Risk Factors for Postural and Functional Balance Impairment in Patients with Chronic Obstructive Pulmonary Disease
Abstract
1. Introduction
2. Materials and methods
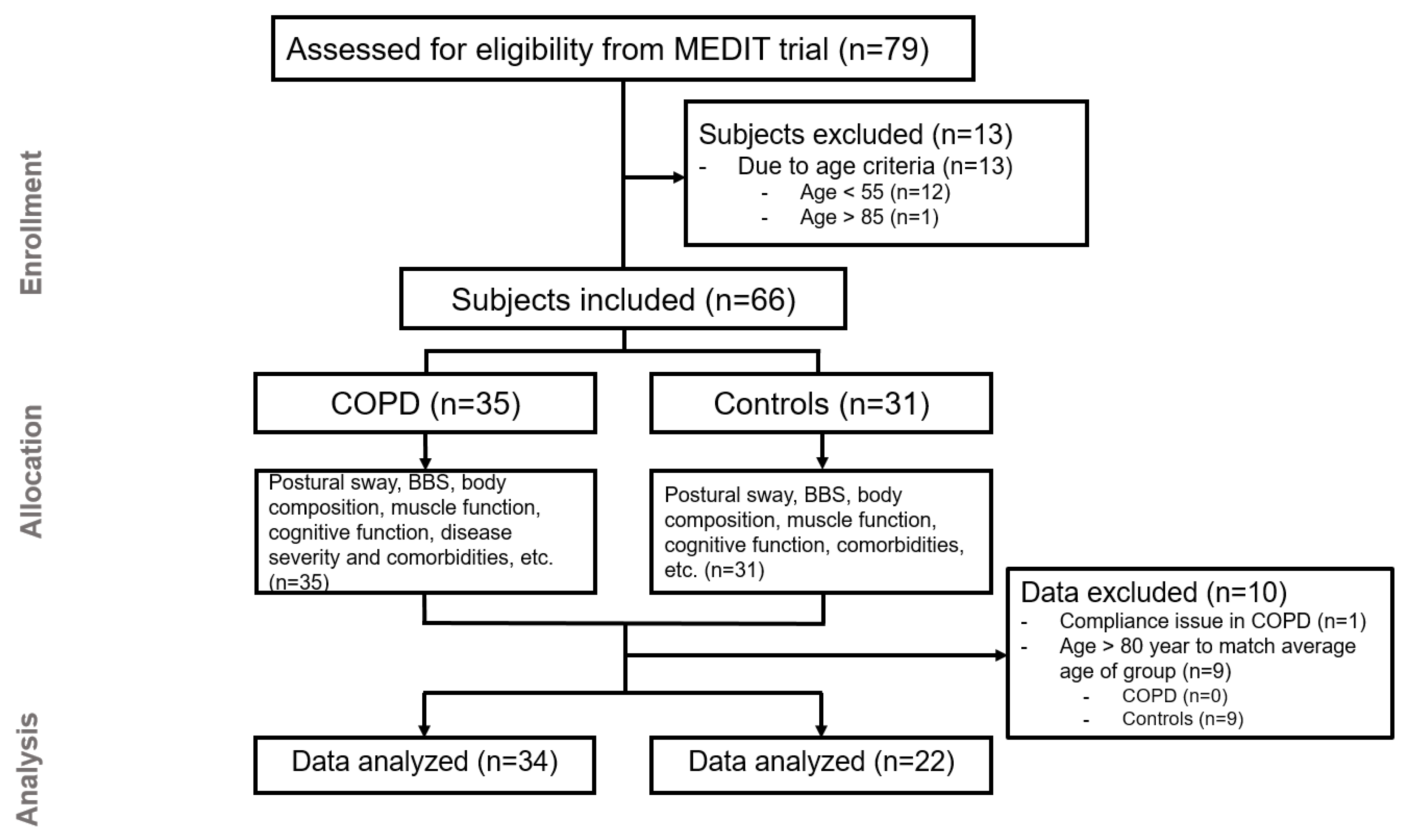
2.1. Subjects
2.2. Anthropometrics, Body Composition, and Lung Function
2.3. Postural Sway Measurement using the Center-of-Pressure
2.4. Functional Balance Measurement Using the Berg Balance Scale (BBS)
2.5. Skeletal Muscle Function and Gait Speed Measurement
2.6. Neurocognitive Function Assessments
2.7. Physical Activity, COPD Severity Questionnaires, and Comorbidity Assessment
2.8. Blood Analysis to Assess Markers of Metabolic/Clinical Health
2.9. Statistical Analysis
3. Results
3.1. General Characteristics
3.2. Balance Function
3.3. Body Composition, Gait Speed, and Muscle and Neurocognitive Functions
3.4. Plasma Clinical Markers and Amino Acid Concentrations
3.5. Relationships Between (Postural and Functional) Balance and Age, Body Composition, Muscle, Physical, and Neurocognitive Function, and Disease Severity
- Age in control (r: 0.56, p = 0.005) and COPD (r: 0.36, p = 0.034).
- Body composition: whole-body fat mass (r: 0.42, p = 0.012) in COPD.
- Neurocognitive function: Stroop interference in control and COPD (r: 0.72, p = 0.0001 and r: 0.59, p = 0.0002, respectively).
- Comorbidity and disease severity: the presence of oxygen therapy (r: 0.3959, p = 0.0204) and CCI (r: 0.4274, p = 0.0117) in COPD, the presence of (pre)diabetes (subanalysis of CCI) (r: 0.492, p = 0.0031) in COPD, and a longer duration of COPD-related symptoms (r: 0.4618, p = 0.0068).
- Plasma markers: fasting glucose concentration in COPD (r: 0.5441, p = 0.0259).
- Age in control (r: −0.49, p = 0.020).
- Blood pressure: systolic blood pressure in control (r: −0.54, p = 0.008).
- Skeletal muscle strength: maximum handgrip strength in COPD (r: 0.4594, p = 0.0479).
- Physical function: gait speed (fast) in control (r: 0.57, p = 0.005) and COPD (r: 0.68, p = 0.001).
- Physical activity level: PASE in COPD (r: 0.52, p = 0.033).
- Neurocognitive function: Stroop interference in COPD (r: -0.45, p = 0.048).
- Comorbidity and disease severity: a greater number of exacerbations in preceding year (r: −0.82, p < 0.0001), the presence of oxygen therapy (r: −0.51, p = 0.0254), and a lower transcutaneous oxygen saturation percentage (r: 0.59, p = 0.0076), and CAT score (r: −0.45, p = 0.02) in COPD.
3.6. Multiple Regression Analysis by Postural and Functional Balance
4. Discussion
4.1. Postural Sway in COPD: Sway Direction and Sway Area
4.2. Demographics, Body Composition, and Muscle Function and Postural Balance in COPD
4.3. Diabetes and Balance Impairment in COPD
4.4. Severity of Hypoxemia and Balance Impairment in COPD
4.5. Comparison between the Postural and Functional Balance Tests
5. Study limitations
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Spruit, M.A.; Singh, S.J.; Garvey, C.; ZuWallack, R.; Nici, L.; Rochester, C.; Hill, K.; Holland, A.E.; Lareau, S.C.; Man, W.D.-C. An official American Thoracic Society/European Respiratory Society statement: Key concepts and advances in pulmonary rehabilitation. Am. J. Respir. Crit. Care Med. 2013, 188, e13–e64. [Google Scholar] [CrossRef]
- Roig, M.; Eng, J.J.; MacIntyre, D.L.; FitzGerald, J.; Burns, J.; Reid, W.D. Falls in people with chronic obstructive pulmonary disease: An observational cohort study. Respir. Med. 2011, 105, 461–469. [Google Scholar] [CrossRef]
- Beauchamp, M.K.; Hill, K.; Goldstein, R.S.; Janaudis-Ferreira, T.; Brooks, D. Impairments in balance discriminate fallers from non-fallers in COPD. Respir. Med. 2009, 103, 1885–1891. [Google Scholar] [CrossRef]
- Beauchamp, M.K. Balance assessment in people with COPD: An evidence-based guide. Chronic Respir. Dis. 2018, 16, 1479973118820311. [Google Scholar] [CrossRef]
- Mancini, M.; Horak, F.B. The relevance of clinical balance assessment tools to differentiate balance deficits. Eur. J. Phys. Rehabil. Med. 2010, 46, 239–248. [Google Scholar]
- Topper, A.; Maki, B.; Holliday, P.J. Are activity-based assessments of balance and gait in the elderly predictive of risk of falling and/or type of fall? J. Am. Geriatr. Soc. 1993, 41, 479–487. [Google Scholar] [CrossRef]
- Schlenstedt, C.; Brombacher, S.; Hartwigsen, G.; Weisser, B.; Möller, B.; Deuschl, G. Comparing the Fullerton Advanced Balance Scale with the Mini-BESTest and Berg Balance Scale to assess postural control in patients with Parkinson disease. Arch. Phys. Med. Rehabil. 2015, 96, 218–225. [Google Scholar] [CrossRef]
- Jácome, C.; Cruz, J.; Oliveira, A.; Marques, A. Validity, Reliability, and Ability to Identify Fall Status of the Berg Balance Scale, BESTest, Mini-BESTest, and Brief-BESTest in Patients With COPD. Phys. Ther. 2016, 96, 1807–1815. [Google Scholar] [CrossRef]
- Lin, D.; Seol, H.; Nussbaum, M.A.; Madigan, M.L. Reliability of COP-based postural sway measures and age-related differences. Gait Posture 2008, 28, 337–342. [Google Scholar] [CrossRef]
- Smith, M.D.; Chang, A.T.; Seale, H.E.; Walsh, J.R.; Hodges, P.W. Balance is impaired in people with chronic obstructive pulmonary disease. Gait Posture 2010, 31, 456–460. [Google Scholar] [CrossRef]
- Janssens, L.; Brumagne, S.; McConnell, A.K.; Claeys, K.; Pijnenburg, M.; Burtin, C.; Janssens, W.; Decramer, M.; Troosters, T. Proprioceptive changes impair balance control in individuals with chronic obstructive pulmonary disease. PLoS ONE 2013, 8, e57949. [Google Scholar] [CrossRef] [PubMed]
- Browne, J.; O’Hare, N. Review of the different methods for assessing standing balance. Physiotherapy 2001, 87, 489–495. [Google Scholar] [CrossRef]
- Voica, A.S.; Oancea, C.; Tudorache, E.; Crisan, A.F.; Fira-Mladinescu, O.; Tudorache, V.; Timar, B. Chronic obstructive pulmonary disease phenotypes and balance impairment. Int. J. Chronic Obstr. Pulm. Dis. 2016, 11, 919–925. [Google Scholar] [CrossRef][Green Version]
- Tudorache, E.; Oancea, C.; Avram, C.; Fira-Mladinescu, O.; Petrescu, L.; Timar, B. Balance impairment and systemic inflammation in chronic obstructive pulmonary disease. Int. J. Chronic Obstr. Pulm. Dis. 2015, 10, 1847. [Google Scholar] [CrossRef] [PubMed]
- Iwakura, M.; Okura, K.; Shibata, K.; Kawagoshi, A.; Sugawara, K.; Takahashi, H.; Shioya, T. Relationship between balance and physical activity measured by an activity monitor in elderly COPD patients. Int. J. Chronic Obstr. Pulm. Dis. 2016, 11, 1505. [Google Scholar] [CrossRef]
- Porto, E.F.; Castro, A.A.M.; Schmidt, V.G.S.; Rabelo, H.M.; Kümpel, C.; Nascimento, O.A.; Jardim, J.R. Postural control in chronic obstructive pulmonary disease: A systematic review. Int. J. Chronic Obstr. Pulm. Dis. 2015, 10, 1233–1239. [Google Scholar] [CrossRef]
- Mochizuki, L.; Duarte, M.; Amadio, A.C.; Zatsiorsky, V.M.; Latash, M.L. Changes in postural sway and its fractions in conditions of postural instability. J. Appl. Biomech. 2006, 22, 51–60. [Google Scholar] [CrossRef]
- Scoppa, F.; Capra, R.; Gallamini, M.; Shiffer, R. Clinical stabilometry standardization: Basic definitions--acquisition interval--sampling frequency. Gait Posture 2013, 37, 290–292. [Google Scholar] [CrossRef]
- Prieto, T.E.; Myklebust, J.; Hoffmann, R.; Lovett, E.; Myklebust, B. Measures of postural steadiness: Differences between healthy young and elderly adults. IEEE Trans. Biomed. Eng. 1996, 43, 956–966. [Google Scholar] [CrossRef]
- Apthorp, D.; Nagle, F.; Palmisano, S. Chaos in balance: Non-linear measures of postural control predict individual variations in visual illusions of motion. PLoS ONE 2014, 9, e113897. [Google Scholar] [CrossRef]
- Berg, K.; Wood-Dauphine, S.; Williams, J.; Gayton, D. Measuring balance in the elderly: Preliminary development of an instrument. Physiother. Can. 1989, 41, 304–311. [Google Scholar] [CrossRef]
- Shumway-Cook, A.; Baldwin, M.; Polissar, N.L.; Gruber, W. Predicting the probability for falls in community-dwelling older adults. Phys. Ther. 1997, 77, 812–819. [Google Scholar] [CrossRef] [PubMed]
- Karpman, C.; Benzo, R. Gait speed as a measure of functional status in COPD patients. Int. J. Chronic Obstr. Pulm. Dis. 2014, 9, 1315. [Google Scholar]
- Sánchez-Cubillo, I.; Perianez, J.; Adrover-Roig, D.; Rodriguez-Sanchez, J.; Rios-Lago, M.; Tirapu, J.; Barcelo, F. Construct validity of the Trail Making Test: Role of task-switching, working memory, inhibition/interference control, and visuomotor abilities. J. Int. Neuropsychol. Soc. 2009, 15, 438–450. [Google Scholar] [CrossRef]
- Lamers, M.J.; Roelofs, A.; Rabeling-Keus, I.M. Selective attention and response set in the Stroop task. Mem. Cogn. 2010, 38, 893–904. [Google Scholar] [CrossRef]
- Stroop, J.R. Studies of interference in serial verbal reactions. J. Exp. Psychol. 1935, 18, 643. [Google Scholar] [CrossRef]
- Valentijn, S.A.; Van Boxtel, M.P.; Van Hooren, S.A.; Bosma, H.; Beckers, H.J.; Ponds, R.W.; Jolles, J. Change in sensory functioning predicts change in cognitive functioning: Results from a 6-year follow-up in the Maastricht Aging Study. J. Am. Geriatr. Soc. 2005, 53, 374–380. [Google Scholar] [CrossRef]
- Deutz, N.E.P.; Thaden, J.J.; ten Have, G.A.M.; Walker, D.K.; Engelen, M.P.K.J. Metabolic phenotyping using kinetic measurements in young and older healthy adults. Metabolism 2018, 78, 167–178. [Google Scholar] [CrossRef]
- Maki, B.E.; Holliday, P.J.; Topper, A.K. A prospective study of postural balance and risk of falling in an ambulatory and independent elderly population. J. Gerontol. 1994, 49, M72–M84. [Google Scholar] [CrossRef]
- Wollseifen, T. Different methods of calculating body sway area. Pharm. Program. 2011, 4, 91–106. [Google Scholar] [CrossRef]
- Hageman, P.A.; Leibowitz, J.M.; Blanke, D. Age and gender effects on postural control measures. Arch. Phys. Med. Rehabil. 1995, 76, 961–965. [Google Scholar] [CrossRef]
- Incalzi, R.A.; Gemma, A.; Marra, C.; Capparella, O.; Fuso, L.; Carbonin, P. Verbal memory impairment in COPD: Its mechanisms and clinical relevance. Chest 1997, 112, 1506–1513. [Google Scholar] [CrossRef] [PubMed]
- Engelen, M.P.; Jonker, R.; Thaden, J.J.; Ten Have, G.A.; Jeon, M.S.; Dasarathy, S.; Deutz, N.E. Comprehensive metabolic flux analysis to explain skeletal muscle weakness in COPD. Clin. Nutr. 2020. [Google Scholar] [CrossRef] [PubMed]
- Engelen, M.P.; van der Meij, B.S.; Deutz, N.E. Protein anabolic resistance in cancer: Does it really exist? Curr. Opin. Clin. Nutr. Metab. Care 2016, 19, 39. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Froese, E.A.; Houston, M.E. Torque-velocity characteristics and muscle fiber type in human vastus lateralis. J. Appl. Physiol. 1985, 59, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Kuo, A.D.; Zajac, F.E. A biomechanical analysis of muscle strength as a limiting factor in standing posture. J. Biomech. 1993, 26, 137–150. [Google Scholar] [CrossRef]
- Said, G. Diabetic neuropathy—A review. Nat. Rev. Neurol. 2007, 3, 331. [Google Scholar] [CrossRef]
- Grp, D.R. Factors in Development of Diabetic Neuropathy-Baseline Analysis of Neuropathy in Feasibility Phase of Diabetes Control and Complications Trial (DCCT). Diabetes 1988, 37, 476–481. [Google Scholar]
- Juster-Switlyk, K.; Smith, A.G. Updates in diabetic peripheral neuropathy. F1000Research 2016, 5. [Google Scholar] [CrossRef]
- Lord, S.R.; Menz, H.B.; Tiedemann, A. A physiological profile approach to falls risk assessment and prevention. Phys. Ther. 2003, 83, 237–252. [Google Scholar] [CrossRef]
- Santos, M.J.; Kanekar, N.; Aruin, A.S. The role of anticipatory postural adjustments in compensatory control of posture: 1. Electromyographic analysis. J. Electromyogr. Kinesiol. 2010, 20, 388–397. [Google Scholar] [CrossRef] [PubMed]
- Simmons, R.W.; Richardson, C.; Pozos, R. Postural stability of diabetic patients with and without cutaneous sensory deficit in the foot. Diabetes Res. Clin. Pract. 1997, 36, 153–160. [Google Scholar] [CrossRef]
- Nakamura, R.; Noritake, M.; Hosoda, Y.; Kamakura, K.; Nagata, N.; Shibasaki, H. Somatosensory conduction delay in central and peripheral nervous system of diabetic patients. Diabetes Care 1992, 15, 532–535. [Google Scholar] [CrossRef] [PubMed]
- Appenzeller, O.; Parks, R.D.; MacGee, J. Peripheral neuropathy in chronic disease of the respiratory tract. Am. J. Med. 1968, 44, 873–880. [Google Scholar] [CrossRef]
- Dodd, J.; Getov, S.; Jones, P.W. Cognitive function in COPD. Eur. Respir. J. 2010, 35, 913–922. [Google Scholar] [CrossRef]
- Donker, S.F.; Roerdink, M.; Greven, A.J.; Beek, P.J. Regularity of center-of-pressure trajectories depends on the amount of attention invested in postural control. Exp. Brain Res. 2007, 181, 1–11. [Google Scholar] [CrossRef]
- Grant, I.; Heaton, R.K.; McSweeny, A.J.; Adams, K.M.; Timms, R.M. Neuropsychologic findings in hypoxemic chronic obstructive pulmonary disease. Arch. Intern. Med. 1982, 142, 1470–1476. [Google Scholar] [CrossRef]
- Alexandre, F.; Heraud, N.; Varray, A. Is nocturnal desaturation a trigger for neuronal damage in chronic obstructive pulmonary disease? Med. Hypotheses 2015, 84, 25–30. [Google Scholar] [CrossRef]
- Esser, R.W.; Stoeckel, M.C.; Kirsten, A.; Watz, H.; Taube, K.; Lehmann, K.; Petersen, S.; Magnussen, H.; von Leupoldt, A. Structural brain changes in patients with COPD. Chest 2016, 149, 426–434. [Google Scholar] [CrossRef]
- Roig, M.; Eng, J.J.; Road, J.D.; Reid, W.D. Falls in patients with chronic obstructive pulmonary disease: A call for further research. Respir. Med. 2009, 103, 1257–1269. [Google Scholar] [CrossRef]
- Downs, S.; Marquez, J.; Chiarelli, P. The Berg Balance Scale has high intra-and inter-rater reliability but absolute reliability varies across the scale: A systematic review. J. Physiother. 2013, 59, 93–99. [Google Scholar] [CrossRef]
- Horak, F.B. Clinical assessment of balance disorders. Gait Posture 1997, 6, 76–84. [Google Scholar] [CrossRef]
- B³aszczyk, J.W. Sway ratio a new measure for quantifying postural stability. Acta Neurobiol. Exp. 2008, 68, 51–57. [Google Scholar]
| Control (n = 22) | COPD (n = 34) | p Value | |
|---|---|---|---|
| General and clinical characteristics of the COPD and Control groups | |||
| Age (years) | 70.44 (1.72) | 68.97 (1.36) | 0.505 |
| Sex (Male/Female) | 11/11 | 14/20 | 0.588 |
| Body Weight (kg) | 82.36 (2.26) | 83.32 (3.47) | 0.838 |
| Height (m) | 1.67 (0.01) | 1.65 (0.01) | 0.326 |
| Body Mass Index (kg/m2) | 29.50 (0.79) | 30.53 (1.20) | 0.528 |
| Charlson comorbidity index (score) | 0.31 (0.12) | 2.08 (0.25) ** | < 0.0001 |
| Physical activity level (PASE score) | 122.0 (18.22) | 106.4 (12.46) | 0.507 |
| Number of subjects who had a fall and/or near fall history within last 12 month 1 | N/A | 21 | - |
| Pulmonary function and COPD-related measures | |||
| FEV1 (% of predicted) | 96.91 (2.96) | 44.18 (3.13) ** | < 0.0001 |
| Transcutaneous oxygen saturation (%) | 97.32 (0.33) | 95.00 (0.70) * | 0.014 |
| Duration of COPD-related symptoms (years) | 10.82 (1.11) | ||
| No. of hospitalizations in last year for exacerbation | 0.26 (0.10) | ||
| No. of exacerbations in the past year | 0.73 (0.24) | ||
| CAT (score) | 21.00 (1.26) | ||
| GOLD Stage | 2.87 (0.13) | ||
| Dyspnea Scale | 2.09 (0.18) | ||
| Oxygen therapy usage (yes/no) | 0/22 | 20/14 | |
| Body Composition | |||
| Lean mass (kg) | 49.00 (2.27) | 48.51 (1.94) | 0.871 |
| Lean mass extremities (kg) | 20.77 (0.91) | 19.41 (0.90) | 0.318 |
| Fat mass (kg) | 28.56 (1.65) | 32.28 (1.91) | 0.178 |
| Fat mass index (kg/m2) | 10.36 (0.68) | 11.95 (0.78) | 0.163 |
| Fat-free mass index (kg/m2) 2 | 19.09 (0.44) | 18.46 (0.63) | 0.478 |
| Appendicular skeletal muscle index (kg/m2) 3 | 7.365 (0.22) | 7.053 (0.25) | 0.4 |
| Fat % android/gynoid (ratio) 4 | 1.101 (0.05) | 1.015 (0.04) | 0.205 |
| Muscle function | |||
| Inspiratory muscle strength (cmH2O) | 83.50 (4.27) | 60.97 (4.22) ** | 0.0007 |
| Expiratory muscle strength (cmH2O) | 100.9 (8.02) | 82.71 (5.90) | 0.068 |
| Maximal handgrip strength (N) | 235.8 (16.58) | 203.4 (10.47) | 0.087 |
| Maximal leg extension force (N) | 257.7 (13.31) | 210.5 (13.86)* | 0.023 |
| Maximal leg extension force per kg fat-free mass (N/kg) | 4.828 (0.20) | 4.152 (0.19)* | 0.026 |
| Physical performance | |||
| Usual gait speed (m/sec) | 1.23 (0.04) | 0.95 (0.04)** | < 0.0001 |
| Fast gait speed (m/sec) | 1.93 (0.07) | 1.33 (0.05)** | < 0.0001 |
| Neurocognitive function | |||
| TMT difference (time B–A) | 24.61 (2.828) | 47.74 (6.370) ** | 0.0032 |
| Stroop interference (score) | 50.95 (4.756) | 61.66 (3.924) | 0.0628 |
| Plasma clinical markers & amino acid concentrations | |||
| EAA (μmol/L) | 589.4 (23.53) | 588.1 (35.14) | 0.975 |
| TRP (μmol/L) | 38.33 (1.91) | 38.13 (1.84) | 0.939 |
| LNAA (μmol/L) | 706.8 (25.29) | 703.9 (36.29) | 0.9476 |
| BCAA (μmol/L) | 322.1 (15.50) | 329.2 (27.17) | 0.818 |
| Glucose concentration (mmol/L) | 5.46 (0.129) | 5.49 (0.113) | 0.6331 |
| Hs-CRP (mg/L) | 1.40 (0.265) | 3.80 (0.872)* | 0.0463 |
| Control (n = 22) | COPD (n = 34) | p Value | |
|---|---|---|---|
| Postural balance—Postural sway measurement by force platform | |||
| AP mean sway velocity (cm/s) | 0.86 (0.061) | 1.11 (0.073) * | 0.0496 |
| ML mean sway velocity (cm/s) | 0.74 (0.051) | 0.86 (0.065) | 0.4485 |
| AP–ML mean sway velocity (cm/s) | 1.27 (0.084) | 1.62 (0.110) | 0.0752 |
| Sway area (cm2) | 3.46 (0.455) | 3.41 (0.319) | 0.9786 |
| Functional balance—Performance oriented measure † | |||
| Berg Balance Scale (score) | 54.71 (0.34) | 53.11 (0.55) * | 0.0253 |
| Coefficients | SE | |t| | p Value | |
|---|---|---|---|---|
| Intercept | −0.4461 | 0.2417 | 1.845 | 0.0752 |
| Presence of oxygen therapy (Y/N) | 0.4062 | 0.125 | 3.249 | 0.0029 |
| Fat mass (kg) | 0.02072 | 0.00556 | 3.726 | 0.0008 |
| Stroop interference (score) | 0.009796 | 0.002222 | 4.409 | 0.0001 |
| Presence of diabetes/prediabetes (Y/N) | 0.451 | 0.1332 | 3.385 | 0.0021 |
| Coefficients | SE | |t| | p Value | |
|---|---|---|---|---|
| Intercept | −5.182 | 21.23 | 0.2441 | 0.8105 |
| Transcutaneous oxygen saturation (%) | 0.5577 | 0.2166 | 2.575 | 0.0211 |
| Exacerbation in the last year (number) | −1.165 | 0.3545 | 3.286 | 0.005 |
| Gait speed (fast) (m/s) | 3.972 | 1.395 | 2.847 | 0.0122 |
| Coefficients | SE | |t| | p Value | |
|---|---|---|---|---|
| Intercept | 0.3956 | 0.1138 | 3.477 | 0.0024 |
| Stroop interference (score) | 0.009227 | 0.002053 | 4.494 | 0.0002 |
| Coefficients | SE | |t| | p Value | |
|---|---|---|---|---|
| Intercept | 71.28 | 6.039 | 11.8 | < 0.0001 |
| Systolic blood pressure (mmHg) | −0.1308 | 0.04482 | 2.919 | 0.0085 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, J.K.; Deutz, N.E.P.; Cruthirds, C.L.; Kirschner, S.K.; Park, H.; Madigan, M.L.; Engelen, M.P.K.J. Risk Factors for Postural and Functional Balance Impairment in Patients with Chronic Obstructive Pulmonary Disease. J. Clin. Med. 2020, 9, 609. https://doi.org/10.3390/jcm9020609
Park JK, Deutz NEP, Cruthirds CL, Kirschner SK, Park H, Madigan ML, Engelen MPKJ. Risk Factors for Postural and Functional Balance Impairment in Patients with Chronic Obstructive Pulmonary Disease. Journal of Clinical Medicine. 2020; 9(2):609. https://doi.org/10.3390/jcm9020609
Chicago/Turabian StylePark, Jaekwan K., Nicolaas E. P. Deutz, Clayton L. Cruthirds, Sarah K. Kirschner, Hangue Park, Michael L. Madigan, and Mariëlle P. K. J. Engelen. 2020. "Risk Factors for Postural and Functional Balance Impairment in Patients with Chronic Obstructive Pulmonary Disease" Journal of Clinical Medicine 9, no. 2: 609. https://doi.org/10.3390/jcm9020609
APA StylePark, J. K., Deutz, N. E. P., Cruthirds, C. L., Kirschner, S. K., Park, H., Madigan, M. L., & Engelen, M. P. K. J. (2020). Risk Factors for Postural and Functional Balance Impairment in Patients with Chronic Obstructive Pulmonary Disease. Journal of Clinical Medicine, 9(2), 609. https://doi.org/10.3390/jcm9020609







