1. Introduction
Dental implants have high survival probabilities with good clinical and radiological findings [
1]. Long-term studies documented a survival rate of approximately 95% over a study period of at least ten years [
2,
3]. One of the important factors leading to late implant loss is peri-implantitis, which is a combination of mucositis and bone resorption around implants [
4,
5,
6]. Pain, implant mobility, crestal bone loss over a half of implant length, and persistent exudate from the peri-implant tissue are the most common reasons for implant removal [
7]. Studies have reported a correlation between autologous cancellous bone grafts and long-term bone resorption, which leads to peri-implantitis and consecutive implant loss [
8,
9].
Bone loss resulting from trauma, disease, aging, or congenital abnormalities, remains a global challenge for health professionals and patients. Maxillofacial or craniofacial bone loss has a high psychological impact on patients. Therefore, aesthetic reconstruction is as valuable as functional reconstruction in this region [
10].
Tissue engineering is a feasible method which can be pursued to perform aesthetic reconstruction. This implies the enhancement of autologous bone grafts using PRP [
11]. In recent years, a staggering development on the use of tissue engineering and biomaterials has shifted beliefs and expectations [
12]. Although recent strategies, such as decellularized matrix, nanoparticles, stem-cell therapies, scaffolds, and the engineering of a whole tooth, have shown some success, older approaches such as platelet-rich plasma (PRP) and platelet-rich fibrin (PRF) still require validation or disproof of their clinical efficacy through long-term follow-ups [
12,
13,
14].
Since the first introduction of the use of PRP in augmentation techniques [
11], various studies have been conducted to evaluate its effectiveness [
15,
16,
17,
18,
19,
20]. Optimized bone healing with enhanced bone mineral density was the most tested hypothesis in these studies. Results were controversial, with positive effects [
15,
18] and no effects [
17,
21] of PRP. Following initial positive effects of PRP, Consolo et al. reported a decrease of the bone mineral density after six months [
18]. In another study, the same period was needed to report the first improvement of bone density by using PRP [
22]. The fact is, if the bone density improves after augmentation using a mainly cancellous bone graft, such from the iliac crest bone, the long-term bone resorption around dental implants will be reduced [
23]. Although the literature has addressed augmentation with and without PRP, long-term follow up has been rarely discussed. Thor et al. compared implant survival, marginal bone level, and implant stability after one year of mastication function of inserted dental implants in the maxilla, following autogenous bone grafting with or without PRP in a split mouth design [
24]. The implant survival and marginal bone level revealed no statistically significant differences. Implant stability was tested by using resonance frequency analysis (RFA). Only implants in the anterior maxilla proved to be more stable on PRP sites [
24]. Recent studies focused on the effect of PRP have had some limitations, such as a lack of a control group or only mid-term follow-up time [
25,
26,
27]. Clinical practitioners judge the validity and feasibility of a given procedure more through a randomized controlled trial with long-term results. To the best of our knowledge, long-term clinical and radiological implant assessments after using an autologous iliac crest bone graft and PRP have not been well addressed in the literature. To investigate the long-term influence of PRP, we used the same collection of patients from the prospective randomized clinical trial (RCT) by Schaaf et al. [
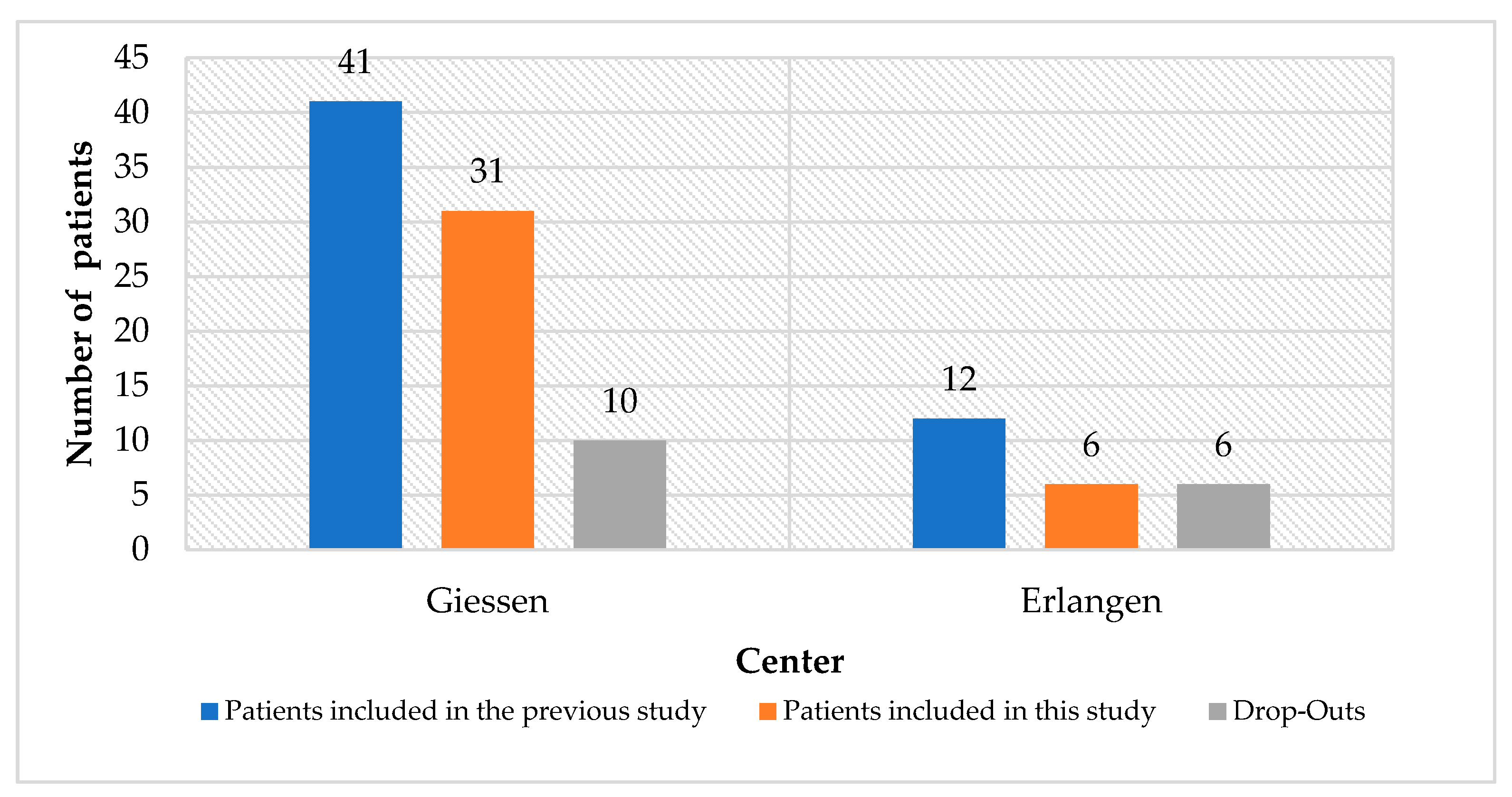
19]. The RCT was conducted between 2001 and 2004 in two centers (Oral and Maxillofacial Departments at the Universities of Giessen and Erlangen, Germany). 53 Patients (34 split-mouth, 19 unilateral) with maxillary atrophy underwent sinus lift surgery and iliac crest augmentation with and without PRP. That study could not determine any significant differences between the test and control group regarding bone density [
19]. In this study, we hypothesized that dental implants placed in PRP-augmented bones have better clinical and radiological outcomes.
4. Discussion
This study aimed to investigate the long-term impact of PRP on the clinical and radiological treatment outcomes of dental implants. A comparison to the treatment outcome without PRP was established by the evaluation of a control group or a control side. The distribution of the patients to the study groups was randomized and the investigators were blinded (patients’ group affiliation was unknown to the investigator). This study represents a non-experimental therapeutic study with retrospective data collection and thus shows a lower level of evidence compared to other prospective studies. Nevertheless, it must be emphasized that there is currently no controlled study reporting the influence of PRP on long-term clinical and radiological outcomes, therefore, this study provides important information in this regard. The randomization and stratification of the previous trial achieved a structural equality within the individual groups. Since the present study is based on the same patient collection, structural equality, despite the drop-outs, was maintained. Additionally, this study was designed as a bicentric study, as patients from both Giessen and Erlangen were examined. Using several centers increased the external validity and improved the comparability of the results of the study [
32]. One limitation of this study is the number of 37 included patients. Statistically, this represents a small population of patients. A greater quantity of patients allows for higher statistical power and can be better communicated to the general public [
33]. However, as this is a long-term follow-up study in which patient population was dependent on the previous study, it is obvious that contact would not be possible for several patients.
The clinical and radiological implant parameters were hard to compare with the current literature, as there is a lack of equivalent research already published. Therefore, studies disregarding PRP were considered and contrasted with the results of the control group. The plaque index according to Mombelli et al. [
30] was recorded in all investigated implants. In the PRP group, 75.3% (split-mouth evaluation) and 78.2% (unilateral evaluation) of the implants showed no or low plaque accumulation (Grade 0 or 1). In the control group, similar results were reported in 65.8% (split-mouth evaluation) and 72.1% (unilateral evaluation) of the implants. This suggests that good oral hygiene was maintained by the majority of patients in both groups. Moderate to massive plaque (Grade 2 or 3) was found in 24.7% (split-mouth evaluation) and 21.8% (unilateral evaluation) of implants in the PRP group, and 34.2% (split-mouth evaluation) and 27.9% (unilateral evaluation) in the control group. There was no significant difference between the groups. Average plaque index was 1 in the PRP group (split-mouth evaluation) and 0.9 (unilateral evaluation) and in the control group it was 1.2 (split-mouth evaluation) or 1.1 (unilateral evaluation). The implants’ plaque index in the study by Attia et al. reported Grade 0 or 1 in 75.2% of cases, while 24.8% of the implants were Grade 2 or 3 [
34]. This corresponds to the results of the control group from the present study. Simonis et al. investigated 131 implants after an observation period of 10 to 16 years [
35]. The average plaque index documented in this study was 1.1, which is comparable with the data of the control group from the present results.
On average, the measured pocket depth in the PRP group was 4.1 mm and 4.2 mm, and in the control group 3.8 mm and 3.6 mm in split-mouth evaluation and unilateral evaluation, respectively. Probing depths of more than 4 mm were considered pathological for implants [
36]. In the present study, 67.1% (split-mouth evaluation) and 63.6% (unilateral evaluation) of the implants had probing depths of up to 4 mm in the PRP group. In the control group, a probing depth exceeding 4 mm was present in 79.8% (split-mouth evaluation) and 88.2% (unilateral evaluation) of implants. Pathological pocket depths of more than 4 mm were documented in the PRP group in 32.9% (split-mouth evaluation) and 36.4% (unilateral evaluation) of total implants, in the control group in 20.2% (split-mouth evaluation) and 11.8% (unilateral evaluation) of total implants. No statistically significant differences between the groups could be demonstrated in the split-mouth analysis with respect to the probing depths. In the unilateral evaluation, however, the PRP group had significantly higher probing depths. Here, it must be taken into consideration that the measurement of probing depths due to pseudo pockets and the design of the crown can be falsified. In case of pseudo pockets, there is no equivalent marginal bone loss despite increased probing depth. In the evaluation of crestal bone loss, no significant difference between the groups was detected in the unilateral evaluation. It can therefore be assumed that the results of the probing depths were worsened by pseudo pockets. In the study by Attia et al., 84.1% of the implants had probing depths of up to 4 mm. Pocket depths of more than 4 mm were measured on 15.7% of all implants. These values confirm the results from the control group of this study [
34]. In addition, Van Velzen et al. reported in their prospective study of 177 patients probing depths of 3.7 mm with 374 implants after 10 years. This result is also comparable with the results of the control group in this study [
37].
In both end points of the PRP group, 60% of implants showed no bleeding on probing. In the control group, 72% of implants did not bleed in both evaluations. No significant difference was found between the two study groups. Becker et al. examined 388 implants after an average of 14 years. They were clinically unable to detect bleeding in 79.1% of the implants. This result is similar to the values of the control group from this study [
38].
No implant loosened at the time of follow-up. Thus, there is no difference between the PRP and the control group with respect to the degree of loosening. Other studies in the literature indicate the same results [
34,
39].
Periotest
® values of −8 to 0 indicate a satisfactory osseointegration of the tested implant. Values of up to 9 should be clinically re-evaluated and values of 10 and above indicate a lack of osseointegration [
40]. In the present study, values ranging from -8 to 0 (68.3 to 76.5%) were observed in both groups in approximately half of the implants. Between 23.5% and 30.4% of the implants, the Periotest
® device delivered values of 1 to 9. Values above 10 were only determined for a maximum of 1.3% of the implants. In the two evaluations, no statistically significant difference could be detected between the PRP and the control group. Periotest
® measurements in this study are not very objective, however, because the supra constructions were not removed. Due to the similar study design, a comparison is merited with the results of Attia et al., who investigated implants in hypodontia patients, and they state that only one of the 148 implants tested had a Periotest
® value greater than 10 [
34]. This corresponds to 0.7%, which is consistent with the values of the control group in the present work.
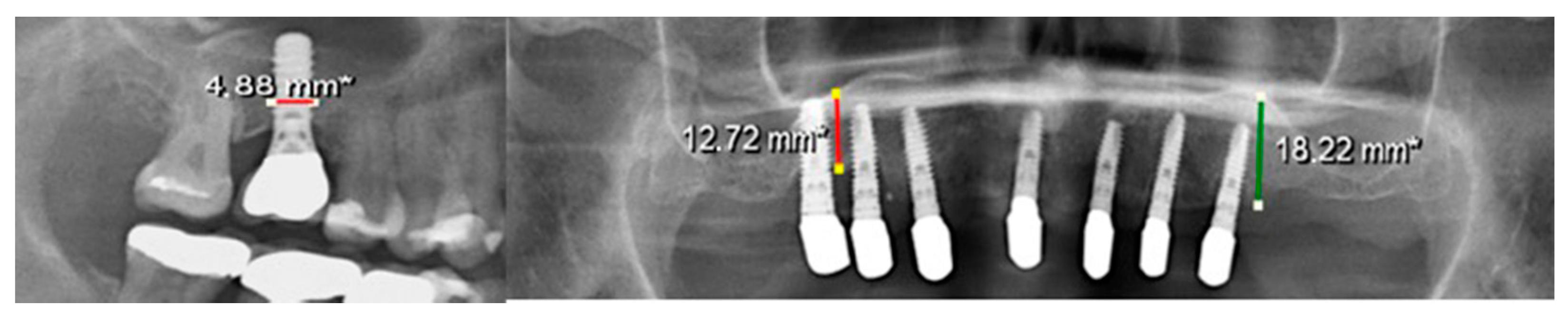
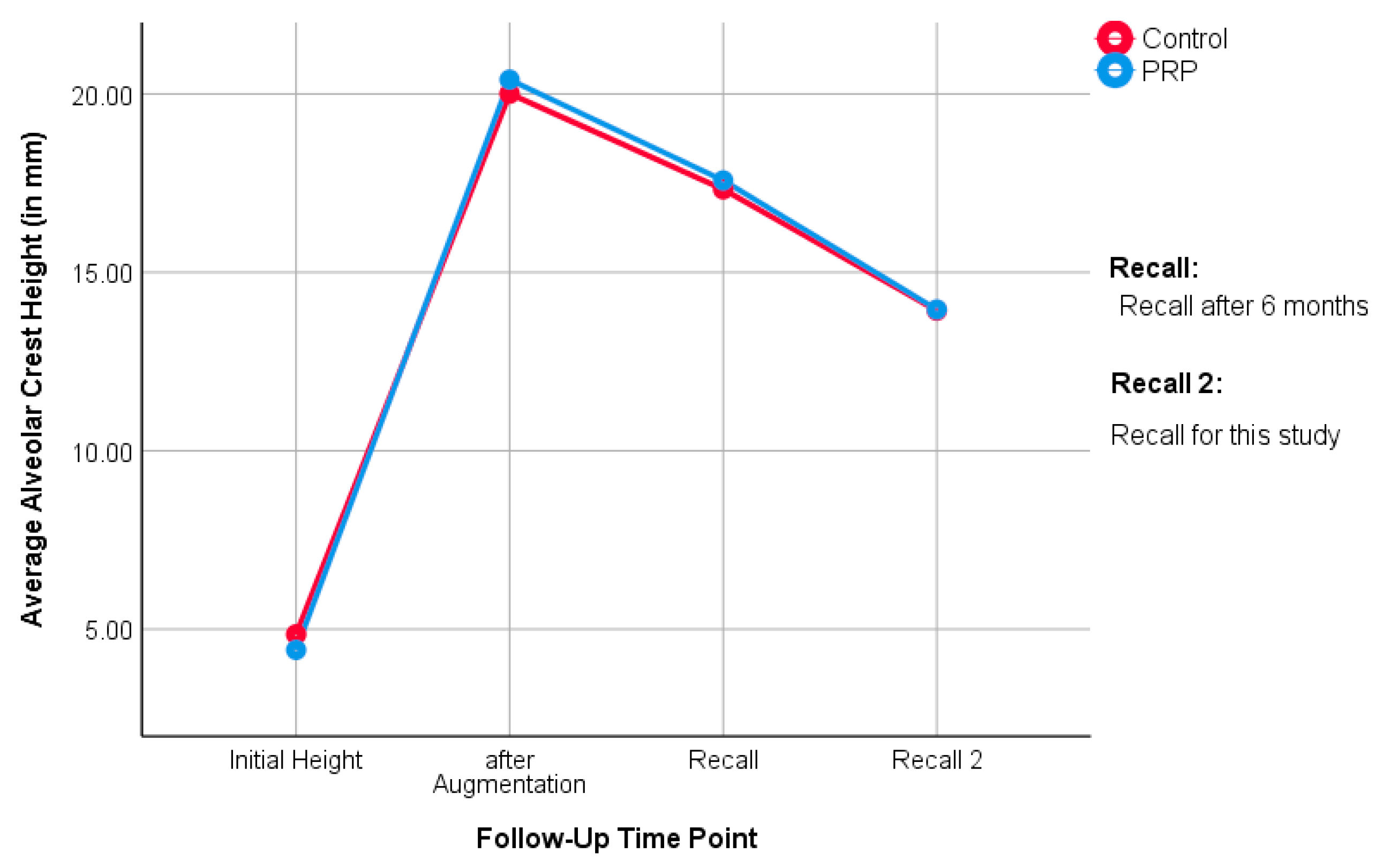
The median alveolar crest height in the PRP group was 13.3 mm (split-mouth evaluation) and 12.8 mm (unilateral evaluation), while it was 12.7 mm (split-mouth evaluation) and 12.8 mm (unilateral evaluation) in the control group. Here, it can be seen that the PRP group had slightly more bone height than the control group, until the first follow-up in the study of Schaaf et al. [
19,
20]. Over time, the groups ended up at equal heights. This is also noticeable when looking at the medians: in the split-mouth evaluation, the medians of the study groups are close to each other. In the unilateral evaluation, they are even identical. This suggests that the influence of PRP on the bone level of the augmented region is, if present, a short-term result. No statistical differences between the groups could be determined in the long-term follow-up.
The percent of bone loss of the augmented region relates absolute bone loss to the baseline. As a result, the percentage change is a meaningful measure for the resorption of the augmented jaw region. The median percentage of bone loss in the PRP group was 31.5% (split-mouth evaluation) and 35% (unilateral evaluation). In the control group, the values were 25.6% (split-mouth evaluation) and 27.2% (unilateral evaluation). The statistical analysis did not show any significant difference between the groups. Schmitt et al. examined 25 patients who had received augmentations in the upper jaw. After ten years, the authors found that 30.2% of augmented bone was resorbed. These findings can be regarded as equivalent to this study [
41].
The majority of the clinical and radiological parameters did not show any difference between the PRP group and the control group. Thus, the hypothesis defined in this study could not be proven. Most of these results correspond to current literature and follow the findings of the previous study by Schaaf et al. [
19,
20]. In the same patient collection, there was no positive effect of PRP in the use of autologous bone grafts at the upper jaw in the short-term outcomes. Now, in this long-term clinical and radiological follow-up, no change could be observed, either.
Ample evidence on the use of regenerative therapy approaches are present in the literature, most of which circumvent one or more limitation of autologous bone grafts and PRP enhancement. Nonetheless, many, if not all, alternative methods open other issues in terms of ethics, feasibility, and specialization.
Using scaffolds in their various forms, decellularized matrix, hydrogels, or spun collagens have certain advantages in handling, and there is the possibility of loading them with mesenchymal stem cells (MSCs). Such scaffolds have shown enhanced osteogenic properties in vitro and enhanced differentiation of loaded stem cells [
27]. In dental treatment, the recent discovery of periapical cyst-mesenchymal stem cells will advance this concept as a local source for stem cells [
42]. To benefit from the scaffolds properties, such as porosity and low coast, osteoconductive rather than osteoinductive approaches have been addressed, especially in disturbed bone healing in cases of tumorous bone such as multiple myeloma [
43], or systemically diseased bone such as in osteoporosis [
44]. Different compounds could enhance osteoblasts or inhibit osteoclasts in vitro such as Donepezil [
45]. The use of minerals such as calcium phosphate cements doped with active ingredients such as strontium [
46] or nanoparticles loaded on scaffolds such as nanosilicates poly(glycerol sebacate) [
13], showed enhanced mineralization in vivo and in vitro.
There are new upcoming development towards smart biomaterials and a faster implementation and integration of materials in patient treatment by modulating the immune response [
47,
48]. The main idea is to implement biomaterials to regulate inflammatory response, one of the major challenges in tissue grafts and an often neglected effect by clinicians. The future aim is to use the immune reaction to manage drug release temporarily and locally according to the need [
49]. One of the methods to regulate the immune response is the regulation of oxidative stress, which is involved in the response to pathogen infection and inflammation [
50,
51]. However, these procedures are still far from daily clinical applications and are costly for the patient.
This paper aimed to fairly assess bone augmentation with PRP after a long-term follow-up, bone augmentation having been the state-of-the-art approach 20 years ago. Besides there being a long way until becoming a certified product, all the above-mentioned technologies are still in the early stages of development with variable unknown factors. Further long-term follow-up studies will be necessary to prove their validity.