Advances and Trends in Pediatric Minimally Invasive Surgery
Abstract
1. Introduction
2. Materials and Methods
3. Review
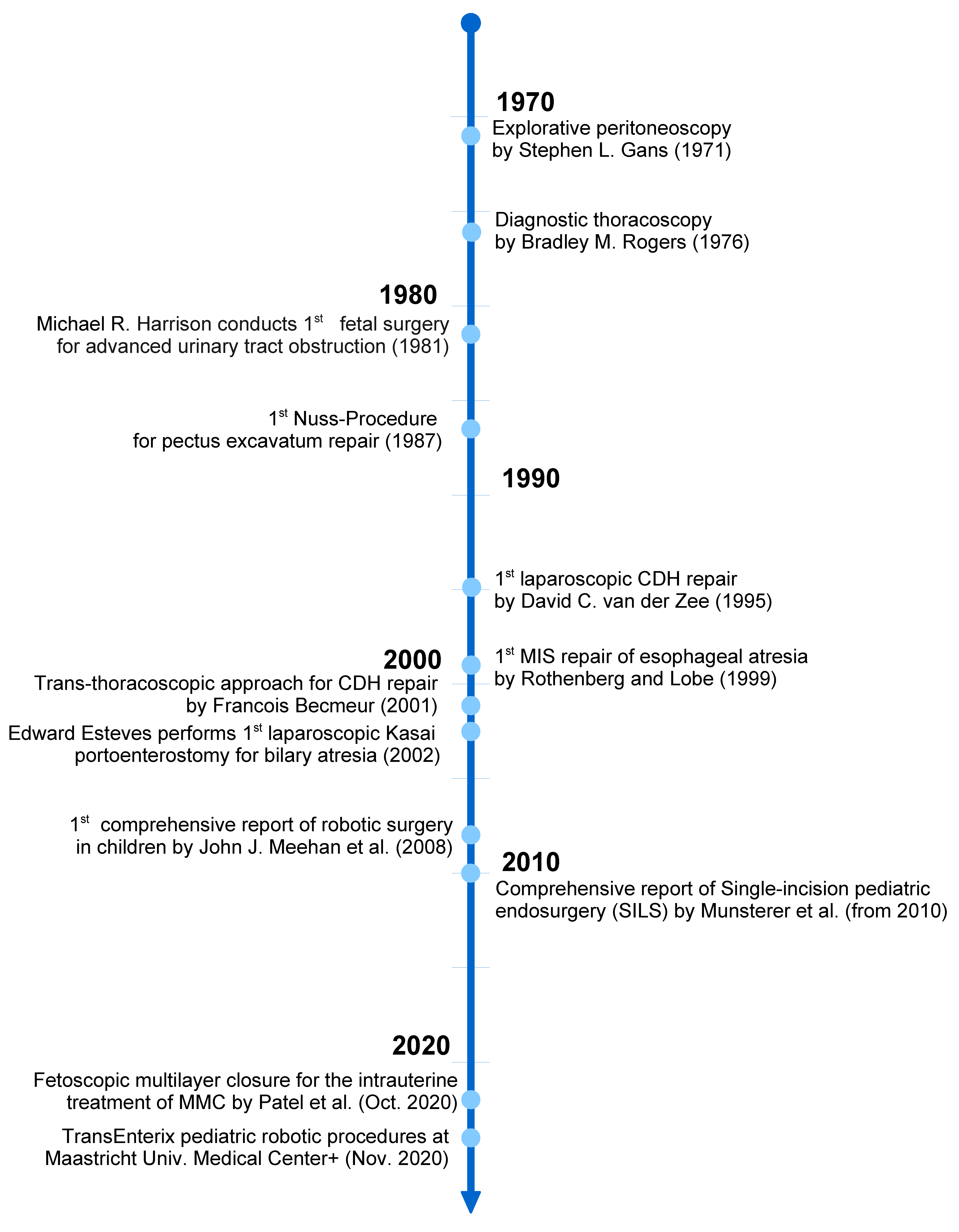
3.1. Milestones of Pediatric Minimally Invasive Surgery
3.2. Technical Developments of Pediatric Laparoscopy
3.3. Natural Orifice Transluminal Endoscopic Surgery (NOTES)
3.4. Single Incision Laparoscopic and Thoracoscopic Surgery
3.5. Endoscopic Pediatric Surgery
3.6. Robotic Pediatric Surgery
3.7. Fetal Surgery
3.8. Outlook: The Future of Pediatric Minimally Invasive Surgery
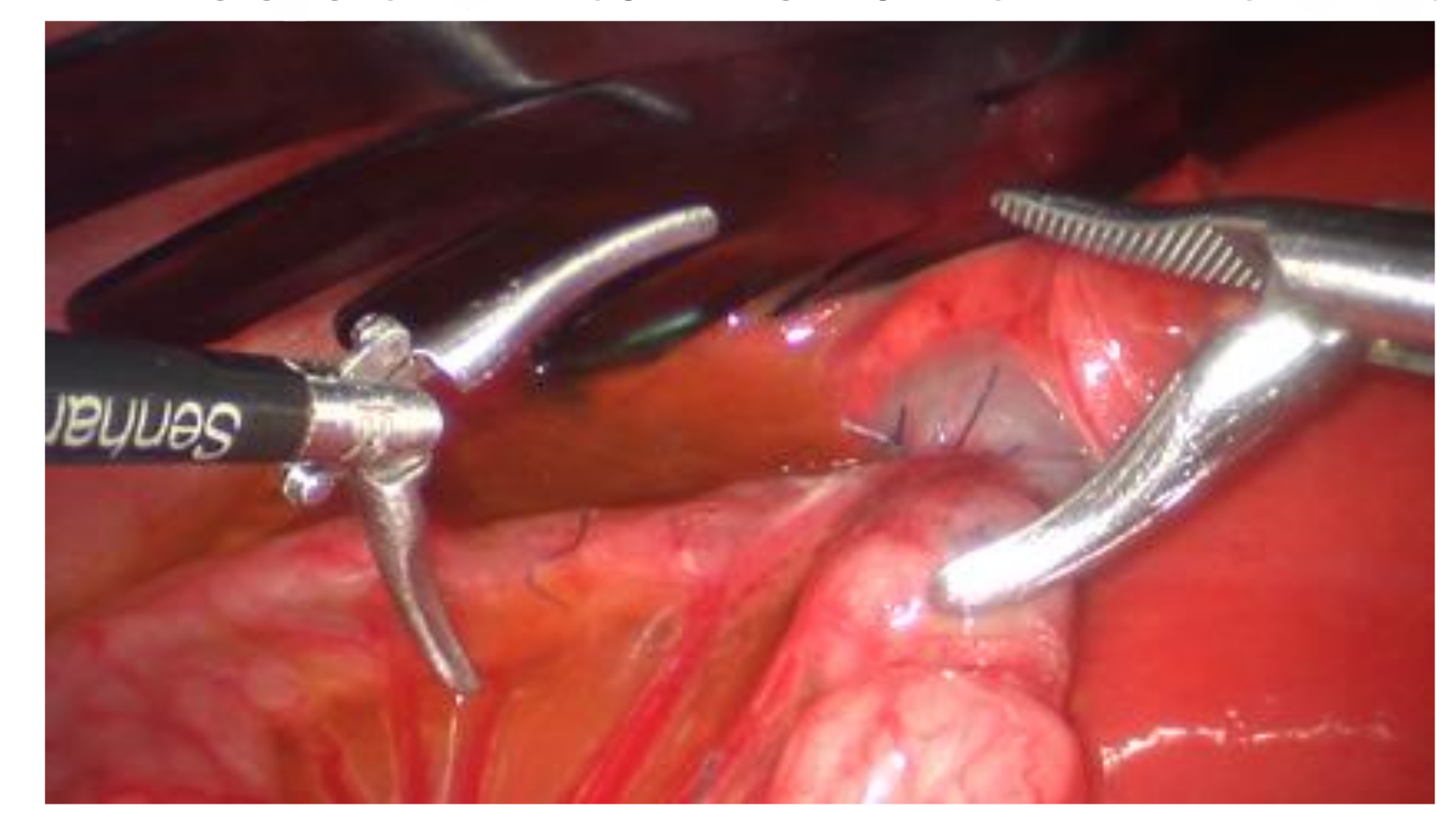
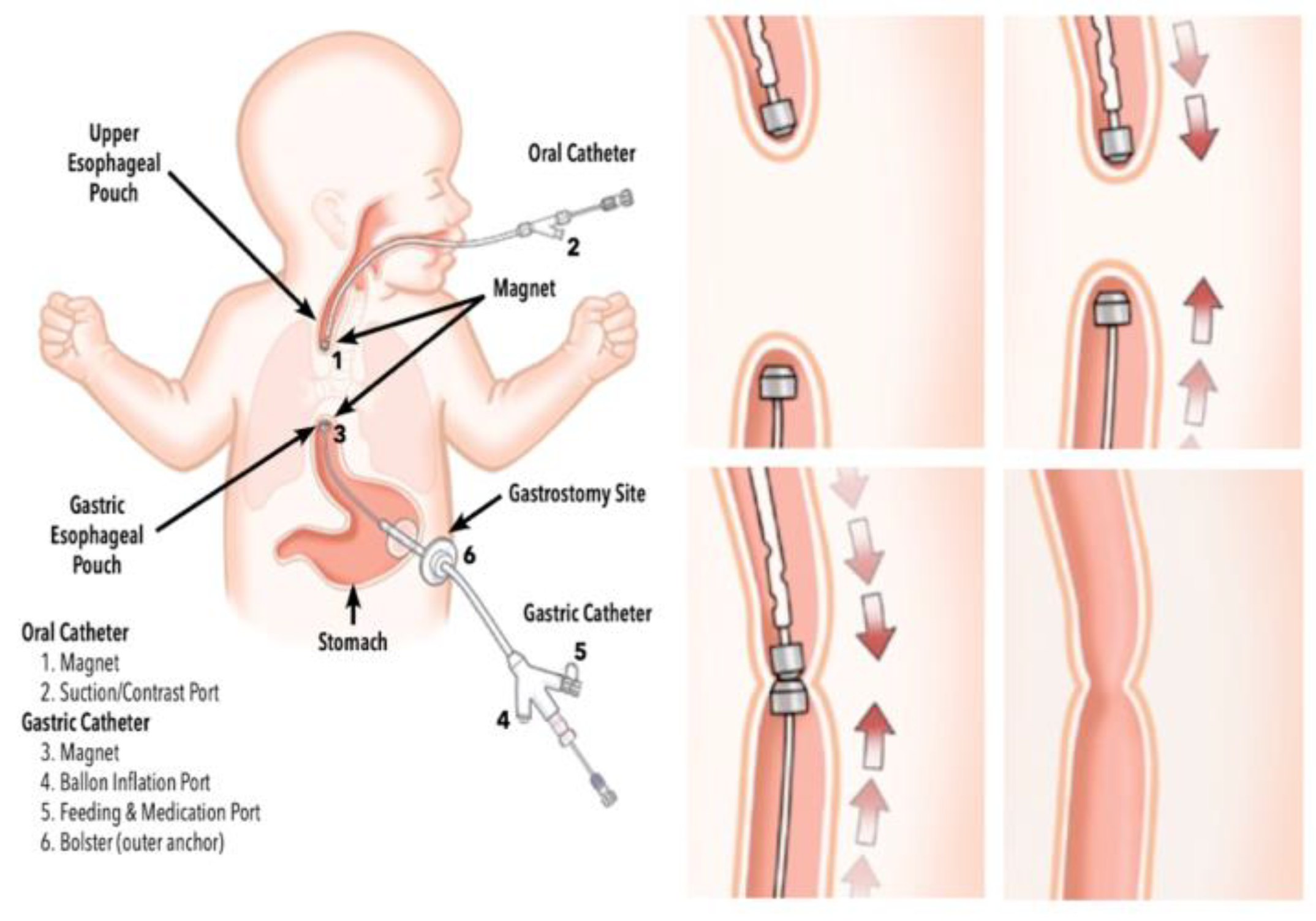
3.8.1. Magnetic Anastomosing Devices
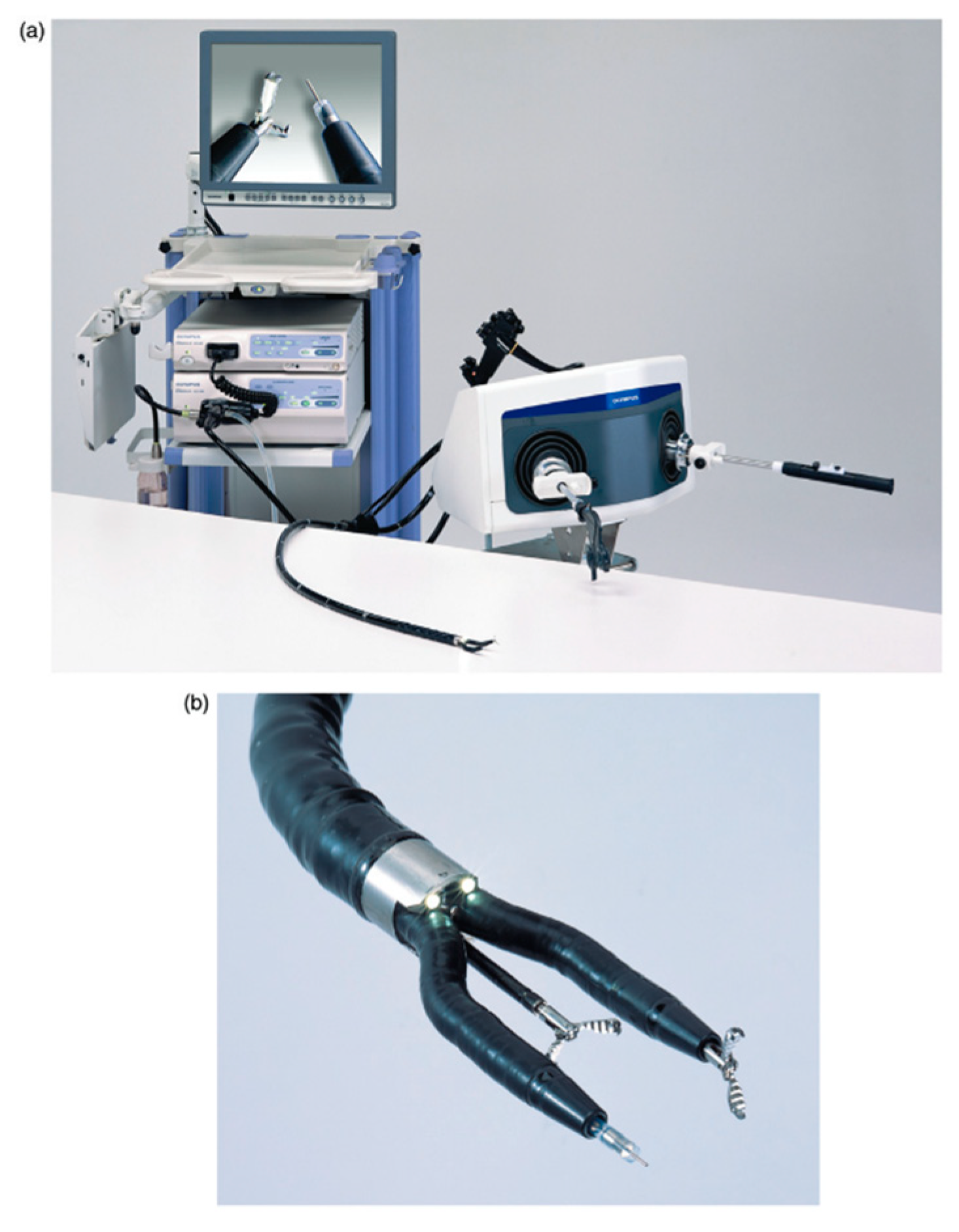
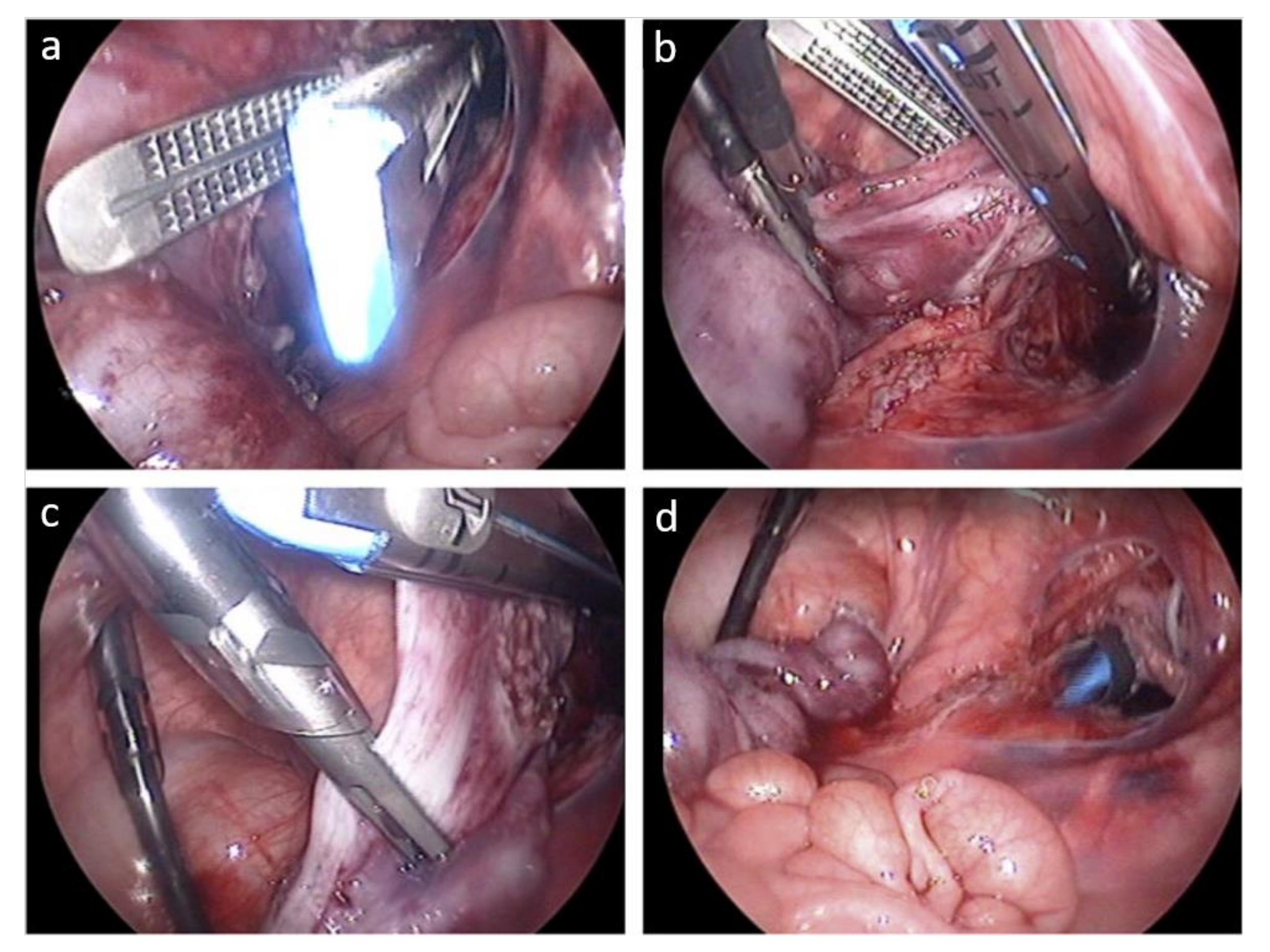
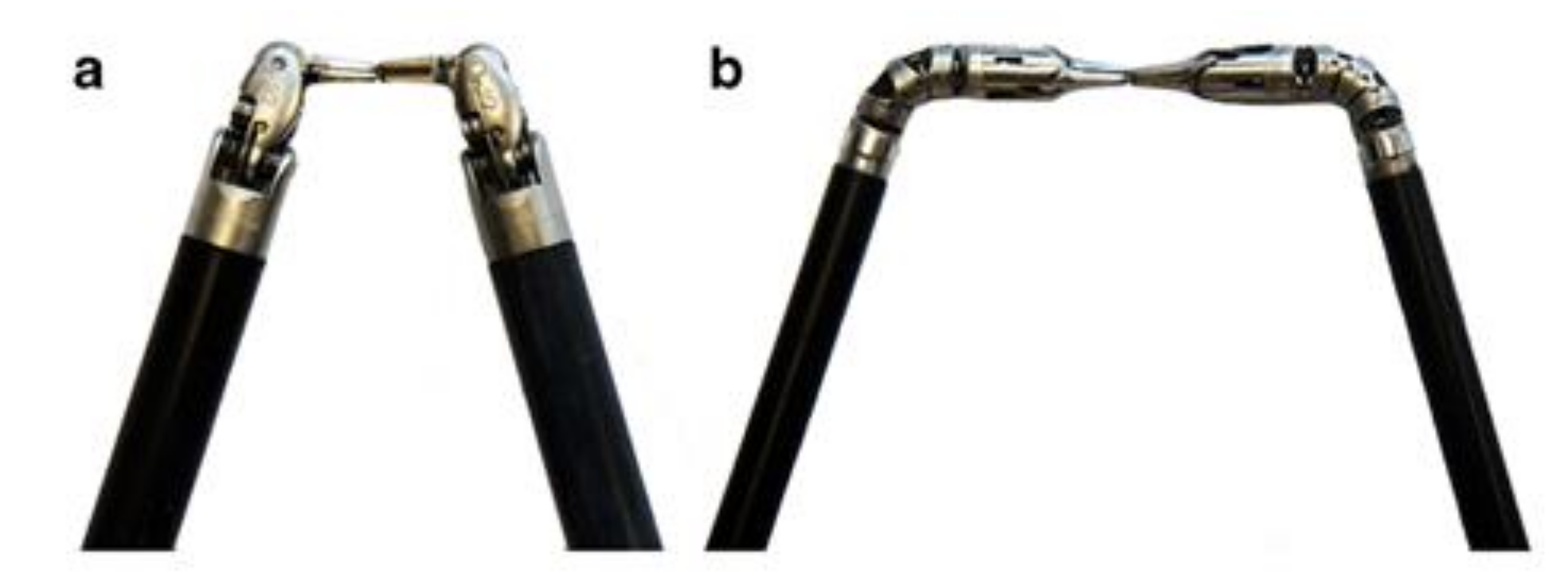
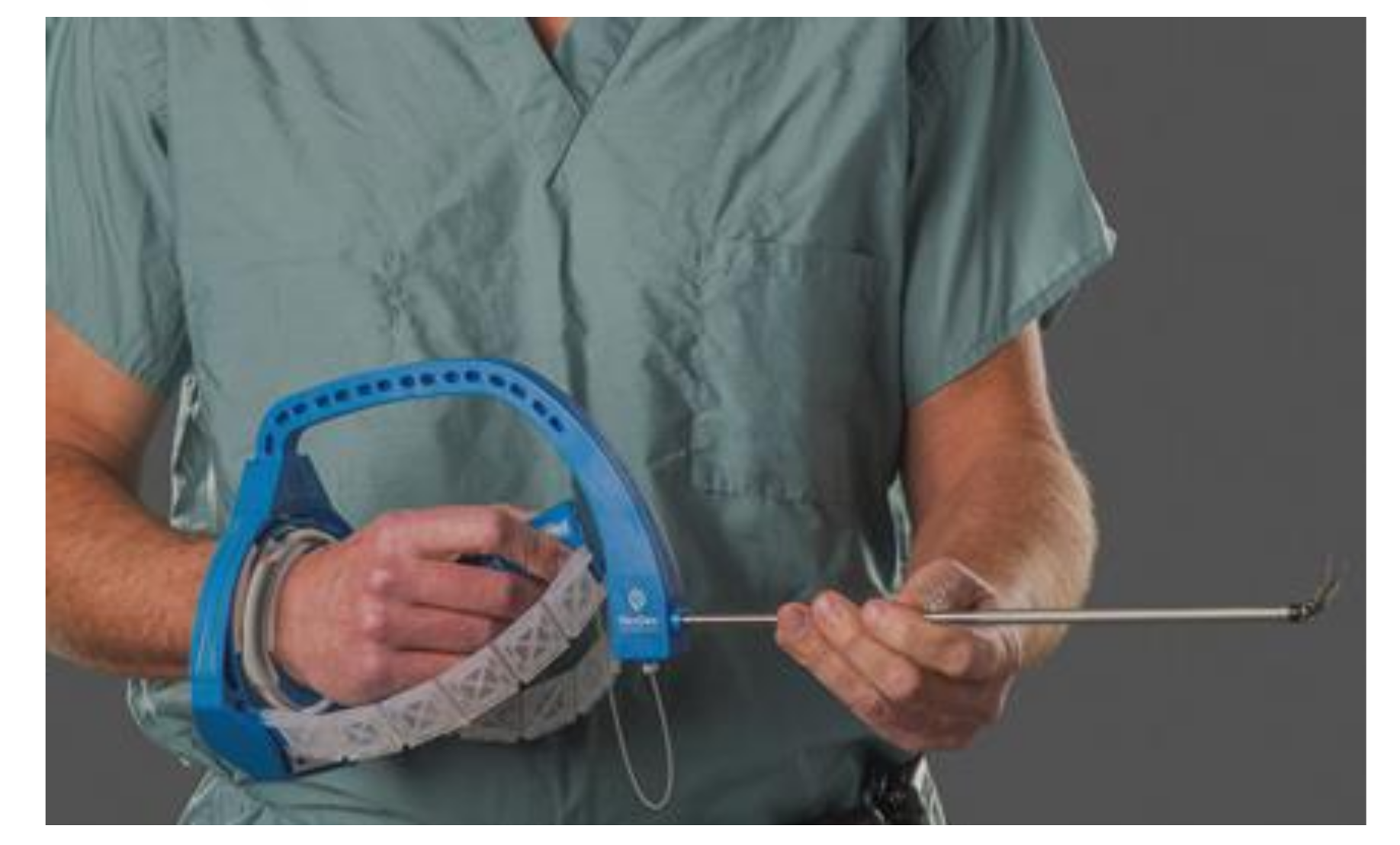
3.8.2. Articulating Laparoscopic Instruments and Devices for Single-Incision Laparoscopic Surgery
3.8.3. New Robots for Children
3.8.4. Deployable Minirobots
3.8.5. Hybrid Procedures
3.8.6. Robots, SILS and NOTES, the Ideal Combination?
3.8.7. Artificial Intelligence and Augmented Reality in Pediatric Surgery
3.8.8. The Artificial Womb
3.9. In the End: Hard Data on Minimally Invasive Surgery
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Jackson, H.T.; Kane, T.D. Advances in minimally invasive surgery in pediatric patients. Adv. Pediatr. 2014, 61, 149–195. [Google Scholar] [CrossRef] [PubMed]
- Schukfeh, N.; Kuebler, J.F.; Dingemann, J.; Ure, B.M. Thirty Years of Minimally Invasive Surgery in Children: Analysis of Meta-Analyses. Eur. J. Pediatr. Surg. 2020, 30, 420–428. [Google Scholar] [CrossRef] [PubMed]
- Semm, K. Endoscopic appendectomy. Endoscopy 1983, 15, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Litynski, G.S. Profiles in laparoscopy: Mouret, Dubois, and Perissat: The laparoscopic breakthrough in Europe (1987-1988). JSLS 1999, 3, 163–167. [Google Scholar]
- Litynski, G.S. Erich Mühe and the Rejection of Laparoscopic Cholecystectomy (1985): A Surgeon Ahead of His Time. JSLS 1998, 2, 341–346. [Google Scholar]
- Carnevale, N.; Baron, N.; Delany, H.M. Peritoneoscopy as an aid in the diagnosis of abdominal trauma: A preliminary report. J. Trauma 1977, 17, 634–641. [Google Scholar] [CrossRef]
- Gans, S.L.; Berci, G. Advances in endoscopy of infants and children. J. Pediatr. Surg. 1971, 6, 199–233. [Google Scholar] [CrossRef]
- Rodgers, B.M.; Talbert, J.L. Thoracoscopy for diagnosis of intrathoracic lesions in children. J. Pediatr. Surg. 1976, 11, 703–708. [Google Scholar] [CrossRef]
- van der Zee, D.C.; Bax, N.M. Laparoscopic repair of congenital diaphragmatic hernia in a 6-month-old child. Surg. Endosc. 1995, 9, 1001–1003. [Google Scholar] [CrossRef]
- Becmeur, F.; Jamali, R.R.; Moog, R.; Keller, L.; Christmann, D.; Donato, L.; Kauffmann, I.; Schwaab, C.; Carrenard, G.; Sauvage, P. Thoracoscopic treatment for delayed presentation of congenital diaphragmatic hernia in the infant. A report of three cases. Surg. Endosc. 2001, 15, 1163–1166. [Google Scholar] [CrossRef]
- Lobe, T.E.; Rothenberg, S.; Waldschmidt, J.; Stroedter, L. Thoracoscopic Repair of Esophageal Atresia in an Infant: A Surgical First. Pediatr. Endosurg. Innovat. Tech. 1999, 3, 141–148. [Google Scholar] [CrossRef]
- Nuss, D.; Kelly, R.E.; Croitoru, D.P.; Katz, M.E. A 10-year review of a minimally invasive technique for the correction of pectus excavatum. J. Pediatr. Surg. 1998, 33, 545–552. [Google Scholar] [CrossRef]
- Farello, G.A.; Cerofolini, A.; Rebonato, M.; Bergamaschi, G.; Ferrari, C.; Chiappetta, A. Congenital choledochal cyst: Video-guided laparoscopic treatment. Surg. Laparosc. Endosc. 1995, 5, 354–358. [Google Scholar] [PubMed]
- Esteves, E.; Clemente Neto, E.; Ottaiano Neto, M.; Devanir, J.; Esteves Pereira, R. Laparoscopic Kasai portoenterostomy for biliary atresia. Pediatr. Surg. Int. 2002, 18, 737–740. [Google Scholar] [CrossRef] [PubMed]
- Meehan, J.J.; Sandler, A. Pediatric robotic surgery: A single-institutional review of the first 100 consecutive cases. Surg. Endosc. 2008, 22, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Muensterer, O.J.; Adibe, O.O.; Harmon, C.M.; Chong, A.; Hansen, E.N.; Bartle, D.; Georgeson, K.E. Single-incision laparoscopic pyloromyotomy: Initial experience. Surg. Endosc. 2010, 24, 1589–1593. [Google Scholar] [CrossRef]
- Hansen, E.N.; Muensterer, O.J.; Georgeson, K.E.; Harmon, C.M. Single-incision pediatric endosurgery: Lessons learned from our first 224 laparoendoscopic single-site procedures in children. Pediatr. Surg. Int. 2011, 27, 643–648. [Google Scholar] [CrossRef]
- O’Donovan, P.J.; McGurgan, P. Microlaparoscopy. Semin. Laparosc. Surg. 1999, 6, 51–57. [Google Scholar] [CrossRef]
- Turial, S.; Schwind, M.; Kohl, M.; Goldinger, B.; Schier, F. Feasibility of microlaparoscopy for surgical procedures of advanced complexity in children. J. Laparoendosc Adv. Surg. Tech. A 2009, 19 Suppl. 1, S103–S105. [Google Scholar] [CrossRef]
- Cuaresma, R.; Benavides, M.; Buela, E.; Bignon, H.; Martínez-Ferro, M. Laparoscopic appendectomies in pediatric patients using Hem-o-lock clips. Cir. Pediatr. 2009, 22, 103–105. [Google Scholar]
- Lv, B.; Zhang, X.; Li, J.; Leng, S.; Li, S.; Zeng, Y.; Wang, B.; Yuan, J.; Fan, J.; Xing, S.; et al. Absorbable polymeric surgical clips for appendicular stump closure: A randomized control trial of laparoscopic appendectomy with lapro-clips. Oncotarget 2016, 7, 41265–41273. [Google Scholar] [CrossRef] [PubMed]
- Harrell, A.G.; Kercher, K.W.; Heniford, B.T. Energy sources in laparoscopy. Semin. Laparosc. Surg. 2004, 11, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Highlights | KARL STORZ Endoskope. Available online: https://www.karlstorz.com/de/en/highlights-tp.htm#mod-9323 (accessed on 12 November 2020).
- Tsin, D.A.; Sequeria, R.J.; Giannikas, G. Culdolaparoscopic cholecystectomy during vaginal hysterectomy. JSLS 2003, 7, 171–172. [Google Scholar] [PubMed]
- Bernhardt, J.; Gerber, B.; Schober, H.-C.; Kähler, G.; Ludwig, K. NOTES—Case report of a unidirectional flexible appendectomy. Int. J. Colorectal. Dis. 2008, 23, 547–550. [Google Scholar] [CrossRef] [PubMed]
- Kaouk, J.H.; Haber, G.-P.; Goel, R.K.; Crouzet, S.; Brethauer, S.; Firoozi, F.; Goldman, H.B.; White, W.M. Pure natural orifice translumenal endoscopic surgery (NOTES) transvaginal nephrectomy. Eur. Urol. 2010, 57, 723–726. [Google Scholar] [CrossRef]
- Horgan, S.; Cullen, J.P.; Talamini, M.A.; Mintz, Y.; Ferreres, A.; Jacobsen, G.R.; Sandler, B.; Bosia, J.; Savides, T.; Easter, D.W.; et al. Natural orifice surgery: Initial clinical experience. Surg. Endosc. 2009, 23, 1512–1518. [Google Scholar] [CrossRef]
- Kume, K. Flexible robotic endoscopy: Current and original devices. Comput. Assist. Surg. 2016, 21, 150–159. [Google Scholar] [CrossRef]
- Zornig, C.; Mofid, H.; Siemssen, L.; Emmermann, A.; Alm, M.; von Waldenfels, H.-A.; Felixmüller, C. Transvaginal NOTES hybrid cholecystectomy: Feasibility results in 68 cases with mid-term follow-up. Endoscopy 2009, 41, 391–394. [Google Scholar] [CrossRef]
- Pulvirenti, E.; Toro, A.; Di Carlo, I. Update on Instrumentations for Cholecystectomies Performed via Transvaginal Route: State of the Art and Future Prospectives. Diagn. Ther. Endosc. 2010, 2010, 405469. [Google Scholar] [CrossRef]
- Our Journey – History from 1945 | KARL STORZ Endoskope | Croatia. Available online: https://www.karlstorz.com/hr/en/history.htm (accessed on 6 December 2020).
- Bulian, D.R.; Knuth, J.; Cerasani, N.; Sauerwald, A.; Lefering, R.; Heiss, M.M. Transvaginal/Transumbilical Hybrid-NOTES-Versus 3-Trocar Needlescopic Cholecystectomy: Short-term Results of a Randomized Clinical Trial. Ann. Surg. 2015, 261, 451–458. [Google Scholar] [CrossRef]
- Ryou, M.; Thompson, C.C. Magnetic retraction in natural-orifice transluminal endoscopic surgery (NOTES): Addressing the problem of traction and countertraction. Endoscopy 2009, 41, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Yang, E.; Nie, D.; Li, Z. Comparison of Major Clinical Outcomes Between Transvaginal NOTES and Traditional Laparoscopic Surgery: A Systematic Review and Meta-analysis. J. Surg Res. 2019, 244, 278–290. [Google Scholar] [CrossRef] [PubMed]
- Lamas-Pinheiro, R.; Henriques-Coelho, T.; Carvalho, J.L.; Correia-Pinto, J. Duhamel pull-through assisted by transrectal port: A hybrid natural orifice transluminal endoscopic surgery approach. J. Pediatr. Surg. 2012, 47, 1962–1965. [Google Scholar] [CrossRef]
- Farr, B.J.; Fox, V.L.; Mooney, D.P. Endoscopic cyst gastrostomy for traumatic pancreatic pseudocysts in children: A case series. Trauma Surg. Acute Care Open 2020, 5, e000456. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.S.; Maharshi, S. Endoscopic management of pancreatic pseudocyst in children-a long-term follow-up. J. Pediatr. Surg. 2008, 43, 1636–1639. [Google Scholar] [CrossRef]
- Oltmann, S.C.; Garcia, N.M.; Ventura, B.; Mitchell, I.; Fischer, A.C. Single-incision laparoscopic surgery: Feasibility for pediatric appendectomies. J. Pediatr. Surg. 2010, 45, 1208–1212. [Google Scholar] [CrossRef]
- Ostlie, D.J.; Miller, K.A.; Woods, R.K.; Holcomb, G.W. Single cannula technique and robotic telescopic assistance in infants and children who require laparoscopic Nissen fundoplication. J. Pediatr. Surg. 2003, 38, 111–115; discussion 111–115. [Google Scholar] [CrossRef]
- Desai, M.M.; Rao, P.P.; Aron, M.; Pascal-Haber, G.; Desai, M.R.; Mishra, S.; Kaouk, J.H.; Gill, I.S. Scarless single port transumbilical nephrectomy and pyeloplasty: First clinical report. BJU Int. 2008, 101, 83–88. [Google Scholar] [CrossRef]
- Martínez-Ferro, M.; Duarte, S.; Laje, P. Single-port thoracoscopy for the treatment of pleural empyema in children. J. Pediatr. Surg. 2004, 39, 1194–1196. [Google Scholar] [CrossRef]
- Frutos, M.D.; Abrisqueta, J.; Lujan, J.; Abellan, I.; Parrilla, P. Randomized Prospective Study to Compare Laparoscopic Appendectomy Versus Umbilical Single-Incision Appendectomy. Ann. Surg. 2013, 257, 413–418. [Google Scholar] [CrossRef]
- Deng, L.; Xiong, J.; Xia, Q. Single-incision versus conventional three-incision laparoscopic appendectomy: A meta-analysis of randomized controlled trials. J. Evid. Based Med. 2017, 10, 196–206. [Google Scholar] [CrossRef]
- Michailidou, M.; Goldstein, S.D.; Sacco Casamassima, M.G.; Salazar, J.H.; Elliott, R.; Hundt, J.; Abdullah, F. Laparoscopic versus open appendectomy in children: The effect of surgical technique on healthcare costs. Am. J. Surg. 2015, 210, 270–275. [Google Scholar] [CrossRef]
- Li, X.; Zhang, J.; Sang, L.; Zhang, W.; Chu, Z.; Li, X.; Liu, Y. Laparoscopic versus conventional appendectomy—A meta-analysis of randomized controlled trials. BMC Gastroenterol. 2010, 10, 129. [Google Scholar] [CrossRef] [PubMed]
- Hermans, B.P.; Otte, J.B. Laparoscopic appendicectomy: Pros & cons--literature review of 4190 cases. Acta. Chir. Belg. 1997, 97, 110–117. [Google Scholar] [PubMed]
- Pelosi, M.A.; Pelosi, M.A., 3rd. Laparoscopic appendectomy using a single umbilical puncture (minilaparoscopy). J. Reprod. Med. 1992, 37, 588–594. [Google Scholar] [PubMed]
- Bergholz, R.; Krebs, T.F.; Klein, I.; Wenke, K.; Reinshagen, K. Transumbilical laparoscopic-assisted versus 3-port laparoscopic and open appendectomy: A case-control study in children. Surg. Laparosc. Endosc. Percutan Tech. 2014, 24, 244–247. [Google Scholar] [CrossRef]
- Bergholz, R.; Klein, I.; Wenke, K.; Boettcher, M.; Reinshagen, K.; Krebs, T. Midterm outcome of transumbilically laparoscopic-assisted versus laparoscopic and open appendectomy in children--a matched prospective study. Eur. J. Pediatr. Surg. 2015, 25, 216–219. [Google Scholar] [CrossRef]
- Esposito, C. One-trocar appendectomy in pediatric surgery. Surg. Endosc. 1998, 12, 177–178. [Google Scholar] [CrossRef]
- Visnjic, S. Transumbilical laparoscopically assisted appendectomy in children: High-tech low-budget surgery. Surg. Endosc. 2008, 22, 1667–1671. [Google Scholar] [CrossRef] [PubMed]
- Cobellis, G.; Torino, G.; Noviello, C.; Cruccetti, A.; Mastroianni, L.; Amici, G.; Martino, A. Versatility of one-trocar surgery in children. J. Laparoendosc. Adv. Surg. Tech. A 2011, 21, 549–554. [Google Scholar] [CrossRef]
- van Lennep, M.; van Wijk, M.P.; Omari, T.I.M.; Salvatore, S.; Benninga, M.A.; Singendonk, M.M.J. European Society for Paediatric Gastroenterology, Hepatology and Nutrition Motility Working Group Clinical Management of Pediatric Achalasia: A Survey of Current Practice. J. Pediatr. Gastroenterol. Nutr. 2019, 68, 521–526. [Google Scholar] [CrossRef] [PubMed]
- Wood, L.S.; Chandler, J.M.; Portelli, K.E.; Taylor, J.S.; Kethman, W.C.; Wall, J.K. Treating children with achalasia using per-oral endoscopic myotomy (POEM): Twenty-one cases in review. J. Pediatr. Surg. 2020, 55, 1006–1012. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.-R.; Ma, X.; Feng, J.; Yang, Z.; Qu, B.; Feng, Z.-T.; Ma, S.-R.; Yin, J.-B.; Sun, R.; Guo, L.-L.; et al. Endoscopic retrograde appendicitis therapy (ERAT): A multicenter retrospective study in China. Surg. Endosc. 2015, 29, 905–909. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.; Zhang, W.; Zeng, L.; Lin, Y.; Wu, J.; Zhang, N.; Xie, X.; Zhang, Y.; Liu, X.; Wang, B.; et al. The modified endoscopic retrograde appendicitis therapy versus antibiotic therapy alone for acute uncomplicated appendicitis in children. Surg. Endosc. 2020. [Google Scholar] [CrossRef] [PubMed]
- Bowen, D.K.; Van Batavia, J.P.; Srinivasan, A.K. Single-Site Laparoscopy and Robotic Surgery in Pediatric Urology. Curr. Urol. Rep. 2018, 19, 42. [Google Scholar] [CrossRef]
- Cave, J.; Clarke, S. Paediatric robotic surgery. Ann. R Coll. Surg. Engl. 2018, 100, 18–21. [Google Scholar] [CrossRef]
- Van Haasteren, G.; Levine, S.; Hayes, W. Pediatric Robotic Surgery: Early Assessment. Pediatrics 2009, 124, 1642–1649. [Google Scholar] [CrossRef]
- Kawal, T.; Srinivasan, A.K.; Shrivastava, D.; Chu, D.I.; Van Batavia, J.; Weiss, D.; Long, C.; Shukla, A.R. Pediatric robotic-assisted laparoscopic pyeloplasty: Does age matter? J. Pediatr. Urol. 2018, 14, 540.e1–540.e6. [Google Scholar] [CrossRef]
- Spinoit, A.-F.; Nguyen, H.; Subramaniam, R. Role of Robotics in Children: A brave New World! Eur. Urol. Focus 2017, 3, 172–180. [Google Scholar] [CrossRef]
- Marcus, H.J.; Hughes-Hallett, A.; Cundy, T.P.; Yang, G.-Z.; Darzi, A.; Nandi, D. da Vinci robot-assisted keyhole neurosurgery: A cadaver study on feasibility and safety. Neurosurg. Rev. 2015, 38, 367–371; discussion 371. [Google Scholar] [CrossRef]
- Thakre, A.A.; Bailly, Y.; Sun, L.W.; Van Meer, F.; Yeung, C.K. Is smaller workspace a limitation for robot performance in laparoscopy? J. Urol. 2008, 179, 1138–1142; discussion 1142–1143. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, N.; Barco-Castillo, C.; ElGhazzaoui, A.; Farhat, W. Effective intracorporeal space in robot-assisted multiquadrant surgery in a pediatric inanimate model. J. Robot. Surg. 2020. [Google Scholar] [CrossRef] [PubMed]
- Finkelstein, J.B.; Levy, A.C.; Silva, M.V.; Murray, L.; Delaney, C.; Casale, P. How to decide which infant can have robotic surgery? Just do the math. J. Pediatr. Urol. 2015, 11, 170.e1–170.e4. [Google Scholar] [CrossRef] [PubMed]
- Bergholz, R.; Botden, S.; Verweij, J.; Tytgat, S.; Van Gemert, W.; Boettcher, M.; Ehlert, H.; Reinshagen, K.; Gidaro, S. Evaluation of a new robotic-assisted laparoscopic surgical system for procedures in small cavities. J. Robot. Surg. 2019, 14, 191–197. [Google Scholar] [CrossRef] [PubMed]
- TransEnterix Announces Maastricht University Medical Center+ in the Netherlands to Initiate Program with the Senhance Surgical System | TransEnterix, Inc. Available online: https://ir.transenterix.com/news-releases/news-release-details/transenterix-announces-maastricht-university-medical-center (accessed on 3 November 2020).
- Looi, T.; Yeung, B.; Umasthan, M.; Drake, J. KidsArm — An image-guided pediatric anastomosis robot. In Proceedings of the 2013 IEEE/RSJ International Conference on Intelligent Robots and Systems, Tokyo, Japan, 3–7 November 2013; pp. 4105–4110. [Google Scholar]
- Leonard, S.; Wu, K.L.; Kim, Y.; Krieger, A.; Kim, P.C.W. Smart tissue anastomosis robot (STAR): A vision-guided robotics system for laparoscopic suturing. IEEE Trans. Biomed. Eng. 2014, 61, 1305–1317. [Google Scholar] [CrossRef]
- Harrison, M.R.; Adzick, N.S.; Flake, A.W.; Jennings, R.W.; Estes, J.M.; MacGillivray, T.E.; Chueh, J.T.; Goldberg, J.D.; Filly, R.A.; Goldstein, R.B. Correction of congenital diaphragmatic hernia in utero: VI. Hard-earned lessons. J. Pediatr. Surg. 1993, 28, 1411–1417; discussion 1417–1418. [Google Scholar] [CrossRef]
- Harrison, M.R.; Golbus, M.S.; Filly, R.A.; Nakayama, D.K.; deLorimier, A.A. Fetal surgical treatment. Pediatr. Ann. 1982, 11, 896–899, 901–903. [Google Scholar]
- Peiró, J.L.; Carreras, E.; Guillén, G.; Arévalo, S.; Sánchez-Durán, M.A.; Higueras, T.; Castillo, F.; Marhuenda, C.; Lloret, J.; Martínez-Ibáñez, V. Therapeutic indications of fetoscopy: A 5-year institutional experience. J. Laparoendosc Adv. Surg. Tech. A 2009, 19, 229–236. [Google Scholar] [CrossRef]
- Diehl, W.; Diemert, A.; Grasso, D.; Sehner, S.; Wegscheider, K.; Hecher, K. Fetoscopic laser coagulation in 1020 pregnancies with twin-twin transfusion syndrome demonstrates improvement in double-twin survival rate. Ultrasound Obstet. Gynecol. 2017, 50, 728–735. [Google Scholar] [CrossRef]
- Ruano, R.; Yoshisaki, C.T.; da Silva, M.M.; Ceccon, M.E.J.; Grasi, M.S.; Tannuri, U.; Zugaib, M. A randomized controlled trial of fetal endoscopic tracheal occlusion versus postnatal management of severe isolated congenital diaphragmatic hernia. Ultrasound Obstet. Gynecol. 2012, 39, 20–27. [Google Scholar] [CrossRef]
- Deprest, J.; Brady, P.; Nicolaides, K.; Benachi, A.; Berg, C.; Vermeesch, J.; Gardener, G.; Gratacos, E. Prenatal management of the fetus with isolated congenital diaphragmatic hernia in the era of the TOTAL trial. Semin. Fetal. Neonatal. Med. 2014, 19, 338–348. [Google Scholar] [CrossRef] [PubMed]
- Ruano, R.; Sananes, N.; Sangi-Haghpeykar, H.; Hernandez-Ruano, S.; Moog, R.; Becmeur, F.; Zaloszyc, A.; Giron, A.M.; Morin, B.; Favre, R. Fetal intervention for severe lower urinary tract obstruction: A multicenter case-control study comparing fetal cystoscopy with vesicoamniotic shunting. Ultrasound Obstet. Gynecol. 2015, 45, 452–458. [Google Scholar] [CrossRef] [PubMed]
- Kabagambe, S.K.; Jensen, G.W.; Chen, Y.J.; Vanover, M.A.; Farmer, D.L. Fetal Surgery for Myelomeningocele: A Systematic Review and Meta-Analysis of Outcomes in Fetoscopic versus Open Repair. Fetal. Diagn. Ther. 2017. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.K.; Habli, M.A.; McKinney, D.N.; Tabbah, S.M.; Lim, F.-Y.; Peiro, J.L.; Stevenson, C.B. Fetoscopic Multilayer, Dural Patch Closure Technique for Intrauterine Myelomeningocele Repair: 2-Dimensional Operative Video. Oper. Neurosurg. 2020. [Google Scholar] [CrossRef]
- Nassr, A.A.; Erfani, H.; Fisher, J.E.; Ogunleye, O.K.; Espinoza, J.; Belfort, M.A.; Shamshirsaz, A.A. Fetal interventional procedures and surgeries: A practical approach. J. Perinat. Med. 2018, 46, 701–715. [Google Scholar] [CrossRef]
- Belfort, M.A.; Whitehead, W.E.; Shamshirsaz, A.A.; Bateni, Z.H.; Olutoye, O.O.; Olutoye, O.A.; Mann, D.G.; Espinoza, J.; Williams, E.; Lee, T.C.; et al. Fetoscopic Open Neural Tube Defect Repair: Development and Refinement of a Two-Port, Carbon Dioxide Insufflation Technique. Obstet. Gynecol. 2017, 129, 734–743. [Google Scholar] [CrossRef]
- Belfort, M.; Deprest, J.; Hecher, K. Current controversies in prenatal diagnosis 1: In utero therapy for spina bifida is ready for endoscopic repair. Prenat. Diagn. 2016, 36, 1161–1166. [Google Scholar] [CrossRef]
- Danzer, E.; Joyeux, L.; Flake, A.W.; Deprest, J. Fetal surgical intervention for myelomeningocele: Lessons learned, outcomes, and future implications. Dev. Med. Child. Neurol. 2020, 62, 417–425. [Google Scholar] [CrossRef]
- Stephenson, J.T.; Pichakron, K.O.; Vu, L.; Jancelewicz, T.; Jamshidi, R.; Grayson, J.K.; Nobuhara, K.K. In utero repair of gastroschisis in the sheep (Ovis aries) model. J. Pediatr. Surg. 2010, 45, 65–69. [Google Scholar] [CrossRef]
- Bergholz, R.; Krebs, T.; Cremieux, B.; Georgi, C.; Fromm, F.; Boettcher, M.; Andreas, T.; Tiemann, B.; Wenke, K.; Reinshagen, K.; et al. Fetoscopic techniques for prenatal covering of gastroschisis in an ovine model are technically demanding and do not lead to permanent anchoring on the fetus until the end of gestation. Surg. Endosc. 2020. [Google Scholar] [CrossRef]
- Bahra, M.; Pratschke, J. Innovations in surgery-How can new technologies be safely implemented in the clinical practice? Chirurg 2020, 91, 553–560. [Google Scholar] [CrossRef] [PubMed]
- Ravitch, M.M. Development of intestinal anastomotic devices. South. Med. J. 1982, 75, 1520–1524. [Google Scholar] [CrossRef] [PubMed]
- Kaidar-Person, O.; Rosenthal, R.J.; Wexner, S.D.; Szomstein, S.; Person, B. Compression anastomosis: History and clinical considerations. Am. J. Surg. 2008, 195, 818–826. [Google Scholar] [CrossRef] [PubMed]
- Graves, C.E.; Co, C.; Hsi, R.S.; Kwiat, D.; Imamura-Ching, J.; Harrison, M.R.; Stoller, M.L. Magnetic Compression Anastomosis (Magnamosis): First-In-Human Trial. J. Am. Coll. Surg. 2017, 225, 676–681.e1. [Google Scholar] [CrossRef] [PubMed]
- Woo, R.; Wong, C.M.; Trimble, Z.; Puapong, D.; Koehler, S.; Miller, S.; Johnson, S. Magnetic Compression Stricturoplasty For Treatment of Refractory Esophageal Strictures in Children: Technique and Lessons Learned. Surg. Innov. 2017, 24, 432–439. [Google Scholar] [CrossRef] [PubMed]
- Dorman, R.M.; Vali, K.; Harmon, C.M.; Zaritzky, M.; Bass, K.D. Repair of esophageal atresia with proximal fistula using endoscopic magnetic compression anastomosis (magnamosis) after staged lengthening. Pediatr. Surg. Int. 2016, 32, 525–528. [Google Scholar] [CrossRef]
- Slater, B.J.; Borobia, P.; Lovvorn, H.N.; Raees, M.A.; Bass, K.D.; Almond, S.; Hoover, J.D.; Kumar, T.; Zaritzky, M. Use of Magnets as a Minimally Invasive Approach for Anastomosis in Esophageal Atresia: Long-Term Outcomes. J. Laparoendosc. Adv. Surg. Tech. A 2019, 29, 1202–1206. [Google Scholar] [CrossRef]
- Zaritzky, M.; Ben, R.; Johnston, K. Magnetic gastrointestinal anastomosis in pediatric patients. J. Pediatr. Surg. 2014, 49, 1131–1137. [Google Scholar] [CrossRef]
- Cook Medical’s FlourishTM Receives Authorization for Pediatric Esophageal Atresia | Newsroom. Available online: https://www.cookmedical.com/newsroom/cook-medicals-flourish-receives-authorization-for-pediatric-esophageal-atresia/ (accessed on 4 November 2020).
- Zaritzky, M.; Ben, R.; Zylberg, G.I.; Yampolsky, B. Magnetic compression anastomosis as a nonsurgical treatment for esophageal atresia. Pediatr. Radiol. 2009, 39, 945–949. [Google Scholar] [CrossRef]
- Wilson-Cook Medical Inc. FlourishTM Pediatric Esophageal Atresia Device-H150003. Food and Drug Administration: Silver Spring, MD, USA, 2019. [Google Scholar]
- Morrow, T. Wilson-Cook’s Flourish Device Uses Magnets To Fix Pediatric Esophageal Atresia. Manag. Care 2017, 26, 32–33. [Google Scholar]
- Criss, C.N.; Jarboe, M.D.; Claflin, J.; Matusko, N.; Rooney, D.M. Evaluating a Solely Mechanical Articulating Laparoscopic Device: A Prospective Randomized Crossover Study. J. Laparoendosc. Adv. Surg. Tech. A 2019, 29, 542–550. [Google Scholar] [CrossRef] [PubMed]
- Criss, C.N.; Ralls, M.W.; Johnson, K.N.; Awtar, S.; Jarboe, M.D.; Geiger, J.D. A Novel Intuitively Controlled Articulating Instrument for Reoperative Foregut Surgery: A Case Report. J. Laparoendosc. Adv. Surg. Tech. A 2017, 27, 983–986. [Google Scholar] [CrossRef] [PubMed]
- FlexDex System—A SAGES Technology and Value Assessment. Available online: https://www.sages.org/publications/tavac/flexdex-system/ (accessed on 6 November 2020).
- Gorgen, A.R.H.; Araldi, M.; de Oliveira Paludo, A.; da Silva, A.G.T.; Ghissi, A.J.; Fernandes, A.S.; Tavares, P.; Rosito, T.E.; Cabral, R.D. Laparoscopic pediatric pyeloplasty using the Flexdex® articulating needle driver: Step-by-step video. J. Pediatr. Urol. 2019, 15, 421–422. [Google Scholar] [CrossRef] [PubMed]
- Frede, T.; Hammady, A.; Klein, J.; Teber, D.; Inaki, N.; Waseda, M.; Buess, G.; Rassweiler, J. The radius surgical system—A new device for complex minimally invasive procedures in urology? Eur. Urol. 2007, 51, 1015–1022. [Google Scholar] [CrossRef]
- Tokas, T.; Gözen, A.S.; Avgeris, M.; Tschada, A.; Fiedler, M.; Klein, J.; Rassweiler, J. Combining of ETHOS Operating Ergonomic Platform, Three-dimensional Laparoscopic Camera, and Radius Surgical System Manipulators Improves Ergonomy in Urologic Laparoscopy: Comparison with Conventional Laparoscopy and da Vinci in a Pelvi Trainer. Eur. Urol. Focus 2017, 3, 413–420. [Google Scholar] [CrossRef]
- LIVSMED LIVSMED. Available online: https://livsmed.com (accessed on 6 November 2020).
- Artisential Articulating Laparoscopic Instrument(s)—A SAGES Technology and Value Assessment. Available online: https://www.sages.org/publications/tavac/artisential-articulating-laparoscopic-instruments/ (accessed on 6 November 2020).
- HandXTM by Human Xtensions—A SAGES Technology and Value Assessment. Available online: https://www.sages.org/publications/tavac/handx-by-human-xtensions/ (accessed on 8 November 2020).
- symphonXTM Surgical Platform—A SAGES Technology and Value Assessment. Available online: https://www.sages.org/publications/tavac/symphonxsurgical-platform/ (accessed on 8 November 2020).
- Datta, R.R.; Dieplinger, G.; Wahba, R.; Kleinert, R.; Thomas, M.; Gebauer, F.; Schiffmann, L.; Stippel, D.L.; Bruns, C.J.; Fuchs, H.F. True single-port cholecystectomy with ICG cholangiography through a single 15-mm trocar using the new surgical platform “symphonX”: First human case study with a commercially available device. Surg. Endosc. 2020, 34, 2722–2729. [Google Scholar] [CrossRef]
- Datta, R.R.; Schönhage, S.; Dratsch, T.; Toader, J.; Müller, D.T.; Wahba, R.; Kleinert, R.; Thomas, M.; Dieplinger, G.; Stippel, D.L.; et al. Learning curve of surgical novices using the single-port platform SymphonX: Minimizing OR trauma to only one 15-mm incision. Surg. Endosc. 2020, 1–14. [Google Scholar] [CrossRef]
- Knight, J.; Tunitsky-Britton, E.; Muffly, T.; Michener, C.M.; Escobar, P.F. Single-port gynecologic surgery with a novel surgical platform. Surg. Innov. 2012, 19, 316–322. [Google Scholar] [CrossRef]
- Leveillee, R.J.; Castle, S.M.; Gorin, M.A.; Salas, N.; Gorbatiy, V. Initial experience with laparoendoscopic single-site simple nephrectomy using the TransEnterix SPIDER surgical system: Assessing feasibility and safety. J. Endourol. 2011, 25, 923–925. [Google Scholar] [CrossRef]
- Pryor, A.D.; Tushar, J.R.; DiBernardo, L.R. Single-port cholecystectomy with the TransEnterix SPIDER: Simple and safe. Surg. Endosc. 2010, 24, 917–923. [Google Scholar] [CrossRef][Green Version]
- Villamizar, N.; Pryor, A.D. SPIDER and flexible laparoscopy: The next frontier in abdominal surgery. Surg. Technol. Int. 2010, 20, 53–58. [Google Scholar] [PubMed]
- Uysal, D.; Gasch, C.; Behnisch, R.; Nickel, F.; Müller-Stich, B.P.; Hohenfellner, M.; Teber, D. Evaluation of new motorized articulating laparoscopic instruments by laparoscopic novices using a standardized laparoscopic skills curriculum. Surg. Endosc. 2020. [Google Scholar] [CrossRef] [PubMed]
- Tuncel, A.; Lucas, S.; Bensalah, K.; Zeltser, I.S.; Jenkins, A.; Saeedi, O.; Park, S.; Cadeddu, J.A. A randomized comparison of conventional vs articulating laparoscopic needle-drivers for performing standardized suturing tasks by laparoscopy-naive subjects. BJU Int. 2008, 101, 727–730. [Google Scholar] [CrossRef] [PubMed]
- Sieber, M.A.; Fellmann-Fischer, B.; Mueller, M. Performance of Kymerax© precision-drive articulating surgical system compared to conventional laparoscopic instruments in a pelvitrainer model. Surg. Endosc. 2017, 31, 4298–4308. [Google Scholar] [CrossRef]
- Sánchez-Margallo, F.M.; Sánchez-Margallo, J.A. Assessment of Postural Ergonomics and Surgical Performance in Laparoendoscopic Single-Site Surgery Using a Handheld Robotic Device. Surg. Innov. 2018, 25, 208–217. [Google Scholar] [CrossRef]
- Madec, F.-X.; Dariane, C.; Cornu, J.-N. Evaluation and comparison of basic gestures in ex vivo laparoscopic surgery using a robotic instrument and traditional laparoscopic instruments. Prog. Urol. 2020, 30, 58–63. [Google Scholar] [CrossRef]
- Varca, V.; Benelli, A.; Pietrantuono, F.; Suardi, N.; Gregori, A.; Gaboardi, F. A robotic needle driver to facilitate vescico-urethral anastomosis during laparoscopic radical prostatectomy. Urologia 2017, 84, 259–262. [Google Scholar] [CrossRef]
- van Mulken, T.J.M.; Schols, R.M.; Scharmga, A.M.J.; Winkens, B.; Cau, R.; Schoenmakers, F.B.F.; Qiu, S.S.; van der Hulst, R.R.W.J. MicroSurgical Robot Research Group First-in-human robotic supermicrosurgery using a dedicated microsurgical robot for treating breast cancer-related lymphedema: A randomized pilot trial. Nat. Commun. 2020, 11, 757. [Google Scholar] [CrossRef]
- MUSA. Available online: http://microsure.nl/musa/ (accessed on 8 November 2020).
- van Mulken, T.J.M.; Boymans, C.A.E.M.; Schols, R.M.; Cau, R.; Schoenmakers, F.B.F.; Hoekstra, L.T.; Qiu, S.S.; Selber, J.C.; van der Hulst, R.R.W.J. Preclinical Experience Using a New Robotic System Created for Microsurgery. Plast. Reconstr. Surg. 2018, 142, 1367–1376. [Google Scholar] [CrossRef]
- Distalmotion › DistalMotion. Available online: https://www.distalmotion.com/ (accessed on 8 November 2020).
- About | Verb Surgical. Available online: https://www.verbsurgical.com/about/ (accessed on 8 November 2020).
- avatera-System—Avateramedical. Available online: https://www.avatera.eu/avatera-system (accessed on 8 November 2020).
- Dixon, F.; O’Hara, R.; Ghuman, N.; Strachan, J.; Khanna, A.; Keeler, B.D. Major colorectal resection is feasible using a new robotic surgical platform: The first report of a case series. Tech. Coloproctol. 2020, 1–5. [Google Scholar] [CrossRef]
- Kelkar, D.; Borse, M.A.; Godbole, G.P.; Kurlekar, U.; Slack, M. Interim safety analysis of the first-in-human clinical trial of the Versius surgical system, a new robot-assisted device for use in minimal access surgery. Surg. Endosc. 2020, 1–10. [Google Scholar] [CrossRef]
- Morton, J.; Hardwick, R.H.; Tilney, H.S.; Gudgeon, A.M.; Jah, A.; Stevens, L.; Marecik, S.; Slack, M. Preclinical evaluation of the versius surgical system, a new robot-assisted surgical device for use in minimal access general and colorectal procedures. Surg. Endosc. 2020. [Google Scholar] [CrossRef]
- Puntambekar, S.P.; Goel, A.; Chandak, S.; Chitale, M.; Hivre, M.; Chahal, H.; Rajesh, K.N.; Manerikar, K. Feasibility of robotic radical hysterectomy (RRH) with a new robotic system. Experience at Galaxy Care Laparoscopy Institute. J. Robot. Surg. 2020. [Google Scholar] [CrossRef] [PubMed]
- Thomas, B.C.; Slack, M.; Hussain, M.; Barber, N.; Pradhan, A.; Dinneen, E.; Stewart, G.D. Preclinical Evaluation of the Versius Surgical System, a New Robot-assisted Surgical Device for Use in Minimal Access Renal and Prostate Surgery. Eur. Urol. Focus 2020. [Google Scholar] [CrossRef] [PubMed]
- Versius For Surgeons—CMR Surgical. Available online: https://cmrsurgical.com/versius/surgeon (accessed on 8 November 2020).
- Forgione, A. In vivo microrobots for natural orifice transluminal surgery. Current status and future perspectives. Surg. Oncol. 2009, 18, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Shah, B.C.; Buettner, S.L.; Lehman, A.C.; Farritor, S.M.; Oleynikov, D. Miniature in vivo robotics and novel robotic surgical platforms. Urol. Clin. N. Am. 2009, 36, 251–263. [Google Scholar] [CrossRef] [PubMed]
- Aoki, M.; Tokioka, S.; Narabayashi, K.; Hakoda, A.; Inoue, Y.; Yorifuji, N.; Chino, Y.; Sato, I.; Egashira, Y.; Takeuchi, T.; et al. Laparoscopic and endoscopic cooperative surgery for intra-mucosal gastric carcinoma adjacent to the ulcer scars. World J. Surg. Oncol. 2018, 16, 53. [Google Scholar] [CrossRef]
- Matsuda, T.; Nunobe, S.; Ohashi, M.; Hiki, N. Laparoscopic endoscopic cooperative surgery (LECS) for the upper gastrointestinal tract. Transl. Gastroenterol. Hepatol. 2017, 2, 40. [Google Scholar] [CrossRef]
- Matsumoto, S.; Hosoya, Y.; Lefor, A.K.; Ino, Y.; Haruta, H.; Kurashina, K.; Saito, S.; Kitayama, J.; Sata, N. Non-exposed endoscopic wall-inversion surgery for pediatric gastrointestinal stromal tumor: A case report. Asian J. Endosc. Surg. 2019, 12, 322–325. [Google Scholar] [CrossRef]
- Peña, A.; Devries, P.A. Posterior sagittal anorectoplasty: Important technical considerations and new applications. J. Pediatr. Surg. 1982, 17, 796–811. [Google Scholar] [CrossRef]
- ENDOSCOPIC-ASSISTED PERCUTANEOUS ANO-RECTO-PLASTY (EPARP). Available online: https://www.ipeg.org/videos/endoscopic-assisted-percutaneous-ano-recto-plasty-eparp/ (accessed on 12 November 2020).
- Atallah, S.; Hodges, A.; Larach, S.W. Direct target NOTES: Prospective applications for next generation robotic platforms. Tech. Coloproctol. 2018, 22, 363–371. [Google Scholar] [CrossRef]
- Seeliger, B.; Diana, M.; Ruurda, J.P.; Konstantinidis, K.M.; Marescaux, J.; Swanström, L.L. Enabling single-site laparoscopy: The SPORT platform. Surg. Endosc. 2019, 33, 3696–3703. [Google Scholar] [CrossRef] [PubMed]
- Misal, M.; Magtibay, P.M.; Yi, J. Robotic LESS and Reduced-Port Hysterectomy Using the da Vinci SP Surgical System: A Single-Institution Case Series. J. Minim. Invasive Gynecol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Khamzina, M.; Parab, K.V.; An, R.; Bullard, T.; Grigsby-Toussaint, D.S. Impact of Pokémon Go on Physical Activity: A Systematic Review and Meta-Analysis. Am. J. Prev. Med. 2020, 58, 270–282. [Google Scholar] [CrossRef] [PubMed]
- Parekh, P.; Patel, S.; Patel, N.; Shah, M. Systematic review and meta-analysis of augmented reality in medicine, retail, and games. Vis. Comput. Ind. Biomed. Art. 2020, 3, 21. [Google Scholar] [CrossRef] [PubMed]
- Kang, X.; Azizian, M.; Wilson, E.; Wu, K.; Martin, A.D.; Kane, T.D.; Peters, C.A.; Cleary, K.; Shekhar, R. Stereoscopic augmented reality for laparoscopic surgery. Surg. Endosc. 2014, 28, 2227–2235. [Google Scholar] [CrossRef]
- Liu, X.; Kang, S.; Plishker, W.; Zaki, G.; Kane, T.D.; Shekhar, R. Laparoscopic stereoscopic augmented reality: Toward a clinically viable electromagnetic tracking solution. J. Med. Imaging 2016, 3, 045001. [Google Scholar] [CrossRef]
- Cercenelli, L.; Carbone, M.; Condino, S.; Cutolo, F.; Marcelli, E.; Tarsitano, A.; Marchetti, C.; Ferrari, V.; Badiali, G. The Wearable VOSTARS System for Augmented Reality-Guided Surgery: Preclinical Phantom Evaluation for High-Precision Maxillofacial Tasks. J. Clin. Med. 2020, 9, 3562. [Google Scholar] [CrossRef]
- Usuda, H.; Watanabe, S.; Miura, Y.; Saito, M.; Musk, G.C.; Rittenschober-Böhm, J.; Ikeda, H.; Sato, S.; Hanita, T.; Matsuda, T.; et al. Successful maintenance of key physiological parameters in preterm lambs treated with ex vivo uterine environment therapy for a period of 1 week. Am. J. Obstet. Gynecol. 2017, 217, 457.e1–457.e13. [Google Scholar] [CrossRef]
- Ignacio, R.C.; Burke, R.; Spencer, D.; Bissell, C.; Dorsainvil, C.; Lucha, P.A. Laparoscopic versus open appendectomy: What is the real difference? Results of a prospective randomized double-blinded trial. Surg. Endosc. 2004, 18, 334–337. [Google Scholar] [CrossRef]
- Katkhouda, N.; Mason, R.J.; Towfigh, S. Laparoscopic versus open appendectomy: A prospective, randomized, double-blind study. Adv. Surg. 2006, 40, 1–19. [Google Scholar] [CrossRef][Green Version]
- Clarke, T.; Katkhouda, N.; Mason, R.J.; Cheng, B.C.; Olasky, J.; Sohn, H.J.; Moazzez, A.; Algra, J.; Chaghouri, E.; Berne, T.V. Laparoscopic versus open appendectomy for the obese patient: A subset analysis from a prospective, randomized, double-blind study. Surg. Endosc. 2011, 25, 1276–1280. [Google Scholar] [CrossRef] [PubMed]
- Ricca, R.; Schneider, J.J.; Brar, H.; Lucha, P.A. Laparoscopic appendectomy in patients with a body mass index of 25 or greater: Results of a double blind, prospective, randomized trial. JSLS 2007, 11, 54–58. [Google Scholar] [PubMed]
- Poprom, N.; Wilasrusmee, C.; Attia, J.; McEvoy, M.; Thakkinstian, A.; Rattanasiri, S. Comparison of postoperative complications between open and laparoscopic appendectomy: An umbrella review of systematic reviews and meta-analyses. J. Trauma Acute. Care Surg. 2020, 89, 813–820. [Google Scholar] [CrossRef]
- Dai, L.; Shuai, J. Laparoscopic versus open appendectomy in adults and children: A meta-analysis of randomized controlled trials. United Europ. Gastroenterol. J. 2017, 5, 542–553. [Google Scholar] [CrossRef] [PubMed]
- Lintula, H.; Kokki, H.; Vanamo, K.; Antila, P.; Eskelinen, M. Laparoscopy in children with complicated appendicitis. J. Pediatr. Surg. 2002, 37, 1317–1320. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lejus, C.; Delile, L.; Plattner, V.; Baron, M.; Guillou, S.; Héloury, Y.; Souron, R. Randomized, single-blinded trial of laparoscopic versus open appendectomy in children: Effects on postoperative analgesia. Anesthesiology 1996, 84, 801–806. [Google Scholar] [CrossRef] [PubMed]
- Hall, N.J.; Pacilli, M.; Eaton, S.; Reblock, K.; Gaines, B.A.; Pastor, A.; Langer, J.C.; Koivusalo, A.I.; Pakarinen, M.P.; Stroedter, L.; et al. Recovery after open versus laparoscopic pyloromyotomy for pyloric stenosis: A double-blind multicentre randomised controlled trial. Lancet 2009, 373, 390–398. [Google Scholar] [CrossRef]
- Leclair, M.-D.; Plattner, V.; Mirallie, E.; Lejus, C.; Nguyen, J.-M.; Podevin, G.; Heloury, Y. Laparoscopic pyloromyotomy for hypertrophic pyloric stenosis: A prospective, randomized controlled trial. J. Pediatr. Surg. 2007, 42, 692–698. [Google Scholar] [CrossRef]
- Sathya, C.; Wayne, C.; Gotsch, A.; Vincent, J.; Sullivan, K.J.; Nasr, A. Laparoscopic versus open pyloromyotomy in infants: A systematic review and meta-analysis. Pediatr. Surg. Int. 2017, 33, 325–333. [Google Scholar] [CrossRef]
- Ramirez, P.T.; Frumovitz, M.; Pareja, R.; Lopez, A.; Vieira, M.; Ribeiro, R.; Buda, A.; Yan, X.; Shuzhong, Y.; Chetty, N.; et al. Minimally Invasive versus Abdominal Radical Hysterectomy for Cervical Cancer. N. Engl. J. Med. 2018, 379, 1895–1904. [Google Scholar] [CrossRef]
- Melamed, A.; Margul, D.J.; Chen, L.; Keating, N.L.; Del Carmen, M.G.; Yang, J.; Seagle, B.-L.L.; Alexander, A.; Barber, E.L.; Rice, L.W.; et al. Survival after Minimally Invasive Radical Hysterectomy for Early-Stage Cervical Cancer. N. Engl. J. Med. 2018, 379, 1905–1914. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meinzer, A.; Alkatout, I.; Krebs, T.F.; Baastrup, J.; Reischig, K.; Meiksans, R.; Bergholz, R. Advances and Trends in Pediatric Minimally Invasive Surgery. J. Clin. Med. 2020, 9, 3999. https://doi.org/10.3390/jcm9123999
Meinzer A, Alkatout I, Krebs TF, Baastrup J, Reischig K, Meiksans R, Bergholz R. Advances and Trends in Pediatric Minimally Invasive Surgery. Journal of Clinical Medicine. 2020; 9(12):3999. https://doi.org/10.3390/jcm9123999
Chicago/Turabian StyleMeinzer, Andreas, Ibrahim Alkatout, Thomas Franz Krebs, Jonas Baastrup, Katja Reischig, Roberts Meiksans, and Robert Bergholz. 2020. "Advances and Trends in Pediatric Minimally Invasive Surgery" Journal of Clinical Medicine 9, no. 12: 3999. https://doi.org/10.3390/jcm9123999
APA StyleMeinzer, A., Alkatout, I., Krebs, T. F., Baastrup, J., Reischig, K., Meiksans, R., & Bergholz, R. (2020). Advances and Trends in Pediatric Minimally Invasive Surgery. Journal of Clinical Medicine, 9(12), 3999. https://doi.org/10.3390/jcm9123999






