Impact of Vitamin C and Thiamine Administration on Delirium-Free Days in Patients with Septic Shock
Abstract
1. Introduction
2. Materials and Methods
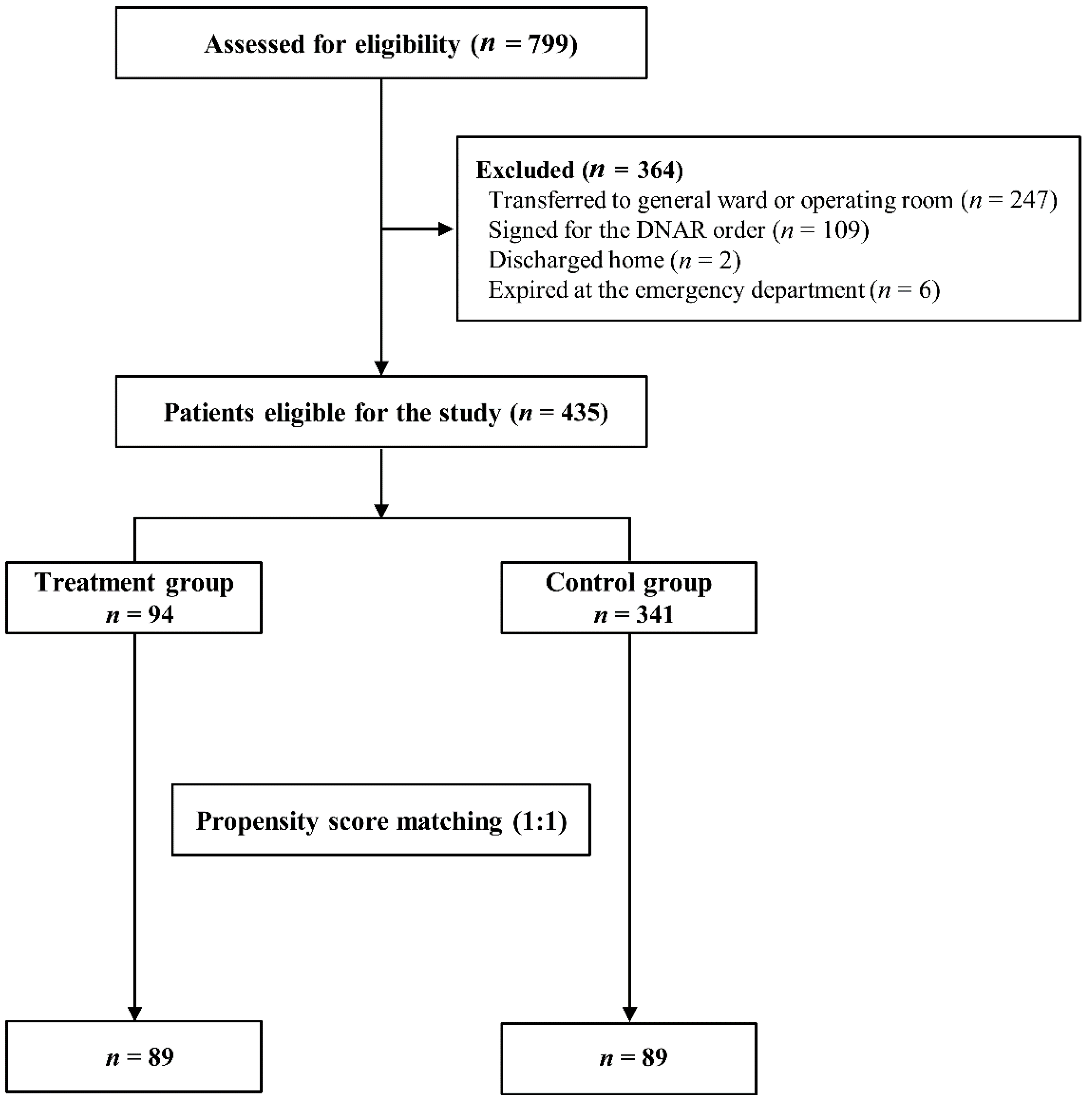
2.1. Study Design and Population
2.2. Vitamin Administration and Patient Management
2.3. Definition and Data Collection
2.4. Outcome Measures
2.5. Primary Data Analysis
3. Results
3.1. Baseline Characteristics
3.2. Outcomes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Association, A.P. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; DSM-5; American Psychiatric Publishing: Arlington, VA, USA, 2013. [Google Scholar]
- Agarwal, V.; O’Neill, P.J.; Cotton, B.A.; Pun, B.T.; Haney, S.; Thompson, J.; Kassebaum, N.; Shintani, A.; Guy, J.; Ely, E.W.; et al. Prevalence and risk factors for development of delirium in burn intensive care unit patients. J. Burn Care Res. Off. Publ. Am. Burn Assoc. 2010, 31, 706–715. [Google Scholar] [CrossRef]
- Salluh, J.I.; Soares, M.; Teles, J.M.; Ceraso, D.; Raimondi, N.; Nava, V.S.; Blasquez, P.; Ugarte, S.; Ibanez-Guzman, C.; Centeno, J.V.; et al. Delirium epidemiology in critical care (DECCA): An international study. Crit. Care 2010, 14, R210. [Google Scholar] [CrossRef] [PubMed]
- Girard, T.D.; Thompson, J.L.; Pandharipande, P.P.; Brummel, N.E.; Jackson, J.C.; Patel, M.B.; Hughes, C.G.; Chandrasekhar, R.; Pun, B.T.; Boehm, L.M.; et al. Clinical phenotypes of delirium during critical illness and severity of subsequent long-term cognitive impairment: A prospective cohort study. Lancet Respir. Med. 2018, 6, 213–222. [Google Scholar] [CrossRef]
- Ely, E.W.; Shintani, A.; Truman, B.; Speroff, T.; Gordon, S.M.; Harrell, F.E., Jr.; Inouye, S.K.; Bernard, G.R.; Dittus, R.S. Delirium as a predictor of mortality in mechanically ventilated patients in the intensive care unit. JAMA 2004, 291, 1753–1762. [Google Scholar] [CrossRef] [PubMed]
- Carr, A.C.; Rosengrave, P.C.; Bayer, S.; Chambers, S.; Mehrtens, J.; Shaw, G.M. Hypovitaminosis C and vitamin C deficiency in critically ill patients despite recommended enteral and parenteral intakes. Crit. Care 2017, 21, 300. [Google Scholar] [CrossRef]
- Donnino, M.W.; Carney, E.; Cocchi, M.N.; Barbash, I.; Chase, M.; Joyce, N.; Chou, P.P.; Ngo, L. Thiamine deficiency in critically ill patients with sepsis. J. Crit. Care 2010, 25, 576–581. [Google Scholar] [CrossRef] [PubMed]
- Paul, E.M.; Joseph, V. Critical Care for the Respiratory Specialist: Sepsis, Delirium and Long-Term Cognitive Dysfunction: Prevention with the Combination of Vitamin C, Hydrocortisone and Thiamine. Curr. Respir. Med. Rev. 2018, 14, 23–28. [Google Scholar]
- Osiezagha, K.; Ali, S.; Freeman, C.; Barker, N.C.; Jabeen, S.; Maitra, S.; Olagbemiro, Y.; Richie, W.; Bailey, R.K. Thiamine deficiency and delirium. Innov. Clin. Neurosci. 2013, 10, 26–32. [Google Scholar] [PubMed]
- Voigt, K.; Kontush, A.; Stuerenburg, H.J.; Muench-Harrach, D.; Hansen, H.C.; Kunze, K. Decreased plasma and cerebrospinal fluid ascorbate levels in patients with septic encephalopathy. Free Radic. Res. 2002, 36, 735–739. [Google Scholar] [CrossRef]
- Jackson, P.; Khan, A. Delirium in critically ill patients. Crit. Care Clin. 2015, 31, 589–603. [Google Scholar] [CrossRef]
- Brown, T.M. Neuropsychiatric scurvy. Psychosomatics 2015, 56, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Torbergsen, A.C.; Watne, L.O.; Frihagen, F.; Wyller, T.B.; Brugaard, A.; Mowe, M. Vitamin deficiency as a risk factor for delirium. Eur. Geriatr. Med. 2015, 6, 314–318. [Google Scholar] [CrossRef]
- Ikeda, K.; Liu, X.; Kida, K.; Marutani, E.; Hirai, S.; Sakaguchi, M.; Andersen, L.W.; Bagchi, A.; Cocchi, M.N.; Berg, K.M.; et al. Thiamine as a neuroprotective agent after cardiac arrest. Resuscitation 2016, 105, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Abdou, E.; Hazell, A.S. Thiamine deficiency: An update of pathophysiologic mechanisms and future therapeutic considerations. Neurochem. Res. 2015, 40, 353–361. [Google Scholar] [CrossRef] [PubMed]
- Woolum, J.A.; Abner, E.L.; Kelly, A.; Thompson Bastin, M.L.; Morris, P.E.; Flannery, A.H. Effect of Thiamine Administration on Lactate Clearance and Mortality in Patients With Septic Shock. Crit. Care Med. 2018, 46, 1747–1752. [Google Scholar] [CrossRef]
- Donnino, M.W.; Andersen, L.W.; Chase, M.; Berg, K.M.; Tidswell, M.; Giberson, T.; Wolfe, R.; Moskowitz, A.; Smithline, H.; Ngo, L.; et al. Randomized, Double-Blind, Placebo-Controlled Trial of Thiamine as a Metabolic Resuscitator in Septic Shock: A Pilot Study. Crit. Care Med. 2016, 44, 360–367. [Google Scholar] [CrossRef]
- Zabet, M.H.; Mohammadi, M.; Ramezani, M.; Khalili, H. Effect of high-dose Ascorbic acid on vasopressor’s requirement in septic shock. J. Res. Pharm. Pract. 2016, 5, 94–100. [Google Scholar]
- Fowler, A.A., 3rd; Syed, A.A.; Knowlson, S.; Sculthorpe, R.; Farthing, D.; DeWilde, C.; Farthing, C.A.; Larus, T.L.; Martin, E.; Brophy, D.F.; et al. Phase I safety trial of intravenous ascorbic acid in patients with severe sepsis. J. Transl. Med. 2014, 12, 32. [Google Scholar] [CrossRef]
- Rhodes, A.; Evans, L.E.; Alhazzani, W.; Levy, M.M.; Antonelli, M.; Ferrer, R.; Kumar, A.; Sevransky, J.E.; Sprung, C.L.; Nunnally, M.E.; et al. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock: 2016. Crit. Care Med. 2017, 45, 486–552. [Google Scholar] [CrossRef]
- Shin, T.G.; Hwang, S.Y.; Kang, G.H.; Kim, W.Y.; Ryoo, S.M.; Kim, K.; Jo, Y.H.; Chung, S.P.; Joo, Y.S.; Beom, J.H.; et al. Korean Shock Society septic shock registry: A preliminary report. Clin. Exp. Emerg. Med. 2017, 4, 146–153. [Google Scholar] [CrossRef]
- Heo, E.Y.; Lee, B.J.; Hahm, B.J.; Song, E.H.; Lee, H.A.; Yoo, C.G.; Kim, Y.W.; Han, S.K.; Shim, Y.S.; Lee, S.M. Translation and validation of the Korean Confusion Assessment Method for the Intensive Care Unit. BMC Psychiatry 2011, 11, 94. [Google Scholar] [CrossRef] [PubMed]
- Jeon, K.; Jeong, B.H.; Ko, M.G.; Nam, J.; Yoo, H.; Chung, C.R.; Suh, G.Y. Impact of delirium on weaning from mechanical ventilation in medical patients. Respirology 2016, 21, 313–320. [Google Scholar] [CrossRef] [PubMed]
- Singer, M.; Deutschman, C.S.; Seymour, C.W.; Shankar-Hari, M.; Annane, D.; Bauer, M.; Bellomo, R.; Bernard, G.R.; Chiche, J.D.; Coopersmith, C.M.; et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016, 315, 801–810. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J. A power primer. Psychol. Bull. 1992, 112, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Sawilowsky, S.S. New effect size rules of thumb. J. Mod. Appl. Stat. Methods 2009, 8, 597–599. [Google Scholar] [CrossRef]
- Ritter, C.; Tomasi, C.D.; Dal-Pizzol, F.; Pinto, B.B.; Dyson, A.; de Miranda, A.S.; Comim, C.M.; Soares, M.; Teixeira, A.L.; Quevedo, J.; et al. Inflammation biomarkers and delirium in critically ill patients. Crit. Care 2014, 18, R106. [Google Scholar] [CrossRef]
- Wood, M.D.; Maslove, D.M.; Muscedere, J.G.; Day, A.G.; Gordon Boyd, J. Low brain tissue oxygenation contributes to the development of delirium in critically ill patients: A prospective observational study. J. Crit. Care 2017, 41, 289–295. [Google Scholar] [CrossRef]
- Van Rompaey, B.; Elseviers, M.M.; Schuurmans, M.J.; Shortridge-Baggett, L.M.; Truijen, S.; Bossaert, L. Risk factors for delirium in intensive care patients: A prospective cohort study. Crit. Care 2009, 13, R77. [Google Scholar] [CrossRef]
- Amrein, K.; Schnedl, C.; Holl, A.; Riedl, R.; Christopher, K.B.; Pachler, C.; Urbanic Purkart, T.; Waltensdorfer, A.; Münch, A.; Warnkross, H.; et al. Effect of High-Dose Vitamin D3on Hospital Length of Stay in Critically Ill Patients With Vitamin D Deficiency. JAMA 2014, 312, 1520–1530. [Google Scholar] [CrossRef]
- Marik, P.E.; Khangoora, V.; Rivera, R.; Hooper, M.H.; Catravas, J. Hydrocortisone, Vitamin C, and Thiamine for the Treatment of Severe Sepsis and Septic Shock: A Retrospective Before-After Study. Chest 2017, 151, 1229–1238. [Google Scholar] [CrossRef]
- O’Keeffe, S.T.; Tormey, W.P.; Glasgow, R.; Lavan, J.N. Thiamine deficiency in hospitalized elderly patients. Gerontology 1994, 40, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Robitaille, L.; Eintracht, S.; Hoffer, L.J. Vitamin C provision improves mood in acutely hospitalized patients. Nutrition 2011, 27, 530–533. [Google Scholar] [CrossRef]
- Brody, S. High-dose ascorbic acid increases intercourse frequency and improves mood: A randomized controlled clinical trial. Biol. Psychiatry 2002, 52, 371–374. [Google Scholar] [CrossRef]
- Amr, M.; El-Mogy, A.; Shams, T.; Vieira, K.; Lakhan, S.E. Efficacy of vitamin C as an adjunct to fluoxetine therapy in pediatric major depressive disorder: A randomized, double-blind, placebo-controlled pilot study. Nutr. J. 2013, 12, 31. [Google Scholar] [CrossRef]
- Carr, A.C.; McCall, C. The role of vitamin C in the treatment of pain: New insights. J. Transl. Med. 2017, 15, 77. [Google Scholar] [CrossRef]
- Marik, P.E. Hydrocortisone, Ascorbic Acid and Thiamine (HAT Therapy) for the Treatment of Sepsis. Focus on Ascorbic Acid. Nutrients 2018, 10, 1762. [Google Scholar] [CrossRef] [PubMed]
- Shin, T.G.; Kim, Y.J.; Ryoo, S.M.; Hwang, S.Y.; Jo, I.J.; Chung, S.P.; Choi, S.H.; Suh, G.J.; Kim, W.Y. Early Vitamin C and Thiamine Administration to Patients with Septic Shock in Emergency Departments: Propensity Score-Based Analysis of a Before-and-After Cohort Study. J. Clin. Med. 2019, 8, 102. [Google Scholar] [CrossRef] [PubMed]
- Litwak, J.J.; Cho, N.; Nguyen, H.B.; Moussavi, K.; Bushell, T. Vitamin C, Hydrocortisone, and Thiamine for the Treatment of Severe Sepsis and Septic Shock: A Retrospective Analysis of Real-World Application. J. Clin. Med. 2019, 8, 478. [Google Scholar] [CrossRef]
- Hager, D.N.; Hooper, M.H.; Bernard, G.R.; Busse, L.W.; Ely, E.W.; Fowler, A.A.; Gaieski, D.F.; Hall, A.; Hinson, J.S.; Jackson, J.C.; et al. The Vitamin C, Thiamine and Steroids in Sepsis (VICTAS) Protocol: A prospective, multi-center, double-blind, adaptive sample size, randomized, placebo-controlled, clinical trial. Trials 2019, 20, 197. [Google Scholar] [CrossRef]
| Variables | Before Matching | After Matching | ||||
|---|---|---|---|---|---|---|
| Treatment (n = 94) | Control (n = 341) | p | Treatment (n = 89) | Control (n = 89) | p | |
| Age, years | 69 (60–76) | 69 (61–76) | 0.943 | 69 (61–76) | 71 (62–78) | 0.653 |
| Sex, male | 55 (58.5) | 229 (67.1) | 0.119 | 52 (58.4) | 55 (61.8) | 0.662 |
| Comorbidities | ||||||
| Diabetes | 32 (34) | 117 (34.3) | 0.961 | 31 (34.8) | 29 (32.6) | 0.768 |
| Hypertension | 36 (38.3) | 139 (40.8) | 0.666 | 34 (38.2) | 35 (39.3) | 0.884 |
| Cardiac disease | 11 (11.7) | 62 (18.2) | 0.137 | 11 (12.4) | 16 (18) | 0.251 |
| Cerebral vascular disease | 9 (9.6) | 27 (7.9) | 0.606 | 9 (10.1) | 8 (9) | 0.782 |
| Chronic lung disease | 8 (8.5) | 36 (10.6) | 0.560 | 8 (9) | 9 (10.1) | 0.808 |
| Chronic renal disease | 7 (7.5) | 23 (10.3) | 0.413 | 7 (7.8) | 9 (10.1) | 0.593 |
| Chronic liver disease | 15 (16) | 47 (14) | 0.593 | 15 (16.9) | 19 (21.4) | 0.465 |
| Hematologic malignancy | 13 (13.8) | 32 (9.4) | 0.210 | 12 (13.5) | 10 (11.2) | 0.655 |
| Metastatic cancer | 20 (21.3) | 92 (27) | 0.263 | 19 (21.4) | 17 (19.1) | 0.706 |
| Infection focus | 0.006 | >0.999 | ||||
| Respiratory | 24 (25.5) | 140 (41.1) | 23 (25.8) | 23 (25.8) | ||
| Non-respiratory | 70 (74.5) | 201 (58.9) | 66 (74.2) | 132 (74.2) | ||
| APACHE II score | 30.2 ± 8.1 | 28.4 ± 9.2 | 0.044 | 30.0 ± 7.9 | 30.0 ± 8.9 | 0.697 |
| SOFA score | 11.5 ± 3.4 | 10.1 ± 3.7 | <0.001 | 11.4 ± 3.5 | 11.5 ± 3.4 | 0.854 |
| Sepsis-3 definition * | 80 (85.1) | 215 (63.1) | <0.001 | 75 (84.3) | 73 (82) | 0.593 |
| Laboratory tests | ||||||
| Lactate (mmol/L) | 5.5 ± 3.1 | 4.6 ± 3.2 | 0.003 | 5.2 ± 2.7 | 4.8 ± 2.9 | 0.431 |
| Albumin (mg/dL) | 3.1 ± 0.6 | 3.2 ± 0.6 | 0.209 | 3.1 ± 0.6 | 3.1 ± 0.6 | 0.742 |
| Creatinine (mg/dL) | 2.1 ± 1.5 | 1.8 ± 1.6 | 0.019 | 1.9 ± 1.4 | 2.2 ± 2.3 | 0.934 |
| Mechanical ventilation use † | 53 (56.4) | 173 (50.7) | 0.332 | 49 (55.1) | 48 (53.9) | 0.876 |
| Medications † ‡ | ||||||
| Steroid use | 57 (60.6) | 162 (47.5) | 0.024 | 52 (58.4) | 52 (58.4) | >0.999 |
| Benzodiazepine | 9 (9.6) | 73 (21.4) | 0.009 | 9 (10.1) | 8 (9) | 0.782 |
| Opioids | 59 (62.8) | 233 (68.3) | 0.309 | 55 (61.8) | 57 (64.0) | 0.746 |
| Propofol | 5 (5.3) | 17 (5) | 0.896 | 5 (5.6) | 2 (2.3) | 0.453 |
| Delirium at the time of initial ICU admission | 36 (38.3) | 87 (25.5) | 0.015 | 32 (36) | 30 (33.7) | 0.715 |
| Outcomes | Before Matching | After Matching | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Total (n = 435) | Treatment (n = 94) | Control (n = 341) | p | Effect Size | Total (n = 178) | Treatment (n = 89) | Control (n = 89) | p | Effect Size | |
| Delirium-free days | 13 (7–14) | 11 (4–14) | 13 (7–14) | 0.042 | 0.188 | 11 (6–14) | 11 (5–14) | 12 (6–14) | 0.894 | 0.035 |
| Delirium-coma-free days | 12 (4–14) | 13 (6–14) | 11 (2–14) | 0.059 | 0.175 | 11 (4–14) | 11 (3–14) | 12 (4–14) | 0.940 | 0.037 |
| Incidence of delirium | 256 (58.9) | 62 (66.0) | 194 (56.9) | 0.113 | 0.141 | 111 (62.4) | 57 (64.0) | 54 (60.7) | 0.622 | 0.046 |
| Duration of delirium, days | 1 (0–4) | 2 (0–6) | 1 (0–3) | 0.032 | 0.196 | 1 (0–4) | 1 (0–5) | 1 (0–4) | 0.604 | 0.082 |
| Hospital LOS, days | 14 (7–26) | 16 (8–27.5) | 13 (7–24) | 0.065 | 0.147 | 15 (8–27) | 16 (8–27) | 13 (7–28) | 0.693 | 0.117 |
| ICU LOS, days | 4 (3–7) | 4 (3–8) | 4 (3–7) | 0.057 | 0.182 | 4 (3–7) | 4 (3–7) | 4 (3–7) | 0.335 | 0.191 |
| 28-day mortality | 95 (21.8) | 23 (24.5) | 72 (21.1) | 0.486 | 0.053 | 37 (20.8) | 21 (23.6) | 16 (18.0) | 0.336 | 0.111 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, J.E.; Shin, T.G.; Jo, I.J.; Jeon, K.; Suh, G.Y.; Park, M.; Won, H.; Chung, C.R.; Hwang, S.Y. Impact of Vitamin C and Thiamine Administration on Delirium-Free Days in Patients with Septic Shock. J. Clin. Med. 2020, 9, 193. https://doi.org/10.3390/jcm9010193
Park JE, Shin TG, Jo IJ, Jeon K, Suh GY, Park M, Won H, Chung CR, Hwang SY. Impact of Vitamin C and Thiamine Administration on Delirium-Free Days in Patients with Septic Shock. Journal of Clinical Medicine. 2020; 9(1):193. https://doi.org/10.3390/jcm9010193
Chicago/Turabian StylePark, Jong Eun, Tae Gun Shin, Ik Joon Jo, Kyeongman Jeon, Gee Young Suh, Minsu Park, Hojeong Won, Chi Ryang Chung, and Sung Yeon Hwang. 2020. "Impact of Vitamin C and Thiamine Administration on Delirium-Free Days in Patients with Septic Shock" Journal of Clinical Medicine 9, no. 1: 193. https://doi.org/10.3390/jcm9010193
APA StylePark, J. E., Shin, T. G., Jo, I. J., Jeon, K., Suh, G. Y., Park, M., Won, H., Chung, C. R., & Hwang, S. Y. (2020). Impact of Vitamin C and Thiamine Administration on Delirium-Free Days in Patients with Septic Shock. Journal of Clinical Medicine, 9(1), 193. https://doi.org/10.3390/jcm9010193






