Treatment of Rheumatoid Arthritis-Associated Interstitial Lung Disease: Lights and Shadows
Abstract
1. Introduction
2. Immunosuppressants
2.1. Corticosteroids
2.2. Cyclophosphamide and Mycophenolate Mofetil
3. Conventional Disease-Modifying Antirheumatic Drugs (cDMARDs)
3.1. Methotrexate
3.2. Leflunomide
3.3. Azathioprine
3.4. Sulfasalazine, Hydroxychloroquine, and Penicillamine
3.5. Calcineurin Inhibitors (Cyclosporin, Tacrolimus)
4. Biological Disease-Modifying Antirheumatic Drugs (bDAMRDs)
4.1. Tumour Necrosis Factor Alpha Inhibitors
4.1.1. Infliximab
4.1.2. Adalimumab
4.1.3. Etanercept
4.1.4. Golimumab and Certolizumab
4.2. Abatacept
4.3. Interleukin-6 Inhibitors
4.3.1. Tocilizumab
4.3.2. Sarilumab
4.4. Rituximab
5. Targeted Synthetic Disease-Modifying Antirheumatic Drugs (tsDMARDs)
6. Antifibrotic Agents
6.1. Pirfenidone
6.2. Nintedanib
7. Conservative Therapy
7.1. Smoking Cessation
7.2. Pulmonary Rehabilitation
7.3. Oxygen Supplementation
7.4. Vaccination
7.5. Comorbidities
8. Lung Transplant
9. Acute Exacerbation of Ra-Ild
10. Biomarkers and Response to Treatment
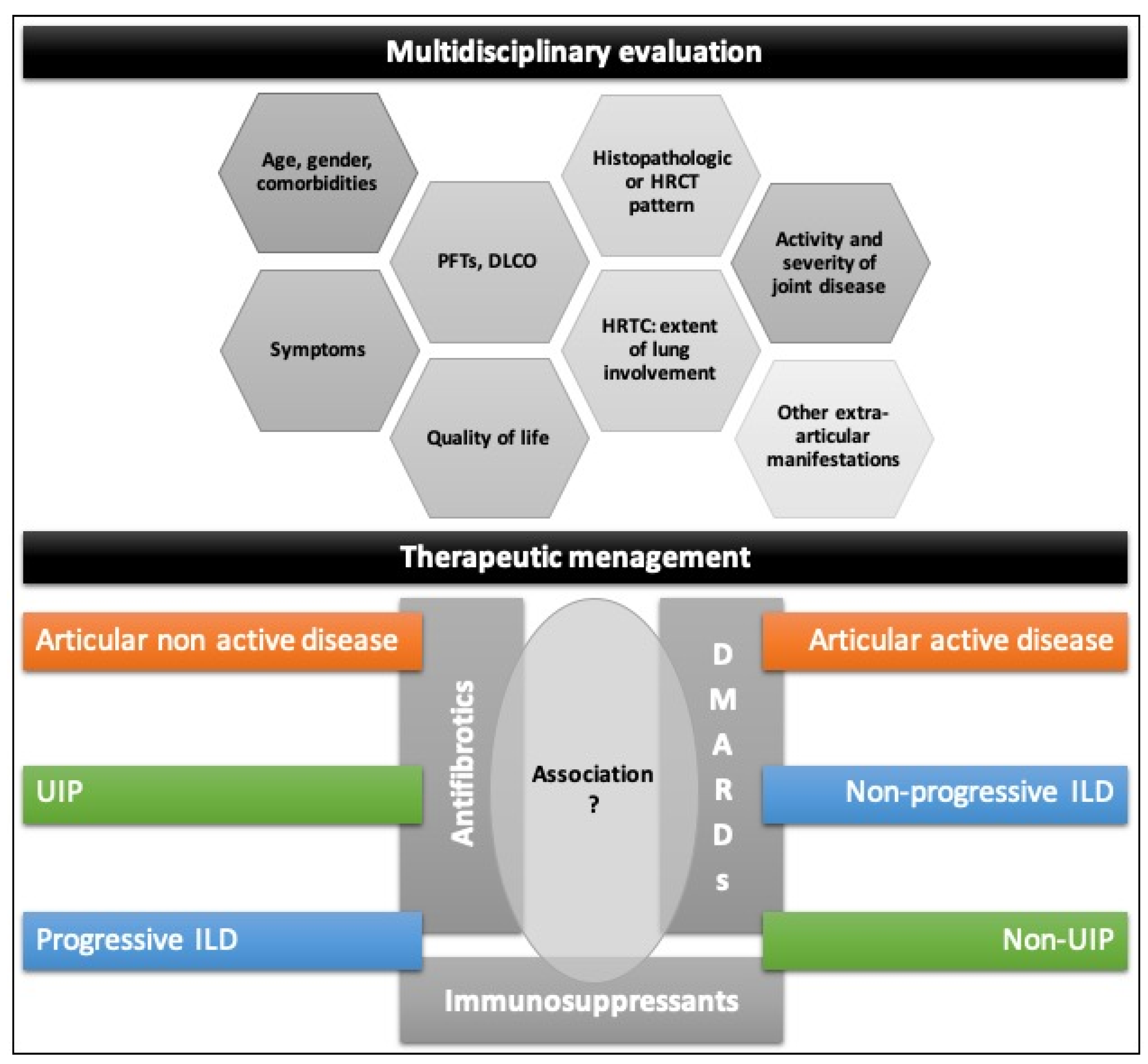
11. Proposal for Patient Management and Treatment
- Therefore, the development of guidelines for RA-ILD treatment remains an open challenge.
- The treatment of RA-associated ILD should be tailored for each patient after the evaluation of:
- -
- age, gender, comorbidities;
- -
- progression and severity of the lung involvement (symptoms, PFTs, DLCO, HRCT);
- -
- histopathologic or HRCT pattern of ILD;
- -
- activity and severity of joint disease;
- -
- other extra-articular manifestations.
12. Conclusions and Research Agenda
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ABA | ABATACEPT |
| AE | Acute exacerbation |
| ANTI-MDA5 | Anti–melanoma differentiation–associated protein 5 |
| AZA | Azathioprine |
| BDMARDS | Biologic disease modifying anti-rheumatic drugs |
| CDMARDS | Conventional disease modifying anti-rheumatic drugs |
| COPD | Chronic obstructive pulmonary disease |
| CTD | Connective tissue diseases |
| CTD-ILD | Connective tissue diseases-realated interstitial lung disease |
| CYC | Cyclophosphamide |
| DLCO | Diffusing capacity of the lungs for carbon monoxide |
| DMARDS | Disease modifying anti-rheumatic drugs |
| FVC | Forced vital capacity |
| HRCT | High-resolution computer tomography |
| ILD | Interstitial lung disease |
| IPF | Idiopathic pulmonary fibrosis |
| JAK | Janus-kinase |
| LEF | Leflunomide |
| MDA5 | Melanoma differentiation–associated protein 5 |
| MDSC | Myeloid-derived suppressor cells |
| MMF | Mycophenolate mofetil |
| MTX | Methotrexate |
| NICE | National Institute for Health and Care Excellence |
| NS-CTLD | Non-scleroderma connective tissue-related lung disease |
| NSIP | Nonspecific interstitial pneumonia |
| OP | Organizing pneumonia |
| PFTS | Pulmonary function tests |
| RA | Rheumatoid arthritis |
| RA-ILD | Rheumatoid arthritis-related interstitial lung disease |
| RTX | Rituximab |
| SSC-ILD | Systemic sclerosis-related interstitial lung disease |
| TCZ | Tocilizumab |
| TNFI | Tumour necrosis factor alpha inhibitor |
| TSDMARDS | Targeted synthetic disease modifying anti-rheumatic drugs |
| UIP | Usual interstitial pneumonia |
References
- Smolen, J.S.; Aletaha, D.; McInnes, I.B. Rheumatoid arthritis. Lancet 2016, 388, 2023–2038. [Google Scholar] [CrossRef]
- Prete, M.; Racanelli, V.; Digiglio, L.; Vacca, A.; Dammacco, F.; Perosa, F. Extra-articular manifestations of rheumatoid arthritis: An update. Autoimmun. Rev. 2011, 11, 123–131. [Google Scholar] [CrossRef]
- Yunt, Z.X.; Solomon, J.J. Lung Disease in Rheumatoid Arthritis. Rheum. Dis. Clin. N. Am. 2015, 41, 225–236. [Google Scholar] [CrossRef]
- Esposito, A.J.; Chu, S.G.; Madan, R.; Doyle, T.J.; Dellaripa, P.F. Thoracic Manifestations of Rheumatoid Arthritis. Clin. Chest Med. 2019, 40, 545–560. [Google Scholar] [CrossRef] [PubMed]
- Bongartz, T.; Nannini, C.; Medina-Velasquez, Y.F.; Achenbach, S.J.; Crowson, C.S.; Ryu, J.; Vassallo, R.; Gabriel, S.E.; Matteson, E.L. Incidence and mortality of interstitial lung disease in rheumatoid arthritis: A population-based study. Arthritis Rheum. 2010, 62, 1583–1591. [Google Scholar] [CrossRef] [PubMed]
- Raimundo, K.; Solomon, J.J.; Olson, A.; Kong, A.M.; Cole, A.L.; Fischer, A.; Swigris, J.J. Rheumatoid Arthritis-Interstitial Lung Disease in the United States: Prevalence, Incidence, and Healthcare Costs and Mortality. J. Rheumatol. 2019, 46, 360–369. [Google Scholar] [CrossRef] [PubMed]
- Hyldgaard, C.; Hilberg, O.; Pedersen, A.B.; Ulrichsen, S.P.; Løkke, A.; Bendstrup, E.; Ellingsen, T. A population-based cohort study of rheumatoid arthritis-associated interstitial lung disease: Comorbidity and mortality. Ann. Rheum. Dis. 2017, 76, 1700–1706. [Google Scholar] [CrossRef]
- Olson, A.; Swigris, J.J.; Sprunger, D.B.; Fischer, A.; Pérez, E.F.; Solomon, J.; Murphy, J.; Cohen, M.; Raghu, G.; Brown, K.K. Rheumatoid arthritis-interstitial lung disease-associated mortality. Am. J. Respir. Crit. Care Med. 2011, 183, 372–378. [Google Scholar] [CrossRef]
- Kelly, C.; Iqbal, K.; Iman-Gutierrez, L.A.; Evans, P.; Manchegowda, K. Lung Involvement in Inflammatory Rheumatic Diseases. Best Pract. Res. Clin. Rheumatol. 2016, 30, 870–888. [Google Scholar] [CrossRef]
- American Thoracic Society/European Respiratory Society. American Thoracic Society/European Respiratory Society international multidisciplinary consensus classification of the idiopathic interstitial pneumonias. Am. J. Respir. Crit. Care Med. 2002, 165, 277–304. [Google Scholar] [CrossRef]
- Travis, W.D.; Costabel, U.; Hansell, D.M.; King, T.E., Jr.; Lynch, D.A.; Nicholson, A.G.; Ryerson, C.J.; Ryu, J.; Selman, M.; Wells, A.U.; et al. ATS/ERS Committee on Idiopathic Interstitial Pneumonias; ATS/ERS Committee on Idiopathic Interstitial Pneumonias. An official American Thoracic Society/European Respiratory Society statement: Update of the international multidisciplinary classification of the idiopathic interstitial pneumonias. Am. J. Respir. Crit. Care Med. 2013, 188, 733–748. [Google Scholar] [CrossRef]
- Balbir-Gurman, A.; Guralnik, L.; Yigla, M.; Braun-Moscovici, J.; Hardak, E. Imaging Aspects of Interstitial Lung Disease in Patients with Rheumatoid Arthritis: Literature Review. Autoimmun. Rev. 2018, 17, 87–93. [Google Scholar] [CrossRef]
- Oliveira, D.S.; Filho, J.D.A.A.; Paiva, A.F.L.; Ikari, E.S.; Chate, R.C.; Nomura, C.H. Idiopathic Interstitial Pneumonias: Review of the Latest American Thoracic Society/European Respiratory Society Classification. Radiol. Bras. 2018, 51, 321–327. [Google Scholar] [CrossRef]
- Assayag, D.; Lee, J.S.; King, T.E., Jr. Rheumatoid arthritis associated interstitial lung disease: A review. Medicina (Buenos Aires) 2014, 74, 158–165. [Google Scholar] [PubMed]
- Duarte, A.C.; Porter, J.C.; Leandro, M.J. The lung in a cohort of rheumatoid arthritis patients-an overview of different types of involvement and treatment. Rheumatology (Oxford) 2019, 58, 2031–2038. [Google Scholar] [CrossRef]
- Hanaka, T.; Kido, T.; Ishimoto, H.; Oda, K.; Noguchi, S.; Nawata, A.; Nakayamada, S.; Sakamoto, N.; Tanaka, Y.; Yatera, K.; et al. Radiological patterns of pulmonary involvement may predict treatment response in rheumatoid arthritis: A retrospective study. Respir. Investig. 2019, 57, 172–182. [Google Scholar] [CrossRef]
- Salaffi, F.; Carotti, M.; Di Carlo, M.; Tardella, M.; Giovagnoni, A. High-resolution computed tomography of the lung in patients with rheumatoid arthritis: Prevalence of interstitial lung disease involvement and determinants of abnormalities. Medicine (Baltimore) 2019, 98, e17088. [Google Scholar] [CrossRef]
- Tanaka, N.; Kunihiro, Y.; Kubo, M.; Kawano, R.; Oishi, K.; Ueda, K.; Gondo, T. HRCT findings of collagen vascular disease-related interstitial pneumonia (CVD-IP): A comparative study among individual underlying diseases. Clin. Radiol. 2018, 73, 833.e1–833.e10. [Google Scholar] [CrossRef]
- Kim, E.J.; Collard, H.R.; King, T.E., Jr. Rheumatoid arthritis-associated interstitial lung disease: The relevance of histopathologic and radiographic pattern. Chest 2009, 136, 1397–1405. [Google Scholar] [CrossRef]
- Brito, J.; Glassberg, M.K.; Ascherman, D.P. Rheumatoid Arthritis-Associated Interstitial Lung Disease: Current Concepts. Curr. Rheumatol. Rep. 2017, 19, 79. [Google Scholar] [CrossRef]
- Spagnolo, P.; Lee, J.S.; Sverzellati, N.; Rossi, G.; Cottin, V. The Lung in Rheumatoid Arthritis: Focus on Interstitial Lung Disease. Arthritis Rheumatol. 2018, 70, 1544–1554. [Google Scholar] [CrossRef]
- Schettino, I.A.; Ab’Saber, A.M.; Vollmer, R.; Saldiva, P.H.N.; Carvalho, C.R.R.; Kairalla, R.; Capelozzi, V.L. Accuracy of high resolution CT in assessing idiopathic pulmonary fibrosis histology by objective morphometric index. Pathol. Res. Pract. 2002, 198, 347–354. [Google Scholar] [CrossRef] [PubMed]
- Jacob, J.; Hirani, N.; Van Moorsel, C.H.M.; Rajagopalan, S.; Murchison, J.T.; Van Es, H.W.; Bartholmai, B.J.; Van Beek, F.T.; Struik, M.H.L.; Stewart, G.A.; et al. Predicting outcomes in rheumatoid arthritis related interstitial lung disease. Eur. Respir. J. 2019, 3, 53. [Google Scholar] [CrossRef]
- Yamakawa, H.; Sato, S.; Tsumiyama, E.; Nishizawa, T.; Kawabe, R.; Oba, T.; Kamikawa, T.; Horikoshi, M.; Akasaka, K.; Amano, M.; et al. Predictive factors of mortality in rheumatoid arthritis-associated interstitial lung disease analysed by modified HRCT classification of idiopathic pulmonary fibrosis according to the 2018 ATS/ERS/JRS/ALAT criteria. J. Thorac. Dis. 2019, 11, 5247–5257. [Google Scholar] [CrossRef]
- Kim, E.J.; Elicker, B.M.; Maldonado, F.; Webb, W.R.; Ryu, J.; Van Uden, J.H.; Lee, J.S.; King, T.E.; Collard, H.R. Usual interstitial pneumonia in rheumatoid arthritis-associated interstitial lung disease. Eur. Respir. J. 2010, 35, 1322–1328. [Google Scholar] [CrossRef]
- Johnson, C. Recent Advances in the Pathogenesis, Prediction, and Management of Rheumatoid Arthritis-Associated Interstitial Lung Disease. Curr. Opin. Rheumatol. 2017, 29, 254–259. [Google Scholar] [CrossRef]
- Paulin, F.; Babini, A.; Mamani, M.; Mercado, J.; Caro, F. Practical Approach to the Evaluation and Management of Rheumatoid Arthritis-Interstitial Lung Disease Based on its Proven and Hypothetical Mechanisms. Rev. Investig. Clin. 2017, 69, 235–242. [Google Scholar] [CrossRef]
- Bes, C. Comprehensive review of current diagnostic and treatment approaches to interstitial lung disease associated with rheumatoid arthritis. Eur. J. Rheumatol. 2018, 6, 146–149. [Google Scholar] [CrossRef]
- Ito, Y.; Arita, M.; Kumagai, S.; Takei, R.; Noyama, M.; Tokioka, F.; Nishimura, K.; Koyama, T.; Tachibana, H.; Ishida, T. Radiological fibrosis score is strongly associated with worse survival in rheumatoid arthritis-related interstitial lung disease. Mod. Rheumatol. 2019, 29, 98–104. [Google Scholar] [CrossRef]
- Nurmi, H.M.; Purokivi, M.K.; Kärkkäinen, M.S.; Kettunen, H.P.; Selander, T.A.; Kaarteenaho, R.L. Variable course of disease of rheumatoid arthritis-associated usual interstitial pneumonia compared to other subtypes. BMC Pulm. Med. 2016, 16, 107. [Google Scholar] [CrossRef]
- Singh, N.; Varghese, J.; England, B.R.; Solomon, J.J.; Michaud, K.; Mikuls, T.R.; Healy, H.; Kimpston, E.M.; Schweizer, M.L. Impact of the pattern of interstitial lung disease on mortality in rheumatoid arthritis: A systematic literature review and meta-analysis. Semin. Arthritis Rheum. 2019, 49, 358–365. [Google Scholar] [CrossRef] [PubMed]
- Kolb, M.; Vašáková, M. The Natural History of Progressive Fibrosing Interstitial Lung Diseases. Respir. Res. 2019, 20, 57. [Google Scholar] [CrossRef]
- Morisset, J.; Lee, J.S. New trajectories in the treatment of interstitial lung disease: Treat the disease or treat the underlying pattern? Curr. Opin. Pulm. Med. 2019, 25, 442–449. [Google Scholar] [CrossRef] [PubMed]
- Roubille, C.; Haraoui, B. Interstitial lung diseases induced or exacerbated by DMARDS and biologic agents in rheumatoid arthritis: A systematic literature review. Semin. Arthritis Rheum. 2014, 43, 613–626. [Google Scholar] [CrossRef] [PubMed]
- Olivas-Flores, E.M.; Bonilla-Lara, D.; Gamez-Nava, J.I.; Rocha-Muñoz, A.D.; Gonzalez-Lopez, L. Interstitial lung disease in rheumatoid arthritis: Current concepts in pathogenesis, diagnosis and therapeutics. World J. Rheumatol. 2015, 5, 1–22. [Google Scholar] [CrossRef]
- Jani, M.; Hirani, N.; Matteson, E.L.; Dixon, W.G. The safety of biologic therapies in RA-associated interstitial lung disease. Nat. Rev. Rheumatol. 2014, 10, 284–294. [Google Scholar] [CrossRef]
- Smolen, J.S.; Landewé, R.; Bijlsma, J.; Burmester, G.; Chatzidionysiou, K.; Dougados, M.; Nam, J.; Ramiro, S.; Voshaar, M.; Van Vollenhoven, R.; et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2016 update. Ann. Rheum. Dis. 2017, 76, 960–977. [Google Scholar] [CrossRef]
- Singh, J.; Saag, K.G.; Bridges, S.L.; Akl, E.A.; Bannuru, R.R.; Sullivan, M.C.; Vaysbrot, E.; McNaughton, C.; Osani, M.; Shmerling, R.; et al. 2015 American College of Rheumatology Guideline for the Treatment of Rheumatoid Arthritis. Arthritis Care Res. (Hoboken) 2016, 68, 1–25. [Google Scholar] [CrossRef]
- Holroyd, C.R.; Seth, R.; Bukhari, M.; Malaviya, A.; Holmes, C.; Curtis, E.; Chan, C.; A Yusuf, M.; Litwic, A.; Smolen, S.; et al. The British Society for Rheumatology Biologic DMARD Safety Guidelines in Inflammatory Arthritis. Rheumatology (Oxford) 2019, 58, e3–e42. [Google Scholar] [CrossRef] [PubMed]
- Balsa Criado A and Grupo GUIPCAR de trabajo de la GPC para el Manejo de Pacientes con Artritis Reumatoide. Guía de Práctica Clínica para el Manejo de los Pacientes con Artritis Reumatoide, actualización 2019. Available online: https://www.ser.es/wp-content/uploads/2019/03/Guia-de-Practica-Clinica-para-el-Manejo-de-Pacientes-con-Artritis-Reumatoide.pdf. (accessed on 22 February 2020).
- Idiopathic Pulmonary Fibrosis Clinical Research Network; Raghu, G.; Anstrom, K.J.; King, T.E., Jr.; Lasky, J.A.; Martinez, F.J. Prednisone, Azathioprine, and N-acetylcysteine for Pulmonary Fibrosis. N. Engl. J. Med. 2012, 366, 1968–1977. [Google Scholar] [CrossRef]
- Tsuchiya, Y.; Takayanagi, N.; Sugiura, H.; Miyahara, Y.; Tokunaga, D.; Kawabata, Y.; Sugita, Y. Lung diseases directly associated with rheumatoid arthritis and their relationship to outcome. Eur. Respir. J. 2011, 37, 1411–1417. [Google Scholar] [CrossRef]
- Vacchi, C.; Sebastiani, M.; Cassone, G.; Cerri, S.; Della Casa, G.; Salvarani, C.; Manfredi, A. Therapeutic Options for the Treatment of Interstitial Lung Disease Related to Connective Tissue Diseases. A Narrative Review. J. Clin. Med. 2020, 9, 407. [Google Scholar] [CrossRef]
- O’Dwyer, D.; Armstrong, M.E.; Cooke, G.; Dodd, J.D.; Veale, D.J.; Donnelly, S.C. Rheumatoid arthritis (RA) associated interstitial lung disease (ILD). Eur. J. Intern. Med. 2013, 24, 597–603. [Google Scholar] [CrossRef]
- Flaherty, K.R.; Travis, W.D.; Colby, T.V.; Toews, G.B.; A Kazerooni, E.; Gross, B.H.; Jain, A.; Strawderman, R.L.; Flint, A.; Lynch, J.P.; et al. Histopathologic variability in usual and nonspecific interstitial pneumonias. Am. J. Respir. Crit. Care Med. 2001, 164, 1722–1727. [Google Scholar] [CrossRef] [PubMed]
- Song, J.W.; Lee, H.K.; Lee, C.K.; Chae, E.J.; Jang, S.J.; Colby, T.V.; Kim, D.S. Clinical course and outcome of rheumatoid arthritis related usual interstitial pneumonia. Sarcoidosis Vasc Diffuse Lung Dis. 2013, 30, 103–112. [Google Scholar] [PubMed]
- Lee, H.K.; Kim, D.S.; Yoo, B.; Seo, J.B.; Rho, J.Y.; Colby, T.V.; Kitaichi, M. Histopathologic pattern and clinical features of rheumatoid arthritis-associated interstitial lung disease. Chest 2005, 127, 2019–2027. [Google Scholar] [CrossRef] [PubMed]
- Yamano, Y.; Taniguchi, H.; Kondoh, Y.; Ando, M.; Kataoka, K.; Furukawa, T.; Johkoh, T.; Fukuoka, J.; Sakamoto, K.; Hasegawa, Y. Multidimensional Improvement in Connective Tissue Disease-Associated Interstitial Lung Disease: Two Courses of Pulse Dose Methylprednisolone Followed by Low-Dose Prednisone and Tacrolimus. Respirology 2018, 23, 1041–1048. [Google Scholar] [CrossRef]
- Zamora-Legoff, J.A.; Krause, M.L.; Crowson, C.D.; Ryu, J.H.; Matteson, E.L. Risk of Serious Infection in Patients with Rheumatoid Arthritis-Associated Interstitial Lung Disease. Clin. Rheumatol. 2016, 35, 2585–2589. [Google Scholar] [CrossRef]
- Sebastiani, M.; Manfredi, A.; Cassone, G.; Sandri, G.; Cerri, S.; Ferri, C. Interstitial lung disease is associated to infections of lower respiratory tract in immunocompromised rheumatoid arthritis patients. Clin. Exp. Rheumatol. 2017, 35, 542. [Google Scholar] [PubMed]
- Tashkin, D.P.; Elashoff, R.; Clements, P.J.; Goldin, J.; Roth, M.D.; Furst, D.E. Cyclophosphamide versus placebo in scleroderma lung disease. N. Engl. J. Med. 2006, 354, 2655–2666. [Google Scholar] [CrossRef]
- Yiannopoulos, G.; Pastromas, V.; Antonopoulos, I.; Katsiberis, G.; Kalliolias, G.; Liossis, S.-N.; Andonopoulos, A.P. Combination of intravenous pulses of cyclophosphamide and methylprednizolone in patients with systemic sclerosis and interstitial lung disease. Rheumatol. Int. 2007, 27, 357–361. [Google Scholar] [CrossRef]
- Tashkin, D.P.; Elashoff, R.; Clements, P.J.; Roth, M.D.; Furst, D.E.; Silver, R.M.; Goldin, J.; Arriola, E.; Strange, C.; Bolster, M.B.; et al. Effects of 1-year treatment with cyclophosphamide on outcomes at 2 years in scleroderma lung disease. Am. J. Respir. Crit. Care Med. 2007, 176, 1026–1034. [Google Scholar] [CrossRef]
- Barnes, H.; Holland, A.E.; Westall, G.P.; Goh, N.S.; Glaspole, I.N. Cyclophosphamide for connective tissue disease-associated interstitial lung disease. Cochrane Database Syst. Rev. 2018, CD010908. [Google Scholar] [CrossRef]
- Nannini, C.; West, C.; Erwin, P.; Matteson, E. Effects of cyclophosphamide on pulmonary function in patients with scleroderma and interstitial lung disease: A systematic review and meta-analysis of randomized controlled trials and observational prospective cohort studies. Arthritis Res. Ther. 2008, 10, R124. [Google Scholar] [CrossRef]
- Zhang, G.; Xu, T.; Zhang, H.; Ye, S.; Wang, Q.; Zhang, L.; Lei, Y.; Luo, R.; Zhang, X. Randomized control multi-center clinical study of mycophenolate mofetil and cyclophosphamide in the treatment of connective tissue disease related interstitial lung disease. Zhonghua Yi Xue Za Zhi 2015, 95, 3641–3645. (In Chinese) [Google Scholar]
- Schupp, J.C.; Köhler, T.; Müller-Quernheim, J. Usefulness of Cyclophosphamide Pulse Therapy in Interstitial Lung Diseases. Respiration 2016, 91, 296–301. [Google Scholar] [CrossRef]
- Wallace, B.I.; Vummidi, D.; Khanna, D. Management of connective tissue diseases associated interstitial lung disease: A review of the published literature. Curr. Opin. Rheumatol. 2016, 28, 236–245. [Google Scholar] [CrossRef]
- Chan, E.; Chapman, K.; Kelly, C. Interstitial lung disease in rheumatoid arthritis: A review. Arthritis Res. Top. Rev. Ser. 2013, 7, 1–4. [Google Scholar]
- Fu, Q.; Wang, L.; Li, L.; Li, Y.; Liu, R.; Zheng, Y. Risk factors for progression and prognosis of rheumatoid arthritis-associated interstitial lung disease: Single center study with a large sample of Chinese population. Clin. Rheumatol. 2019, 38, 1109–1116. [Google Scholar] [CrossRef]
- Menard, T.; Salciccioli, L.; Clark, L.; Lazar, J.M.; Swigris, J.J.; Olson, A.; Fischer, A.; Lynch, D.A.; Cosgrove, G.P.; Frankel, S.K.; et al. Mycophenolate mofetil is safe, well tolerated, and preserves lung function in patients with connective tissue disease-related interstitial lung disease. Chest 2006, 130, 30–36. [Google Scholar] [CrossRef]
- Fischer, A.; Brown, K.K.; Du Bois, R.M.; Frankel, S.K.; Cosgrove, G.P.; Pérez, E.F.; Huie, T.J.; Krishnamoorthy, M.; Meehan, R.T.; Olson, A.; et al. Mycophenolate mofetil improves lung function in connective tissue disease-associated interstitial lung disease. J. Rheumatol. 2013, 40, 640–646. [Google Scholar] [CrossRef]
- Saketkoo, L.A.; Espinoza, L.R. Rheumatoid arthritis interstitial lung disease: Mycophenolate mofetil as antifibrotic and disease-modifying antirheumatic drug. Arch. Intern. Med. 2008, 168, 1718–1719. [Google Scholar] [CrossRef] [PubMed]
- Kelly, C.; Young, A.; Ahmad, Y.; Dawson, J.; Carty, S.; Nisar, M.; Woodhead, F. The effect of steroids, azathioprine and Mycophenolate on the risk of death in rheumatoid arthritis. Rheumatology 2016, 55, i99. [Google Scholar]
- Rojas-Serrano, J.; González-Velásquez, E.; Mejía, M.; Sánchez-Rodríguez, A.; Carrillo, G. Interstitial lung disease related to rheumatoid arthritis: Evolution after treatment. Reumatol. Clin. 2012, 8, 68–71. [Google Scholar] [CrossRef]
- Saravanan, V.; Kelly, C. Drug-related pulmonary problems in patients with rheumatoid arthritis. Rheumatology (Oxford) 2006, 45, 787–789. [Google Scholar] [CrossRef]
- Fragoulis, G.E.; Nikiphorou, E.; Larsen, J.; Korsten, P.; Conway, R. Methotrexate-Associated Pneumonitis and Rheumatoid Arthritis-Interstitial Lung Disease: Current Concepts for the Diagnosis and Treatment. Front. Med. (Lausanne) 2019, 6, 238. [Google Scholar] [CrossRef]
- Conway, R.; Carey, J.J. Methotrexate and lung disease in rheumatoid arthritis. Panminerva. Med. 2017, 59, 33–46. [Google Scholar] [CrossRef]
- Gaffo, A.; Alarcón, G. Methotrexate is not associated with progression of interstitial lung disease in rheumatoid arthritis. Arch. Intern. Med. 2008, 168, 1927–1928. [Google Scholar] [CrossRef]
- Kiely, P.; Busby, A.D.; Nikiphorou, E.; Sullivan, K.; Walsh, D.A.; Creamer, P.; Dixey, J.; Young, A. Is incident rheumatoid arthritis interstitial lung disease associated with methotrexate treatment? Results from a multivariate analysis in the ERAS and ERAN inception cohorts. BMJ Open. 2019, 9, e028466. [Google Scholar] [CrossRef]
- Conway, R.; Low, C.; Coughlan, R.J.; O’Donnell, M.J.; Carey, J.J. Methotrexate and lung disease in rheumatoid arthritis: A meta-analysis of randomized controlled trials. Arthritis Rheumatol. 2014, 66, 803–812. [Google Scholar] [CrossRef]
- Rojas-Serrano, J.; Herrera-Bringas, D.; Pérez-Román, D.I.; Pérez-Dorame, R.; Mateos-Toledo, H.; Mejía, M. Rheumatoid arthritis-related interstitial lung disease (RA-ILD): Methotrexate and the severity of lung disease are associated to prognosis. Clin. Rheumatol. 2017, 36, 1493–1500. [Google Scholar] [CrossRef] [PubMed]
- Saag, K.G.; Teng‡, G.G.; Patkar, N.M.; Anuntiyo, J.; Finney, C.; Curtis, J.R.; Paulus, H.E.; Mudano, A.; Pisu, M.; Elkins-Melton, M.; et al. American College of Rheumatology 2008 recommendations for the use of nonbiologic and biologic disease-modifying antirheumatic drugs in rheumatoid arthritis. Arthritis Rheum. 2008, 59, 762–784. [Google Scholar] [CrossRef] [PubMed]
- Sawada, T.; Inokuma, S.; Sato, T.; Otsuka, T.; Saeki, Y.; Takeuchi, T.; Matsuda, T.; Takemura, T.; Sagawa, A. Leflunomide-induced interstitial lung disease: Prevalence and risk factors in Japanese patients with rheumatoid arthritis. Rheumatology (Oxford) 2009, 48, 1069–1072. [Google Scholar] [CrossRef]
- Ju, J.H.; Kim, S.-I.; Lee, J.-H.; Lee, S.-I.; Yoo, W.-H.; Choe, J.-Y.; Chung, S.-H.; Lee, J.; Lee, Y.-H.; Lee, S.-S.; et al. Risk of interstitial lung disease associated with leflunomide treatment in Korean patients with rheumatoid arthritis. Arthritis Rheum. 2007, 56, 2094–2096. [Google Scholar] [CrossRef]
- Suissa, S.; Hudson, M.; Ernst, P. Leflunomide use and the risk of interstitial lung disease in rheumatoid arthritis. Arthritis Rheum. 2006, 54, 1435–1439. [Google Scholar] [CrossRef]
- Conway, R.; Low, C.; Coughlan, R.J.; O’Donnell, M.J.; Carey, J.J. Leflunomide Use and Risk of Lung Disease in Rheumatoid Arthritis: A Systematic Literature Review and Metaanalysis of Randomized Controlled Trials. J. Rheumatol. 2016, 43, 855–860. [Google Scholar] [CrossRef]
- Raj, R.; Nugent, K. Leflunomide-induced interstitial lung disease (a systematic review). Sarcoidosis Vasc. Diffuse Lung Dis. 2013, 30, 167–176. [Google Scholar] [PubMed]
- Cohen, J.M.; Miller, A.; Spiera, H. Interstitial pneumonitis complicating rheumatoid arthritis. Sustained remission with azathioprine therapy. Chest 1977, 72, 521–524. [Google Scholar] [CrossRef]
- Ishida, T.; Kotani, T.; Takeuchi, T.; Makino, S. Pulmonary toxicity after initiation of azathioprine for treatment of interstitial pneumonia in a patient with rheumatoid arthritis. J. Rheumatol. 2012, 39, 1104–1105. [Google Scholar] [CrossRef][Green Version]
- Oldham, J.M.; Lee, C.; Valenzi, E.; Witt, L.J.; Adegunsoye, A.; Hsu, S.; Chen, L.; Montner, S.; Chung, J.H.; Noth, I.; et al. Azathioprine response in patients with fibrotic connective tissue disease-associated interstitial lung disease. Respir. Med. 2016, 121, 117–122. [Google Scholar] [CrossRef]
- Parry, S.D.; Barbatzas, C.; Peel, E.T.; Barton, J.R. Sulphasalazine and lung toxicity. Eur. Respir. J. 2002, 19, 756–764. [Google Scholar] [CrossRef]
- Kumar, A.; Bhat, A.; Gupta, D.K.; Goel, A.; Malaviya, A.N. D-penicillamine-induced acute hypersensitivity pneumonitis and cholestatic hepatitis in a patient with rheumatoid arthritis. Clin. Exp. Rheumatol. 1985, 3, 337–339. [Google Scholar] [PubMed]
- Scott, D.L.; Bradby, G.V.; Aitman, T.J.; Zaphiropoulos, G.C.; Hawkins, C.F. Relationship of gold and penicillamine therapy to diffuse interstitial lung disease. Ann. Rheum. Dis. 1981, 40, 136–141. [Google Scholar] [CrossRef]
- Van der Schee, A.C.; Dinkla, B.A.; Festen, J.J. Penicillamine for interstitial lung disease in rheumatoid arthritis. Respiration 1989, 56, 134–136. [Google Scholar] [CrossRef]
- Chang, H.K.; Park, W.; Ryu, D.S. Successful treatment of progressive rheumatoid interstitial lung disease with cyclosporine: A case report. J. Korean Med. Sci. 2002, 17, 270–273. [Google Scholar] [CrossRef]
- Ogawa, D.; Hashimoto, H.; Wada, J.; Ueno, A.; Yamasaki, Y.; Yamamura, M.; Makino, H. Successful use of cyclosporin A for the treatment of acute interstitial pneumonitis associated with rheumatoid arthritis. Rheumatology (Oxford) 2000, 39, 1422–1424. [Google Scholar] [CrossRef][Green Version]
- Puttick, M.P.; Klinkhoff, A.V.; Chalmers, A.; Ostrow, D.N. Treatment of progressive rheumatoid interstitial lung disease with cyclosporine. J. Rheumatol. 1995, 22, 2163–2165. [Google Scholar] [PubMed]
- Okano, Y.; Ogasawara, H.; Ando, S.; Fujii, T.; Kaneko, H.; Tamura, N.; Yano, T.; Hirokawa, K.; Fukazawa, T.; Murashima, A.; et al. Cyclosporin A therapy for interstitial pneumonitis associated with rheumatic disease. Mod. Rheumatol. 2002, 12, 305–310. [Google Scholar] [CrossRef]
- Wilkes, M.R.; Sereika, S.M.; Fertig, N.; Lucas, M.R.; Oddis, C.V. Treatment of antisynthetaseassociated interstitial lung disease with tacrolimus. Arthritis Rheum. 2005, 52, 2439–2446. [Google Scholar] [CrossRef]
- Sharma, N.; Putman, M.S.; Vij, R.; Strek, M.E.; Dua, A. Myositis-associated Interstitial Lung Disease: Predictors of Failure of Conventional Treatment and Response to Tacrolimus in a US Cohort. J. Rheumatol. 2017, 44, 1612–1618. [Google Scholar] [CrossRef]
- Guglielmi, S.; Merz, T.M.; Gugger, M.; Suter, C.; Nicod, L.P. Acute respiratory distress syndrome secondary to antisynthetase syndrome is reversible with tacrolimus. Eur. Respir. J. 2008, 31, 213–217. [Google Scholar] [CrossRef] [PubMed]
- Sueoka, N.; Sueoka, E.; Miyazaki, Y.; Okabe, S.; Kurosumi, M.; Takayama, S.; Fujiki, H. Molecular pathogenesis of interstitial pneumonitis with TNF-alpha transgenic mice. Cytokine 1998, 10, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Hou, J.; Ma, T.; Cao, H.; Chen, Y.; Wang, C.; Chen, X.; Xiang, Z.; Han, X. TNF-α-induced NF-κB activation promotes myofibroblast differentiation of LR-MSCs and exacerbates bleomycin-induced pulmonary fibrosis. J. Cell Physiol. 2018, 233, 2409–2419. [Google Scholar] [CrossRef]
- Ortiz, L.A.; Lasky, J.; Hamilton, R.F., Jr.; Holian, A.; Hoyle, G.W.; Banks, W.; Peschon, J.J.; Brody, A.R.; Lungarella, G.; Friedman, M. Expression of TNF and the necessity of TNF receptors in bleomycin-induced lung injury in mice. Exp. Lung Res. 1998, 24, 721–743. [Google Scholar] [CrossRef]
- Dixon, W.; Hyrich, K.; Watson, K.; Lunt, M.; BSRBR Control Centre Consortium; Symmons, D.P.; British Society for Rheumatology Biologics Register. Influence of anti-TNF therapy on mortality in patients with rheumatoid arthritis associated interstitial lung disease: Results from the British Society for Rheumatology Biologics Register. Ann. Rheum. Dis. 2010, 69, 1086–1091. [Google Scholar] [CrossRef]
- Hadjinicolaou, A.V.; Nisar, M.K.; Bhagat, S.; Parfrey, H.; Chilvers, E.R.; Ostör, A.J. Non-infectious pulmonary complications of newer biological agents for rheumatic diseases—A systematic literature review. Rheumatology (Oxford) 2011, 50, 2297–2305. [Google Scholar] [CrossRef]
- Koike, T.; Harigai, M.; Ishiguro, N.; Inokuma, S.; Takei, S.; Takeuchi, T.; Yamanaka, H.; Tanaka, Y. Safety and effectiveness of adalimumab in Japanese rheumatoid arthritis patients: Postmarketing surveillance report of the first 3, 000 patients. Mod. Rheumatol. 2012, 22, 498–508. [Google Scholar] [CrossRef]
- Panopoulos, S.; Sfikakis, P. Biological treatments and connective tissue disease associated interstitial lung disease. Curr. Opin. Pulmon. Med. 2011, 17, 362–367. [Google Scholar] [CrossRef]
- Pearce, F.; Johnson, S.; Courtney, P. Interstitial lung disease following certolizumab pegol. Rheumatology (Oxford) 2012, 51, 578–580. [Google Scholar] [CrossRef][Green Version]
- Perez-Alvarez, R.; Perez-de-Lis, M.; Diaz-Lagares, C.; Pego-Reigosa, J.M.; Retamozo, S.; Bove, A.; Brito-Zeron, P.; Bosch, X.; Ramos-Casals, M. Interstitial lung disease induced or exacerbated by TNF-targeted therapies: Analysis of 122 cases. Semin. Arthritis Rheum. 2011, 41, 256–264. [Google Scholar] [CrossRef]
- Schuller, A.; Coudurier, M.; Lega, J.C.; Khouatra, C.; Cottin, V.; Cordier, J.F. Interstitial lung disease and anti-TNF-alpha therapy in rheumatoid arthritis: Two different patterns? Rev. Mal. Respir. 2010, 27, 232–237. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, T.; Tatsuki, Y.; Nogami, Y.; Ishiguro, N.; Tanaka, Y.; Yamanaka, H.; Kamatani, N.; Harigai, M.; Ryu, J.; Inoue, K.; et al. Postmarketing surveillance of the safety profile of infliximab in 5000 Japanese patients with rheumatoid arthritis. Ann. Rheum. Dis. 2008, 67, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Koo, B.S.; Hong, S.; Kim, Y.J.; Kim, Y.G.; Lee, C.K.; Yoo, B. Mortality in patients with rheumatoid arthritis-associated interstitial lung disease treated with an anti-tumor necrosis factor agent. Korean J. Intern. Med. 2015, 30, 104–109. [Google Scholar] [CrossRef] [PubMed]
- Ledingham, J.; Deighton, C.; British Society for Rheumatology Standards, Guidelines and Audit Working Group. Update on the British Society for Rheumatology guidelines for prescribing TNFalpha blockers in adults with rheumatoid arthritis (update of previous guidelines of April 2001). Rheumatology 2005, 44, 157–163. [Google Scholar] [CrossRef]
- Herrinton, L.J.; Harrold, L.R.; Liu, L.; Raebel, M.A.; Taharka, A.; Winthrop, K.L.; Solomon, D.H.; Curtis, J.R.; Lewis, J.D.; Saag, K.G. Association between anti-TNF—A therapy and interstitial lung disease. Pharmacoepidemiol. Drug Saf. 2013, 22, 394–402. [Google Scholar] [CrossRef]
- Vassallo, R.; Matteson, E.; Thomas, C.F., Jr. Clinical response of rheumatoid arthritis associated pulmonary fibrosis to tumor necrosis factor-alpha inhibition. Chest 2002, 122, 1093–1096. [Google Scholar] [CrossRef]
- Detorakis, E.E.; Magkanas, E.; Lasithiotaki, I.; Sidiropoulos, P.; Boumpas, D.T.; Gourtsoyiannis, N.; Antoniou, K.; Raissaki, M. Evolution of imaging findings, laboratory and functional parameters in rheumatoid arthritis patients after one year of treatment with anti-TNF-α agents. Clin. Exp. Rheumatol. 2017, 35, 43–52. [Google Scholar] [PubMed]
- Nakashita, T.; Ando, K.; Kaneko, N.; Takahashi, K.; Motojima, S. Potential risk of TNF inhibitors on the progression of interstitial lung disease in patients with rheumatoid arthritis. BMJ Open. 2014, 4, e005615. [Google Scholar] [CrossRef]
- Curtis, J.R.; Sarsour, K.; Napalkov, P.; Costa, L.A.; Schulman, K.L. Incidence and Complications of Interstitial Lung Disease in Users of Tocilizumab, Rituximab, Abatacept and Anti-Tumor Necrosis Factor α Agents, a Retrospective Cohort Study. Arthritis Res. Ther. 2015, 17, 319. [Google Scholar] [CrossRef]
- Bargagli, E.; Galeazzi, M.; Rottoli, P. Infliximab treatment in a patient with rheumatoid arthritis and pulmonary fibrosis. Eur. Respir. J. 2004, 24, 708. [Google Scholar] [CrossRef]
- Antoniou, K.M.; Mamoulaki, M.; Malagari, K.; Kritikos, H.D.; Bouros, D.; Siafakas, N.M.; Boumpas, D.T. Infliximab therapy in pulmonary fibrosis associated with collagen vascular disease. Clin. Exp. Rheumatol. 2007, 25, 23–28. [Google Scholar] [PubMed]
- Kaneko, A.; Hirano, Y.; Fujibayashi, T.; Hattori, Y.; Terabe, K.; Kojima, T.; Ishiguro, N. Twenty-four-week clinical results of adalimumab therapy in Japanese patients with rheumatoid arthritis: Retrospective analysis for the best use of adalimumab in daily practice. Mod. Rheumatol. 2013, 23, 466–477. [Google Scholar] [CrossRef] [PubMed]
- Komiya, K.; Ishii, H.; Fujita, N.; Oka, H.; Iwata, A.; Sonoda, H.; Kadota, J. Adalimumab-induced interstitial pneumonia with an improvement of pre-existing rheumatoid arthritis-associated lung involvement. Intern. Med. 2011, 50, 749–751. [Google Scholar] [CrossRef]
- Dias, O.M.; Pereira, D.A.; Baldi, B.G.; Costa, A.N.; Athanazio, R.A.; Kairalla, R.A.; Carvalho, C.R. Adalimumab-induced acute interstitial lung disease in a patient with rheumatoid arthritis. J. Bras. Pneumol. 2014, 40, 77–81. [Google Scholar] [CrossRef][Green Version]
- Yamazaki, H.; Isogai, S.; Sakurai, T.; Nagasaka, K. A case of adalimumab-associated interstitial pneumonia with rheumatoid arthritis. Mod. Rheumatol. 2010, 20, 518–521. [Google Scholar] [CrossRef]
- Raghu, G.; Brown, K.K.; Costabel, U.; Cottin, V.; du Bois, R.M.; Lasky, J.A.; Thomeer, M.; Utz, J.P.; Khandker, R.K.; McDermott, L.; et al. Treatment of idiopathic pulmonary fibrosis with etanercept: An exploratory, placebo-controlled trial. Am. J. Respir. Crit. Care Med. 2008, 178, 948–955. [Google Scholar] [CrossRef]
- Schultz, R.; Mattila, J.; Gappa, M.; Verronen, P. Development of progressive pulmonary interstitial and intra-alveolar cholesterol granulomas (PICG) associated with therapy-resistant chronic systemic juvenile arthritis (CJA). Pediatr. Pulmonol. 2001, 32, 397–402. [Google Scholar] [CrossRef]
- Wang, Y.; Xu, S.Q.; Xu, J.H.; Ding, C. Treatment with etanercept in a patient with rheumatoid arthritis-associated interstitial lung disease. Clin. Med. Insights Case Rep. 2011, 4, 49–52. [Google Scholar] [CrossRef]
- Horai, Y.; Miyamura, T.; Shimada, K.; Takahama, S.; Minami, R.; Yamamoto, M.; Suematsu, E. Eternacept for the treatment of patients with rheumatoid arthritis and concurrent interstitial lung disease. J. Clin. Pharm. Ther. 2012, 37, 117–121. [Google Scholar] [CrossRef]
- Tournadre, A.; Ledoux-Eberst, J.; Poujol, D.; Dubost, J.J.; Ristori, J.M.; Soubrier, M. Exacerbation of interstitial lung disease during etanercept therapy: Two cases. Joint Bone Spine 2008, 75, 215–218. [Google Scholar] [CrossRef]
- Hagiwara, K.; Sato, T.; Takagi-Kobayashi, S.; Hasegawa, S.; Shigihara, N.; Akiyama, O. Acute exacerbation of preexisting interstitial lung disease after administration of etanercept for rheumatoid arthritis. J. Rheumatol. 2007, 34, 1151–1154. [Google Scholar] [PubMed]
- Lindsay, K.; Melsom, R.; Jacob, B.K.; Mestry, N. Acute progression of interstitial lung disease: A complication of etanercept particularly in the presence of rheumatoid lung and methotrexate treatment. Rheumatology (Oxford) 2006, 45, 1048–1049. [Google Scholar] [CrossRef] [PubMed]
- Koike, T.; Harigai, M.; Inokuma, S.; Ishiguro, N.; Ryu, J.; Takeuchi, T.; Tanaka, Y.; Yamanaka, H.; Fujii, K.; Yoshinaga, T.; et al. Postmarketing surveillance of safety and effectiveness of etanercept in Japanese patients with rheumatoid arthritis. Mod. Rheumatol. 2011, 21, 343–351. [Google Scholar] [CrossRef] [PubMed]
- Koike, T.; Harigai, M.; Inokuma, S.; Ishiguro, N.; Ryu, J.; Takeuchi, T.; Tanaka, Y.; Yamanaka, H.; Hirose, T.; Yoshinaga, T.; et al. Safety and effectiveness of 6 months’ etanercept monotherapy and combination therapy in Japanese patients with rheumatoid arthritis: Effect of concomitant disease-modifying antirheumatic drugs. J. Rheumatol. 2013, 40, 1658–1668. [Google Scholar] [CrossRef] [PubMed]
- Migita, K.; Tsuji, Y.; Hisatomi, K.; Shigeno, R.; Izumi, Y.; Iwanaga, N.; Koga, T. Acute exacerbation of rheumatoid interstitial lung disease during the maintenance therapy with certolizumab pegol. Mod. Rheumatol. 2017, 27, 1079–1082. [Google Scholar] [CrossRef] [PubMed]
- Savage, E.M.; Millar, A.M.; Taggart, A.J. Comment on: A case of certolizumab-induced interstitial lung disease in a patient with rheumatoid arthritis. Rheumatology (Oxford) 2014, 53, 1154–1155. [Google Scholar] [CrossRef][Green Version]
- Lager, J.; Hilberg, O.; Løkke, A.; Bendstrup, E. Severe interstitial lung disease following treatment with certolizumab pegol: A case report. Eur. Respir. Rev. 2013, 22, 414–416. [Google Scholar] [CrossRef]
- Glaspole, I.N.; Hoy, R.F.; Ryan, P.F. A case of certolizumab-induced interstitial lung disease in a patient with rheumatoid arthritis. Rheumatology (Oxford) 2013, 52, 2302–2304. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kameda, H.; Nishida, K.; Nannki, T.; Watanabe, A.; Oshima, Y.; Momohara, S. Safety and effectiveness of certolizumab pegol in patients with rheumatoid arthritis: Interim analysis of post-marketing surveillance. Nihon Rinsho Meneki Gakkai Kaishi. 2017, 40, 196–205. [Google Scholar] [CrossRef]
- Kurata, I.; Tsuboi, H.; Terasaki, M.; Shimizu, M.; Toko, H.; Honda, F.; Ohyama, A.; Yagishita, M.; Osada, A.; Ebe, H.; et al. Effect of Biological Disease-modifying Anti-rheumatic Drugs on Airway and Interstitial Lung Disease in Patients with Rheumatoid Arthritis. Intern. Med. 2019, 58, 1703–1712. [Google Scholar] [CrossRef]
- Boleto, G.; Guignabert, C.; Pezet, S.; Cauvet, A.; Sadoine, J.; Tu, L.; Nicco, C.; Gobeaux, C.; Batteux, F.; Allanore, Y.; et al. T-cell costimulation blockade is effective in experimental digestive and lung tissue fibrosis. Arthritis Res. Ther. 2018, 20, 197. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Alvarez, L.; Arreola, J.L.; Ramírez-Martínez, G.; Ortiz-Quintero, B.; Gaxiola, M.; Reynoso-Robles, R.; Avila-Moreno, F.; Urrea, F.; Pardo, A.; Selman, M.; et al. The effect of CTLA-4Ig, a CD28/B7 antagonist, on the lung inflammation and T cell subset profile during murine hypersensitivity pneumonitis. Exp. Mol. Pathol. 2011, 91, 718–722. [Google Scholar] [CrossRef] [PubMed]
- Mera-Varela, A.; Pérez-Pampín, E. Abatacept therapy in rheumatoid arthritis with interstitial lung disease. J. Clin. Rheumatol. 2014, 20, 445–446. [Google Scholar] [CrossRef] [PubMed]
- Wada, T.; Akiyama, Y.; Yokota, K.; Sato, K.; Funakubo, Y.; Mimura, T. A case of rheumatoid arthritis complicated with deteriorated interstitial pneumonia after the administration of abatacept. Nihon Rinsho Meneki Gakkai Kaishi 2012, 35, 433–438. [Google Scholar] [CrossRef]
- Ye, W.; Fifield, M.C.; Östör, A.J. Refractory Rheumatoid Arthritis and Associated Interstitial Lung Disease: Could Abatacept be the Answer? J. Clin. Rheumatol. 2017, 23, 125–126. [Google Scholar] [CrossRef] [PubMed]
- Nakashita, T.; Ando, K.; Takahashi, K.; Motojima, S. Possible effect of abatacept on the progression of interstitial lung disease in rheumatoid arthritis patients. Respir. Investig. 2016, 54, 376–379. [Google Scholar] [CrossRef]
- Fernández-Díaz, C.; Loricera, J.; Castañeda, S.; López-Mejías, R.; Ojeda-García, C.; Olivé, A.; Rodríguez-Muguruza, S.; Carreira, P.E.; Pérez-Sandoval, T.; Retuerto, M.; et al. Abatacept in patients with rheumatoid arthritis and interstitial lung disease: A national multicenter study of 63 patients. Semin. Arthritis Rheum. 2018, 48, 22–27. [Google Scholar] [CrossRef]
- Mochizuki, T.; Ikari, K.; Yano, K.; Sato, M.; Okazaki, K. Long-term deterioration of interstitial lung disease in patients with rheumatoid arthritis treated with abatacept. Mod. Rheumatol. 2019, 29, 413–417. [Google Scholar] [CrossRef]
- Cassone, G.; Manfredi, A.; Atzeni, F.; Venerito, V.; Vacchi, C.; Picerno, V.; Furini, F.; Erre, G.L.; Tomietto, P.; Fedele, A.L.; et al. Safety of Abatacept in Italian Patients With Rheumatoid Arthritis and Interstitial Lung Disease: A Multicenter Retrospective Study. J. Clin. Med. 2020, 9, E277. [Google Scholar] [CrossRef]
- Gallelli, L.; Falcone, D.; Pelaia, G.; Renda, T.; Terracciano, R.; Malara, N.; Vatrella, A.; Sanduzzi, A.; D’Agostino, B.; Rossi, F.; et al. Interleukin-6 receptor superantagonist Sant7 inhibits TGFbeta-induced proliferation of human lung fibroblasts. Cell Prolif. 2008, 41, 393–407. [Google Scholar] [CrossRef]
- Manfredi, A.; Sebastiani, M.; Cassone, G.; Colaci, M.; Sandri, G.; Ferri, C. Tocilizumab for the treatment of patients with rheumatoid arthritis and interstitial lung diseases: A case series. Clin. Exp. Rheumatol. 2018, 36, 342. [Google Scholar] [PubMed]
- Picchianti Diamanti, A.; Markovic, M.; Argento, G.; Giovagnoli, S.; Ricci, A.; Laganà, B.; D’Amelio, R. Therapeutic management of patients with rheumatoid arthritis and associated interstitial lung disease: Case report and literature review. Ther. Adv. Respir. Dis. 2017, 11, 64–72. [Google Scholar] [CrossRef] [PubMed]
- Mohr, M.; Jacobi, A.M. Interstitial lung disease in rheumatoid arthritis: Response to IL-6R blockade. Scand. J. Rheumatol. 2011, 40, 400–401. [Google Scholar] [CrossRef]
- Manfredi, A.; Cassone, G.; Furini, F.; Gremese, E.; Venerito, V.; Atzeni, F.; Arrigoni, E.; Della Casa, G.; Cerri, S.; Govoni, M.; et al. Tocilizumab therapy in rheumatoid arthritis with interstitial lung disease: A multicenter retrospective study. Intern. Med. J. 2019. [Google Scholar] [CrossRef] [PubMed]
- Wendling, D.; Vidon, C.; Godfrin-Valnet, M.; Rival, G.; Guillot, X.; Prati, C. Exacerbation of combined pulmonary fibrosis and emphysema syndrome during tocilizumab therapy for rheumatoid arthritis. Joint Bone Spine 2013, 80, 670–671. [Google Scholar] [CrossRef] [PubMed]
- Kawashiri, S.Y.; Kawakami, A.; Sakamoto, N.; Ishimatsu, Y.; Eguchi, K. A fatal case of acute exacerbation of interstitial lung disease in a patient with rheumatoid arthritis during treatment with tocilizumab. Rheumatol. Int. 2012, 32, 4023–4026. [Google Scholar] [CrossRef] [PubMed]
- Akiyama, M.; Kaneko, Y.; Yamaoka, K.; Kondo, H.; Takeuchi, T. Association of disease activity with acute exacerbation of interstitial lung disease during tocilizumab treatment in patients with rheumatoid arthritis: A retrospective, case-control study. Rheumatol. Int. 2016, 36, 881–889. [Google Scholar] [CrossRef]
- Koike, T.; Harigai, M.; Inokuma, S.; Ishiguro, N.; Ryu, J.; Takeuchi, T.; Takei, S.; Tanaka, Y.; Ito, K.; Yamanaka, H. Postmarketing surveillance of tocilizumab for rheumatoid arthritis in Japan: Interim analysis of 3881 patients. Ann. Rheum. Dis. 2011, 70, 2148–2151. [Google Scholar] [CrossRef]
- Koike, T.; Harigai, M.; Inokuma, S.; Ishiguro, N.; Ryu, J.; Takeuchi, T.; Takei, S.; Tanaka, Y.; Sano, Y.; Yaguramaki, H.; et al. Effectiveness and safety of tocilizumab: Postmarketing surveillance of 7901 patients with rheumatoid arthritis in Japan. J. Rheumatol. 2014, 41, 15–23. [Google Scholar] [CrossRef]
- Iqbal, K.; Carty, S.; Dawson, J.; Woodhead, F.; Young, A.; Kelly, C. Survival in rheumatoid lung disease is longer in patients treated with rituximab than in those receiving anti tumour necrosis factor therapy. Rheumatology 2016, 55, i86. [Google Scholar]
- Druce, K.L.; Iqbal, K.; Watson, K.D.; Symmons, D.P.M.; Hyrich, K.L.; Kelly, C. Mortality in Patients With Interstitial Lung Disease Treated With Rituximab or TNFi as a First Biologic. RDM Open 2017, 3, e000473. [Google Scholar] [CrossRef] [PubMed]
- Hartung, W.; Maier, J.; Pfeifer, M.; Fleck, M. Effective Treatment of Rheumatoid Arthritis-Associated Interstitial Lung Disease by B-Cell Targeted Therapy with Rituximab. Case Rep. Immunol. 2012, 2012, 272303. [Google Scholar] [CrossRef] [PubMed]
- Md Yusof, M.Y.; Kabia, A.; Darby, M.; Lettieri, G.; Beirne, P.; Vital, E.M.; Dass, S.; Emery, P. Effect of Rituximab on the Progression of Rheumatoid Arthritis-Related Interstitial Lung Disease: 10 Years’ Experience at a Single Centre. Rheumatology (Oxford) 2017, 56, 1348–1357. [Google Scholar] [CrossRef] [PubMed]
- Braun-Moscovici, Y.; Butbul-Aviel, Y.; Guralnik, L.; Toledano, K.; Markovits, D.; Rozin, A.; Nahir, M.A.; Balbir-Gurman, A. Rituximab: Rescue therapy in life threatening complications or refractory autoimmune diseases: A single center experience. Rheumatol. Int. 2013, 33, 1495–1504. [Google Scholar] [CrossRef]
- Hadjinicolaou, A.V.; Nisar, M.K.; Parfrey, H.; Chilvers, E.R.; Ostor, A.J. Non-infectious pulmonary toxicity of rituximab: A systematic review. Rheumatology (Oxford) 2012, 51, 653–662. [Google Scholar] [CrossRef]
- Wagner, S.A.; Mehta, A.C.; Laber, D.A. Rituximab-induced interstitial lung disease. Am. J. Hematol. 2007, 82, 916–919. [Google Scholar] [CrossRef]
- Naqibullah, M.; Shaker, S.B.; Bach, K.S.; Bendstrup, E. Rituximab-induced interstitial lung disease: Five case reports. Eur. Clin. Respir. J. 2015, 2, 27178. [Google Scholar] [CrossRef]
- Park, S.Y.; Kim, M.Y.; Choi, W.J.; Yoon, D.H.; Lee, S.O.; Choi, S.H.; Kim, Y.S.; Suh, C.; Woo, J.H.; Kim, S.H. Pneumocystis pneumonia versus rituximab-induced interstitial lung disease in lymphoma patients receiving rituximab-containing chemotherapy. Med. Mycol. 2017, 55, 349–357. [Google Scholar] [CrossRef]
- Zhou, T.; Shen, Q.; Peng, H.; Chao, T.; Zhang, L.; Huang, L.; Yang, K.; Thapa, S.; Yu, S.; Jiang, Y. Incidence of interstitial pneumonitis in non-Hodgkin’s lymphoma patients receiving immunochemotherapy with pegylated liposomal doxorubicin and rituximab. Ann. Hematol. 2018, 97, 141–147. [Google Scholar] [CrossRef]
- Franzen, D.; Ciurea, A.; Bratton, D.J.; Clarenbach, C.F.; Latshang, T.D.; Russi, E.W.; Kyburz, D.; Kohler, M. Effect of rituximab on pulmonary function in patients with rheumatoid arthritis. Pulm. Pharmacol. Ther. 2016, 37, 24–29. [Google Scholar] [CrossRef]
- Keir, G.J.; Maher, T.M.; Ming, D.; Abdullah, R.; de Lauretis, A.; Wickremasinghe, M.; Nicholson, A.G.; Hansell, D.M.; Wells, A.U.; Renzoni, E.A. Rituximab in severe, treatment-refractory interstitial lung disease. Respirology 2014, 19, 353–359. [Google Scholar] [CrossRef]
- Chartrand, S.; Swigris, J.J.; Peykova, L.; Fischer, A. Rituximab for the treatment of connective tissue disease-associated interstitial lung disease. Sarcoidosis Vasc Diffuse Lung Dis. 2016, 32, 296–304. [Google Scholar] [PubMed]
- Fui, A.; Bergantini, L.; Selvi, E.; Mazzei, M.A.; Bennett, D.; Pieroni, M.G.; Rottoli, P.; Bargagli, E. Rituximab Therapy in Interstitial Lung Disease associated with Rheumatoid Arthritis. Intern. Med. J. 2020, 50, 330–336. [Google Scholar] [CrossRef] [PubMed]
- Matteson, E.L.; Bongartz, T.; Ryu, J.H.; Crowson, C.S.; Hartman, T.E.; Dellaripa, P.F. Open-Label, Pilot Study of the Safety and Clinical Effects of Rituximab in Patients with Rheumatoid Arthritis-Associated Interstitial Pneumonia. Open J. Rheum. Autoimmune Dis. 2012, 2, 53–58. [Google Scholar] [CrossRef]
- Becerra, E.; Leandro, M.J. Safety and efficacy of rituximab in patients with rheumatoid arthritis and lung involvement (abstract 502). Arthritis Rheum. 2012, 64, S220. [Google Scholar]
- Dass, S. Safety of rituximab in patients with rheumatoid arthritis and concomitant lung disease (abstract 205). Rheumatology (Oxford) 2011, 50, iii121. [Google Scholar]
- Kabia, A.; Yusof, Y.; Dass, S.; Emery, P. Efficacy and Safety of Rituximab in Rheumatoid Arthritis Patients with Concomitant Interstitial Lung Disease: 10-Year Experience at Single Centre. Rheumatology 2015, 54, i86. [Google Scholar] [CrossRef][Green Version]
- Harigai, M. Growing evidence of the safety of JAK inhibitors in patients with rheumatoid arthritis. Rheumatology (Oxford) 2019, 58, i34–i42. [Google Scholar] [CrossRef]
- Elli_Lilly_and_Company. Japanese Package Inserts of Baricitinib (in Japanese). Available online: http://www.info.pmda.go.jp/go/pack/3999043F1020_1_02/ (accessed on 16 July 2018).
- Pfizer. Japanese Package Inserts of Tofacitinib (in Japanese). Available online: http://www.info.pmda.go.jp/go/pack/3999034F1020_1_09/ (accessed on 16 July 2018).
- Chen, Z.; Wang, X.; Ye, S. Tofacitinib in Amyopathic Dermatomyositis-Associated Interstitial Lung Disease. N. Engl. J. Med. 2019, 381, 291–293. [Google Scholar] [CrossRef]
- Kato, M.; Ikeda, K.; Kageyama, T.; Kasuya, T.; Kumagai, T.; Furuya, H.; Furuta, S.; Tamachi, T.; Suto, A.; Suzuki, K.; et al. Successful Treatment for Refractory Interstitial Lung Disease and Pneumomediastinum With Multidisciplinary Therapy Including Tofacitinib in a Patient With Anti-MDA5 Antibody-Positive Dermatomyositis. J. Clin. Rheumatol. 2019. [Google Scholar] [CrossRef]
- Van Vollenhoven, R.F.; Fleischmann, R.; Cohen, S.; Lee, E.B.; García Meijide, J.A.; Wagner, S. Tofacitinib or adalimumab versus placebo in rheumatoid arthritis. N. Engl. J. Med. 2012, 367, 508–519. [Google Scholar] [CrossRef] [PubMed]
- Fleischmann, R.; Kremer, J.; Cush, J.; Schulze-Koops, H.; Connell, C.A.; Bradley, J.D.; Gruben, D.; Wallenstein, G.V.; Zwillich, S.H.; Kanik, K.S.; et al. Placebo-controlled trial of tofacitinib monotherapy in rheumatoid arthritis. N. Engl. J. Med. 2012, 367, 495–507. [Google Scholar] [CrossRef] [PubMed]
- Fleischmann, R.; Wollenhaupt, J.; Takiya, L.; Maniccia, A.; Kwok, K.; Wang, L.; van Vollenhoven, R.F. Safety andmaintenance of response for tofacitinib monotherapy andcombination therapy in rheumatoid arthritis: An analysis ofpooled data from open-label long-term extension studies. RMD Open 2017, 3, e000491. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.; Radominski, S.C.; Gomez-Reino, J.J.; Wang, L.; Krishnaswami, S.; Wood, S.P.; Soma, K.; Nduaka, C.I.; Kwok, K.; Valdez, H.; et al. Analysisof infections and all-cause mortality in phase II, phase III, and long-term extension studies of tofacitinib in patientswith rheumatoid arthritis. Arthritis Rheumatol. 2014, 66, 2924–2937. [Google Scholar] [CrossRef]
- Mimori, T.; Harigai, M.; Atsumi, T.; Kuwana, M.; Takei, S.; Tamura, N.; Fujii, T.; Matsuno, H.; Momohara, S.; Yamamoto, K.; et al. Post-marketing sur-veillance of tofacitinib in Japanese patients with rheuma-toid arthritis: An interim report of safety data (Abstract 431). Arthritis Rheumatol 2017, 69, 431. [Google Scholar]
- Saldarriaga-Rivera, L.M.; López-Villegas, V.J. Janus kinase inhibitors as a therapeutic option in rheumatoid arthritis and associated interstitial lung disease. Report of four cases. Rev. Colomb. Reumatol. 2019, 26, 137–139. [Google Scholar] [CrossRef]
- Sendo, S.; Saegusa, J.; Yamada, H.; Nishimura, K.; Morinobu, A. Tofacitinib facilitates the expansion of myeloid-derived suppressor cells and ameliorates interstitial lung disease in SKG mice. Arthritis Res. Ther. 2019, 6, 21, 184. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Wang, D.; Wang, L.; Wang, S.; Roden, A.C.; Zhao, H.; Li, X.; Prakash, Y.S.; Matteson, E.L.; Tschumperlin, D.J.; et al. Pro-fibrotic effects of IL-17A and elevated IL-17RA in IPF and RA-ILD support a direct role for IL-17A/IL-17RA in human fibrotic interstitial lung disease. Am. J. Physiol Lung Cell Mol Physiol. 2019, 316, L487–L497. [Google Scholar] [CrossRef] [PubMed]
- Paulin, F.; Doyle, T.J.; Fletcher, E.A.; Ascherman, D.P.; Rosas, I.O. Rheumatoid arthritis-associated interstitial lung disease and idiopathic pulmonary fibrosis: Shared mechanistic and phenotypic traits suggest overlapping disease mechanisms. Rev. Investig. Clin. 2015, 67, 280–286. [Google Scholar]
- Juge, P.A.; Lee, J.S.; Ebstein, E.; Furukawa, H.; Dobrinskikh, E.; Gazal, S.; Kannengiesser, C.; Ottaviani, S.; Oka, S.; Tohma, S.; et al. MUC5B promoter variant and rheumatoid arthritis with interstitial lung disease. N. Engl. J. Med. 2018, 379, 2209–2219. [Google Scholar] [CrossRef]
- Newton, C.A.; Oldham, J.M.; Ley, B.; Anand, V.; Adegunsoye, A.; Liu, G.; Batra, K.; Torrealba, J.; Kozlitina, J.; Glazer, C.; et al. Telomere length and genetic variant associations with interstitial lung disease progression and survival. Eur. Respir. J. 2019, 53, 1801641. [Google Scholar] [CrossRef] [PubMed]
- Flaherty, K.R.; Wells, A.U.; Cottin, V.; Devaraj, A.; Walsh, S.L.F.; Inoue, Y.; Richeldi, L.; Kolb, M.; Tetzlaff, K.; Stowasser, S.; et al. Nintedanib in Progressive Fibrosing Interstitial Lung Diseases. N. Engl. J. Med. 2019, 381, 1718–1727. [Google Scholar] [CrossRef] [PubMed]
- Behr, J.; Neuser, P.; Prasse, A.; Kreuter, M.; Rabe, K.; Schade-Brittinger, C.; Wagner, J.; Günther, A. Exploring efficacy and safety of oral Pirfenidone for progressive, non-IPF lung fibrosis (RELIEF)—A randomized, doubleblind, placebo-controlled, parallel group, multi-center, phase II trial. BMC Pulm. Med. 2017, 17, 122. [Google Scholar] [CrossRef]
- Solomon, J.J.; Danoff, S.K.; Goldberg, H.J.; Woodhead, F.; Kolb, M.; Chambers, D.C.; DiFranco, D.; Spino, C.; Haynes-Harp, S.; Hurwitz, S.; et al. The Design and Rationale of the Trail1 Trial: A Randomized Double-Blind Phase 2 Clinical Trial of Pirfenidone in Rheumatoid Arthritis-Associated Interstitial Lung Disease. Adv. Ther. 2019, 36, 3279–3287. [Google Scholar] [CrossRef]
- An Expanded Access Program to Provide Nintedanib to Patients With Non-IPF ILD Who Have no Alternative Treatment Possibilities. Available online: https://clinicaltrials.gov/ct2/show/study/NCT03843892 (accessed on 22 February 2020).
- Raghu, G.; Rochwerg, B.; Zhang, Y.; Garcia, C.A.; Azuma, A.; Behr, J.; Brozek, J.L.; Collard, H.R.; Cunningham, W.; Homma, S.; et al. An official ATS/ERS/JRS/ALAT clinical practice guideline: Treatment of idiopathic pulmonary fibrosis. an update of the 2011 clinical practice guideline. Am. J. Respir. Crit. Care Med. 2015, 192, e3–e19. [Google Scholar] [CrossRef]
- Schaefer, C.J.; Ruhrmund, D.W.; Pan, L.; Seiwert, S.D.; Kossen, K. Anti-fibrotic activities of pirfenidone in animal models. Eur. Respir. Rev. 2011, 20, 85–97. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Lin, H.; Zhang, X. Inhibitory effects of pirfenidone on fibroblast to myofibroblast transition in rheumatoid arthritis-associated interstitial lung disease via the downregulation of activating transcription factor 3 (ATF3). Int. Immunopharmacol. 2019, 74, 105700. [Google Scholar] [CrossRef] [PubMed]
- Redente, E.F.; Aguilar, M.A.; Black, B.P.; Edelman, B.L.; Bahadur, A.N.; Humphries, S.M.; Lynch, D.A.; Wollin, L.; Riches, D.W.H. Nintedanib reduces pulmonary fibrosis in a model of rheumatoid arthritis-associated interstitial lung disease. Am. J. Physiol. Lung Cell Mol. Physiol. 2018, 314, L998–L1009. [Google Scholar] [CrossRef] [PubMed]
- Kakuwa, T.; Izumi, S.; Sakamoto, K.; Suzuki, T.; Iikura, M.; Sugiyama, H. A successful treatment of rheumatoid arthritis-related interstitial pneumonia with nintedanib. Respir. Med. Case Rep. 2018, 26, 50–52. [Google Scholar] [CrossRef] [PubMed]
- Chang, K.; Yang, S.M.; Kim, S.H.; Han, K.H.; Park, S.J.; Shin, J.I. Smoking and rheumatoid arthritis. Int. J. Mol. Sci. 2014, 15, 22279–22295. [Google Scholar] [CrossRef]
- Szekanecz, Z.; Koncz, Á.; Dunkel, J.; Vencovský, J. CIMDORA collaborators. Cigarette smoking and clinical response to certolizumab pegol treatment in Hungarian, Czech, and Slovak patients with rheumatoid arthritis: 104-week data from the CIMDORA prospective, non-interventional study. Clin. Exp. Rheumatol. 2019, 37, 1010–1018. [Google Scholar] [PubMed]
- Roelsgaard, I.K.; Esbensen, B.A.; Østergaard, M.; Rollefstad, S.; Semb, A.G.; Christensen, R.; Thomsen, T. Smoking cessation intervention for reducing disease activity in chronic autoimmune inflammatory joint diseases. Cochrane Database Syst. Rev. 2019, 9, CD012958. [Google Scholar] [CrossRef] [PubMed]
- Naji, N.A.; Connor, M.C.; Donnelly, S.C.; McDonnell, T.J. Effectiveness of pulmonary rehabilitation in restrictive lung disease. J. Cardiopulm. Rehabil. 2006, 26, 237–243. [Google Scholar] [CrossRef] [PubMed]
- Holland, A.E.; Hill, C.J.; Conron, M.; Munro, P.; McDonald, C.F. Short term improvement in exercise capacity and symptoms following exercise training in interstitial lung disease. Thorax 2008, 63, 549–554. [Google Scholar] [CrossRef] [PubMed]
- Cottin, V.; Nunes, H.; Mouthon, L.; Gamondes, D.; Lazor, R.; Hachulla, E.; Revel, D.; Valeyre, D.; Cordier, J.F.; Groupe d’Etudes et de Recherche sur les Maladies “Orphelines” Pulmonaires. Combined pulmonary fibrosis and emphysema syndrome in connective tissue disease. Arthritis Rheum. 2011, 63, 295–304. [Google Scholar] [CrossRef] [PubMed]
- Antoniou, K.M.; Walsh, S.L.; Hansell, D.M.; Rubens, M.R.; Marten, K.; Tennant, R.; Hansel, T.; Desai, S.R.; Siafakas, N.M.; du Bois, R.M.; et al. Smoking-related emphysema is associated with idiopathic pulmonary fibrosis and rheumatoid lung. Respirology 2013, 18, 1191–1196. [Google Scholar] [CrossRef]
- Yazdani, A.; Singer, L.G.; Strand, V.; Gelber, A.C.; Williams, L.; Mittoo, S. Survival and quality of life in rheumatoid arthritis-associated interstitial lung disease after lung transplantation. J. Heart Lung Transplant. 2014, 33, 514–520. [Google Scholar] [CrossRef]
- Prieto-Peña, D.; Martínez-Meñaca, A.; Calderón-Goercke, M.; Mora-Cuesta, V.M.; Fernández-Rozas, S.; Iturbe-Fernández, D.; Gómez-Román, J.J.; Cifrián-Martínez, J.M.; Castañeda, S.; Hernández, J.L.; et al. Long-term survival of lung transplantation for interstitial lung disease associated with connective tissue diseases: A study of 26 cases from a referral centre. Clin. Exp. Rheumatol. 2019, 17. [Google Scholar] [PubMed]
- Courtwright, A.M.; El-Chemaly, S.; Dellaripa, P.F.; Goldberg, H.J. Survival and outcomes after lung transplantation for non-scleroderma connective tissue-related interstitial lung disease. J. Heart Lung Transplant. 2017, 36, 763–769. [Google Scholar] [CrossRef]
- Leuschner, G.; Behr, J. Acute Exacerbation in Interstitial Lung Disease. Front. Med. (Lausanne) 2017, 4, 176. [Google Scholar] [CrossRef]
- Park, I.N.; Kim, D.S.; Shim, T.S.; Lim, C.M.; Lee, S.D.; Koh, Y.; Kim, W.S.; Kim, W.D.; Jang, S.J.; Colby, T.V. Acute exacerbation of interstitial pneumonia other than idiopathic pulmonary fibrosis. Chest 2007, 132, 214–220. [Google Scholar] [CrossRef] [PubMed]
- Rice, A.J.; Wells, A.U.; Bouros, D.; du Bois, R.M.; Hansell, D.M.; Polychronopoulos, V.; Vassilakis, D.; Kerr, J.R.; Evans, T.W.; Nicholson, A.G. Terminal diffuse alveolar damage in relation to interstitial pneumonias. An autopsy study. Am. J. Clin. Pathol. 2003, 119, 709–714. [Google Scholar] [CrossRef] [PubMed]
- Parambil, J.G.; Myers, J.L.; Ryu, J.H. Diffuse alveolar damage: Uncommon manifestation of pulmonary involvement in patients with connective tissue diseases. Chest 2006, 130, 553–586. [Google Scholar] [CrossRef] [PubMed]
- Suda, T.; Kaida, Y.; Nakamura, Y.; Enomoto, N.; Fujisawa, T.; Imokawa, S.; Hashizume, H.; Naito, T.; Hashimoto, D.; Takehara, Y.; et al. Acute exacerbation of interstitial pneumonia associated with collagen vascular diseases. Respir. Med. 2009, 103, 846–853. [Google Scholar] [CrossRef] [PubMed]
- Toyoda, Y.; Hanibuchi, M.; Kishi, J.; Kawano, H.; Morizumi, S.; Sato, S.; Kondo, M.; Takikura, T.; Tezuka, T.; Goto, H.; et al. Clinical features and outcome of acute exacerbation of interstitial pneumonia associated with connective tissue disease. J. Med. Investig. 2016, 63, 294–299. [Google Scholar] [CrossRef] [PubMed]
- Silva, C.I.; Müller, N.L.; Fujimoto, K.; Kato, S.; Ichikado, K.; Taniguchi, H.; Kondoh, Y.; Johkoh, T.; Churg, A. Acute exacerbation of chronic interstitial pneumonia: High-resolution computed tomography and pathologic findings. J. Thorac. Imaging 2007, 22, 221–229. [Google Scholar] [CrossRef]
- Manfredi, A.; Sebastiani, M.; Cerri, S.; Vacchi, C.; Tonelli, R.; Della Casa, G.; Cassone, G.; Spinella, A.; Fabrizio, P.; Luppi, F.; et al. Acute Exacerbation of Interstitial Lung Diseases Secondary to Systemic Rheumatic Diseases: A Prospective Study and Review of the Literature. J. Thorac. Dis. 2019, 11, 1621–1628. [Google Scholar] [CrossRef]
- Hozumi, H.; Nakamura, Y.; Johkoh, T.; Sumikawa, H.; Colby, T.V.; Kono, M.; Hashimoto, D.; Enomoto, N.; Fujisawa, T.; Inui, N.; et al. Acute exacerbation in rheumatoid arthritis-associated interstitial lung disease: A retrospective case control study. BMJ Open 2013, 3, e003132. [Google Scholar] [CrossRef]
- Morawiec, E.; Tillie-Leblond, I.; Pansini, V.; Salleron, J.; Remy-Jardin, M.; Wallaert, B. Exacerbations of idiopathic pulmonary fibrosis treated with corticosteroids and cyclophosphamide pulses. Eur. Respir. J. 2011, 38, 1487–1489. [Google Scholar] [CrossRef]
- Ota, M.; Iwasaki, Y.; Harada, H.; Sasaki, O.; Nagafuchi, Y.; Nakachi, S.; Sumitomo, S.; Shoda, H.; Tohma, S.; Fujio, K.; et al. Efficacy of intensive immunosuppression in exacerbated rheumatoid arthritis-associated interstitial lung disease. Mod. Rheumatol. 2017, 27, 22–28. [Google Scholar] [CrossRef]
- Ito, Y.; Tazaki, G.; Kondo, Y.; Takahashi, G.; Sakamaki, F. Therapeutic Effect of Nintedanib on Acute Exacerbation of Interstitial Lung Diseases. Respir. Med. Case Rep. 2019, 26, 317–320. [Google Scholar] [CrossRef]
- Tomioka, H.; Takata, H. Treatment With Nintedanib for Acute Exacerbation of Idiopathic Pulmonary Fibrosis. Respirol. Case Rep. 2017, 5, e00215. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.S.; Kim, H.C.; Lee, B.Y.; Lee, C.K.; Kim, M.Y.; Jang, S.J.; Lee, H.S.; Moon, J.; Colby, T.V.; Kim, D.S. The Value of Biomarkers as Predictors of Outcome in Patients with Rheumatoid Arthritis-Associated Usual Interstitial Pneumonia. Sarcoidosis Vasc. Diffuse Lung Dis. 2016, 33, 216–223. [Google Scholar] [PubMed]
- Kim, H.C.; Choi, K.H.; Jacob, J.; Song, J.W. Prognostic role of blood KL-6 in rheumatoid arthritis-associated interstitial lung disease. PLoS ONE 2020, 15, e0229997. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, K.; Nakamura, H.; Takenouchi, K.; Iizawa, N.; Koiwa, M.; Sato, A.; Mochizuki, Y.; Watanabe, H.; Takai, S. Serum KL-6 elevation and possible pulmonary involvement in patients with rheumatoid arthritis treated with biological agents. J. Nippon. Med. Sch. 2014, 81, 364–371. [Google Scholar] [CrossRef] [PubMed]
- Doyle, T.J.; Patel, A.S.; Hatabu, H.; Nishino, M.; Wu, G.; Osorio, J.C.; Golzarri, M.F.; Traslosheros, A.; Chu, S.G.; Frits, M.L.; et al. Detection of Rheumatoid Arthritis-Interstitial Lung Disease Is Enhanced by Serum Biomarkers. Am. J. Respir. Crit. Care Med. 2015, 191, 1403–1412. [Google Scholar] [CrossRef]
- Harlow, L.; Gochuico, B.R.; Rosas, I.O.; Doyle, T.J.; Osorio, J.C.; Travers, T.S.; Camacho, C.C.; Oddis, C.V.; Ascherman, D.P. Anti-citrullinated heat shock protein 90 antibodies identified in bronchoalveolar lavage fluid are a marker of lung-specific immune responses. Clin. Immunol. 2014, 155, 60–70. [Google Scholar] [CrossRef]
- Walsh, S.L.F.; Wells, A.U.; Desai, S.R.; Poletti, V.; Piciucchi, S.; Dubini, A.; Nunes, H.; Valeyre, D.; Brillet, P.Y.; Kambouchner, M.; et al. Multicentre Evaluation of Multidisciplinary Team Meeting Agreement on Diagnosis in Diffuse Parenchymal Lung Disease: A Case-Cohort Study. Lancet Respir. Med. 2016, 4, 557–565. [Google Scholar] [CrossRef]
- Manfredi, A.; Cassone, G.; Cerri, S.; Venerito, V.; Fedele, A.L.; Trevisani, M.; Furini, F.; Addimanda, O.; Pancaldi, F.; Della Casa, G.; et al. Diagnostic Accuracy of a Velcro Sound Detector (VECTOR) for Interstitial Lung Disease in Rheumatoid Arthritis Patients: The InSPIRAtE Validation Study (INterStitial Pneumonia in Rheumatoid ArThritis with an Electronic Device). BMC Pulm. Med. 2019, 19, 111. [Google Scholar] [CrossRef]
- Maher, T.M.; Corte, T.J.; Fischer, A.; Kreuter, M.; Lederer, D.J.; Molina-Molina, M.; Axmann, J.; Kirchgaessler, K.U.; Cottin, V. Pirfenidone in patients with unclassifiable progressive fibrosing interstitial lung disease: Design of a double-blind, randomised, placebo-controlled phase II trial. BMJ Open Respir. Res. 2018, 5, e000289. [Google Scholar] [CrossRef]
- London, J.; Ait, E.L.; Ghaz, S. Pilot study of Pirfenidone in Pulmonary Fibrosis with Anti-Myeloperoxydase Antibodies (PIRFENIVAS). Available online: https://clinicaltrials.gov/ct2/show/NCT03385668 (accessed on 22 February 2020).
- Baughman, R.P.; Reeves, R. Pirfenidone for Progressive Fibrotic Sarcoidosis (PirFS). Available online: https://clinicaltrials.gov/ct2/show/NCT03260556 (accessed on 22 February 2020).
- Zhang, L. A Phase III, Randomized, Double-blind, Placebo Controlled, Multicenter Clinical Trial to Evaluate the Efficacy and Safety of Pirfenidone in Subjects With Dermatomyositis Interstitial Lung Disease (Dm-ILD). Available online: https://clinicaltrials.gov/ct2/show/NCT03857854 (accessed on 22 February 2020).

| Interstitial lung disease | |
| UIP | |
| NSIP, OP, DIP, LIP, mixed disease | |
| Airways disease | |
| Bronchiectasis | |
| Bronchiolitis | |
| Bronchiolitis obliterans | |
| Follicular bronchiolitis | |
| Panbronchiolitis | |
| Chronic small airway obstruction | |
| Cricoarytenoid arthritis | |
| Rheumatoid nodules | |
| generally, in subpleural areas, single or multiple, solid or cavitary, range in size | |
| Pleural disease | |
| Pleuritis | |
| Pleural effusion | |
| Pleural thickening | |
| Lung entrapment and trapped lung | |
| Pneumothorax | |
| Vascular disease | |
| Pulmonary hypertension | |
| Primary (related to underlying vasculitis) | |
| Secondary (associated to ILD) | |
| Vasculitis | |
| Haemorrhagic alveolitis | |
| Venous thromboembolism | |
| Caplan syndrome | |
| it occurs in patients with both RA and pneumoconiosis | |
| Lower respiratory tract infection | |
| Common bacterial | |
| Opportunistic infection (pneumocystis jirovecii) | |
| Fungal | |
| Mycobacterial | |
| Amyloidosis | |
| Apical fibrobullous disease | |
| Lung cancer | |
| Drug toxicity | |
| Nsaids | |
| Diffuse pulmonary infiltration | |
| Eosinophilic pneumonia | |
| ARDS | |
| Bronchospasm | |
| Infection/Pneumonitis | |
| Noncardiogenic pulmonary edema | |
| Glucocorticoids | |
| Infection/Pneumonitis | |
| Cyclophosphamide and | |
| Mycophenolate mofetil | Infection/Pneumonitis |
| Fibrosis | |
| Noncardiogenic pulmonary edema | |
| Methotrexate | |
| Hypersensitivity pneumonitis | |
| Infection | |
| New onset or exacerbation of ILD | |
| Noncardiogenic pulmonary edema | |
| Bronchospasm | |
| Leflunomide | |
| Hypersensitivity pneumonitis | |
| Infection | |
| New onset or exacerbation of ILD | |
| Other conventional DMARDs | |
| Infection/Pneumonitis | |
| Obliterative bronchiolitis | |
| New onset or exacerbation of ILD | |
| Drug-induced lupus | |
| Biologic DMARDs | |
| Infection/Pneumonitis | |
| Noncardiogenic pulmonary edema | |
| New onset or exacerbation of ILD | |
| Drug-induced lupus | |
| Histologic Pattern | Prevalence in RA | Pattern of Distribution | Radiographic Findings |
|---|---|---|---|
| UIP: Usual interstitial pneumonia | 8–66% | Peripheral, subpleural, basal | Reticular opacities; honeycombing; minimal ground-glass opacity; architectural distortion |
| NSIP: Nonspecific interstitial pneumonia | 19–57% | Peripheral, basal, symmetric | Extensive ground-glass opacity; irregular linear opacities; traction bronchiectasis; subpleural preservation |
| RB: Respiratory bronchiolitis | 0–42% | Principally upper fields, centrilobular | Bronchial wall thickening; centrilobular nodules; ground-glass opacities |
| Mixed forms and unclassifiable interstitial pneumonia | 0–11% | Coexisting patterns of interstitial fibrosing and other lung disease, e.g., emphysema | |
| OP: Organizing pneumonia | 0–11% | Subpleural, peribronchial | Focal ground-glass opacities; consolidations; reversed halo sign |
| DAD: Diffuse alveolar damage | 0–11% | Diffuse or focal | Consolidations; ground-glass opacities; traction bronchiectasis |
| DIP: Desquamative interstitial pneumonia | rare | Lower fields, predominantly peripheral | Ground-glass attenuation; cysts; reticular opacities |
| LIP: Lymphoid interstitial pneumonia | rare | Predominantly in the upper lung fields | Thin-walled cysts; centrilobular nodules; ground-glass attenuation; peribronchovascular septal thickening |
| PPFE: Idiopathic pleuroparenchymal fibroelastosis | rare | Peripheral, upper fields | Pleural thickening; subpleural fibrotic changes |
| Cyclophosphamide | ||
| Number of patients 89 | ||
| Author, year (Ref) | Article type | |
| Chang HK, 2002 [86] | case report | 1 |
| Schupp JC, 2016 [57] | retrospective study | 7 |
| Fu Q, 2018 [60] | retrospective study | 81 |
| Other articles * | ||
| Song JW, 2013 [46] | na | 84 |
| Zhang G, 2015 [56] | na | 23 CTD-ILD |
| Mycophenolate Mofetil | ||
| Number of patients 29 | ||
| Author, year (Ref) | Article type | |
| Saketkoo LA, 2008 [63] | case series | 3 |
| Fischer A, 2013 [62] | retrospective study | 18 |
| Oldham JM, 2016 [81] | retrospective study | 8 |
| Other articles * | ||
| Zhang G, 2015 [56] | na | 23 CTD-ILD |
| Methotrexate | ||
| Number of patients 72 | ||
| Author, year (Ref) | Article type | |
| Rojas-Serrano J, 2012 [65] | retrospective study | 18 |
| Rojas-Serrano J, 2017 [72] | retrospective study | 54 |
| Leflunomide | ||
| Number of patients 12 | ||
| Author, year (Ref) | Article type | |
| Rojas-Serrano J, 2012 [65] | retrospective study | 12 |
| Azathioprine | ||
| Number of patients 27 | ||
| Author, year (Ref) | Article type | |
| Cohen JM, 1977 [79] | case report | 1 |
| Ishida T, 2012 [80] | case report | 1 |
| Rojas-Serrano J, 2012 [65] | retrospective study | 10 |
| Oldham JM, 2016 [81] | retrospective study | 15 |
| Other articles * | ||
| Song JW, 2013 [46] | na | 84 |
| Penicillamine | ||
| Number of patients 7 | ||
| Author, year (Ref) | Article type | |
| van der Schee AC, 1989 [85] | open trial | 7 |
| Cyclosporine | ||
| Number of patients 8 | ||
| Author, year (Ref) | Article type | |
| Puttick MP, 1995 [88] | case report | 1 |
| Ogawa D, 2000 [87] | case report | 1 |
| Tokano Y, 2002 [89] | pilot study | 4 |
| Chang HK, 2002 [86] | case report | 1 |
| Ishida T, 2012 [80] | case report | 1 |
| Other articles * | ||
| Song JW, 2013 [46] | na | 84 |
| Tacrolimus | ||
| Number of patients 11 | ||
| Author, year (Ref) | Article type | |
| Yamano Y, 2018 [48] | retrospective case series | 11 |
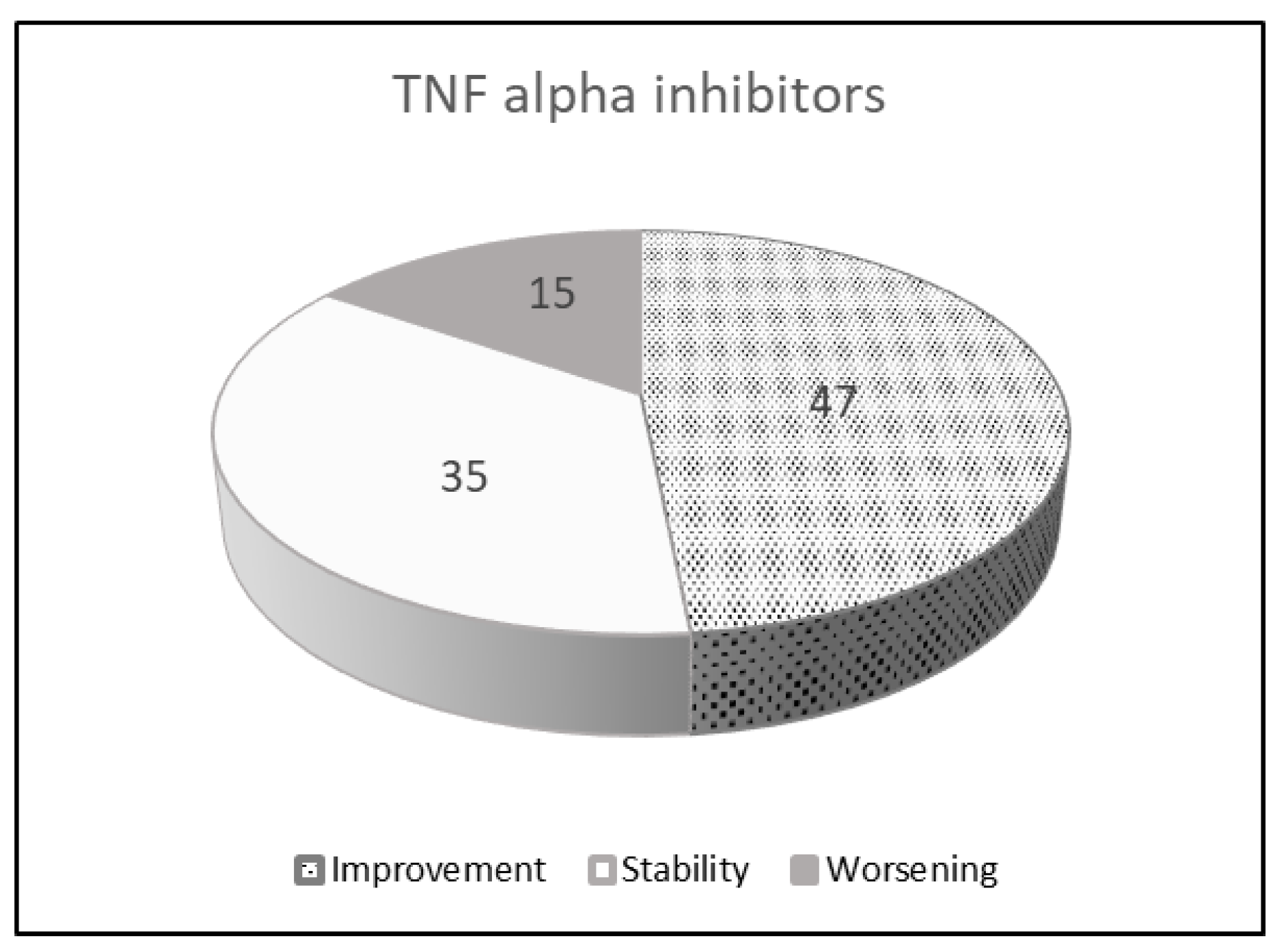
| TNF Alpha Inhibitors | ||
|---|---|---|
| Number of patients 96 | ||
| Improvement | 47 | 48.4% |
| Stability | 35 | 36.1% |
| Worsening | 15 | 15.5% |
| Author, year (Ref) | Article type | |
| Schultz R, 2001 [118] | case report | 1 |
| Vassallo R, 2002 [107] | case report | 1 |
| Bargagli E, 2004 [111] | case report | 1 |
| Antoniou KM, 2007 [112] | prospective case series | 3 |
| Wang Y, 2011 [119] | case report | 1 |
| Komiya K, 2011 [114] | case report | 1 |
| Nakashita T, 2014 [109] | retrospective review | 46 |
| Detorakis EE, 2017 [108] | prospective study | 42 |
| Other articles * | ||
| Kurata I, 2019 [131] | retrospective study | 30 |
| Abatacept | ||
|---|---|---|
| Number of patients 187 | ||
| Improvement | 31 | 16.6% |
| Stability | 140 | 74.9% |
| Worsening | 16 | 8.5% |
| Author, year (Ref) | Article type | |
| Wada T, 2012 [135] | case report | 1 |
| Mera-Varela A, 2014 [134] | case series | 4 |
| Nakashita T, 2014 [109] | retrospective review | 3 |
| Nakashita T, 2016 [137] | retrospective study | 16 |
| Ye W, 2017 [136] | case report | 1 |
| Fernández-Díaz C, 2018 [138] | retrospective study | 63 |
| Mochizuki T, 2019 [139] | retrospective study | 55 |
| Cassone G, 2020 [140] | retrospective study | 44 |
| Other articles * | ||
| Kurata I, 2019 [131] | retrospective study | 12 |
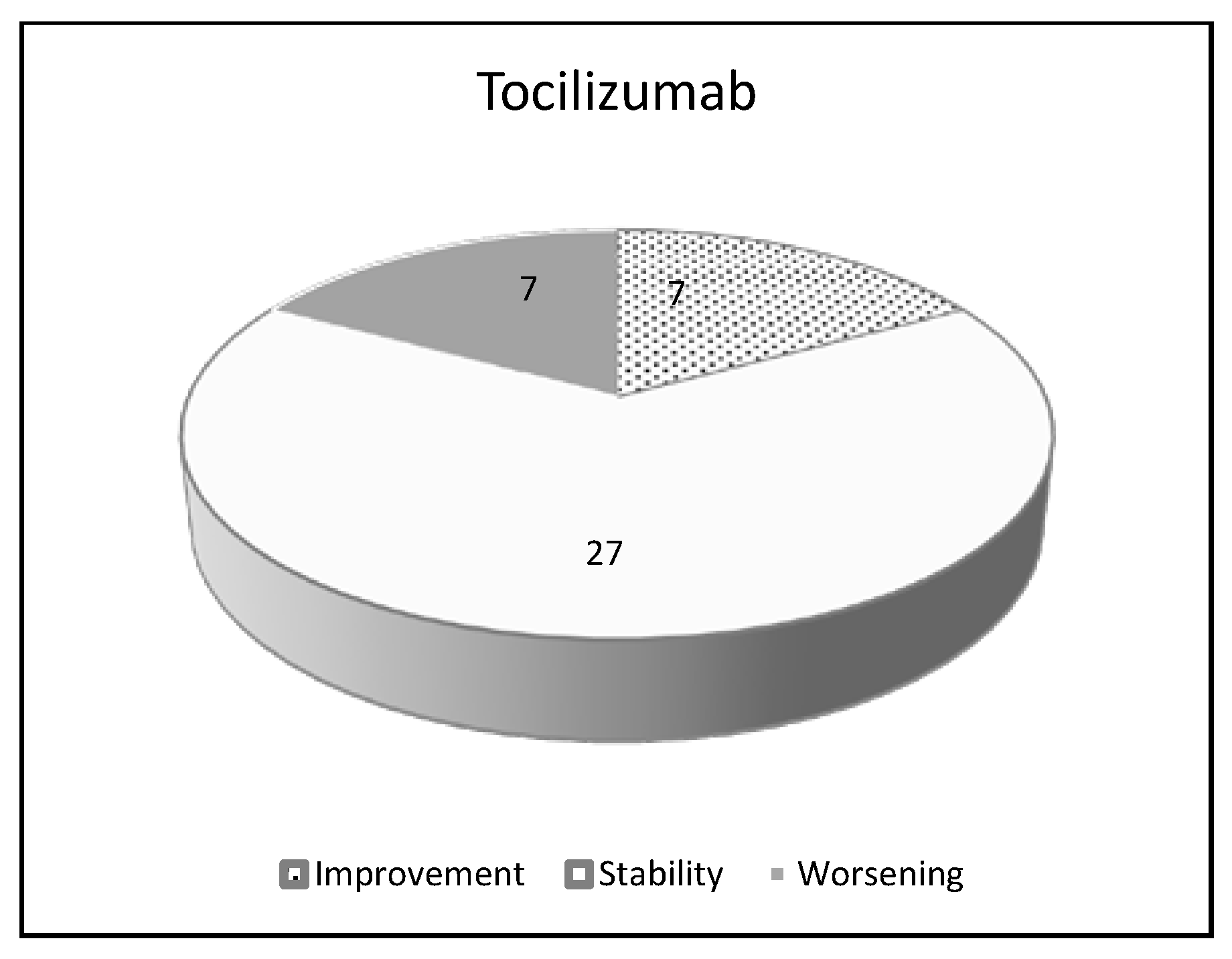
| Tocilizumab | ||
|---|---|---|
| Number of patients 41 | ||
| Improvement | 7 | 17.0% |
| Stability | 27 | 65.8% |
| Worsening | 7 | 17.0% |
| Author, year (Ref) | Article type | |
| Mohr M, 2011 [144] | case report | 1 |
| Wendling D, 2013 [146] | case report | 1 |
| Nakashita T, 2014 [109] | retrospective review | 9 |
| Picchianti Diamanti A, 2017 [143] | case report | 1 |
| Manfredi A, 2018 [142] | case series | 4 |
| Manfredi A, 2019 [145] | retrospective study | 28 |
| Other articles * | ||
| Koike T, 2014 [150] | Post-marketing data | 22 |
| Kurata I, 2019 [131] | retrospective study | 7 |
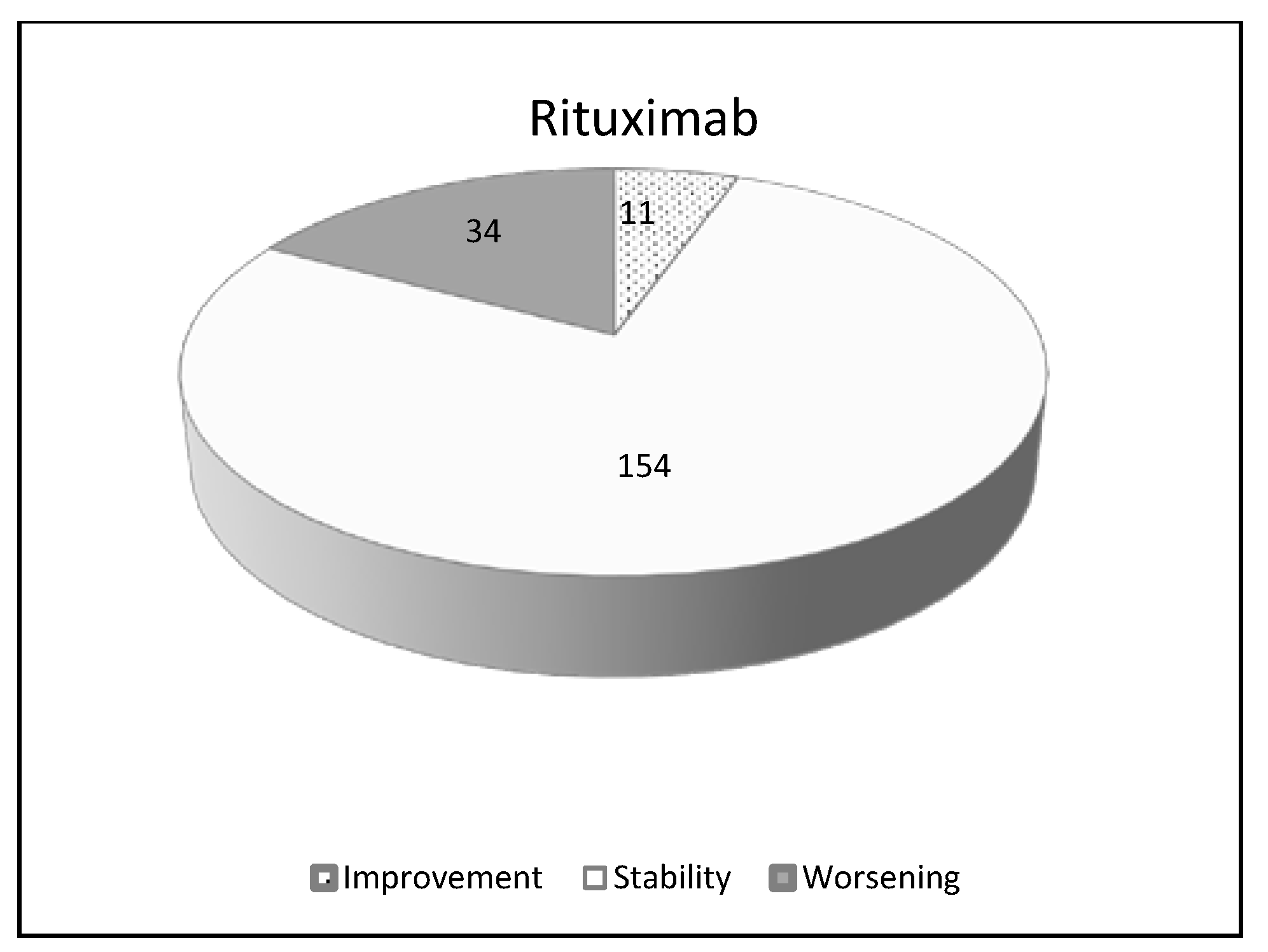
| Rituximab | ||
|---|---|---|
| Number of patients 201 | ||
| Improvement | 11 | 5.4% |
| Stability | 154 | 76.6% |
| Worsening | 34 | 16.9% |
| Author, year (Ref) | Article type | |
| Dass S, 2011 [167] | abstract | 48 |
| Matteson EL, 2012 [165] | open-label pilot study | 7 |
| Hartung W, 2012 [153] | case report | 1 |
| Kabia A, 2015 [168] | abstract | 53 |
| Chartrand S, 2016 [163] | case series | 15 |
| Yusof, 2017 [154] | retrospective observational study | 44 |
| Fui A, 2019 [164] | retrospective study | 14 |
| Duarte AC, 2019 [15] | retrospective study | 17 |
| Other articles * | ||
| Becerra E, 2012 [166] | abstract | 19 |
| Keir GJ, 2014 [162] | retrospective study | 2 |
| Trial Number (Ref) | Study Name | Phase, Design, Population | Patients | Duration | State |
|---|---|---|---|---|---|
| NCT02999178 (extension NCT03820726) [185] | Inbuild | Phase III efficacy and safety of nintedanib in patients with PF-ILD | 663 | 52 w | Completed Extension in fieri |
| EudraCT 2014–000861-32 DRKS00009822 [186] | Relief | Phase II Efficacy and safety of pirfenidone as an add-on to existing treatment for progressive, non-IPF lung fibrosis | 374 | 48 w | Completed |
| NCT02808871 [187] | Trail1 | Phase II Efficacy and safety of pirfenidone as an add-on to existing treatment in patients with RA-ILD | 270 estimated | 52 w | Recruiting |
| NCT03843892 [188] | na | Expanded access program to provide nintedanib to patients with non-IPF ILD who have no alternative treatment possibilities | na | na | Available |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cassone, G.; Manfredi, A.; Vacchi, C.; Luppi, F.; Coppi, F.; Salvarani, C.; Sebastiani, M. Treatment of Rheumatoid Arthritis-Associated Interstitial Lung Disease: Lights and Shadows. J. Clin. Med. 2020, 9, 1082. https://doi.org/10.3390/jcm9041082
Cassone G, Manfredi A, Vacchi C, Luppi F, Coppi F, Salvarani C, Sebastiani M. Treatment of Rheumatoid Arthritis-Associated Interstitial Lung Disease: Lights and Shadows. Journal of Clinical Medicine. 2020; 9(4):1082. https://doi.org/10.3390/jcm9041082
Chicago/Turabian StyleCassone, Giulia, Andreina Manfredi, Caterina Vacchi, Fabrizio Luppi, Francesca Coppi, Carlo Salvarani, and Marco Sebastiani. 2020. "Treatment of Rheumatoid Arthritis-Associated Interstitial Lung Disease: Lights and Shadows" Journal of Clinical Medicine 9, no. 4: 1082. https://doi.org/10.3390/jcm9041082
APA StyleCassone, G., Manfredi, A., Vacchi, C., Luppi, F., Coppi, F., Salvarani, C., & Sebastiani, M. (2020). Treatment of Rheumatoid Arthritis-Associated Interstitial Lung Disease: Lights and Shadows. Journal of Clinical Medicine, 9(4), 1082. https://doi.org/10.3390/jcm9041082







