Anterior Chamber Angle Assessment Techniques: A Review
Abstract
1. Introduction
2. Contact Techniques
2.1. Gonioscopy
- Direct, in which light from the anterior chamber passes through the cornea and through a contact gonioscopy lens, allowing direct view of the iridocorneal angle.
- Indirect, the gold standard technique, in which light from the anterior chamber is reflected in a mirror allowing an inverted view of the angle [19].
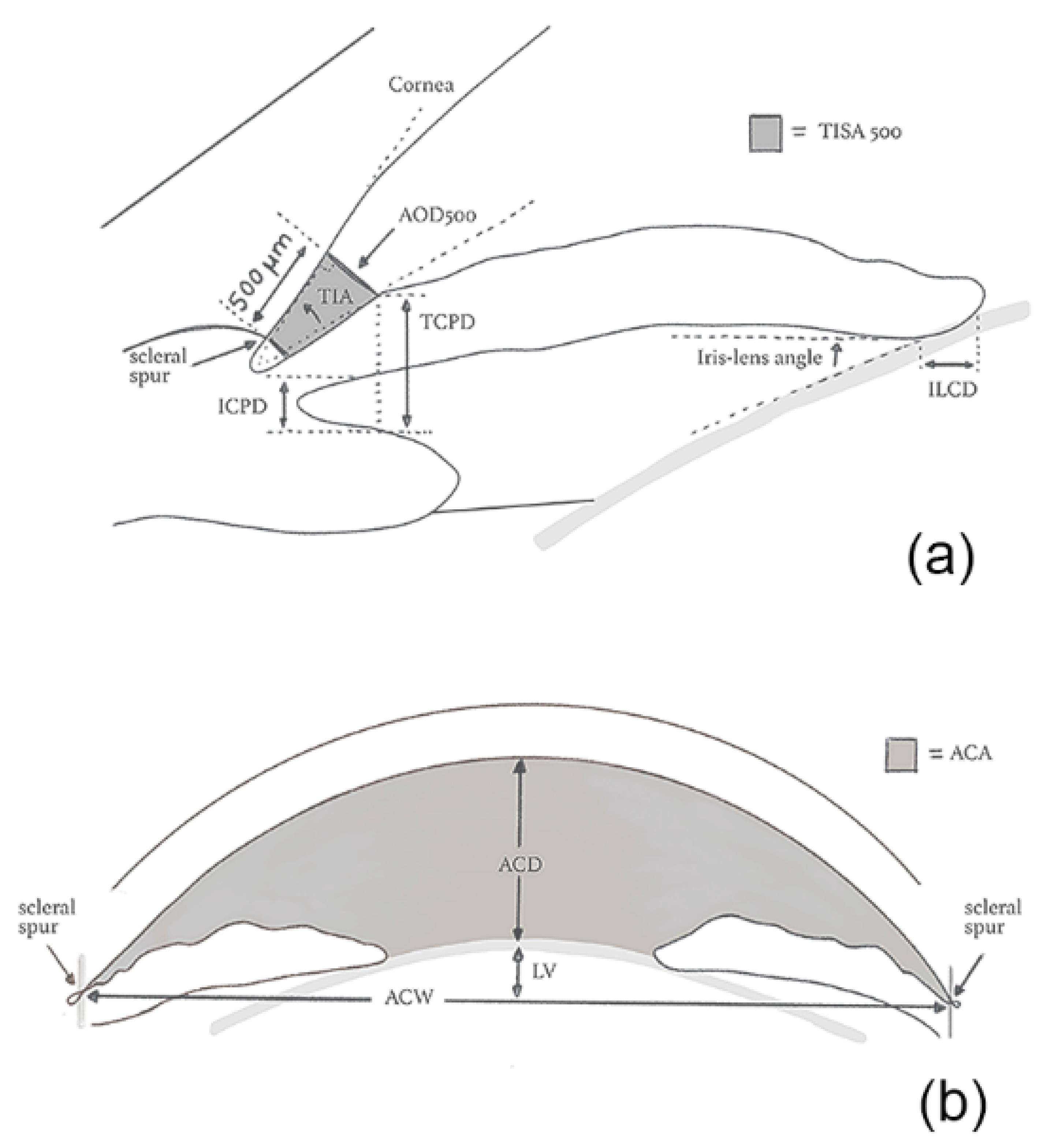
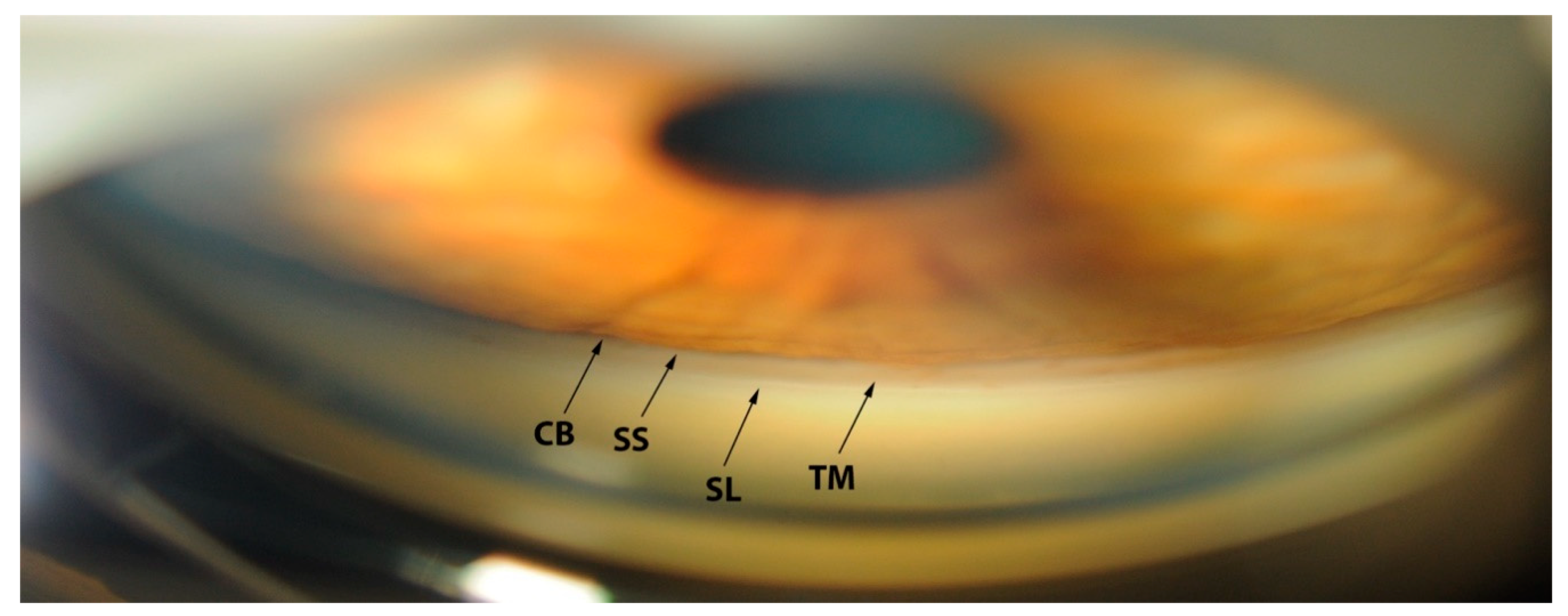
Gonioscopic Irido-Corneal Angle Anatomy
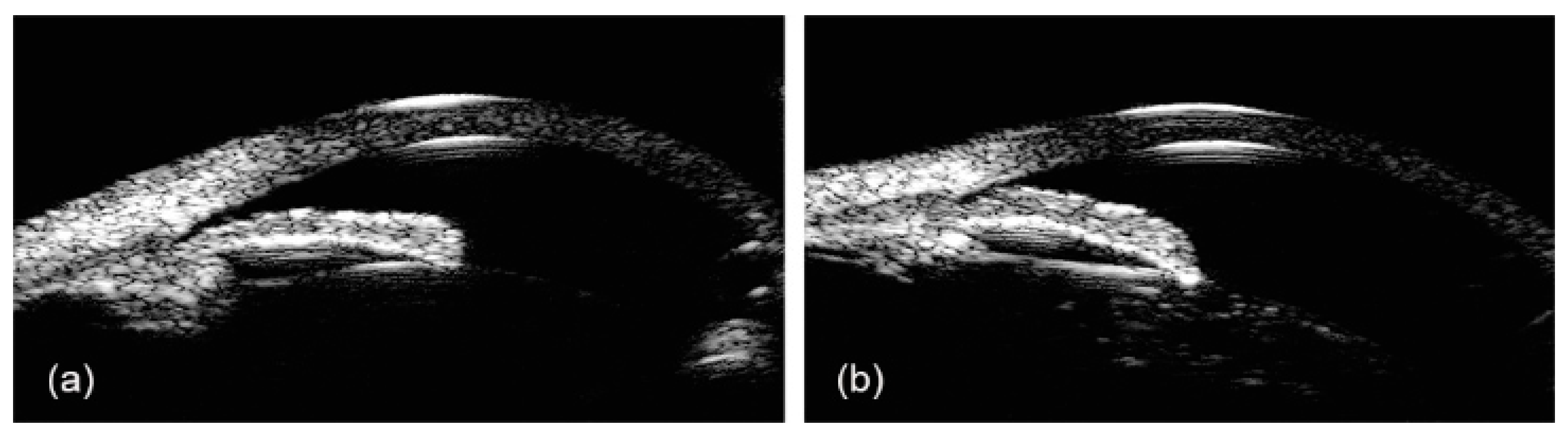
2.2. Ultrasound Biomicroscopy (UBM)
2.3. Gonio-Photographic Systems (GPS)
3. Non-Contact Techniques
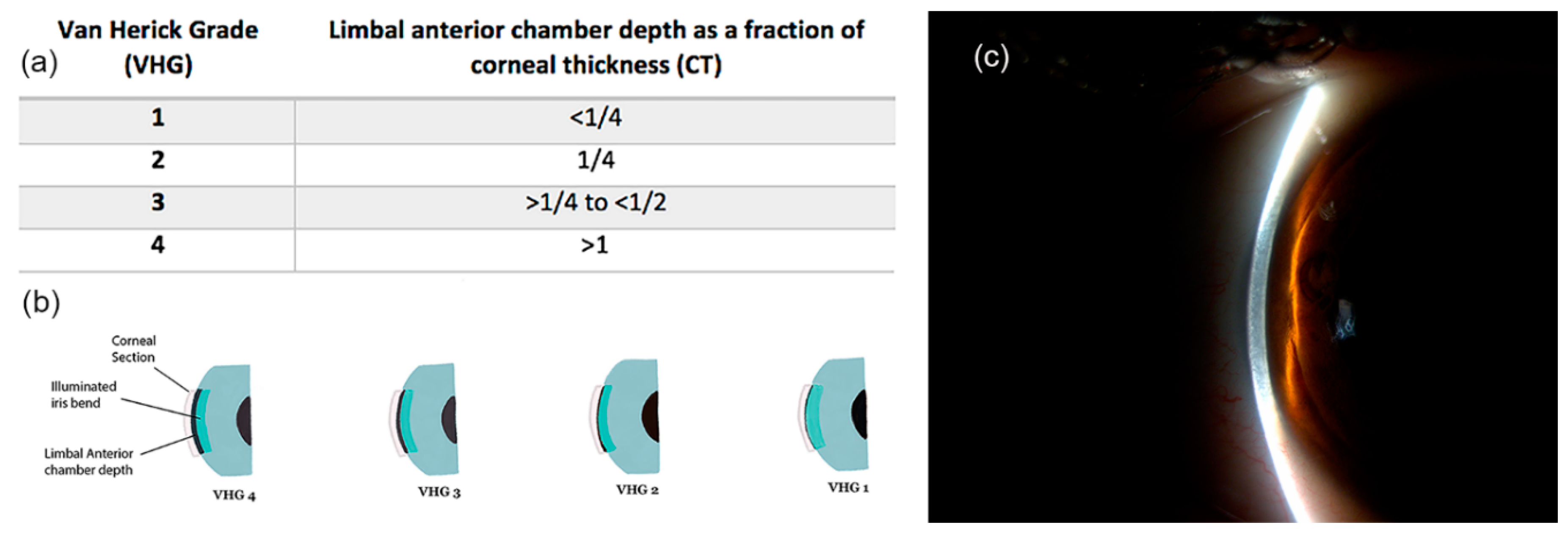
3.1. Limbal Anterior Chamber Depth Measurement (LACDM)
3.2. Scanning Peripheral Anterior Chamber Depth Analyzer (SPAC)
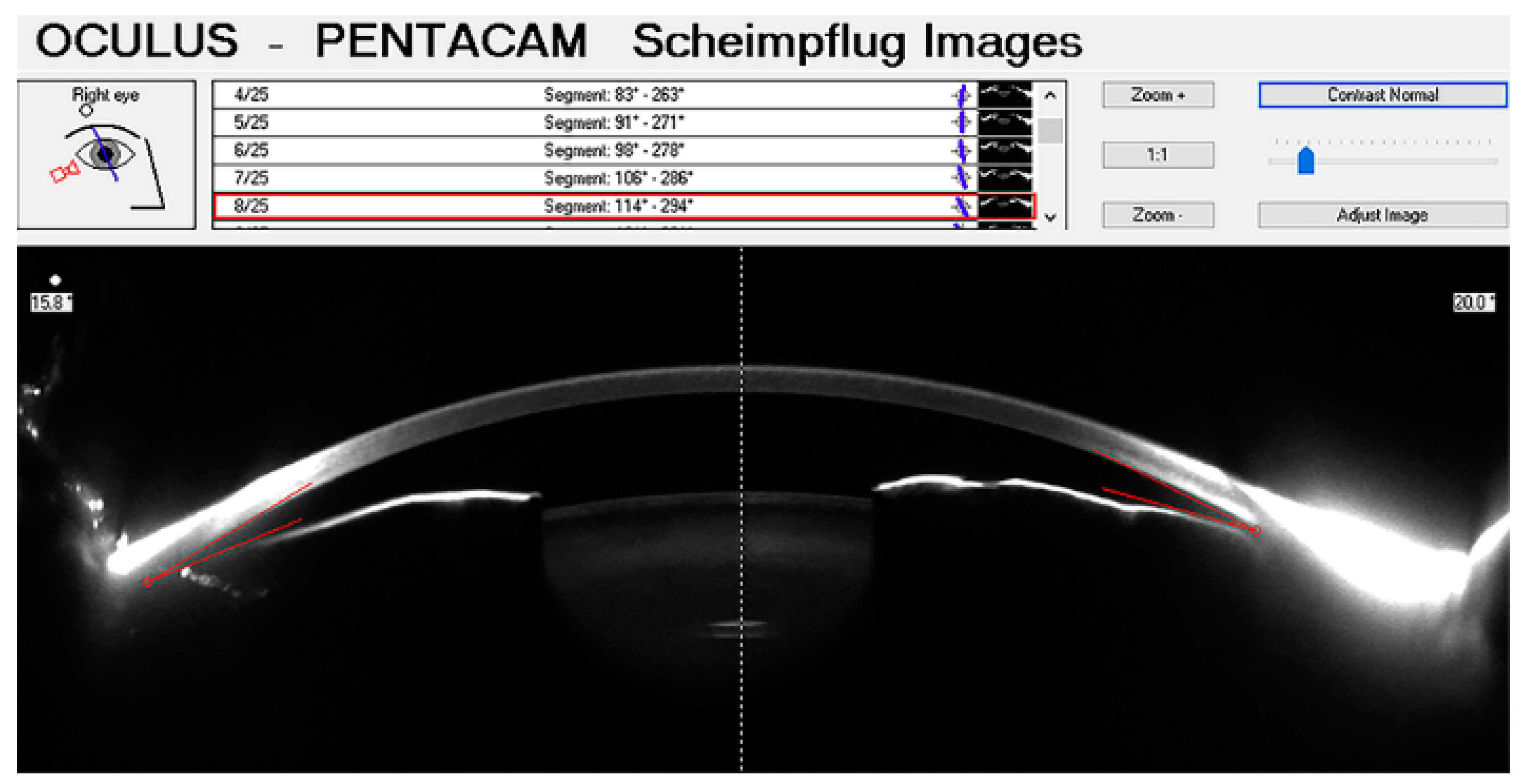
3.3. Scheimpflug Photography (SP)
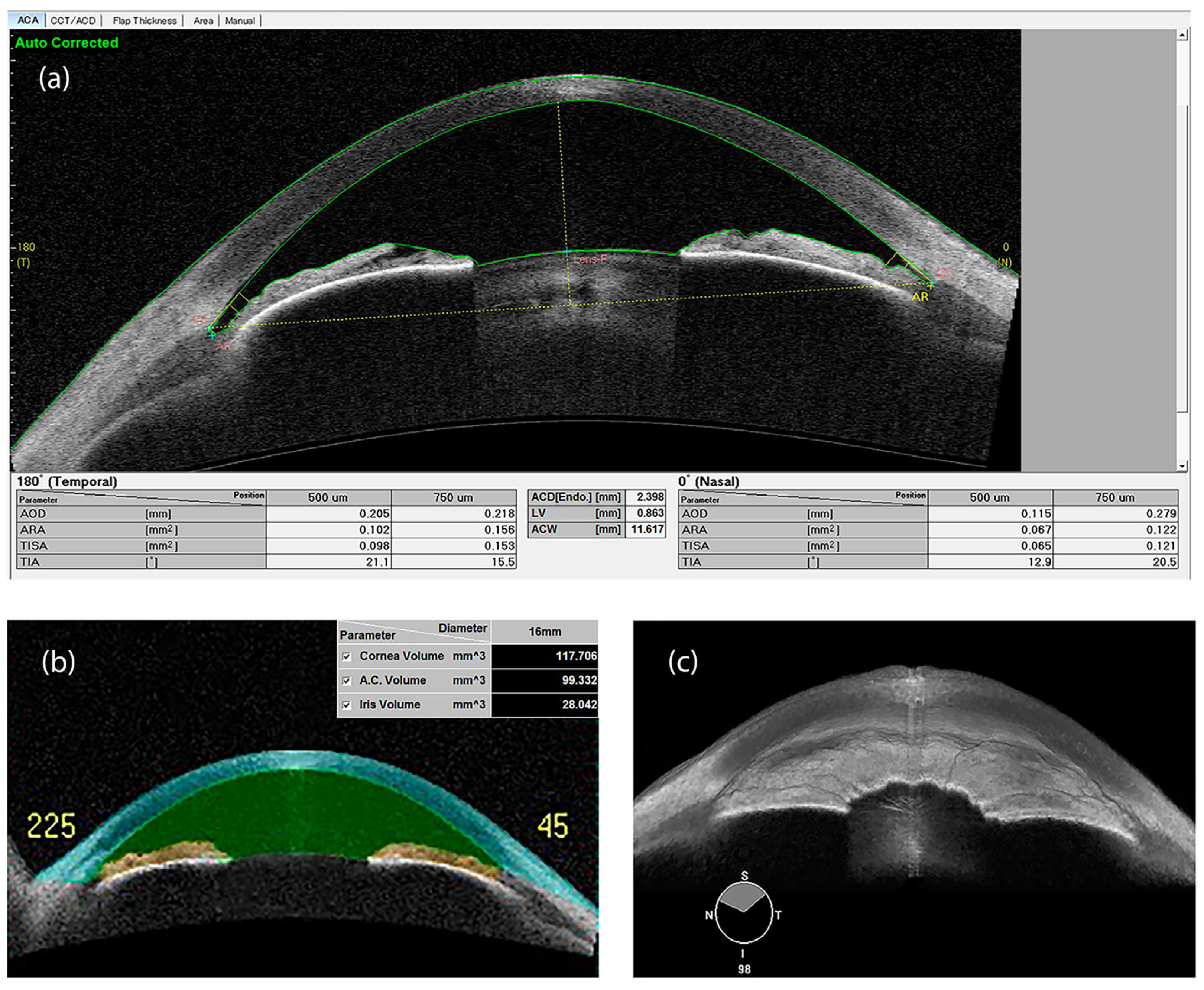
3.4. Anterior Segment Optical Coherence Tomography (AS-OCT)
3.5. Deep Learning
4. Conclusions and Future Directions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviation
| ACA | Anterior Chamber Angle |
| ACD | Anterior Chamber Depth |
| ACV | Anterior Chamber Volume |
| ACW | Anterior Chamber Width |
| AOD | Angle Opening Distance |
| ARA | Angle Recess Area |
| AS-OCT | Anterior Segment Optical Coherence Tomography |
| AUC | Area Under the Curve |
| BELL | Band of extra-canalicular limbal lamina |
| CB | Ciliary Body Band |
| GPS | Gonio-Photographic Systems |
| IC | Iris Curvature |
| ICC | Intraclass correlation coefficient |
| ICPD | Iris-ciliary process distance |
| ILAθ | Iris-lens angle |
| ILCD | Iris-lens contact distance |
| IOL | Intra-Ocular Lens |
| IT | Iris thickness |
| IV | Iris volume |
| ITC | IridoTrabecular Contact |
| IZD | Iris-zonule distance |
| LACDM | Limbal Anterior Chamber Depth Measurement |
| LV | Lens Vault |
| PAS | Peripheral Anterior Synechiae |
| SC | Schlemm’s Canal |
| SD-OCT | Spectral-Domain Optical Coherence Tomography |
| SL | Schwalbe’s line |
| SL-AOD | Schwalbe’s line-angle opening distance |
| SL-TISA | Schwalbe’s line trabecular-iris space area |
| SP | Scheimpflug Photography |
| SPAC | Scanning Peripheral Anterior Chamber depth analyzer |
| SS | Scleral spur |
| SS-OCT | Swept-Source Optical Coherence Tomography |
| TCPD | Trabecular-Ciliary Process Distance |
| TD-OCT | Time-Domain Optical Coherence Tomography |
| TIA | Trabecular-Iris Angle |
| TISA | Trabecular-Iris Space Area |
| TM | Trabecular Meshwork |
| UBM | Ultrasound Biomicroscopy |
| VHG | Van Herick Grading |
References
- Pillunat, L.E.; Erb, C.; Junemann, A.G.M.; Kimmich, F. Micro-Invasive Glaucoma Surgery (Migs): A Review of Surgical Procedures Using Stents. Clin. Ophthalmol. 2017, 11, 1583–1600. [Google Scholar] [CrossRef] [PubMed]
- Schacknow, P.N.; Samples, J.R. The Glaucoma Book: A Practical, Evidence-Based Approach to Patient Care; Springer: New York, NY, USA, 2010. [Google Scholar]
- Teixeira, F.; Sousa, D.C.; Leal, I.; Barata, A.; Neves, C.M.; Pinto, L.A. Automated Gonioscopy Photography for Iridocorneal Angle Grading. Eur. J. Ophthalmol. 2020, 30, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Tham, Y.C.; Li, X.; Wong, T.Y.; Quigley, H.A.; Aung, T.; Cheng, C.Y. Global Prevalence of Glaucoma and Projections of Glaucoma Burden through 2040: A Systematic Review and Meta-Analysis. Ophthalmology 2014, 121, 2081–2090. [Google Scholar] [CrossRef] [PubMed]
- Foster, P.J.; Johnson, G.J. Glaucoma in China: How Big Is the Problem? Br. J. Ophthalmol. 2001, 85, 1277–1282. [Google Scholar] [CrossRef]
- Arkell, S.M.; Lightman, D.A.; Sommer, A.; Taylor, H.R.; Korshin, O.M.; Tielsch, J.M. The Prevalence of Glaucoma among Eskimos of Northwest Alaska. Arch Ophthalmol. 1987, 105, 482–485. [Google Scholar] [CrossRef]
- Foster, P.J.; Buhrmann, R.; Quigley, H.A.; Johnson, G.J. The Definition and Classification of Glaucoma in Prevalence Surveys. Br. J. Ophthalmol. 2002, 86, 238–242. [Google Scholar] [CrossRef]
- Foster, P.J.; Aung, T.; Nolan, W.P.; Machin, D.; Baasanhu, J.; Khaw, P.T.; Alsbirk, P.H.; Lee, P.S.; Seah, S.K.; Johnson, G.J. Defining Occludable Angles in Population Surveys: Drainage Angle Width, Peripheral Anterior Synechiae, and Glaucomatous Optic Neuropathy in East Asian People. Br. J. Ophthalmol. 2004, 88, 486–490. [Google Scholar] [CrossRef]
- He, M.; Jiang, Y.; Huang, S.; Chang, D.S.; Munoz, B.; Aung, T.; Foster, P.J.; Friedman, D.S. Laser Peripheral Iridotomy for the Prevention of Angle Closure: A Single-Centre, Randomised Controlled Trial. Lancet 2019, 393, 1609–1618. [Google Scholar] [CrossRef]
- Day, A.C.; Baio, G.; Gazzard, G.; Bunce, C.; Azuara-Blanco, A.; Munoz, B.; Friedman, D.S.; Foster, P.J. The Prevalence of Primary Angle Closure Glaucoma in European Derived Populations: A Systematic Review. Br. J. Ophthalmol. 2012, 96, 1162–1167. [Google Scholar] [CrossRef]
- Bonomi, L.; Marchini, G.; Marraffa, M.; Bernardi, P.; de Franco, I.; Perfetti, S.; Varotto, A. Epidemiology of Angle-Closure Glaucoma: Prevalence, Clinical Types, and Association with Peripheral Anterior Chamber Depth in the Egna-Neumarket Glaucoma Study. Ophthalmology 2000, 107, 998–1003. [Google Scholar] [CrossRef]
- Varma, D.K.; Simpson, S.M.; Rai, A.S.; Ahmed, I.I.K. Undetected Angle Closure in Patients with a Diagnosis of Open-Angle Glaucoma. Can. J. Ophthalmol. 2017, 52, 373–378. [Google Scholar] [CrossRef] [PubMed]
- Quigley, H.A.; Friedman, D.S.; Hahn, S.R. Evaluation of Practice Patterns for the Care of Open-Angle Glaucoma Compared with Claims Data: The Glaucoma Adherence and Persistency Study. Ophthalmology 2007, 114, 1599–1606. [Google Scholar] [CrossRef] [PubMed]
- Campbell, P.; Redmond, T.; Agarwal, R.; Marshall, L.R.; Evans, B.J. Repeatability and Comparison of Clinical Techniques for Anterior Chamber Angle Assessment. Ophthalmic Physiol. Opt. 2015, 35, 170–178. [Google Scholar] [CrossRef] [PubMed]
- Phu, J.; Wang, H.; Khuu, S.K.; Zangerl, B.; Hennessy, M.P.; Masselos, K.; Kalloniatis, M. Anterior Chamber Angle Evaluation Using Gonioscopy: Consistency and Agreement between Optometrists and Ophthalmologists. Optom. Vis. Sci. 2019, 96, 751–760. [Google Scholar] [CrossRef] [PubMed]
- de Leon, J.M.S.; Tun, T.A.; Perera, S.A.; Aung, T. Angle Closure Imaging: A Review. Curr. Ophthalmol. Rep. 2013, 1, 80–88. [Google Scholar] [CrossRef]
- Porporato, N.; Baskaran, M.; Husain, R.; Aung, T. Recent Advances in Anterior Chamber Angle Imaging. Eye 2020, 34, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Shinoj, V.K.; Hong, X.J.J.; Murukeshan, V.M.; Baskaran, M.; Aung, T. Progress in Anterior Chamber Angle Imaging for Glaucoma Risk Prediction—A Review on Clinical Equipment, Practice and Research. Med. Eng. Phys. 2016, 38, 1383–1391. [Google Scholar]
- Raluca, M.; Mircea, F.; Andrei, F.; Carmen, D.; Miruna, N.; Grigorios, T.; Ileana, U. Old and New in Exploring the Anterior Chamber Angle. Rom. J. Ophthalmol. 2015, 59, 208–216. [Google Scholar] [PubMed]
- Liu, S.; Li, H.; Dorairaj, S.; Cheung, C.Y.; Rousso, J.; Liebmann, J.; Ritch, R.; Lam, D.S.; Leung, C.K. Assessment of Scleral Spur Visibility with Anterior Segment Optical Coherence Tomography. J. Glaucoma 2010, 19, 132–135. [Google Scholar] [CrossRef]
- Inomata, H.; Tawara, A. Anterior and Posterior Parts of Human Trabecular Meshwork. Jpn. J. Ophthalmol. 1984, 28, 339–348. [Google Scholar]
- Ozeki, H.; Shirai, S.; Majima, A.; Sano, M.; Ikeda, K. Clinical Evaluation of Posterior Embryotoxon in One Institution. Jpn. J. Ophthalmol. 1997, 41, 422–425. [Google Scholar] [CrossRef]
- Rennie, C.A.; Chowdhury, S.; Khan, J.; Rajan, F.; Jordan, K.; Lamb, R.J.; Vivian, A.J. The Prevalence and Associated Features of Posterior Embryotoxon in the General Ophthalmic Clinic. Eye 2005, 19, 396–399. [Google Scholar] [CrossRef]
- Prum, B.E., Jr.; Herndon, L.W., Jr.; Moroi, S.E.; Mansberger, S.L.; Stein, J.D.; Lim, M.C.; Rosenberg, L.F.; Gedde, S.J.; Williams, R.D. Primary Angle Closure Preferred Practice Pattern((R)) Guidelines. Ophthalmology 2016, 123, P1–P40. [Google Scholar] [CrossRef]
- Phu, J.; Wong, B.; Lim, T.; Kalloniatis, M. Assessment of Angle Closure Spectrum Disease as a Continuum of Change Using Gonioscopy and Anterior Segment Optical Coherence Tomography. Ophthalmic Physiol. Opt. 2020, 40, 617–631. [Google Scholar] [CrossRef] [PubMed]
- Pavlin, C.J.; Harasiewicz, K.; Sherar, M.D.; Foster, F.S. Clinical Use of Ultrasound Biomicroscopy. Ophthalmology 1991, 98, 287–295. [Google Scholar] [CrossRef]
- Dada, T.; Gadia, R.; Sharma, A.; Ichhpujani, P.; Bali, S.J.; Bhartiya, S.; Panda, A. Ultrasound Biomicroscopy in Glaucoma. Surv. Ophthalmol. 2011, 56, 433–450. [Google Scholar] [CrossRef] [PubMed]
- Pavlin, C.J.; Sherar, M.D.; Foster, F.S. Subsurface Ultrasound Microscopic Imaging of the Intact Eye. Ophthalmology 1990, 97, 244–250. [Google Scholar] [CrossRef]
- Mansoori, T.; Balakrishna, N. Anterior Segment Morphology in Primary Angle Closure Glaucoma Using Ultrasound Biomicroscopy. J. Curr. Glaucoma Pract. 2017, 11, 86–91. [Google Scholar] [CrossRef]
- He, M.; Wang, D.; Jiang, Y. Overview of Ultrasound Biomicroscopy. J. Curr. Glaucoma Pract. 2012, 6, 25–53. [Google Scholar] [CrossRef]
- Quek, D.T.; Nongpiur, M.E.; Perera, S.A.; Aung, T. Angle Imaging: Advances and Challenges. Indian J. Ophthalmol. 2011, 59, S69–S75. [Google Scholar]
- Potop, V.; Coviltir, V.; Schmitzer, S.; Corbu, C.G.; Ionescu, C.I.; Burcel, M.G.; Ciocalteu, A.; Dascalescu, D. Ultrasound Biomicroscopy as a Vital Tool in Occult Phacomorphic Glaucoma. Rom. J. Ophthalmol. 2019, 63, 311–314. [Google Scholar] [CrossRef] [PubMed]
- Friedman, D.S.; He, M. Anterior Chamber Angle Assessment Techniques. Surv. Ophthalmol. 2008, 53, 250–273. [Google Scholar] [CrossRef] [PubMed]
- Girija, K.; Smitha, V.K.; Ashok, A. Ultrasound Biomicroscopy: An Overview. Kerala J. Ophthalmol. 2018, 30, 216–218. [Google Scholar] [CrossRef]
- Barkana, Y.; Dorairaj, S.K.; Gerber, Y.; Liebmann, J.M.; Ritch, R. Agreement between Gonioscopy and Ultrasound Biomicroscopy in Detecting Iridotrabecular Apposition. Arch. Ophthalmol. 2007, 125, 1331–1335. [Google Scholar] [CrossRef] [PubMed]
- Kong, X.; Foster, P.J.; Huang, Q.; Zheng, Y.; Huang, W.; Cai, X.; He, M. Appositional Closure Identified by Ultrasound Biomicroscopy in Population-Based Primary Angle-Closure Glaucoma Suspects: The Liwan Eye Study. Investig. Ophthalmol. Vis. Sci. 2011, 52, 3970–3975. [Google Scholar] [CrossRef]
- Radhakrishnan, S.; Goldsmith, J.; Huang, D.; Westphal, V.; Dueker, D.K.; Rollins, A.M.; Izatt, J.A.; Smith, S.D. Comparison of Optical Coherence Tomography and Ultrasound Biomicroscopy for Detection of Narrow Anterior Chamber Angles. Arch. Ophthalmol. 2005, 123, 1053–1059. [Google Scholar] [CrossRef]
- Urbak, S.F.; Pedersen, J.K.; Thorsen, T.T. Ultrasound Biomicroscopy. Ii. Intraobserver and Interobserver Reproducibility of Measurements. Acta Ophthalmol. Scand. 1998, 76, 546–549. [Google Scholar] [CrossRef]
- Pavlin, C.J.; Harasiewicz, K.; Foster, F.S. Ultrasound Biomicroscopy of Anterior Segment Structures in Normal and Glaucomatous Eyes. Am. J. Ophthalmol. 1992, 113, 381–389. [Google Scholar] [CrossRef]
- Wang, N.; Ye, T.; Lai, M.; Ou, Y.; Zeng, M.; Yang, C. Comparison of Results of Chamber Angle Examination by Ultrasound Biomicroscopy and Gonioscopy. Zhonghua Yan Ke Za Zhi 1999, 35, 10, 174–178. (In Chinese) [Google Scholar]
- Foster, F.S.; Pavlin, C.J.; Harasiewicz, K.A.; Christopher, D.A.; Turnbull, D.H. Advances in Ultrasound Biomicroscopy. Ultrasound Med. Biol. 2000, 26, 1–27. [Google Scholar] [CrossRef]
- Ishikawa, H.; Schuman, J.S. Anterior Segment Imaging: Ultrasound Biomicroscopy. Ophthalmol. Clin. N. Am. 2004, 17, 7–20. [Google Scholar] [CrossRef]
- Pavlin, C.J.; Macken, P.; Trope, G.E.; Harasiewicz, K.; Foster, F.S. Accommodation and Iridotomy in the Pigment Dispersion Syndrome. Ophthalmic Surg. Lasers 1996, 27, 113–120. [Google Scholar] [PubMed]
- Qian, Y.; Liu, L.; Shi, Y.; Wang, M.; Li, M.; Zou, J. Assessment of Anterior Chamber by Ultrasound Biomicroscopy and Anterior Segment Optical Coherence Tomography in Patients with Inflammatory Glaucoma. J. Int. Med. Res. 2019, 47, 5950–5956. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Huang, J.; Lin, J.; Liang, X.; Cai, X.; Ge, J. Quantitative Measurements of the Ciliary Body in Eyes with Malignant Glaucoma after Trabeculectomy Using Ultrasound Biomicroscopy. Ophthalmology 2014, 121, 862–869. [Google Scholar] [CrossRef] [PubMed]
- Bell, N.P.; Nagi, K.S.; Cumba, R.J.; Chuang, A.Z.; Lee, D.A.; Prager, T.C.; Rao, K.; Feldman, R.M. Age and Positional Effect on the Anterior Chamber Angle: Assessment by Ultrasound Biomicroscopy. ISRN Ophthalmol. 2013, 2013, 706201. [Google Scholar] [CrossRef]
- Spaeth, G.L.; Azuara-Blanco, A.; Araujo, S.V.; Augsburger, J.J. Intraobserver and Interobserver Agreement in Evaluating the Anterior Chamber Angle Configuration by Ultrasound Biomicroscopy. J. Glaucoma 1997, 6, 13–17. [Google Scholar] [CrossRef]
- Yang, H.; Lin, Z.; Chen, X.; Wang, N. Intraobserver Reproducibility Study of Parameters for Measurement of Position and Height of Ciliary Process by Ultrasound Biomicroscopy. Yan Ke Xue Bao 1999, 15, 103–106, 123. [Google Scholar]
- Zhang, Q.; Jin, W.; Wang, Q. Repeatability, Reproducibility, and Agreement of Central Anterior Chamber Depth Measurements in Pseudophakic and Phakic Eyes: Optical Coherence Tomography Versus Ultrasound Biomicroscopy. J. Cataract Refract. Surg. 2010, 36, 941–946. [Google Scholar] [CrossRef]
- Tian, J.; Marziliano, P.; Baskaran, M.; Wong, H.T.; Aung, T. Automatic Anterior Chamber Angle Assessment for Hd-Oct Images. IEEE Trans. Biomed. Eng. 2011, 58, 3242–3249. [Google Scholar] [CrossRef]
- Baskaran, M.; Cheng, J.; Perera, S.A.; Tun, T.A.; Liu, J.; Aung, T. Automated Analysis of Angle Closure from Anterior Chamber Angle Images. Investig. Ophthalmol. Vis Sci. 2014, 55, 7669–7673. [Google Scholar] [CrossRef]
- Li, W.; Chen, Q.; Jiang, Z.; Deng, G.; Zong, Y.; Shi, G.; Jiang, C.; Sun, X. Automatic Anterior Chamber Angle Measurement for Ultrasound Biomicroscopy Using Deep Learning. J. Glaucoma 2020, 29, 81–85. [Google Scholar] [CrossRef] [PubMed]
- Perera, S.A.; Baskaran, M.; Friedman, D.S.; Tun, T.A.; Htoon, H.M.; Kumar, R.S.; Aung, T. Use of Eyecam for Imaging the Anterior Chamber Angle. Investig. Ophthalmol. Vis. Sci. 2010, 51, 2993–2997. [Google Scholar] [CrossRef] [PubMed]
- Baskaran, M.; Aung, T.; Friedman, D.S.; Tun, T.A.; Perera, S.A. Comparison of Eyecam and Anterior Segment Optical Coherence Tomography in Detecting Angle Closure. Acta Ophthalmol. 2012, 90, e621–e625. [Google Scholar] [CrossRef]
- Baskaran, M.; Perera, S.A.; Nongpiur, M.E.; Tun, T.A.; Park, J.; Kumar, R.S.; Friedman, D.S.; Aung, T. Angle Assessment by Eyecam, Goniophotography, and Gonioscopy. J. Glaucoma 2012, 21, 493–497. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.Y.; Pardeshi, A.A.; Burkemper, B.; Richter, G.M.; Lin, S.C.; McKean-Cowdin, R.; Varma, R. Differences in Anterior Chamber Angle Assessments between Gonioscopy, Eyecam, and Anterior Segment Oct: The Chinese American Eye Study. Transl. Vis. Sci. Technol. 2019, 8, 5. [Google Scholar] [CrossRef] [PubMed]
- Murakami, Y.; Wang, D.; Burkemper, B.; Lin, S.C.; Varma, R. A Population-Based Assessment of the Agreement between Grading of Goniophotographic Images and Gonioscopy in the Chinese-American Eye Study (Ches). Investig. Ophthalmol. Vis. Sci. 2016, 57, 4512–4516. [Google Scholar] [CrossRef] [PubMed]
- Ritch, R. Pigment Dispersion Syndrome. Am. J. Ophthalmol. 1998, 126, 442–445. [Google Scholar] [CrossRef]
- Yue, S.; Yang, X.; Marion, K.M.; Francis, B.A.; Sadda, S.R.; Chopra, V. Novel and Semiautomated 360-Degree Gonioscopic Anterior Chamber Angle Imaging in under 60 Seconds. Ophthalmol. Glaucoma 2019, 2, 215–223. [Google Scholar]
- Shinoj, V.K.; Murukeshan, V.M.; Baskaran, M.; Aung, T. Integrated Flexible Handheld Probe for Imaging and Evaluation of Iridocorneal Angle. J. Biomed. Opt. 2015, 20, 016014. [Google Scholar] [CrossRef]
- Perinchery, S.M.; Shinde, A.; Fu, C.Y.; Hong, X.J.J.; Baskaran, M.; Aung, T.; Murukeshan, V.M. High Resolution Iridocorneal Angle Imaging System by Axicon Lens Assisted Gonioscopy. Sci. Rep. 2016, 6, 30844. [Google Scholar] [CrossRef]
- Van Herick, W.; Shaffer, R.N.; Schwartz, A. Estimation of Width of Angle of Anterior Chamber. Incidence and Significance of the Narrow Angle. Am. J. Ophthalmol. 1969, 68, 626–629. [Google Scholar] [CrossRef]
- Thomas, R.; George, T.; Braganza, A.; Muliyil, J. The Flashlight Test and Van Herick’s Test Are Poor Predictors for Occludable Angles. Aust. N. Z. J. Ophthalmol. 1996, 24, 251–256. [Google Scholar] [CrossRef] [PubMed]
- Congdon, N.G.; Spaeth, G.L.; Augsburger, J.; Klancnik, J., Jr.; Patel, K.; Hunter, D.G. A Proposed Simple Method for Measurement in the Anterior Chamber Angle: Biometric Gonioscopy. Ophthalmology 1999, 106, 2161–2167. [Google Scholar] [CrossRef]
- Kashiwagi, K.; Tokunaga, T.; Iwase, A.; Yamamoto, T.; Tsukahara, S. Agreement between Peripheral Anterior Chamber Depth Evaluation Using the Van Herick Technique and Angle Width Evaluation Using the Shaffer System in Japanese. Jpn. J. Ophthalmol. 2005, 49, 134–136. [Google Scholar] [CrossRef] [PubMed]
- Foster, P.J.; Devereux, J.G.; Alsbirk, P.H.; Lee, P.S.; Uranchimeg, D.; Machin, D.; Johnson, G.J.; Baasanhu, J. Detection of Gonioscopically Occludable Angles and Primary Angle Closure Glaucoma by Estimation of Limbal Chamber Depth in Asians: Modified Grading Scheme. Br. J. Ophthalmol. 2000, 84, 186–192. [Google Scholar] [CrossRef]
- Baskaran, M.; Oen, F.T.; Chan, Y.H.; Hoh, S.T.; Ho, C.L.; Kashiwagi, K.; Foster, P.J.; Aung, T. Comparison of the Scanning Peripheral Anterior Chamber Depth Analyzer and the Modified Van Herick Grading System in the Assessment of Angle Closure. Ophthalmology 2007, 114, 501–506. [Google Scholar] [CrossRef]
- Sihota, R.; Kamble, N.; Sharma, A.K.; Bhari, A.; Gupta, A.; Midha, N.; Selvan, H.; Dada, T.; Gupta, V.; Pandey, R.M. ‘Van Herick Plus’: A Modified Grading Scheme for the Assessment of Peripheral Anterior Chamber Depth and Angle. Br. J. Ophthalmol. 2019, 103, 960–965. [Google Scholar] [CrossRef]
- Javed, A.; Loutfi, M.; Kaye, S.; Batterbury, M. Interobserver Reliability When Using the Van Herick Method to Measure Anterior Chamber Depth. Oman J. Ophthalmol. 2017, 10, 9–12. [Google Scholar] [CrossRef]
- Xu, B.Y.; Pardeshi, A.A.; Burkemper, B.; Richter, G.M.; Lin, S.C.; McKean-Cowdin, R.; Varma, R. Quantitative Evaluation of Gonioscopic and Eyecam Assessments of Angle Dimensions Using Anterior Segment Optical Coherence Tomography. Transl. Vis. Sci. Technol. 2018, 7, 33. [Google Scholar] [CrossRef]
- Phu, J.; Tong, J.; Zangerl, B.; Le, J.L.; Kalloniatis, M. Cluster Analysis Reveals Patterns of Age-Related Change in Anterior Chamber Depth for Gender and Ethnicity: Clinical Implications. Ophthalmic Physiol. Opt. 2020, 40, 632–649. [Google Scholar] [CrossRef]
- Jindal, A.; Ctori, I.; Virgili, G.; Lucenteforte, E.; Lawrenson, J.G. Non-Contact Tests for Identifying People at Risk of Primary Angle Closure Glaucoma. Cochrane Database Syst. Rev. 2020, 5, CD012947. [Google Scholar] [PubMed]
- Pettersson, L.; Källmark, F. Difference in the Anterior Chamber Angle of the Four Meridians. J. Appl. Med. Sci. 2012, 1, 1–13. [Google Scholar]
- Källmark, F.P.; Sakhi, M. Evaluation of Nasal and Temporal Anterior Chamber Angle with Four Different Techniques. Int. J. Clin. Med. 2013, 4, 548–555. [Google Scholar]
- Kashiwagi, K.; Kashiwagi, F.; Hiejima, Y.; Tsukahara, S. Finding Cases of Angle-Closure Glaucoma in Clinic Setting Using a Newly Developed Instrument. Eye 2006, 20, 319–324. [Google Scholar] [CrossRef] [PubMed]
- Lavanya, R.; Foster, P.J.; Sakata, L.M.; Friedman, D.S.; Kashiwagi, K.; Wong, T.-Y.; Aung, H.T.; Alfred, T.; Gao, H.; Ee, A.G.; et al. Screening for Narrow Angles in the Singapore Population: Evaluation of New Noncontact Screening Methods. Ophthalmology 2008, 115, 1720–1727.e2. [Google Scholar] [CrossRef] [PubMed]
- Faria-Correia, F. and Ambrósio Jr, R. Clinical Applications of the Scheimpflug Principle in Ophthalmology. Rev. Bras. Oftalmol. 2016, 75, 160–165. [Google Scholar] [CrossRef]
- Brown, N. An Advanced Slit-Image Camera. Br. J. Ophthalmol. 1972, 56, 624. [Google Scholar] [CrossRef][Green Version]
- Rabsilber, T.M.; Khoramnia, R.; Auffarth, G.U. Anterior Chamber Measurements Using Pentacam Rotating Scheimpflug Camera. J. Cataract Refract. Surg. 2006, 32, 456–459. [Google Scholar] [CrossRef]
- Li, X.; Wang, Z.; Cao, Q.; Hu, L.; Tian, F.; Dai, H. Pentacam Could Be a Useful Tool for Evaluating and Qualifying the Anterior Chamber Morphology. Int. J. Clin. Exp. Med. 2014, 7, 1878–1882. [Google Scholar]
- Masoud, M.; Livny, E.; Bahar, I. Repeatability and Intrasession Reproducibility Obtained by the Sirius Anterior Segment Analysis System. Eye Contact Lens 2015, 41, 107–110. [Google Scholar] [CrossRef]
- Bedei, A.; Appolloni, I.; Madesani, A.; Pietrelli, A.; Franceschi, S.; Barabesi, L. Repeatability and Agreement of 2 Scheimpflug Analyzers in Measuring the Central Corneal Thickness and Anterior Chamber Angle, Volume, and Depth. Eur. J. Ophthalmol. 2012, 22 (Suppl. S7), S29–S32. [Google Scholar] [CrossRef] [PubMed]
- Prakash, G.; Srivastava, D. Single Session, Intrauser Repeatability of Anterior Chamber Biometric and Corneal Pachy-Volumetric Parameters Using a New Scheimpflug+Placido Device. J. Ophtalmol. 2016, 9, 85–92. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Grewal, D.S.; Brar, G.S.; Jain, R. and Grewal, S.P.S. Comparison of Scheimpflug Imaging and Spectral Domain Anterior Segment Optical Coherence Tomography for Detection of Narrow Anterior Chamber Angles. Eye (Lond.) 2011, 25, 603–611. [Google Scholar] [CrossRef] [PubMed]
- Kurita, N.; Mayama, C.; Tomidokoro, A.; Aihara, M.; Araie, M. Potential of the Pentacam in Screening for Primary Angle Closure and Primary Angle Closure Suspect. J. Glaucoma 2009, 18, 506–512. [Google Scholar] [CrossRef]
- Jain, R.; Grewal, D.; Grewal, S.P.S. Predictive Value of Anterior Chamber Volume on Scheimpflug Imaging in Eyes with Narrow Angle. Asian J. Ophtalmol. 2007, 9, 58. [Google Scholar]
- Yi, J.-H.; Lee, H.; Hong, S.; Seong, G.J.; Kang, S.Y.; Ma, K.T.; Kim, C.Y. Anterior Chamber Measurements by Pentacam and as-Oct in Eyes with Normal Open Angles. Korean J. Ophthalmol. 2008, 22, 242–245. [Google Scholar] [CrossRef]
- Nguyen, A.T.; Liu, T.; Liu, J. Applications of Scheimpflug Imaging in Glaucoma Management: Current and Potential Applications. J. Ophthalmol. 2016, 2016, 3062381. [Google Scholar] [CrossRef]
- Ruiz-Belda, C.; Piñero, D.P.; Ruiz-Fortes, P.; Soto-Negro, R.; Moya, M.; Pérez-Cambrodí, R.J.; Artola, A. Intra-Session Repeatability of Iridocorneal Angle Measurements Provided by a Scheimpflug Photography-Based System in Healthy Eyes. Graefe’s Arch. Clin. Exp. Ophthalmol. 2016, 254, 169–175. [Google Scholar] [CrossRef]
- Porporato, N.; Baskaran, M.; Tun, T.A.; Sultana, R.; Tan, M.; Quah, J.H.; Allen, J.C.; Perera, S.; Friedman, D.S.; Cheng, C.Y.; et al. Understanding Diagnostic Disagreement in Angle Closure Assessment between Anterior Segment Optical Coherence Tomography and Gonioscopy. Br. J. Ophthalmol. 2020, 104, 795–799. [Google Scholar] [CrossRef]
- Li, P.; Johnstone, M.; Wang, R.K. Full Anterior Segment Biometry with Extended Imaging Range Spectral Domain Optical Coherence Tomography at 1340 Nm. J. Biomed. Opt. 2014, 19, 046013. [Google Scholar] [CrossRef]
- Ang, M.; Baskaran, M.; Werkmeister, R.M.; Chua, J.; Schmidl, D.; Santos, V.A.D.; Garhofer, G.; Mehta, J.S.; Schmetterer, L. Anterior Segment Optical Coherence Tomography. Prog. Retin. Eye Res. 2018, 66, 132–156. [Google Scholar] [CrossRef] [PubMed]
- Maslin, J.S.; Barkana, Y.; Dorairaj, S.K. Anterior Segment Imaging in Glaucoma: An Updated Review. Indian J. Ophthalmol. 2015, 63, 630–640. [Google Scholar] [PubMed]
- Nolan, W.P.; See, J.L.; Chew, P.T.; Friedman, D.S.; Smith, S.D.; Radhakrishnan, S.; Zheng, C.; Foster, P.J.; Aung, T. Detection of Primary Angle Closure Using Anterior Segment Optical Coherence Tomography in Asian Eyes. Ophthalmology 2007, 114, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Sakata, L.M.; Lavanya, R.; Friedman, D.S.; Aung, H.T.; Gao, H.; Kumar, R.S.; Foster, P.J.; Aung, T. Comparison of Gonioscopy and Anterior Segment Ocular Coherence Tomography in Detecting Angle Closure in Different Quadrants of the Anterior Chamber Angle. Ophthalmology 2008, 115, 769–774. [Google Scholar] [CrossRef] [PubMed]
- Chansangpetch, S.; Tran, B.; Perez, C.I.; Siguan-Bell, C.; Lau, K.; Nguyen, A.H.; Thai, A.; He, M.; Wang, D.; Nguyen, N.; et al. Comparison of Anterior Segment Optical Coherence Tomography Parameters among Vietnamese, Chinese, and Whites. Am. J. Ophthalmol. 2018, 195, 72–82. [Google Scholar] [CrossRef] [PubMed]
- Baskaran, M.; Iyer, J.V.; Narayanaswamy, A.K.; He, Y.; Sakata, L.M.; Wu, R.; Liu, D.; Nongpiur, M.E.; Friedman, D.S.; Aung, T. Anterior Segment Imaging Predicts Incident Gonioscopic Angle Closure. Ophthalmology 2015, 122, 2380–2384. [Google Scholar] [CrossRef]
- Nongpiur, M.E.; Aboobakar, I.F.; Baskaran, M.; Narayanaswamy, A.; Sakata, L.M.; Wu, R.; Atalay, E.; Friedman, D.S.; Aung, T. Association of Baseline Anterior Segment Parameters with the Development of Incident Gonioscopic Angle Closure. JAMA Ophthalmol. 2017, 135, 252–258. [Google Scholar] [CrossRef]
- Chang, D.S.; Sakata, L.M.; Aung, T.; He, M.G.; Lavanya, R.; Kashiwagi, K.; Friedman, D.S. Single Versus Sequential Testing with Scanning Peripheral Anterior Chamber Depth Analyser, Iolmaster and Anterior Segment Optical Coherence Tomography for the Detection of Narrow Angles. Br. J. Ophthalmol. 2011, 95, 1410–1414. [Google Scholar] [CrossRef]
- Nongpiur, M.E.; Sakata, L.M.; Friedman, D.S.; He, M.; Chan, Y.-H.; Lavanya, R.; Wong, T.Y.; Aung, T. Novel Association of Smaller Anterior Chamber Width with Angle Closure in Singaporeans. Ophthalmology 2010, 117, 1967–1973. [Google Scholar] [CrossRef]
- Nongpiur, M.E.; He, M.; Amerasinghe, N.; Friedman, D.S.; Tay, W.T.; Baskaran, M.; Smith, S.D.; Wong, T.Y.; Aung, T. Lens Vault, Thickness, and Position in Chinese Subjects with Angle Closure. Ophthalmology 2011, 118, 474–479. [Google Scholar] [CrossRef]
- Wang, B.; Sakata, L.M.; Friedman, D.S.; Chan, Y.H.; He, M.; Lavanya, R.; Wong, T.Y.; Aung, T. Quantitative Iris Parameters and Association with Narrow Angles. Ophthalmology 2010, 117, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Foo, L.L.; Nongpiur, M.E.; Allen, J.C.; Perera, S.A.; Friedman, D.S.; He, M.; Cheng, C.Y.; Wong, T.Y.; Aung, T. Determinants of Angle Width in Chinese Singaporeans. Ophthalmology 2012, 119, 278–282. [Google Scholar] [CrossRef]
- Cheung, C.Y.; Liu, S.; Weinreb, R.N.; Liu, J.; Li, H.; Leung, D.Y.; Dorairaj, S.; Liebmann, J.; Ritch, R.; Lam, D.S.; et al. Dynamic Analysis of Iris Configuration with Anterior Segment Optical Coherence Tomography. Investig. Ophthalmol. Vis. Sci. 2010, 51, 4040–4046. [Google Scholar] [CrossRef]
- Baskaran, M.; Ho, S.W.; Tun, T.A.; How, A.C.; Perera, S.A.; Friedman, D.S.; Aung, T. Assessment of Circumferential Angle-Closure by the Iris-Trabecular Contact Index with Swept-Source Optical Coherence Tomography. Ophthalmology 2013, 120, 2226–2231. [Google Scholar] [CrossRef] [PubMed]
- Lai, I.; Mak, H.; Lai, G.; Yu, M.; Lam, D.S.; Leung, C.K. Anterior Chamber Angle Imaging with Swept-Source Optical Coherence Tomography: Measuring Peripheral Anterior Synechia in Glaucoma. Ophthalmology 2013, 120, 1144–1149. [Google Scholar] [CrossRef] [PubMed]
- Porporato, N.; Baskaran, M.; Tun, T.A.; Sultana, R.; Tan, M.C.L.; Quah, J.H.M.; Allen, J.; Friedman, D.S.; Cheng, C.-Y.; Aung, A. Assessment of Circumferential Angle Closure with Swept-Source Optical Coherence Tomography: A Community Based Study. Am. J. Ophthalmol. 2019, 199, 133–139. [Google Scholar] [CrossRef]
- Melese, E.K.; Chan, J.D.; Blieden, L.S.; Chuang, A.Z.; Baker, L.A.; Bell, N.P.; Feldman, R.M. Determination and Validation of Thresholds of Anterior Chamber Parameters by Dedicated Anterior Segment Optical Coherence Tomography. Am. J. Ophthalmol. 2016, 169, 208–217. [Google Scholar] [CrossRef]
- Rigi, M.; Bell, N.P.; Lee, D.A.; Baker, L.A.; Chuang, A.Z.; Nguyen, D.; Minnal, V.R.; Feldman, R.M.; Blieden, L.S. Agreement between Gonioscopic Examination and Swept Source Fourier Domain Anterior Segment Optical Coherence Tomography Imaging. J. Ophthalmol. 2016, 2016, 1727039. [Google Scholar] [CrossRef]
- Tun, T.A.; Baskaran, M.; Perera, S.A.; Htoon, H.M.; Aung, T.; Husain, R. Swept-Source Optical Coherence Tomography Assessment of Iris-Trabecular Contact after Phacoemulsification with or without Goniosynechialysis in Eyes with Primary Angle Closure Glaucoma. Br. J. Ophthalmol. 2015, 99, 927–931. [Google Scholar] [CrossRef]
- Li, F.; Zhou, R.; Gao, K.; Jin, L.; Zhang, X. Volumetric Parameters-Based Differentiation of Narrow Angle from Open Angle and Classification of Angle Configurations: An Ss-Oct Study. Br. J. Ophthalmol. 2020, 104, 92–97. [Google Scholar] [CrossRef]
- Quigley, H.A.; Silver, D.M.; Friedman, D.S.; He, M.; Plyler, R.J.; Eberhart, C.G.; Jampel, H.D.; Ramulu, P. Iris Cross-Sectional Area Decreases with Pupil Dilation and Its Dynamic Behavior Is a Risk Factor in Angle Closure. J. Glaucoma 2009, 18, 173–179. [Google Scholar] [CrossRef]
- Gold, M.E.; Kansara, S.; Nagi, K.S.; Bell, N.P.; Blieden, L.S.; Chuang, A.Z.; Baker, L.A.; Mankiewicz, K.A.; Feldman, R.M. Age-Related Changes in Trabecular Meshwork Imaging. Biomed. Res. Int. 2013, 2013, 295204. [Google Scholar] [CrossRef]
- Kim, D.Y.; Sung, K.R.; Kang, S.Y.; Cho, J.W.; Lee, K.S.; Park, S.B.; Kim, S.T.; Kook, M.S. Characteristics and Reproducibility of Anterior Chamber Angle Assessment by Anterior-Segment Optical Coherence Tomography. Acta Ophthalmol. 2011, 89, 435–441. [Google Scholar] [CrossRef]
- Li, H.; Leung, C.K.; Cheung, C.Y.; Wong, L.; Pang, C.P.; Weinreb, R.N.; Lam, D.S. Repeatability and Reproducibility of Anterior Chamber Angle Measurement with Anterior Segment Optical Coherence Tomography. Br. J. Ophthalmol. 2007, 91, 1490–1492. [Google Scholar] [CrossRef]
- Muller, M.; Dahmen, G.; Porksen, E.; Geerling, G.; Laqua, H.; Ziegler, A.; Hoerauf, H. Anterior Chamber Angle Measurement with Optical Coherence Tomography: Intraobserver and Interobserver Variability. J. Cataract Refract. Surg. 2006, 32, 1803–1808. [Google Scholar] [CrossRef]
- Marion, K.M.; Dastiridou, A.; Niemeyer, M.; Francis, B.A.; Sadda, S.R.; Chopra, V. Anterior Chamber Angle Morphometry Measurement Changes to Ambient Illumination Scaling in Visante Time Domain Optical Coherence Tomography. Curr. Eye Res. 2017, 42, 386–393. [Google Scholar] [CrossRef]
- Marion, K.M.; Niemeyer, M.; Francis, B.; Sadda, S.R.; Chopra, V. Effects of Light Variation on Schwalbe’s Line-Based Anterior Chamber Angle Metrics Measured with Cirrus Spectral Domain Optical Coherence Tomography. Clin. Exp. Ophthalmol. 2016, 44, 455–464. [Google Scholar] [CrossRef]
- Sakata, L.M.; Lavanya, R.; Friedman, D.S.; Aung, H.T.; Seah, S.K.; Foster, P.J.; Aung, T. Assessment of the Scleral Spur in Anterior Segment Optical Coherence Tomography Images. Arch. Ophthalmol. 2008, 126, 181–185. [Google Scholar] [CrossRef]
- Wang, B.S.; Narayanaswamy, A.; Amerasinghe, N.; Zheng, C.; He, M.; Chan, Y.H.; Nongpiur, M.E.; Friedman, D.S.; Aung, T. Increased Iris Thickness and Association with Primary Angle Closure Glaucoma. Br. J. Ophthalmol. 2011, 95, 46–50. [Google Scholar] [CrossRef]
- Console, J.W.; Sakata, L.M.; Aung, T.; Friedman, D.S.; He, M. Quantitative Analysis of Anterior Segment Optical Coherence Tomography Images: The Zhongshan Angle Assessment Program. Br. J. Ophthalmol. 2008, 92, 1612–1616. [Google Scholar] [CrossRef]
- Cumba, R.J.; Radhakrishnan, S.; Bell, N.P.; Nagi, K.S.; Chuang, A.Z.; Lin, S.C.; Mankiewicz, K.A.; Feldman, R.M. Reproducibility of Scleral Spur Identification and Angle Measurements Using Fourier Domain Anterior Segment Optical Coherence Tomography. J. Ophthalmol. 2012, 2012, 487309. [Google Scholar] [CrossRef]
- Cheung, C.Y.; Zheng, C.; Ho, C.L.; Tun, T.A.; Kumar, R.S.; Sayyad, F.E.; Wong, T.Y.; Aung, T. Novel Anterior-Chamber Angle Measurements by High-Definition Optical Coherence Tomography Using the Schwalbe Line as the Landmark. Br. J. Ophthalmol. 2011, 95, 955–959. [Google Scholar] [CrossRef]
- Crowell, E.L.; Baker, L.; Chuang, A.Z.; Feldman, R.M.; Bell, N.P.; Chevez-Barrios, P.; Blieden, L.S. Characterizing Anterior Segment Oct Angle Landmarks of the Trabecular Meshwork Complex. Ophthalmology 2018, 125, 994–1002. [Google Scholar] [CrossRef]
- Goodfellow, I.; Bengio, Y.; Courville, A. Deep Learning; MIT Press: Cambridge, MA, USA, 2016. [Google Scholar]
- Gobert, L.; Hiroshi, F. Deep Learning in Medical Image Analysis: Challenges and Applications; Springer: Berlin/Heidelberg, Germany, 2020. [Google Scholar]
- Fu, H.; Baskaran, M.; Xu, Y.; Lin, S.; Wong, D.W.K.; Liu, J.; Tun, T.A.; Mahesh, M.; Perera, S.A.; Aung, T. A Deep Learning System for Automated Angle-Closure Detection in Anterior Segment Optical Coherence Tomography Images. Am. J. Ophthalmol. 2019, 203, 37–45. [Google Scholar] [CrossRef]
- Xu, B.Y.; Chiang, M.; Chaudhary, S.; Kulkarni, S.; Pardeshi, A.A. and Varma, R. Deep Learning Classifiers for Automated Detection of Gonioscopic Angle Closure Based on Anterior Segment Oct Images. Am. J. Ophthalmol. 2019, 208, 273–280. [Google Scholar] [CrossRef]
- Pham, T.H.; Devalla, S.K.; Ang, A.; Soh, Z.D.; Thiery, A.H.; Boote, C.; Cheng, C.Y.; Girard, M.J.A.; Koh, V. Deep Learning Algorithms to Isolate and Quantify the Structures of the Anterior Segment in Optical Coherence Tomography Images. Br. J. Ophthalmol. 2020. [Google Scholar] [CrossRef]
- Xin, C.; Bian, G.B.; Zhang, H.; Liu, W.; Dong, Z. Optical Coherence Tomography-Based Deep Learning Algorithm for Quantification of the Location of the Intraocular Lens. Ann. Transl. Med. 2020, 8, 872. [Google Scholar] [CrossRef]
- Xu, B.Y.; Chiang, M.; Pardeshi, A.A.; Moghimi, S.; Varma, R. Deep Neural Network for Scleral Spur Detection in Anterior Segment Oct Images: The Chinese American Eye Study. Transl. Vis. Sci. Technol. 2020, 9, 18. [Google Scholar] [CrossRef]
- Napier, M.L.; Azuara-Blanco, A. Changing Patterns in Treatment of Angle Closure Glaucoma. Curr. Opin. Ophthalmol. 2018, 29, 130–134. [Google Scholar] [CrossRef]
- Smith, D.S.; Singh, K.; Lin, S.C.; Chen, P.P.; Chen, T.C.; Francis, B.A.; Jampel, H.D. Evaluation of the Anterior Chamber Angle in Glaucoma: A Report by the American Academy of Ophthalmology. Ophthalmology 2013, 120, 1985–1997. [Google Scholar] [CrossRef]
- Saleem, M.S.; Pasquale, L.R.; Sidoti, P.A.; Tsai, J.C. Virtual Ophthalmology: Telemedicine in a Covid-19 Era. Am. J. Ophthalmol. 2020, 216, 237–242. [Google Scholar] [CrossRef]
- Phu, J.; Wang, H.; Khou, V.; Zhang, S.; Kalloniatis, M. Remote Grading of the Anterior Chamber Angle Using Goniophotographs and Optical Coherence Tomography: Implications for Telemedicine or Virtual Clinics. Transl. Vis. Sci. Technol. 2019, 8, 16. [Google Scholar] [CrossRef] [PubMed]
| Parameter | Description | Technology |
|---|---|---|
| Anterior chamber depth (ACD) | Distance between the corneal endothelium and the anterior surface of the lens | OCT |
| Anterior chamber width (ACW) | Distance between the scleral spurs in the nasal and temporal quadrants | OCT |
| Angle opening distance (AOD) | Distance between the trabecular meshwork and the iris at 500 (AOD 500) or 750 μm (AOD 750), anteriorly to the scleral spur | UBM/OCT |
| Angle recess area (ARA) | The triangular area (ARA 500 or 750) bounded by the AOD 500 or 750, the anterior iris surface and the inner corneo-scleral wall | OCT |
| Iris-ciliary process distance (ICPD) | Distance between the iris and the ciliary process along the line of TCPD | UBM |
| Iris thickness 1 (ID1) | Iris thickness at 500 μm anterior to the scleral spur | UBM |
| Iris thickness 2 (ID2) | Iris thickness at 2 mm from the iris roof | UBM |
| Iris thickness 3 (ID3) | Maximum iris thickness near the pupillary edge | UBM |
| Iris-lens angle (ILAθ) | Angle between the iris and the lens near the pupillary edge | UBM |
| Iris-lens contact distance (ILCD) | Contact distance between the iris and the lens | UBM |
| Iris thickness (IT) | Iris thickness measured at 750 um (IT 750) or 2000 μm (IT2000) from the scleral spur | OCT |
| Iris-zonule distance (IZD) | Distance between the iris and the zonule along the line of TCPD | UBM |
| Lens vault (LV) | The perpendicular distance between the anterior pole of the lens and the horizontal line joining the 2 scleral spurs on horizontal AS-OCT scans | OCT |
| Trabecular-ciliary process distance (TCPD) | Distance between the trabecular meshwork and the ciliary process at 500 μm anterior to the scleral spur | UBM |
| Trabecular-iris angle (TIAθ1) | Angle of the angle recess | UBM |
| Trabecular iris space area (TISA) | Trapezoidal area (TISA 500 or 750) bounded by the AOD 500 or 750, the anterior iris surface and the inner corneo-scleral wall | OCT |
| Technique | AUC | 95% CI | Main Ethnicity (%) | Mean Age (SD) (y) |
|---|---|---|---|---|
| Angle closure detection | ||||
| EyeCam [54] | 0.98 | 0.93–1.00 | Chinese (70.4%) | 60.7 (12.6) |
| Manual grading EyeCam [51] | 0.88 | 0.81–0.96 | Chinese (72.9%) | 60.5 (12.9) |
| Automated grading EyeCam [51] | 0.74 | 0.63–0.85 | Chinese (72.9%) | 60.5 (12.9) |
| Visante AS-OCT [54] | 0.85 | 0.76–0.92 | Chinese (70.4%) | 60.7 (12.6) |
| Visante AS-OCT [98] | 0.76 | 0.74–0.78 | Chinese (86.8%) | 60.8 (6.8) |
| CASIA SS-1000 SS-OCT [107] | 0.84 | 0.81–0.88 | Chinese (87.3%) | 61.8 (6.7) |
| Deep Learning algorithm (ResNet-18) [128] | 0.93 | 0.92–0.94 | Chinese (100%) | 61.1 (8.1) |
| Narrow angle detection | ||||
| Ultrasound Biomicroscopy (ARA 750) [37] | 0.97 | 0.92–1.00 | White (58.3%) | 42.9 (n/a) |
| Scheimpflug Photography [84] | 0.93 | 0.90–0.96 | Indian (100%) | 56.2 (6.5) |
| Scanning peripheral ACD analyzer [76] | 0.79 | 0.70–0.87 | Chinese (94.5%) | 65.5 (8.2) |
| Modified Van Herick [76] | 0.87 | 0.80–0.94 | Chinese (94.5%) | 65.5 (8.2) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Riva, I.; Micheletti, E.; Oddone, F.; Bruttini, C.; Montescani, S.; De Angelis, G.; Rovati, L.; Weinreb, R.N.; Quaranta, L. Anterior Chamber Angle Assessment Techniques: A Review. J. Clin. Med. 2020, 9, 3814. https://doi.org/10.3390/jcm9123814
Riva I, Micheletti E, Oddone F, Bruttini C, Montescani S, De Angelis G, Rovati L, Weinreb RN, Quaranta L. Anterior Chamber Angle Assessment Techniques: A Review. Journal of Clinical Medicine. 2020; 9(12):3814. https://doi.org/10.3390/jcm9123814
Chicago/Turabian StyleRiva, Ivano, Eleonora Micheletti, Francesco Oddone, Carlo Bruttini, Silvia Montescani, Giovanni De Angelis, Luigi Rovati, Robert N. Weinreb, and Luciano Quaranta. 2020. "Anterior Chamber Angle Assessment Techniques: A Review" Journal of Clinical Medicine 9, no. 12: 3814. https://doi.org/10.3390/jcm9123814
APA StyleRiva, I., Micheletti, E., Oddone, F., Bruttini, C., Montescani, S., De Angelis, G., Rovati, L., Weinreb, R. N., & Quaranta, L. (2020). Anterior Chamber Angle Assessment Techniques: A Review. Journal of Clinical Medicine, 9(12), 3814. https://doi.org/10.3390/jcm9123814