Abstract
From monoclonal antibodies (mAbs) to Chimeric Antigen Receptor (CAR) T cells, immunotherapies have enhanced the efficacy of treatments against B cell malignancies. The same has not been true for Acute Myeloid Leukemia (AML). Hematologic toxicity has limited the potential of modern immunotherapies for AML at preclinical and clinical levels. Gemtuzumab Ozogamicin has demonstrated hematologic toxicity, but the challenge of preserving normal hematopoiesis has become more apparent with the development of increasingly potent immunotherapies. To date, no single surface molecule has been identified that is able to differentiate AML from Hematopoietic Stem and Progenitor Cells (HSPC). Attempts have been made to spare hematopoiesis by targeting molecules expressed only on later myeloid progenitors as well as AML or using toxins that selectively kill AML over HSPC. Other strategies include targeting aberrantly expressed lymphoid molecules or only targeting monocyte-associated proteins in AML with monocytic differentiation. Recently, some groups have accepted that stem cell transplantation is required to access potent AML immunotherapy and envision it as a rescue to avoid severe hematologic toxicity. Whether it will ever be possible to differentiate AML from HSPC using surface molecules is unclear. Unless true specific AML surface targets are discovered, stem cell transplantation could be required to harness the true potential of immunotherapy in AML.
1. Introduction
Until recently, therapeutic options for Acute Myeloid Leukemia (AML) had changed very little. In the last decade, there has been a substantial increase in knowledge of the molecular landscape of AML, which has led to several new targeted therapies [1]. However, many of these molecular therapies have limited duration of action when not combined with conventional chemotherapy [2]. Since the FDA approval of Rituximab in 1997, immunotherapy has become an increasingly important part of the management of hematologic malignancies [3]. Unconjugated antibodies, Antibody Drug Conjugates (ADC), Bispecific T Cell Engagers (BiTEs), and Chimeric Antigen Receptor (CAR) T cells are all now part of accepted therapy.
More potent forms of immunotherapy, such as Chimeric Antigen Receptor (CAR) T cells, lead to target cell aplasia, which is tolerable in B cell malignancies [4,5,6]. Indeed, nearly all the benefits of targeted immunotherapy are in the setting of B cell malignancies. Aside from Gemtuzumab Ozogamicin (GO), no immunotherapies have been accepted for hematologic malignancies of myeloid origin [7]. The limited efficacy in AML to date highlights the challenges presented.
Most surface molecules expressed on AML are also expressed on Hematopoietic Stem and Progenitor Cells (HSPC), and potent immunotherapies against traditional AML molecules (CD33 and CD123) have led to hematologic toxicity, both in preclinical models and in clinical trials [8,9,10]. An additional issue is that the complex subclonal architecture and plasticity of surface molecules makes tumor escape a significant concern [11,12,13,14]. Despite these challenges, new immunotherapies against AML continue to be tested. Allogeneic Hematopoietic Stem Cell Transplantation (allo-HSCT) is an established cellular therapy in AML, but the toxicity and limited efficacy, especially in high risk patients, allows for potential improvement. In this procedure, patients undergo conditioning chemotherapy with or without radiotherapy followed by transfusion of donor hematopoietic cells. Immunosuppression is required after the transplant to reduce the chance of Graft Versus Host Disease (GVHD). Allo-HSCT has been shown to reduce the relapse rate of AML and is the only potentially curative therapy in those with refractory disease [15,16]. The decision on whether to offer an allo-HSCT is complex and considers patient fitness, risk of AML relapse, and availability of donors. Generally, if a suitable well-matched donor is available allo-HSCT is recommended for those with intermediate or adverse risk disease [15]. The major limitation is that most patients are not fit for the procedure due to their age at diagnosis and other comorbidities. Autologous HSCT is an alternative to allo-HSCT that is associated with reduced toxicity, but is only effective in patients without high risk disease and is not widely utilized in all jurisdictions [17].
While the majority of the emerging anti-AML immunotherapies seek to control disease without allo-HSCT by sparing HSPC (Figure 1), others look to targeted immunotherapy to act as a component of allo-HSCT and accept that depleting HSPC is required to control AML (Figure 2).
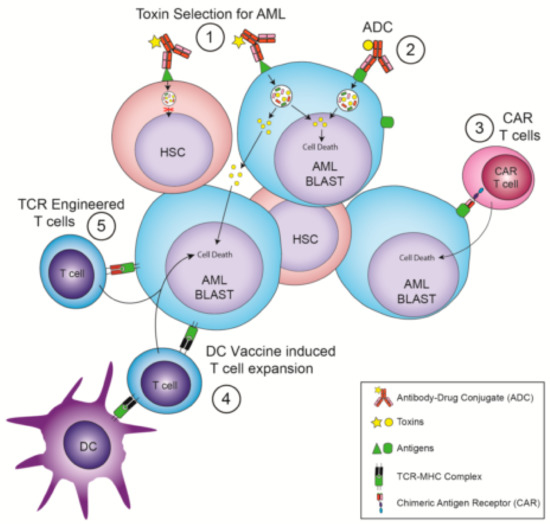
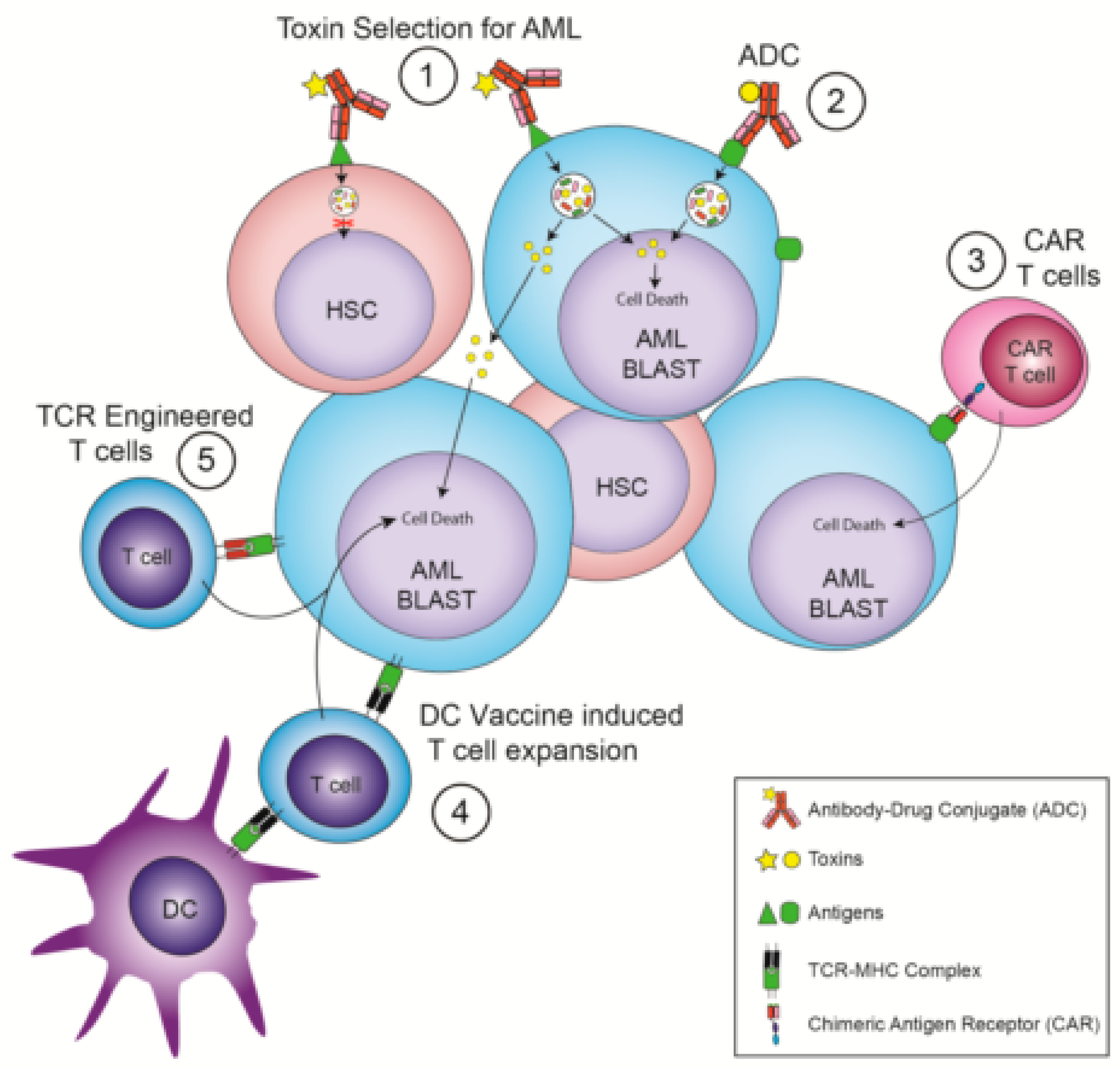
Figure 1.
Immunotherapies that avoid the need for hematopoietic stem cell transplantation. (1) Antibody Drug Conjugates (ADC) that recognize Hematopoietic Stem Cells (HSC) and Acute Myeloid Leukemia (AML) but whose toxin selectively kills AML cells. (2) ADC that recognize AML-specific surface targets. (3) Chimeric Antigen Receptor (CAR) T cells that recognize AML-specific surface targets. (4) Dendritic cell (DC) vaccination leading to a T cell response against AML-specific intracellular targets. (5) T cells expressing T-cell receptors (TCRs) engineered to recognize AML-specific intracellular antigens displayed by a Major Histocompatibility Complex (MHC) molecule.
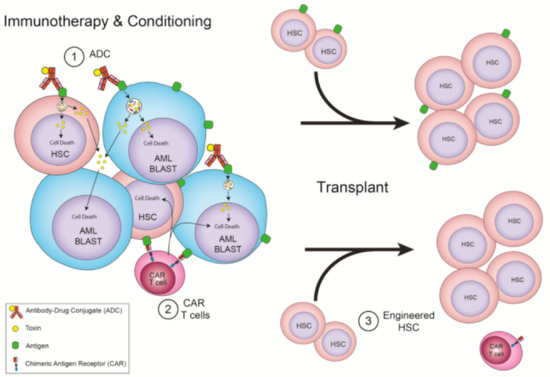
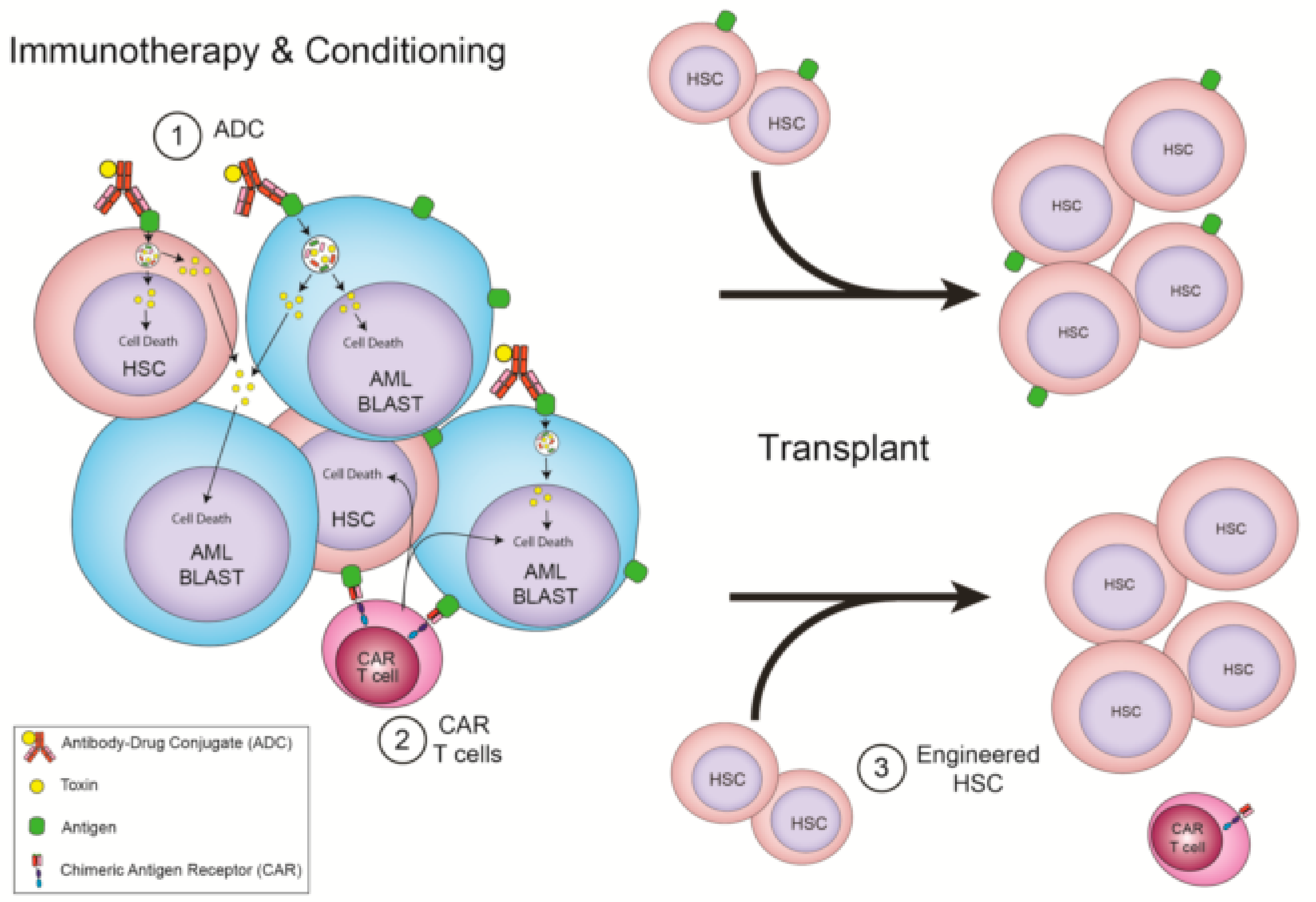
Figure 2.
Immunotherapies that require allo-HSCT. (1) ADC that recognize HSC and AML shared antigens as conditioning for a transplant. (2) CAR T cells that recognize HSC and AML shared antigens and are depleted with conditioning for a transplant. (3) CAR T cells that recognize HSC and AML shared antigens as conditioning for a transplant, but whose donor cells have the target removed, allowing for CAR T cell persistence.
Graft Versus Leukemia (GVL) in allo-HSCT provides evidence that immune mechanisms can reduce relapse risk in AML. The evidence of GVL was seen in preclinical models and confirmed clinically with the observations of reduced relapse rates in patients who develop GVHD, and an increased relapse rate in patients who receive T cell depleted grafts [18,19]. The immune mechanism of GVL is complex; incompletely understood; and involves T, NK, and antigen-presenting cells with likely multiple leukemia antigens [20]. This contrasts to current immunotherapies, which primarily target a single molecule. Immunotherapy can often play a complementary role with allo-HSCT, acting as bridge to transplant. This is seen in B-cell Acute Lymphoblastic Leukemia (B-ALL), where the ADC, Inotuzumab Ozogamicin, and Bispecific T Cell Engagers (BiTE), Blinatumomab, have been shown to eliminate leukemic cells capable of achieving Minimal Residual Disease (MRD) negativity prior to allo-HSCT [21,22]. The emergence of immunotherapies altering the treatment landscape in regard to allo-HSCT can be seen in the field of B-ALL. CD19 CAR T cells are capable of inducing sustained Complete Remission (CR), thus potentially minimizing the role of allo-HSCT in children and young adults with relapsed ALL [23]. Despite the emergence of CAR T cells in B-ALL, it is unclear if the need for allo-HSCT will be reduced over time or if these therapies will play complementary roles. Given that relapse occurs post CD19 CAR T cell therapy, the role of consolidation with an allo-HSCT is not currently established [24,25].
Optimal disease control prior to allo-HSCT in AML is an important part of reducing relapse risk. Patients with refractory disease going into to transplant have a much higher risk of relapse compared to those in CR [26,27]. Those who are MRD-positive at the time of transplant have worse outcomes [28,29,30]. Even though MRD positivity is associated with a high relapse rate post allo-HSCT, these patients may still benefit from transplant [31]. Immunotherapy in AML may contribute as a bridge to transplant, as has been established in B-ALL [21,22]. There are already emerging strategies reported of adding targeted AML agents to conventional conditioning regimens, which can reduce the relapse risk post-transplant in high-risk patients with AML [32]. As AML and HSPC share antigens, it is possible that the step of disease depletion with immunotherapy prior to allo-HSCT can also act as a form of conditioning. This application would have the advantage of depleting residual disease and reducing nonhematologic toxicity by potentially replacing nonspecific conditioning agents that eliminate host HSPC. The disadvantage of this strategy is the requirement of an allo-HSCT.
Allo-HSCT carries significant limitations. The median age of diagnosis of AML is 72, which prevents a majority of patients from accessing allo-HSCT due to treatment related toxicity [33]. Allo-HSCT requires chemotherapeutic agents as conditioning to suppress the host’s immune system while removing residual AML and HSPC. These factors are critical to allow for the safe engraftment of donor cells. The development of nonmyeloablative and Reduced Intensity Conditioning (RIC) regimens has allowed more patients to benefit from allo-HSCT by reducing the treatment-related mortality [34]. These developments have made transplantation in selected patients over the age of 65 and increasingly over the age of 70 possible, but not without significant morbidity and mortality [35,36]. In attempts to broaden the benefits of allo-HSCT, there is an emerging effort to reduce treatment related mortality from allo-HSCT by replacing traditional conditioning immunotherapy [37]. Clinical trials (NCT02963064) have been established in children with severe combined immunodeficiency receiving chemotherapy and radiation free conditioning with a CD117 antibody, with early reports demonstrating partial engraftment [38]. An antibody-based conditioning strategy against CD117 has been shown to be effective in eliminating myelodysplastic syndrome HSPC and allowing for engraftment of healthy HSPC preclinically [39]. This emerging field will likely converge with AML immunotherapy due to shared targets.
Is Disease Control Linked to Hematologic Toxicity? Implications from Targeting CD33
CD33/Siglec-3 is a protein from the immunoglobulin superfamily, with expression that ranges from myeloid progenitors to mature myeloid cells [40]. CD33 is expressed in 90–95% of AML [41,42]. Positivity is often defined by a specific median fluorescent intensity ratio by flow cytometry, using either an external control or an internal negative control such as lymphocytes [42,43,44]. Despite the vast majority of cases meeting positive median fluorescent intensity ratio criteria, a substantial proportion of cases have less than 70% AML cells expressing CD33 when examined on an individual cell level and this impacts the efficacy of immunotherapy [44]. It is accepted that CD33 is expressed on myeloid progenitors but there have been conflicting reports on the expression on Hematopoietic Stem Cells (HSC) [45,46]. Functional studies demonstrating CD33+ cells are capable of serial engraftment in immunodeficient mice suggests its expression on HSC [45].
Gemtuzumab Ozogamicin (GO) is an ADC using a calicheamicin warhead and the only currently listed form of immunotherapy for AML. Its history with regulatory authorities is long and complex. GO was given accelerated approval in 2000 by the FDA only for it to be withdrawn in 2010 [47,48]. The results of the ALFA-0701 study and a meta-analysis demonstrating improved survival in low to intermediate risk patients led to a subsequent GO approval by the FDA in 2017 for the treatment of AML in the upfront setting with a fractionated dosing schedule [48,49,50,51]. While the increased rates of sinusoidal obstruction syndrome (SOS), especially with higher doses, are the best known adverse effect of GO, hematological toxicity has been a feature of GO across several studies [48]. The results from the ALFA-0701 study using the recommended fractionated dosing schedule during induction showed prolonged neutropenia after consolidation phases as well as prolonged and persistent thrombocytopenia after induction [50]. When used outside of intensive chemotherapy, GO was associated with universal pancytopenia but did increase survival compared to best supportive care [52]. While GO has tolerable hematologic toxicity and is capable of benefiting some patients with AML, there is still a very large proportion of patients who do not benefit from it. More potent forms of immunotherapy have been targeted against CD33, and while these may have theoretical advantages in efficacy, they come at the price of increased hematologic toxicity.
Vadastuximab talirine (VT), SGN33a, is an anti-CD33 ADC utilizing a PyrroloBenzodiazepine Dimer (PBD) toxin. VT has a number of theoretical advantages over GO, including uniform drug loading, effectiveness in multidrug resistance positive cell lines, and activity against AML with adverse cytogenetics [53]. VT has displayed impressive CR rates, including MRD negativity when used in the upfront setting as well as a higher rate of response when combined with hypomethylating agents compared to historical controls [54,55]. The potential for significant myelosuppression by targeting CD33 was supported by a dose limiting toxicity (60 ug/kg) of hypocellular marrow in a phase I trial for VT [10]. The outcomes of older patients with newly diagnosed AML treated with VT monotherapy revealed that prolonged neutropenia (median 6.1 weeks) and thrombocytopenia (median 5.1 weeks) were common in those who achieved CR or CR with incomplete recovery [56]. A phase 3 trial of hypomethylating agents +/- VT was suspended after excess deaths due to infection were observed in the VT arm [57]. With increasingly potent therapeutics available, avoiding hematologic toxicity while remaining effective against AML may require an allo-HSCT as a rescue.
2. Strategies Trying to Mitigate Hematologic Toxicity
2.1. Alternate Surface Targets
While there remains several ongoing strategies to target CD33 in AML, the challenges in the development of VT highlight some of the difficulties with this target [58]. Alternate targets with reduced expression across HSPC have become appealing given the potential for reduced hematologic toxicity.
C-Type Lectin-Like Molecule 1 (CLL-1, CD371) is a surface molecule found on AML and committed myeloid progenitors [59]. A CLL-1xCD3 BiTE was broadly effective against a range of AML cell lines [60]. It induced neutropenia that recovered by day 22 in cynomolgus monkeys. This finding confirms the potential for neutropenia, but the recovery suggests a potential to spare HSC. An ADC targeting CLL-1 was effective against in AML cell lines in vitro and in vivo [61]. The ADC significantly reduces granulocytic Colony Forming Units (CFU) while sparing erythroid CFUs. This ADC does not reduce the number of human CD45+ cells in a humanized mouse model. A different ADC against CLL-1 with a PBD toxin depleted AML cell lines and primary AML in vitro and prolonged survival of mice xenografted with an AML cell line [62]. Limited preclinical hematologic toxicity was demonstrated, as the ADC caused reversible neutropenia in cynomolgus monkeys without a reduction in platelets or lymphocytes. These results suggest that an anti-CLL-1 immunotherapy may lead to transient neutropenia with a predictable recovery. While neutropenia is a significant toxicity, it is common amongst AML therapeutics and the consistent recovery across different therapeutics suggests that HSC and early progenitors are unlikely to be targeted by an anti-CLL-1 therapeutic.
T cell immunoglobulin mucin-3 (TIM-3, CD366) has emerged as a potential selective target against AML while sparing normal hematopoiesis. TIM-3 is expressed on AML blasts and is upregulated on T cells of AML patients [63,64]. TIM-3 appears to play a role in Leukemic Stem Cell (LSC) establishment, as TIM-3 antibodies can block engraftment of AML in immunocompromised mice [65]. Within HSPC, only a subset of Granulocyte/Macrophage Progenitors express TIM-3 when assessed by flow cytometry [65]. A TIM-3 antibody was able to deplete TIM-3+ monocytes in a humanized mouse model but did not impair the development of B or myeloid cells [65]. An early report of a TIM-3 antibody in combination with decitabine in hypomethylating agent naive patients has demonstrated limited toxicity but also limited efficacy to date with a CR rate of 14% (2/14) [66]. A CAR T cell has been developed that selectively kills targets expressing CD13 and TIM-3 [67]. This strategy still depleted HSPC but not as much as targeting CAR T cells directed against only CD13. Weather a limited long-term depletion of HSPC has clinically significant impacts is unknown.
Natural Killer Group 2D (NKG2D) is a surface receptor that is expressed normally on NK and CD8+ T cells, but is also expressed on stressed and malignant cells, including the majority of AML [68,69]. Its absence on resting healthy tissues makes it an attractive target. A case report from a Phase I/II trial (NCT03018405) with a patient who had relapsed FLT3+ AML who underwent NKG2D CAR T cell therapy demonstrates initial promise [70]. The patient achieved a complete morphological response but had evidence of clonal evolution with a new IDH2 mutation. Despite this finding, he had normal trilineage hematopoiesis, providing evidence of the lack of HSPC expression of NKG2D. He subsequently underwent allo-HSCT, leading to CR with normal molecular studies. It is unclear if AML subclones may have variable NKG2D expression; this case highlights the challenges in targeting a heterogeneous disease like AML with a single surface molecule. More data are required to determine if NKG2D targeted therapy can be a standalone therapy or a bridge to transplant.
CD70 is the ligand of CD27, and this interaction helps to regulate lymphocyte and HSPC activity [71]. CD70 has minimal expression on healthy HSPC and is substantially upregulated in AML as well as other malignancies [72,73]. The blockade of CD70 with a monoclonal antibody inhibits self-renewal of AML and LSC while extending survival in a xenograft model of primary AML [72]. Hypomethylating agents have been shown to induce CD70 expression further on AML, thus suggesting synergistic potential with CD70 antibodies, and a phase II trial using this combination in previously untreated AML is underway (NCT04023526) [74]. What remains unclear is if blocking the CD70/CD27 axis will affect hematopoiesis even if the HSPC themselves do not express CD70.
While many of the targets to date have been expressed on a majority of AML, there have also been efforts to target surface proteins that are aberrantly expressed on AML even if they constitute a minority of cases. CD7 is a cell surface glycoprotein that is normally expressed on T and NK cells as well as their progenitors [75]. CD7 is expressed in 30% of AML cases [76]. The lack of CD7 expression on HSPC reduces the chance for severe hematologic toxicity resulting from targeting CD7. A CAR T cell directed against CD7 depleted AML cell lines in vitro and in vivo but did not reduce CFU formation from normal cord blood [77]. The possibility of CAR T cell fratricide is reduced by editing the CD7 gene on the CAR T cells so they do not display their target [78]. This strategy would only be suitable for a minority of patients with AML, and the impact of immunosuppression from potentially long-term healthy T cell depletion is unclear.
Another method of target selection that may reduce hematologic toxicity is to selectively target markers specific for monocytic differentiation. Leukocyte immunoglobulin-like receptor-B 4 (LILRB4, CD85k, ILT3) is expressed from promonocytes to mature monocytes [79]. LILRB4 is also expressed on AML with monocytic differentiation i.e., M4 and M5 by FAB classification [80]. A CAR T cell directed against LILRB4 demonstrated efficacy against M5 AML cell lines and primary M5 AML in vitro as well as a M5 AML cell line in vivo [81]. It did not reduce CFU numbers and in a humanized mouse model did not deplete CD34+, CD33+, or CD19+ cells. M5 AML was chosen as the target, as M4 AML did not uniformly express LILRB4. The targeting of M5 AML limits the number of potential patients who may benefit as AML with monocytic differentiation accounts for only 5-10% overall, though the proportion is up to 40% in children [82,83].
CD300f is another surface target expressed across AML, mature myeloid cells, and HSPC [84,85]. There are seven isoforms of CD300f described, and it has been shown that exon 4 expression of the protein is selectively upregulated in AML with monocytic differentiation compared to healthy HSPC [85]. In addition, a monoclonal antibody can enact a conformational change of CD300f allowing a second antibody to bind to AML with monocytic differentiation at high affinity, but not to HSPC. CD300f is a promising target in AML with monocytic differentiation, but more work remains to develop a comprehensive strategy to utilize the selective expression and spare HSPC from future therapeutics.
2.2. Intracellular Targets
While AML-specific surface targets have been difficult to validate, intracellular targets have been described that are substantially upregulated in AML compared to normal populations. The best known upregulated AML antigens are WT1 and PRAME, which are found in the majority of AML samples [86]. Despite their suitability for targeting, the difficulty accessing intracellular antigens has limited their development in immunotherapy.
Dendritic cell (DC) vaccines have been the most common strategy to target AML intracellular antigens, with a wide range of manufacturing strategies and targets explored [87]. DCs loaded with AML antigens are able to stimulate T and NK cells to induce an antileukemia effect [88]. The main advantage of DC vaccination is the demonstrated favorable safety profile, which is especially important given the advanced average age at presentation [88]. The main barriers to entering routine clinical practice are the variability across different strategies and limited efficacy [87]. The type of DC used may be one of the limiting factors in vaccination efficacy. Most studies have used monocyte-derived DCs, which are less efficient at antigen presentation and the generation of T cell responses [89,90]. Using blood DC has the potential to overcome some of the limitations in DC vaccine efficacy [91].
An alternate way to target intracellular antigens and possibly increase the efficacy of treatment over DC vaccination is to use T-cell receptor (TCR)-engineered T cells. Engineered T cells can be designed to recognize intracellular antigens expressed by MHC on AML [92]. CD8+ T cells reactive against WT1 on HLA-A*02:01 are functional in vitro, are home to the bone marrow of patients, and persist after reinfusion [93]. Persistence of engineered T cells against WT1 expressed on HLA-A*24:02 has been demonstrated in an MDS setting [94]. The limited patient numbers published make the assessment of clinical efficacy difficult. A central limitation with this approach is that engineered T cells must be made to recognize a specific HLA allele, thereby restricting utility to only those patients who express the targeted HLA.
2.3. Alternate Effector
A separate strategy to reduce hematologic toxicity is to use a toxin as part of an ADC that demonstrates increased efficacy against AML compared to HSPC. It may be possible to find an effective therapeutic window where an ADC can deplete AML while sparing HSPC. IMGN632 is an ADC that targets CD123 and uses an indolinobenzodiazepine pseudodimer alkylator payload that demonstrates activity against AML cell lines and primary AML in vitro as well as AML cell lines in vivo [95]. IMGN632 had an increased therapeutic window when compared to GO with myeloid progenitor and AML CFU assays. The antibody clone of IMGN632, G4723A, was also conjugated to an alternate more potent toxin that had similar IC90 values between AML and myeloid progenitors. The alternate ADC was >40x more toxic to myeloid progenitors than IMGN632, which demonstrates the importance of the toxin over the target in providing an extended therapeutic window.
3. Strategies Incorporating Allogeneic Hematopoietic Stem Cell Transplantation
Due to the difficulties in avoiding HSPC depletion with anti-AML immunotherapies, an alternative strategy would be to incorporate these therapies into allo-HSCT, which is an established therapy for AML. Attempts to target CD123 with CAR T cells demonstrate how immunotherapies against AML may be incorporated into a transplant model. CAR T cells have shown preclinical in vitro and in vivo efficacy against AML, but there are conflicting reports on the potential for toxicity against HSPC. Some studies demonstrated no effect on myeloid progenitors, while others displayed complete myeloablation in humanized mouse models [8,96]. Depleting CAR T cells after a fixed period is a potential strategy to circumvent destruction of donor HSPC during allo-HSCT. CAR T cells can be depleted by targeting their natural or engineered surface molecules. CAR T elimination has been demonstrated with alemtuzumab, or rituximab when CD20 is introduced during CAR T cell manufacturing [97]. A functional proof of CAR T potential in allo-HSCT was demonstrated by using CD123 CAR T cells to deplete human cells in a mouse sequentially engrafted with human T cell deplete bone marrow and a human AML cell line [97]. This was followed by rituximab administration to remove the CAR T cells before a subsequent sex mismatched human donor graft [97]. “Bio-degradable” CD123 CAR T cells, which lack the ability for indefinite self-perpetuation, were administered to patients as part of a Phase I trial to exclude toxicity [92]. These CAR T cells were well tolerated but did not display efficacy against AML [92]. The correlation between persistence of CD19 CAR T cells and continued response in B-ALL suggests that a prolonged presence of CAR T cells may be required for efficacy; this may limit the effectiveness of short-term CAR T cells. There are currently clinical trials using CD123 CAR T cells in AML that do (NCT03766126) and do not (NCT04109482) require a nominated transplant donor prior to enrolling, as a precaution in the event of bone marrow aplasia.
An innovative way to allow anti-AML CAR T cells to persist is to remove a common target from HSPC. This strategy has been explored by developing CAR T cells against CD33 and then removing the CD33 from an allo-HSCT donor graft [98]. CD33-depleted myeloid cells did not display any significant developmental or functional differences, and they were able to be manufactured from both human and nonhuman primates HSPC [98]. The CD33 CAR T cells were able to deplete an AML cell line in CD33 KO humanized mice without reducing normal myelopoiesis. While this strategy may circumvent many theoretical problems with immunotherapy in AML, practical issues including cost may become limiting. CAR T persistence would likely increase efficacy but the possibility of antigen escape remains, as seen with CD19- relapses in ALL [99]. To combat this issue, immunotherapies targeting multiple antigens have been developed.
A compound CAR T cell targeting CD33 and CD123 has been developed that demonstrates independent activity against cells expressing either molecule [100]. These CAR T cells were effective in vitro against AML cells lines and were able to prolong survival of xenografted mice with AML cell lines displaying CD33, CD123, or both CD33 and CD123. To enhance safety and reduce the chance of prolonged bone marrow aplasia, the compound CAR T cells were able to be depleted in vivo using alemtuzumab. Results from Phase 1 trial (NCT03795779) of a compound CLL-1 CD33 CAR T cell have been presented. The first patient reported conversion from active disease to MRD negative with pancytopenia; this was followed by a successful matched sibling transplant [101]. A second case presentation with this construct demonstrated ablation of AML and pancytopenia as a result of CAR T cell administration followed by a allo-HSCT [101]. Given the limited efficacy detailed in anti-AML CAR T cell case reports and early phase trial results, compound targeting may represent a significant improvement on sustained efficacy against AML [92]. By incorporating an increasing number of shared targets, this strategy makes it even more likely to have significant hematological toxicity without allo-HSCT rescue.
In addition to CAR T cells, there have been reports of ADCs against CD45 and CD117 to reduce AML and conditions for an allo-HSCT [102]. These antibodies use the ribosomal inhibiting toxin amanitin and are effective at prolonging survival in mice bearing AML xenografts. Both the CD45 and CD117 amanitin ADC depleted human and nonhuman primate HSPC to confirm the potential utility in conditioning [103,104]. These results are still preliminary but offer a possible pathway to AML immunotherapy incorporating allo-HSCT that avoids the complexities of CAR T cell generation.
4. Conclusions
Effective immunotherapy for most patients with AML remains elusive. The experience with GO suggests that immunotherapy may be able to play a role in AML treatment, but the clinical trials with VT illustrate the difficulty in managing more potent therapeutics against AML with the associated increase in hematologic toxicity. Unless a true leukemia specific epitope is found that is consistently expressed across at an individual cell level, then targeting single molecules may continue to show limited efficacy. Multiantigen targeting increases the risks of hematologic toxicity but would likely reduce the chance of tumor escape.
Requiring an allo-HSCT is a major disadvantage of any potential future therapy, as most patients with AML are ineligible for allo-HSCT. The concept of bridging to allo-HSCT may change with AML immunotherapy. In other hematologic malignancies, bridging therapy occurs before conditioning; therefore, the treatment is naturally longer. In AML therapy, it may be possible to combine bridging with conditioning, which may reduce the toxicity of the individual elements combined. Another potential benefit of incorporating immunotherapy in allo-HSCT conditioning for AML is that these therapies may replace traditional conditioning agents. The consistent presentation of AML targets on HSPC may allow for an extension immunotherapy to conditioning in other diseases, including myelodysplastic syndromes as well as inherited disorders of hematopoiesis and immunity that respond poorly to traditional conditioning agents.
The design and trials of emerging AML immunotherapy must be carefully considered to have a chance of success, given the high morbidity and mortality associated with the disease itself. Tolerable and effective immunotherapy that can reduce the need for allo-HSCT would constitute a major change in the treatment of AML, but to date this has been elusive. Finally, the alternative of enhancing the efficacy while reducing the toxicity of allo-HSCT with immunotherapy would constitute substantial progress in finding better outcomes for patients with AML.
Author Contributions
This review article was conceptualized by E.A. and G.J.C.; the original draft was written by E.A.; preparation of original figures by P.A.S.; writing-review and editing by R.E.G., P.A.S., S.L. and G.J.C.; funding acquisition and supervision by S.L. and G.J.C. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Cancer Institute NSW Translational Program Grant number 2017/TPG002.
Conflicts of Interest
G.J.C. is a director of Dendrocyte Biotech Pty Ltd. and Kira Biotech Pty Ltd. There are no other conflicts of interest to declare.
References
- Coombs, C.C.; Tallman, M.S.; Levine, R.L. Molecular therapy for acute myeloid leukaemia. Nat. Rev. Clin. Oncol. 2016, 13, 305–318. [Google Scholar] [CrossRef]
- Kayser, S.; Levis, M.J. Advances in targeted therapy for acute myeloid leukaemia. Br. J. Haematol. 2018, 180, 484–500. [Google Scholar] [CrossRef] [PubMed]
- Leget, G.A.; Czuczman, M.S. Use of rituximab, the new FDA-approved antibody. Curr. Opin. Oncol. 1998, 10, 548–551. [Google Scholar] [CrossRef] [PubMed]
- Davila, M.L.; Kloss, C.C.; Gunset, G.; Sadelain, M. CD19 CAR-targeted T cells induce long-term remission and B Cell Aplasia in an immunocompetent mouse model of B cell acute lymphoblastic leukemia. PLoS ONE 2013, 8, e61338. [Google Scholar] [CrossRef] [PubMed]
- Turtle, C.J.; Hanafi, L.-A.; Berger, C.; Gooley, T.A.; Cherian, S.; Hudecek, M.; Sommermeyer, D.; Melville, K.; Pender, B.; Budiarto, T.M. CD19 CAR–T cells of defined CD4+: CD8+ composition in adult B cell ALL patients. J. Clin. Investig. 2016, 126, 2123–2138. [Google Scholar] [CrossRef] [PubMed]
- Locke, F.L.; Neelapu, S.S.; Bartlett, N.L.; Siddiqi, T.; Chavez, J.C.; Hosing, C.M.; Ghobadi, A.; Budde, L.E.; Bot, A.; Rossi, J.M. Phase 1 results of ZUMA-1: A multicenter study of KTE-C19 anti-CD19 CAR T cell therapy in refractory aggressive lymphoma. Mol. Ther. 2017, 25, 285–295. [Google Scholar] [CrossRef]
- Im, A.; Pavletic, S.Z. Immunotherapy in hematologic malignancies: Past, present, and future. J. Hematol. Oncol. 2017, 10, 94. [Google Scholar] [CrossRef]
- Gill, S.; Tasian, S.K.; Ruella, M.; Shestova, O.; Li, Y.; Porter, D.L.; Carroll, M.; Danet-Desnoyers, G.; Scholler, J.; Grupp, S.A. Preclinical targeting of human acute myeloid leukemia and myeloablation using chimeric antigen receptor–modified T cells. Blood 2014, 123, 2343–2354. [Google Scholar] [CrossRef]
- Kenderian, S.S.; Ruella, M.; Shestova, O.; Klichinsky, M.; Aikawa, V.; Morrissette, J.J.; Scholler, J.; Song, D.; Porter, D.L.; Carroll, M. CD33-specific chimeric antigen receptor T cells exhibit potent preclinical activity against human acute myeloid leukemia. Leukemia 2015, 29, 1637–1647. [Google Scholar] [CrossRef]
- Stein, E.M.; Walter, R.B.; Erba, H.P.; Fathi, A.T.; Advani, A.S.; Lancet, J.E.; Ravandi, F.; Kovacsovics, T.; DeAngelo, D.J.; Bixby, D. A phase 1 trial of vadastuximab talirine as monotherapy in patients with CD33-positive acute myeloid leukemia. Blood 2018, 131, 387–396. [Google Scholar] [CrossRef]
- Paguirigan, A.L.; Smith, J.; Meshinchi, S.; Carroll, M.; Maley, C.; Radich, J.P. Single-cell genotyping demonstrates complex clonal diversity in acute myeloid leukemia. Sci. Transl. Med. 2015, 7, 281re2. [Google Scholar] [CrossRef] [PubMed]
- Potter, N.; Miraki-Moud, F.; Ermini, L.; Titley, I.; Vijayaraghavan, G.; Papaemmanuil, E.; Campbell, P.; Gribben, J.; Taussig, D.; Greaves, M. Single cell analysis of clonal architecture in acute myeloid leukaemia. Leukemia 2019, 33, 1113–1123. [Google Scholar] [CrossRef] [PubMed]
- Ferrell, P.B., Jr.; Diggins, K.E.; Polikowsky, H.G.; Mohan, S.R.; Seegmiller, A.C.; Irish, J.M. High-dimensional analysis of acute myeloid leukemia reveals phenotypic changes in persistent cells during induction therapy. PLoS ONE 2016, 11, e0153207. [Google Scholar] [CrossRef] [PubMed]
- Majzner, R.G.; Mackall, C.L. Tumor antigen escape from CAR T-cell therapy. Cancer Discov. 2018, 8, 1219–1226. [Google Scholar] [CrossRef] [PubMed]
- Dohner, H.; Estey, E.; Grimwade, D.; Amadori, S.; Appelbaum, F.R.; Buchner, T.; Dombret, H.; Ebert, B.L.; Fenaux, P.; Larson, R.A.; et al. Diagnosis and management of AML in adults: 2017 ELN recommendations from an international expert panel. Blood 2017, 129, 424–447. [Google Scholar] [CrossRef] [PubMed]
- Gyurkocza, B.; Lazarus, H.; Giralt, S.J.B.M.T. Allogeneic hematopoietic cell transplantation in patients with AML not achieving remission: Potentially curative therapy. Bone Marrow Transplant. 2017, 52, 1083–1090. [Google Scholar] [CrossRef]
- Cornelissen, J.J.; Blaise, D. Hematopoietic stem cell transplantation for patients with AML in first complete remission. Blood 2016, 127, 62–70. [Google Scholar] [CrossRef]
- Kolb, H.-J. Graft-versus-leukemia effects of transplantation and donor lymphocytes. Blood 2008, 112, 4371–4383. [Google Scholar] [CrossRef]
- Negrin, R.S. Graft-versus-host disease versus graft-versus-leukemia. Ash Educ. Program Book 2015, 2015, 225–230. [Google Scholar] [CrossRef]
- Dickinson, A.M.; Norden, J.; Li, S.; Hromadnikova, I.; Schmid, C.; Schmetzer, H.; Jochem-Kolb, H. Graft-versus-leukemia effect following hematopoietic stem cell transplantation for leukemia. Front. Immunol. 2017, 8, 496. [Google Scholar] [CrossRef] [PubMed]
- Marks, D.I.; Kebriaei, P.; Stelljes, M.; Gökbuget, N.; Kantarjian, H.; Advani, A.S.; Merchant, A.; Stock, W.; Cassaday, R.D.; Wang, T. Outcomes of Allogeneic Stem Cell Transplantation after Inotuzumab Ozogamicin Treatment for Relapsed or Refractory Acute Lymphoblastic Leukemia. Biol. Blood Marrow Transplant. 2019, 9, 1720–1729. [Google Scholar] [CrossRef] [PubMed]
- Keating, A.K.; Gossai, N.; Phillips, C.L.; Maloney, K.; Campbell, K.; Doan, A.; Bhojwani, D.; Burke, M.J.; Verneris, M.R. Reducing minimal residual disease with blinatumomab prior to HCT for pediatric patients with acute lymphoblastic leukemia. Blood Adv. 2019, 3, 1926–1929. [Google Scholar] [CrossRef] [PubMed]
- Pehlivan, K.C.; Duncan, B.B.; Lee, D.W. CAR-T Cell Therapy for Acute Lymphoblastic Leukemia: Transforming the Treatment of Relapsed and Refractory Disease. Curr. Hematol. Malig. Rep. 2018, 13, 396–406. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Rivière, I.; Gonen, M.; Wang, X.; Sénéchal, B.; Curran, K.J.; Sauter, C.; Wang, Y.; Santomasso, B.; Mead, E. Long-term follow-up of CD19 CAR therapy in acute lymphoblastic leukemia. N. Engl. J. Med. 2018, 378, 449–459. [Google Scholar] [CrossRef]
- Kansagra, A.J.; Frey, N.V.; Bar, M.; Laetsch, T.W.; Carpenter, P.A.; Savani, B.N.; Heslop, H.E.; Bollard, C.M.; Komanduri, K.V.; Gastineau, D.A. Clinical utilization of Chimeric Antigen Receptors T-cells (CAR-T) in B-cell acute lymphoblastic leukemia (ALL)–an expert opinion from the European Society for Blood and Marrow Transplantation (EBMT) and the American Society for Blood and Marrow Transplantation (ASBMT). Elsevier 2018, 25, e76–e85. [Google Scholar]
- Duval, M.; Klein, J.P.; He, W.; Cahn, J.-Y.; Cairo, M.; Camitta, B.M.; Kamble, R.; Copelan, E.; De Lima, M.; Gupta, V. Hematopoietic stem-cell transplantation for acute leukemia in relapse or primary induction failure. J. Clin. Oncol. 2010, 28, 3730–3738. [Google Scholar] [CrossRef]
- Craddock, C.; Labopin, M.; Pillai, S.; Finke, J.; Bunjes, D.; Greinix, H.; Ehninger, G.; Steckel, N.; Zander, A.; Schwerdtfeger, R. Factors predicting outcome after unrelated donor stem cell transplantation in primary refractory acute myeloid leukaemia. Leukemia 2011, 25, 808–813. [Google Scholar] [CrossRef]
- Walter, R.B.; Gooley, T.A.; Wood, B.L.; Milano, F.; Fang, M.; Sorror, M.L.; Estey, E.H.; Salter, A.I.; Lansverk, E.; Chien, J.W. Impact of pretransplantation minimal residual disease, as detected by multiparametric flow cytometry, on outcome of myeloablative hematopoietic cell transplantation for acute myeloid leukemia. J. Clin. Oncol. 2011, 29, 1190–1197. [Google Scholar] [CrossRef]
- Delsing Malmberg, E.; Johansson Alm, S.; Nicklasson, M.; Lazarevic, V.; Ståhlman, S.; Samuelsson, T.; Lenhoff, S.; Asp, J.; Ehinger, M.; Palmqvist, L. Minimal residual disease assessed with deep sequencing of NPM1 mutations predicts relapse after allogeneic stem cell transplant in AML. Leuk. Lymphoma 2019, 60, 409–417. [Google Scholar] [CrossRef]
- Press, R.; Eickelberg, G.; Froman, A.; Yang, F.; Stentz, A.; Flatley, E.M.; Fan, G.; Lim, J.Y.; Meyers, G.; Maziarz, R. Next-generation sequencing-defined minimal residual disease before stem cell transplantation predicts acute myeloid leukemia relapse. Am. J. Hematol. 2019, 94, 902–912. [Google Scholar] [CrossRef]
- Versluis, J.; Kalin, B.; Zeijlemaker, W.; Passweg, J.; Graux, C.; Manz, M.G.; Vekemans, M.-C.; Biemond, B.J.; Legdeur, M.-C.J.; Kooy, M.v.M. Graft-versus-leukemia effect of allogeneic stem-cell transplantation and minimal residual disease in patients with acute myeloid leukemia in first complete remission. JCO Precis. Oncol. 2017, 1, 1–13. [Google Scholar] [CrossRef]
- Shelikhova, L.; Khismatullina, R.; Maschan, M.; Maschan, A.A.; Kalinina, I.; Kurnikova, E.; Muzalevsky, Y.; Kazachenok, A.; Pershin, D.; Popov, A. Targeting Bcl-2 and CD38 As Part of Personalized HSCT Conditioning Regimen in Chemorefractory Acute Leukemia in Children. Blood 2019, 132, 2084. [Google Scholar] [CrossRef]
- Juliusson, G.; Lazarevic, V.; Hörstedt, A.-S.; Hagberg, O.; Höglund, M. Acute myeloid leukemia in the real world: Why population-based registries are needed. Blood 2012, 119, 3890–3899. [Google Scholar] [CrossRef] [PubMed]
- Scott, B.L.; Pasquini, M.C.; Logan, B.R.; Wu, J.; Devine, S.M.; Porter, D.L.; Maziarz, R.T.; Warlick, E.D.; Fernandez, H.F.; Alyea, E.P. Myeloablative versus reduced-intensity hematopoietic cell transplantation for acute myeloid leukemia and myelodysplastic syndromes. J. Clin. Oncol. 2017, 35, 1154–1161. [Google Scholar] [CrossRef] [PubMed]
- Rashidi, A.; Ebadi, M.; Colditz, G.A.; DiPersio, J.F. Outcomes of allogeneic stem cell transplantation in elderly patients with acute myeloid leukemia: A systematic review and meta-analysis. Biol. Blood Marrow Transplant. 2016, 22, 651–657. [Google Scholar] [CrossRef]
- Brunner, A.M.; Kim, H.T.; Coughlin, E.; Alyea III, E.P.; Armand, P.; Ballen, K.K.; Cutler, C.; Dey, B.R.; Glotzbecker, B.; Koreth, J. Outcomes in patients age 70 or older undergoing allogeneic hematopoietic stem cell transplantation for hematologic malignancies. Biol. Blood Marrow Transplant. 2013, 19, 1374–1380. [Google Scholar] [CrossRef]
- Abadir, E.; Bryant, C.; Larsen, S.; Clark, G.J. Targeting the niche: Depleting haemopoietic stem cells with targeted therapy. Bone Marrow Transplant. 2019, 54, 961–968. [Google Scholar] [CrossRef]
- Agarwal, R.; Dvorak, C.C.; Prohaska, S.; Long-Boyle, J.; Kwon, H.-S.; Weinberg, K.I.; Le, A.; Guttman-Klein, A.; Logan, A.C.; Weissman, I.L. Toxicity-free hematopoietic stem cell engraftment achieved with anti-CD117 monoclonal antibody conditioning. Biol. Blood Marrow Transplant. 2019, 25, S92. [Google Scholar] [CrossRef]
- Pang, W.W.; Czechowicz, A.; Logan, A.C.; Bhardwaj, R.; Poyser, J.; Park, C.Y.; Weissman, I.L.; Shizuru, J.A. Anti-CD117 antibody depletes normal and myelodysplastic syndrome human hematopoietic stem cells in xenografted mice. Blood 2019, 133, 2069–2078. [Google Scholar] [CrossRef]
- Freeman, S.D.; Kelm, S.; Barber, E.K.; Crocker, P.R. Characterization of CD33 as a new member of the sialoadhesin family of cellular interaction molecules. Blood 1995, 85, 2005–2012. [Google Scholar] [CrossRef]
- Dinndorf, P.A.; Andrews, R.G.; Benjamin, D.; Ridgway, D.; Wolff, L.; Bernstein, I.D. Expression of normal myeloid-associated antigens by acute leukemia cells. Blood 1986, 67, 1048–1053. [Google Scholar] [CrossRef] [PubMed]
- Schwonzen, M.; Diehl, V.; Dellanna, M.; Staib, P. Immunophenotyping of surface antigens in acute myeloid leukemia by flow cytometry after red blood cell lysis. Leuk. Res. 2007, 31, 113–116. [Google Scholar] [CrossRef]
- Ehninger, A.; Kramer, M.; Röllig, C.; Thiede, C.; Bornhäuser, M.; Von Bonin, M.; Wermke, M.; Feldmann, A.; Bachmann, M.; Ehninger, G. Distribution and levels of cell surface expression of CD33 and CD123 in acute myeloid leukemia. Blood Cancer J. 2014, 4, e218. [Google Scholar] [CrossRef] [PubMed]
- Olombel, G.; Guerin, E.; Guy, J.; Perrot, J.-Y.; Dumezy, F.; de Labarthe, A.; Bastie, J.-N.; Legrand, O.; Raffoux, E.; Plesa, A. The level of blast CD33 expression positively impacts the effect of gemtuzumab ozogamicin in patients with acute myeloid leukemia. Blood 2016, 127, 2157–2160. [Google Scholar] [CrossRef]
- Taussig, D.C.; Pearce, D.J.; Simpson, C.; Rohatiner, A.Z.; Lister, T.A.; Kelly, G.; Luongo, J.L.; Gwenn-aël, H.; Bonnet, D. Hematopoietic stem cells express multiple myeloid markers: Implications for the origin and targeted therapy of acute myeloid leukemia. Blood 2005, 106, 4086–4092. [Google Scholar] [CrossRef]
- Laszlo, G.S.; Estey, E.H.; Walter, R.B. The past and future of CD33 as therapeutic target in acute myeloid leukemia. Blood Rev. 2014, 28, 143–153. [Google Scholar] [CrossRef]
- Egan, P.C.; Reagan, J.L. The return of gemtuzumab ozogamicin: A humanized anti-CD33 monoclonal antibody–drug conjugate for the treatment of newly diagnosed acute myeloid leukemia. Oncotargets Ther. 2018, 11, 8265. [Google Scholar] [CrossRef]
- Godwin, C.; Gale, R.; Walter, R. Gemtuzumab ozogamicin in acute myeloid leukemia. Leukemia 2017, 31, 1855–1868. [Google Scholar] [CrossRef]
- Jen, E.Y.; Ko, C.-W.; Lee, J.E.; Del Valle, P.L.; Aydanian, A.; Jewell, C.; Norsworthy, K.J.; Przepiorka, D.; Nie, L.; Liu, J. FDA Approval: Gemtuzumab ozogamicin for the treatment of adults with newly diagnosed CD33-positive acute myeloid leukemia. Clin. Cancer Res. 2018, 24, 3242–3246. [Google Scholar] [CrossRef]
- Castaigne, S.; Pautas, C.; Terré, C.; Raffoux, E.; Bordessoule, D.; Bastie, J.-N.; Legrand, O.; Thomas, X.; Turlure, P.; Reman, O. Effect of gemtuzumab ozogamicin on survival of adult patients with de-novo acute myeloid leukaemia (ALFA-0701): A randomised, open-label, phase 3 study. Lancet 2012, 379, 1508–1516. [Google Scholar] [CrossRef]
- Hills, R.K.; Castaigne, S.; Appelbaum, F.R.; Delaunay, J.; Petersdorf, S.; Othus, M.; Estey, E.H.; Dombret, H.; Chevret, S.; Ifrah, N. Addition of gemtuzumab ozogamicin to induction chemotherapy in adult patients with acute myeloid leukaemia: A meta-analysis of individual patient data from randomised controlled trials. Lancet Oncol. 2014, 15, 986–996. [Google Scholar] [CrossRef]
- Amadori, S.; Suciu, S.; Selleslag, D.; Aversa, F.; Gaidano, G.; Musso, M.; Annino, L.; Venditti, A.; Voso, M.T.; Mazzone, C. Gemtuzumab ozogamicin versus best supportive care in older patients with newly diagnosed acute myeloid leukemia unsuitable for intensive chemotherapy: Results of the randomized phase III EORTC-GIMEMA AML-19 trial. J. Clin. Oncol. 2016, 34, 972–979. [Google Scholar] [CrossRef] [PubMed]
- Sutherland, M.S.K.; Walter, R.B.; Jeffrey, S.C.; Burke, P.J.; Yu, C.; Kostner, H.; Stone, I.; Ryan, M.C.; Sussman, D.; Lyon, R.P. SGN-CD33A: A novel CD33-targeting antibody–drug conjugate using a pyrrolobenzodiazepine dimer is active in models of drug-resistant AML. Blood 2013, 122, 1455–1463. [Google Scholar] [CrossRef] [PubMed]
- Erba, H.P.; Levy, M.Y.; Vasu, S.; Stein, A.S.; Fathi, A.T.; Maris, M.B.; Advani, A.; Faderl, S.; Smith, S.E.; Wood, B.L. A phase 1b study of vadastuximab talirine in combination with 7 + 3 induction therapy for patients with newly diagnosed acute myeloid leukemia (AML). Am. Soc. Hematol. 2016, 128, 211. [Google Scholar] [CrossRef]
- Fathi, A.T.; Erba, H.P.; Lancet, J.E.; Stein, E.M.; Ravandi, F.; Faderl, S.; Walter, R.B.; Advani, A.; DeAngelo, D.J.; Kovacsovics, T.J. Vadastuximab talirine plus hypomethylating agents: A well-tolerated regimen with high remission rate in frontline older patients with acute myeloid leukemia (AML). Am. Soc. Hematol. 2016, 128, 591. [Google Scholar] [CrossRef]
- Bixby, D.L.; Stein, A.S.; Fathi, A.T.; Kovacsovics, T.J.; Levy, M.Y.; Erba, H.P.; Lancet, J.E.; Jillella, A.P.; Ravandi, F.; Walter, R.B. Vadastuximab talirine monotherapy in older patients with treatment naive CD33-positive acute myeloid leukemia (AML). Am. Soc. Hematol. 2016, 128, 590. [Google Scholar] [CrossRef]
- Assi, R.; Kantarjian, H.; Ravandi, F.; Daver, N. Immune therapies in acute myeloid leukemia: A focus on monoclonal antibodies and immune checkpoint inhibitors. Curr. Opin. Hematol. 2018, 25, 136–145. [Google Scholar] [CrossRef]
- Walter, R.B. Investigational CD33-targeted therapeutics for acute myeloid leukemia. Expert Opin. Investig. Drugs 2018, 27, 339–348. [Google Scholar] [CrossRef]
- Bakker, A.B.; van den Oudenrijn, S.; Bakker, A.Q.; Feller, N.; van Meijer, M.; Bia, J.A.; Jongeneelen, M.A.; Visser, T.J.; Bijl, N.; Geuijen, C.A. C-type lectin-like molecule-1: A novel myeloid cell surface marker associated with acute myeloid leukemia. Cancer Res. 2004, 64, 8443–8450. [Google Scholar] [CrossRef]
- Leong, S.R.; Sukumaran, S.; Hristopoulos, M.; Totpal, K.; Stainton, S.; Lu, E.; Wong, A.; Tam, L.; Newman, R.; Vuillemenot, B.R. An anti-CD3/anti–CLL-1 bispecific antibody for the treatment of acute myeloid leukemia. Blood 2017, 129, 609–618. [Google Scholar] [CrossRef]
- Jiang, Y.-P.; Liu, B.Y.; Zheng, Q.; Panuganti, S.; Chen, R.; Zhu, J.; Mishra, M.; Huang, J.; Dao-Pick, T.; Roy, S. CLT030, a leukemic stem cell–targeting CLL1 antibody-drug conjugate for treatment of acute myeloid leukemia. Blood Adv. 2018, 2, 1738–1749. [Google Scholar] [CrossRef] [PubMed]
- Zheng, B.; Yu, S.-F.; Del Rosario, G.; Leong, S.R.; Lee, G.Y.; Vij, R.; Chiu, C.; Liang, W.-C.; Wu, Y.; Chalouni, C. An Anti–CLL-1 Antibody–Drug Conjugate for the Treatment of Acute Myeloid Leukemia. Clin. Cancer Res. 2019, 25, 1358–1368. [Google Scholar] [CrossRef] [PubMed]
- Haubner, S.; Perna, F.; Köhnke, T.; Schmidt, C.; Berman, S.; Augsberger, C.; Schnorfeil, F.; Krupka, C.; Lichtenegger, F.; Liu, X. Coexpression profile of leukemic stem cell markers for combinatorial targeted therapy in AML. Leukemia 2019, 33, 64–74. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Chen, X.; Yu, X.; Zhu, Y.; Ma, C.; Xia, R.; Ma, J.; Gu, C.; Ye, L.; Wu, D. Tim-3 is highly expressed in T cells in acute myeloid leukemia and associated with clinicopathological prognostic stratification. Int. J. Clin. Exp. Pathol. 2014, 7, 6880–6888. [Google Scholar]
- Kikushige, Y.; Shima, T.; Takayanagi, S.-I.; Urata, S.; Miyamoto, T.; Iwasaki, H.; Takenaka, K.; Teshima, T.; Tanaka, T.; Inagaki, Y. TIM-3 is a promising target to selectively kill acute myeloid leukemia stem cells. Cell Stem Cell 2010, 7, 708–717. [Google Scholar] [CrossRef]
- Borate, U.; Esteve, J.; Porkka, K.; Knapper, S.; Vey, N.; Scholl, S.; Garcia-Manero, G.; Wermke, M.; Janssen, J.; Traer, E. Phase Ib Study of the Anti-TIM-3 Antibody MBG453 in Combination with Decitabine in Patients with High-Risk Myelodysplastic Syndrome (MDS) and Acute Myeloid Leukemia (AML); American Society of Hematology: Washington, DC, USA, 2019; Volume 134, 590p. [Google Scholar]
- He, X.; Feng, Z.; Ma, J.; Ling, S.; Cao, Y.; Gurung, B.; Wu, Y.; Katona, B.W.; O’Dwyer, K.P.; Siegel, D.L.J.B.J. Bi-specific and Split CAR T Cells Targeting CD13 and TIM3 Eradicate Acute Myeloid Leukemia. Blood J. 2020, 2019002779. [Google Scholar] [CrossRef]
- Raulet, D.H.; Gasser, S.; Gowen, B.G.; Deng, W.; Jung, H. Regulation of ligands for the NKG2D activating receptor. Annu. Rev. Immunol. 2013, 31, 413–441. [Google Scholar] [CrossRef]
- Hilpert, J.; Grosse-Hovest, L.; Grünebach, F.; Buechele, C.; Nuebling, T.; Raum, T.; Steinle, A.; Salih, H.R. Comprehensive analysis of NKG2D ligand expression and release in leukemia: Implications for NKG2D-mediated NK cell responses. J. Immunol. 2012, 189, 1360–1371. [Google Scholar] [CrossRef]
- Sallman, D.A.; Brayer, J.; Sagatys, E.M.; Lonez, C.; Breman, E.; Agaugué, S.; Verma, B.; Gilham, D.E.; Lehmann, F.F.; Davila, M.L. NKG2D-based chimeric antigen receptor therapy induced remission in a relapsed/refractory acute myeloid leukemia patient. Haematologica 2018, 103, e424–e426. [Google Scholar] [CrossRef]
- Nolte, M.A.; Van Olffen, R.W.; Van Gisbergen, K.P.; Van Lier, R.A. Timing and tuning of CD27–CD70 interactions: The impact of signal strength in setting the balance between adaptive responses and immunopathology. Immunol. Rev. 2009, 229, 216–231. [Google Scholar] [CrossRef]
- Riether, C.; Schürch, C.M.; Bührer, E.D.; Hinterbrandner, M.; Huguenin, A.-L.; Hoepner, S.; Zlobec, I.; Pabst, T.; Radpour, R.; Ochsenbein, A.F.J.J.o.E.M. CD70/CD27 signaling promotes blast stemness and is a viable therapeutic target in acute myeloid leukemia. J. Exp. Med. 2016, 214, 359–380. [Google Scholar] [CrossRef]
- Grewal, I.S. CD70 as a therapeutic target in human malignancies. Expert Opin. Ther. Targets 2008, 12, 341–351. [Google Scholar] [CrossRef]
- Ochsenbein, A.F.; Pabst, T.; Höpner, S.; Bacher, V.U.; Hinterbrandner, M.; Banz, Y.; Müller, R.; Manz, M.G.; Gharib, W.; Francisco, D. Targeting CD70 with Cusatuzumab Eliminates Acute Myeloid Leukemia Stem Cells in Humans; American Society of Hematology: Washington, DC, USA, 2019. [Google Scholar]
- Hoebeke, I.; De Smedt, M.; Stolz, F.; Pike-Overzet, K.; Staal, F.; Plum, J.; Leclercq, G. T-, B-and NK-lymphoid, but not myeloid cells arise from human CD34+ CD38− CD7+ common lymphoid progenitors expressing lymphoid-specific genes. Leukemia 2007, 21, 311–319. [Google Scholar] [CrossRef]
- Ogata, K.; Yokose, N.; Shioi, Y.; Ishida, Y.; Tomiyama, J.; Hamaguchi, H.; Yagasaki, F.; Bessyo, M.; Sakamaki, H.; Dan, K. Reappraisal of the clinical significance of CD7 expression in association with cytogenetics in de novo acute myeloid leukaemia. Br. J. Haematol. 2001, 115, 612–615. [Google Scholar] [CrossRef]
- Gomes-Silva, D.; Atilla, E.; Atilla, P.A.; Mo, F.; Tashiro, H.; Srinivasan, M.; Lulla, P.; Rouce, R.H.; Cabral, J.M.; Ramos, C.A. CD7 CAR T cells for the therapy of acute myeloid leukemia. Mol. Ther. 2019, 27, 272–280. [Google Scholar] [CrossRef]
- Gomes-Silva, D.; Srinivasan, M.; Sharma, S.; Lee, C.M.; Wagner, D.L.; Davis, T.H.; Rouce, R.H.; Bao, G.; Brenner, M.K.; Mamonkin, M. CD7-edited T cells expressing a CD7-specific CAR for the therapy of T-cell malignancies. Blood 2017, 130, 285–296. [Google Scholar] [CrossRef]
- Kang, X.; Kim, J.; Deng, M.; John, S.; Chen, H.; Wu, G.; Phan, H.; Zhang, C.C. Inhibitory leukocyte immunoglobulin-like receptors: Immune checkpoint proteins and tumor sustaining factors. Cell Cycle 2016, 15, 25–40. [Google Scholar] [CrossRef]
- Dobrowolska, H.; Gill, K.Z.; Serban, G.; Ivan, E.; Li, Q.; Qiao, P.; Suciu-Foca, N.; Savage, D.; Alobeid, B.; Bhagat, G. Expression of immune inhibitory receptor ILT3 in acute myeloid leukemia with monocytic differentiation. Cytom. Part B Clin. Cytom. 2013, 84, 21–29. [Google Scholar] [CrossRef]
- John, S.; Chen, H.; Deng, M.; Gui, X.; Wu, G.; Chen, W.; Li, Z.; Zhang, N.; An, Z.; Zhang, C.C. A novel anti-LILRB4 CAR-T cell for the treatment of monocytic AML. Mol. Ther. 2018, 26, 2487–2495. [Google Scholar] [CrossRef]
- Swerdlow, S.H. WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues; International Agency for Research on Cancer: Lyon, France, 2017. [Google Scholar]
- Szalontay, L.; Shad, A.T. Pediatric Acute Myeloid Leukemia: How to Improve Outcome? Curr. Pediatrics Rep. 2014, 2, 26–37. [Google Scholar] [CrossRef]
- Korver, W.; Zhao, X.; Singh, S.; Pardoux, C.; Zhao, J.; Guzman, M.; Sen, S.; Yonkovich, S.; Liu, S.; Zhan, X. Monoclonal antibodies against IREM-1: Potential for targeted therapy of AML. Leukemia 2009, 23, 1587–1597. [Google Scholar] [CrossRef] [PubMed]
- Abadir, E.; Gasiorowski, R.E.; Lai, K.; Kupresanin, F.; Romano, A.; Silveira, P.A.; Lo, T.H.; Fromm, P.D.; Kennerson, M.L.; Iland, H.J. CD300f epitopes are specific targets for acute myeloid leukemia with monocytic differentiation. Mol. Oncol. 2019, 13, 2107–2120. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; Zhu, H.; Jiang, B.; Li, J.; Lu, X.; Li, L.; Ruan, G.; Liu, Y.; Chen, S.; Huang, X. Expression patterns of WT1 and PRAME in acute myeloid leukemia patients and their usefulness for monitoring minimal residual disease. Leuk. Res. 2009, 33, 384–390. [Google Scholar] [CrossRef] [PubMed]
- Van Acker, H.H.; Versteven, M.; Lichtenegger, F.S.; Roex, G.; Campillo-Davo, D.; Lion, E.; Subklewe, M.; Van Tendeloo, V.F.; Berneman, Z.N.; Anguille, S. Dendritic cell-based immunotherapy of acute myeloid leukemia. J. Clin. Med. 2019, 8, 579. [Google Scholar] [CrossRef] [PubMed]
- Anguille, S.; Willemen, Y.; Lion, E.; Smits, E.L.; Berneman, Z.N. Dendritic cell vaccination in acute myeloid leukemia. Cytotherapy 2012, 14, 647–656. [Google Scholar] [CrossRef]
- Fromm, P.D.; Papadimitrious, M.S.; Hsu, J.L.; Van Kooten Losio, N.; Verma, N.D.; Lo, T.H.; Silveira, P.A.; Bryant, C.E.; Turtle, C.J.; Prue, R.L. CMRF-56+ blood dendritic cells loaded with mRNA induce effective antigen-specific cytotoxic T-lymphocyte responses. Oncoimmunology 2016, 5, e1168555. [Google Scholar] [CrossRef]
- Osugi, Y.; Vuckovic, S.; Hart, D.N. Myeloid blood CD11c+ dendritic cells and monocyte-derived dendritic cells differ in their ability to stimulate T lymphocytes. Blood 2002, 100, 2858–2866. [Google Scholar] [CrossRef]
- Hsu, J.L.; Bryant, C.E.; Papadimitrious, M.S.; Kong, B.; Gasiorowski, R.E.; Orellana, D.; McGuire, H.M.; Groth, B.F.d.S.; Joshua, D.E.; Ho, P.J. A blood dendritic cell vaccine for acute myeloid leukemia expands anti-tumor T cell responses at remission. Oncoimmunology 2018, 7, e1419114. [Google Scholar] [CrossRef]
- Cummins, K.D.; Gill, S. Chimeric antigen receptor T-cell therapy for acute myeloid leukemia: How close to reality? Haematologica 2019, 104, 1302–1308. [Google Scholar] [CrossRef]
- Chapuis, A.G.; Ragnarsson, G.B.; Nguyen, H.N.; Chaney, C.N.; Pufnock, J.S.; Schmitt, T.M.; Duerkopp, N.; Roberts, I.M.; Pogosov, G.L.; Ho, W.Y. Transferred WT1-reactive CD8+ T cells can mediate antileukemic activity and persist in post-transplant patients. Sci. Transl. Med. 2013, 5, ra127–ra174. [Google Scholar] [CrossRef]
- Tawara, I.; Kageyama, S.; Miyahara, Y.; Fujiwara, H.; Nishida, T.; Akatsuka, Y.; Ikeda, H.; Tanimoto, K.; Terakura, S.; Murata, M. Safety and persistence of WT1-specific T-cell receptor gene− transduced lymphocytes in patients with AML and MDS. Blood 2017, 130, 1985–1994. [Google Scholar] [CrossRef] [PubMed]
- Kovtun, Y.; Jones, G.E.; Adams, S.; Harvey, L.; Audette, C.A.; Wilhelm, A.; Bai, C.; Rui, L.; Laleau, R.; Liu, F. A CD123-targeting antibody-drug conjugate, IMGN632, designed to eradicate AML while sparing normal bone marrow cells. Blood Adv. 2018, 2, 848–858. [Google Scholar] [CrossRef] [PubMed]
- Mardiros, A.; Dos Santos, C.; McDonald, T.; Brown, C.E.; Wang, X.; Budde, L.E.; Hoffman, L.; Aguilar, B.; Chang, W.-C.; Bretzlaff, W. T cells expressing CD123-specific chimeric antigen receptors exhibit specific cytolytic effector functions and antitumor effects against human acute myeloid leukemia. Blood 2013, 122, 3138–3148. [Google Scholar] [CrossRef] [PubMed]
- Tasian, S.K.; Kenderian, S.S.; Shen, F.; Ruella, M.; Shestova, O.; Kozlowski, M.; Li, Y.; Schrank-Hacker, A.; Morrissette, J.J.; Carroll, M. Optimized depletion of chimeric antigen receptor T cells in murine xenograft models of human acute myeloid leukemia. Blood 2017, 129, 2395–2407. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.Y.; Yu, K.-R.; Kenderian, S.S.; Ruella, M.; Chen, S.; Shin, T.-H.; Aljanahi, A.A.; Schreeder, D.; Klichinsky, M.; Shestova, O. Genetic inactivation of CD33 in hematopoietic stem cells to enable CAR T cell immunotherapy for acute myeloid leukemia. Cell 2018, 173, 1439–1453. [Google Scholar] [CrossRef]
- Ruella, M.; Maus, M.V. Catch me if you can: Leukemia escape after CD19-directed T cell immunotherapies. Comput. Struct. Biotechnol. J. 2016, 14, 357–362. [Google Scholar] [CrossRef]
- Petrov, J.C.; Wada, M.; Pinz, K.G.; Yan, L.E.; Chen, K.H.; Shuai, X.; Liu, H.; Chen, X.; Leung, L.-H.; Salman, H. Compound CAR T-cells as a double-pronged approach for treating acute myeloid leukemia. Leukemia 2018, 32, 1317–1326. [Google Scholar] [CrossRef]
- Liu, F.; Cao, Y.; Pinz, K.; Ma, Y.; Wada, M.; Chen, K.; Ma, G.; Shen, J.; Tse, C.O.; Su, Y. First-in-human CLL1-CD33 compound CAR T cell therapy induces complete remission in patients with refractory acute myeloid leukemia: Update on phase 1 clinical trial. Am. Soc. Hematol. 2018, 132, 901. [Google Scholar] [CrossRef]
- Proctor, J.L.; Hyzy, S.L.; Adams, H.L.; Brooks, M.; Gabros, A.D.; McDonough, S.M.; Kien, L.; Aslanian, S.; Pearse, B.R.; Palchaudhuri, R. Single Doses of Antibody Drug Conjugates (ADCs) Targeted to CD117 or CD45 Have Potent In Vivo Anti-Leukemia Activity and Survival Benefit in Patient Derived AML Models. Biol. Blood Marrow Transplant. 2019, 25, S100–S101. [Google Scholar] [CrossRef]
- Palchaudhuri, R.; Pearse, B.R.; Proctor, J.L.; Hyzy, S.L.; Aslanian, S.; Bhat, A.; Gillard, G.O.; McDonough, S.M.; Sarma, G.N.; Ladwig, D. Non-Genotoxic Conditioning Using Amanitin Antibody-Drug Conjugates Targeting CD45 Effectively Deplete Human and Non-Human Primate Hematopoietic Stem Cells and Immune Cells. Biol. Blood Marrow Transplant. 2019, 25, S32. [Google Scholar] [CrossRef]
- Pearse, B.R.; McDonough, S.M.; Proctor, J.L.; Panwar, R.; Sarma, G.N.; Kien, L.; Dushime, J.; Adams, H.L.; Hyzy, S.L.; Brooks, M. A CD117-Amanitin Antibody Drug Conjugate (ADC) Effectively Depletes Human and Non-Human Primate Hematopoietic Stem and Progenitor Cells (HSPCs): Targeted Non-Genotoxic Conditioning for Bone Marrow Transplant. Biol. Blood Marrow Transplant. 2019, 25, S29–S30. [Google Scholar] [CrossRef][Green Version]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).