Elevated Procalcitonin as a Risk Factor for Postoperative Delirium in the Elderly after Cardiac Surgery—A Prospective Observational Study
Abstract
1. Introduction
2. Experimental Section
2.1. Procalcitonin Measurement
2.2. Preoperative Assessment
2.3. Anesthesia and Surgery
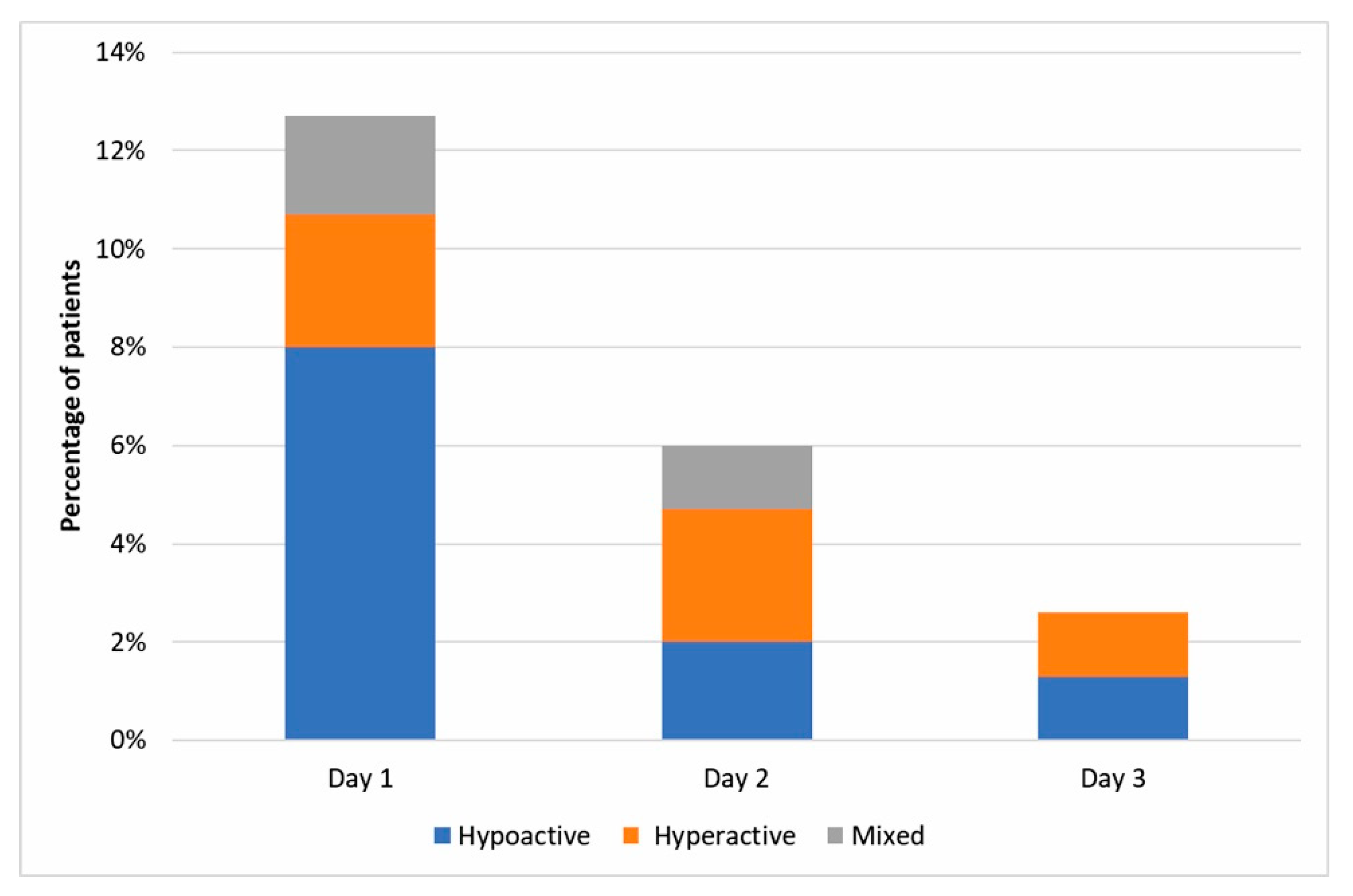
2.4. Delirium Assessment
2.5. Statistical Analysis
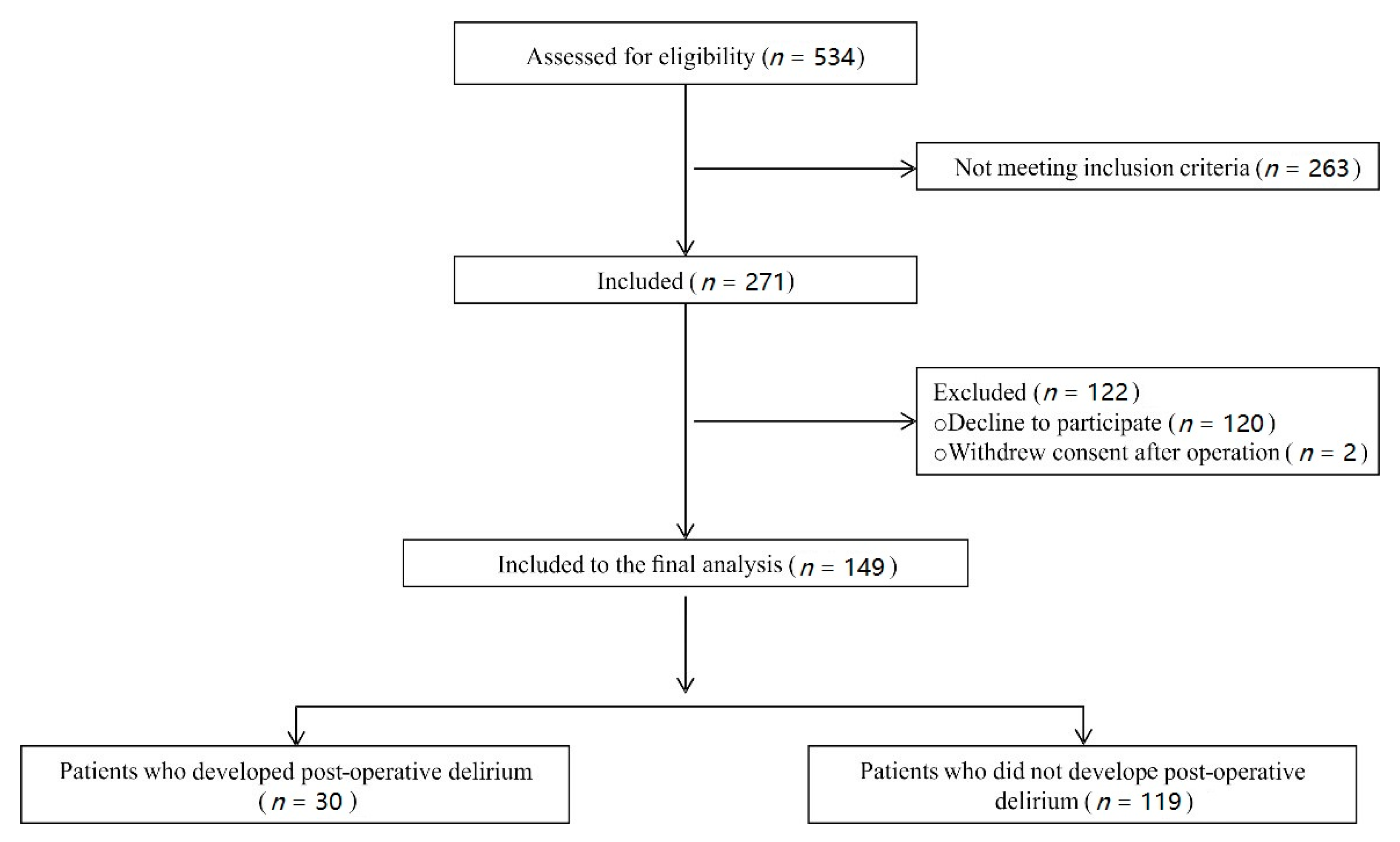
3. Results
3.1. Study Sample
3.2. Risk Factors for Delirium
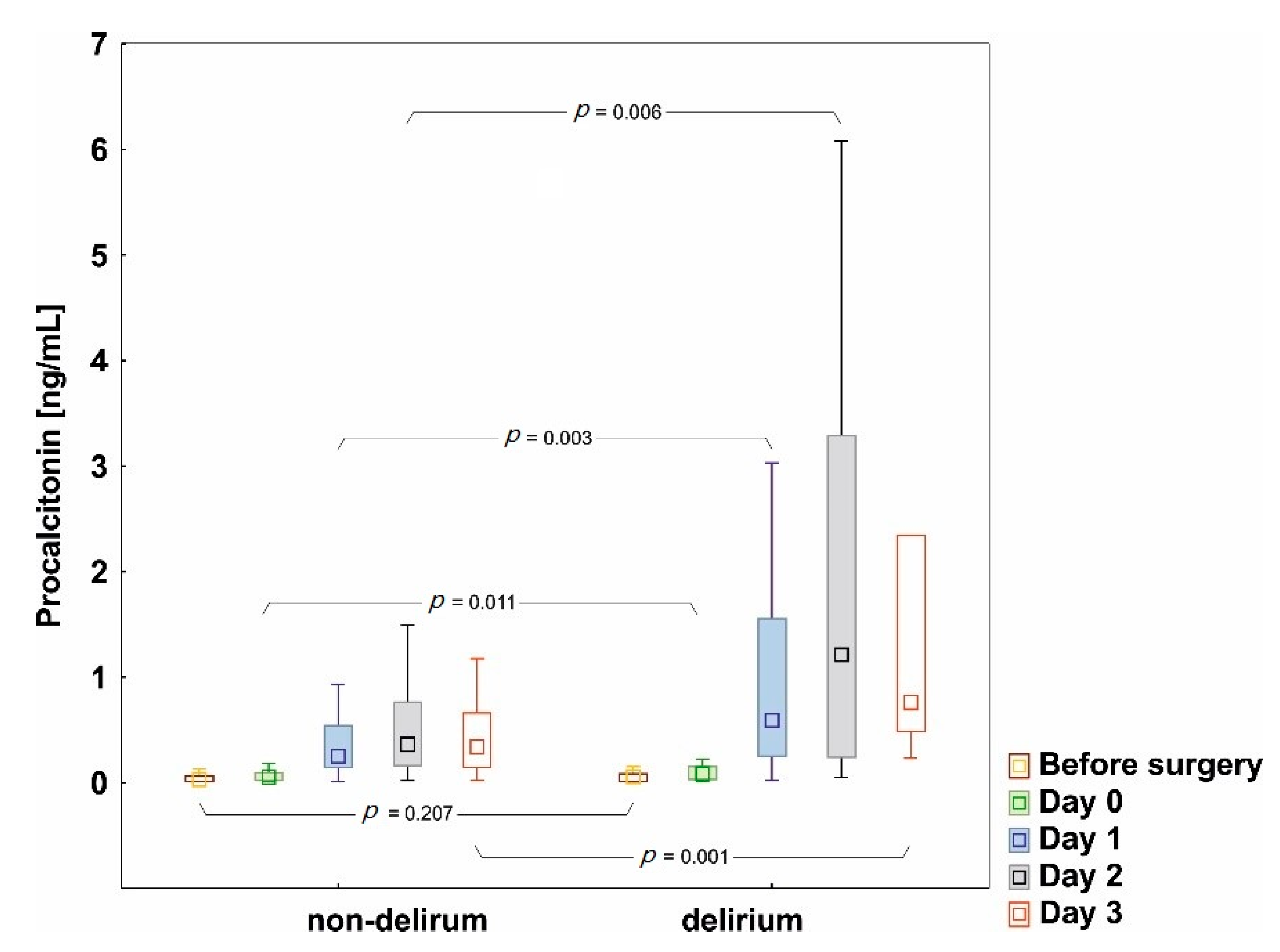
3.2.1. Procalcitonin
3.2.2. Age
3.2.3. Comorbidities
3.2.4. Euroscore II
3.2.5. Preoperative Geriatric Assessment Scores
3.2.6. Surgery and Postoperative Indices
3.3. Prediction Model of Postoperative Delirium
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Benjamin, E.J.; Virani, S.S.; Callaway, C.W.; Chamberlain, A.M.; Chang, A.R.; Cheng, S.; de Ferranti, S.D.; Floyd, J.; Fornage, M.; Gillespie, C.; et al. Heart Disease and Stroke Statistics—2017 Update. Circulation 2017, 135, e146–e603. [Google Scholar] [CrossRef]
- Iung, B.; Baron, G.; Butchart, E.G.; Delahaye, F.; Gohlke-Bärwolf, C.; Levang, O.W.; Tornos, P.; Vanoverschelde, J.-L.; Vermeer, F.; Boersma, E.; et al. A prospective survey of patients with valvular heart disease in Europe: The Euro Heart Survey on valvular heart disease. Eur. Heart J. 2003, 24, 1231–1243. [Google Scholar] [CrossRef]
- Ranucci, M.; Castelvecchio, S.; Menicanti, L.; Frigiola, A.; Pelissero, G. Risk of assessing mortality risk in elective cardiac operations: Age, creatinine, ejection fraction, and the law of parsimony. Circulation 2009, 119, 3053–3061. [Google Scholar] [CrossRef]
- Kupiec, A.; Adamik, B.; Forkasiewicz-Gardynik, K.; Goździk, W. Intra-operative hyperoxia and the risk of delirium in elderly patients after cardiac surgery. Aging 2020, 12, 7006–7014. [Google Scholar] [CrossRef]
- Lin, Y.; Chen, J.; Wang, Z. Meta-analysis of factors which influence delirium following cardiac surgery. J. Card. Surg. 2012, 27, 481–492. [Google Scholar] [CrossRef]
- Bakker, R.C.; Osse, R.J.; Tulen, J.H.M.; Kappetein, A.P.; Bogers, A.J.J.C. Preoperative and operative predictors of delirium after cardiac surgery in elderly patients. Eur. J. Cardio Thorac. Surg. 2012, 41, 544–549. [Google Scholar] [CrossRef]
- Koster, S.; Hensens, A.G.; Schuurmans, M.J.; Van Der Palen, J. Consequences of delirium after cardiac operations. Ann. Thorac. Surg. 2012, 93, 705–711. [Google Scholar] [CrossRef]
- Sauër, A.C.; Veldhuijzen, D.S.; Ottens, T.H.; Slooter, A.J.C.; Kalkman, C.J.; Van Dijk, D. Association between delirium and cognitive change after cardiac surgery. Br. J. Anaesth. 2017, 119, 308–315. [Google Scholar] [CrossRef]
- Maldonado, J.R. Delirium pathophysiology: An updated hypothesis of the etiology of acute brain failure. Int. J. Geriatr. Psychiatry 2018, 33, 1428–1457. [Google Scholar] [CrossRef]
- Paparella, D.; Yau, T.M.; Young, E. Cardiopulmonary bypass induced inflammation: Pathophysiology and treatment. An update. Eur. J. Cardio Thorac. Surg. 2002, 21, 232–244. [Google Scholar] [CrossRef]
- Gozdzik, W.; Adamik, B.; Gozdzik, A.; Rachwalik, M.; Kustrzycki, W.; Kübler, A. Unchanged plasma levels of the soluble urokinase plasminogen activator receptor in elective coronary artery bypass graft surgery patients and cardiopulmonary bypass use. PLoS ONE 2014, 9, e98923. [Google Scholar] [CrossRef]
- Li, Q.; Zheng, S.; Zhou, P.Y.; Xiao, Z.; Wang, R.; Li, J. The diagnostic accuracy of procalcitonin in infectious patients after cardiac surgery: A systematic review and meta-analysis. J. Cardiovasc. Med. 2020. [Google Scholar] [CrossRef]
- Brocca, A.; Virzì, G.M.; de Cal, M.; Giavarina, D.; Carta, M.; Ronco, C. Elevated Levels of Procalcitonin and Interleukin-6 are Linked with Postoperative Complications in Cardiac Surgery. Scand. J. Surg. 2017, 106, 318–324. [Google Scholar] [CrossRef]
- Clementi, A.; Virzì, G.M.; Muciño-Bermejo, M.J.; Nalesso, F.; Giavarina, D.; Carta, M.; Brocca, A.; de Cal, M.; Danesi, T.H.; Zanella, M.; et al. Presepsin and Procalcitonin Levels as Markers of Adverse Postoperative Complications and Mortality in Cardiac Surgery Patients. Blood Purif. 2019, 47, 140–148. [Google Scholar] [CrossRef]
- Clementi, A.; Brocca, A.; Virzì, G.M.; De Cal, M.; Giavarina, D.; Carta, M.; Muciño-Bermejo, M.-J.; Danesi, T.H.; Salvador, L.; Ronco, C. Procalcitonin and Interleukin-6 Levels: Are They Useful Biomarkers in Cardiac Surgery Patients? Blood Purif. 2017, 43, 290–297. [Google Scholar] [CrossRef]
- Partridge, J.S.L.; Harari, D.; Martin, F.C.; Dhesi, J.K. The impact of pre-operative comprehensive geriatric assessment on postoperative outcomes in older patients undergoing scheduled surgery: A systematic review. Anaesthesia 2014, 69, 8–16. [Google Scholar] [CrossRef]
- Katz, S. Assessing self-maintenance: Activities of daily living, mobility, and instrumental activities of daily living. J. Am. Geriatr. Soc. 1983, 31, 721–727. [Google Scholar] [CrossRef]
- Vittengl, J.R.; White, C.N.; McGovern, R.J.; Morton, B.J. Comparative validity of seven scoring systems for the instrumental activities of daily living scale in rural elders. Aging Ment. Health 2006, 10, 40–47. [Google Scholar] [CrossRef]
- Foley, T.; McKinlay, A.; Warren, N.; Stolwyk, R.J. Assessing the sensitivity and specificity of cognitive screening measures for people with Parkinson’s disease. NeuroRehabilitation 2019, 43, 491–500. [Google Scholar] [CrossRef]
- Wancata, J.; Alexandrowicz, R.; Marquart, B.; Weiss, M.; Friedrich, F. The criterion validity of the geriatric depression scale: A systematic review. Acta Psychiatr. Scand. 2006, 114, 398–410. [Google Scholar] [CrossRef]
- WHO. Haemoglobin Concentrations for the Diagnosis of Anaemia and Assessment of Severity. Vitamin and Mineral Nutrition Information System; World Health Organization: Geneva, Switzerland, 2011; pp. 1–6. Available online: http://scholar.google.com/scholar?hl=en&btnG=Search&q=intitle:Haemoglobin+concentrations+for+the+diagnosis+of+anaemia+and+assessment+of+severity#1 (accessed on 26 November 2020).
- Kellum, J.A.; Lameire, N.; Aspelin, P.; Barsoum, R.S.; Burdmann, E.A.; Goldstein, S.L.; Herzog, C.A.; Joannidis, M.; Kribben, A.; Levey, A.S.; et al. Kidney disease: Improving global outcomes (KDIGO) acute kidney injury work group. KDIGO clinical practice guideline for acute kidney injury. Kidney Int. Suppl. 2012, 2, 1–138. [Google Scholar]
- Chanques, G.; Ely, E.W.; Garnier, O.; Perrigault, F.; Eloi, A.; Carr, J.; Rowan, C.M.; Prades, A.; de Jong, A.; Moritz-Gasser, S.; et al. The 2014 updated version of the Confusion Assessment Method for the Intensive Care Unit compared to the 5th version of the Diagnostic and Statistical Manual of Mental Disorders and other current methods used by intensivists. Ann. Intensive Care. 2018, 8, 33. [Google Scholar] [CrossRef] [PubMed]
- Keranov, S.; Kim, W.K.; Arsalan, M.; Renker, M.; Keller, T.; Bauer, T.; Dörr, O.; Nef, H.M.; Gaede, L.; Möllmann, H.; et al. Predictive value of preprocedural procalcitonin for short- and long-term mortality after transfemoral transcatheter aortic valve implantation. Heart Vessels 2019, 34, 1993–2001. [Google Scholar] [CrossRef] [PubMed]
- Cereghetti, C.; Siegemund, M.; Schaedelin, S.; Fassl, J.; Seeberger, M.D.; Eckstein, F.S.; Steiner, L.A.; Goettel, N. Independent Predictors of the Duration and Overall Burden of Postoperative Delirium After Cardiac Surgery in Adults: An Observational Cohort Study. J. Cardiothorac. Vasc. Anesth. 2017, 31, 1966–1973. [Google Scholar] [CrossRef]
- Eshmawey, M.; Arlt, S.; Ledschbor-Frahnert, C.; Guenther, U.; Popp, J. Preoperative Depression and Plasma Cortisol Levels as Predictors of Delirium after Cardiac Surgery. Dement Geriatr. Cogn. Disord. 2020, 48, 207–214. [Google Scholar] [CrossRef]
- Oldham, M.A.; Hawkins, K.; Lin, I.; Deng, Y.; Hao, Q.; Scoutt, L.M.; Yuh, D.D.; Lee, H.B. Depression predicts delirium after coronary artery bypass graft surgery independent of cognitive impairment and cerebrovascular disease: An analysis of the NOAHS study. HHS Public Access. 2020, 27, 476–486. [Google Scholar]
- Scheel-Hincke, L.L.; Möller, S.; Lindahl-Jacobsen, R.; Jeune, B.; Ahrenfeldt, L.J. Cross-national comparison of sex differences in ADL and IADL in Europe: Findings from SHARE. Eur. J. Ageing 2020, 17, 69–79. [Google Scholar] [CrossRef]
- Bleijenberg, N.; Zuithoff, N.P.A.; Smith, A.K.; de Wit, N.J.; Schuurmans, M.J. Disability in the individual ADL, IADL, and mobility among older adults: A prospective cohort study. J. Nutr. Health Aging 2017, 21, 897–903. [Google Scholar] [CrossRef]
- Sponholz, C.; Sakr, Y.; Reinhart, K.; Brunkhorst, F. Diagnostic value and prognostic implications of serum procalcitonin after cardiac surgery: A systematic review of the literature. Crit. Care 2006, 10, R145. [Google Scholar] [CrossRef]
- Cascella, M.; Muzio, M.R.; Bimonte, S.; Cuomo, A.; Jakobsson, J.G. Postoperative delirium and postoperative cognitive dysfunction: Updates in pathophysiology, potential translational approaches to clinical practice and further research perspectives. Minerva Anestesiol. 2018, 84, 246–260. [Google Scholar]
- Klingele, M.; Bomberg, H.; Schuster, S.; Schäfers, H.J.; Groesdonk, H.V. Prognostic value of procalcitonin in patients after elective cardiac surgery: A prospective cohort study. Ann. Intensive Care 2016, 6, 116. [Google Scholar] [CrossRef] [PubMed]
- McGrane, S.; Girard, T.D.; Thompson, J.L.; Shintani, A.K.; Woodworth, A.; Ely, E.W.; Pandharipande, P.P. Procalcitonin and C-reactive protein levels at admission as predictors of duration of acute brain dysfunction in critically ill patients. Crit. Care 2011, 15, R78. Available online: http://ccforum.com/content/15/2/R78 (accessed on 26 November 2020). [CrossRef] [PubMed]
- Nemeth, E.; Vig, K.; Racz, K.; Koritsanszky, K.B.; Ronkay, K.I.; Hamvas, F.P.; Borbély, C.; Eory, A.; Merkely, B.; Gal, J. Influence of the postoperative inflammatory response on cognitive decline in elderly patients undergoing on-pump cardiac surgery: A controlled, prospective observational study. BMC Anesthesiol. 2017, 17, 113. [Google Scholar] [CrossRef] [PubMed]
- Guenther, U.; Theuerkauf, N.; Frommann, I.; Brimmers, K.; Malik, R.; Stori, S.; Scheidemann, M.; Putensen, C.; Popp, J. Predisposing and precipitating factors of delirium after cardiac surgery: A prospective observational cohort study. Ann. Surg. 2013, 257, 1160–1167. [Google Scholar] [CrossRef]
- Fowler, A.J.; Ahmad, T.; Abbott, T.E.F.; Torrance, H.D.; Wouters, P.F.; De Hert, S.; Lobo, S.M.; Rasmussen, L.S.; della Rocca, G.; Beattie, W.S.; et al. Association of preoperative anaemia with postoperative morbidity and mortality: An observational cohort study in low-, middle-, and high-income countries. Br. J. Anaesth. 2018, 121, 1227–1235. [Google Scholar] [CrossRef]
- Kunz, J.V.; Spies, C.D.; Bichmann, A.; Sieg, M.; Mueller, A. Postoperative anaemia might be a risk factor for postoperative delirium and prolonged hospital stay: A secondary analysis of a prospective cohort study. PLoS ONE 2020, 15, e0229325. [Google Scholar] [CrossRef]
- Gosselt, A.N.C.; Slooter, A.J.C.; Boere, P.R.Q.; Zaal, I.J. Risk factors for delirium after on-pump cardiac surgery: A systematic review. Crit. Care 2015, 19, 1–8. [Google Scholar] [CrossRef]
- Smulter, N.; Engström, K.G.; Appelblad, M.; Gustafson, Y.; Lingehall, H.C.; Svenmarker, S.; Svenmarker, S. Disturbances in Oxygen Balance During Cardiopulmonary Bypass: A Risk Factor for Postoperative Delirium. J. Cardiothorac. Vasc. Anesth. 2017, 32, 684–690. [Google Scholar] [CrossRef]
| Preoperative Characteristics | Delirium | Non-Delirium | p |
|---|---|---|---|
| n = 30 | n = 119 | ||
| Age | 73.5 (70–76) | 69 (67–74) | 0.038 |
| Gender, men, n (%) | 18 (60) | 79 (66) | 0.511 |
| BMI, (kg/m2) | 28.4 (24.2–29.4) | 27.5 (25.3–31.0) | 0.597 |
| Euroscore II, (%) | 2.5 (1.6–5.7) | 1.8 (1.2–2.8) | 0.009 |
| Comorbidities, n (%) | |||
| Cerebrovascular disease | 7 (23) | 13 (10) | 0.138 |
| Arterial hypertension | 26 (86) | 107 (89) | 0.607 |
| Diabetes mellitus | 11 (36) | 44 (36) | 0.975 |
| Continuous atrial fibrillation | 2 (6) | 11 (9) | 0.654 |
| Chronic renal insufficiency | 8 (26) | 20 (16) | 0.216 |
| Anemia, n (%) | 13(43) | 23 (19) | 0.006 |
| Ejection fraction, n (%) | 60 (50–65) | 60 (50–65) | 0.471 |
| Nicotine abuse, n (%) | 6 (20) | 26 (22) | 0.825 |
| Delirium | Non-Delirium | p | |
|---|---|---|---|
| n = 30 | n = 119 | ||
| MMSE, n (%) | 0.123 | ||
| no cognitive impairment (≥27 pts.) | 8 (27) | 50 (42) | |
| cognitive impairment (≤26 pts.) | 22 (73) | 69 (58) | |
| ADL, n (%) | 0.103 | ||
| full functioning (6 pts.), n (%) | 28 (93) | 118 (99) | |
| compromised functioning (<6 pts.), n (%) | 2 (7) | 1(1) | |
| IADL, n (%) | 0.046 | ||
| full functioning (24 pts) | 16 (53) | 86 (72) | |
| compromised functioning (total score < 24 pts.): | |||
| (1) use phone | 2 (7) | 3 (3) | 0.471 |
| (2) shopping | 5 (16) | 6(5) | 0.041 |
| (3) housekeeping | 9 (30) | 4 (4) | <0.001 |
| (4) handle finances | 7 (23) | 23 (19) | 0.235 |
| (5) a mode of transportation | 10 (33) | 15(12) | 0.014 |
| (6) responsibility for own medication | 5 (16) | 5 (5) | 0.035 |
| (7) preparation of food | 5 (16) | 4 (3) | 0.022 |
| (8) doing laundry | 7 (23) | 5 (5) | 0.001 |
| GDS-15, n (%) | 0.005 | ||
| no depression (<5 pts.) | 18 (60) | 99 (83) | |
| depressive symptoms (≥5 pts.) | 12 (40) | 20 (17) |
| Delirium | Non-Delirium | p | |
|---|---|---|---|
| n = 30 | n = 119 | ||
| Intra-operative characteristics | |||
| Type of operation, n (%) | 0.009 | ||
| CABG | 16 (53) | 84 (71) | |
| Valve only | 2 (7) | 18 (15) | |
| CABG + valve | 12 (40) | 17 (14) | |
| CBP time (min) | 102 (81–127) | 79 (67–94) | <0.001 |
| AoX time (min) | 57 (43–81) | 43 (34–60) | 0.004 |
| Anesthesia time (min) | 272 (240–305) | 255 (235–280) | 0.101 |
| Post-operative characteristics | |||
| Respiratory support (minutes) | 290 (210–525) | 200 (175–270) | 0.002 |
| AKI, n (%) | 6 (20) | 8 (7) | 0.025 |
| ICU stay (days) | 3 (2–6) | 3 (2–5) | 0.133 |
| Hospital stay (days) | 11 (10–15) | 9 (8–11) | 0.002 |
| In-hospital mortality, n (%) | 1 (3.3) | 1 (0.8) | 0.323 |
| Univariate Analysis | Multivariate Analysis | |||||
|---|---|---|---|---|---|---|
| Odds Ratio | 95% CI | p | Odds Ratio | 95% CI | p | |
| PCT baseline | 2.70 | 1.15–6.32 | 0.022 | 3.05 | 1.01–9.19 | 0.048 |
| IADL | 0.81 | 0.70–0.92 | 0.002 | 0.76 | 0.63–0.91 | 0.003 |
| Depressive symptoms | 3.30 | 1.37–7.91 | 0.008 | 5.02 | 1.67–15.10 | 0.004 |
| Age | 1.08 | 1.01–1.17 | 0.031 | 1.14 | 1.02–1.26 | 0.016 |
| CPB time | 1.02 | 1.01–1.04 | <0.001 | 1.04 | 1.02–1.06 | <0.001 |
| AoX time | 1.02 | 1.01–1.04 | 0.003 | |||
| Euroscore | 1.21 | 1.06–1.37 | 0.004 | |||
| Gender | 0.75 | 0.33–1.73 | 0.511 | |||
| MMSE | 0.96 | 0.84–1.09 | 0.595 | |||
| ADL | 0.34 | 0.10–1.16 | 0.086 | |||
| Anemia before surgery | 3.19 | 1.35–7.49 | 0.007 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kupiec, A.; Adamik, B.; Kozera, N.; Gozdzik, W. Elevated Procalcitonin as a Risk Factor for Postoperative Delirium in the Elderly after Cardiac Surgery—A Prospective Observational Study. J. Clin. Med. 2020, 9, 3837. https://doi.org/10.3390/jcm9123837
Kupiec A, Adamik B, Kozera N, Gozdzik W. Elevated Procalcitonin as a Risk Factor for Postoperative Delirium in the Elderly after Cardiac Surgery—A Prospective Observational Study. Journal of Clinical Medicine. 2020; 9(12):3837. https://doi.org/10.3390/jcm9123837
Chicago/Turabian StyleKupiec, Anna, Barbara Adamik, Natalia Kozera, and Waldemar Gozdzik. 2020. "Elevated Procalcitonin as a Risk Factor for Postoperative Delirium in the Elderly after Cardiac Surgery—A Prospective Observational Study" Journal of Clinical Medicine 9, no. 12: 3837. https://doi.org/10.3390/jcm9123837
APA StyleKupiec, A., Adamik, B., Kozera, N., & Gozdzik, W. (2020). Elevated Procalcitonin as a Risk Factor for Postoperative Delirium in the Elderly after Cardiac Surgery—A Prospective Observational Study. Journal of Clinical Medicine, 9(12), 3837. https://doi.org/10.3390/jcm9123837






