High Plasma Branched-Chain Amino Acids Are Associated with Higher Risk of Post-Transplant Diabetes Mellitus in Renal Transplant Recipients
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Population
2.2. Data Collection
2.3. Quantification of BCAAs
2.4. Clinical Endpoints
2.5. Statisical Analyses
3. Results
3.1. Patient Characteristics in Whole Cohort (n = 518)
3.2. Patient Characteristics in Subgroup of Non-Diabetic Renal Transplant Recipients (n = 386)
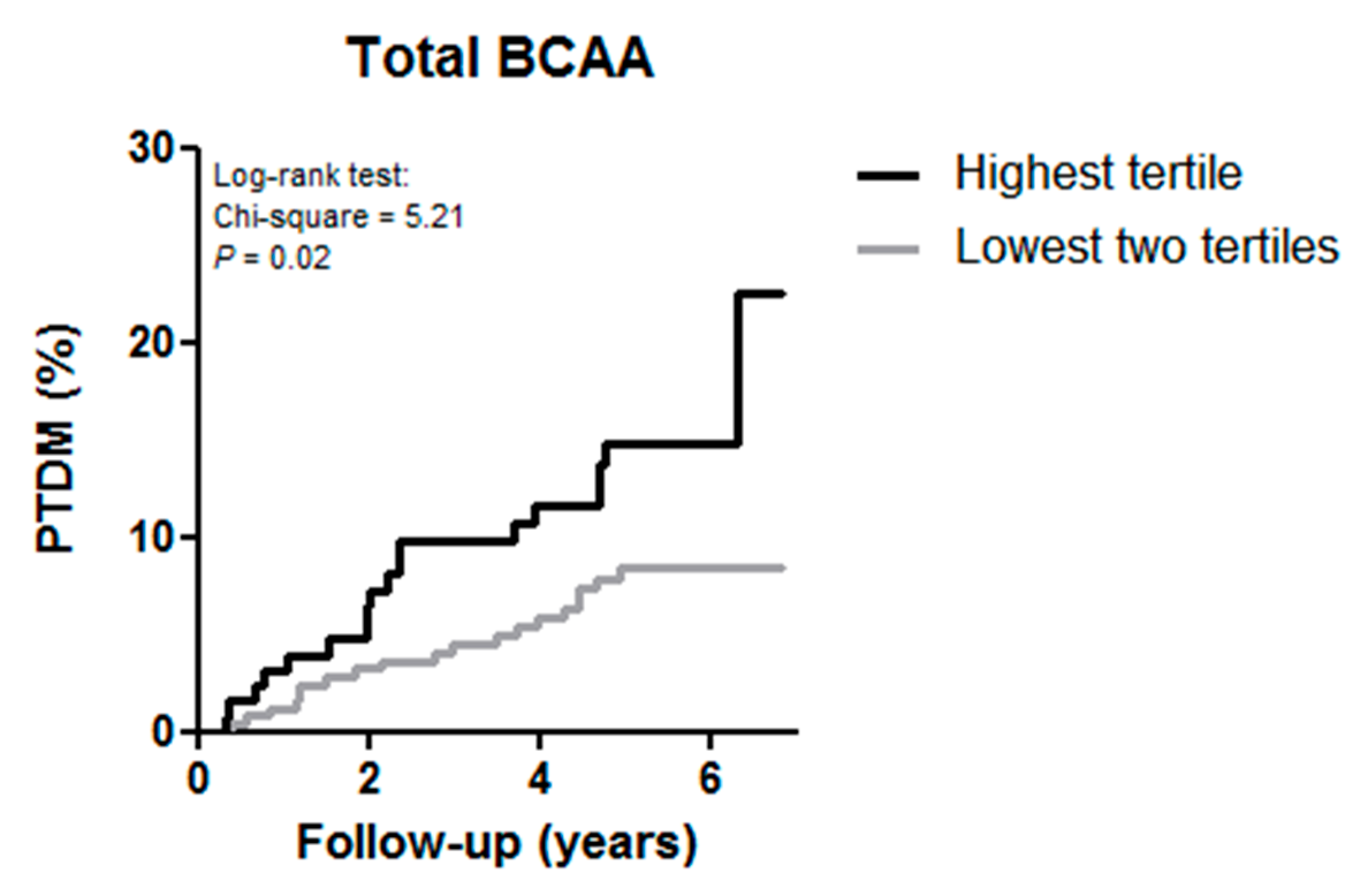
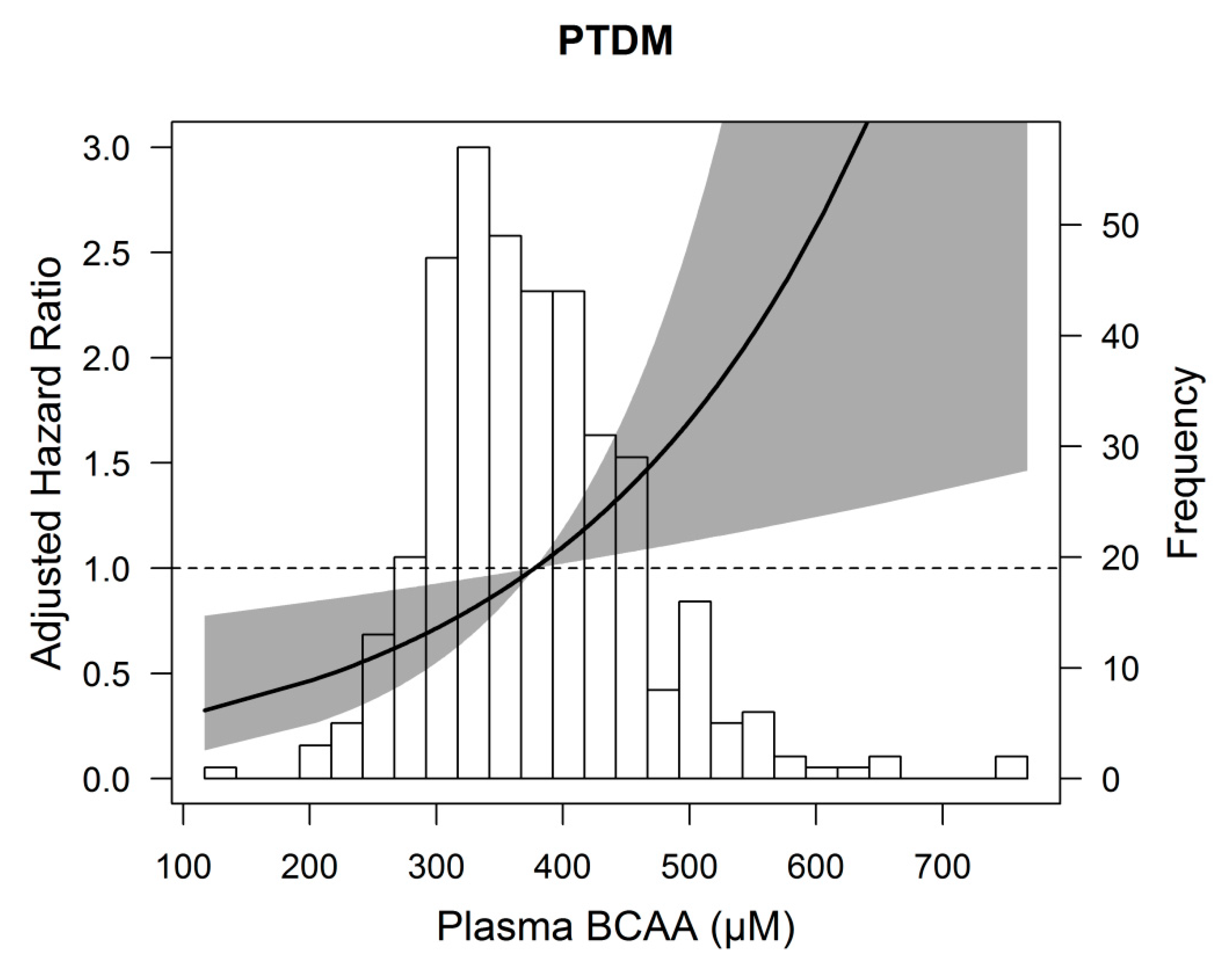
3.3. BCAAs and Risk of Developing PTDM
3.4. Secondary Analyses
3.5. BCAAs and Risk of All-Cause Mortality and Death-Censored Graft Failure
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hjelmesaeth, J.; Midtvedt, K.; Jenssen, T.; Hartmann, A. Insulin resistance after renal transplantation: Impact of immunosuppressive and antihypertensive therapy. Diabetes Care 2001, 24, 2121–2126. [Google Scholar] [CrossRef] [PubMed]
- Cosio, F.G.; Pesavento, T.E.; Osei, K.; Henry, M.L.; Ferguson, R.M. Post-transplant diabetes mellitus: Increasing incidence in renal allograft recipients transplanted in recent years. Kidney Int. 2001, 59, 732–737. [Google Scholar] [CrossRef] [PubMed]
- Jenssen, T.; Hartmann, A. Post-transplant diabetes mellitus in patients with solid organ transplants. Nat. Rev. Endocrinol. 2019, 15, 172–188. [Google Scholar] [CrossRef] [PubMed]
- Montori, V.M.; Basu, A.; Erwin, P.J.; Velosa, J.A.; Gabriel, S.E.; Kudva, Y.C. Posttransplantation diabetes. Diabetes Care 2002, 25, 583–592. [Google Scholar] [CrossRef]
- Rodrigo, E.; Fernandez-Fresnedo, G.; Valero, R.; Ruiz, J.C.; Pinera, C.; Palomar, R.; Gonzalez-Cotorruelo, J.; Gomez-Alamillo, C.; Arias, M. New-Onset Diabetes after Kidney Transplantation: Risk Factors. J. Am. Soc. Nephrol. 2006, 17, S291–S295. [Google Scholar] [CrossRef]
- Kaposztas, Z.; Gyurus, E.; Kahan, B.D. New-onset diabetes after renal transplantation: Diagnosis, incidence, risk factors, impact on outcomes, and novel implications. Transplant. Proc. 2011, 43, 1375–1394. [Google Scholar] [CrossRef]
- Wauters, R.P.; Cosio, F.G.; Suarez Fernandez, M.L.; Kudva, Y.; Shah, P.; Torres, V.E. Cardiovascular consequences of new-onset hyperglycemia after kidney transplantation. Transplantation 2012, 94, 377–382. [Google Scholar] [CrossRef]
- Revanur, V.K.; Jardine, A.G.; Kingsmore, D.B.; Jaques, B.C.; Hamilton, D.H.; Jindal, R.M. Influence of diabetes mellitus on patient and graft survival in recipients of kidney transplantation. Clin. Transplant. 2001, 15, 89–94. [Google Scholar] [CrossRef]
- Cole, E.H.; Johnston, O.; Rose, C.L.; Gill, J.S. Impact of acute rejection and new-onset diabetes on long-term transplant graft and patient survival. Clin. J. Am. Soc. Nephrol. 2008, 3, 814–821. [Google Scholar] [CrossRef]
- Hjelmesæth, J.; Hartmann, A.; Leivestad, T.; Holdaas, H.; Sagedal, S.; Olstad, M.; Jenssen, T. The impact of early-diagnosed new-onset post-transplantation diabetes mellitus on survival and major cardiac events. Kidney Int. 2006, 69, 588–595. [Google Scholar] [CrossRef]
- Hoftman, N.; Prunean, A.; Dhillon, A.; Danovitch, G.M.; Lee, M.S.; Gritsch, H.A. Revised Cardiac Risk Index (RCRI) is a useful tool for evaluation of perioperative cardiac morbidity in kidney transplant recipients. Transplantation 2013, 96, 639–643. [Google Scholar] [CrossRef] [PubMed]
- Karthikeyan, V.; Ananthasubramaniam, K. Coronary risk assessment and management options in chronic kidney disease patients prior to kidney transplantation. Curr. Cardiol. Rev. 2009, 5, 177–186. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pita-Fernandez, S.; Pertega-Diaz, S.; Valdes-Canedo, F.; Seijo-Bestilleiro, R.; Seoane-Pillado, T.; Fernandez-Rivera, C.; Alonso-Hernandez, A.; Lorenzo-Aguiar, D.; Lopez-Calvino, B.; Lopez-Muniz, A. Incidence of cardiovascular events after kidney transplantation and cardiovascular risk scores: Study protocol. BMC Cardiovasc. Disord. 2011, 11, 2. [Google Scholar] [CrossRef] [PubMed]
- Heleniak, Z.; Komorowska-Jagielska, K.; Dębska-Ślizień, A. Assessment of Cardiovascular Risk in Renal Transplant Recipients: Preliminary Results. Transplant. Proc. 2018, 50, 1813–1817. [Google Scholar] [CrossRef]
- Woodward, R.S.; Schnitzler, M.A.; Baty, J.; Lowell, J.A.; Lopez-Rocafort, L.; Haider, S.; Woodworth, T.G.; Brennan, D.C. Incidence and cost of new onset diabetes mellitus among U.S. wait-listed and transplanted renal allograft recipients. Am. J. Transplant. 2003, 3, 590–598. [Google Scholar] [CrossRef]
- Shivaswamy, V.; Boerner, B.; Larsen, J. Post-Transplant Diabetes Mellitus: Causes, Treatment, and Impact on Outcomes. Endocr. Rev. 2016, 37, 37–61. [Google Scholar] [CrossRef]
- Layman, D.K. The role of leucine in weight loss diets and glucose homeostasis. J. Nutr. 2003, 133, 261S–267S. [Google Scholar] [CrossRef]
- Wu, G. Functional amino acids in nutrition and health. Amino Acids 2013, 45, 407–411. [Google Scholar] [CrossRef]
- Saha, A.K.; Xu, X.J.; Lawson, E.; Deoliveira, R.; Brandon, A.E.; Kraegen, E.W.; Ruderman, N.B. Downregulation of AMPK accompanies leucine- and glucose-induced increases in protein synthesis and insulin resistance in rat skeletal muscle. Diabetes 2010, 59, 2426–2434. [Google Scholar] [CrossRef]
- Lynch, C.J.; Adams, S.H. Branched-chain amino acids in metabolic signalling and insulin resistance. Nat. Rev. Endocrinol. 2014, 10, 723–736. [Google Scholar] [CrossRef]
- Wang, T.J.; Larson, M.G.; Vasan, R.S.; Cheng, S.; Rhee, E.P.; McCabe, E.; Lewis, G.D.; Fox, C.S.; Jacques, P.F.; Fernandez, C.; et al. Metabolite profiles and the risk of developing diabetes. Nat. Med. 2011, 17, 448–453. [Google Scholar] [CrossRef] [PubMed]
- Guasch-Ferré, M.; Hruby, A.; Toledo, E.; Clish, C.B.; Martínez-González, M.A.; Salas-Salvadó, J.; Hu, F.B. Metabolomics in prediabetes and diabetes: A systematic review and meta-analysis. Diabetes Care 2016, 39, 833–846. [Google Scholar] [CrossRef] [PubMed]
- Newgard, C.B.; An, J.; Bain, J.R.; Muehlbauer, M.J.; Stevens, R.D.; Lien, L.F.; Haqq, A.M.; Shah, S.H.; Arlotto, M.; Slentz, C.A.; et al. A Branched-Chain Amino Acid-Related Metabolic Signature that Differentiates Obese and Lean Humans and Contributes to Insulin Resistance. Cell Metab. 2009, 9, 311–326. [Google Scholar] [CrossRef] [PubMed]
- Honda, T.; Kobayashi, Y.; Togashi, K.; Hasegawa, H.; Iwasa, M.; Taguchi, O.; Takei, Y.; Sumida, Y. Associations among circulating branched-chain amino acids and tyrosine with muscle volume and glucose metabolism in individuals without diabetes. Nutrition 2016, 32, 531–538. [Google Scholar] [CrossRef]
- Connelly, M.A.; Wolak-Dinsmore, J.; Dullaart, R.P.F. Branched Chain Amino Acids Are Associated with Insulin Resistance Independent of Leptin and Adiponectin in Subjects with Varying Degrees of Glucose Tolerance. Metab. Syndr. Relat. Disord. 2017, 15, 183–186. [Google Scholar] [CrossRef]
- Flores-Guerrero, J.; Osté, M.; Kieneker, L.; Gruppen, E.; Wolak-Dinsmore, J.; Otvos, J.; Connelly, M.; Bakker, S.; Dullaart, R. Plasma Branched-Chain Amino Acids and Risk of Incident Type 2 Diabetes: Results from the PREVEND Prospective Cohort Study. J. Clin. Med. 2018, 7, 513. [Google Scholar] [CrossRef]
- Boulet, M.M.; Chevrier, G.; Grenier-Larouche, T.; Pelletier, M.; Nadeau, M.; Scarpa, J.; Prehn, C.; Marette, A.; Adamski, J.; Tchernof, A. Alterations of plasma metabolite profiles related to adipose tissue distribution and cardiometabolic risk. Am. J. Physiol. Endocrinol. Metab. 2015, 309, E736–E746. [Google Scholar] [CrossRef]
- Pedersen, H.K.; Gudmundsdottir, V.; Nielsen, H.B.; Hyotylainen, T.; Nielsen, T.; Jensen, B.A.H.; Forslund, K.; Hildebrand, F.; Prifti, E.; Falony, G.; et al. Human gut microbes impact host serum metabolome and insulin sensitivity. Nature 2016, 535, 376–381. [Google Scholar] [CrossRef]
- Swarte, J.C.; Douwes, R.M.; Hu, S.; Vila, A.V.; Eisenga, M.F.; Van Londen, M.; Gomes-neto, A.W.; Weersma, R.K. Characteristics and Dysbiosis of the Gut Microbiome in Renal Transplant Recipients. J. Clin. Med. 2020, 9, 386. [Google Scholar] [CrossRef]
- Lee, J.R.; Muthukumar, T.; Dadhania, D.; Toussaint, N.C.; Ling, L.; Pamer, E.; Suthanthiran, M. Gut microbial community structure and complications after kidney transplantation: A pilot study. Transplantation 2014, 98, 697–705. [Google Scholar] [CrossRef]
- Klont, F.; Kieneker, L.M.; Gomes-Neto, A.W.; Stam, S.P.; Ten Hacken, N.H.T.; Kema, I.P.; van Beek, A.P.; van den Berg, E.; Horvatovich, P.; Bischoff, R.; et al. Female Specific Association of Low Insulin-Like Growth Factor 1 (IGF1) Levels with Increased Risk of Premature Mortality in Renal Transplant Recipients. J. Clin. Med. 2020, 9, 293. [Google Scholar] [CrossRef] [PubMed]
- Michel, R.N.; Dunn, S.E.; Chin, E.R. Calcineurin and skeletal muscle growth. Proc. Nutr. Soc. 2004, 63, 341–349. [Google Scholar] [CrossRef] [PubMed]
- Shah, S.H.; Sun, J.-L.; Stevens, R.D.; Bain, J.R.; Muehlbauer, M.J.; Pieper, K.S.; Haynes, C.; Hauser, E.R.; Kraus, W.E.; Granger, C.B.; et al. Baseline metabolomic profiles predict cardiovascular events in patients at risk for coronary artery disease. Am. Heart J. 2012, 163, 844–850. [Google Scholar] [CrossRef] [PubMed]
- Fischer, K.; Kettunen, J.; Würtz, P.; Haller, T.; Havulinna, A.S.; Kangas, A.J.; Soininen, P.; Esko, T.; Tammesoo, M.L.; Mägi, R.; et al. Biomarker Profiling by Nuclear Magnetic Resonance Spectroscopy for the Prediction of All-Cause Mortality: An Observational Study of 17,345 Persons. PLoS Med. 2014, 11. [Google Scholar] [CrossRef]
- Welsh, P.; Rankin, N.; Li, Q.; Mark, P.B.; Würtz, P.; Ala-Korpela, M.; Marre, M.; Poulter, N.; Hamet, P.; Chalmers, J.; et al. Circulating amino acids and the risk of macrovascular, microvascular and mortality outcomes in individuals with type 2 diabetes: Results from the ADVANCE trial. Diabetologia 2018, 61, 1581–1591. [Google Scholar] [CrossRef]
- Cano, N.J.M.; Fouque, D.; Leverve, X.M. Application of branched-chain amino acids in human pathological states: Renal failure. J. Nutr. 2006, 136, 299S–307S. [Google Scholar] [CrossRef]
- Kumar, M.A.; Bitla, A.R.R.; Raju, K.V.N.; Manohar, S.M.; Kumar, V.S.; Narasimha, S.R.P.V.L. Branched chain amino acid profile in early chronic kidney disease. Saudi J. Kidney Dis. Transpl. 2012, 23, 1202–1207. [Google Scholar]
- van den Berg, E.; Engberink, M.F.; Brink, E.J.; van Baak, M.A.; Joosten, M.M.; Gans, R.O.B.; Navis, G.; Bakker, S.J.L. Dietary acid load and metabolic acidosis in renal transplant recipients. Clin. J. Am. Soc. Nephrol. 2012, 7, 1811–1818. [Google Scholar] [CrossRef]
- van den Berg, E.; Geleijnse, J.M.; Brink, E.J.; van Baak, M.A.; Homan van der Heide, J.J.; Gans, R.O.B.; Navis, G.; Bakker, S.J.L. Sodium intake and blood pressure in renal transplant recipients. Nephrol. Dial. Transplant. 2012, 27, 3352–3359. [Google Scholar] [CrossRef]
- Myers, M.G.; Valdivieso, M.; Kiss, A. Use of automated office blood pressure measurement to reduce the white coat response. J. Hypertens. 2009, 27, 280–286. [Google Scholar] [CrossRef]
- Wendel-Vos, G.C.W.; Schuit, A.J.; Saris, W.H.M.; Kromhout, D. Reproducibility and relative validity of the short questionnaire to assess health-enhancing physical activity. J. Clin. Epidemiol. 2003, 56, 1163–1169. [Google Scholar] [CrossRef]
- Feunekes, I.J.; Van Staveren, W.A.; Graveland, F.; De Vos, J.; Burema, J. Reproducibility of a semiquantitative food frequency questionnaire to assess the intake of fats and cholesterol in The Netherlands. Int. J. Food Sci. Nutr. 1995, 46, 117–123. [Google Scholar] [CrossRef]
- Levey, A.S.; Stevens, L.A.; Schmid, C.H.; Zhang, Y.L.; Castro, A.F.; Feldman, H.I.; Kusek, J.W.; Eggers, P.; Van Lente, F.; Greene, T.; et al. A new equation to estimate glomerular filtration rate. Ann. Intern. Med. 2009, 150, 604–612. [Google Scholar] [CrossRef] [PubMed]
- Wolak-Dinsmore, J.; Gruppen, E.G.; Shalaurova, I.; Matyus, S.P.; Grant, R.P.; Gegen, R.; Bakker, S.J.L.; Otvos, J.D.; Connelly, M.A.; Dullaart, R.P.F. A novel NMR-based assay to measure circulating concentrations of branched-chain amino acids: Elevation in subjects with type 2 diabetes mellitus and association with carotid intima media thickness. Clin. Biochem. 2018, 54, 92–99. [Google Scholar] [CrossRef]
- Davidson, J.; Wilkinson, A.; Dantal, J.; Dotta, F.; Haller, H.; Hernandez, D.; Kasiske, B.L.; Kiberd, B.; Krentz, A.; Legendre, C.; et al. New-onset diabetes after transplantation: 2003 International consensus guidelines. Transplantation 2003, 75, SS3–SS24. [Google Scholar] [CrossRef] [PubMed]
- Sharif, A.; Hecking, M.; de Vries, A.P.J.; Porrini, E.; Hornum, M.; Rasoul-Rockenschaub, S.; Berlakovich, G.; Krebs, M.; Kautzky-Willer, A.; Schernthaner, G.; et al. Proceedings from an international consensus meeting on posttransplantation diabetes mellitus: Recommendations and future directions. Am. J. Transplant. 2014, 14, 1992–2000. [Google Scholar] [CrossRef]
- Kidney Disease: Improving Global Outcomes (KDIGO) Transplant Work Group. KDIGO clinical practice guideline for the care of kidney transplant recipients. Am. J. Transplant. 2009, 9, S1–S155. [Google Scholar] [CrossRef]
- Peduzzi, P.; Concato, J.J.; Feinstein, A.R.; Holford, T.R. Importance of events per independent variable in proportional hazards regression analysis. II. Accuracy and precision of regression estimates. J. Clin. Epidemiol. 1995, 48, 1503–1510. [Google Scholar] [CrossRef]
- Grambsch, P.M. Therneau TM Proportional hazards tests and diagnostics based on weighted residuals. Biometrika 1994, 81, 515–526. [Google Scholar] [CrossRef]
- Committee, I.E. International expert committee report on the role of the A1C assay in the diagnosis of diabetes. Diabetes Care 2009, 32, 1237–1334. [Google Scholar]
- American Diabetes Association Diagnosis and Classification of Diabetes Mellitus Mellitus. Diabetes Care 2010, 33, S62–S69. [CrossRef] [PubMed]
- Tingley, D.; Yamamoto, T.; Hirose, K.; Keele, L.; Imai, K. Mediation: R package for causal mediation analysis. J. Stat. Softw. 2014, 59, 1–38. [Google Scholar] [CrossRef]
- Noordzij, M.; Leffondré, K.; Van Stralen, K.J.; Zoccali, C.; Dekker, F.W.; Jager, K.J. When do we need competing risks methods for survival analysis in nephrology? Nephrol. Dial. Transplant. 2013, 28, 2670–2677. [Google Scholar] [CrossRef] [PubMed]
- Fine, J.P.; Gray, R.J. A Proportional Hazards Model for the Subdistribution of a Competing Risk. J. Am. Stat. Assoc. 1999, 94, 496–509. [Google Scholar] [CrossRef]
- Xu, F.; Tavintharan, S.; Sum, C.F.; Woon, K.; Lim, S.C.; Ong, C.N. Metabolic signature shift in type 2 diabetes mellitus revealed by mass spectrometry-based metabolomics. J. Clin. Endocrinol. Metab. 2013, 98, E1060–E1065. [Google Scholar] [CrossRef]
- Fiehn, O.; Garvey, W.T.; Newman, J.W.; Lok, K.H.; Hoppel, C.L.; Adams, S.H. Plasma metabolomic profiles reflective of glucose homeostasis in non-diabetic and type 2 diabetic obese African-American women. PLoS ONE 2010, 5, e15234. [Google Scholar] [CrossRef]
- Zheng, Y.; Li, Y.; Qi, Q.; Hruby, A.; Manson, J.E.; Willett, W.C.; Wolpin, B.M.; Hu, F.B.; Qi, L. Cumulative consumption of branched-chain amino acids and incidence of type 2 diabetes. Int. J. Epidemiol. 2016, 45, 1482–1492. [Google Scholar] [CrossRef]
- Maida, A.; Chan, J.S.K.; Sjoberg, K.A.; Zota, A.; Schmoll, D.; Kiens, B.; Herzig, S.; Rose, A.J. Repletion of branched chain amino acids reverses mTORC1 signaling but not improved metabolism during dietary protein dilution. Mol. Metab. 2017, 6, 873–881. [Google Scholar] [CrossRef]
- Karusheva, Y.; Koessler, T.; Strassburger, K.; Markgraf, D.; Mastrototaro, L.; Jelenik, T.; Simon, M.-C.; Pesta, D.; Zaharia, O.-P.; Bodis, K.; et al. Short-term dietary reduction of branched-chain amino acids reduces meal-induced insulin secretion and modifies microbiome composition in type 2 diabetes: A randomized controlled crossover trial. Am. J. Clin. Nutr. 2019, 110, 1098–1107. [Google Scholar] [CrossRef]
- Rietman, A.; Schwarz, J.; Tome, D.; Kok, F.J.; Mensink, M. High dietary protein intake, reducing or eliciting insulin resistance? Eur. J. Clin. Nutr. 2014, 68, 973–979. [Google Scholar] [CrossRef]
- Newgard, C.B. Interplay between lipids and branched-chain amino acids in development of insulin resistance. Cell Metab. 2012, 15, 606–614. [Google Scholar] [CrossRef] [PubMed]
- Holecek, M. Branched-chain amino acids in health and disease: Metabolism, alterations in blood plasma, and as supplements. Nutr. Metab. 2018, 15, 33. [Google Scholar] [CrossRef] [PubMed]
- Tom, A.; Nair, K.S. Assessment of branched-chain amino Acid status and potential for biomarkers. J. Nutr. 2006, 136, 324S–330S. [Google Scholar] [CrossRef]
- Lee, C.C.; Watkins, S.M.; Lorenzo, C.; Wagenknecht, L.E.; Il’Yasova, D.; Chen, Y.D.I.; Haffner, S.M.; Hanley, A.J. Branched-chain amino acids and insulin metabolism: The Insulin Resistance Atherosclerosis Study (IRAS). Diabetes Care 2016, 39, 582–588. [Google Scholar] [CrossRef] [PubMed]
- Allam-Ndoul, B.; Guénard, F.; Garneau, V.; Barbier, O.; Pérusse, L.; Vohl, M.-C. Associations between branched chain amino acid levels, obesity and cardiometabolic complications. Integr. Obes. Diabetes 2015, 1, 157–162. [Google Scholar] [CrossRef]
- Bostom, A.G.; Carpenter, M.A.; Kusek, J.W.; Levey, A.S.; Hunsicker, L.; Pfeffer, M.A.; Selhub, J.; Jacques, P.F.; Cole, E.; Gravens-Mueller, L.; et al. Homocysteine-lowering and cardiovascular disease outcomes in kidney transplant recipients: Primary results from the folic acid for vascular outcome reduction in transplantation trial. Circulation 2011, 123, 1763–1770. [Google Scholar] [CrossRef]
- Leibowitz, G.; Cerasi, E.; Ketzinel-Gilad, M. The role of mTOR in the adaptation and failure of beta-cells in type 2 diabetes. Diabetes. Obes. Metab. 2008, 10, 157–169. [Google Scholar] [CrossRef]
- Hecking, M.; Haidinger, M.; Doller, D.; Werzowa, J.; Tura, A.; Zhang, J.; Tekoglu, H.; Pleiner, J.; Wrba, T.; Rasoul-Rockenschaub, S.; et al. Early basal insulin therapy decreases new-onset diabetes after renal transplantation. J. Am. Soc. Nephrol. 2012, 23, 739–749. [Google Scholar] [CrossRef]
- Chakkera, H.A.; Weil, E.J.; Castro, J.; Heilman, R.L.; Reddy, K.S.; Mazur, M.J.; Hamawi, K.; Mulligan, D.C.; Moss, A.A.; Mekeel, K.L.; et al. Hyperglycemia during the immediate period after kidney transplantation. Clin. J. Am. Soc. Nephrol. 2009, 4, 853–859. [Google Scholar] [CrossRef]
- Lorent, M.; Foucher, Y.; Kerleau, K.; Brouard, S.; Baayen, C.; Lebouter, S.; Naesens, M.; Bestard Matamoros, O.; Åsberg, A.; Giral, M. The EKiTE network (epidemiology in kidney transplantation—A European validated database): An initiative epidemiological and translational European collaborative research. BMC Nephrol. 2019, 20, 1–10. [Google Scholar] [CrossRef]
- Kramer, A.; Pippias, M.; Noordzij, M.; Stel, V.S.; Afentakis, N.; Ambuhl, P.M.; Andrusev, A.M.; Fuster, E.A.; Arribas Monzon, F.E.; Asberg, A.; et al. The European Renal Association—European Dialysis and Transplant Association (ERA-EDTA) Registry Annual Report 2015: A summary. Clin. Kidney J. 2018, 11, 108–122. [Google Scholar] [CrossRef] [PubMed]
- Ussif, A.M.; Åsberg, A.; Halden, T.A.S.; Nordheim, E.; Hartmann, A.; Jenssen, T. Validation of diagnostic utility of fasting plasma glucose and HbA1c in stable renal transplant recipients one year after transplantation. BMC Nephrol. 2019, 20, 1–6. [Google Scholar] [CrossRef] [PubMed]
| Total (n = 518) | Diabetic RTR (n = 132) | Non-Diabetic RTR (n = 386) | P-Value | |
|---|---|---|---|---|
| General characteristics | ||||
| Age, years | 52.7 ± 13.0 | 57.2 ± 10.1 | 51.1 ± 13.6 | <0.001 |
| Male sex, n (%) | 278 (53.7) | 71 (46.2) | 207 (53.6) | 0.97 |
| Race (white), n (%) | 515 (99.4) | 132 (100.0) | 383 (99.2) | 0.31 |
| BMI, kg/m2 | 26.6 ± 4.8 | 28.7 ± 5.3 | 25.9 ± 4.5 | <0.001 |
| Physical activity score (time x intensity) | 5075 (2190–8100) | 3690 (1215–6563) | 5590 (2810–8715) | <0.001 |
| Smoking status, n (%) | 0.16 | |||
| Never | 203 (39.2) | 49 (37.1) | 154 (39.9) | |
| Former | 228 (44.0) | 63 (47.7) | 165 (42.7) | |
| Current | 63 (12.2) | 10 (7.6) | 53 (13.7) | |
| Alcohol consumption, g/d | 2.3 (0.0–11.1) | 1.6 (0.0–7.4) | 2.6 (0.0–11.7) | 0.05 |
| Total energy intake, kcal/d | 2139 ± 634 | 2106 ± 599 | 2152 ± 647 | 0.49 |
| Urea excretion, mmol/24h | 389.4 ± 117.2 | 391.2 ± 124.3 | 388.8 ± 114.9 | 0.83 |
| Circulation | ||||
| Heart rate, b.p.m. | 68.6 ± 12.1 | 72.2 ± 11.3 | 67.3 ± 12.1 | <0.001 |
| SBP, mmHg | 135.9 ± 17.7 | 138.2 ± 18.5 | 135.1 ± 17.4 | 0.07 |
| DBP, mmHg | 82.1 ± 11.0 | 82.0 ± 10.3 | 82.1 ± 11.3 | 0.93 |
| Transplant characteristics | ||||
| Transplant vintage, years | 5.0 (1.7–11.9) | 6.2 (1.7–11.8) | 4.9 (1.7–12.0) | 0.59 |
| Living donor, n (%) | 182 (35.1) | 37 (28.0) | 145 (37.6) | 0.05 |
| Pre-emptive transplant, n (%) | 91 (17.6) | 14 (10.6) | 77 (19.9) | 0.02 |
| Dialysis duration, months | 42.0 (18.5–59.0) | 36.0 (18.5–52.5) | 44.0 (17.0–59.8) | 0.58 |
| Age donor, years | 43.7 ± 15.2 | 41.9 ± 15.2 | 44.3 ± 15.2 | 0.12 |
| Renal allograft function | ||||
| Serum creatinine, µmol/L | 127.0 (101.0–167.3) | 133.0 (102.3–166.0) | 126.0 (101.0–168.0) | 0.77 |
| eGFR, ml/min per 1.73 m2 | 50.6 ± 19.9 | 49.5 ± 20.8 | 51.0 ± 19.6 | 0.45 |
| Proteinuria, n (%) | 117 (22.6) | 35 (26.5) | 82 (21.2) | 0.19 |
| Glucose homeostasis | ||||
| Plasma glucose (mmol/L) | 5.2 (4.8–6.0) | 7.0 (5.4–8.1) | 5.1 (4.7–5.5) | <0.001 |
| HbA1c (%) | 6.0 ± 0.8 | 6.9 ± 1.1 | 5.7 ± 0.4 | <0.001 |
| Lipids and lipoproteins | ||||
| Total cholesterol, mmol/L | 5.2 ± 1.2 | 5.2 ± 1.2 | 5.1 ± 1.1 | 0.64 |
| HDL-cholesterol, mmol/L | 1.4 ± 0.5 | 1.3 ± 0.4 | 1.4 ± 0.5 | 0.03 |
| LDL-cholesterol, mmol/L | 3.0 ± 0.9 | 3.0 ± 1.0 | 3.0 ± 0.9 | 0.59 |
| Triglycerides, mmol/L | 1.7 (1.3–2.4) | 1.9 (1.4–3.0) | 1.7 (1.2–2.2) | <0.001 |
| Medication | ||||
| Calcineurin inhibitor, n (%) | 0.19 | |||
| Cyclosporine | 218 (42.1) | 61 (46.2) | 157 (40.7) | |
| Tacrolimus | 93 (18.0) | 27 (20.5) | 66 (17.1) | |
| Trough level cyclosporine (µg/L) | 108.0 (77.0–144.0) | 102.5 (74.0–156.0) | 111.0 (77.5–142.0) | 0.78 |
| Trough level tacrolimus (µg/L) | 6.8 (5.0–9.0) | 6.6 (5.4–9.9) | 7.2 (4.9–9.0) | 0.78 |
| Proliferation inhibitor, n (%) | 0.20 | |||
| Azathioprine | 100 (19.3) | 22 (16.7) | 78 (20.2) | |
| Mycofenol | 333 (64.3) | 82 (62.1) | 251 (65.0) | |
| Prednisolone, n (%) | 513 (99.0) | 131 (99.2) | 382 (99.0) | 0.78 |
| Prednisolone dose, mg/24h | 10.0 (7.5–10.0) | 10.0 (7.5–10.0) | 10.0 (7.5–10.0) | 0.71 |
| Antihypertensive drugs, n (%) | 462 (89.2) | 121 (91.7) | 341 (88.3) | 0.29 |
| Statins, n (%) | 270 (52.1) | 84 (63.6) | 186 (48.2) | 0.002 |
| Amino acids | ||||
| Total BCAA, µM | 389.6 ± 89.0 | 424.6 ± 97.9 | 377.6 ± 82.5 | <0.001 |
| Valine, µM | 203.0 ± 44.7 | 217.0 ± 48.9 | 198.2 ± 42.2 | <0.001 |
| Leucine, µM | 141.7 ± 37.3 | 157.1 ± 42.1 | 136.5 ± 34.0 | <0.001 |
| Isoleucine, µM | 44.9 ± 19.1 | 51.8 ± 20.0 | 43.5 ± 17.8 | <0.001 |
| Tertile 1 (n = 127) | Tertile 2 (n = 130) | Tertile 3 (n = 129) | P-Value | |
|---|---|---|---|---|
| General characteristics | ||||
| Age, years | 49.9 ± 13.3 | 52.8 ± 14.4 | 50.6 ± 12.9 | 0.19 |
| Male sex, n (%) | 81 (63.8) | 73 (56.2) | 88 (68.2) | <0.001 |
| Race (white), n (%) | 126 (99.2) | 129 (99.2) | 128 (99.2) | 1.00 |
| BMI, kg/m2 | 25.4 ± 5.2 | 26.0 ± 4.2 | 26.3 ± 3.9 | 0.27 |
| Physical activity score (time x intensity) | 4930 (2100–7260) | 5905 (3315–8625) | 6100 (3120–9860) | 0.05 |
| Smoking status, n (%) | 0.10 | |||
| Never | 54 (42.5) | 58 (44.6) | 42 (32.6) | |
| Former | 47 (37.0) | 51 (39.2) | 67 (51.9) | |
| Current | 21 (16.5) | 17 (13.1) | 15 (11.6) | |
| Alcohol consumption, g/d | 1.6 (0.0–8.9) | 2.9 (0.1–11.3) | 4.3 (0.1–15.8) | 0.03 |
| Total energy intake, kcal/d | 2178 ± 631 | 2184 ± 724 | 2096 ± 578 | 0.51 |
| Urea excretion, mmol/24h | 335.1 ± 92.5 | 406.6 ± 117.2 | 423.2 ± 114.2 | <0.001 |
| Circulation | ||||
| Heart rate, b.p.m. | 70.3 ± 12.3 | 66.0 ± 12.5 | 65.8 ± 11.0 | 0.005 |
| SBP, mmHg | 134.9 ± 17.8 | 133.9 ± 18.2 | 136.4 ± 16.1 | 0.51 |
| DBP, mmHg | 81.7 ± 11.8 | 80.4 ± 11.4 | 84.1 ± 10.4 | 0.03 |
| Transplant characteristics | ||||
| Transplant vintage, years | 5.0 (1.8–14.9) | 4.7 (1.7–12.0) | 4.9 (1.3–10.8) | 0.37 |
| Living donor, n (%) | 56 (44.1) | 39 (30.0) | 50 (38.8) | 0.06 |
| Pre-emptive transplant, n (%) | 33 (26.0) | 22 (16.9) | 22 (17.1) | 0.12 |
| Dialysis duration, months | 34.5 (11.0–63.0) | 47.0 (14.0–60.5) | 37.0 (22.5–58.5) | 0.81 |
| Age donor, years | 42.7 ± 15.5 | 44.7 ± 15.6 | 45.5 ± 14.4 | 0.33 |
| Renal allograft function | ||||
| Serum creatinine, µmol/L | 124.0 (98.0–175.0) | 123.0 (99.8–154.5) | 134.0 (104.0–180.5) | 0.23 |
| eGFR, ml/min per 1.73 m2 | 50.1 ± 21.1 | 52.8 ± 19.3 | 50.1 ± 18.2 | 0.44 |
| Proteinuria, n (%) | 28 (22.0) | 28 (21.5) | 26 (20.2) | 0.92 |
| Glucose homeostasis | ||||
| Plasma glucose (mmol/L) | 5.1 (4.6–5.5) | 5.1 (4.7–5.4) | 5.0 (4.7–5.6) | 1.00 |
| HbA1c (%) | 5.6 ± 0.3 | 5.6 ± 0.4 | 5.7 ± 0.4 | 0.10 |
| Lipids and lipoproteins | ||||
| Total cholesterol, mmol/L | 5.2 ± 1.0 | 5.1 ± 1.1 | 5.2 ± 1.3 | 0.62 |
| HDL-cholesterol, mmol/L | 1.5 ± 0.5 | 1.4 ± 0.4 | 1.3 ± 0.4 | <0.001 |
| LDL-cholesterol, mmol/L | 3.0 ± 0.8 | 3.0 ± 1.0 | 3.1 ± 1.0 | 0.43 |
| Triglycerides, mmol/L | 1.6 (1.2–2.1) | 1.6 (1.2–2.2) | 1.7 (1.3–2.4) | 0.19 |
| Medication | ||||
| Calcineurin inhibitor, n (%) | 0.57 | |||
| Cyclosporine | 53 (41.7) | 54 (41.5) | 50 (38.8) | |
| Tacrolimus | 19 (15.0) | 19 (14.6) | 28 (21.7) | |
| Trough level cyclosporine (µg/L) | 112.0 (78.3–143.3) | 102.0 (74.8–141.5) | 105.0 (74.5–156.5) | 0.85 |
| Trough level tacrolimus (µg/L) | 5.5 (3.9–8.0) | 7.7 (6.0–9.7) | 7.4 (6.0–9.6) | 0.08 |
| Proliferation inhibitor, n (%) | 0.04 | |||
| Azathioprine | 34 (26.8) | 25 (19.2) | 19 (14.7) | |
| Mycofenol | 71 (55.9) | 84 (64.6) | 96 (74.4) | |
| Prednisolone, n (%) | 127 (100.0) | 128 (98.5) | 127 (98.4) | 0.37 |
| Prednisolone dose, mg/24h | 10.0 (7.5–10.0) | 10.0 (7.5–10.0) | 10.0 (7.5–10.0) | 0.19 |
| Antihypertensive drugs, n (%) | 107 (84.3) | 117 (90.0) | 117 (90.7) | 0.21 |
| Statins, n (%) | 53 (41.7) | 66 (50.8) | 67 (51.9) | 0.20 |
| Amino acids | ||||
| Total BCAA, µM | 297.1 ± 33.7 | 366.6 ± 20.4 | 467.9 ± 64.7 | <0.001 |
| Valine, µM | 159.1 ± 20.7 | 194.5 ± 18.4 | 240.5 ± 35.3 | <0.001 |
| Leucine, µM | 107.6 ± 21.5 | 133.3 ± 17.3 | 168.2 ± 29.3 | <0.001 |
| Isoleucine, µM | 31.6 ± 10.0 | 39.1 ± 11.2 | 59.1 ± 18.0 | <0.001 |
| Per SD as Continuous Variable (µmol/L) | Highest Tertile vs. Lower Two Tertiles | ||||
|---|---|---|---|---|---|
| BCAA | |||||
| No. of events | 38 | 19 | 19 | ||
| HR (95% CI) | P | Reference | HR (95% CI) | P | |
| Crude | 1.43 (1.09–1.88) | 0.009 | 1.00 | 2.06 (1.09–3.90) | 0.03 |
| Model 1 | 1.43 (1.08–1.89) | 0.01 | 1.00 | 2.07 (1.07–3.99) | 0.03 |
| Model 2 | 1.43 (1.07–1.90) | 0.02 | 1.00 | 1.97 (1.02–3.82) | 0.05 |
| Model 3 | 1.37 (1.03–1.84) | 0.03 | 1.00 | 1.82 (0.93–3.57) | 0.08 |
| Model 4 | 1.42 (1.06–1.90) | 0.02 | 1.00 | 1.90 (0.95–3.80) | 0.07 |
| Model 5 | 1.47 (1.10–1.96) | 0.009 | 1.00 | 2.09 (1.05–4.17) | 0.04 |
| Model 6 | 1.42 (1.08–1.85) | 0.01 | 1.00 | 2.12 (1.09–4.12) | 0.03 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Osté, M.C.J.; Flores-Guerrero, J.L.; Gruppen, E.G.; Kieneker, L.M.; Connelly, M.A.; Otvos, J.D.; Dullaart, R.P.F.; Bakker, S.J.L. High Plasma Branched-Chain Amino Acids Are Associated with Higher Risk of Post-Transplant Diabetes Mellitus in Renal Transplant Recipients. J. Clin. Med. 2020, 9, 511. https://doi.org/10.3390/jcm9020511
Osté MCJ, Flores-Guerrero JL, Gruppen EG, Kieneker LM, Connelly MA, Otvos JD, Dullaart RPF, Bakker SJL. High Plasma Branched-Chain Amino Acids Are Associated with Higher Risk of Post-Transplant Diabetes Mellitus in Renal Transplant Recipients. Journal of Clinical Medicine. 2020; 9(2):511. https://doi.org/10.3390/jcm9020511
Chicago/Turabian StyleOsté, Maryse C. J., Jose L. Flores-Guerrero, Eke G. Gruppen, Lyanne M. Kieneker, Margery A. Connelly, James D. Otvos, Robin P. F. Dullaart, and Stephan J. L. Bakker. 2020. "High Plasma Branched-Chain Amino Acids Are Associated with Higher Risk of Post-Transplant Diabetes Mellitus in Renal Transplant Recipients" Journal of Clinical Medicine 9, no. 2: 511. https://doi.org/10.3390/jcm9020511
APA StyleOsté, M. C. J., Flores-Guerrero, J. L., Gruppen, E. G., Kieneker, L. M., Connelly, M. A., Otvos, J. D., Dullaart, R. P. F., & Bakker, S. J. L. (2020). High Plasma Branched-Chain Amino Acids Are Associated with Higher Risk of Post-Transplant Diabetes Mellitus in Renal Transplant Recipients. Journal of Clinical Medicine, 9(2), 511. https://doi.org/10.3390/jcm9020511







