Patient-Reported Outcomes Following Total Knee Replacement in Patients <65 Years of Age—A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Material and Methods
2.1. Search Strategy
2.2. Inclusion and Exclusion Criteria
2.3. Study Selection
2.4. Data Extraction
2.5. Statistical Analysis
2.6. Assessment of Methodological Quality
2.7. Clinically Meaningful Improvements
3. Results
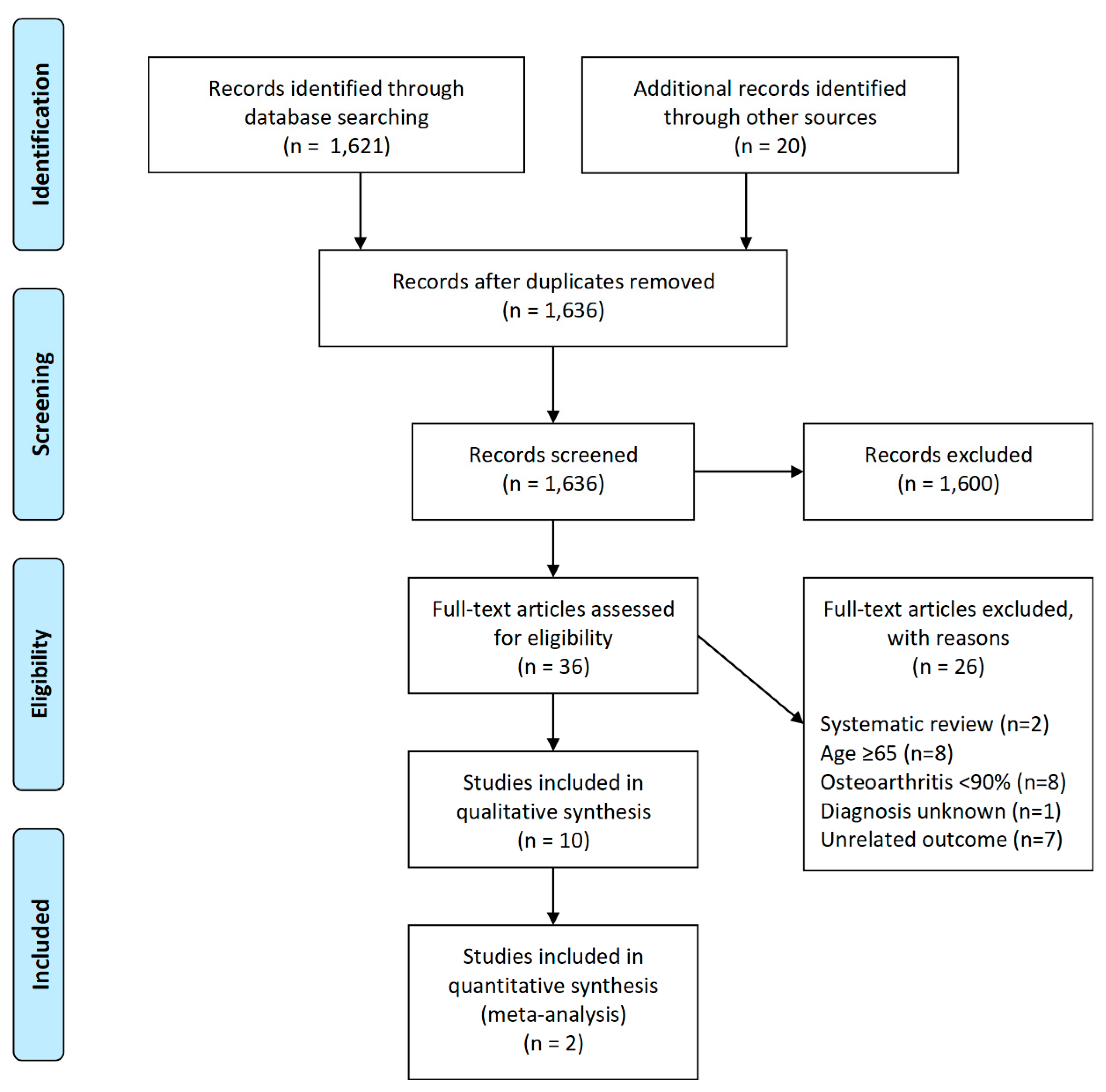
3.1. Search Strategy
3.2. Study, Patient, and Surgery Characteristics
3.3. Disease-Specific Instruments
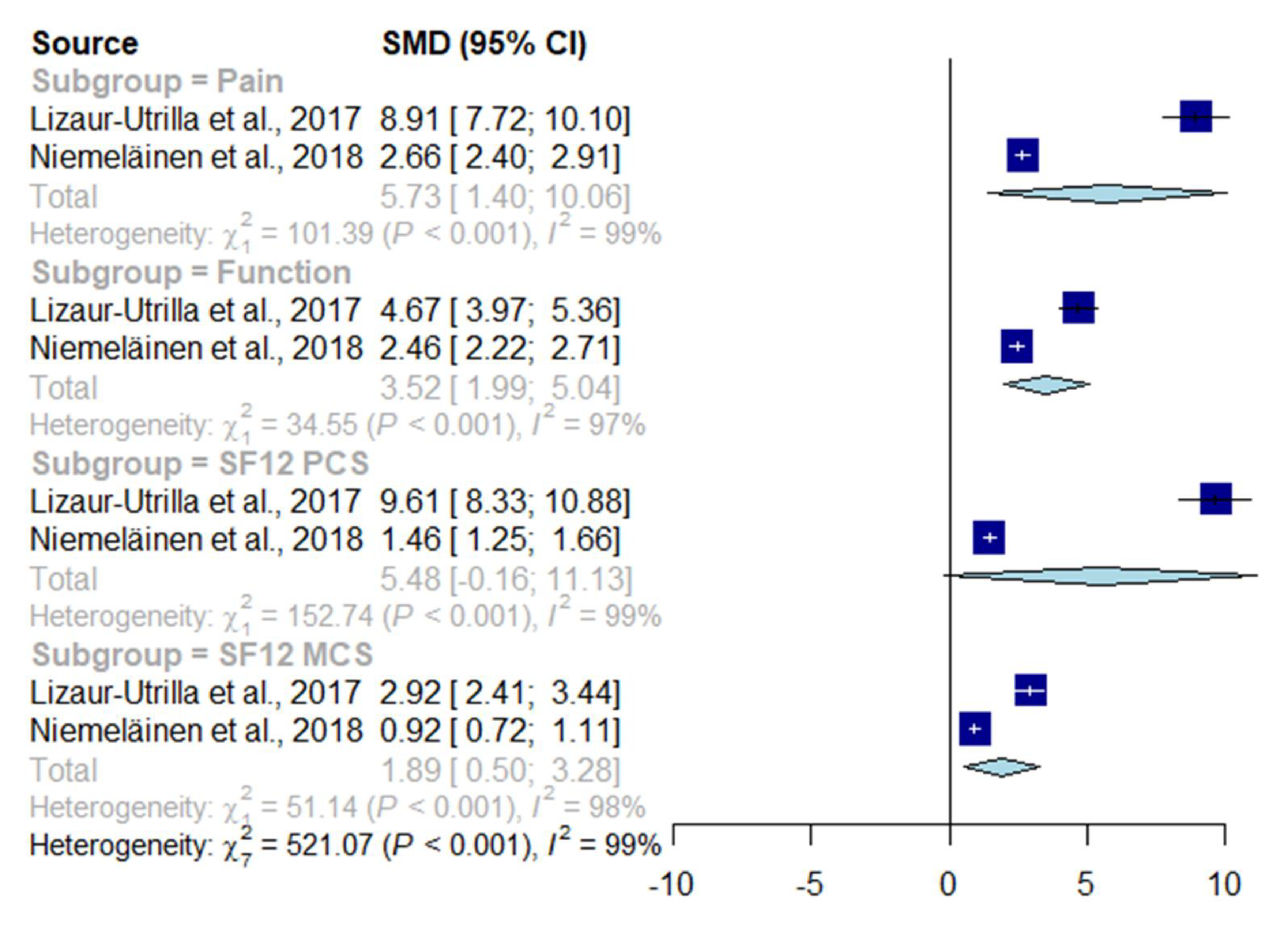
3.4. Generic Health-Related Quality of Life Instruments
3.5. Satisfaction
3.6. Methodological Quality
4. Discussion
4.1. Implications for Practice and Research
4.2. Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Carr, A.J.; Robertsson, O.; Graves, S.; Price, A.J.; Arden, N.K.; Judge, A.; Beard, D.J. Knee replacement. Lancet 2012, 379, 1331–1340. [Google Scholar] [CrossRef]
- Kurtz, S.; Ong, K.; Lau, E.; Mowat, F.; Halpern, M. Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J. Bone Joint Surg. Am. 2007, 89, 780–785. [Google Scholar] [CrossRef] [PubMed]
- Pabinger, C.; Lothaller, H.; Geissler, A. Utilization rates of knee-arthroplasty in OECD countries. Osteoarthr. Cartil. 2015, 23, 1664–1673. [Google Scholar] [CrossRef] [PubMed]
- Ackerman, I.N.; Bohensky, M.A.; Zomer, E.; Tacey, M.; Gorelik, A.; Brand, C.A.; de Steiger, R. The projected burden of primary total knee and hip replacement for osteoarthritis in Australia to the year 2030. BMC Musculoskelet. Disord. 2019, 20, 90. [Google Scholar] [CrossRef]
- Culliford, D.; Maskell, J.; Judge, A.; Cooper, C.; Prieto-Alhambra, D.; Arden, N.K. Future projections of total hip and knee arthroplasty in the UK: Results from the UK Clinical Practice Research Datalink. Osteoarthr. Cartil. 2015, 23, 594–600. [Google Scholar] [CrossRef]
- Mody, B.S.; Mody, K. Arthroplasty in young adults: Options, techniques, trends, and results. Curr. Rev. Musculoskelet. Med. 2014, 7, 131–135. [Google Scholar] [CrossRef]
- Kurtz, S.M.; Lau, E.; Ong, K.; Zhao, K.; Kelly, M.; Bozic, K.J. Future young patient demand for primary and revision joint replacement: National projections from 2010 to 2030. Clin. Orthop. Relat. Res. 2009, 467, 2606–2612. [Google Scholar] [CrossRef]
- Losina, E.; Katz, J.N. Total knee arthroplasty on the rise in younger patients: Are we sure that past performance will guarantee future success? Arthritis Rheum. 2012, 64, 339–341. [Google Scholar] [CrossRef]
- Bayliss, L.E.; Culliford, D.; Monk, A.P.; Glyn-Jones, S.; Prieto-Alhambra, D.; Judge, A.; Cooper, C.; Carr, A.J.; Arden, N.K.; Beard, D.J.; et al. The effect of patient age at intervention on risk of implant revision after total replacement of the hip or knee: A population-based cohort study. Lancet 2017, 389, 1424–1430. [Google Scholar] [CrossRef]
- Wilson, I.; Bohm, E.; Lübbeke, A.; Lyman, S.; Overgaard, S.; Rolfson, O.; W-Dahl, A.; Wilkinson, M.; Dunbar, M. Orthopaedic registries with patient-reported outcome measures. EFORT Open Rev. 2019, 4, 357–367. [Google Scholar] [CrossRef]
- Keeney, J.A.; Eunice, S.; Pashos, G.; Wright, R.W.; Clohisy, J.C. What is the evidence for total knee arthroplasty in young patients?: A systematic review of the literature. Clin. Orthop. Relat. Res. 2011, 469, 574–583. [Google Scholar] [CrossRef] [PubMed]
- Aujla, R.S.; Esler, C.N. Total Knee Arthroplasty for Osteoarthritis in Patients Less Than Fifty-Five Years of Age: A Systematic Review. J. Arthroplast. 2017, 32, 2598–2603.e1. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, S.; Niki, Y.; Harato, K.; Nagura, T.; Nakamura, M.; Matsumoto, M. Rheumatoid Arthritis Patients Achieve Better Satisfaction but Lower Functional Activities as Compared to Osteoarthritis Patients After Total Knee Arthroplasty. J. Arthroplast. 2019, 34, 478.e471–482.e471. [Google Scholar] [CrossRef] [PubMed]
- Ayers, D.C.; Bozic, K.J. The importance of outcome measurement in orthopaedics. Clin. Orthop. Relat. Res. 2013, 471, 3409–3411. [Google Scholar] [CrossRef] [PubMed]
- Losina, E.; Thornhill, T.S.; Rome, B.N.; Wright, J.; Katz, J.N. The dramatic increase in total knee replacement utilization rates in the United States cannot be fully explained by growth in population size and the obesity epidemic. J. Bone Joint Surg. Am. 2012, 94, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. J. Clin. Epidemiol. 2009, 62, e1–e34. [Google Scholar] [CrossRef]
- Singh, J.; Sloan, J.A.; Johanson, N.A. Challenges with health-related quality of life assessment in arthroplasty patients: Problems and solutions. J. Am. Acad. Orthop. Surg. 2010, 18, 72–82. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.J.; Welch, V. Cochrane Handbook for Systematic Reviews of Interventions Version 6.0 (Updated July 2019). Cochrane 2019. Available online: www.training.cochrane.org/handbook (accessed on 1 July 2020).
- Borenstein, M.; Hedges, L.V.; Higgins, J.P.; Rothstein, H.R. A basic introduction to fixed-effect and random-effects models for meta-analysis. Res. Synth. Methods 2010, 1, 97–111. [Google Scholar] [CrossRef]
- Balduzzi, S.; Rücker, G.; Schwarzer, G. How to perform a meta-analysis with R: A practical tutorial. Evid. Based Ment Health 2019, 22, 153–160. [Google Scholar] [CrossRef]
- Sterne, J.A.; Hernan, M.A.; Reeves, B.C.; Savovic, J.; Berkman, N.D.; Viswanathan, M.; Henry, D.; Altman, D.G.; Ansari, M.T.; Boutron, I.; et al. ROBINS-I: A tool for assessing risk of bias in non-randomised studies of interventions. BMJ 2016, 355, i4919. [Google Scholar] [CrossRef]
- Higgins, J.P.; Altman, D.G.; Sterne, J.A.; Group, C.S.M.; Group, C.B.M. Chapter 8: Assessing Risk of Bias in Included Studies. Cochrane Handbook for Systematic Reviews of Interventions. 2011. Available online: www.handbook.cochrane.org (accessed on 1 December 2019).
- Engel, L.; Beaton, D.E.; Touma, Z. Minimal Clinically Important Difference: A Review of Outcome Measure Score Interpretation. Rheum. Dis. Clin. N. Am. 2018, 44, 177–188. [Google Scholar] [CrossRef] [PubMed]
- Jaeschke, R.; Singer, J.; Guyatt, G.H. Measurement of health status. Ascertaining the minimal clinically important difference. Control Clin. Trials 1989, 10, 407–415. [Google Scholar] [CrossRef]
- Maredupaka, S.; Meshram, P.; Chatte, M.; Kim, W.H.; Kim, T.K. Minimal clinically important difference of commonly used patient-reported outcome measures in total knee arthroplasty: Review of terminologies, methods and proposed values. Knee Surg. Relat. Res. 2020, 32, 19. [Google Scholar] [CrossRef] [PubMed]
- Faraone, S.V. Interpreting estimates of treatment effects: Implications for managed care. Pharmacol. Ther. 2008, 33, 700–711. [Google Scholar]
- Lizaur-Utrilla, A.; Gonzalez-Parreno, S.; Martinez-Mendez, D.; Miralles-Munoz, F.A.; Lopez-Prats, F.A. Minimal clinically important differences and substantial clinical benefits for Knee Society Scores. Knee Surg. Sports Traumatol. Arthrosc. 2019, 28, 1473–1478. [Google Scholar] [CrossRef] [PubMed]
- Beard, D.J.; Harris, K.; Dawson, J.; Doll, H.; Murray, D.W.; Carr, A.J.; Price, A.J. Meaningful changes for the Oxford hip and knee scores after joint replacement surgery. J. Clin. Epidemiol. 2015, 68, 73–79. [Google Scholar] [CrossRef]
- Clement, N.D.; Bardgett, M.; Weir, D.; Holland, J.; Gerrand, C.; Deehan, D.J. What is the Minimum Clinically Important Difference for the WOMAC Index After TKA? Clin. Orthop. Relat. Res. 2018, 476, 2005–2014. [Google Scholar] [CrossRef]
- Clement, N.D.; MacDonald, D.; Simpson, A.H.R.W. The minimal clinically important difference in the Oxford knee score and Short Form 12 score after total knee arthroplasty. Knee Surg. Sports Traumatol. Arthrosc. 2014, 22, 1933–1939. [Google Scholar] [CrossRef]
- Monticone, M.; Ferrante, S.; Salvaderi, S.; Motta, L.; Cerri, C. Responsiveness and minimal important changes for the Knee Injury and Osteoarthritis Outcome Score in subjects undergoing rehabilitation after total knee arthroplasty. Am. J. Phys. Med. Rehabil. 2013, 92, 864–870. [Google Scholar] [CrossRef]
- Clement, N.D.; Weir, D.; Holland, J.; Gerrand, C.; Deehan, D.J. Meaningful changes in the Short Form 12 physical and mental summary scores after total knee arthroplasty. Knee 2019, 26, 861–868. [Google Scholar] [CrossRef]
- Lizaur-Utrilla, A.; Martinez-Mendez, D.; Miralles-Munoz, F.A.; Marco-Gomez, L.; Lopez-Prats, F.A. Comparable outcomes after total knee arthroplasty in patients under 55 years than in older patients: A matched prospective study with minimum follow-up of 10 years. Knee Surg. Sports Traumatol. Arthrosc. Off. J. ESSKA 2017, 25, 3396–3402. [Google Scholar] [CrossRef] [PubMed]
- Clement, N.D.; Walker, L.C.; Bardgett, M.; Weir, D.; Holland, J.; Gerrand, C.; Deehan, D.J. Patient age of less than 55 years is not an independent predictor of functional improvement or satisfaction after total knee arthroplasty. Arch. Orthop. Trauma Surg. 2018, 138, 1755–1763. [Google Scholar] [CrossRef] [PubMed]
- Townsend, L.A.; Roubion, R.C.; Bourgeois, D.M.; Leonardi, C.; Fox, R.S.; Dasa, V.; Pollock, G.R. Impact of Age on Patient-Reported Outcome Measures in Total Knee Arthroplasty. J. Knee Surg. 2018, 31, 580–584. [Google Scholar] [CrossRef] [PubMed]
- Garabano, G.; Lopreite, F.; del Sel, H. Total Knee Arthroplasty for gonarthrosis in patients 55 years old or younger. 2-13 years follow-up. Rev. Asoc. Argent. Ortop. Traumatol. 2017, 82, 94–101. [Google Scholar] [CrossRef]
- Long, W.J.; Bryce, C.D.; Hollenbeak, C.S.; Benner, R.W.; Scott, W.N. Total knee replacement in young, active patients: Long-term follow-up and functional outcome: A concise follow-up of a previous report. J. Bone Jt. Surg. Am. Vol. 2014, 96, e159. [Google Scholar] [CrossRef]
- McCalden, R.W.; Robert, C.E.; Howard, J.L.; Naudie, D.D.; McAuley, J.P.; MacDonald, S.J. Comparison of outcomes and survivorship between patients of different age groups following TKA. J. Arthroplast. 2013, 28, 83–86. [Google Scholar] [CrossRef]
- Kim, Y.H.; Kim, J.S.; Choe, J.W.; Kim, H.J. Long-term comparison of fixed-bearing and mobile-bearing total knee replacements in patients younger than fifty-one years of age with osteoarthritis. J Bone Jt. Surg. Am. 2012, 94, 866–873. [Google Scholar] [CrossRef]
- Price, A.J.; Longino, D.; Rees, J.; Rout, R.; Pandit, H.; Javaid, K.; Arden, N.; Cooper, C.; Carr, A.J.; Dodd, C.A.; et al. Are pain and function better measures of outcome than revision rates after TKR in the younger patient? Knee 2010, 17, 196–199. [Google Scholar] [CrossRef]
- Tai, C.C.; Cross, M.J. Five- to 12-year follow-up of a hydroxyapatite-coated, cementless total knee replacement in young, active patients. J. Bone Jt. Surg. Br. Vol. 2006, 88, 1158–1163. [Google Scholar] [CrossRef]
- Niemelainen, M.; Moilanen, T.; Huhtala, H.; Eskelinen, A. Outcome of knee arthroplasty in patients aged 65 years or less: A prospective study of 232 patients with 2-year follow-up. Scand. J. Surg. SJS Off. Organ Finn. Surg. Soc. Scand. Surg. Soc. 2018. [Google Scholar] [CrossRef]
- Kim, Y.H.; Yoon, S.H.; Kim, J.S. The long-term results of simultaneous fixed-bearing and mobile-bearing total knee replacements performed in the same patient. J. Bone Jt. Surg. Br. Vol. 2007, 89, 1317–1323. [Google Scholar] [CrossRef] [PubMed]
- Scott, C.E.; Howie, C.R.; MacDonald, D.; Biant, L.C. Predicting dissatisfaction following total knee replacement: A prospective study of 1217 patients. J. Bone Jt. Surg. Br. Vol. 2010, 92, 1253–1258. [Google Scholar] [CrossRef] [PubMed]
- Scott, C.E.H.; Oliver, W.M.; MacDonald, D.; Wade, F.A.; Moran, M.; Breusch, S.J. Predicting dissatisfaction following total knee arthroplasty in patients under 55 years of age. Bone Jt. J. 2016, 98-B, 1625–1634. [Google Scholar] [CrossRef] [PubMed]
- Bourne, R.B.; Chesworth, B.M.; Davis, A.M.; Mahomed, N.N.; Charron, K.D. Patient satisfaction after total knee arthroplasty: Who is satisfied and who is not? Clin. Orthop. Relat. Res. 2010, 468, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Prodinger, B.; Taylor, P. Improving quality of care through patient-reported outcome measures (PROMs): Expert interviews using the NHS PROMs Programme and the Swedish quality registers for knee and hip arthroplasty as examples. BMC Health Serv. Res. 2018, 18, 87. [Google Scholar] [CrossRef]
- Devlin, N.J.; Appleby, J. Getting the most out of PROMs: Putting health outcomes at the heart of NHS decision-making. In The King’s Fund, Office of Health Economics; The King’s Fund: London, UK, 2010. [Google Scholar]
- Association, A.O. Hip, Knee & Shoulder Arthroplasty. In Annual Report; Australian Orthopaedic Association National Joint Replacement Registry: Adelaide, Australia, 2019. [Google Scholar]
- Sun, H.; Zhou, L.; Li, F.; Duan, J. Comparison between Closing-Wedge and Opening-Wedge High Tibial Osteotomy in Patients with Medial Knee Osteoarthritis: A Systematic Review and Meta-analysis. J. Knee Surg. 2017, 30, 158–165. [Google Scholar] [CrossRef]
- Dowsey, M.M.; Smith, A.J.; Choong, P.F.M. Latent Class Growth Analysis predicts long term pain and function trajectories in total knee arthroplasty: A study of 689 patients. Osteoarthr. Cartil. 2015, 23, 2141–2149. [Google Scholar] [CrossRef]
- Tew, M.; Dalziel, K.; Clarke, P.; Smith, A.; Choong, P.F.; Dowsey, M. Patient-reported outcome measures (PROMs): Can they be used to guide patient-centered care and optimize outcomes in total knee replacement? Qual. Life Res. 2020. [Google Scholar] [CrossRef]
- Williams, K.; Sansoni, J.; Morris, D.; Grootemaat, P.; Thompson, C. Patient-reported outcome measures: Literature review. Sydney ACSQHC 2016. [Google Scholar]
- de Steiger, R.N.; Graves, S.E. Orthopaedic registries: The Australian experience. EFORT Open Rev. 2019, 4, 409–415. [Google Scholar] [CrossRef]
- Jiang, Y.; Sanchez-Santos, M.T.; Judge, A.D.; Murray, D.W.; Arden, N.K. Predictors of Patient-Reported Pain and Functional Outcomes Over 10 Years After Primary Total Knee Arthroplasty: A Prospective Cohort Study. J. Arthroplast. 2017, 32, 92–100.e2. [Google Scholar] [CrossRef] [PubMed]
- Judge, A.; Arden, N.K.; Cooper, C.; Kassim Javaid, M.; Carr, A.J.; Field, R.E.; Dieppe, P.A. Predictors of outcomes of total knee replacement surgery. Rheumatology 2012, 51, 1804–1813. [Google Scholar] [CrossRef] [PubMed]
| Outcomes | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Study | Pre-Operative | Post-Operative | Change | Minimal Clinically Important Difference | |||||
| Mean | SD | Mean | SD | Difference | SD | MCID (95% CI) | Method | Reference | |
| Knee Society Score (KSS) | |||||||||
| McCalden et al. | N/A | N/A | N/A | N/A | 78.9 | N/A | N/A | N/A | N/A |
| Tai et al. | 49.0 | N/A | 86.5 | N/A | 37.5 | N/A | |||
| KSS (Knee) | |||||||||
| Garabano et al. | 46.8 | N/A | 91.4 | N/A | 44.6 | N/A | 7.2 (5.1–7.8) 7.2 (5.3–9.0) | Anchor Distribution | Lizaur-Utrilla et al., 2019 [27] |
| Kim et al. | 25.5 | N/A | 94.5 | N/A | 69.0 | N/A | |||
| Lizaur-Utrilla et al. | 34.7 | 11.8 | 87.4 | 8.1 | 52.7 | N/A | |||
| Long et al. | N/A | N/A | 87.4 | 17.6 | N/A | N/A | |||
| Price at al. | N/A | N/A | 74.8 | N/A | N/A | N/A | |||
| Tai et al. | 57.0 | N/A | 85.0 | N/A | 28.0 | N/A | |||
| KSS (Function) | |||||||||
| Garabano et al. | 49.3 | N/A | 92.6 | N/A | 43.3 | N/A | 9.7 (7.3–10.2) 6.3 (5.0–8.1) | Anchor Distribution | Lizaur-Utrilla et al., 2019 |
| Kim et al. | 30.0 | N/A | 84.5 | N/A | 54.5 | N/A | |||
| Lizaur-Utrilla et al. | 38.1 | 17.0 | 86.3 | 11.4 | 48.2 | N/A | |||
| Long et al. | N/A | N/A | 62.1 | 32.2 | N/A | N/A | |||
| Price et al. | N/A | N/A | 56.8 | N/A | N/A | N/A | |||
| Tai et al. | 43.0 | N/A | 90.0 | N/A | 47.0 | N/A | |||
| Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) | |||||||||
| Clement et al. | 31.7 | 16.1 | 69.1 | 24.0 | 37.5 | 15.3 | 10 | Anchor | Clement et al., 2018 |
| Kim et al. | 29.2 | N/A | 63.5 | N/A | 34.3 | N/A | |||
| McCalden et al. | N/A | N/A | N/A | N/A | 33.3 | N/A | |||
| Townsend et al. | 40.3 | N/A | 54.1 | N/A | 13.8 | N/A | |||
| WOMAC (Pain) | |||||||||
| Clement et al. | 28.8 | 16.2 | 74.6 | 24.4 | 45.8 | 17.5 | 11 | Anchor | Clement et al., 2018 |
| Garabano et al. | N/A | N/A | 88.5 | N/A | N/A | N/A | |||
| Kim et al. | 45.0 | N/A | 88.5 | N/A | 43.5 | N/A | |||
| Lizaur-Utrilla et al. | 39.4 | 6.3 | 88.5 | 4.5 | 49.1 | N/A | |||
| WOMAC (Function) | |||||||||
| Clement et al. | 32.7 | 17.1 | 68.0 | 25.0 | 35.3 | 15.6 | 9 | Anchor | Clement et al., 2018 |
| Garabano et al. | N/A | N/A | 59.9 | N/A | N/A | N/A | |||
| Kim et al. | 24.3 | N/A | 56.0 | N/A | 31.7 | N/A | |||
| Lizaur-Utrilla et al. | 49.2 | 8.1 | 83.9 | 6.6 | 34.7 | N/A | |||
| WOMAC (Stiffness) | |||||||||
| Clement et al. | 30.0 | 20.6 | 65.5 | 24.8 | 35.2 | 19.1 | 8 | Anchor | Clement et al., 2018 |
| Garabano et al. | N/A | N/A | 67.5 | N/A | N/A | N/A | |||
| Kim et al. | 31.9 | N/A | 65.6 | N/A | 33.7 | N/A | |||
| Oxford Knee Score (OKS) | |||||||||
| Niemeläinen et al. | 45.8 | 16.0 | 85.4 | 16.0 | 39.6 | N/A | 19.2 | Anchor | Beard et al., 2015 [28] |
| Price at al. | N/A | N/A | 64.4 | N/A | N/A | N/A | |||
| Townsend et. al. | 30.0 | N/A | 44.4 | N/A | 14.4 | N/A | |||
| OKS (Pain) | |||||||||
| Townsend et al. | 25.6 | N/A | 40.1 | N/A | 14.5 | N/A | 25 (22–27.5) | Anchor | Clement et al., 2014 [29] |
| OKS (Function) | |||||||||
| Townsend et al. | 32.4 | N/A | 47.5 | N/A | 15.2 | N/A | 15.4 (13.6–17.1) | Anchor | Clement et al., 2014 [30] |
| Hospital for Special Surgery Knee Score (HSS) | |||||||||
| Kim et al. | 48.0 | N/A | 91.5 | N/A | 43.5 | N/A | N/A | N/A | N/A |
| Long et al. | 57.9 | 10.3 | 85.3 | 13.2 | 27.4 | N/A | |||
| High Activity Arthroplasty Score (HAAS) | |||||||||
| Niemeläinen et al. | 33.3 | 21.1 | 61.1 | 21.1 | 27.8 | N/A | N/A | N/A | N/A |
| Knee Injury and Osteoarthritis Outcome Score (KOOS) Pain | |||||||||
| Niemeläinen et al. | 45.0 | 15.4 | 86.0 | 15.4 | 41.0 | N/A | 16.7 | Anchor | Monticone et al., 2013 [31] |
| Knee Injury and Osteoarthritis Outcome Score (KOOS) Symptoms | |||||||||
| Niemeläinen et al. | 43.0 | 19.2 | 79.0 | 19.2 | 36.0 | N/A | 10.7 | Anchor | Monticone et al., 2013 |
| Knee Injury and Osteoarthritis Outcome Score (KOOS) QOL | |||||||||
| Niemeläinen et al. | 21.0 | 15.4 | 70.0 | 23.1 | 49.0 | N/A | 15.6 | Anchor | Monticone et al., 2013 |
| Knee Injury and Osteoarthritis Outcome Score (KOOS) ADLs function | |||||||||
| Niemeläinen et al. | 50.0 | 15.4 | 88.0 | 15.4 | 38.0 | N/A | 18.4 | Anchor | Monticone et al., 2013 |
| Knee Injury and Osteoarthritis Outcome Score (KOOS) Sport/recreation function | |||||||||
| Niemeläinen et al. | 14.0 | 19.2 | 55.0 | 30.7 | 41.0 | N/A | 12.5 | Anchor | Monticone et al., 2013 |
| Short Form-12 Physical Component Score (SF-12 PCS) | |||||||||
| Clement et al. | 26.9 | 7.0 | 38.1 | 12.2 | 11.2 | 7.8 | 1.8 (0.1–3.5) | Anchor | Clement et al., 2019 [32] |
| Lizaur-Utrilla et al. | 20.1 | 6.0 | 82.6 | 6.9 | N/A | N/A | |||
| Short Form-12 Mental Component Score (SF-12 MCS) | |||||||||
| Clement et al. | 41.6 | 13.9 | 44.5 | 14.7 | 2.9 | 9.4 | Nil significant | Anchor | Clement et al., 2019 |
| Lizaur-Utrilla et al. | 47.2 | 10.4 | 74.6 | 8.1 | N/A | N/A | |||
| Research and Development-36 Physical Component Score (RAND-36 PCS) | |||||||||
| Niemeläinen et al. | 37.0 | 19.2 | 68.0 | 23.1 | 31.0 | N/A | N/A | N/A | N/A |
| Research and Development-36 Mental Component Score (RAND-36 MCS) | |||||||||
| Niemeläinen et al. | 61.0 | 23.1 | 79.0 | 15.4 | 18.0 | N/A | N/A | N/A | N/A |
| Study Characteristics | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Study | Design | TKRs | Prosthesis | Patellar Resurfacing | Age | Osteoarthritis (%) | Surgery Period | Follow-Up (Range) | Follow-Up % | Disease-Specific Instruments | Generic Health Instruments | Satisfaction Instruments | Difference in Outcomes Compared to Older Patients | Summary of Statistical Significance | Summary of Clinical Significance |
| Clement et al., 2018 | Retrospective | 224 | N/A | N/A | <55 | 100 | 2003–2013 | 1 year | N/A | WOMAC | SF-12 (PCS & MCS) | Four-point Likert scale | WOMAC total: −0.8 (p = 0.57) WOMAC pain: −1.2 (p = 0.46) WOMAC function: −1.6 (p = 0.29) SF-12 PCS: 1.3 (p = 0.06) SF-12 MCS: 0.8 (p = 0.37) Satisfaction: 83.4% (<55 years) vs. 92.0% (≥55 years) (p = 0.001) no statistical difference for above scores following regression adjustment for patient demographics and pre-operative scores | WOMAC total: equivalent WOMAC pain: equivalent WOMAC function: equivalent SF-12 PCS: equivalent SF-12 MCS: equivalent Satisfaction: inferior satisfaction considered equivalent by authors after attribution to higher prevalence of mental health disorders in younger age group | No difference as did not exceed MCID |
| Niemeläinen et al., 2018 | Prospective | 227 | CR 96%, PS 4% | 12/227 | Mean 58 (<65) | 100 | 2012–2014 | 2 years (22 to 26 months) | 93% | OKS, HAAS, KOOS | RAND-36 (PCS & MCS) | 100-point Visual Analogue Scale | N/A | N/A | N/A |
| Lizaur-Utrilla et al., 2017 | Prospective | 61 | CR 100% | 61/61 | Median 53 (30–55) | 100 | 2001–2005 | 12 (10 to 14) years | 100% | KSS, WOMAC | SF-12 (PCS & MCS) | Satisfaction assessed but instrument not specified | Five year follow-up KSS knee: −0.6 (p > 0.05) KSS function: −0.2 (p > 0.05) WOMAC pain: −3.3 (p > 0.05) WOMAC function: 4.0 (p = 0.001) SF-12 PCS: 0.5 (p > 0.05) SF-12 MCS: −0.6 (p > 0.05) Latest follow-up KSS knee: 2.5 (p > 0.05) KSS function: 5.2 (p = 0.018) WOMAC pain: −3.1 (p > 0.05) WOMAC function: 2.7 (p = 0.028) SF-12 PCS: 2.4 (p = 0.001) SF-12 MCS: 2.4 (p = 0.035) Satisfaction: 90.2% (<55 years) vs. 87.9% (≥55 years) (p > 0.05) | Five year follow-up KSS knee: equivalent KSS function: equivalent WOMAC pain: equivalent WOMAC function: superior SF-12 PCS: equivalent SF-12 MCS: equivalent Latest follow-up KSS knee: equivalent KSS function: superior WOMAC pain: equivalent WOMAC function: superior SF-12 PCS: superior SF-12 MCS: superior Satisfaction: equivalent | Difference in KSS function within 95% CI of MCID by distribution method Difference in SF-12 PCS and MCS within 95% CI of MCID by anchor method |
| Townsend et. al., 2017 | Retrospective | 100 | N/A | N/A | <60 | 100 | 2008–2014 | 6 months | N/A | WOMAC, OKS | N/A | N/A | WOMAC total: −3.3 (p = 0.0007) OKS total: −0.8 (p < 0.0001) outcomes are weighted by sample size comparing patients <60 years vs. patients ≥70 years; one subgroup was age 60–69 years which was excluded from comparison, p values are after adjustment for baseline scores by authors | WOMAC total: inferior OKS total: inferior | No difference as did not exceed MCID |
| Garabano et al., 2016 | Retrospective | 53 | PS 100% | 40/53 | Mean 49 (26–54) | 96 | 1997–2011 | 6.5 (2 to 15) years | N/A | KSS, WOMAC | N/A | Satisfaction assessed but instrument not specified | N/A | N/A | N/A |
| Long et al., 2014 | Prospective | 38 | PS 100% | N/A | Mean 51 (22–55) | 100 | 1977–1992 | 25.1 (20 to 35) years | 95% | KSS, HSS | N/A | N/A | N/A | N/A | N/A |
| McCalden et al., 2013 | Retrospective | 673 | CR 12%, PS 88% | 660/673 | Mean 50 (<55) | 90 | 1996–2009 | 2+ years | N/A | KSS, WOMAC | SF-12 (PCS & MCS) | N/A | KSS total: 9.9 (p < 0.001) KSS knee: greater improvement but value N/A (p < 0.001) KSS function: greater improvement but value N/A (p < 0.001) WOMAC total: 6.0 (p < 0.001) WOMAC pain: greater improvement but value N/A (p < 0.001) WOMAC function: greater improvement but value N/A (p < 0.001) WOMAC stiffness: greater improvement but value N/A (p < 0.001) SF-12 PCS: no significant difference (p > 0.05) SF-12 MCS: no significant difference (p > 0.05) comparing patients ≤55 years vs. patients >70 years; one subgroup was age 55–70 years which was excluded from comparison | KSS total: superior KSS knee: superior KSS function: superior WOMAC total: superior WOMAC pain: superior WOMAC function: superior WOMAC stiffness: superior SF-12 PCS: equivalent SF-12 MCS: equivalent | Values or MCID unavailable to enable determination |
| Kim et al., 2012 | Prospective | 216 | CR 50%, PS 50% | N/A | Mean 45 (<51) | 100 | 1993–1996 | 16.8 (15 to 18) years | 95% | KSS, WOMAC, HSS | N/A | N/A | N/A | N/A | N/A |
| Price at al., 2010 | Prospective | 37 | CR 100% | 5/37 | Mean 55.4 (32–59.5) | 100 | 1987–1993 | 15.7 (12 to 19) years | 60% | KSS, OKS | N/A | N/A | N/A | N/A | N/A |
| Tai et al., 2006 | Prospective | 118 | CR 100% | 43/118 | Mean 50.7 (32–55) | 100 | 1992–2000 | 7.9 (5 to 12.5) years | 99% | KSS | N/A | N/A | N/A | N/A | N/A |
| Satisfaction | ||||||
|---|---|---|---|---|---|---|
| Authors | TKRs | Satisfaction | Domain | Instrument | Criteria | Follow-Up |
| Clement et al. | 224 | 83.4% 85.1% 80.1% 72.4% | Overall Pain Work Recreation | Four-point Likert scale: Very satisfied Somewhat satisfied Very dissatisfied Somewhat dissatisfied | Very satisfied Somewhat satisfied | 1 year |
| Niemeläinen et al. | 227 | 81% at 1 year 84% at 2 years | Overall | Visual Analogue Scale: Very satisfied (76–100 points) Satisfied (51–75 points) Unsure (26 to 50 points) Dissatisfied (0–25 points) | Very satisfied (76–100 points) Satisfied (51–75 points) | 1 year 2 years |
| Garabano et al. | 53 | 93.5% | Overall | N/A | N/A | 6.5 years |
| Lizaur-Utrilla et al. | 61 | 90.2% | Overall | N/A | N/A | 10 years |
| Study | Confounding | Selection of Participants | Classification of Interventions | Missing Data | Outcome Measurement | Selective Reporting | Overall Risk of Bias |
|---|---|---|---|---|---|---|---|
| Clement et al., 2018 | |||||||
| Niemeläinen et al., 2018 | |||||||
| Lizaur-Utrilla et al., 2017 | |||||||
| Townsend et. al., 2017 | |||||||
| Garabano et al., 2016 | |||||||
| Long et al., 2014 | |||||||
| McCalden et al., 2013 | |||||||
| Kim et al., 2012 | |||||||
| Price at al., 2010 | |||||||
| Tai et al., 2006 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Trieu, J.; Gould, D.J.; Schilling, C.; Spelman, T.; Dowsey, M.M.; Choong, P.F. Patient-Reported Outcomes Following Total Knee Replacement in Patients <65 Years of Age—A Systematic Review and Meta-Analysis. J. Clin. Med. 2020, 9, 3150. https://doi.org/10.3390/jcm9103150
Trieu J, Gould DJ, Schilling C, Spelman T, Dowsey MM, Choong PF. Patient-Reported Outcomes Following Total Knee Replacement in Patients <65 Years of Age—A Systematic Review and Meta-Analysis. Journal of Clinical Medicine. 2020; 9(10):3150. https://doi.org/10.3390/jcm9103150
Chicago/Turabian StyleTrieu, Jason, Daniel J. Gould, Chris Schilling, Tim Spelman, Michelle M. Dowsey, and Peter F. Choong. 2020. "Patient-Reported Outcomes Following Total Knee Replacement in Patients <65 Years of Age—A Systematic Review and Meta-Analysis" Journal of Clinical Medicine 9, no. 10: 3150. https://doi.org/10.3390/jcm9103150
APA StyleTrieu, J., Gould, D. J., Schilling, C., Spelman, T., Dowsey, M. M., & Choong, P. F. (2020). Patient-Reported Outcomes Following Total Knee Replacement in Patients <65 Years of Age—A Systematic Review and Meta-Analysis. Journal of Clinical Medicine, 9(10), 3150. https://doi.org/10.3390/jcm9103150





