rhizomes | Oral | [62] |
| Philippines | Gastrointestinal, rheumatism | [56] | ||
| Russia | Typhoid, syphilis, baldness, fever, cholera | [63] | ||
| Thailand | Blood purifier, fever | [64] | ||
| Turkey | Wound healing, cough, tuberculosis | External and oral | [61] | |
| Gastrointestinal | Oral | [65,66] | ||
| Arab countries | Gastrointestinal, tuberculosis | [67,68] | ||
| Brazil | Destroys parasitic worms | [68] | ||
| Argentina | Dysmenorrhea | [69] | ||
| United States | Gastrointestinal, abortifacient, stimulant, tonic, respiratory disorder | Rhizomes | [70] | |
| Korea | Improves memory and life span | [71] | ||
| Sri Lanka | Cough, worm infestation | Rhizomes paste are given with milk | [72] |
| Medicine/Formulations | Indications/Use | Manufacturers |
|---|---|---|
| Pilochek tablets | Hemorrhoids | Dabur India Limited |
| Brahm Rasayan | Nervine tonic | |
| Mahasudarsan Churna | Malaria | |
| Janma Ghunti Honey | Babies growth, Constipation, Diarrhea | |
| Brahmi Pearls capsules | Brain Nourisher | Kerala Ayurveda |
| GT capsules | Osteoarthritis, osteoporosis, hyperlipidemia | |
| Histantin tablets | Anti-allergic | |
| Santhwanam oil | Antioxidant, rejuvenate | |
| Mahathikthaka Ghrita capsules | Skin disease, malabsorption syndrome | |
| Calamus root tincture | Stimulates the digestive system | Florida Herbal Pharmacy |
| Vacha capsules | Food supplements | DR Wakde’s Natural Health Care, London |
| Mentat tablets and syrup | Nervine tonic | Himalaya Herbal Healthcare |
| Abana | Cardiovascular disorders, hyperlipidemia, dyslipidemia | |
| Mentat tablets and Syrup | Anxiety, depression, insomnia | |
| Muscle & Joint Rub | Backaches, muscular sprains, pain | |
| Anxocare | Anxiety | |
| Erina-EP | Ectoparasites | |
| Himpyrin, Himpyrin Vet | Analgesic and anti-inflammatory | |
| Scavon Vet | Anti-bacterial, anti-fungal | |
| Vacha powder | Brain tonic, improves digestion, and prevents nausea | Bixa Botanical |
| Amalth | Herbal supplements | Mcnow Biocare Private Limited |
| Sunarin capsules | Anal fissures, piles, rectal inflammation, congestion | SG Phyto Pharma |
| Dr Willmar Schwabe India Acorus calamus mother tincture | Intestinal worms and stomach disorders, fever, nausea | Dr Willmar Schwabe India Pvt Ltd. |
| Himalayan calamus root essential oil | Pain relief and calm mind | Naturalis Essence of Nature |
| Calamus oil | Body, skin care, hair growth | Kazima Perfumers |
| Calamus root powder | Mental health problems | Heilen Biopharm |
| Winton tablets and syrup | Reduce tension, stress, and anxiety | Scortis Healthcare |
| Chesol syrup | Muscular aches and pains, chest colds, and bronchitis | J & J Dechane Laboratories Private Limited |
| Enzo Fast | Acidity, gastritis, flatulence, indigestion | Naturava |
| Dark Forest Vekhand powder | Abdomen pain, worms (infants) | Simandhar Herbal Pvt. Ltd. |
| Nervocare | Insomnia | Deep Ayurveda |
| Antress tablets | Anxiety and stress disorders | Ayursun Pharma |
| Grapzone syrup | Mental wellness | Alna Biotech Pvt Ltd. |
| Memoctive syrup | Improves memory power | Aayursh Herbal India |
| Smrutihills capsules | Stress, anxiety, adaptogenic | Ayush Arogyam |
| Gastrin capsules | Gastritis, dyspepsia | Sarvana Marundhagam |
| Pigmento tablets | Leukoderma or vitiligo | Charak Pharma |
| Paedritone drops | Digestive functions | |
| Vacha Churna | Brain tonic, digestion, nausea | Sadvaidyasala |
| Alert capsules | Immunomodulator, anxiety | Vasu Healthcare |
| Brento tablets | Increasing cognitive functions | Zandu Realty Limited |
| Livotrit Forte | Hepatitis, jaundice | |
| Zanduzyme | Indigestion and dyspepsia | |
| Vedic Slim | Anti-obesity | Vedic Bio-Labs Pvt. Ltd. |
| Hinguvachaadi Gulika | Anorexia, indigestion, appetite loss | Nagarajuna Pvt. Ltd. |
| Nilsin capsules | Sinusitis and allergic rhinitis | Phytomarketing |
| Norbeepee tablet | Hypertension | AVN Formulations |
| Sooktyn tablet | Antacid, antispasmodic | Alarsin Pharma Pvt. Ltd. |
| Deonac oil | Pain reliving oil | Doux Healthcare Pvt. Ltd. |
| Smrutisagar Rasa | Memory enhancer | Shree Dhootpapeshwar Limited |
| Yogaraj Guggul | Vitiligo, anorexia, indigestion, loss of appetite | |
| Kankayan Bati | Gastritis, flatulence, dyspepsia | Baidyanath Pvt. Ltd. |
| Brahmi Ghrita | Insanity and memory issues | |
| Fat Go | Controls high cholesterol level | Jolly Healthcare |
| Divya Medha Vati | Improves memory power | Patanjali Ayurveda |
| Divya Mukta Vati | High blood pressure |
| Classification | Compound No. | Chemical Ingredient | Methods of Characterization | Parts/Extract | References |
|---|---|---|---|---|---|
| Phenylpropanoids | 1 | α-Asarone | GC-FID, GC-MS | Rhizomes/n-hexane, aqueous, methanol, ethanol | [74,78,84,89,90,91] |
| 2 | β-Asarone | ||||
| 3 | γ-Asarone | ||||
| 4 | Eugenyl acetate | GC-MS | Rhizomes/aqueous extract | [74,78,91] | |
| 5 | Eugenol | ||||
| 6 | Isoeugenol | ||||
| 7 | Methyl eugenol | Rhizomes/n-hexane, ethyl acetate | [92] | ||
| 8 | Methyl isoeugenol | Rhizomes/hexane | [74,78,91,94] | ||
| 9 | Calamol | Rhizomes/aqueous extract | [74,78,91] | ||
| 10 | Azulene | ||||
| 11 | Eugenol methyl ether | ||||
| 12 | Dipentene | ||||
| 13 | Asaronaldehyde | ||||
| 14 | Terpinolene | ||||
| 15 | 1,8-cineole | ||||
| 16 | (E)-isoeugenol acetate | GC-FID, GC-MS | [89] | ||
| 17 | (E)-methyl isoeugenol | ||||
| 18 | Cis-methyl isoeugenol | Rhizomes/n-hexane, ethyl acetate | [92] | ||
| 19 | Euasarone | ||||
| 20 | Cinnamaldehyde | ||||
| 21 | Cyclohexanone | GC-MS | Rhizomes/hexane | [94] | |
| 22 | Acorin | NMR | Rhizomes/chloroform | [95] | |
| 23 | Isoasarone | ||||
| 24 | Safrole | ||||
| 25 | Z-3-(2,4,5-trimethoxyphenyl)-2-propenal | FTIR, NMR | Rhizomes/ethanol | [96] | |
| 26 | 2,3-dihydro-4,5,7-trimethoxy-1-ethyl-2-methyl-3 (2,4,5-trimethoxyphenyl) indene | ||||
| 27 | (Z)-asarone | GC-MS | Leaves/n-hexane | [97] | |
| 28 | (E)-caryophyllene | ||||
| 29 | Estragole | Rhizomes/aqueous | [98] | ||
| 30 | Carvacrol | ||||
| 31 | 2-cyclohexane-1-one | ||||
| 32 | Naphthalene | ||||
| 33 | γ-Cadinene | ||||
| 34 | Aristolene | ||||
| 35 | 1(5),3-aromadenedradiene | ||||
| 36 | 5-n-butyltetraline | ||||
| 37 | 4,5-dehydro-isolongifolene | ||||
| 38 | Calarene | ||||
| 39 | Isohomogenol | ||||
| 40 | Zingiberene | ||||
| 41 | α-Calacorene | ||||
| 42 | 5,8-dimethyl isoquinoline | ||||
| 43 | Cyclohexane methanol | ||||
| 44 | Longifolene | ||||
| 45 | Isoelemicin | ||||
| Sesquiterpenoids | 46 | Calamene | [74,78,91] | ||
| 47 | Calamenenol | ||||
| 48 | Calameone | ||||
| 49 | Preisocalamendiol | ||||
| 50 | 1,4-(trans)1,7(trans)-acorenone | [93] | |||
| 51 | 1,4-(cis)-1,7-(trans)-acorenone | ||||
| 52 | 2,6 diepishyobunone | ||||
| 53 | α-Gurjunene | ||||
| 54 | β-Gurjunene | ||||
| 55 | α-Cedrene | [98] | |||
| 56 | β-Elemene | ||||
| 57 | β-Cedrene | [93] | |||
| 58 | β-Caryophyllene | ||||
| 59 | Valencene | ||||
| 60 | Viridiflorene | ||||
| 61 | α-Selinene | GC-FID, GC-MS | [89,93] | ||
| 62 | δ-Cadinene | GC-MS | [93] | ||
| 63 | α-Curcumene | ||||
| 64 | Shyobunone | [84,93,99,100] | |||
| 65 | Isoshyobunone | [93,99,101] | |||
| 66 | Caryophyllene oxide | [93] | |||
| 67 | Humulene oxide II | GC-FID, GC-MS | [89,93] | ||
| 68 | Elemol | GC-MS | [93] | ||
| 69 | Cedrol | ||||
| 70 | Spathulenol | ||||
| 71 | Acorenone | ||||
| 72 | α-Cadinol | ||||
| 73 | Humulene epoxide II | GC-FID, GC-MS | [89] | ||
| 74 | α-Bisabolol | ||||
| 75 | Asaronaldehyde | NMR | Rhizomes/chloroform | [95] | |
| 76 | Calamusenone | GLC, IR, NMR | Rhizomes/petroleum ether | [99] | |
| 77 | Isocalamendiol | ||||
| 78 | Dehydroxyiso-calamendiol | ||||
| 79 | Epishyobunone | ||||
| 80 | Acorone | NMR | Rhizomes/hydro alcoholic | [100] | |
| 81 | Neo-acorane A | Rhizomes/ethanol | [102] | ||
| 82 | Acoric acid | ||||
| 83 | Calamusin D | ||||
| 84 | 1β,5α-Guaiane-4β,10α-diol-6-one | [103] | |||
| 85 | Dioxosarcoguaiacol | HPLC | Rhizomes/petroleum ether | [101] | |
| 86 | 7-tetracycloundecanol,4,4,11,11-tetramethyl | GC-MS | Rhizomes/ethanol | [84] | |
| 87 | 4α,7-Methano-4α-naphth[1,8a-b] oxirene, | ||||
| 88 | Spathulenol | Rhizomes/aqueous | [98] | ||
| 89 | Vulgarol B | ||||
| 90 | Tatanan A | HPLC, NMR | Rhizomes/95% ethanol | [104] | |
| 91 | Acoramone | ||||
| 92 | 2-hydroxyacorenone | ||||
| 93 | 4-(2-formyl-5-methoxymethyl pyrrol-1-yl) butyric acid methyl ester | ||||
| 94 | 2-acetoxyacorenone | ||||
| 95 | Acoramol | ||||
| 96 | N-transferuloyl tyramine | ||||
| 97 | Tatarinoid A | ||||
| 98 | Tatarinoid B | ||||
| 99 | Acortatarin A | ||||
| Monoterpenes | 100 | α-Pinene | GC-MS | Rhizomes, roots/aqueous | [74,78,91,93] |
| 101 | β-Pinene | ||||
| 102 | Camphene | [74,78,91,93,98] | |||
| 103 | o-Cymol | [98] | |||
| 104 | p-Cymene | GC-FID, GC-MS | [89,93,98] | ||
| 105 | γ-Terpinene | GC-MS | [98] | ||
| 106 | α-Terpinolene | ||||
| 107 | Anethole | ||||
| 108 | Thymol | ||||
| 109 | Isoaromadendrene epoxide | ||||
| 110 | Camphor | Rhizome, leaves, roots/aqueous, hexane | [93,97] | ||
| 111 | Sabinene | Roots/aqueous | [93] | ||
| 112 | 2-hexenal | ||||
| 113 | Limonene | [93,98] | |||
| 114 | Cis-linaloloxide | [93] | |||
| 115 | Cis-sabinene hydrate | ||||
| 116 | Trans-linalol oxide | ||||
| 117 | Linalool | [93,97] | |||
| 118 | Terpinen-4-ol | [93] | |||
| 119 | α-Acoradiene | ||||
| 120 | β-Acoradiene | ||||
| 121 | α-Terpineol | ||||
| 122 | Isoborneol | Leaves/hexane | [97] | ||
| Xanthone glycosides | 123 | 4,5,8-trimethoxy-xanthone-2-O-β-D-glucopyranosyl (1-2)-O-β-D-galactopyranoside | NMR | Rhizome/ethanol | [83] |
| Triterpenoid saponins | 124 | 1β,2α,3β, 19α-Tetrahydroxyurs-12-en-28-oic acid-28-O- {(β-D-glucopyranosyl (1-2)}-β-D galactopyranoside | [82] | ||
| 125 | 3-β, 22-α-24,29-Tetrahydroxyolean-12-en-3-O-(β-Darabinosyl (1,3)}-β-D-arabinopyranoside | ||||
| Alkaloids | 126 | Trimethoxyamphetamine,2,3,5 | GC-MS | [84] | |
| 127 | Pyrimidin-2-one,4-[N-methylureido]-1-[4methyl amino carbonloxy methy] | ||||
| Triterpene glycoside | 128 | 22-[(6-deoxy-α-L-rhamnopyranosyl) oxy]-3,23-dihydroxy-, methyl ester, (3β,4β,20α,22β) | NMR | Root, Rhizomes/ethyl acetate | [85] |
| Steroids/Sterols | 129 | β-daucosterol | |||
| Amino acids | 130 | Arginine | HPLC | Roots/ethanol | [86,87] |
| 131 | Lysine | ||||
| 132 | Phenylalanine | ||||
| 133 | Threonine | ||||
| 134 | Tryptophan | ||||
| 135 | α-alanine | ||||
| 136 | Asparagine | ||||
| 137 | Aspartic acid | ||||
| 138 | Norvaline | ||||
| 139 | Proline | ||||
| 140 | Tyrosine | ||||
| 141 | Glutamic acid | ||||
| Fatty acids | 142 | Palmitic acid | GLC | Rhizome/petroleum ether | [88] |
| 143 | Myristic acid | ||||
| 144 | Palmitoleic acid | ||||
| 145 | Stearic acid |
| Action | Parts of Plant | Extract/Compound | Animal Model | Dosage | Results | References |
|---|---|---|---|---|---|---|
| Antidiabetic effects | Rhizomes | Methanol | STZ-induced | 50, 100, and 200 mg/kg, p.o. to rats | ↓ Lipid profile and blood glucose, while ↑ levels of plasma insulin, tissue glycogen, and G6PD | [105] |
| Alloxan-induced | 150 and 200 mg/kg, p.o. to rat | ↓ Blood glucose level | [106] | |||
| Ethyl acetate | Genetically obese diabetic C57BL/Ks db/db mice | 100 mg/kg, p.o. | ↓ Levels of triglycerides and serum glucose | [107] | ||
| GLP-1 expression and secretion with STZ-induced | 100 mg/kg, i.g. | ↑ Secretion of GLP-1 and ↓ blood glucose levels | [108] | |||
| In vitro HIT-T15 cell line and alpha-glucosidase enzyme | 6.25, 12.5, and 25 µg/mL | ↑ Insulin secretion in HIT-T15 cells | [109] | |||
| Glucose tolerance | 400 and 800 mg/kg, p.o. to mice | ↓ Serum glucose, and abolished the ↑ level of blood glucose | ||||
| Anti-obesity effects | Ethanol and aqueous | HFD-induced | 100 and 200 mg/kg to rats | ↓ Levels of serum cholesterol and triglycerides, ↑ lipoprotein fraction | [110] | |
| Diethyl ether | HFD-induced | 20 and 40 mg/kg, p.o. to rats | ↓ Total cholesterol and low-density lipoprotein levels, ↑ plasma fibrinogen levels | [111] | ||
| Methanol | Triton-X-100-induced hyperlipidemic | 250 and 500 mg/kg to rats | Dose-dependent anti-hyperlipidemic effect | [112] | ||
| HFD-induced | 250 and 500 mg/kg, p.o. to rats | ↓ Level of total cholesterol, triglycerides, and LDL, ↑ HDL cholesterol | [113] | |||
| Aqueous | HFD-induced | 100, 200, and 300 mg/kg, p.o. to rats | ↓ Levels of serum glucose, leptin, and insulin along with ↓ triglyceride, low-density lipoprotein, very LDL cholesterol, total cholesterol, phospholipids, and free fatty acid increased levels | [114] | ||
| Antihypertensive effects | Ethyl acetate | Clamping the left kidney artery for 4 h | 250 mg/kg, p.o. to rats | ↓SBP and DBP, blood urea nitrogen, creatinine and LPO, ↑ level of nitric oxide, SOD, CAT, GPX | [115] | |
| Crude extract, ethyl acetate and n-hexane | Blood pressure lowering effect in normotensive | 10, 30, and 50 mg/kg to anesthetized rats | Relaxant effects mediated through Ca+2 antagonism and NO pathways | [116] | ||
| Ethanol and α-asarone | Dimethyl sulfoxide-induced noise stress to rats | 100 and 9 mg/kg, p.o. to rats | ↓ Destructive effect of stress enlightening the morphological changes of hippocampus | [117] | ||
| Anti-inflammatory effects | Leaves | Ethanol | Carrageenan-induced paw edema | 100 and 200 mg/kg to rats | ↓ Histamine, 5-HT, and kinins | [118] |
| Antioxidant effects | Rhizomes | α-asarone | Noise stress induced to rats | 3, 6, and 9 mg/kg, i.p. to rats | ↑ SOD and LPO, decreased ↓ CAT, GPX, GSH, vitamins C and E, and protein thiol levels | [119] |
| Leaves and rhizomes | Ethyl acetate and methanol | DPPH radical scavenging chelating ferrous ions, FRAP | 200, 100, 80, 60, 40, 20, 10, and 5 μg/mL | Prominent DPPH scavenging activity, chelating ferrous ions, and reducingpower | [120,121] | |
| Rhizomes | Ethanol | Acetaminophen-induced | 250, 500 mg/kg, p.o. to rats | ↓ MDA and ↑ SOD, CAT, GPX, GSH levels | [122] | |
| Anticonvulsant effects | Roots | Ethanol and β-asarone | Kainic acid-induced convulsion | 35 and 20 mg/kg | ↓ Epileptic seizure, neuroprotective, and regenerative ability | [123] |
| Methanol | PTZ-induced convulsion | 100 and 200 mg/kg, p.o. to mice | ↑ Latency period and ↓ PTZ-induced seizure time | [124] | ||
| Rhizomes | Calamus oil | MES, PTZ, and MCS model | 30, 100, and 300 mg/kg, p.o. to mice | Calamus oil is found stable | [125] | |
| Ethanol | MES and PTZ-induced convulsion | 250, 500 mg/kg, p.o. to mice | ↓ Hind limb extension and tonic flexion of forelimbs | [126] | ||
| Methanol | MES and PTZ-induced | 250 and 150 mg/kg, p.o. to rats | ↓ Immobility time at 250 mg/kg; however, ineffective at 150 mg/kg | [127] | ||
| Antidepressant effects | TST and FST | 50 and 100 mg/kg, i.p. to mice | ↓ Immobility time in a dose-dependent manner | [128] | ||
| Leaves | TST and FST | 50 and 100 mg/kg | ↓ Immobility time | [129] | ||
| Roots | Aqueous | TST and FST | 100, 150, 200 mg/kg, p.o. to mice | ↓ Immobility time | [130] | |
| Rhizomes | Hydro-alcoholic extract | TST and FST | 75 and 150 mg/kg, p.o. to mice | ↓ Corticosteroid levels | [131] | |
| Ethanol | OFB and HPM test | 72 mg/kg, p.o. | No stimulation of postsynaptic 5-HT1A receptors | [132] | ||
| Methanol and acetone | Behavioral despair test | 5, 20, and 50 mg/kg, p.o. | ↓ Spontaneous locomotor activity | [133] | ||
| β-asarone | EPM and FST | 25, 50, and 100 mg/kg, p.o. | ↓ Immobility time | [134] | ||
| Neuroprotective effects | Hydro-alcoholic | CCI of sciatic nerve-induced neuropathic pain | 10 mg/kg to rats | Significantly ameliorated CCI-induced nociceptive pain | [135] | |
| CCI of sciatic nerve-induced peripheral neuropathy | 100 and 200 mg/kg to rats | Prevented CCI-induced neuropathy through ↓ oxidation and inflammation | [136] | |||
| Leaves | Methanol and acetone | Apomorphine-induced stereotypy and haloperidol-induced catalepsy | 20 and 50 mg/kg to mice | Reversed stereotypy induced by apomorphine and significantly potentiated catalepsy induced by haloperidol | [137] | |
| Rhizomes | Ethanol | Spontaneous electrical activity and monoamine levels of the brain | 200 and 300 mg/ kg to rats | Depressive response by altering electrical activity, including changing brain monoamine levels | [138] | |
| Hydro-alcoholic | MCAo-produced brain ischemia | 25 mg/kg to rats | Improvement in neurobehavioral performance, ↓ levels of GSH, SOD, and ↑ LPO level | [139] | ||
| Ethanol | Methotrexate-induced stress | 5, 10, 15, 20, 25 ppm concentration to fruit flies | ↓ Elevated ROS, SOD, CAT, and GPX levels | [140] | ||
| Cardioprotective effects | Whole plant | DOX-induced myocardial toxicity | 100 and 200 mg/kg to rats | ↓ Serum enzyme levels and protected the myocardium from the toxic effect of DOX | [141] | |
| Rhizomes | Crude, n-hexane, ethyl acetate | Guinea pig tracheal segments | 0.01 mg/mL | ↓ Force and rate of contractions at higher concentrations | [142] |
| Formulations/Dosage forms A. calamus | Subjects | Study Design | Intervention | Primary Endpoint | Outcome | Evidence Quality | Reference |
|---|---|---|---|---|---|---|---|
| A. calamus rhizome powder | 24 patients of both sexes with hyperlipidemia | Randomized single-blind controlled study | 500 mg twice daily after meal for 1 month | BMI, body perimeter, skinfold depth | Significant reduction in skinfold depth, fatigue, and excessive hunger | III | [178] |
| Davaie Loban capsules (A. calamus, nut grass, incense, ginger, and black pepper) | 24 patients of both sexes with Alzheimer’s disease | Double-blind randomized clinical study | 500 mg capsule thrice daily for 3 months | ADAS-cog and CDR-SOB scores | At 4 weeks and 12 weeks: significant reduction in the ADAS-cog and CDR-SOB scores | III | [179] |
| 70% hydro-alcoholic extract of A. calamus | 33 patients of both sexes (20 male and 13 female) with anxiety disorder | Non-randomized, open-label, single-arm study | 500 mg extract of one capsule twice daily after meal for 2 months | BPRS score | Significant reduction of anxiety and stress-related disorder | III | [180] |
| Vachadi Churna (A. calamus, Cyperus rotundus, Cedrus deodara, ginger, Aconitum Heterophyllum, T. chebula) | 30 obese patients of both sexes aged 14–50 years | Non-randomized, open-label, single-arm study | 3 g powder twice daily with lukewarm water before meal for 1 month | BMI, girth measurements of mid-thigh, abdomen, hip, chest | Significant improvement in extreme sleep, body heaviness, fatigue, and excessive hunger | III | [181] |
| Guduchyadi Medhya Rasayana, (A. calamus, Tinospora cordifolia, Achyranthes aspera, Embelia ribes, Convolvulus pluricaulis, T. chebula, S. lappa, Asparagus racemosus, cow ghee, and sugar) | 138 patients of both sexes aged 55–75 years with senile memory impairment | Randomized, two-parallel-group study | 3 g granule thrice daily after meal for 3 months | Mini–Mental State Examination, BPRS score, and estimation of serum acetylcholinesterase | Significant improvement in terms of recall memory, cognitive impairment, amnesia, concentration ability, depression, and stress | III | [182] |
| Dried aqueous extract of A. calamus | 40 healthy volunteers, both sexes aged 18–50 years with a premedicant for anesthesia | Open-label randomized, two- parallel-group study | 90 min before anesthesia; In the control group: 0.2 mg intramuscular (IM) glycopyrrolate and a 0.2 mg IM 50 mg tablet of promethazine hydrochloride with water; In the second group: 0.2 mg IM glycopyrrolate and 100 mg A. calamus extract | Pulse rate, blood pressure, respiratory rate, body temperature | The dried aqueous extract exhibited anti-hyperthermic and sedative effect without producing any respiratory depression | III | [183] |
| Shankhapushpyadi Ghana Vati (A. calamus, C. pluricaulis, Bacopa monnieri, T. cordifolia, C. fistula, A. indica, S. lappa, Tribulus terrestris) | 20 hypertensive patients of both sexes | Randomized single-blind controlled study | 1 g twice daily after meal for 2 months | SBP and DBP | Significant relief in raised SBP and DBP | III | [184] |
| Brahmyadiyoga (A. calamus, Centella asiatica, Rauvolfia serpentina, Saussurea lappa, Nardostachys jatamansi) | 10 schizophrenia patients of both sexes aged 18–40 years | Non-randomized, open-label, single- arm study | 4 tablets thrice daily for three months after meal | Symptoms rating scale | Significant effect as a brain tonic, tranquillizer, hypnotic, and sedative | III | [185] |
| Bala compound (A. calamus, Emblica officinalis, E. ribes, T. cordifolia, Piper longum, Glycyrrhiza glabra, C. rotundus, A. heterophyllum) | 24 neonates, both sexes, 2.5–3 kg body weight | Randomized single-blind controlled study | 5 oral drops twice daily for 6 months | Change in serum immunoglobulins (IgG, IgM, and IgA) levels | Significant improvement in immunoglobulin levels after 6 months | Ib | [186] |
| Vachadi Ghrita (A. calamus, T. cordifolia, Hedychium spicatum, C. pluricaulis, E. ribes, ginger, A. aspera, T. chebula, and cow ghee) | 90 healthy individuals of both sexes aged 40–50 years for assessment of cognition | Non-randomized positive-controlled study | 10 g twice daily for 1 month with lukewarm water | Post Graduate Institute Memory Scale (PGIMS) test | Significant change in the mental balance score, holding of like and different pairs, late-immediate memory, and also improved digestion | III | [187] |
| Bramhi Vati (A. calamus, B. monnieri, C. pluricaulis, Onosma bracteatum, copper pyrite, iron pyrite, mercuric sulphide, Piper nigrum, N. jatamansi) | 68 essential hypertension patients of both sexes aged 20–70 years | Randomized, double-blind, parallel-group comparative study | 500 mg tablets twice daily for 1 month | Hamilton anxiety rating scale, SBP and DBP, and MAP | Significant improvement in the Hamilton anxiety rating scale, SBP and DBP, and MAP | III | [188] |
| Tagaradi Yoga (A. calamus, Valeriana wallichii, N. jatamansi) | 24 insomnia patients of both sexes aged 18–75 years | Non-randomized positive-controlled study | 500 mg hydro-alcoholic extract capsule twice daily after meal for 15 days | Sleep duration, initiating time of sleep, quality of sleep | Significant improvement in sleep duration, in the initiating time of sleep, and in quality of sleep | III | [189] |
| Acorus calamus rhizome powder | 20 obese patients of both sexes | Randomized single-blind study | 250 mg rhizome powder twice daily for 1 month | Body weight, height according to age, waist-hip ratio, and BMI | Significant improvement in extreme sleep, body heaviness, fatigue, and excessive hunger | III | [190] |
| Acorus calamus rhizome powder | 45 ischemic heart disease patients | Non-randomized positive-controlled study | 3 gm rhizome powder twice daily for 3 months | ECG, serum cholesterol level | Improvement of chest pain, dyspnea on effort, reduction of the body mass index, improved ECG: reduced serum cholesterol, reduced serum LDL, and increased serum HDL | Ib | [191] |
| Study | Compound | Model | Increased Level | Decreased Level | References |
|---|---|---|---|---|---|
| Anti-Parkinson | β-Asarone | 6-OHDA parkinsonian | Bcl-2 expression | GRP78, p-PERK, CHOP, and Beclin-1 expression | [192] |
| 6-OHDA parkinsonian | - | mRNA levels of GRP78 and CHOP and p-IRE1and XBP1 | [193] | ||
| Dopamine in the striatum | TH plasma concentrations | Striatal COMT levels | [194] | ||
| 6-OHDA parkinsonian | L-DOPA, DA, DOPAC, and HVA levels | P-gp, ZO-1, occludin, actin, and claudin-5 | [195] | ||
| Alzheimer’s | Aβ25-35-induced inflammation | Bcl-2 level | TNF-α, IL-1β, IL-6, Beclin-1, and LC3B level | [196] | |
| NG108 cells | - | Upregulated SYP and GluR1 expression | [197] | ||
| PC12 cells | - | Aβ-induced JNK activation, Bcl-w and Bcl-xL levels, cytochrome c release, and caspase-3 activation | [198] | ||
| Aβ-induced cytotoxicity | Cell viability, p-Akt and p-mTOR | NSE levels, Beclin-1 expression | [199] | ||
| Neuroprotective | Pb-induced impairments | NR2B protein expression along with Arc/Arg3.1 and Wnt7a mRNA levels | - | [200] | |
| β-Asarone, eugenol | Scopolamine-induced | Improvement of neuron organelles and synaptic structure | APP expression | [201] | |
| Neotatarine | MTT reduction assay | - | Aβ25-35–induced PC12 cell death | [202] | |
| β-asarone, paeonol | MCAo model | Cholecystokinin and NF-κB signaling | TNF-α, IL-1β, IL-6 production | [203] | |
| β-Asarone | Cultured rat astrocytes | NGF, BDNF, and GDNF expression | - | [204] | |
| SN4741 cells | p62, Bcl-2 expression | JNK, p-JNK and Beclin-1 expressions | [205] | ||
| Tatarinolactone | hSERT-HEK293 cell line | - | SERTs activity | [206] | |
| β-Asarone | RSC96 Schwann cells | GDNF, BDNF, and CNTF expression | - | [207] | |
| Aβ-induced | p-mTOR and p62 expression | AChE and Aβ42 levels, p-Akt, Beclin-1, and LC3B expression, APP mRNA and Beclin-1 mRNA levels | [208] | ||
| Aβ1–42-induced injury | - | GFAP, AQP4, IL-1β, and TNF-α expression | [209] | ||
| Anti-depression | Chronic unpredictable mild stress | BDNF expression | Blocked ERK1/2-CREB signaling | [210] | |
| α-Asarone | Noradrenergic and serotonergic neuromodulators in TST | α1 and α2 adrenoceptors and 5-HT1A receptors | - | [211] | |
| Anticonvulsant and sedative | Eudesmin | MES and PTZ | GABA contents, expressions of GAD65, GABAA, and Bcl-2 | Glu contents and ratio of Glu/GABA, caspase-3 | [212] |
| Anti-anxiety | α-Asarone | BLA or CFA-induced | Down-regulation of GABAA receptors | Up-regulation of GluR1-containing AMPA, NMDA receptors | [213] |
| Anti-epilepsy | Temporal lobe epilepsy | Levels of GABA, GAD67, and GABAAR-mRNA expression | GABA-T | [214] | |
| Mitral cells | Down-regulation of GABAA receptors | Na+ channel blockade | [215] | ||
| β-Asarone | KA-induced | GABA | Glu | [216] | |
| Anti-inflammatory | α-Asarone | Spinal cord injury | IL-4, IL-10, and arginase 1 levels | TNF-α, IL-1β, IL-6, MCP-1, MIP-2, iNOS levels | [217] |
| Cytoprotective | β-Asarone | tBHP-induced astrocyte injury | GST, GCLM, GCLC, NQO1, Akt phosphorylation | - | [218] |
| Cardioprotective | Cultured neonate rat cardiac myocytes | Viability of cardiac myocytes | Pulse frequency | [219] | |
| Arteriosclerosis | ECV304 cell strain | Apoptotic rate of ECV304 cells | Apoptotic rate of MMP, stabilized MMP and VSMC proliferation | [220] | |
| Anti-adipogenic | 3T3-L1 preadipocytes | - | C/EBPβ, C/EBPα, and PPARγ expression levels, ERK1/2 phosphorylation | [89] | |
| Antioxidant | Cerebral artery occlusion | Antioxidant activity | Focal cerebral ischemic/reperfusion injury | [221] | |
| Anti-diabetic | α-Asarone + β-asarone + metformin HCl | STZ-induced | Insulin level | Glucose, glycosylated hemoglobin level, liver dysfunction, and tumor biomarkers | [222] |
| Asarone | 3T3-L1 preadipocytes | Hormone-sensitive lipase phosphorylation | Intracellular triglyceride levels, down-regulation of PPARγ and C/EBPα | [223] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sharma, V.; Sharma, R.; Gautam, D.S.; Kuca, K.; Nepovimova, E.; Martins, N. Role of Vacha (Acorus calamus Linn.) in Neurological and Metabolic Disorders: Evidence from Ethnopharmacology, Phytochemistry, Pharmacology and Clinical Study. J. Clin. Med. 2020, 9, 1176. https://doi.org/10.3390/jcm9041176
Sharma V, Sharma R, Gautam DS, Kuca K, Nepovimova E, Martins N. Role of Vacha (Acorus calamus Linn.) in Neurological and Metabolic Disorders: Evidence from Ethnopharmacology, Phytochemistry, Pharmacology and Clinical Study. Journal of Clinical Medicine. 2020; 9(4):1176. https://doi.org/10.3390/jcm9041176
Chicago/Turabian StyleSharma, Vineet, Rohit Sharma, DevNath Singh Gautam, Kamil Kuca, Eugenie Nepovimova, and Natália Martins. 2020. "Role of Vacha (Acorus calamus Linn.) in Neurological and Metabolic Disorders: Evidence from Ethnopharmacology, Phytochemistry, Pharmacology and Clinical Study" Journal of Clinical Medicine 9, no. 4: 1176. https://doi.org/10.3390/jcm9041176
APA StyleSharma, V., Sharma, R., Gautam, D. S., Kuca, K., Nepovimova, E., & Martins, N. (2020). Role of Vacha (Acorus calamus Linn.) in Neurological and Metabolic Disorders: Evidence from Ethnopharmacology, Phytochemistry, Pharmacology and Clinical Study. Journal of Clinical Medicine, 9(4), 1176. https://doi.org/10.3390/jcm9041176









