Apical Periodontitis and Diabetes Mellitus Type 2: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Methodology
2.1. Focused Question
2.2. Search Strategy
2.3. Data Extraction and Analysis
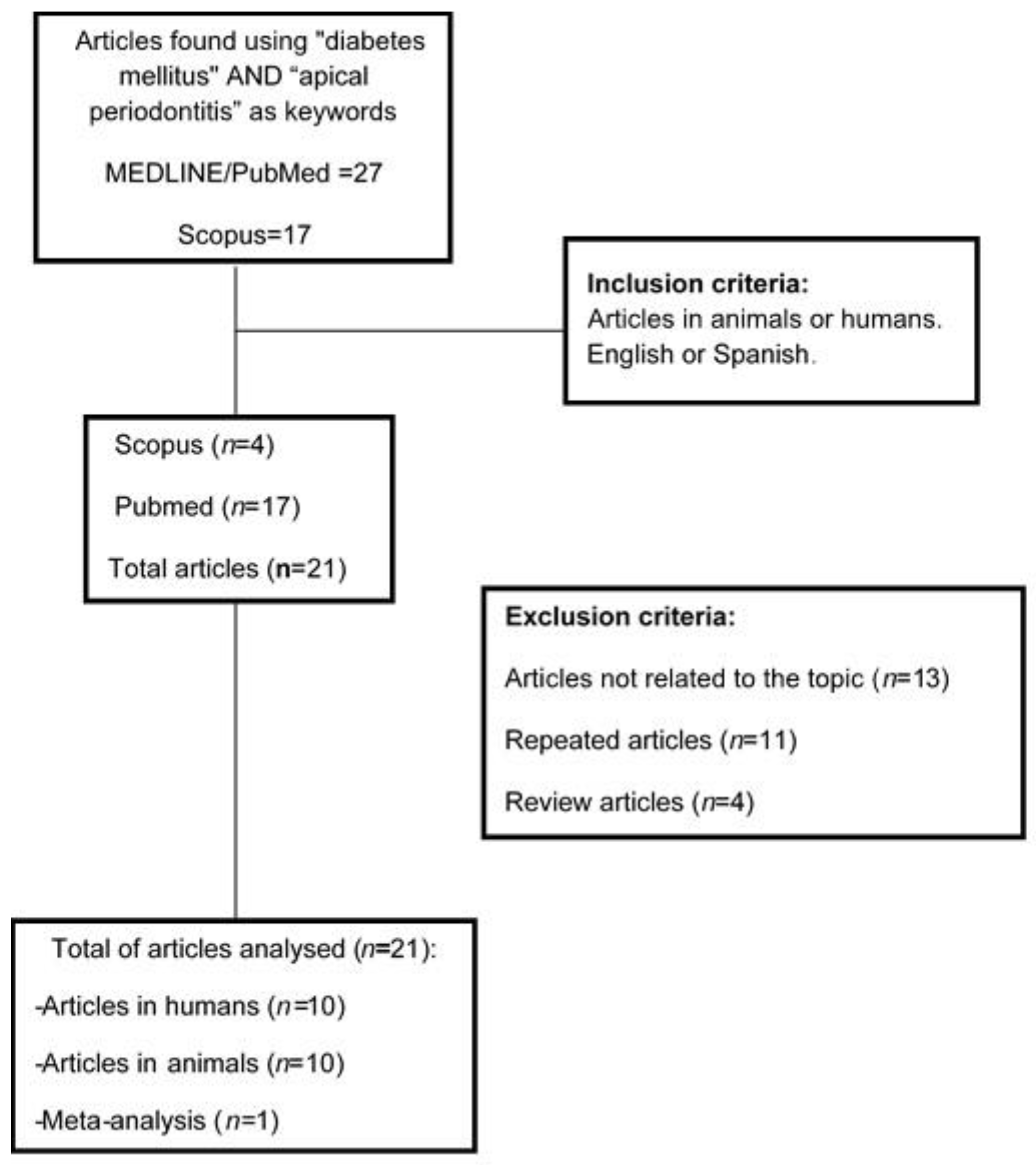
3. Results
3.1. Study Selection
3.2. Quality Assessment
4. Discussion
4.1. Animal Studies
4.2. Human Studies
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zheng, Y.; Ley, S.H.; Hu, F.B. Global aetiology and epidemiology of type 2 diabetes mellitus and its complications. Nat. Rev. Endocrinol. 2018, 14, 88–98. [Google Scholar] [CrossRef] [PubMed]
- Grant, R.W.; Kirkman, M.S. Trends in the evidencelevel for the american diabetes association’s ‘standards of medical care in diabetes’ from 2005 to 2014. Diabetes Care 2015, 38, 6–8. [Google Scholar] [CrossRef] [PubMed]
- Segura-Egea, J.J.; Jiménez-Pinzón, A.; Poyato-Ferrera, M.; Velasco-Ortega, E.; Ríos-Santos, J.V. Periapical status and quality of root fillings and coronal restorations in an adult Spanish population. Int. Endod. J. 2004, 37, 525–530. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association. Standards of medical care in diabetes—2014. Diabetes Care 2014, 37 (Suppl. 1), S14–S80. [Google Scholar] [CrossRef]
- Espelt, A.; Borrell, C.; Palència, L.; Goday, A.; Spadea, T.; Gnavi, R.; Font-Ribera, L.; Kunst, A.E. Socioeconomic inequalities in the incidence and prevalence of type 2 diabetes mellitus in Europe. Gac. Sanit. 2013, 27, 494–501. [Google Scholar] [CrossRef]
- Tunes, R.S.; Foss-Freitas, M.C.; Da Rocha Nogueira-Filho, G. Impact of periodontitis on the diabetes-related inflammatory status. J. Can. Dent. Assoc. 2010, 76, a35. [Google Scholar]
- Montoya-Carralero, J.M.; Saura-Pérez, M.; Canteras-Jordana, M.; Morata-Murcia, I.M. Reduction of HbA1c levels following nonsurgical treatment of periodontal disease in type 2 diabetics. Med. Oral Patol. Oral Cir. Bucal 2010, 15, e808–e812. [Google Scholar] [CrossRef]
- Segura-Egea, J.J.; Martín-González, J.; Castellanos-Cosano, L. Endodontic medicine: Connections between apical periodontitis and systemic diseases. Int. Endod. J. 2015, 48, 933–951. [Google Scholar] [CrossRef]
- Segura-Egea, J.-J.; Castellanos-Cosano, L.; Machuca, G.; López-López, J.; Martín-González, J.; Velasco-Ortega, E.; Sánchez-Domínguez, B.; López-Frías, F.J. Diabetes mellitus, periapical inflammation and endodontic treatment outcome. Med. Oral Patol. Oral Cir. Bucal. 2012, 17, e356–e361. [Google Scholar] [CrossRef]
- Verhulst, M.J.L.; Loos, B.G.; Gerdes, V.E.A.; Teeuw, W.J. Evaluating All Potential Oral Complications of Diabetes Mellitus. Front. Endocrinol. (Lausanne) 2019, 10, 56. [Google Scholar] [CrossRef]
- Segura-Egea, J.J.; Martín-González, J.; Cabanillas-Balsera, D.; Fouad, A.F.; Velasco-Ortega, E.; López-López, J. Association between diabetes and the prevalence of radiolucent periapical lesions in root-filled teeth: Systematic review and meta-analysis. Clin. Oral Investig. 2016, 20, 1133–1141. [Google Scholar] [CrossRef] [PubMed]
- Khalighinejad, N.; Aminoshariae, M.R.; Aminoshariae, A.; Kulild, J.C.; Mickel, A.; Fouad, A.F. Association between Systemic Diseases and Apical Periodontitis. J. Endod. 2016, 42, 1427–1434. [Google Scholar] [CrossRef] [PubMed]
- Khalighinejad, N.; Aminoshariae, A.; Kulild, J.C.; Mickel, A. Apical Periodontitis, a Predictor Variable for Preeclampsia: A Case-control Study. J. Endod. 2017, 43, 1611–1614. [Google Scholar] [CrossRef] [PubMed]
- Cintra, L.T.A.; Estrela, C.; Azuma, M.M.; Queiroz, Í.O.A.; Kawai, T.; Gomes-Filho, J.E. Endodontic medicine: Interrelationships among apical periodontitis, systemic disorders, and tissue responses of dental materials. Braz. Oral Res. 2018, 32, e68. [Google Scholar] [CrossRef]
- Poyato-Borrego, M.; Segura-Sampedro, J.J.; Martín-González, J.; Torres-Domínguez, Y.; Velasco-Ortega, E.; Segura-Egea, J.J. High Prevalence of Apical Periodontitis in Patients With Inflammatory Bowel Disease: An Age- and Gender- matched Case-control Study. Inflamm. Bowel Dis. 2020, 26, 273–279. [Google Scholar] [CrossRef]
- Castellanos-Cosano, L.; Machuca-Portillo, G.; Sánchez-Domínguez, B.; Torrés-Lagares, D.; López-López, J.; Segura-Egea, J.J. High prevalence of radiolucent periapical lesions amongst patients with inherited coagulation disorders. Haemophilia 2013, 19, 110–115. [Google Scholar] [CrossRef] [PubMed]
- Segura-Egea, J.J.; Cabanillas-Balsera, D.; Jiménez-Sánchez, M.C.; Martín-González, J. Endodontics and diabetes: Association versus causation. Int. Endod. J. 2019, 52, 790–802. [Google Scholar] [CrossRef]
- López-López, J.; Jané-Salas, E.; Estrugo-Devesa, A.; Castellanos-Cosano, L.; Martín-González, J.; Velasco-Ortega, E.; Segura-Egea, J.J. Frequency and distribution of root-filled teeth and apical periodontitis in an adult population of Barcelona, Spain. Int. Dent. J. 2012, 62, 40–46. [Google Scholar]
- Janket, S.J.; Jones, J.A.; Meurman, J.H.; Baird, A.E.; van Dyke, T.E. Oral infection, hyperglycemia, and endothelial dysfunction. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 2008, 105, 173–179. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Centre for Evidence Based Medicine. Critical Appraisal for Therapy Articles; University of Oxford Medical: Oxford, UK, 2011. Available online: https://www.cebm.net/wp-content/uploads/2014/06/CEBM-Levels-of-Evidence-2.1.pdf (accessed on 2 October 2019).
- Nakahara, Y.; Sano, T.; Kodama, Y.; Ozaki, K.; Matsuura, T. Alloxan-induced hyperglycemia causes rapid-onset and progressive dental caries and periodontitis in F344 rats. Histol. Histopathol. 2012, 27, 1297–1306. [Google Scholar] [PubMed]
- Wolle, C.F.; Zollmann, L.A.; Bairros, P.O.; Etges, A.; Leite, C.E.; Morrone, F.B.; Campos, M.M. Outcome of periapical lesions in a rat model of type 2 diabetes: Refractoriness to systemic antioxidant therapy. J. Endod. 2013, 39, 643–647. [Google Scholar] [CrossRef] [PubMed]
- Cintra, L.T.; Samuel, R.O.; Facundo, A.C.; Prieto, A.K.; Sumida, D.H.; Bomfim, S.R.; Souza, J.C.; Dezan-Júnior, E.; Gomes-Filho, J.E. Relationships between oral infections and blood glucose concentrations or HbA1c levels in normal and diabetic rats. Int. Endod. J. 2014, 47, 228–237. [Google Scholar] [CrossRef] [PubMed]
- Cintra, L.T.; Samuel, R.O.; Azuma, M.M.; Ribeiro, C.P.; Narciso, L.G.; de Lima, V.M.; Sumida, D.H.; Coclete, G.A.; Dezan-Júnior, E.; Gomes-Filho, J.E. Apical periodontitis and periodontal disease increase serum IL-17 levels in normoglycemic and diabetic rats. Clin. Oral Investig. 2014, 18, 2123–2128. [Google Scholar] [CrossRef]
- Cintra, L.T.; da Silva Facundo, A.C.; Prieto, A.K.; Sumida, D.H.; Narciso, L.G.; Bomfim, S.R.; e Silva, C.O.; Dezan-Júnior, E.; Gomes-Filho, J.E. Blood profile and histology in oral infections associated with diabetes. J. Endod. 2014, 40, 1139–1144. [Google Scholar] [CrossRef]
- Azuma, M.M.; Gomes-Filho, J.E.; Prieto, A.K.; Samuel, R.O.; de Lima, V.M.F.; Sumida, D.H.; Ervolino, E.; Cintra, L.T.A. Diabetes increases interleukin-17 levels in periapical, hepatic, and renal tissues in rats. Arch. Oral Biol. 2017, 83, 230–235. [Google Scholar] [CrossRef]
- Prieto, A.K.C.; Gomes-Filho, J.E.; Azuma, M.M.; Sivieri-Araújo, G.; Narciso, L.G.; Souza, J.C.; Ciarlini, P.C.; Cintra, L.T. Influence of Apical Periodontitis on Stress Oxidative Parameters in Diabetic Rats. J. Endod. 2017, 43, 1651–1656. [Google Scholar] [CrossRef]
- Ferreira, L.L.; Gomes Filho, J.E.F.; Sumida, D.H.; Bonfim, S.R.; Sivieri-Araújo, G.; Guimarães, G.; Cintra, L.T.A. Diabetic Rats Present High Mean Platelet Count in the Presence of Oral Infections. Braz. Dent. J. 2017, 28, 548–551. [Google Scholar] [CrossRef]
- Ng, Y.-L.; Mann, V.; Gulabivala, K. A prospective study of the factors affecting outcomes of non-surgical root canal treatment: Part 2: Tooth survival. Int. Endod. J. 2011, 44, 610–625. [Google Scholar] [CrossRef]
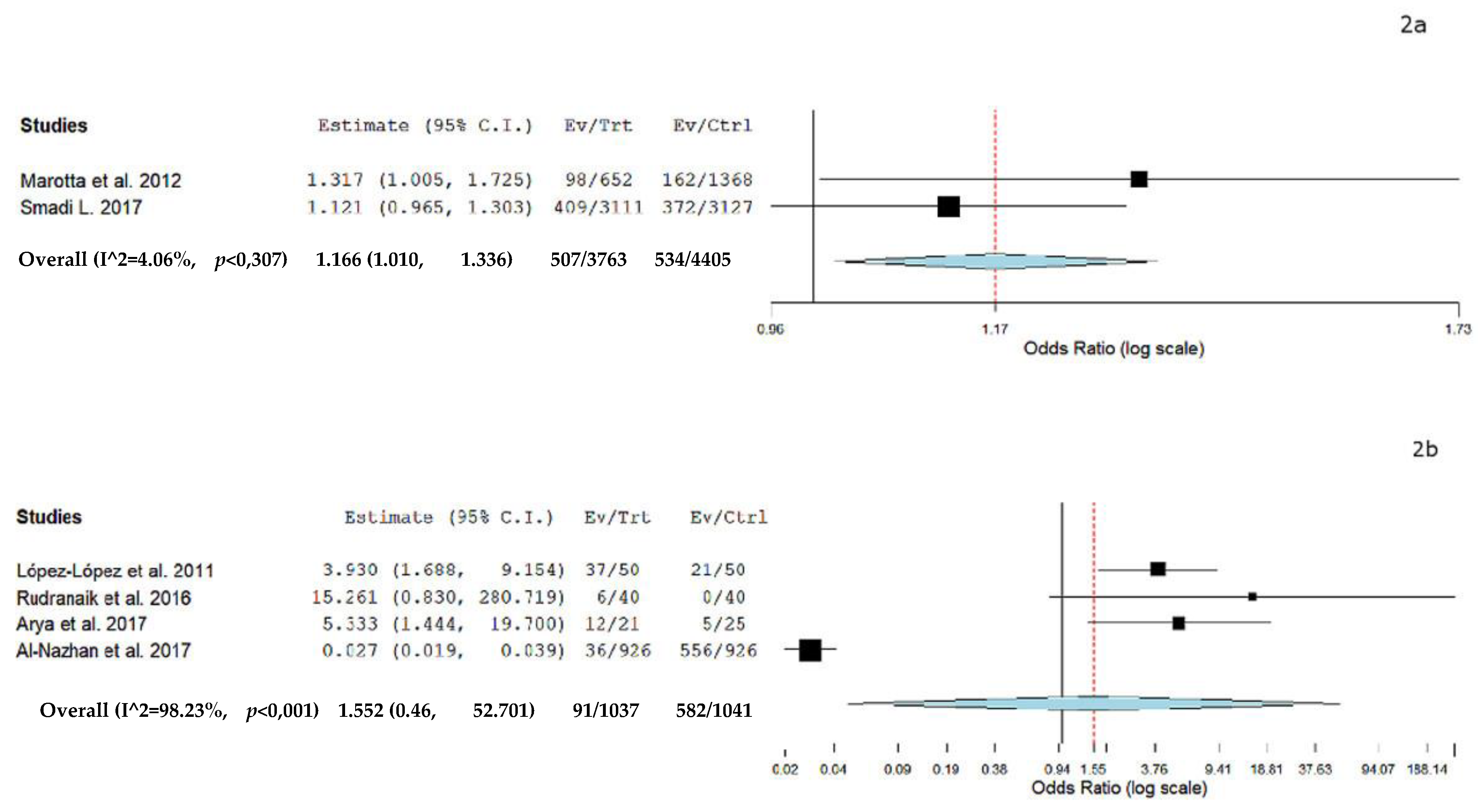
- Marotta, P.S.; Fontes, T.V.; Armada, L.; Lima, K.C.; Rôças, I.N.; Siqueira, J.F., Jr. Type 2 diabetes mellitus and the prevalence of apical periodontitis and endodontic treatment in an adult brazilian population. J. Endod. 2012, 38, 297–300. [Google Scholar] [CrossRef]
- Ferreira, M.M.; Carrilho, E.; Carrilho, F. Diabetes mellitus and its influence on the success of endodontic treatment: A retrospective clinical study. Acta Med. Port. 2014, 27, 15–22. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sánchez-Domínguez, B.; López-López, J.; Jané-Salas, E.; Castellanos-Cosano, L.; Velasco-Ortega, E.; Segura-Egea, J.J. Glycated hemoglobin levels and prevalence of apical periodontitis in type 2 diabetic patients. J. Endod. 2015, 41, 601–606. [Google Scholar] [CrossRef] [PubMed]
- Rudranaik, S.; Nayak, M.; Babshet, M. Periapical healing outcome following single visit endodontic treatment in patients with type 2 diabetes mellitus. J. Clin. Exp. Dent. 2016, 8, e498–e504. [Google Scholar] [CrossRef] [PubMed]
- Smadi, L. Apical Periodontitis and Endodontic Treatment in Patients with Type II Diabetes Mellitus: Comparative Cross-sectional Survey. J. Contemp. Dent. Pract. 2017, 18, 358–362. [Google Scholar] [PubMed]
- Arya, S.; Duhan, J.; Tewari, S.; Sangwan, P.; Ghalaut, V.; Aggarwal, S. Healing of Apical Periodontitis after Nonsurgical Treatment in Patients with Type 2 Diabetes. J. Endod. 2017, 43, 1623–1627. [Google Scholar] [CrossRef] [PubMed]
- Al-Nazhan, S.A.; Alsaeed, S.A.; Al-Attas, H.A.; Dohaithem, A.J.; Al-Serhan, M.S.; Al-Maflehi, N.S. Prevalence of apical periodontitis and quality of root canal treatment in an adult Saudi population. Saudi Med. J. 2017, 38, 413–421. [Google Scholar] [CrossRef] [PubMed]
- Sano, T.; Matsuura, T.; Ozaki, K.; Narama, I. Dental Caries and Caries-Related Periodontitis in Type 2 Diabetic Mice. Vet. Pathol. 2011, 48, 506–512. [Google Scholar] [CrossRef]
- Kodama, Y.; Matsuura, M.; Sano, T.; Nakahara, Y.; Ozaki, K.; Narama, I.; Matsuura, T. Diabetes enhances dental caries and apical periodontitis in caries-susceptible WBN/KobSlc rats. Comp. Med. 2011, 61, 53–59. [Google Scholar]
- López-López, J.; Jané-Salas, E.; Estrugo-Devesa, A.; Velasco-Ortega, E.; Martín-González, J.; Segura-Egea, J.J. Periapical and endodontic status of type 2 diabetic patients in Catalonia, Spain: A cross-sectional study. J. Endod. 2011, 37, 598–601. [Google Scholar] [CrossRef]
- Mesgarani, A.; Eshkevari, N.; Ehsani, M.; Khafri, S.; Nafarzade, S.; Damankesh, Z. Frequency of odontogenic periradicular lesions in diabetic patients. Casp. J. Intern. Med. 2014, 5, 22–25. [Google Scholar]
- Samuel, R.O.; Ervolino, E.; Queiroz, Í.O.; Azuma, M.M.; Ferreira, G.T.; Cintra, L.T.A. Th1/Th2/Th17/Treg Balance in Apical Periodontitis of Normoglycemic and Diabetic Rats. J. Endod. 2019, 45, 1009–1015. [Google Scholar] [CrossRef] [PubMed]
- Cabanillas-Balsera, D.; Martín-González, J.; Montero-Miralles, P.; Sánchez-Domínguez, B.; Jiménez-Sánchez, M.C.; Segura-Egea, J.J. Association between diabetes and nonretention of root filled teeth: A systematic review and meta-analysis. Int. Endod. J. 2019, 52, 297–306. [Google Scholar] [CrossRef] [PubMed]
- Nagendrababu, V.; Segura-Egea, J.J.; Fouad, A.F.; Pulikkotil, S.J.; Dummer, P.M.H. Association between diabetes and the outcome of root canal treatment in adults: An umbrella review. Int. Endod. J. 2019. [Google Scholar] [CrossRef] [PubMed]
| Author, Year | Level of Evidence/Degree of Recommendation | Sample and Groups | Association AP and DM | Results |
|---|---|---|---|---|
| Kodama et al., 2011 [39] | 2b/B | F344 Mice, n = 40 DM Mice: ♂ (n = 10); ♀ (n = 10) Non-DM Mice: ♂ (n = 10); ♀ (n = 10) | Yes | The incidence and severity of caries, alveolar bone resorption and periapical lesions were higher in rats with chronic DM. |
| Sano et al., 2011 [38] | 3b/B | n = 88 Mice Mice ♂: n = 68 (DM and no-DM) Mice ♀: n = 20 (DB and no-DM) | Yes | Diabetes increase dental caries and suggest that apical periodontitis is secondary to dental caries in non-DM mice. |
| Nakahara et al., 2012 [22] | 3b/B | ♀ F344 Mice, n = 47 TG: n = 30 (1 dose of Aloxane, 35 mg/kg corporal weight) CG: n = 17 untreated | Yes | Mice treated with Aloxane developed more carious lesions and progressive periodontitis. |
| Wolle et al., 2013 [23] | 1b/A | ♂ Wistar Mice, n = 20 TG: n = 15; mice with DM (receive 20% D-Glucosein drinking water, 10 mL/kg/9 weeks) GC: n = 5; no-DM mice (receive filtered/9 weeks) | No | No significant differences were found between groups. Tempol does not improve the outcome of injuries associated with endodontic teeth. |
| Cintra, et al., 2014 [24] | 2b/B | Albinos Wistar Mice, n = 80 (10 each group) # G1: GC; G2:AP; G3: PD; G4: AP + PD; G5: DM, G6: DM + AP; G7:DM + PD; G8: DM + AP + PD | Yes | Mice with oral pathology (AP + PD) had a significant increase in IL-17 compared to mice without oral pathology. |
| Cintra, et al., 2014 [25] | 2b/B | Albinos Wistar Mice, n = 80 (10 each group) # G1: GC; G2:AP; G3: PD; G4: AP + PD; G5: DM, G6: DM + AP; G7:DM + PD; G8: DM + AP + PD | Yes | Oral infections affect blood sugar levels in DM mice and increase HbA1c levels in DM and normoglycemic mice. |
| Cintra, et al., 2014 [26] | 2b/B | Albinos Wistar Mice, n = 80 (10 each group) # G1: GC; G2:AP; G3: PD; G4: AP + PD; G5: DM, G6: DM + AP; G7:DM + PD; G8: DM + AP + PD | Yes | DM increases the development and progression of AP and PD, causing an increase in the cellular mean of erythrocytes, leukocytes and neutrophils. Both oral infections increased the total number of leukocytes, neutrophils, lymphocytes and glucose concentrations in mice with DM. |
| Azuma et al., 2017 [27] | 2b/B | ♀ Winstar mice n = 40 (10 each group) Normoglycemic rats (N); Normoglycemic rats with apical periodontitis (N-AP); rats with experimental diabetes (ED), and rats with experimental diabetes and apical periodontitis (ED-AP) | Nr | AP did not impact the levels of IL-17 in hepatic and renal tissues, irrespective of the presence or absence of diabetes. There is an increase of IL-17 levels in the periapical region of diabetic rats without AP (ED) as compared to control rats (N), and an increase of IL-17 levels in AP rats with experimental diabetes (ED-AP) was observed as compared to the N-AP. |
| Prieto et al., 2017 [28] | 2b/B | ♀ Winstar mice n = 40 (10 each group) G1:CG, G2: AP, G3: DM, G4: DM + AP | Yes | Microscopically in the groups with AP (AP and DM + AP), an increase in the intensity and extent of the inflammatory infiltrate was noted, periapical lesions in the diabetic rats were higher and more aggressive compared with that in normoglycemic rats, AP associated with diabetes reduced the serum levels of albumin and increased the endogenous antioxidant uric acid. |
| Ferreira et al., 2017 [29] | 2b/B | ♀ Winstar mice n = 80 (10 each group) G1:CG, G2: AP, G3:PD, G4:AP + PD, G5:DM, G6: DM + AP, G7: DM + PD, G8:DM + AP + PD | Nr | The presence of oral infections increased blood glucose concentrations in diabetic rats. DM + PD and DM + AP + PD groups had higher mean values of platelet count with statistical difference compared to CG and AP groups. |
| Author, Year | Level of Evidence/Degree of Recommendation | Sample and Groups | Association AP and DM | Results |
|---|---|---|---|---|
| López-López et al., 2011 [40] | 3b/B/CS | n = 100 TG: n = 50, patients with DM2 CG: n = 50, patients without DM | Yes | DM2 is significantly associated with a higher prevalence of AP and root canal treatment. |
| NG YL et al., 2011 [30] | 2b/B/ PCT | n = 1617 teeth in 1214 patients. Group 1: Primary RCT Group 2: Secondary RCT | Nr | A 95% survival of primary and secondary root canal treatment after 4 years was found with 13 common factors, DM being one of the significant patient’s factors. |
| Marota et al., 2012 [31] | 3b/B/CS | n = 90 TG: n = 30, patients with DM2 CG: n = 60, patients without DM | Yes | A higher prevalence was found in patients with DM than in patients without previous history of DM. |
| Ferreira et al., 2014 [32] | 3b/B/RCT | n = 62 TG: n = 37 teeth CG: n = 25 teeth | No | No significant difference was found between both groups for AP. |
| Mesgarani et al., 2014 [41] | 3b/B/CS | n = 122 Long-term DM patients (>48 months): n = 85 Short-term DM patients (<48 months): n = 37 | Yes | The frequency of AP was more significant in patients with long-term DM than in those with short-term DM. |
| Sánchez-Dominguez et al., 2015 [33] | 3b/B/CS | n = 83 CG: n = 24, good control DM (HbA1c < 6.5%) TG: n = 59, poor control DM (HbA1c > 6.5%) | Yes | AP is significantly associated with HbA1c levels in patients with DM and root canal treatment. |
| Rudranaik et al., 2016 [34] | 2b/B/PCT | n = 80 Group 1: n = 40, patients without DM Group 2: n = 40, patients with DM2 | Nr | Patients with diabetes were more prone for chronic periapical disease with larger lesions. Healing outcome at one year was unsuccessful in poor controlled patients with diabetes when compared to fair and good controlled patients in group 2. |
| Segura-Egea et al., 2016 [11] | 1a/A/M | n = 7 Epidemiological studies: 1593 teeth with root canal treatment GT: n = 582 patients with DM GC: n = 1011 patients without DM | Yes | AP is significantly associated with diabetes with a higher prevalence of periapical lesions on the teeth with root canal treatment. |
| Smadi L. 2017 [35] | 3b/B/CS | n = 291 patients Group 1: n = 145 patients; n = 3111; 409 teeth with DM + AP Group 2: n = 146 patients; n = 3127; 372 teeth with No DM + AP | Nr | A higher prevalence of AP in DM patients but this difference was not statistically significant. |
| Arya et al., 2017 [36] | 2b/B/PCT | n = 46 Group 1: 21 patients with DM/12 with DM + AP Group 2: 25 patients without DM/5 No DM + AP | Nr | Periapical healing showed a significantly lower success rate at 1 year follow up in the type 2 diabetic group than the nondiabetic group. However, even among type 2 diabetics, 90% of cases showed an improved periapical status. No significant difference in healing between good controlled and poor controlled patients or those with raised HbA1c levels was found. |
| Al-Nazhan et al., 2017 [37] | 3b/B/CS | n = 926 patients 36 patients with DM + AP 556 with no DM + AP | Nr | This study revealed a higher prevalence of AP in diabetic subjects, although the sample of diabetic patients was small. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pérez-Losada, F.d.L.; Estrugo-Devesa, A.; Castellanos-Cosano, L.; Segura-Egea, J.J.; López-López, J.; Velasco-Ortega, E. Apical Periodontitis and Diabetes Mellitus Type 2: A Systematic Review and Meta-Analysis. J. Clin. Med. 2020, 9, 540. https://doi.org/10.3390/jcm9020540
Pérez-Losada FdL, Estrugo-Devesa A, Castellanos-Cosano L, Segura-Egea JJ, López-López J, Velasco-Ortega E. Apical Periodontitis and Diabetes Mellitus Type 2: A Systematic Review and Meta-Analysis. Journal of Clinical Medicine. 2020; 9(2):540. https://doi.org/10.3390/jcm9020540
Chicago/Turabian StylePérez-Losada, Flor de Liz, Albert Estrugo-Devesa, Lissett Castellanos-Cosano, Juan José Segura-Egea, José López-López, and Eugenio Velasco-Ortega. 2020. "Apical Periodontitis and Diabetes Mellitus Type 2: A Systematic Review and Meta-Analysis" Journal of Clinical Medicine 9, no. 2: 540. https://doi.org/10.3390/jcm9020540
APA StylePérez-Losada, F. d. L., Estrugo-Devesa, A., Castellanos-Cosano, L., Segura-Egea, J. J., López-López, J., & Velasco-Ortega, E. (2020). Apical Periodontitis and Diabetes Mellitus Type 2: A Systematic Review and Meta-Analysis. Journal of Clinical Medicine, 9(2), 540. https://doi.org/10.3390/jcm9020540








