Physical Activity and Sedentary Behaviour Patterns in 326 Persons with COPD before Starting a Pulmonary Rehabilitation: A Cluster Analysis
Abstract
1. Introduction
2. Methods
2.1. Study Population
2.2. Assessments
2.2.1. Physical Activity and Sedentary Behaviour
2.2.2. Secondary Outcomes
2.3. Assessments
2.3.1. Processing of Accelerometer Data
2.3.2. Determining the Number of Clusters
2.3.3. Comparison of the Different Clusters
3. Results
3.1. Number of Different Movement Behaviour Clusters
3.2. Characterisation of the Clusters: Physical Activity and Sedentary Behaviour
3.3. Characterisation of the Clusters: Other Parameters
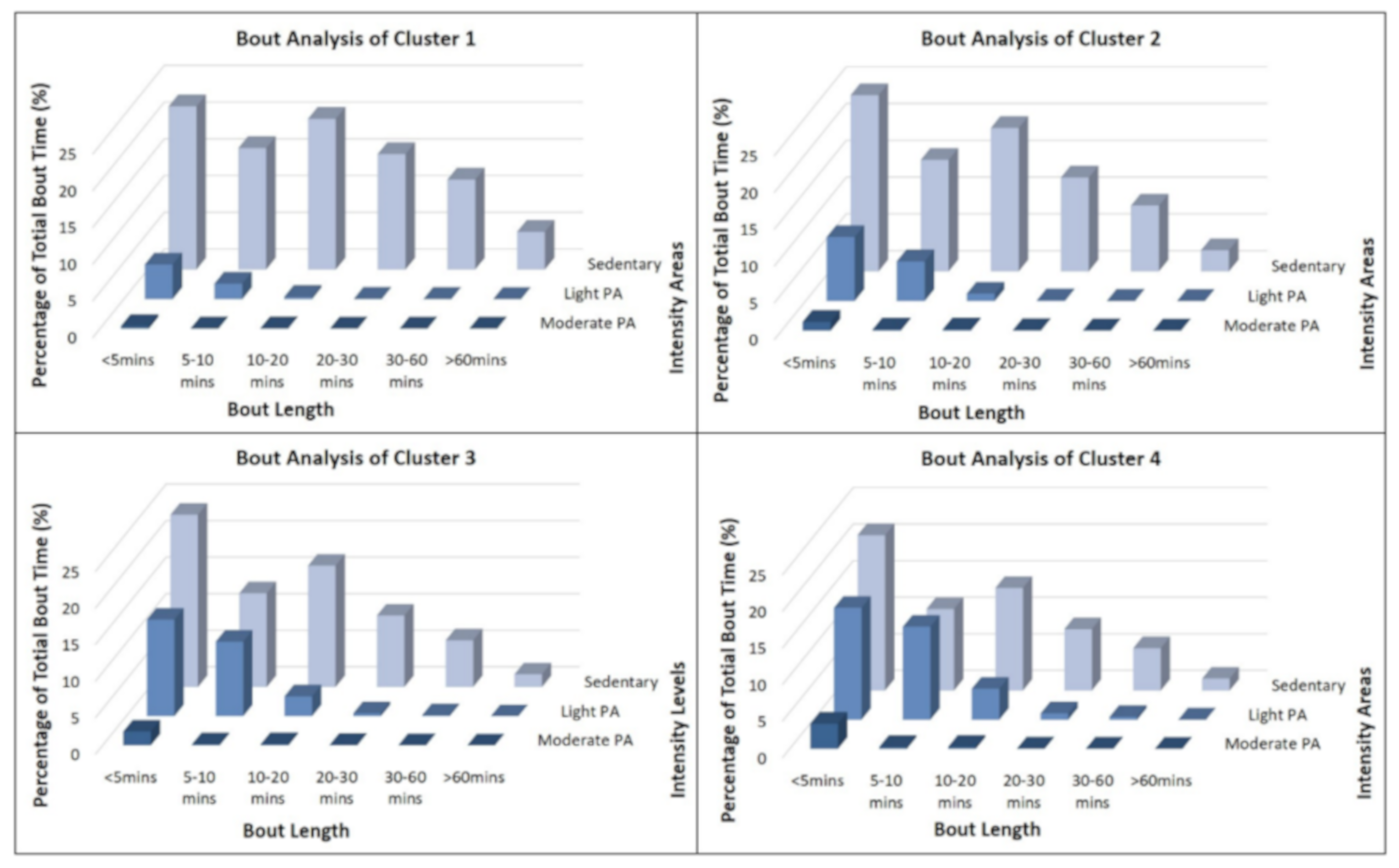
3.4. Activity Bout Analysis
4. Discussion
4.1. Cluster Differences Regarding PA
4.2. Cluster Differences Regarding Sedentary Behaviour
4.3. Cluster Differences Regarding Clinical and Sociodemographic Parameters
4.4. Implications for Pulmonary Rehabilitation
4.5. Future Research
4.6. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Pedersen, B.K.; Saltin, B. Exercise as medicine-evidence for prescribing exercise as therapy in 26 different chronic diseases. Scand. J. Med. Sci. Sports 2015, 25, 1–72. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Global Recommendations on Physical Activity for Health; World Health Organization: Geneva, Switzerland, 2010. [Google Scholar]
- Spruit, M.A.; Singh, S.J.; Garvey, C.; ZuWallack, R.; Nici, L.; Rochester, C.; Hill, K.; Holland, A.E.; Lareau, S.C.; Man, W.D.-C.; et al. An official American Thoracic Society/European Respiratory Society statement: Key concepts and advances in pulmonary rehabilitation. Am. J. Respir. Crit. Care Med. 2013, 188, e13–e64. [Google Scholar] [CrossRef] [PubMed]
- Tremblay, M.S.; Chaput, J.-P.; Adamo, K.B.; Aubert, S.; Barnes, J.D.; Choquette, L.; Duggan, M.; Faulkner, G.; Goldfield, G.S.; Gray, C.E.; et al. Canadian 24-hour movement guidelines for the early years (0–4 years) An integration of physical activity, sedentary behaviour, and sleep. BMC Public Health 2017, 17, 874. [Google Scholar] [CrossRef] [PubMed]
- Gimeno-Santos, E.; Frei, A.; Steurer-Stey, C.; De Batlle, J.; Rabinovich, R.A.; Raste, Y.; Hopkinson, N.S.; Polkey, M.I.; Van Remoortel, H.; Troosters, T.; et al. Determinants and outcomes of physical activity in patients with COPD: A systematic review. Thorax 2014, 69, 731–739. [Google Scholar] [CrossRef] [PubMed]
- Donaire-Gonzalez, D.; Gimeno-Santos, E.; Balcells, E.; Batlle, d.J.; Ramon, M.A.; Rodriguez, E.; Farrero, E.; Benet, M.; Guerra, S.; Sauleda, J.; et al. Benefits of physical activity on COPD hospitalisation depend on intensity. Eur. Respir. J. 2015, 46, 1281–1289. [Google Scholar] [CrossRef] [PubMed]
- Ekelund, U.; Steene-Johannessen, J.; Brown, W.J.; Fagerland, M.W.; Owen, N.; Powell, K.E.; Bauman, A.; Lee, I.-M. Does physical activity attenuate, or even eliminate, the detrimental association of sitting time with mortality? A harmonised meta-analysis of data from more than 1 million men and women. Lancet 2016, 388, 1302–1310. [Google Scholar] [CrossRef]
- Furlanetto, K.C.; Donária, L.; Schneider, L.P.; Lopes, J.R.; Ribeiro, M.; Fernandes, K.B.; Hernandes, N.A.; Pitta, F. Sedentary behavior is an independent predictor of mortality in subjects with COPD. Respir. Care 2017, 62, 579–587. [Google Scholar] [CrossRef]
- Dogra, S.; Good, J.; Buman, M.P.; Gardiner, P.A.; Copeland, J.L.; Stickland, M.K. Physical activity and sedentary time are related to clinically relevant health outcomes among adults with obstructive lung disease. BMC Pulm. Med. 2018, 18, 98. [Google Scholar] [CrossRef]
- Spruit, M.A.; Pitta, F.; McAuley, E.; Zuwallack, R.L.; Nici, L. Pulmonary rehabilitation and physical activity in patients with COPD. Am. J. Respir. Crit. Care Med. 2015, 192, 924–933. [Google Scholar] [CrossRef]
- Vorrink, S.N.W.; Kort, H.S.M.; Troosters, T.; Lammers, J.-W.J. Level of daily physical activity in individuals with COPD compared with healthy controls. Respir. Res. 2011, 12, 33. [Google Scholar] [CrossRef]
- Mantoani, L.C.; Rubio, N.; McKinstry, B.; MacNee, W.; Rabinovich, R.A. Interventions to modify physical activity in patients with COPD: A systematic review. Eur. Respir. J. 2016, 48, 69–81. [Google Scholar] [CrossRef] [PubMed]
- Evenson, K.R.; Herring, A.H.; Wen, F. Accelerometry-assessed latent class patterns of physical activity and sedentary behavior with mortality. Am. J. Prev. Med. 2017, 52, 135–143. [Google Scholar] [CrossRef] [PubMed]
- Mesquita, R.; Spina, G.; Pitta, F.; Donaire-Gonzalez, D.; Deering, B.M.; Patel, M.S.; Mitchell, K.E.; Alison, J.; Van Gestel, A.J.R.; Zogg, S.; et al. Physical activity patterns and clusters in 1001 patients with COPD. Chronic Respir. Dis. 2017, 2, 147997231668720. [Google Scholar] [CrossRef] [PubMed]
- Geidl, W.; Semrau, J.; Streber, R.; Lehbert, N.; Wingart, S.; Tallner, A.; Wittmann, M.; Wagner, R.; Schultz, K.; Pfeifer, K. Effects of a brief, pedometer-based behavioral intervention for individuals with COPD during inpatient pulmonary rehabilitation on 6-week and 6-month objectively measured physical activity: Study protocol for a randomized controlled trial. Trials 2017, 18, 396. [Google Scholar] [CrossRef] [PubMed]
- Ozemek, C.; Kirschner, M.M.; Wilkerson, B.S.; Byun, W.; Kaminsky, L.A. Intermonitor reliability of the GT3X + accelerometer at hip, wrist and ankle sites during activities of daily living. Physiol. Meas. 2014, 35, 129–138. [Google Scholar] [CrossRef] [PubMed]
- Aadland, E.; Ylvisåker, E.; López Lluch, G. Reliability of the actigraph GT3X + accelerometer in adults under free-living conditions. PLoS ONE 2015, 10, e0134606. [Google Scholar] [CrossRef]
- Rabinovich, R.A.; Louvaris, Z.; Raste, Y.; Langer, D.; Van Remoortel, H.; Giavedoni, S.; Burtin, C.; Regueiro, E.M.G.; Vogiatzis, I.; Hopkinson, N.S.; et al. Validity of physical activity monitors during daily life in patients with COPD. Eur. Respir. J. 2013, 42, 1205–1215. [Google Scholar] [CrossRef]
- Van Remoortel, H.; Raste, Y.; Louvaris, Z.; Giavedoni, S.; Burtin, C.; Langer, D.; Wilson, F.; Rabinovich, R.; Vogiatzis, I.; Hopkinson, N.S.; et al. Validity of six activity monitors in chronic obstructive pulmonary disease: A comparison with indirect calorimetry. PLoS ONE 2012, 7, e39198. [Google Scholar] [CrossRef]
- Byrom, B.; Rowe, D.A. Measuring free-living physical activity in COPD patients: Deriving methodology standards for clinical trials through a review of research studies. Contemp. Clin. Trials 2016, 47, 172–184. [Google Scholar] [CrossRef]
- Jones, P.W.; Harding, G.; Berry, P.; Wiklund, I.; Chen, W.-H.; Kline Leidy, N. Development and first validation of the COPD Assessment Test. Eur. Respir. J. 2009, 34, 648–654. [Google Scholar] [CrossRef]
- Holland, A.E.; Spruit, M.A.; Troosters, T.; Puhan, M.A.; Pepin, V.; Saey, D.; McCormack, M.C.; Carlin, B.W.; Sciurba, F.C.; Pitta, F.; et al. An official European Respiratory Society/American Thoracic Society technical standard: Field walking tests in chronic respiratory disease. Eur. Respir. J. 2014, 44, 1428–1446. [Google Scholar] [CrossRef] [PubMed]
- Vogelmeier, C.F.; Criner, G.J.; Martinez, F.J.; Anzueto, A.; Barnes, P.J.; Bourbeau, J.; Celli, B.R.; Chen, R.; Decramer, M.; Fabbri, L.M.; et al. Global strategy for the diagnosis, management, and prevention of chronic obstructive lung disease 2017 report GOLD executive summary. Am. J. Respir. Crit. Care Med. 2017, 49. [Google Scholar] [CrossRef]
- Vestbo, J.; Hurd, S.S.; Agustí, A.G.; Jones, P.W.; Vogelmeier, C.; Anzueto, A.; Barnes, P.J.; Fabbri, L.M.; Martinez, F.J.; Nishimura, M.; et al. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease GOLD executive summary. Am. J. Respir. Crit. Care Med. 2013, 187, 347–365. [Google Scholar] [CrossRef] [PubMed]
- Freedson, P.S.; Melanson, E.; Sirard, J. Calibration of the computer science and applications, inc. accelerometer. Med. Sci. Sports Exerc. 1998, 30, 777–781. [Google Scholar] [CrossRef] [PubMed]
- Gorman, E.; Hanson, H.M.; Yang, P.H.; Khan, K.M.; Liu-Ambrose, T.; Ashe, M.C. Accelerometry analysis of physical activity and sedentary behavior in older adults: A systematic review and data analysis. Eur. Rev. Aging Phys. Act. 2014, 11, 35–49. [Google Scholar] [CrossRef] [PubMed]
- Cain, K.L.; Geremia, C.M. Accelerometer Data Collection and Scoring Manual for Adult & Senior Studies; San Diego State University: San Diego, CA, USA, 2012; Available online: http://sallis.ucsd.edu/measures.html (accessed on 12 March 2018).
- Backhaus, K.; Erichson, B.; Plinke, W.; Weiber, R. Clusteranalyse. In Multivariate Analysemethoden: Eine Anwendungsorientierte Einführung; Backhaus, K., Erichson, B., Plinke, W., Weiber, R., Eds.; Springer: Berlin, Germany, 2016; ISBN 978-3-662-46075-7. [Google Scholar]
- Milligan, G.W.; Cooper, M.C. An Examination of Procedures for Determining the Number of Clusters in a Data Set. Psychometrika 1985, 50, 159–179. [Google Scholar] [CrossRef]
- Calinski, R.B.; Harabasz, J. A dendrite method for cluster analysis. Commun. Stat. 1974, 3, 1–27. [Google Scholar]
- Duda, R.O.; Hart, P.E. Pattern Classification and Scene Analysis; Wiley: New York, NY, USA, 1973; ISBN 978-0471223610. [Google Scholar]
- McVeigh, J.A.; Winkler, E.A.H.; Howie, E.K.; Tremblay, M.S.; Smith, A.; Abbott, R.A.; Eastwood, P.R.; Healy, G.N.; Straker, L.M. Objectively measured patterns of sedentary time and physical activity in young adults of the Raine study cohort. Int. J. Behav. Nutr. Phys. Act. 2016, 13, 41. [Google Scholar] [CrossRef]
- Charrad, M.; Ghazzali, N.; Boiteau, V.; Niknafs, A. NbClust: An R package for determining the relevant number of clusters in a data set. J. Stat. Softw. 2014, 61. [Google Scholar] [CrossRef]
- Depew, Z.S.; Novotny, P.J.; Benzo, R.P. How many steps are enough to avoid severe physical inactivity in patients with chronic obstructive pulmonary disease? Respirology 2012, 17, 1026–1027. [Google Scholar] [CrossRef]
- Depew, Z.S.; Garofoli, A.C.; Novotny, P.J.; Benzo, R.P. Screening for severe physical inactivity in chronic obstructive pulmonary disease: The value of simple measures and the validation of two physical activity questionnaires. Chronic Respir. Dis. 2013, 10, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Garber, C.E.; Blissmer, B.; Deschenes, M.R.; Franklin, B.A.; LAMONTE, M.J.; Lee, I.-M.; Nieman, D.C.; Swain, D.P. American College of Sports Medicine position stand. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults Guidance for prescribing exercise. Med. Sci. Sports Exerc. 2011, 43, 1334–1359. [Google Scholar] [CrossRef] [PubMed]
- Pfeifer, K.; Rütten, A. National Recommendations for Physical Activity and Physical Activity Promotion; FAU University Press: Erlangen, Germany, 2016. [Google Scholar]
- U.S. Department of Health and Human Services. Physical Activity Guidelines for Americans, 2nd ed.; Department of Health and Human Services: Washington, DC, USA, 2018.
- Van Remoortel, H.; Camillo, C.A.; Langer, D.; Hornikx, M.; Demeyer, H.; Burtin, C.; Decramer, M.; Gosselink, R.; Janssens, W.; Troosters, T.; et al. Moderate intense physical activity depends on selected metabolic equivalent of task (MET) cut-off and type of data analysis. PLoS ONE 2013, 8, e84365. [Google Scholar] [CrossRef] [PubMed]
- English, C.; Healy, G.N.; Coates, A.; Lewis, L.; Olds, T.; Bernhardt, J. Sitting and activity time in people with stroke. Phys. Ther. 2016, 96, 193–201. [Google Scholar] [CrossRef] [PubMed]
- Cooper, A.R.; Sebire, S.; Montgomery, A.A.; Peters, T.J.; Sharp, D.J.; Jackson, N.; Fitzsimons, K.; Dayan, C.M.; Andrews, R.C. Sedentary time, breaks in sedentary time and metabolic variables in people with newly diagnosed type 2 diabetes. Diabetologia 2012, 55, 589–599. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, J.E.; Hickey, A.; Staudenmayer, J.; John, D.; Kent, J.A.; Freedson, P.S. Performance of activity classification algorithms in free-living older adults. Med. Sci. Sports Exerc. 2016, 48, 941–950. [Google Scholar] [CrossRef]
- Prince, S.A.; Blanchard, C.M.; Grace, S.L.; Reid, R.D. Objectively-measured sedentary time and its association with markers of cardiometabolic health and fitness among cardiac rehabilitation graduates. Eur. J. Prev. Cardiol. 2016, 23, 818–825. [Google Scholar] [CrossRef]
- Hill, K.; Gardiner, P.A.; Cavalheri, V.; Jenkins, S.C.; Healy, G.N. Physical activity and sedentary behaviour: Applying lessons to chronic obstructive pulmonary disease. Intern. Med. J. 2015, 45, 474–482. [Google Scholar] [CrossRef]
- Dunstan, D.W.; Kingwell, B.A.; Larsen, R.; Healy, G.N.; Cerin, E.; Hamilton, M.T.; Shaw, J.E.; Bertovic, D.A.; Zimmet, P.Z.; Salmon, J.; et al. Breaking up prolonged sitting reduces postprandial glucose and insulin responses. Diabetes Care 2012, 35, 976–983. [Google Scholar] [CrossRef]
- O’Donnell, D.; Gebke, K. Activity restriction in mild COPD: A challenging clinical problem. COPD 2014, 9, 577–588. [Google Scholar] [CrossRef]
- Silva Junior, J.L.; Conde, M.B.; De Sousa Correa, K.; Da Silva, C.; Da Silva Prestes, L.; Rabahi, M.F. COPD Assessment Test (CAT) score as a predictor of major depression among subjects with chronic obstructive pulmonary disease and mild hypoxemia: A case-control study. BMC Pulm. Med. 2014, 14, 186. [Google Scholar] [CrossRef] [PubMed]
- Ghobadi, H.; Ahari, S.S.; Kameli, A.; Lari, S.M. The Relationship between COPD Assessment Test (CAT) Scores and Severity of Airflow Obstruction in Stable COPD Patients. Tanaffos 2012, 11, 22–26. [Google Scholar] [PubMed]
- Ramon, M.A.; Ter Riet, G.; Carsin, A.-E.; Gimeno-Santos, E.; Agustí, A.; Antó, J.M.; Donaire-Gonzalez, D.; Ferrer, J.; Rodríguez, E.; Rodriguez-Roisin, R.; et al. The dyspnoea-inactivity vicious circle in COPD: Development and external validation of a conceptual model. Eur. Respir. J. 2018, 52. [Google Scholar] [CrossRef] [PubMed]
- Shirazipour, C.H.; Evans, M.B.; Leo, J.; Lithopoulos, A.; Martin Ginis, K.A.; Latimer-Cheung, A.E. Program conditions that foster quality physical activity participation experiences for people with a physical disability A systematic review. Disabil. Rehabilit. 2018. [Google Scholar] [CrossRef] [PubMed]
- Arena, R.; McNeil, A.; Street, S.; Bond, S.; Laddu, D.R.; Lavie, C.J.; Hills, A.P. Let us talk about moving: Reframing the exercise and physical activity discussion. Curr. Probl. Cardiol. 2018, 43, 154–179. [Google Scholar] [CrossRef] [PubMed]
- Moy, M.L.; Gould, M.K.; Liu, I.-L.A.; Lee, J.S.; Nguyen, H.Q. Physical activity assessed in routine care predicts mortality after a COPD hospitalisation. ERJ Open Res. 2016, 2. [Google Scholar] [CrossRef] [PubMed]
- Warburton, D.E.R.; Bredin, S.S.D. Reflections on Physical Activity and Health: What Should We Recommend? Can. J. Cardiol. 2016, 32, 495–504. [Google Scholar] [CrossRef] [PubMed]
- Blondeel, A.; Demeyer, H.; Janssens, W.; Troosters, T. The role of physical activity in the context of pulmonary rehabilitation. COPD 2019. [Google Scholar] [CrossRef]
- Burge, A.T.; Cox, N.S.; Abramson, M.J.; Holland, A.E. Interventions for promoting physical activity in people with COPD. Cochrane Database Syst. Rev. 2017, 5, 20415. [Google Scholar] [CrossRef]
- Shioya, T.; Sato, S.; Iwakura, M.; Takahashi, H.; Terui, Y.; Uemura, S.; Satake, M. Improvement of physical activity in chronic obstructive pulmonary disease by pulmonary rehabilitation and pharmacological treatment. Respir. Investig. 2018, 56, 292–306. [Google Scholar] [CrossRef]
- Benatti, F.B.; Ried-Larsen, M. The Effects of Breaking up Prolonged Sitting Time: A Review of Experimental Studies. Med. Sci. Sports Exerc. 2015, 47, 2053–2061. [Google Scholar] [CrossRef] [PubMed]
- Cavalheri, V.; Straker, L.; Gucciardi, D.F.; Gardiner, P.A.; Hill, K. Changing physical activity and sedentary behaviour in people with COPD. Respirology 2016, 21, 419–426. [Google Scholar] [CrossRef] [PubMed]
- Lewthwaite, H.; Effing, T.W.; Olds, T.; Williams, M.T. Physical activity, sedentary behaviour and sleep in COPD guidelines: A systematic review. Chronic Respir. Dis. 2017, 14, 231–244. [Google Scholar] [CrossRef] [PubMed]
- Demeyer, H.; Burtin, C.; Van Remoortel, H.; Hornikx, M.; Langer, D.; Decramer, M.; Gosselink, R.; Janssens, W.; Troosters, T. Standardizing the analysis of physical activity in patients with COPD following a pulmonary rehabilitation program. Chest 2014, 146, 318–327. [Google Scholar] [CrossRef] [PubMed]
- Alahmari, A.D.; Mackay, A.J.; Patel, A.R.C.; Kowlessar, B.S.; Singh, R.; Brill, S.E.; Allinson, J.P.; Wedzicha, J.A.; Donaldson, G.C. Influence of weather and atmospheric pollution on physical activity in patients with COPD. Respir. Res. 2015, 16, 786. [Google Scholar] [CrossRef] [PubMed]
- Pitta, F.; Breyer, M.-K.; Hernandes, N.A.; Teixeira, D.; Sant’Anna, T.J.P.; Fontana, A.D.; Probst, V.S.; Brunetto, A.F.; Spruit, M.A.; Wouters, E.F.M.; et al. Comparison of daily physical activity between COPD patients from Central Europe and South America. Respir. Med. 2009, 103, 421–426. [Google Scholar] [CrossRef] [PubMed]
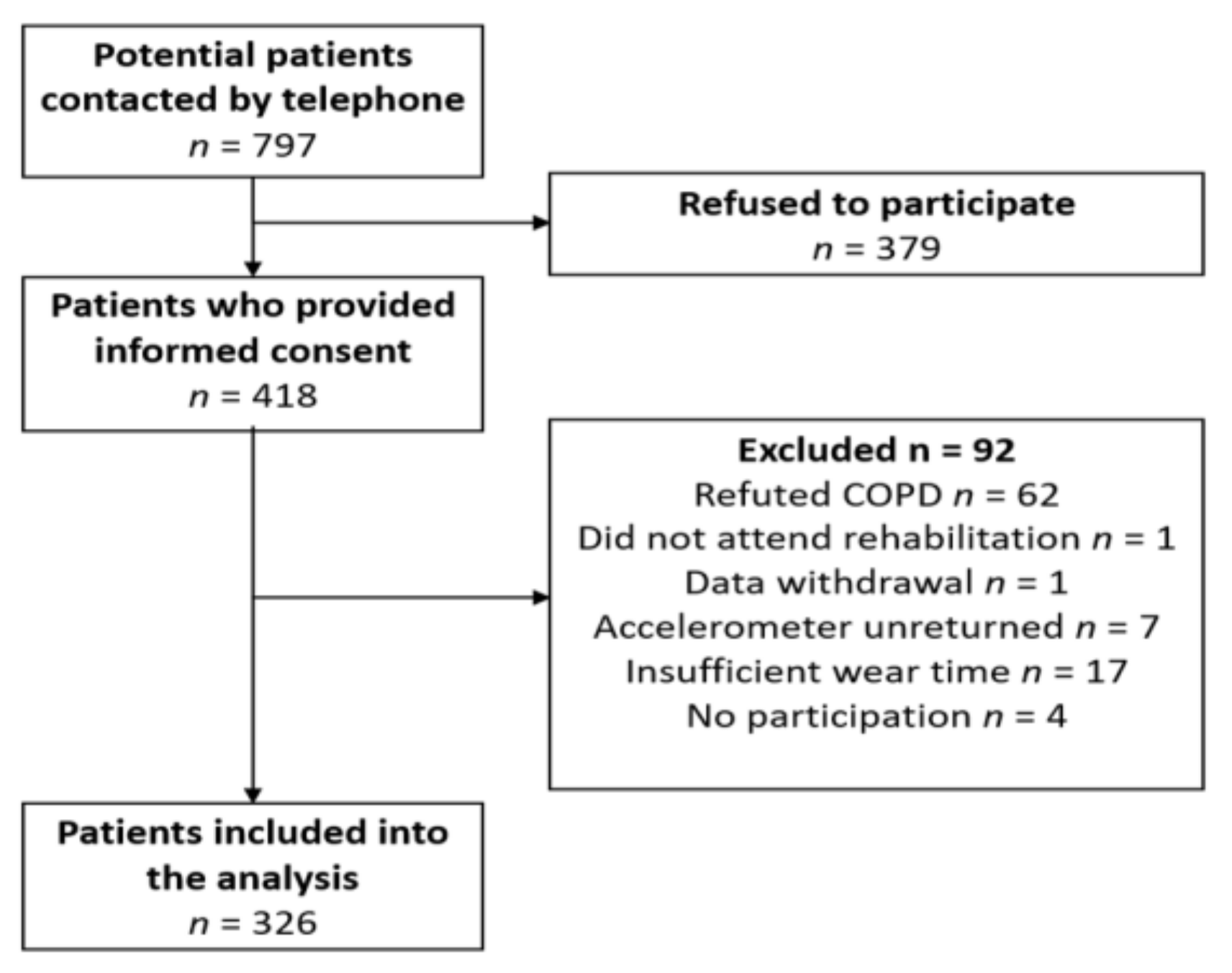

| n | 326 |
|---|---|
| Age (years) | 58.2 ± 5.6 |
| Sex (% male) | 67.8% |
| Height (cm) | 170 ± 9.2 |
| Weight (kg) | 80.2 ± 20.9 |
| BMI (kg/m2) | 27.4 ± 6.5 |
| FEV1 (%) | 53.7 ± 18.2 |
| GOLD Severity Classification 1/2/3/4 (%) | 9.3/44.4/38.0/8.3 |
| GOLD Classification A/B/C/D (%) | 1.7/44.2/0/53.6 |
| COPD Assessment Test (CAT Score) | 23.37 ± 6.71 |
| Number of Comorbidities | 4.48 ± 2.58 |
| Percentage of Current Smokers | 45.8% |
| Employed | 75.3% |
| Sedentary Behaviour (min/day) | 559.4 ± 92.9 |
| Moderate-to-Vigorous Physical Activity (min/week) | 204.3 ± 160.9 |
| Number of Steps (per day) | 5803 ± 3051 |
| Cluster 1 (Sedentary Non-Movers) | Cluster 2 (Sedentary occasional Movers) | Cluster 3 (Sedentary Movers) | Cluster 4 (Sedentary Exercisers) | df | F | p | Effect Size η2 | |
|---|---|---|---|---|---|---|---|---|
| General Characteristics | ||||||||
| N | 93 | 136 | 64 | 33 | ||||
| % | 28.5% | 41.7% | 19.6% | 10.1% | ||||
| Age (years) | 59.5 (6.1) | 58.0 (6.0) | 57.1 (4.1) | 57.4 (4.5) | 3, 322 | 2.70 | 0.046 | 0.025 |
| Sex (% male) a | 72.0 | 68.1 | 64.1 | 65.6 | 3 | 1.24 a | 0.744 a | - |
| BMI (kg/m2) | 27.39 (7.27) | 27.44 (6.68) | 27.40 (5.81) | 27.19 (5.14) | 3, 312 | 0.012 | 0.998 | - |
| FEV1 (%) | 43.10 (13.81) | 54.25 (17.95) | 61.82 (17.47) | 64.89 (16.71) | 3, 113.0 | 25.61 | <0.001 | 0.175 |
| GOLD 1/2/3/4 (%) a | 0/31.8/50.0/18.2 | 7.7/46.9/39.2/6.2 | 19.4/50.0/29.0/1.6 | 21.2/57.6/18.2/3.0 | 9 | 49.082 a | <0.001 | V = 0.226 a |
| GOLD A/B/C/D (%) a | 0/37.6/0/62.4 | 1.6/49.2/0/49.2 | 1.8/49.1/0/49.1 | 3.3/46.7/0/50.0 | 6 | 5.81 a | 0.445 | - |
| Comorbidities | 4.37 (2.77) | 4.29 (2.47) | 4.92 (2.60) | 4.76 (2.42) | 3, 322 | 1.05 | 0.371 | - |
| COPD Assessment Test | 26.37 (5.84) | 22.34 (6.74) | 22.63 (7.37) | 20.59 (4.68) | 3, 118.7 | 12.76 | <0.001 | 0.087 |
| 6-Min-Walk-Test (in m) | 386.1 (103.8) | 458.7 (97.6) | 496.7 (71.5) | 503.3 (79.1) | 3, 298 | 22.23 | <0.001 | 0.183 |
| Physical Activity and Sedentary Behaviour (min/day) | ||||||||
| Very Light PA | 102.97 (26.78) | 147.82 (32.29) | 191.61 (35.20) | 210.15 (55.3) | 3, 106.6 | 123.8 | <0.001 | 0.533 |
| Light PA | 25.00 (10.89) | 44.81 (10.95) | 70.22 (12.46) | 93.92 (18.55) | 3, 106.7 | 265.0 | <0.001 | 0.760 |
| Moderate PA | 9.23 (6.09) | 27.60 (16.12) | 38.49 (11.88) | 69.76 (23.38) | 3, 102.9 | 193.3 | <0.001 | 0.600 |
| Vigorous PA | 0.13 (0.21) | 0.34 (1.50) | 0.36 (0.88) | 1.64 (3.76) | 3, 94.4 | 3.94 | 0.011 | 0.067 |
| Steps (per day) | 2749 (1064) | 5649 (1826) | 7866 (1786) | 11045 (2621) | 3, 106.1 | 243.3 | <0.001 | 0.678 |
| Overall Sedentary Time | 644.33 (76.02) | 561.46 (59.93) | 490.22 (60.43) | 445.58 (68.54) | 3, 112.8 | 94.5 | <0.001 | 0.501 |
| Wear Time (min/day) | 780.1 (87.2) | 781.4 (73.7) | 793.1 (82.2) | 819.7 (68.0) | 3, 322 | 2.46 | 0.063 | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Geidl, W.; Carl, J.; Cassar, S.; Lehbert, N.; Mino, E.; Wittmann, M.; Wagner, R.; Schultz, K.; Pfeifer, K. Physical Activity and Sedentary Behaviour Patterns in 326 Persons with COPD before Starting a Pulmonary Rehabilitation: A Cluster Analysis. J. Clin. Med. 2019, 8, 1346. https://doi.org/10.3390/jcm8091346
Geidl W, Carl J, Cassar S, Lehbert N, Mino E, Wittmann M, Wagner R, Schultz K, Pfeifer K. Physical Activity and Sedentary Behaviour Patterns in 326 Persons with COPD before Starting a Pulmonary Rehabilitation: A Cluster Analysis. Journal of Clinical Medicine. 2019; 8(9):1346. https://doi.org/10.3390/jcm8091346
Chicago/Turabian StyleGeidl, Wolfgang, Johannes Carl, Samuel Cassar, Nicola Lehbert, Eriselda Mino, Michael Wittmann, Rupert Wagner, Konrad Schultz, and Klaus Pfeifer. 2019. "Physical Activity and Sedentary Behaviour Patterns in 326 Persons with COPD before Starting a Pulmonary Rehabilitation: A Cluster Analysis" Journal of Clinical Medicine 8, no. 9: 1346. https://doi.org/10.3390/jcm8091346
APA StyleGeidl, W., Carl, J., Cassar, S., Lehbert, N., Mino, E., Wittmann, M., Wagner, R., Schultz, K., & Pfeifer, K. (2019). Physical Activity and Sedentary Behaviour Patterns in 326 Persons with COPD before Starting a Pulmonary Rehabilitation: A Cluster Analysis. Journal of Clinical Medicine, 8(9), 1346. https://doi.org/10.3390/jcm8091346





