Essentials of Insulinoma Localization with Selective Arterial Calcium Stimulation and Hepatic Venous Sampling
Abstract
1. Introduction
2. Non-Invasive Pre-Operative Localization
3. Invasive Pre-Operative Localization
4. Intraoperative Localization
5. Selective Arterial Calcium Stimulation and Hepatic Venous Sampling
6. Pancreatic Vascular Anatomy
7. Pre-Procedural Workup
8. Procedural Description
9. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Service, F.J.; McMahon, M.M.; O’Brien, P.C.; Ballard, D.J. Functioning insulinoma--incidence, recurrence, and long-term survival of patients: A 60-year study. Mayo Clin. Proc. 1991, 66, 711–719. [Google Scholar] [CrossRef]
- Grant, C.S. Insulinoma. Best Pract. Res. Clin. Gastroenterol. 2005, 19, 783–798. [Google Scholar] [CrossRef] [PubMed]
- Service, F.J. Hypoglycemic disorders. N. Engl. J. Med. 1995, 332, 1144–1152. [Google Scholar] [CrossRef] [PubMed]
- Cryer, P.E. Mechanisms of hypoglycemia-associated autonomic failure and its component syndromes in diabetes. Diabetes 2005, 54, 3592–3601. [Google Scholar] [CrossRef] [PubMed]
- Thomspon, G.B.; Service, F.J. Insulinoma. In Common Surgical Diseases: An Algorithmic Approach to Problem Solving, 2nd ed.; Saclarides, T.J., Millikan, K.W., Godellas, C.V., Eds.; Springer: New York, NY, USA, 2008. [Google Scholar]
- Rostambeigi, N.; Thompson, G.B. What should be done in an operating room when an insulinoma cannot be found? Clin. Endocrinol. 2009, 70, 512–515. [Google Scholar] [CrossRef]
- Okabayashi, T.; Shima, Y.; Sumiyoshi, T.; Kozuki, A.; Ito, S.; Ogawa, Y.; Kobayashi, M.; Hanazaki, K. Diagnosis and management of insulinoma. World J. Gastroenterol. 2013, 19, 829–837. [Google Scholar] [CrossRef]
- Service, F.J.; Natt, N. The prolonged fast. J. Clin. Endocrinol. Metab. 2000, 85, 3973–3974. [Google Scholar] [CrossRef]
- Hirshberg, B.; Livi, A.; Bartlett, D.L.; Libutti, S.K.; Alexander, H.R.; Doppman, J.L.; Skarulis, M.C.; Gorden, P. Forty-eight-hour fast: The diagnostic test for insulinoma. J. Clin. Endocrinol. Metab. 2000, 85, 3222–3226. [Google Scholar] [CrossRef]
- Vezzosi, D.; Bennet, A.; Fauvel, J.; Caron, P. Insulin, C-peptide and proinsulin for the biochemical diagnosis of hypoglycaemia related to endogenous hyperinsulinism. Eur. J. Endocrinol. 2007, 157, 75–83. [Google Scholar] [CrossRef]
- Crippa, S.; Zerbi, A.; Boninsegna, L.; Capitanio, V.; Partelli, S.; Balzano, G.; Pederzoli, P.; Di Carlo, V.; Falconi, M. Surgical management of insulinomas: Short-and long-term outcomes after enucleations and pancreatic resections. Arch. Surg. 2012, 147, 261–266. [Google Scholar] [CrossRef]
- Mele, C.; Brunani, A.; Damascelli, B.; Ticha, V.; Castello, L.; Aimaretti, G.; Scacchi, M.; Marzullo, P. Non-surgical ablative therapies for inoperable benign insulinoma. J. Endocrinol. Investig. 2018, 41, 153–162. [Google Scholar] [CrossRef] [PubMed]
- Chavan, A.; Kirchhoff, T.D.; Brabant, G.; Scheumann, G.F.; Wagner, S.; Galanski, M. Role of the intra-arterial calcium stimulation test in the preoperative localization of insulinomas. Eur. Radiol. 2000, 10, 1582–1586. [Google Scholar] [CrossRef] [PubMed]
- Lo, C.Y.; Chan, F.L.; Tam, S.C.; Cheng, P.W.; Fan, S.T.; Lam, K.S. Value of intra-arterial calcium stimulated venous sampling for regionalization of pancreatic insulinomas. Surgery 2000, 128, 903–909. [Google Scholar] [CrossRef] [PubMed]
- Guettier, J.M.; Kam, A.; Chang, R.; Skarulis, M.C.; Cochran, C.; Alexander, H.R.; Libutti, S.K.; Pingpank, J.F.; Gorden, P. Localization of insulinomas to regions of the pancreas by intraarterial calcium stimulation: The NIH experience. J. Clin. Endocrinol. Metab. 2009, 94, 1074–1080. [Google Scholar]
- Goh, B.K.; Ooi, L.L.; Cheow, P.C.; Tan, Y.M.; Ong, H.S.; Chung, Y.F.; Chow, P.K.; Wong, W.K.; Soo, K.C. Accurate preoperative localization of insulinomas avoids the need for blind resection and reoperation: Analysis of a single institution experience with 17 surgically treated tumors over 19 years. J. Gastrointest Surg. 2009, 13, 1071–1077. [Google Scholar] [CrossRef] [PubMed]
- Ravi, K.; Britton, B.J. Surgical approach to insulinomas: Are pre-operative localisation tests necessary? Ann. R. Coll. Surg. Engl. 2007, 89, 212–217. [Google Scholar] [CrossRef]
- McAuley, G.; Delaney, H.; Colville, J.; Lyburn, I.; Worsley, D.; Govender, P.; Torreggiani, W.C. Multimodality preoperative imaging of pancreatic insulinomas. Clin. Radiol. 2005, 60, 1039–1450. [Google Scholar] [CrossRef]
- Gorman, B.; Charboneau, J.W.; James, E.M.; Reading, C.C.; Galiber, A.K.; Grant, C.S.; van Heerden, J.A.; Telander, R.L.; Service, F.J. Benign pancreatic insulinoma: Preoperative and intraoperative sonographic localization. Am. J. Roentgenol. 1986, 147, 929–934. [Google Scholar] [CrossRef]
- Galiber, A.K.; Reading, C.C.; Charboneau, J.W.; Sheedy, P.F., 2nd; James, E.M.; Gorman, B.; Grant, C.S.; van Heerden, J.A.; Telander, R.L. Localization of pancreatic insulinoma: Comparison of pre- and intraoperative US with CT and angiography. Radiology 1988, 166, 405–408. [Google Scholar] [CrossRef]
- Doppman, J.L.; Chang, R.; Fraker, D.L.; Norton, J.A.; Alexander, H.R.; Miller, D.L.; Collier, E.; Skarulis, M.C.; Gorden, P. Localization of insulinomas to regions of the pancreas by intra-arterial stimulation with calcium. Ann. Intern. Med. 1995, 123, 269–273. [Google Scholar] [CrossRef]
- Fidler, J.L.; Fletcher, J.G.; Reading, C.C.; Andrews, J.C.; Thompson, G.B.; Grant, C.S.; Service, F.J. Preoperative Detection of Pancreatic Insulinomas on Multiphasic Helical CT. Am. J. Roentgenol. 2003, 181, 775–780. [Google Scholar] [CrossRef] [PubMed]
- Stark, D.D.; Moss, A.A.; Goldberg, H.I.; Deveney, C.W. CT of pancreatic islet cell tumors. Radiology 1984, 150, 491–494. [Google Scholar] [CrossRef]
- Gouya, H.; Vignaux, O.; Augui, J.; Dousset, B.; Palazzo, L.; Louvel, A.; Chaussade, S.; Legmann, P. CT, endoscopic sonography, and a combined protocol for preoperative evaluation of pancreatic insulinomas. Am. J. Roentgenol. 2003, 181, 987–992. [Google Scholar] [CrossRef] [PubMed]
- Thoeni, R.F.; Mueller-Lisse, U.G.; Chan, R.; Do, N.K.; Shyn, P.B. Detection of small, functional islet cell tumors in the pancreas: Selection of MR imaging sequences for optimal sensitivity. Radiology 2000, 214, 483–490. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Xue, H.; Sun, Z.; Li, P.; Qian, T.; Xing, X.; Li, N.; Zhao, Y.; Wu, W.; Jin, Z. Prospective comparison of biphasic contrast-enhanced CT, volume perfusion CT, and 3 Tesla MRI with diffusion-weighted imaging for insulinoma detection. J. Magn. Reson. Imaging 2017, 46, 1648–1655. [Google Scholar] [CrossRef]
- Briganti, V.; Matteini, M.; Ferri, P.; Vaggelli, L.; Castagnoli, A.; Pieroni, C. Octreoscan SPET Evaluation in the Diagnosis of Pancreas Neuroendocrine Tumors. Cancer Biother. Radiopharm. 2001, 16, 515–524. [Google Scholar] [CrossRef]
- Chiti, A.; Fanti, S.; Savelli, G.; Romeo, A.; Bellanova, B.; Rodari, M.; van Graafeiland, B.J.; Monetti, N.; Bombardieri, E. Comparison of somatostatin receptor imaging, computed tomography and ultrasound in the clinical management of neuroendocrine gastro-entero-pancreatic tumours. Eur. J. Nucl. Med. 1998, 25, 1396–1403. [Google Scholar] [CrossRef] [PubMed]
- Nockel, P.; Babic, B.; Millo, C.; Herscovitch, P.; Patel, D.; Nilubol, N.; Sadowski, S.M.; Cochran, C.; Gorden, P.; Kebebew, E. Localization of Insulinoma Using 68Ga-DOTATATE PET/CT Scan. J. Clin. Endocrinol. Metab. 2017, 102, 195–199. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Arora, S.; Karunanithi, S.; Khadgawat, R.; Durgapal, P.; Sharma, R.; Kandasamy, D.; Bal, C.; Kumar, R. Somatostatin receptor based PET/CT imaging with 68Ga-DOTA-Nal3-octreotide for localization of clinically and biochemically suspected insulinoma. Q. J. Nucl. Med. Mol. Imaging 2016, 60, 69–76. [Google Scholar] [PubMed]
- Sotoudehmanesh, R.; Hedayat, A.; Shirazian, N.; Shahraeeni, S.; Ainechi, S.; Zeinali, F.; Kolahdoozan, S. Endoscopic ultrasonography (EUS) in the localization of insulinoma. Endocrine 2007, 31, 238–241. [Google Scholar] [CrossRef]
- Tseng, L.M.; Chen, J.Y.; Won, J.G.; Tseng, H.S.; Yang, A.H.; Wang, S.E.; Lee, C.H. The role of intra-arterial calcium stimulation test with hepatic venous sampling (IACS) in the management of occult insulinomas. Ann. Surg. Oncol. 2007, 14, 2121–2127. [Google Scholar] [CrossRef]
- Gimm, O.; Konig, E.; Thanh, P.N.; Brauckhoff, M.; Karges, W.; Dralle, H. Intra-operative quick insulin assay to confirm complete resection of insulinomas guided by selective arterial calcium injection (SACI). Langenbecks Arch. Surg. 2007, 392, 679–684. [Google Scholar] [CrossRef]
- Balci, N.C.; Semelka, R.C. Radiologic features of cystic, endocrine and other pancreatic neoplasms. Eur. J. Radiol. 2001, 38, 113–119. [Google Scholar] [CrossRef]
- Anaye, A.; Mathieu, A.; Closset, J.; Bali, M.A.; Metens, T.; Matos, C. Successful preoperative localization of a small pancreatic insulinoma by diffusion-weighted MRI. J. Pancreas 2009, 10, 528–531. [Google Scholar]
- Mirallie, E.; Pattou, F.; Malvaux, P.; Filoche, B.; Godchaux, J.M.; Maunoury, V.; Palazzo, L.; Lefebvre, J.; Huglo, D.; Paris, J.C.; et al. Value of endoscopic ultrasonography and somatostatin receptor scintigraphy in the preoperative localization of insulinomas and gastrinomas. Experience of 54 cases. Gastroenterol. Clin. Biol. 2002, 26, 360–366. [Google Scholar]
- Dromain, C.; Deandreis, D.; Scoazec, J.Y.; Goere, D.; Ducreux, M.; Baudin, E.; Tselikas, L. Imaging of neuroendocrine tumors of the pancreas. Diagn. Interv. Imaging 2016, 97, 1241–1257. [Google Scholar] [CrossRef]
- Reubi, J.C.; Waser, B. Concomitant expression of several peptide receptors in neuroendocrine tumours: Molecular basis for in vivo multireceptor tumour targeting. Eur. J. Nucl. Med. Mol. Imaging 2003, 30, 781–793. [Google Scholar] [CrossRef]
- Antwi, K.; Fani, M.; Heye, T.; Nicolas, G.; Rottenburger, C.; Kaul, F.; Merkle, E.; Zech, C.J.; Boll, D.; Vogt, D.R.; et al. Comparison of glucagon-like peptide-1 receptor (GLP-1R) PET/CT, SPECT/CT and 3T MRI for the localisation of occult insulinomas: Evaluation of diagnostic accuracy in a prospective crossover imaging study. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 2318–2327. [Google Scholar] [CrossRef]
- Hayward, I.; Mindelzun, R.E.; Jeffrey, R.B. Intrapancreatic accessory spleen mimicking pancreatic mass on CT. J. Comput. Assist. Tomogr. 1992, 16, 984–985. [Google Scholar] [CrossRef]
- Uchiyama, S.; Chijiiwa, K.; Hiyoshi, M.; Ohuchida, J.; Imamura, N.; Nagano, M.; Hidaka, H.; Yorita, K.; Akiyama, Y.; Nishiura, M. Intrapancreatic accessory spleen mimicking endocrine tumor of the pancreas: Case report and review of the literature. J. Gastrointest. Surg. 2008, 12, 1471–1473. [Google Scholar] [CrossRef]
- Hwang, H.S.; Lee, S.S.; Kim, S.C.; Seo, D.W.; Kim, J. Intrapancreatic accessory spleen: Clinicopathologic analysis of 12 cases. Pancreas 2011, 40, 956–965. [Google Scholar] [CrossRef]
- Sahani, D.V.; Bonaffini, P.A.; Fernandez-Del Castillo, C.; Blake, M.A. Gastroenteropancreatic neuroendocrine tumors: Role of imaging in diagnosis and management. Radiology 2013, 266, 38–61. [Google Scholar] [CrossRef]
- Shah, M.; McClelland, A.; Moadel, R.; Javed, A.A.; Freeman, L.M. Splenule disguised as pancreatic mass: Elucidated with SPECT liver-spleen scintigraphy. Clin. Nucl. Med. 2014, 39, 405–406. [Google Scholar] [CrossRef]
- Kann, P.H.; Rothmund, M.; Zielke, A. Endoscopic ultrasound imaging of insulinomas: Limitations and clinical relevance. Exp. Clin. Endocrinol. Diabetes 2005, 113, 471–474. [Google Scholar] [CrossRef]
- Kann, P.H.; Ivan, D.; Pfutzner, A.; Forst, T.; Langer, P.; Schaefer, S. Preoperative diagnosis of insulinoma: Low body mass index, young age, and female gender are associated with negative imaging by endoscopic ultrasound. Eur. J. Endocrinol. 2007, 157, 209–213. [Google Scholar] [CrossRef]
- Tucker, O.N.; Crotty, P.L.; Conlon, K.C. The management of insulinoma. Br. J. Surg. 2006, 93, 264–275. [Google Scholar] [CrossRef]
- Roche, A.; Raisonnier, A.; Gillon-Savouret, M.C. Pancreatic venous sampling and arteriography in localizing insulinomas and gastrinomas: Procedure and results in 55 cases. Radiology 1982, 145, 621–627. [Google Scholar] [CrossRef]
- Gunther, R.W.; Klose, K.J.; Ruckert, K.; Beyer, J.; Kuhn, F.P.; Klotter, H.J. Localization of small islet-cell tumors. Preoperative and intraoperative ultrasound, computed tomography, arteriography, digital subtraction angiography, and pancreatic venous sampling. Gastrointest. Radiol. 1985, 10, 145–152. [Google Scholar] [CrossRef]
- Jackson, J.E. Angiography and arterial stimulation venous sampling in the localization of pancreatic neuroendocrine tumours. Best Pract. Res. Clin. Endocrinol. Metab. 2005, 19, 229–239. [Google Scholar] [CrossRef]
- van Treijen, M.J.C.; van Beek, D.J.; van Leeuwaarde, R.S.; Vriens, M.R.; Valk, G.D. Diagnosing Nonfunctional Pancreatic NETs in MEN1: The Evidence Base. J. Endocr. Soc. 2018, 2, 1067–1088. [Google Scholar] [CrossRef]
- Shin, L.K.; Brant-Zawadzki, G.; Kamaya, A.; Jeffrey, R.B. Intraoperative ultrasound of the pancreas. Ultrasound Q. 2009, 25, 39–48. [Google Scholar] [CrossRef]
- Ritzel, R.A.; Isermann, B.; Schilling, T.; Knaebel, H.P.; Buchler, M.W.; Nawroth, P.P. Diagnosis and localization of insulinoma after negative laparotomy by hyperinsulinemic, hypoglycemic clamp and intra-arterial calcium stimulation. Rev. Diabet Stud. 2004, 1, 42–46. [Google Scholar] [CrossRef][Green Version]
- Wong, M.; Isa, S.H.; Zahiah, M.; Azmi, K.N. Intraoperative ultrasound with palpation is still superior to intra-arterial calcium stimulation test in localising insulinoma. World J. Surg. 2007, 31, 586–592. [Google Scholar] [CrossRef]
- Kalafat, H.; Mihmanli, I.; Saribeyoglu, K.; Belli, A. Intraoperative doppler ultrasound: A reliable diagnostic method in insulinoma. Hepatogastroenterology 2007, 54, 1256–1258. [Google Scholar]
- Gianello, P.; Gigot, J.F.; Berthet, F.; Dardenne, A.N.; Lambotte, L.; Rahier, J.; Otte, J.B.; Kestens, P.J. Pre-and intraoperative localization of insulinomas: Report of 22 observations. World J. Surg. 1988, 12, 389–397. [Google Scholar] [CrossRef]
- Nakamura, Y.; Matsushita, A.; Katsuno, A.; Yamahatsu, K.; Sumiyoshi, H.; Mizuguchi, Y.; Uchida, E. Clinical outcomes of 15 consecutive patients who underwent laparoscopic insulinoma resection: The usefulness of monitoring intraoperative blood insulin during laparoscopic pancreatectomy. Asian J. Endosc. Surg. 2015, 8, 303–309. [Google Scholar] [CrossRef]
- Morita, S.; Machida, H.; Kuwatsuru, R.; Saito, N.; Suzuki, K.; Iihara, M.; Obara, T.; Mitsuhashi, N. Preoperative localization of pancreatic insulinoma by super selective arterial stimulation with venous sampling. Abdom. Imaging 2007, 32, 126–128. [Google Scholar] [CrossRef]
- Winer, J.H.; Choi, H.S.; Gibbs-Strauss, S.L.; Ashitate, Y.; Colson, Y.L.; Frangioni, J.V. Intraoperative localization of insulinoma and normal pancreas using invisible near-infrared fluorescent light. Ann. Surg. Oncol. 2010, 17, 1094–1100. [Google Scholar] [CrossRef]
- Thompson, S.M.; Vella, A.; Service, F.J.; Grant, C.S.; Thompson, G.B.; Andrews, J.C. Impact of variant pancreatic arterial anatomy and overlap in regional perfusion on the interpretation of selective arterial calcium stimulation with hepatic venous sampling for preoperative localization of occult insulinoma. Surgery 2015, 158, 162–172. [Google Scholar] [CrossRef][Green Version]
- Thompson, S.M.; Vella, A.; Thompson, G.B.; Rumilla, K.M.; Service, F.J.; Grant, C.S.; Andrews, J.C. Selective Arterial Calcium Stimulation with Hepatic Venous Sampling Differentiates Insulinoma From Nesidioblastosis. J. Clin. Endocrinol. Metab. 2015, 100, 4189–4197. [Google Scholar] [CrossRef]
- Service, G.J.; Thompson, G.B.; Service, F.J.; Andrews, J.C.; Collazo-Clavell, M.L.; Lloyd, R.V. Hyperinsulinemic hypoglycemia with nesidioblastosis after gastric-bypass surgery. N. Engl. J. Med. 2005, 353, 249–254. [Google Scholar] [PubMed]
- Bertelli, E.; Di Gregorio, F.; Mosca, S.; Bastianini, A. Selective Arterial Calcium Stimulation with Hepatic Venous Sampling in Immune-Mediated Hypoglycemia. J. Endocr. Soc. 2017, 1, 671–675. [Google Scholar]
- Bertelli, E.; Di Gregorio, F.; Mosca, S.; Bastianini, A. The arterial blood supply of the pancreas: A review. V. The dorsal pancreatic artery. An. anatomic review and a radiologic study. Surg. Radiol. Anat. 1998, 20, 445–452. [Google Scholar]
- Woodburne, R.T.; Olsen, L.L. The arteries of the pancreas. Anat. Rec. 1951, 111, 255–270. [Google Scholar] [CrossRef] [PubMed]
- Lewis, M.P.; Reber, H.A.; Ashley, S.W. Pancreatic blood flow and its role in the pathophysiology of pancreatitis. J. Surg. Res. 1998, 75, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Dolensek, J.; Rupnik, M.S.; Stozer, A. Structural similarities and differences between the human and the mouse pancreas. Islets 2015, 7, e1024405. [Google Scholar] [CrossRef]
- Sakuhara, Y.; Kodama, Y.; Abo, D.; Hasegawa, Y.; Shimizu, T.; Omatsu, T.; Kamishika, T.; Onodera, Y.; Terae, S.; Shirato, H. Evaluation of the vascular supply to regions of the pancreas on CT during arteriography. Abdom. Imaging 2008, 33, 563–570. [Google Scholar] [CrossRef]
- Tanaka, T.; Sakaguchi, H.; Anai, H.; Yamamoto, K.; Morimoto, K.; Nishiofuku, H.; Kichikawa, K. Catheter position for adequate intra-arterial chemotherapy for advanced pancreatic cancer: Evaluation with CT during arterial injection of contrast material. J. Vasc. Interv. Radiol. 2004, 15, 1089–1097. [Google Scholar] [CrossRef]
- Patel, I.J.; Rahim, S.; Davidson, J.C.; Hanks, S.E.; Tam, A.L.; Walker, T.G.; Wilkins, L.R.; Sarode, R.; Weinberg, I. Society of Interventional Radiology Consensus Guidelines for the Periprocedural Management of Thrombotic and Bleeding Risk in Patients Undergoing Percutaneous Image-Guided Interventions-Part. II: Recommendations: Endorsed by the Canadian Association for Interventional Radiology and the Cardiovascular and Interventional Radiological Society of Europe. J. Vasc. Interv. Radiol. 2019, 30, 1168–1184. [Google Scholar]
- Morera, J.; Guillaume, A.; Courtheoux, P.; Palazzo, L.; Rod, A.; Joubert, M.; Reznik, Y. Preoperative localization of an insulinoma: Selective arterial calcium stimulation test performance. J. Endocrinol. Investig. 2016, 39, 455–463. [Google Scholar]
- Thompson, S.M.; Vella, A.; Service, F.J.; Thompson, G.; Andrews, J.C. Selective Arterial Calcium Stimulation with Hepatic Venous Sampling in Patients with Recurrent Endogenous Hyperinsulinemic Hypoglycemia and Metastatic Insulinoma: Evaluation in Five Patients. J. Vasc. Interv. Radiol. 2017, 28, 1745–1749. [Google Scholar] [CrossRef] [PubMed]
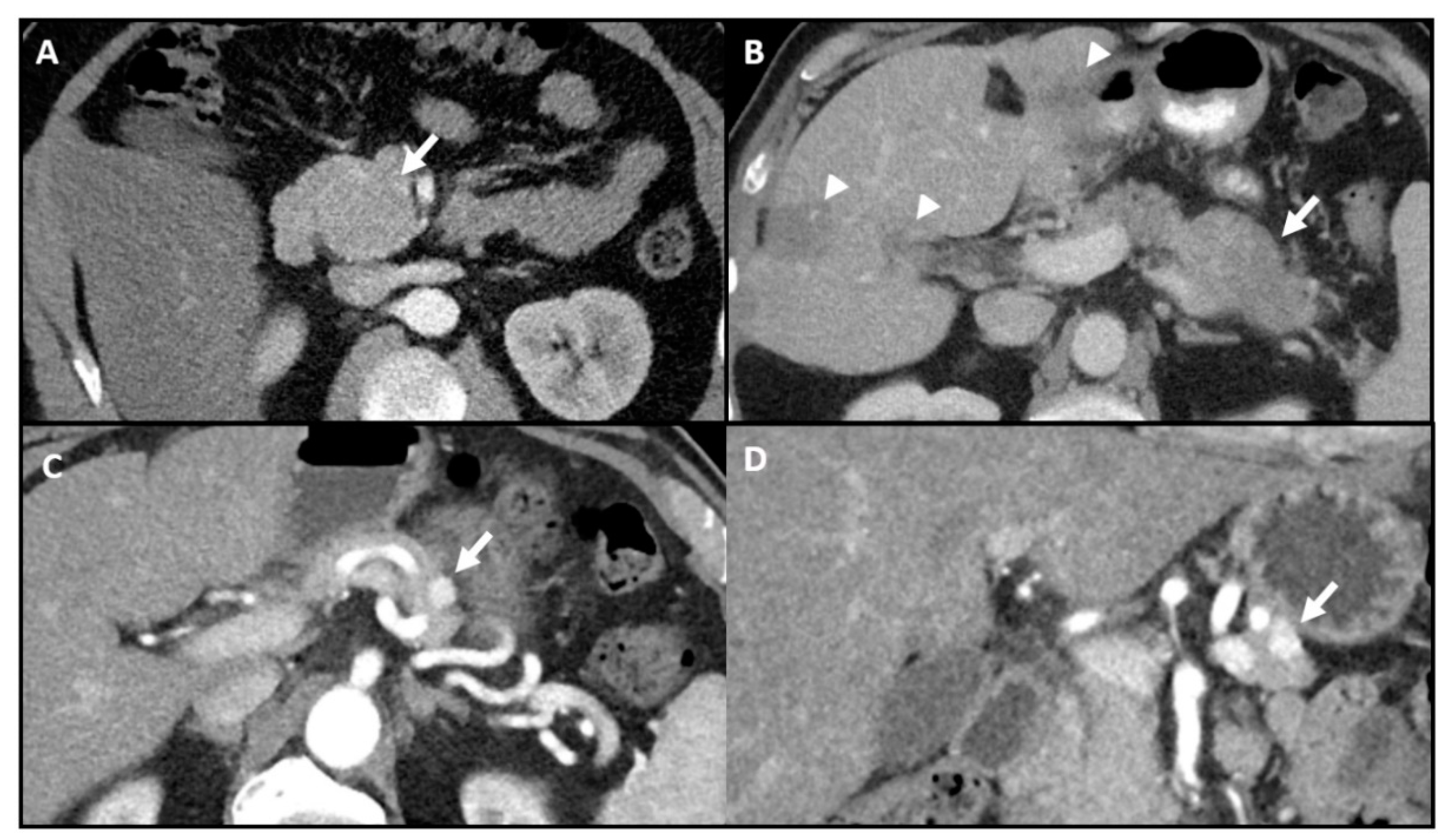
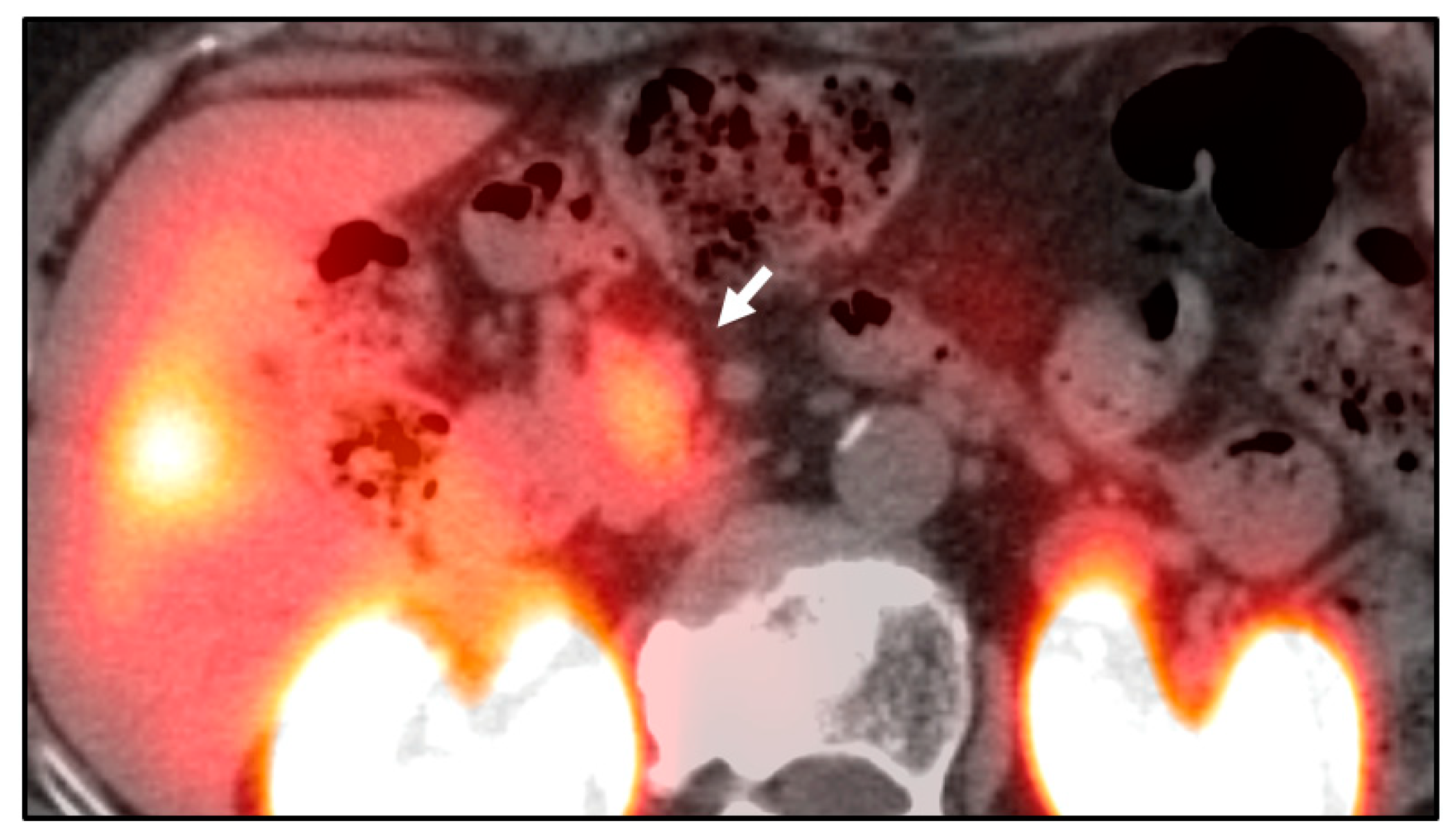
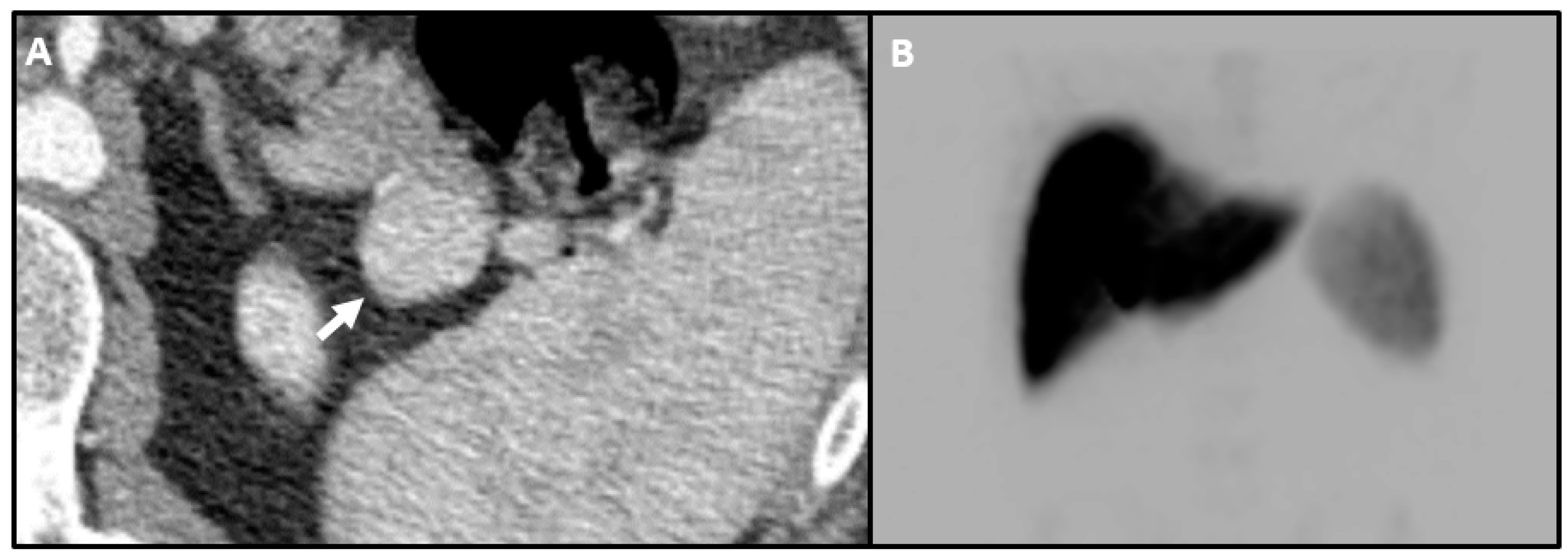
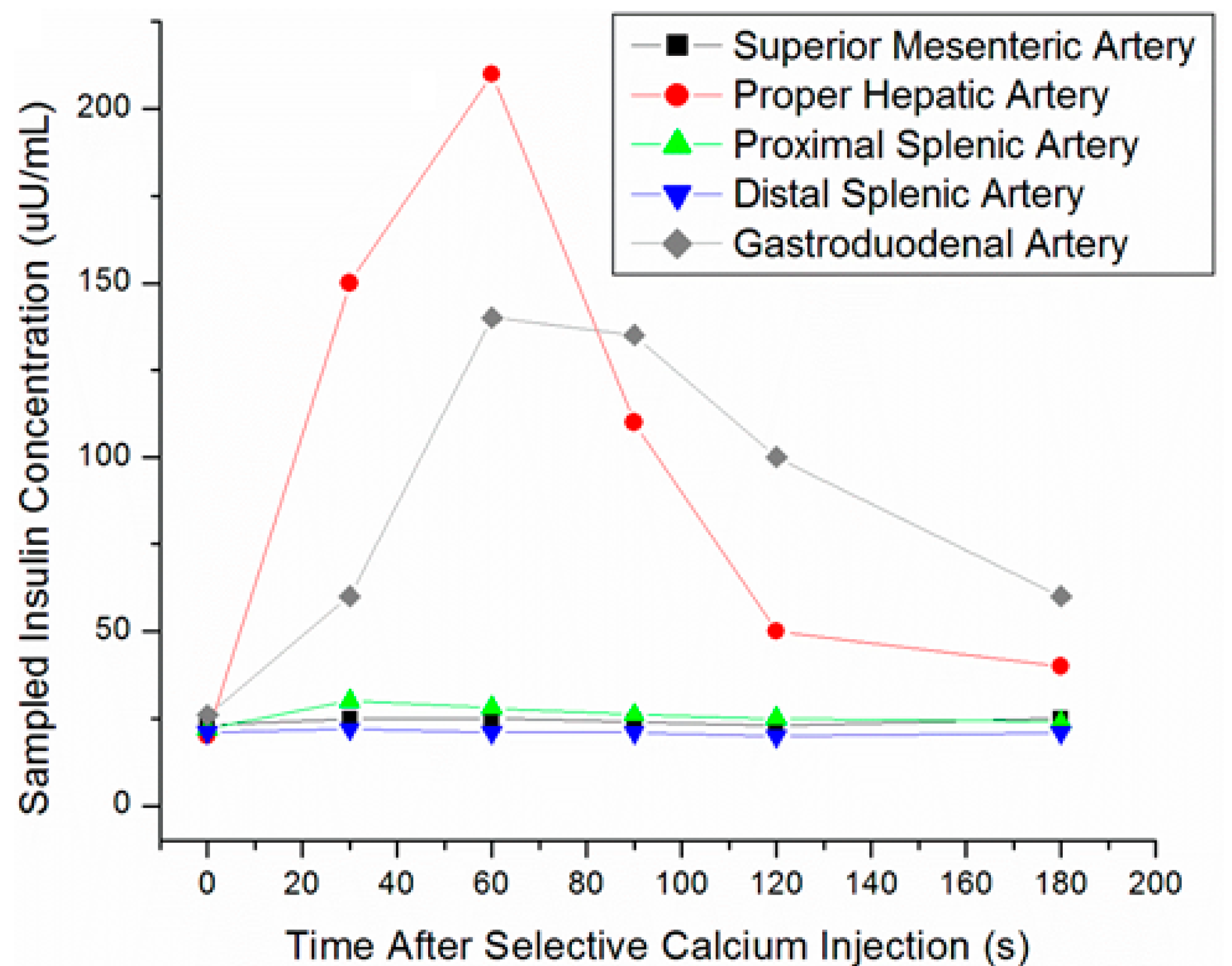
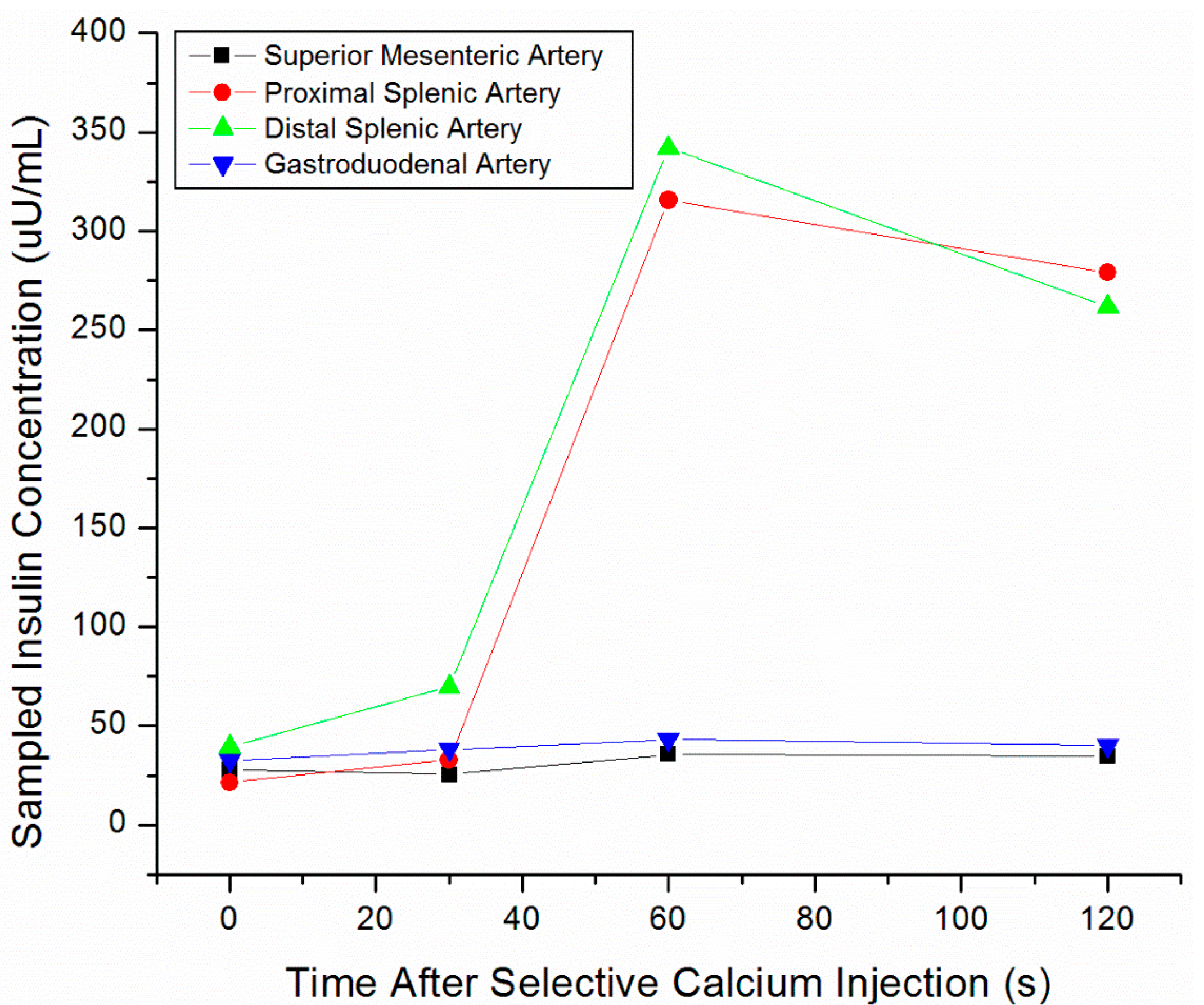
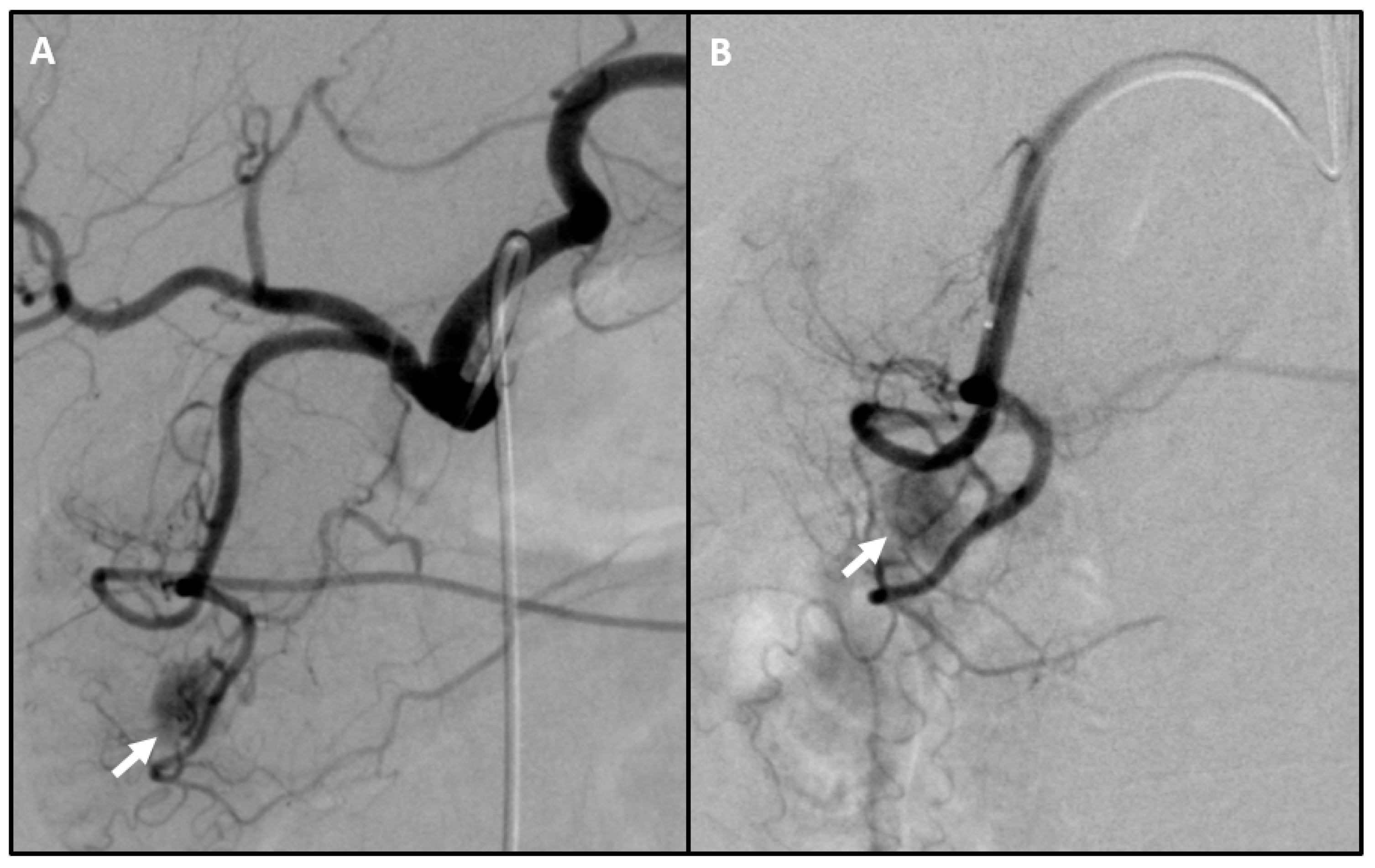
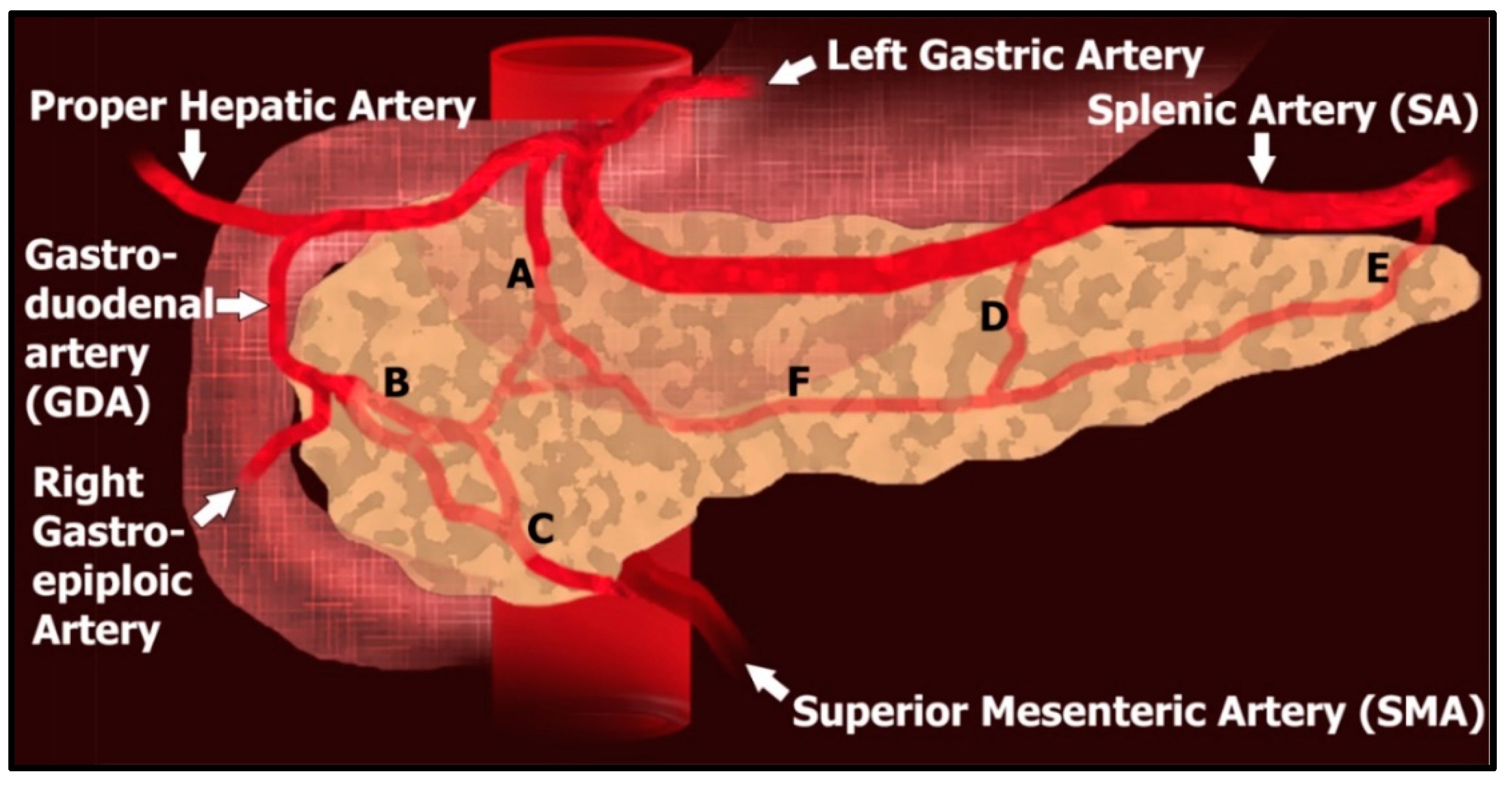
| Method | Detection Rate | Pros | Cons |
|---|---|---|---|
| Transabdominal US [19,20,21] | 9–63% | Inexpensive. Widely available. | Limited by body habitus and bowel gas. Poor visualization of pancreatic tail and small tumors. |
| Contrast Enhanced CT [22,23,24] | 63–94% | Rapid imaging. High spatial resolution. Visualizes anatomy and potential metastatic disease. | Ionizing radiation. Iodinated contrast. |
| Contrast-Enhanced MRI [25,26] | 60–90% | No ionizing radiation. No iodinated contrast. | Expensive. Confined space. Prone to motion artifact. |
| Somatostatin Receptor Imaging [27,28,29,30] | 47–60% or 32–90%, per radiotracer | Useful for troubleshooting when other imaging is negative or equivocal. | Ionizing radiation. Long image acquisition time. Poor spatial resolution. |
| Endoscopic Ultrasound [31] | 40–93%, per pancreatic region | Excellent visualization of pancreatic head. May obtain concurrent biopsies. | Invasive. Poor visualization of pancreatic tail. |
| SACST [14,16,17,32,33] | 67–100% | High sensitivity. | Invasive. Ionizing radiation. Requires technical expertise. |
| Operative Inspection and Palpation [16,17] | 77–91% | Highly effective when performed by expert surgeon. | Requires open surgery and operative experience. |
| Hepatic Venous Sampling |
| 6 Fr short sheath (right internal jugular vein), no flush |
| 6 Fr 65 cm Cobra catheter, with added side hole |
| 42 labeled 10 cc syringes |
| Selective Arterial Calcium Stimulation |
| 5 Fr short sheath (right common femoral artery), normal saline flush |
| 5 Fr 100 cm Sim 1 catheter |
| 130 cm Progreat microcatheter and Fathom microwire |
| 10% calcium gluconate solution (940 mg/10 cc) diluted to 5 cc aliquots of 1 mg/kg (0.025 mEq Ca/kg) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, K.; Patel, N.; Kulkarni, K.; Gross, J.S.; Taslakian, B. Essentials of Insulinoma Localization with Selective Arterial Calcium Stimulation and Hepatic Venous Sampling. J. Clin. Med. 2020, 9, 3091. https://doi.org/10.3390/jcm9103091
Zhao K, Patel N, Kulkarni K, Gross JS, Taslakian B. Essentials of Insulinoma Localization with Selective Arterial Calcium Stimulation and Hepatic Venous Sampling. Journal of Clinical Medicine. 2020; 9(10):3091. https://doi.org/10.3390/jcm9103091
Chicago/Turabian StyleZhao, Ken, Nihal Patel, Kopal Kulkarni, Jonathan S. Gross, and Bedros Taslakian. 2020. "Essentials of Insulinoma Localization with Selective Arterial Calcium Stimulation and Hepatic Venous Sampling" Journal of Clinical Medicine 9, no. 10: 3091. https://doi.org/10.3390/jcm9103091
APA StyleZhao, K., Patel, N., Kulkarni, K., Gross, J. S., & Taslakian, B. (2020). Essentials of Insulinoma Localization with Selective Arterial Calcium Stimulation and Hepatic Venous Sampling. Journal of Clinical Medicine, 9(10), 3091. https://doi.org/10.3390/jcm9103091




