A Mechanistic and Pathophysiological Approach for Stroke Associated with Drugs of Abuse
Abstract
1. Introduction
1.1. Stroke Definitions
1.2. Epidemiology of Illicit Drugs of Abuse Use and Stroke
1.3. Classic Concept of Stroke Pathophysiology
2. Stroke Linked to Illicit Drugs of Abuse
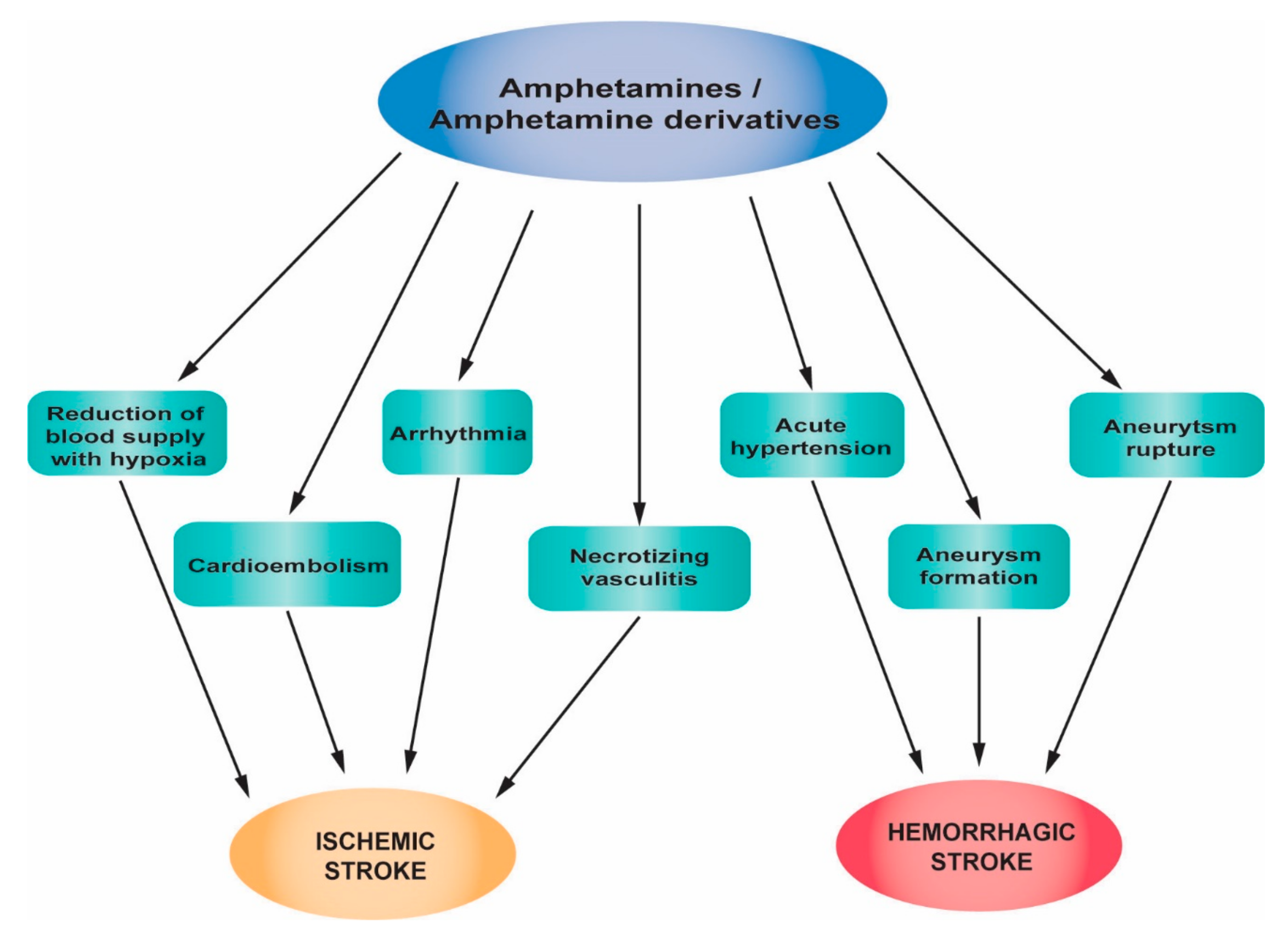
2.1. Amphetamines and Amphetamine Derivatives
2.1.1. Mechanisms of Actions of Amphetamines and Amphetamine Derivatives
2.1.2. Influence of Amphetamines and Amphetamine Derivatives on Stroke
2.1.3. Clinical Studies, Case Reports and Epidemiology of Stroke Related to Amphetamines and Amphetamine Derivatives abuse
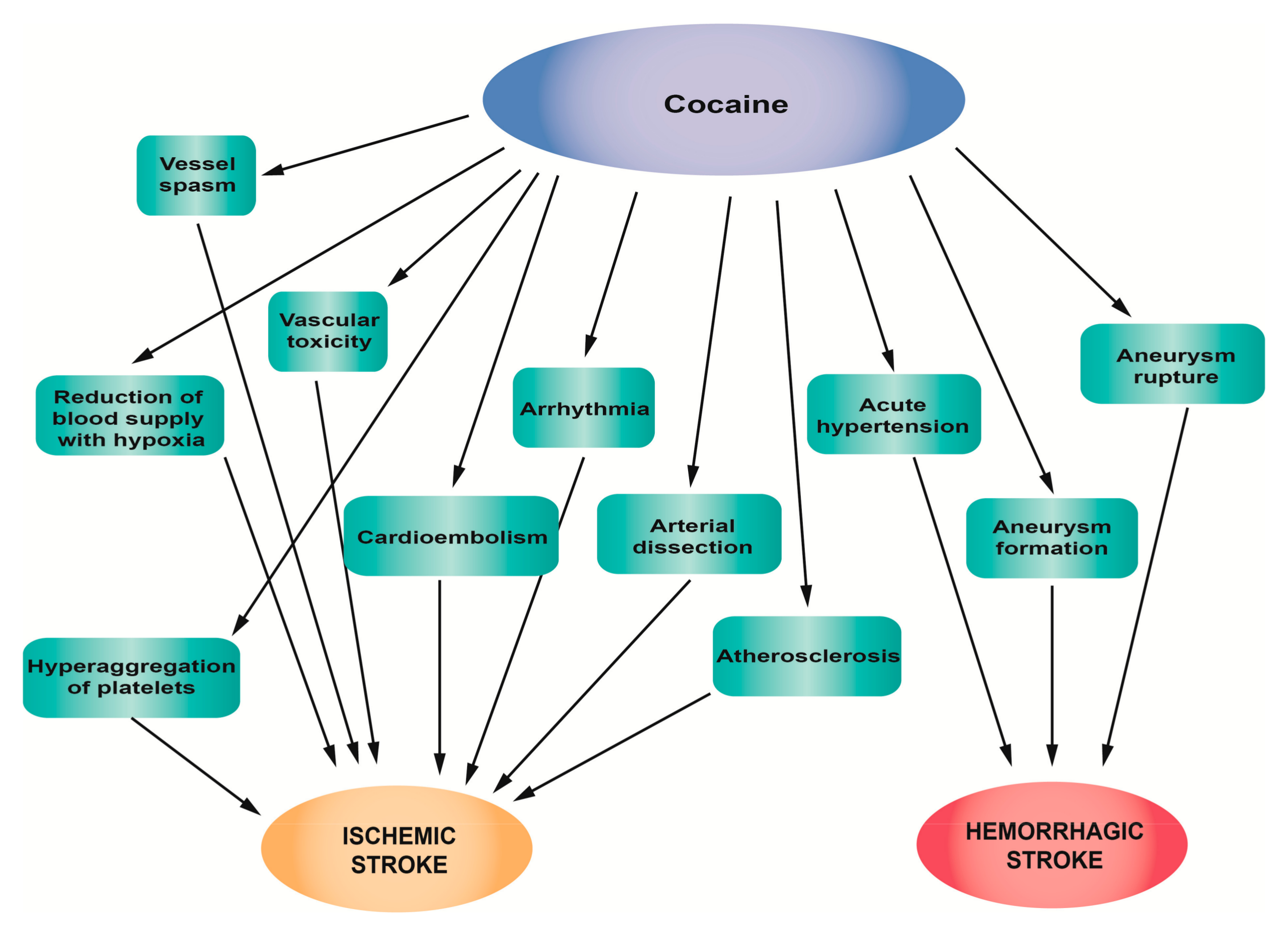
2.2. Cocaine
2.2.1. The Mechanism of Action of Cocaine
2.2.2. Influence of Cocaine on Stroke
2.2.3. Clinical Studies, Case Reports and Epidemiology of Stroke Related to Cocaine Abuse
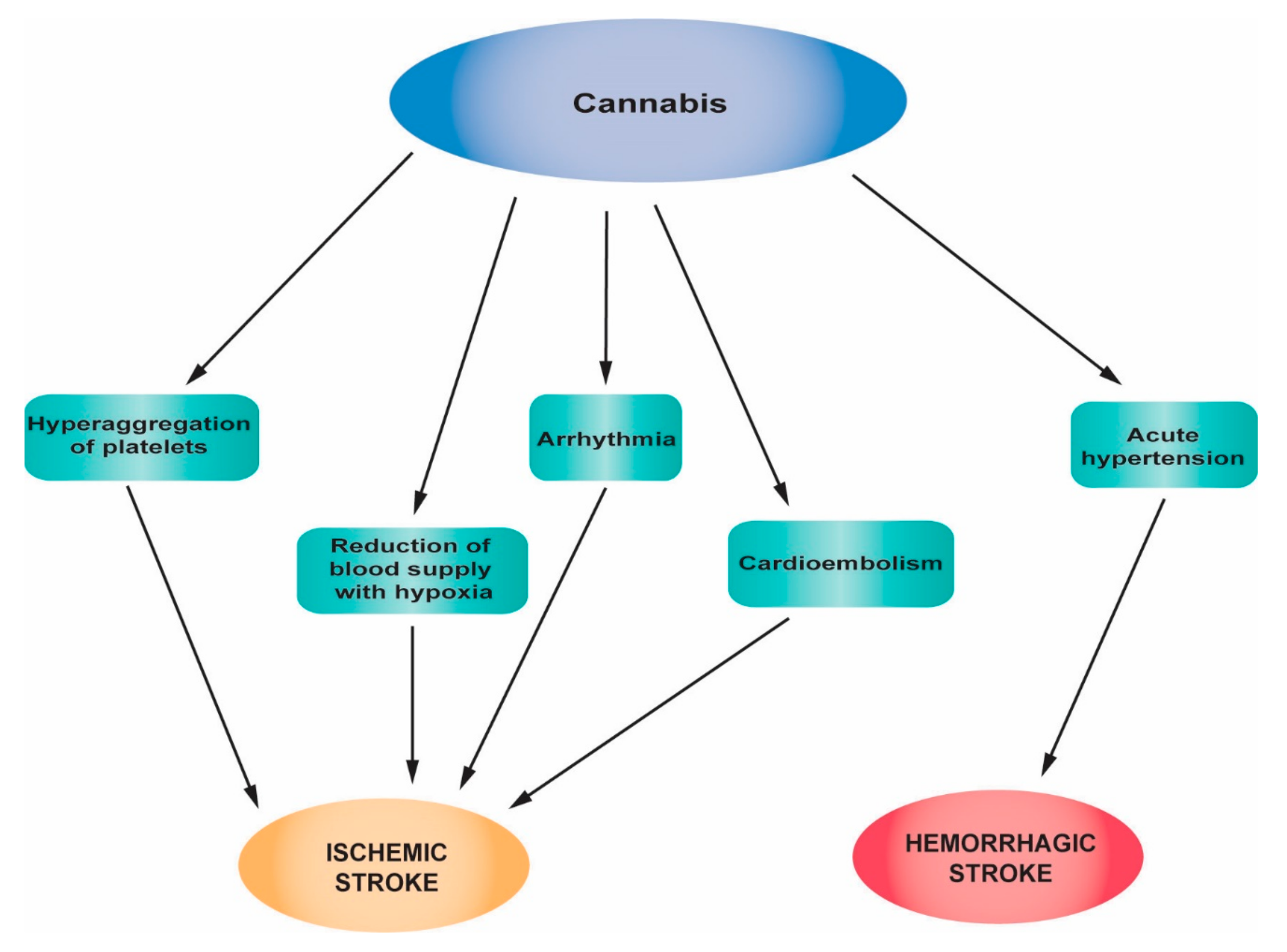
2.3. Cannabis
2.3.1. The Mechanism of Action of Cannabis
2.3.2. The Influence of Cannabis on Stroke
2.3.3. Clinical Studies, Case Reports and Epidemiology of Stroke Related to Cannabis
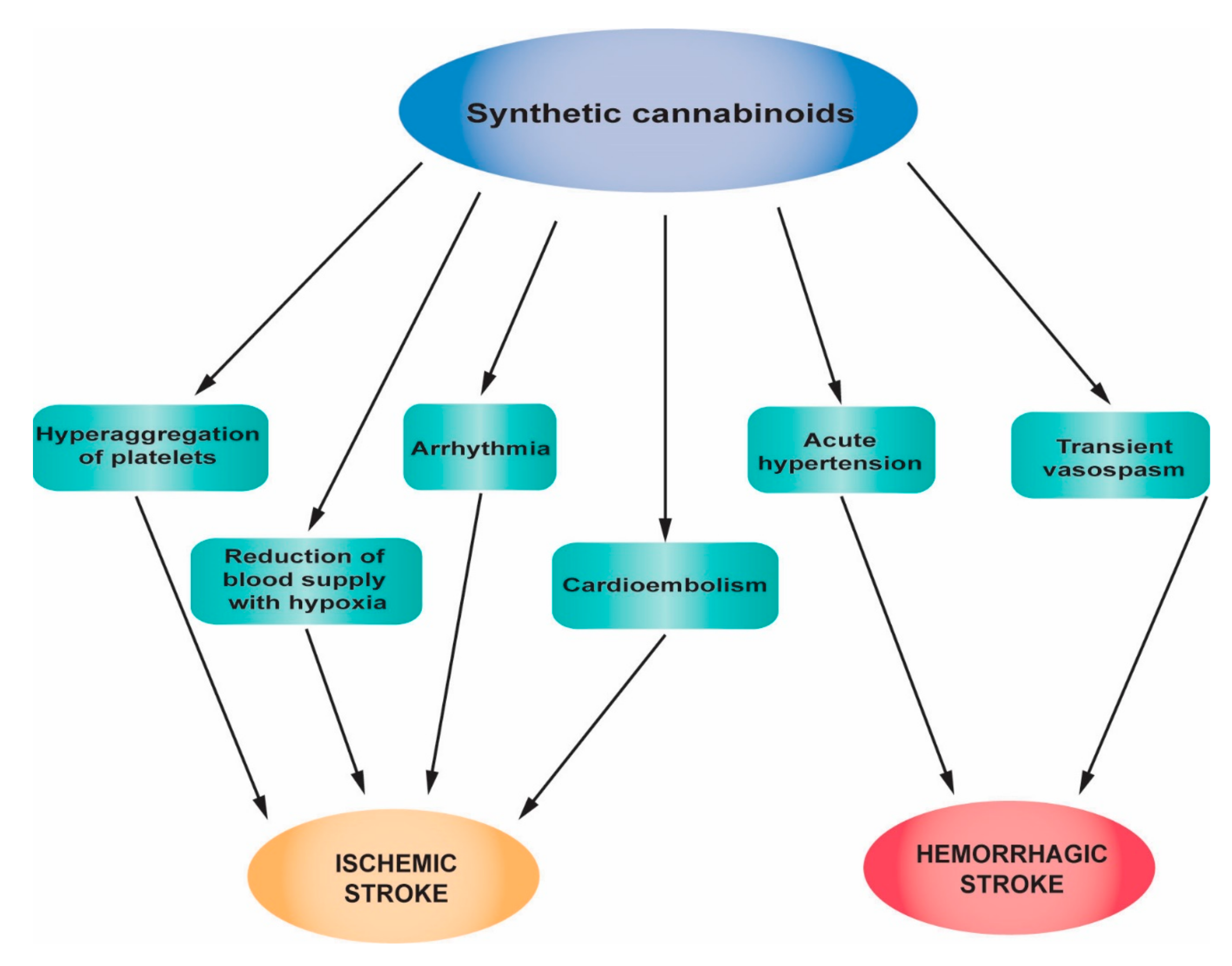
2.4. Synthetic Cannabinoids
2.4.1. The Mechanism of Action of the Synthetic Cannabinoids
2.4.2. The Influence of Synthetic Cannabinoids on Stroke
2.4.3. Clinical Studies, Case Reports and Epidemiology of Stroke Related to Synthetic Cannabinoid Abuse
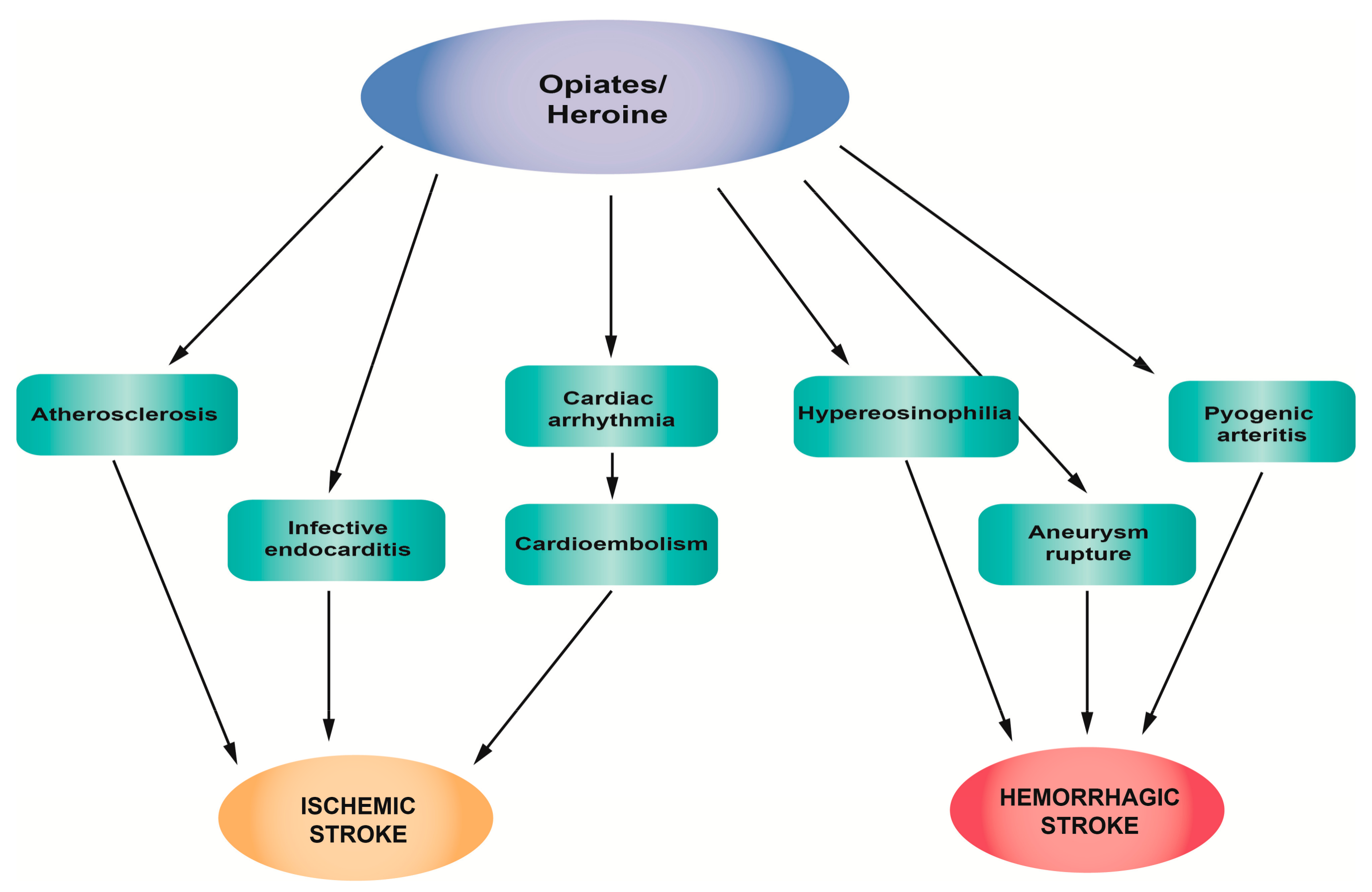
2.5. Opiates/Heroin
2.5.1. The Mechanism of Action of Opiates/Heroin
2.5.2. The Influence of Opiates/Heroin on Stroke
2.5.3. Clinical Studies, Case Reports and Epidemiology of Stroke Related to Opiates/Heroin Abuse
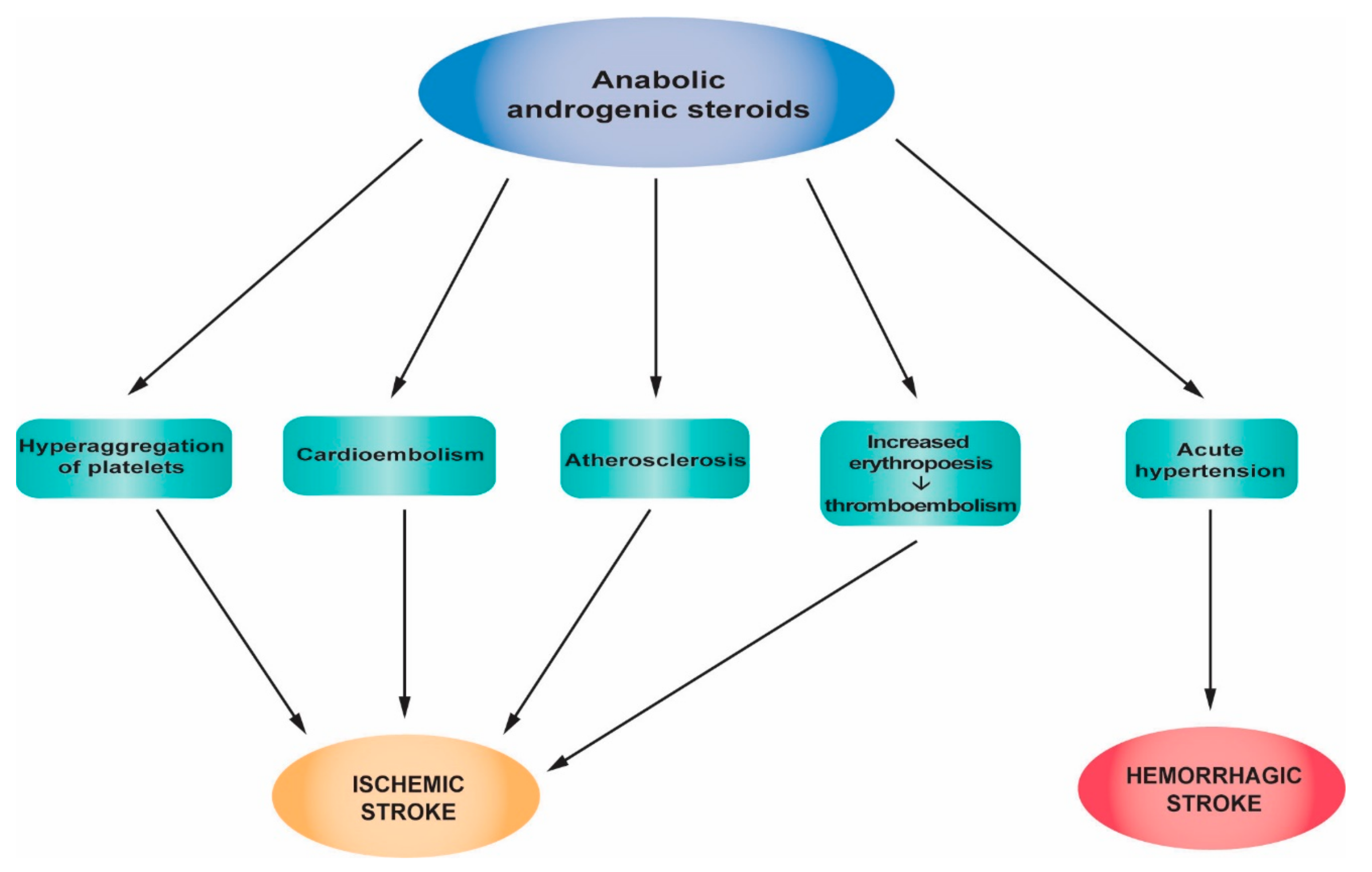
2.6. Androgenic Anabolic Steroids
2.6.1. The Mechanism of Action of AASs
2.6.2. The Influence of AASs on Stroke
2.6.3. Clinical Studies, Case Reports and Epidemiology of Stroke Related to AAS Abuse
3. Management
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Easton, J.D.; Saver, J.L.; Albers, G.W.; Alberts, M.J.; Chaturvedi, S.; Feldmann, E.; Hatsukami, T.S.; Higashida, R.T.; Johnston, S.C.; Kidwell, C.S.; et al. Definition and evaluation of transient ischemic attack: A scientific statement for healthcare professionals from the American Heart Association/American Stroke Association Stroke Council; Council on Cardiovascular Surgery and Anesthesia; Council on Cardiovascular Radiology and Intervention; Council on Cardiovascular Nursing; and the Interdisciplinary Council on Peripheral Vascular Disease. The American Academy of Neurology affirms the value of this statement as an educational tool for neurologists. Stroke 2009, 40, 2276–2293. [Google Scholar] [CrossRef] [PubMed]
- National Collaborating Centre for Chronic Conditions. National Collaborating Centre for Chronic Conditions. National Institute for Health and Clinical Excellence: Guidance. In Stroke: National Clinical Guideline for Diagnosis and Initial Management of Acute Stroke and Transient Ischaemic Attack (TIA); Royal College of Physicians (UK) Royal College of Physicians of London: London, UK, 2008. [Google Scholar]
- Johnson, W.; Onuma, O.; Owolabi, M.; Sachdev, S. Stroke: A global response is needed. Bull. World Health Organ. 2016, 94. [Google Scholar] [CrossRef] [PubMed]
- United Nations Oddice on Drugs and Crime. World Drug Report 2017. Available online: https://www.unodc.org/wdr2017/field/WDR_2017_presentation_lauch_version.pdf (accessed on 26 February 2019).
- Smajlovic, D. Strokes in young adults: Epidemiology and prevention. Vasc. Health Risk Manag. 2015, 11, 157–164. [Google Scholar] [CrossRef] [PubMed]
- Mendis, S. Stroke disability and rehabilitation of stroke: World Health Organization perspective. Int. J. Stroke 2013, 8, 3–4. [Google Scholar] [CrossRef] [PubMed]
- Jivan, K.; Ranchod, K.; Modi, G. Management of ischaemic stroke in the acute setting: Review of the current status. Cardiovasc. J. Afr. 2013, 24, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Falkstedt, D.; Wolff, V.; Allebeck, P.; Hemmingsson, T.; Danielsson, A.K. Cannabis, Tobacco, Alcohol Use, and the Risk of Early Stroke: A Population-Based Cohort Study of 45,000 Swedish Men. Stroke 2017, 48, 265–270. [Google Scholar] [CrossRef] [PubMed]
- Sloan, M.A. Illicit drug use/abuse and stroke. Handb. Clin. Neurol. 2009, 93, 823–840. [Google Scholar] [CrossRef] [PubMed]
- Nieswandt, B.; Kleinschnitz, C.; Stoll, G. Ischaemic stroke: A thrombo-inflammatory disease? J. Physiol. 2011, 589, 4115–4123. [Google Scholar] [CrossRef]
- Stoll, G.; Nieswandt, B. Thrombo-inflammation in acute ischaemic stroke—Implications for treatment. Nat. Rev. Neurol. 2019, 15, 473–481. [Google Scholar] [CrossRef]
- Slomka, A.; Switonska, M.; Sinkiewicz, W.; Zekanowska, E. Assessing Circulating Factor VIIa-Antithrombin Complexes in Acute Ischemic Stroke: A Pilot Study. Clin. Appl. Thromb. Hemost. 2017, 23, 351–359. [Google Scholar] [CrossRef]
- Switonska, M.; Slomka, A.; Sinkiewicz, W.; Zekanowska, E. Tissue-factor-bearing microparticles (MPs-TF) in patients with acute ischaemic stroke: The influence of stroke treatment on MPs-TF generation. Eur. J. Neurol. 2015, 22, e328–e399. [Google Scholar] [CrossRef] [PubMed]
- Brouwers, H.B.; Greenberg, S.M. Hematoma expansion following acute intracerebral hemorrhage. Cerebrovasc. Dis. 2013, 35, 195–201. [Google Scholar] [CrossRef] [PubMed]
- Sonni, S.; Lioutas, V.A.; Selim, M.H. New avenues for treatment of intracranial hemorrhage. Curr. Treat. Options Cardiovasc. Med. 2014, 16, 277. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Dardiotis, E.; Aloizou, A.M.; Markoula, S.; Siokas, V.; Tsarouhas, K.; Tzanakakis, G.; Libra, M.; Kyritsis, A.P.; Brotis, A.G.; Aschner, M.; et al. Cancer-associated stroke: Pathophysiology, detection and management (Review). Int. J. Oncol. 2019, 54, 779–796. [Google Scholar] [CrossRef]
- Teodoro, M.; Briguglio, G.; Fenga, C.; Costa, C. Genetic polymorphisms as determinants of pesticide toxicity: Recent advances. Toxicol. Rep. 2019. [Google Scholar] [CrossRef] [PubMed]
- Kaye, S.; Darke, S.; Duflou, J.; McKetin, R. Methamphetamine-related fatalities in Australia: Demographics, circumstances, toxicology and major organ pathology. Addiction 2008, 103, 1353–1360. [Google Scholar] [CrossRef]
- Kaku, D.A.; Lowenstein, D.H. Emergence of recreational drug abuse as a major risk factor for stroke in young adults. Ann. Intern. Med. 1990, 113, 821–827. [Google Scholar] [CrossRef]
- Ho, E.L.; Josephson, S.A.; Lee, H.S.; Smith, W.S. Cerebrovascular complications of methamphetamine abuse. Neurocrit. Care 2009, 10, 295–305. [Google Scholar] [CrossRef]
- Buttner, A. Review: The neuropathology of drug abuse. Neuropathol. Appl. Neurobiol. 2011, 37, 118–134. [Google Scholar] [CrossRef]
- Sloan, M.A.; Kittner, S.J.; Rigamonti, D.; Price, T.R. Occurrence of stroke associated with use/abuse of drugs. Neurology 1991, 41, 1358–1364. [Google Scholar] [CrossRef]
- Parrott, A.C.; Milani, R.M.; Gouzoulis-Mayfrank, E.; Daumann, J. Cannabis and Ecstasy/MDMA (3,4-methylenedioxymethamphetamine): An analysis of their neuropsychobiological interactions in recreational users. J. Neural Transm. (Vienna, Austria: 1996) 2007, 114, 959–968. [Google Scholar] [CrossRef] [PubMed]
- Heal, D.J.; Smith, S.L.; Gosden, J.; Nutt, D.J. Amphetamine, past and present—A pharmacological and clinical perspective. J. Psychopharmacol. (Oxford, UK) 2013, 27, 479–496. [Google Scholar] [CrossRef] [PubMed]
- Walker-Batson, D.; Mehta, J.; Smith, P.; Johnson, M. Amphetamine and other pharmacological agents in human and animal studies of recovery from stroke. Prog. Neuropsychopharmacol. Biol. Psychiatry 2016, 64, 225–230. [Google Scholar] [CrossRef] [PubMed]
- Calipari, E.S.; Ferris, M.J. Amphetamine mechanisms and actions at the dopamine terminal revisited. J. Neurosci. 2013, 33, 8923–8925. [Google Scholar] [CrossRef] [PubMed]
- Christophersen, A.S. Amphetamine designer drugs—An overview and epidemiology. Toxicol. Lett. 2000, 112–113, 127–131. [Google Scholar] [CrossRef]
- Albertson, T.E.; Derlet, R.W.; Van Hoozen, B.E. Methamphetamine and the expanding complications of amphetamines. West. J. Med. 1999, 170, 214–219. [Google Scholar] [PubMed]
- Westover, A.N.; McBride, S.; Haley, R.W. Stroke in young adults who abuse amphetamines or cocaine: A population-based study of hospitalized patients. Arch. Gen. Psychiatry 2007, 64, 495–502. [Google Scholar] [CrossRef]
- Phillips, M.C.; Leyden, J.M.; Chong, W.K.; Kleinig, T.; Czapran, P.; Lee, A.; Koblar, S.A.; Jannes, J. Ischaemic stroke among young people aged 15 to 50 years in Adelaide, South Australia. Med. J. Aust. 2011, 195, 610–614. [Google Scholar] [CrossRef]
- Indave, B.I.; Sordo, L.; Bravo, M.J.; Sarasa-Renedo, A.; Fernandez-Balbuena, S.; De la Fuente, L.; Sonego, M. Risk of stroke in prescription and other amphetamine-type stimulants use: A systematic review. Drug Alcohol Rev. 2018, 37, 56–69. [Google Scholar] [CrossRef]
- Ohta, K.; Mori, M.; Yoritaka, A.; Okamoto, K.; Kishida, S. Delayed ischemic stroke associated with methamphetamine use. J. Emerg. Med. 2005, 28, 165–167. [Google Scholar] [CrossRef]
- De Silva, D.A.; Wong, M.C.; Lee, M.P.; Chen, C.L.; Chang, H.M. Amphetamine-associated ischemic stroke: Clinical presentation and proposed pathogenesis. J. Stroke Cerebrovasc. Dis. 2007, 16, 185–186. [Google Scholar] [CrossRef] [PubMed]
- Weiss, S.R.; Raskind, R.; Morganstern, N.L.; Pytlyk, P.J.; Baiz, T.C. Intracerebral and subarachnoid hemorrhage following use of methamphetamine (“speed”). Int. J. Surg. 1970, 53, 123–127. [Google Scholar]
- Yakoot, M. Phenylpropanolamine and the hemorrhagic stroke: A new search for the culprit. J. Pharmacol. Pharmacother. 2012, 3, 4–6. [Google Scholar] [CrossRef] [PubMed]
- Polesskaya, O.; Silva, J.; Sanfilippo, C.; Desrosiers, T.; Sun, A.; Shen, J.; Feng, C.; Polesskiy, A.; Deane, R.; Zlokovic, B.; et al. Methamphetamine causes sustained depression in cerebral blood flow. Brain Res. 2011, 1373, 91–100. [Google Scholar] [CrossRef] [PubMed]
- Berlit, P. Diagnosis and treatment of cerebral vasculitis. Ther. Adv. Neurol. Disord. 2010, 3, 29–42. [Google Scholar] [CrossRef] [PubMed]
- McGee, S.M.; McGee, D.N.; McGee, M.B. Spontaneous intracerebral hemorrhage related to methamphetamine abuse: Autopsy findings and clinical correlation. Am. J. Forensic Med. Pathol. 2004, 25, 334–337. [Google Scholar] [CrossRef] [PubMed]
- Pilgrim, J.L.; Gerostamoulos, D.; Drummer, O.H.; Bollmann, M. Involvement of amphetamines in sudden and unexpected death. J. Forensic Sci. 2009, 54, 478–485. [Google Scholar] [CrossRef] [PubMed]
- Lappin, J.M.; Darke, S.; Farrell, M. Stroke and methamphetamine use in young adults: A review. J. Neurol. Neurosurg. Psychiatry 2017, 88, 1079–1091. [Google Scholar] [CrossRef]
- Huang, M.C.; Yang, S.Y.; Lin, S.K.; Chen, K.Y.; Chen, Y.Y.; Kuo, C.J.; Hung, Y.N. Risk of Cardiovascular Diseases and Stroke Events in Methamphetamine Users: A 10-Year Follow-Up Study. J. Clin. Psychiatry 2016, 77, 1396–1403. [Google Scholar] [CrossRef]
- Christensen, M.R.; Lesnikova, I.; Madsen, L.B.; Rosendal, I.; Banner, J. Drug-induced bilateral ischemic infarction in an amphetamine addict. Forensic Sci. Med. Pathol. 2013, 9, 458–461. [Google Scholar] [CrossRef]
- Kahn, D.E.; Ferraro, N.; Benveniste, R.J. 3 cases of primary intracranial hemorrhage associated with “Molly”, a purified form of 3,4-methylenedioxymethamphetamine (MDMA). J. Neurol. Sci. 2012, 323, 257–260. [Google Scholar] [CrossRef] [PubMed]
- Wijers, C.H.W.; Visser, M.C.; van Litsenburg, R.T.H.; Niesink, R.J.M.; Willemse, R.B.; Croes, E.A. Haemorrhagic stroke related to the use of 4-fluoroamphetamine. J. Neurol. 2018, 265, 1607–1611. [Google Scholar] [CrossRef] [PubMed]
- Chiu, Z.K.; Bennett, I.E.; Chan, P.; Rosenfeld, J.V. Methamphetamine-related brainstem haemorrhage. J. Clin. Neurosci. 2016, 32, 137–139. [Google Scholar] [CrossRef] [PubMed]
- Kapetanovic, S.; Kim, M.A. Hemorrhagic stroke in a patient recently started on mixed amphetamine salts. Am. J. Psychiatry 2010, 167, 1277–1278. [Google Scholar] [CrossRef] [PubMed]
- Lyson, T.; Kochanowicz, J.; Rutkowski, R.; Turek, G.; Lewko, J. Cerebral vasospasm in patient with hemorrhagic stroke after amphetamine intake—Case report. Pol. Merkur. Lek. Organ Pol. Tow. Lek. 2008, 24, 265–267. [Google Scholar]
- Muntan, C.D.; Tuckler, V. Cerebrovascular accident following MDMA ingestion. J. Med. Toxicol. 2006, 2, 16–18. [Google Scholar] [CrossRef] [PubMed][Green Version]
- McIntosh, A.; Hungs, M.; Kostanian, V.; Yu, W. Carotid artery dissection and middle cerebral artery stroke following methamphetamine use. Neurology 2006, 67, 2259–2260. [Google Scholar] [CrossRef]
- Treadwell, S.D.; Robinson, T.G. Cocaine use and stroke. Postgrad. Med. J. 2007, 83, 389–394. [Google Scholar] [CrossRef]
- Kishi, T.; Matsuda, Y.; Iwata, N.; Correll, C.U. Antipsychotics for cocaine or psychostimulant dependence: Systematic review and meta-analysis of randomized, placebo-controlled trials. J. Clin. Psychiatry 2013, 74, e1169–e1180. [Google Scholar] [CrossRef]
- Riezzo, I.; Fiore, C.; De Carlo, D.; Pascale, N.; Neri, M.; Turillazzi, E.; Fineschi, V. Side effects of cocaine abuse: Multiorgan toxicity and pathological consequences. Curr. Med. Chem. 2012, 19, 5624–5646. [Google Scholar] [CrossRef]
- Bachi, K.; Mani, V.; Jeyachandran, D.; Fayad, Z.A.; Goldstein, R.Z.; Alia-Klein, N. Vascular disease in cocaine addiction. Atherosclerosis 2017, 262, 154–162. [Google Scholar] [CrossRef] [PubMed]
- Sordo, L.; Indave, B.I.; Barrio, G.; Degenhardt, L.; de la Fuente, L.; Bravo, M.J. Cocaine use and risk of stroke: A systematic review. Drug Alcohol Depend. 2014, 142, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, P.; Taraman, S.; Shankar, L.; Chaturvedi, S.; Madhavan, R. Clinical Profiles, Complications, and Disability in Cocaine-Related Ischemic Stroke. J. Stroke Cerebrovasc. Dis. 2011, 20, 443–449. [Google Scholar] [CrossRef] [PubMed]
- Pradhan, L.; Mondal, D.; Chandra, S.; Ali, M.; Agrawal, K.C. Molecular analysis of cocaine-induced endothelial dysfunction: Role of endothelin-1 and nitric oxide. Cardiovasc. Toxicol. 2008, 8, 161–171. [Google Scholar] [CrossRef] [PubMed]
- He, G.Q.; Zhang, A.; Altura, B.T.; Altura, B.M. Cocaine-induced cerebrovasospasm and its possible mechanism of action. J. Pharmacol. Exp. Ther. 1994, 268, 1532–1539. [Google Scholar] [PubMed]
- You, J.; Du, C.; Volkow, N.D.; Pan, Y. Optical coherence Doppler tomography for quantitative cerebral blood flow imaging. Biomed. Opt. Express 2014, 5, 3217–3230. [Google Scholar] [CrossRef] [PubMed]
- Toossi, S.; Hess, C.P.; Hills, N.K.; Josephson, S.A. Neurovascular Complications of Cocaine Use at a Tertiary Stroke Center. J. Stroke Cerebrovasc. Dis. 2010, 19, 273–278. [Google Scholar] [CrossRef]
- Narvaez, J.C.; Magalhaes, P.V.; Fries, G.R.; Colpo, G.D.; Czepielewski, L.S.; Vianna, P.; Chies, J.A.; Rosa, A.R.; Von Diemen, L.; Vieta, E.; et al. Peripheral toxicity in crack cocaine use disorders. Neurosci. Lett. 2013, 544, 80–84. [Google Scholar] [CrossRef]
- Fox, H.C.; D’Sa, C.; Kimmerling, A.; Siedlarz, K.M.; Tuit, K.L.; Stowe, R.; Sinha, R. Immune system inflammation in cocaine dependent individuals: Implications for medications development. Hum. Psychopharmacol. 2012, 27, 156–166. [Google Scholar] [CrossRef]
- Lucas, G.M.; Atta, M.G.; Fine, D.M.; McFall, A.M.; Estrella, M.M.; Zook, K.; Stein, J.H. HIV, Cocaine Use, and Hepatitis C Virus: A Triad of Nontraditional Risk Factors for Subclinical Cardiovascular Disease. Arterioscler. Thromb. Vasc. Biol. 2016, 36, 2100–2107. [Google Scholar] [CrossRef]
- Harsch, H.H.; Pankiewicz, J.; Bloom, A.S.; Rainey, C.; Cho, J.K.; Sperry, L.; Stein, E.A. Hepatitis C virus infection in cocaine users—A silent epidemic. Community Ment. Health J. 2000, 36, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Kalichman, S.C.; Washington, C.; Kegler, C.; Grebler, T.; Kalichman, M.O.; Cherry, C.; Eaton, L. Continued Substance Use Among People Living With HIV-Hepatitis-C Co-Infection and Receiving Antiretroviral Therapy. Subst. Use Misuse 2015, 50, 1536–1543. [Google Scholar] [CrossRef] [PubMed]
- Dash, S.; Balasubramaniam, M.; Villalta, F.; Dash, C.; Pandhare, J. Impact of cocaine abuse on HIV pathogenesis. Front. Microbiol. 2015, 6, 1111. [Google Scholar] [CrossRef] [PubMed]
- Boehme, A.K.; Esenwa, C.; Elkind, M.S. Stroke Risk Factors, Genetics, and Prevention. Circ. Res. 2017, 120, 472–495. [Google Scholar] [CrossRef] [PubMed]
- Gretarsdottir, S.; Thorleifsson, G.; Manolescu, A.; Styrkarsdottir, U.; Helgadottir, A.; Gschwendtner, A.; Kostulas, K.; Kuhlenbaumer, G.; Bevan, S.; Jonsdottir, T.; et al. Risk variants for atrial fibrillation on chromosome 4q25 associate with ischemic stroke. Ann. Neurol. 2008, 64, 402–409. [Google Scholar] [CrossRef] [PubMed]
- Kouzarides, T. Histone acetylases and deacetylases in cell proliferation. Curr. Opin. Genet. Dev. 1999, 9, 40–48. [Google Scholar] [CrossRef]
- Penrod, R.D.; Carreira, M.B.; Taniguchi, M.; Kumar, J.; Maddox, S.A.; Cowan, C.W. Novel role and regulation of HDAC4 in cocaine-related behaviors. Addict. Biol. 2018, 23, 653–664. [Google Scholar] [CrossRef] [PubMed]
- Lanktree, M.B.; Hegele, R.A.; Yusuf, S.; Anand, S.S. Multi-ethnic genetic association study of carotid intima-media thickness using a targeted cardiovascular SNP microarray. Stroke 2009, 40, 3173–3179. [Google Scholar] [CrossRef]
- Desai, R.; Patel, U.; Rupareliya, C.; Singh, S.; Shah, M.; Patel, R.S.; Patel, S.; Mahuwala, Z. Impact of Cocaine Use on Acute Ischemic Stroke Patients: Insights from Nationwide Inpatient Sample in the United States. Cureus 2017, 9, e1536. [Google Scholar] [CrossRef]
- Cheng, Y.C.; Ryan, K.A.; Qadwai, S.A.; Shah, J.; Sparks, M.J.; Wozniak, M.A.; Stern, B.J.; Phipps, M.S.; Cronin, C.A.; Magder, L.S.; et al. Cocaine Use and Risk of Ischemic Stroke in Young Adults. Stroke 2016, 47, 918–922. [Google Scholar] [CrossRef]
- Daras, M.; Tuchman, A.J.; Marks, S. Central nervous system infarction related to cocaine abuse. Stroke 1991, 22, 1320–1325. [Google Scholar] [CrossRef] [PubMed]
- Martin-Schild, S.; Albright, K.C.; Hallevi, H.; Barreto, A.D.; Philip, M.; Misra, V.; Grotta, J.C.; Savitz, S.I. Intracerebral hemorrhage in cocaine users. Stroke 2010, 41, 680–684. [Google Scholar] [CrossRef] [PubMed]
- Lucerna, A.; Espinosa, J.; Zaman, T.; Hertz, R.; Stranges, D. Limb Pain as Unusual Presentation of a Parietal Intraparenchymal Bleeding Associated with Crack Cocaine Use: A Case Report. Case Rep. Neurol. Med. 2018, 2018, 9598675. [Google Scholar] [CrossRef] [PubMed]
- Mullaguri, N.; Battineni, A.; Narayan, A.; Guddeti, R. Cocaine Induced Bilateral Posterior Inferior Cerebellar Artery and Hippocampal Infarction. Cureus 2018, 10, e2576. [Google Scholar] [CrossRef] [PubMed]
- Rico-Mesa, J.S.; Rico-Mesa, M.A.; Berrouet, M.C. Ischemic stroke related to acute consumption of cocaine. CES Medicina 2017, 31, 207–214. [Google Scholar] [CrossRef][Green Version]
- Vidale, S.; Peroni, R.; Di Palma, F.; Sampietro, A.; Gozzi, G.; Arnaboldi, M. Intra-arterial thrombolysis in a young patient with cocaine-associated stroke. Neurol. Sci. 2014, 35, 1465–1466. [Google Scholar] [CrossRef] [PubMed]
- Renard, D.; Brunel, H.; Gaillard, N. Bilateral haemorrhagic infarction of the globus pallidus after cocaine and alcohol intoxication. Acta Neurol. Belg. 2009, 109, 159–161. [Google Scholar] [PubMed]
- Sein Anand, J.; Chodorowski, Z.; Wisniewski, M.; Golska, A. A cocaine-associated quadriplegia and motor aphasia after first use of cocaine. Przegl. Lek. 2007, 64, 316–317. [Google Scholar] [PubMed]
- Wolff, V.; Armspach, J.P.; Lauer, V.; Rouyer, O.; Bataillard, M.; Marescaux, C.; Geny, B. Cannabis-related stroke: Myth or reality? Stroke 2013, 44, 558–563. [Google Scholar] [CrossRef]
- Zou, S.; Kumar, U. Cannabinoid Receptors and the Endocannabinoid System: Signaling and Function in the Central Nervous System. Int. J. Mol. Sci. 2018, 19, 833. [Google Scholar] [CrossRef]
- Howlett, A.C.; Fleming, R.M. Cannabinoid inhibition of adenylate cyclase. Pharmacology of the response in neuroblastoma cell membranes. Mol. Pharmacol. 1984, 26, 532–538. [Google Scholar] [PubMed]
- Pertwee, R.G. The pharmacology of cannabinoid receptors and their ligands: An overview. Int. J. Obes. 2006, 30, S13–S18. [Google Scholar] [CrossRef] [PubMed]
- Wolff, V.; Armspach, J.P.; Lauer, V.; Rouyer, O.; Ducros, A.; Marescaux, C.; Geny, B. Ischaemic strokes with reversible vasoconstriction and without thunderclap headache: A variant of the reversible cerebral vasoconstriction syndrome? Cerebrovasc. Dis. 2015, 39, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Hemachandra, D.; McKetin, R.; Cherbuin, N.; Anstey, K.J. Heavy cannabis users at elevated risk of stroke: Evidence from a general population survey. Aust. N. Z. J. Public Health 2016, 40, 226–230. [Google Scholar] [CrossRef] [PubMed]
- Rumalla, K.; Reddy, A.Y.; Mittal, M.K. Recreational marijuana use and acute ischemic stroke: A population-based analysis of hospitalized patients in the United States. J. Neurol. Sci. 2016, 364, 191–196. [Google Scholar] [CrossRef] [PubMed]
- Atchaneeyasakul, K.; Torres, L.F.; Malik, A.M. Large Amount of Cannabis Ingestion Resulting in Spontaneous Intracerebral Hemorrhage: A Case Report. J. Stroke Cerebrovasc. Dis. 2017, 26, e138–e139. [Google Scholar] [CrossRef] [PubMed]
- Ince, B.; Benbir, G.; Yuksel, O.; Koseoglu, L.; Uluduz, D. Both hemorrhagic and ischemic stroke following high doses of cannabis consumption. Presse Med. 2015, 44, 106–107. [Google Scholar] [CrossRef] [PubMed]
- Deusch, E.; Kress, H.G.; Kraft, B.; Kozek-Langenecker, S.A. The procoagulatory effects of delta-9-tetrahydrocannabinol in human platelets. Anesth. Analg. 2004, 99, 1127–1130. [Google Scholar] [CrossRef]
- Levy, R.; Schurr, A.; Nathan, I.; Dvilanski, A.; Livn, A. Impairment of ADP-Induced Platelet Aggregation by Hashish Components. Thromb. Haemost. 1976, 36, 634–640. [Google Scholar] [CrossRef]
- Thanvi, B.R.; Treadwell, S.D. Cannabis and stroke: Is there a link? Postgrad. Med. J. 2009, 85, 80–83. [Google Scholar] [CrossRef]
- Mateo, I.; Pinedo, A.; Gomez-Beldarrain, M.; Basterretxea, J.M.; Garcia-Monco, J.C. Recurrent stroke associated with cannabis use. J. Neurol. Neurosurg. Psychiatry 2005, 76, 435–437. [Google Scholar] [CrossRef] [PubMed]
- Bailly, C.; Merceron, O.; Hammoudi, N.; Dorent, R.; Michel, P.L. Cannabis induced acute coronary syndrome in a young female. Int. J. Cardiol. 2010, 143, e4–e6. [Google Scholar] [CrossRef] [PubMed]
- EMCDDA. Medical Use of Cannabis and Cannabinoids: Questions and Answers for Policymaking; Publications Office of the European Union: Luxembourg, Luxembourg, 2018. [Google Scholar]
- Barber, P.A.; Pridmore, H.M.; Krishnamurthy, V.; Roberts, S.; Spriggs, D.A.; Carter, K.N.; Anderson, N.E. Cannabis, ischemic stroke, and transient ischemic attack: A case-control study. Stroke 2013, 44, 2327–2329. [Google Scholar] [CrossRef] [PubMed]
- Jamil, M.; Zafar, A.; Adeel Faizi, S.; Zawar, I. Stroke from Vasospasm due to Marijuana Use: Can Cannabis Synergistically with Other Medications Trigger Cerebral Vasospasm? Case Rep. Neurol. Med. 2016, 2016, 5313795. [Google Scholar] [CrossRef] [PubMed]
- Trojak, B.; Leclerq, S.; Meille, V.; Khoumri, C.; Chauvet-Gelinier, J.C.; Giroud, M.; Bonin, B.; Gisselmann, A. Stroke with neuropsychiatric sequelae after cannabis use in a man: A case report. J. Med. Case Rep. 2011, 5, 264. [Google Scholar] [CrossRef]
- Wolff, V.; Lauer, V.; Rouyer, O.; Sellal, F.; Meyer, N.; Raul, J.S.; Sabourdy, C.; Boujan, F.; Jahn, C.; Beaujeux, R.; et al. Cannabis use, ischemic stroke, and multifocal intracranial vasoconstriction: A prospective study in 48 consecutive young patients. Stroke 2011, 42, 1778–1780. [Google Scholar] [CrossRef]
- Shere, A.; Goyal, H. Cannabis can augment thrombolytic properties of rtPA: Intracranial hemorrhage in a heavy cannabis user. Am. J. Emerg. Med. 2017, 35, 1988-e1. [Google Scholar] [CrossRef]
- Volpon, L.C.; Sousa, C.; Moreira, S.K.K.; Teixeira, S.R.; Carlotti, A. Multiple Cerebral Infarcts in a Young Patient Associated With Marijuana Use. J. Addict. Med. 2017, 11, 405–407. [Google Scholar] [CrossRef]
- Tirkey, N.K.; Gupta, S. Acute Antero-Inferior Wall Ischaemia with Acute Ischaemic Stroke Caused by Oral Ingestion of Cannabis in a Young Male. J. Assoc. Physicians India 2016, 64, 93–94. [Google Scholar]
- Baharnoori, M.; Kassardjian, C.D.; Saposnik, G. Cannabis use associated with capsular warning syndrome and ischemic stroke. Can. J. Neurol. Sci. 2014, 41, 272–273. [Google Scholar] [CrossRef]
- Renard, D.; Taieb, G.; Gras-Combe, G.; Labauge, P. Cannabis-related myocardial infarction and cardioembolic stroke. J. Stroke Cerebrovasc. Dis. 2012, 21, 82–83. [Google Scholar] [CrossRef] [PubMed]
- Castaneto, M.S.; Gorelick, D.A.; Desrosiers, N.A.; Hartman, R.L.; Pirard, S.; Huestis, M.A. Synthetic cannabinoids: Epidemiology, pharmacodynamics, and clinical implications. Drug Alcohol Depend. 2014, 144, 12–41. [Google Scholar] [CrossRef] [PubMed]
- Seely, K.A.; Lapoint, J.; Moran, J.H.; Fattore, L. Spice drugs are more than harmless herbal blends: A review of the pharmacology and toxicology of synthetic cannabinoids. Prog. Neuropsychopharmacol. Biol. Psychiatry 2012, 39, 234–243. [Google Scholar] [CrossRef] [PubMed]
- Tai, S.; Fantegrossi, W.E. Synthetic Cannabinoids: Pharmacology, Behavioral Effects, and Abuse Potential. Curr. Addict. Rep. 2014, 1, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Brents, L.K.; Reichard, E.E.; Zimmerman, S.M.; Moran, J.H.; Fantegrossi, W.E.; Prather, P.L. Phase I hydroxylated metabolites of the K2 synthetic cannabinoid JWH-018 retain in vitro and in vivo cannabinoid 1 receptor affinity and activity. PLoS ONE 2011, 6, e21917. [Google Scholar] [CrossRef] [PubMed]
- Freeman, M.J.; Rose, D.Z.; Myers, M.A.; Gooch, C.L.; Bozeman, A.C.; Burgin, W.S. Ischemic stroke after use of the synthetic marijuana “spice”. Neurology 2013, 81, 2090–2093. [Google Scholar] [CrossRef] [PubMed]
- Rose, D.Z.; Guerrero, W.R.; Mokin, M.V.; Gooch, C.L.; Bozeman, A.C.; Pearson, J.M.; Burgin, W.S. Hemorrhagic stroke following use of the synthetic marijuana “spice”. Neurology 2015, 85, 1177–1179. [Google Scholar] [CrossRef] [PubMed]
- Hillard, C.J. Endocannabinoids and vascular function. J. Pharmacol. Exp. Ther. 2000, 294, 27–32. [Google Scholar] [PubMed]
- Brents, L.K.; Prather, P.L. The K2/Spice phenomenon: Emergence, identification, legislation and metabolic characterization of synthetic cannabinoids in herbal incense products. Drug Metab. Rev. 2014, 46, 72–85. [Google Scholar] [CrossRef] [PubMed]
- Bernson-Leung, M.E.; Leung, L.Y.; Kumar, S. Synthetic cannabis and acute ischemic stroke. J. Stroke Cerebrovasc. Dis. 2014, 23, 1239–1241. [Google Scholar] [CrossRef] [PubMed]
- Faroqui, R.; Mena, P.; Wolfe, A.R.; Bibawy, J.; Visvikis, G.A.; Mantello, M.T. Acute carotid thrombosis and ischemic stroke following overdose of the synthetic cannabinoid K2 in a previously healthy young adult male. Radiol. Case Rep. 2018, 13, 747–752. [Google Scholar] [CrossRef] [PubMed]
- Takematsu, M.; Hoffman, R.S.; Nelson, L.S.; Schechter, J.M.; Moran, J.H.; Wiener, S.W. A case of acute cerebral ischemia following inhalation of a synthetic cannabinoid. Clin. Toxicol. 2014, 52, 973–975. [Google Scholar] [CrossRef] [PubMed]
- Lachenmeier, D.W.; Sproll, C.; Musshoff, F. Poppy seed foods and opiate drug testing—Where are we today? Ther. Drug Monit. 2010, 32, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Mars, S.G.; Bourgois, P.; Karandinos, G.; Montero, F.; Ciccarone, D. The Textures of Heroin: User Perspectives on “Black Tar” and Powder Heroin in Two U.S. Cities. J. Psychoact. Drugs 2016, 48, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Freye, E.; Levy, J. Opioids in Medicine—A Comprehensive Review on the Mode of Action and the Use of Analgesics in Different Clinical Pain States; Springer Science + Business Media BV: Dordrecht, The Netherlands, 2008. [Google Scholar]
- Dinis-Oliveira, R.J.; Carvalho, F.; Moreira, R.; Duarte, J.A.; Proenca, J.B.; Santos, A.; Magalhaes, T. Clinical and forensic signs related to opioids abuse. Curr. Drug Abus. Rev. 2012, 5, 273–290. [Google Scholar] [CrossRef]
- Yeh, S.Y. Urinary excretion of morphine and its metabolites in morphine-dependent subjects. J. Pharmacol. Exp. Ther. 1975, 192, 201–210. [Google Scholar] [PubMed]
- Meyer, M.R.; Schutz, A.; Maurer, H.H. Contribution of human esterases to the metabolism of selected drugs of abuse. Toxicol. Lett. 2015, 232, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Fonseca, A.C.; Ferro, J.M. Drug abuse and stroke. Curr. Neurol. Neurosci. Rep. 2013, 13, 325. [Google Scholar] [CrossRef]
- Niehaus, L.; Roricht, S.; Meyer, B.U.; Sander, B. Nuclear magnetic resonance tomography detection of heroin-associated CNS lesions. Aktuelle Radiol. 1997, 7, 309–311. [Google Scholar]
- Enevoldson, T.P. Recreational drugs and their neurological consequences. J. Neurol. Neurosurg. Psychiatry 2004, 75, iii9–iii15. [Google Scholar] [CrossRef]
- Bolz, J.; Meves, S.H.; Kara, K.; Reinacher-Schick, A.; Gold, R.; Krogias, C. Multiple cerebral infarctions in a young patient with heroin-induced hypereosinophilic syndrome. J. Neurol. Sci. 2015, 356, 193–195. [Google Scholar] [CrossRef] [PubMed]
- Prick, J.J.; Gabreels-Festen, A.A.; Korten, J.J.; van der Wiel, T.W. Neurological manifestations of the hypereosinophilic syndrome (HES). Clin. Neurol. Neurosurg. 1988, 90, 269–273. [Google Scholar] [CrossRef]
- Hamzei Moqaddam, A.; Ahmadi Musavi, S.M.R.; Khademizadeh, K. Relationship of opium dependency and stroke. Addict. Health 2009, 1, 6–10. [Google Scholar] [PubMed]
- Kumar, N.; Bhalla, M.C.; Frey, J.A.; Southern, A. Intraparenchymal hemorrhage after heroin use. Am. J. Emerg. Med. 2015, 33, 1109-e3. [Google Scholar] [CrossRef] [PubMed]
- Alquist, C.R.; McGoey, R.; Bastian, F.; Newman, W., 3rd. Bilateral globus pallidus lesions. J. La. State Med. Soc. 2012, 164, 145–146. [Google Scholar] [PubMed]
- Vila, N.; Chamorro, A. Ballistic movements due to ischemic infarcts after intravenous heroin overdose: Report of two cases. Clin. Neurol. Neurosurg. 1997, 99, 259–262. [Google Scholar] [CrossRef]
- Brust, J.C.; Richter, R.W. Stroke associated with addiction to heroin. J. Neurol. Neurosurg. Psychiatry 1976, 39, 194–199. [Google Scholar] [CrossRef]
- Lee, C.W.; Muo, C.H.; Liang, J.A.; Sung, F.C.; Kao, C.H. Association of intensive morphine treatment and increased stroke incidence in prostate cancer patients: A population-based nested case-control study. Jpn. J. Clin. Oncol. 2013, 43, 776–781. [Google Scholar] [CrossRef]
- Hamzei-Moghaddam, A.; Shafa, M.A.; Khanjani, N.; Farahat, R. Frequency of Opium Addiction in Patients with Ischemic Stroke and Comparing their Cerebrovascular Doppler Ultrasound Changes to Non-Addicts. Addict. Health 2013, 5, 95–101. [Google Scholar]
- Benoilid, A.; Collongues, N.; de Seze, J.; Blanc, F. Heroin inhalation-induced unilateral complete hippocampal stroke. Neurocase 2013, 19, 313–315. [Google Scholar] [CrossRef]
- Hsu, W.Y.; Chiu, N.Y.; Liao, Y.C. Rhabdomyolysis and brain ischemic stroke in a heroin-dependent male under methadone maintenance therapy. Acta Psychiatr. Scand. 2009, 120, 76–79. [Google Scholar] [CrossRef] [PubMed]
- Brenu, E.W.; McNaughton, L.; Marshall-Gradisnik, S.M. Is there a potential immune dysfunction with anabolic androgenic steroid use: A review. Mini Rev. Med. Chem. 2011, 11, 438–445. [Google Scholar] [CrossRef] [PubMed]
- Kenna, G.A.; Lewis, D.C. Risk factors for alcohol and other drug use by healthcare professionals. Subst. Abus. Treat. Prev. Policy 2008, 3, 3. [Google Scholar] [CrossRef] [PubMed]
- Hartgens, F.; Rietjens, G.; Keizer, H.A.; Kuipers, H.; Wolffenbuttel, B.H. Effects of androgenic-anabolic steroids on apolipoproteins and lipoprotein (a). Br. J. Sports Med. 2004, 38, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Santamarina, R.D.; Besocke, A.G.; Romano, L.M.; Ioli, P.L.; Gonorazky, S.E. Ischemic stroke related to anabolic abuse. Clin. Neuropharmacol. 2008, 31, 80–85. [Google Scholar] [CrossRef] [PubMed]
- Shamloul, R.M.; Aborayah, A.F.; Hashad, A.; Abd-Allah, F. Anabolic steroids abuse-induced cardiomyopathy and ischaemic stroke in a young male patient. BMJ Case Rep. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Youssef, M.Y.; Alqallaf, A.; Abdella, N. Anabolic androgenic steroid-induced cardiomyopathy, stroke and peripheral vascular disease. BMJ Case Rep. 2011, 2011. [Google Scholar] [CrossRef] [PubMed]
- Pearson, A.C.; Schiff, M.; Mrosek, D.; Labovitz, A.J.; Williams, G.A. Left ventricular diastolic function in weight lifters. Am. J. Cardiol. 1986, 58, 1254–1259. [Google Scholar] [CrossRef]
- Lippi, G.; Banfi, G. Doping and thrombosis in sports. Semin. Thromb. Hemost. 2011, 37, 918–928. [Google Scholar] [CrossRef]
- Beutel, A.; Bergamaschi, C.T.; Campos, R.R. Effects of chronic anabolic steroid treatment on tonic and reflex cardiovascular control in male rats. J. Steroid Biochem. Mol. Biol. 2005, 93, 43–48. [Google Scholar] [CrossRef]
- Santos, M.A.; Oliveira, C.V.; Silva, A.S. Adverse cardiovascular effects from the use of anabolic-androgenic steroids as ergogenic resources. Subst. Use Misuse 2014, 49, 1132–1137. [Google Scholar] [CrossRef] [PubMed]
- Achar, S.; Rostamian, A.; Narayan, S.M. Cardiac and metabolic effects of anabolic-androgenic steroid abuse on lipids, blood pressure, left ventricular dimensions, and rhythm. Am. J. Cardiol. 2010, 106, 893–901. [Google Scholar] [CrossRef] [PubMed]
- D’Ascenzo, S.; Millimaggi, D.; Di Massimo, C.; Saccani-Jotti, G.; Botre, F.; Carta, G.; Tozzi-Ciancarelli, M.G.; Pavan, A.; Dolo, V. Detrimental effects of anabolic steroids on human endothelial cells. Toxicol. Lett. 2007, 169, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Hartgens, F.; Kuipers, H. Effects of androgenic-anabolic steroids in athletes. Sports Med. 2004, 34, 513–554. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.Y.; Death, A.K.; Handelsman, D.J. Androgens and cardiovascular disease. Endocr. Rev. 2003, 24, 313–340. [Google Scholar] [CrossRef] [PubMed]
- Rocha, F.L.; Carmo, E.C.; Roque, F.R.; Hashimoto, N.Y.; Rossoni, L.V.; Frimm, C.; Aneas, I.; Negrao, C.E.; Krieger, J.E.; Oliveira, E.M. Anabolic steroids induce cardiac renin-angiotensin system and impair the beneficial effects of aerobic training in rats. Am. J. Physiol. Heart Circ. Physiol. 2007, 293, H3575–H3583. [Google Scholar] [CrossRef] [PubMed]
- Do Carmo, E.C.; Fernandes, T.; Koike, D.; Da Silva, N.D., Jr.; Mattos, K.C.; Rosa, K.T.; Barretti, D.; Melo, S.F.; Wichi, R.B.; Irigoyen, M.C.; et al. Anabolic steroid associated to physical training induces deleterious cardiac effects. Med. Sci. Sports Exerc. 2011, 43, 1836–1848. [Google Scholar] [CrossRef] [PubMed]
- Schooling, C.M.; Luo, S.; Au Yeung, S.L.; Thompson, D.J.; Karthikeyan, S.; Bolton, T.R.; Mason, A.M.; Ingelsson, E.; Burgess, S. Genetic predictors of testosterone and their associations with cardiovascular disease and risk factors: A Mendelian randomization investigation. Int. J. Cardiol. 2018, 267, 171–176. [Google Scholar] [CrossRef] [PubMed]
- Cole, A.P.; Hanske, J. Impact of testosterone replacement therapy on thromboembolism, heart disease and obstructive sleep apnoea in men. BJU Int. 2018, 121, 811–818. [Google Scholar] [CrossRef] [PubMed]
- Normann, S.; de Veber, G.; Fobker, M.; Langer, C.; Kenet, G.; Bernard, T.J.; Fiedler, B.; Sträter, R.; Goldenberg, N.A.; Nowak-Göttl, U. Role of endogenous testosterone concentration in pediatric stroke. Ann. Neurol. 2009, 66, 754–758. [Google Scholar] [CrossRef] [PubMed]
- Bhasin, S.; Brito, J.P.; Cunningham, G.R.; Hayes, F.J.; Hodis, H.N.; Matsumoto, A.M.; Snyder, P.J.; Swerdloff, R.S.; Wu, F.C.; Yialamas, M.A. Testosterone Therapy in Men With Hypogonadism: An Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 2018, 103, 1715–1744. [Google Scholar] [CrossRef] [PubMed]
- El Scheich, T.; Weber, A.A.; Klee, D.; Schweiger, D.; Mayatepek, E.; Karenfort, M. Adolescent ischemic stroke associated with anabolic steroid and cannabis abuse. J. Pediatr. Endocrinol. Metab. 2013, 26, 161–165. [Google Scholar] [CrossRef] [PubMed]
- Juhan-Vague, I.; Pyke, S.D.; Alessi, M.C.; Jespersen, J.; Haverkate, F.; Thompson, S.G. Fibrinolytic factors and the risk of myocardial infarction or sudden death in patients with angina pectoris. ECAT Study Group. European Concerted Action on Thrombosis and Disabilities. Circulation 1996, 94, 2057–2063. [Google Scholar] [CrossRef] [PubMed]
- Siokas, V.; Dardiotis, E.; Sokolakis, T.; Kotoula, M.; Tachmitzi, S.V.; Chatzoulis, D.Z.; Almpanidou, P.; Stefanidis, I.; Hadjigeorgiou, G.M.; Tsironi, E.E. Plasminogen Activator Inhibitor Type-1 Tag Single-Nucleotide Polymorphisms in Patients with Diabetes Mellitus Type 2 and Diabetic Retinopathy. Curr. Eye Res. 2017, 42, 1048–1053. [Google Scholar] [CrossRef] [PubMed]
- Winkler, U.H. Effects of androgens on haemostasis. Maturitas 1996, 24, 147–155. [Google Scholar] [CrossRef]
- Nieminen, M.S.; Ramo, M.P.; Viitasalo, M.; Heikkila, P.; Karjalainen, J.; Mantysaari, M.; Heikkila, J. Serious cardiovascular side effects of large doses of anabolic steroids in weight lifters. Eur. Heart J. 1996, 17, 1576–1583. [Google Scholar] [CrossRef] [PubMed]
- Kalin, M.F.; Zumoff, B. Sex hormones and coronary disease: A review of the clinical studies. Steroids 1990, 55, 330–352. [Google Scholar] [CrossRef]
- Bachman, E.; Feng, R.; Travison, T.; Li, M.; Olbina, G.; Ostland, V.; Ulloor, J.; Zhang, A.; Basaria, S.; Ganz, T.; et al. Testosterone suppresses hepcidin in men: A potential mechanism for testosterone-induced erythrocytosis. J. Clin. Endocrinol. Metab. 2010, 95, 4743–4747. [Google Scholar] [CrossRef]
- Bachman, E.; Travison, T.G.; Basaria, S.; Davda, M.N.; Guo, W.; Li, M.; Connor Westfall, J.; Bae, H.; Gordeuk, V.; Bhasin, S. Testosterone induces erythrocytosis via increased erythropoietin and suppressed hepcidin: Evidence for a new erythropoietin/hemoglobin set point. J. Gerontol. A Biol. Sci. Med. Sci. 2014, 69, 725–735. [Google Scholar] [CrossRef]
- Young, N.S. Telomere biology and telomere diseases: Implications for practice and research. Hematol. Am. Soc. Hematol. Educ. Program 2010, 2010, 30–35. [Google Scholar] [CrossRef]
- Tsarouhas, K.; Kioukia-Fougia, N.; Papalexis, P.; Tsatsakis, A.; Kouretas, D.; Bacopoulou, F.; Tsitsimpikou, C. Use of nutritional supplements contaminated with banned doping substances by recreational adolescent athletes in Athens, Greece. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2018, 115, 447–450. [Google Scholar] [CrossRef] [PubMed]
- Tsitsimpikou, C.; Chrisostomou, N.; Papalexis, P.; Tsarouhas, K.; Tsatsakis, A.; Jamurtas, A. The use of nutritional supplements among recreational athletes in Athens, Greece. Int. J. Sport Nutr. Exerc. Metab. 2011, 21, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Vasilaki, F.; Tsitsimpikou, C.; Tsarouhas, K.; Germanakis, I.; Tzardi, M.; Kavvalakis, M.; Ozcagli, E.; Kouretas, D.; Tsatsakis, A.M. Cardiotoxicity in rabbits after long-term nandrolone decanoate administration. Toxicol. Lett. 2016, 241, 143–151. [Google Scholar] [CrossRef] [PubMed]
- Baggish, A.L.; Weiner, R.B.; Kanayama, G.; Hudson, J.I.; Picard, M.H.; Hutter, A.M., Jr.; Pope, H.G., Jr. Long-term anabolic-androgenic steroid use is associated with left ventricular dysfunction. Circ. Heart Fail. 2010, 3, 472–476. [Google Scholar] [CrossRef] [PubMed]
- Sattler, F.R.; Jaque, S.V.; Schroeder, E.T.; Olson, C.; Dube, M.P.; Martinez, C.; Briggs, W.; Horton, R.; Azen, S. Effects of pharmacological doses of nandrolone decanoate and progressive resistance training in immunodeficient patients infected with human immunodeficiency virus. J. Clin. Endocrinol. Metab. 1999, 84, 1268–1276. [Google Scholar] [CrossRef] [PubMed]
- Shimada, Y.; Yoritaka, A.; Tanaka, Y.; Miyamoto, N.; Ueno, Y.; Hattori, N.; Takao, U. Cerebral Infarction in a Young Man Using High-dose Anabolic Steroids. J. Stroke Cerebrovasc. Dis. 2012, 21, 906-e9. [Google Scholar] [CrossRef] [PubMed]
- D’Andrea, A.; Caso, P.; Salerno, G.; Scarafile, R.; De Corato, G.; Mita, C.; Di Salvo, G.; Severino, S.; Cuomo, S.; Liccardo, B.; et al. Left ventricular early myocardial dysfunction after chronic misuse of anabolic androgenic steroids: A Doppler myocardial and strain imaging analysis. Br. J. Sports Med. 2007, 41, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Esperon, C.; Hervas-Garcia, J.V.; Jimenez-Gonzalez, M.; Perez de la Ossa-Herrero, N.; Gomis-Cortina, M.; Dorado-Bouix, L.; Lopez-Cancio Martinez, E.; Castano-Duque, C.H.; Millan-Torne, M.; Davalos, A. [Ingestion of anabolic steroids and ischaemic stroke. A clinical case report and review of the literature]. Rev. Neurol. 2013, 56, 327–331. [Google Scholar]
- Low, M.S.; Vilcassim, S.; Fedele, P.; Grigoriadis, G. Anabolic androgenic steroids, an easily forgotten cause of polycythaemia and cerebral infarction. Intern. Med. J. 2016, 46, 497–499. [Google Scholar] [CrossRef]
- Jouanjus, E.; Lapeyre-Mestre, M.; Micallef, J. Cannabis use: Signal of increasing risk of serious cardiovascular disorders. J. Am. Heart Assoc. 2014, 3, e000638. [Google Scholar] [CrossRef]
| Subject/Age | Substance Exposure | Symptoms | Diagnostic Approach | Diagnosis | Intervention | Evolution | Reference |
|---|---|---|---|---|---|---|---|
| Female, 23, no previous medical history | Took 4-fluoroamphetamine 4 h before, concomitant use of cannabis 7 h before | Collapsed at a dance event, no neurological deficits, sleepy and headache, decreased consciousness 1.5 h later, weakness of the right arm and leg | Plain computed tomography scan (computed tomography (CT) scan); CT angiography | Intracerebral hemorrhage in the left hemisphere; dilated non-responsive right pupil (false localizing sign) | Acute neurosurgical intervention: Hematoma evacuation, removal of the bone flap due to persistent intraoperative brain swelling | Right-sided hemiparalysis and severe aphasia. Replacement of the autologous bone graft after 4 months without complications. Able to talk in her native language and walk with supportive measures | [44] |
| Female,23, no medical history | Took 110 mg 4-fluoroamphetamine the night before, concomitant use of four units of alcohol | Severe headache, nausea, followed by vomiting 5 h after the intake, dizziness, photophobia | CT scan | Small subarachnoid hemorrhage at the right frontal side | Discharged after 24 h | Headache for weeks that gradually declined cognitive problems. Inability to work for several months | [44] |
| Female, 29, progressive headache and diplopia for 2 weeks, no medical history | Intravenous methamphetamine use | A 2-day history of left-sided hemiparesis and dysarthria | Cranial nerve examination, CT brain imaging without contrast medium, magnetic resonance imaging (MRI), angiogram | A 25 × 25 × 20-mm hyperdense lesion within the right cerebellopontine angle Initially thought to represent an extra-axial mass (meningioma), confirmed to be a large brainstem hemorrhage, extended from the inferior midbrain to the pontomedullary junction | Transferred to rehabilitation | Deterioration of left hemiparesis, dysarthria and dysphagia after 1 month. No underlying vascular abnormality observed | [45] |
| Male, Caucasian, 53, history of head and neck squamous cell carcinoma post-surgery and radiation (13 years before), hypothyroidism, hyperlipidemia, gastrointestinal reflux disease | Treatment for Attention Deficit Hyperactivity Disorder (ADHD) with mixed amphetamine salts, starting 5 mg/day to 15 mg/day over 4 months | Posterior headache with left-face numbness, diplopia 2.5 months after last dosing scheme | Head CT without contrast agent; MRI; transthoracic echocardiogram | Right posterior paramedian midbrain hematoma with cerebral aqueduct effacement and mild ventriculomegaly. No hypertension, arteriovenous malformation, cavernous malformation, or aneurysms | - | - | [46] |
| Male, 31 | Amphetamine abuse | - | Transcranial color-coded Doppler sonography; angiography | Intracerebral hemorrhage, diffuse cerebral vasospasm | Surgical removal of intracerebral hemorrhage, pharmaceutical treatment | - | [47] |
| Male, African-American, 20 | Took 3,4-methylenedioxymeth-amphetamine (MDMA), concomitant use of marijuana and beer | Non-verbal, vomiting and aphasic upon presentation, no sign of trauma, 18 h after ingestion developed right-sided weakness, left-sided facial droop and bilateral hyperreflexia in the lower extremities | MRI; carotid ultrasound; magnetic resonance angiogram of the brain | Left middle cerebral artery complete infarction, no significant stenosis, mild to moderate stenosis observed on the distal left internal carotid artery | Transferred to rehabilitation | - | [48] |
| Female, 36, history of migraine | Methamphetamine use, concomitant use of oral contraceptives | Sudden onset of speech difficulty and right-sided weakness | Head CT; MRI of the brain; MR angiography | Small infarct in the left frontal lobe, focal narrowing in the left internal carotid artery | Pharmaceutical treatment: IV heparin, discharged on warfarin 5 days after stroke; after 8 months, warfarin was replaced with aspirin 81 mg/day | Recovered after 4 months with only mild expressive aphasia | [49] |
| Female, 29 | History of methamphetamine use for 10 years | Sudden right-sided weakness and speech difficulty 4 days after last use of methamphetamine | Head CT, MRI, MR angiography | Large left middle cerebral artery (MCA) infarct, MCA infarct with hemorrhagic transformation | Discharged after 4 days on aspirin treatment, on day 5th showed worsening deficit, hospitalized; stent-assisted transformation applied | Recovered only with moderate expression aphasia and mild right-hand weakness within 4 months | [49] |
| Male,31 | Methamphetamine ingestion approximately 0.25 and 0.5 g Urine screen positive also for tetrahydrocannabinol (THC) | Severe headache, nausea, vomiting, left-side of the body felt numb, slurred speech, died the next day | Autopsy | Cerebral edema, subarachnoid hemorrhage over the cerebral convexities bilaterally, intracerebral hemorrhage lateral to the basal ganglia extending to involve the lateral aspect of the putamen, external capsule claustrum, insula, and superior longitudinal fasciculus of the right cerebral hemisphere (3.5 cm by 4.5 cm) No evidence of inflammation or vasculitis | Death | [38] | |
| Male, Caucasian, 33, amphetamine addict | Amphetamine and methamphetamine ingestion. Low concentrations of methadone and codeine in the blood | Bilateral cerebral infarction associated with multi-organ failure | CT scan, autopsy | Extensive infarction of both cerebral hemispheres; symmetrical necrosis of the white matter of both cerebral hemispheres in the autopsy | Died 19 days after hospital admission | [42] | |
| Female, 30, no significant medical history, non-smoker, very light alcohol consumer | Ecstasy ingestion one night before the presentation | Right-sided weakness, global aphasia, right neglect, and right hemiparesis | Brain CT scan; ultrasound of the extracranial carotid arteries; transcranial color-coded Doppler (TCCD); MRI | Left parietal hypodensity consistent with left middle cerebral artery (MCA) infarction; irregularity of the left MCA | Aspirin 100 mg/day | TCCD studies showed normal velocities in the MCA 3 months after onset | [33] |
| Female, 19, duodenal ulcer at 16, no other medical history, no family history of stroke | Methamphetamine intravenously four times over 2 months, wash-out for 3 months, concomitant use of cigarettes and alcohol | Severe right-sided headache, blurred vision on the left side and numbness of the left arm and leg upon admission, severe headache every time associated with use | Brain CT, MRI and magnetic resonance angiography | Right occipital infarction, segmental narrowing of the right posterior cerebral artery with characteristics of vasculitis | Discharged one week after admission | The right occipital infarction faded with mild atrophy, left superior quadrant hemianopia remained and had persistent headaches 4 months later | [32] |
| Subject/Age | Substance Exposure | Symptoms | Diagnostic Approach | Diagnosis | Intervention | Evolution | Reference |
|---|---|---|---|---|---|---|---|
| Male, African American, 65, diabetes, heart diseases, hepatitis C | Smoking crack cocaine before symptom onset, admitted to intermittent cocaine abuse | Left arm pain described as feeling like “jumping out of the window” | Head CT scan; carotid ultrasound; CT angiography of head and neck | Acute 2.2-cm intraparenchymal hemorrhage that presented in the posterior right parietal lobe vasogenic edema | Send to the rehabilitation unit | Left arm pain resolved after 24 h | [75] |
| Female, African-American, 66, multi-substance abuser, hepatitis C, heart diseases | Urine samples positive for cocaine | Somnolent a day prior to admission, confused in the day of admission, short-term memory loss, unable to perform usual daily activities | Brain CT; CT angiogram of the head and neck; MRI of the brain associated with MR venogram | Infarction in bilateral posterior inferior cerebellar artery and hippocamp showing multifocal punctate infarcts in the basal ganglia and bilateral posterior cerebral artery secondary to severe vasoconstriction | Neurosurgery consult for possible external ventricular drain placement and posterior fossa decompression | Mental status improved during hospitalization; discharged to a rehabilitation center after 7 days with persistent problems of memory and inability to recognize faces | [76] |
| Male,22, hypertension and cocaine abuser | Positive for cocaine and tetrahydrocannabinol | Right hemiplegia associated with motor and sensitive aphasia | CT scan | The ischemic region in the left medial cerebral artery region with increased cerebral edema and cerebral midline displacement of 9 mm on the subfalcine region | Not suitable for surgery due to complications | Died in the hospital | [77] |
| Female, 39, smoker, no other risk factors for stroke | Urine screening positive for cocaine | Global aphasia, left-side total gaze paresis, 7th cranial nerve right-side partial paresis and right hemiplegia | Non-contrast brain CT | Left ischemic stroke—hyperdensity in the left middle cerebral artery (MCA); occlusion in the left and right MCA and an irregular profile of the left internal carotid artery (ICA) | Endovascular treatment, intra-arterial administration of 40 mg of recombinant tissue plasminogen activator (rtPA) associated with a self-expandable and retrievable stent | After 3 months from the event, ischemia at the left basal ganglia | [78] |
| Male, 31, no medical history | Positive urine screening for cocaine and negative for other drugs | Found unresponsive 6 h after excessive alcohol and intranasal cocaine abuse | MRI; intra- and extracranial CT angiography | Globus pallidus and the vascular watershed zones presents acute bilateral ischemia | - | Consciousness improved progressively; clinical improvements, but mental slowing, executive dysfunction, hypophonia, and verbal fluency deficit persisted | [79] |
| Female, 31, no medical history, occasional alcohol consumer and smoker | First time snorted cocaine hydrochloride associated with 500 mL of vodka | Acute onset of right hemiplegia and left hemiparesis evolving into quadriplegia | MRI | Thickened pons with focus localized in his central part on the left side (20 mm) (ischemic change) | After 17 days of hospitalization, transferred to rehabilitation | The movements of the left side of the body improved slowly and the rehabilitation continues in ambulatory | [80] |
| Subject/Age | Substance Exposure | Symptoms | Diagnostic Approach | Diagnosis | Intervention | Evolution | Reference |
|---|---|---|---|---|---|---|---|
| Female, 51, asthma | Long-term cannabis user, positive urine screening for cannabis, a large amount of cannabis was consumed prior to the onset of symptoms | Left-side upper and lower extremities weakness | Head CT scan | Acute right cerebral infarct; after 30 min from arrival, developed in the left pons new hemorrhage associated with decompression on the lateral and left ventricles | Pharmaceutical treatment: Labetalol, recombinant tissue plasminogen activator | Died | [100] |
| Male, 27, without any known medical history | Single raw cannabis consumption (confirmed by a blood test) just before symptom onset | Sudden progressive left-sided weakness, degradation in mentation, nausea, and vomiting | Brain CT without contrast media; CT angiography; MRI of the brain | Right basal ganglia ICH measuring 32 × 24 mm with extension into the ventricles with mild hydrocephalus, no vasculature abnormality | Intubation and placement of an external ventricular drain, treatment on recombinant tissue plasminogen activator | Improvement of motor function, left hemiparesis | [88] |
| Female, 14, no remarkable medical history | Toxicological screening positive for cannabis 2-year history of daily cannabis use | Generalized tonic–clonic seizures | Head CT, electroencephalography (EEG); MRI | Multiple ischemic infarcts located in basal ganglia, left frontal lobe, and genu of corpus callosum, which had both chronic and acute features | After stabilization, transferred to rehabilitation | Complained of chronic headache, learning disabilities | [101] |
| Male, 25 | Cannabis ingestion one night before Concomitant ingestion of alcohol | Drowsy, talking irrelevantly and the state degraded | Non-contrast CT of the brain; Coronary CT angiogram | Acute infarct in the right frontoparietal region | After hospitalization was discharged in a stable condition | Left-sided weakness improved | [102] |
| Male | Marijuana History of smoking marijuana from the age of 1 | Presented with weakness of leg, arm and face associated with slurred speech 90 min after smoking marijuana Recurrence of the symptoms twice | Brain CT scan, CT angiogram and MRI | Right lentiform nucleus presents subtle hypodensity; no evidence of vasospasm, thrombus or dissection | Heparin treatment after a recurrent episode of focal neurological deficits | After 2 months, he presented residual weakness in the left arm and leg, left facial droop and spastic tone | [103] |
| Male, 33, smoker | Urine toxicologic screening positive for cannabis Heavy user of cannabis for 15 years | Transient left hemiparesis and dysarthria, no altered consciousness, chest pain one day before | Brain MRI and CT angiography | The presence of multi focal acute infarctions in the bilateral watershed zones between middle and anterior cerebral artery territories and the right middle cerebral artery territory. Cardioembolic stroke produced by acute myocardial infarction (likely related to cannabis use) | - | No recurrence in the following 6 months of cardiac or neurologic symptoms | [104] |
| Male, Caucasian French, 24, no medical history | Urine toxicology positive for cannabis; heavy cannabis use one night before admission Regular cannabis smoker for four years | Non-reactive state, with seizures | Cerebral CT scan, EEG, MRI, Doppler examination, magnetic resonance angiography, and angiography | Infarcts in the insular mantle and the lenticular and caudate nuclear structures exclude all other causes of stroke in young people | Treated in the hospital until recovery and transferred to the psychiatric department to be treated for behavioral disorders | In the following 1 and a half years, he returned on seven occasions for generalized tonic–clonic seizures | [98] |
| Male, 36, with no history of migraine or other known vascular risk factors | Urine toxicological screening positive for cannabis Heavy hashish consumption and alcohol before the symptoms Sporadically hashish user | An acute episode of isolated aphasia, followed by convulsive seizures | Cranial MRI and MR angiography | Had 2 acute ischemic infarcts, one on the left temporal lobe and another area of silent ischemia in the right parietal lobe | Treatment with ticlopidine | After 1 year, a new episode of aphasia and right hemiparesis immediately after hashish smoking and a new episode after 1 and a half years again after hashish use Between the two episodes, he denied consumption | [93] |
| Subject/Age | Substance Exposure | Symptoms | Diagnostic Approach | Diagnosis | Intervention | Evolution | Reference |
|---|---|---|---|---|---|---|---|
| Male, African American, 36, no history of stroke or coagulopathy or blood disorders | Reported taking K2 on the night before symptom onsetConcomitant use of marijuana in the past | Had a 1-day history of aphasia and weakness in the right side of the body | Non-contrast CT of the head; computed tomography angiography (CTA); MRI; MR angiography | A thrombotic event that lead to an acute ischemic infarct with left MCA distribution characterized by hypodensity in the left basal ganglia and a left hyperdense MCA; a large filling defect observed from the origin of the left ICA into the intracranial portions of the ICA | Aspirin, clopidogrel and enoxaparin | After 10 days, the patient was discharged for short-term rehabilitation after gradual improvement | [114] |
| Female, 22, in treatment with atomoxetine and estrogen-containing oral contraceptive | Smoked K2; concomitant use of THC, benzodiazepine and salicylates as they were positive at urine toxicological test | While smoking K2 presented dyspnea, palpitations and angor animi. Few hours later after smoking K2, developed dysarthria and difficulty standing | Head CT, MRI, and CT angiogram | Right middle cerebral artery AIS; proximal right M1 occlusion with distal reconstruction | Aspirin | In follow-up, presented limited ambulation and no use of her spastic left arm | [113] |
| Female, 26, smoker, used estrogen-containing oral contraceptive, suffering from migraine with aura | Smoked ‘Peak Extreme’ | The next morning after smoking drugs, presented with felt-sided numbness, left facial weakness and dysfluency | CT angiogram, MRI, and head CT | Near occlusion of the right M1 segment with extensive infarction in the middle cerebral artery territory | Warfarin | Improved speech and comprehension | [113] |
| Male, 33, no medical history | Smoked two “joints” of synthetic cannabinoid product 10 min prior to the onset of symptoms; urine positive also for opiates; synthetic cannabinoid XLR-11-1-(5-fl uoropentyl)-1H-indol-3-yl) (2,2,3,3-tetramethylcyclopropyl) methanone was confirmed in the product used | Right-sided weakness and aphasia | Non-contrast head CT, and electrocardiography | Acute infarction located in the left insular cortex | Aspirin | The neurological problems were completely resolved in 3 days in the hospital; no return to follow-up | [115] |
| Male, 26, no family history of any stroke risk factors, non-smoker, non-alcohol consumer | Smoked spice “a few hours prior” to his symptom onset; concomitant use of marijuana in the past but not recent | Weakness of right side of face and arm, dysarthria, expressive aphasia that occur suddenly | Non-contrast head CT; CT perfusion; CT angiography; MRI | Hyperdense left middle cerebral artery (MCA); a large area of penumbra without core infarction; left MCA clot | Received IV tissue plasminogen activator (t-PA) | Improved clinically and did not return to follow-up | [109] |
| Female, 19, smoker, anxiety disorder and panic attacks | Smoked spice; urine drug screening positive for cannabinoids and confirmed for JWH-018 | A few minutes after smoking spice, the patient lost consciousness and started vomiting; mental status was persistently altered for several hours; presented with “shaking movements” of the legs and arms according to witnesses | CT angiogram and MRI | Infarctions in the left MCA with large distribution associated with punctate infarcts localized in the right cerebral hemisphere | - | She stabilized neurologically, but right hemiparesis and expressive aphasia remained at a follow-up office visit | [109] |
| Male, 31 | Smoked spice; toxicological tests confirmed XLR-11 | Generalized seizure | Head CT and digital subtraction angiography (DSA) | Hemorrhage in the bifrontal subarachnoid associated with left frontal and right parieto-occipital intraparenchymal hemorrhage | Intra-arterial verapamil | After 10 days from the event the paralysis of left leg, left homonymous hemianopsia and mentation improved | [110] |
| Female, 25, preeclampsia | Smoked synthetic marijuana; concomitant use of marijuana | Seizure after smoking synthetic and nonsynthetic marijuana; left leg monoplegia | CT, MRI, and DSA | SAH in the bilateral Sylvian fissures and interpeduncular and prepontine cisterns; restricted diffusion localized in the right frontal lobe, left cerebellum, left temporal lobe and bilateral parietal and occipital lobes, which is consistent with the diagnosis of multifocal AIS | Intra-arterial verapamil | Follow-up DSA showed worsening vertebrobasilar vasospasm | [110] |
| Subject/Age | Substance Exposure | Symptoms | Diagnostic Approach | Diagnosis | Intervention | Evolution | Reference |
|---|---|---|---|---|---|---|---|
| Female, 28 | Admitted to using heroin | Altered mental status | Head CT | A large 5.1 × 5-cm intraparenchymal hemorrhage in the left frontal lobe, vasogenic edema, and a 5-mm midline shift | Surgical intervention was unnecessary. After discharge, was transferred to rehabilitation | Improvement in cognitive function was mild; the patient continue to be confused and presented significant memory loss | [128] |
| Male, 29, without cardiovascular risk factors | Sniffed heroin with regularity in the last seven years | Left-sided hemihypesthesia and gait disturbance | MRI and MR angiography | Multiple cerebral and cerebellar areas of diffusion restriction in different territories; heroin-induced eosinophilia | Steroid pulse treatment (methylprednisolone 250 mg IV) in the first three days followed by another 21 days of oral prednisolone (60 mg)—for eosinophilia and antiplatelet therapy with aspirin | A slight improvement in his sensorium and gait but only incomplete recovery | [125] |
| Male, 33 | Heroin inhalation | Amnesia 48 h after first heroin inhalation | MRI | Cortical laminar necrosis of the left hippocampus without vascular abnormality | - | Impaired performance on the verbal and visual level | [134] |
| Male, 33 | Used heroin for 13 years Concomitant use of methamphetamine. For 6 months, started methadone treatment to quit heroin | Found unconsciousness | Brain CT and MRI | Acute ischemic strokes localized in bilateral fronto-parieto-temporal white matter and in bilateral corona radiate. Damage was noted in the bilateral globus pallidus and left cerebral peduncle; rhabdomyolysis | Active treatment in the intensive care unit | - | [135] |
| Subject/Age | Substance Exposure | Symptoms | Diagnostic Approach | Diagnosis | Reference |
|---|---|---|---|---|---|
| Male, 27, with an American father and a mother who was half Japanese, no known stroke risk factors, regularly training, AAS user | Methasterone, prostanozol for the past 6 months | Sudden right hemiparalysis, homonymous hemianopia, dysarthria, tinnitus, and double vision in the middle of muscle training | MRI with and without gadolinium enhancement, MR angiography, three-dimensional CT angiography, carotid ultrasonography, transcranial Doppler and transesophageal echocardiography, and duplex ultrasonography | Cardiogenic embolism and atrial septal aneurysm and large patent foramen ovule, suspected deep vein thrombosis | [170] |
| Male, 37, no history of alcohol or any other substance abuse, negative medical and family histories | Methandienone, methenolone acetate for the past 2 years | Acute right-sided hemiparesis (grade 3) with right-sided facial weakness, associated with a confused state followed a first-ever experience of generalized tonic–clonic seizure | Brain CT and MRI, ECG, chest X-ray, abdominal ultrasound, and echocardiography | Chronic infarction in the left frontal lobe and subacute left temporoparietal infarction Dilated cardiomyopathy and multiple thrombi in the left ventricle Hepatomegaly, mild ascites and bilateral pleural effusion in addition to a grade I nephropathy | [140] |
| Male, 16, healthy bodybuilder (weight 87 kg and height 181 cm), unremarkable past medical record | Concomitant use of cannabis (up to 1.5 g/day) and methandrostenolone (40 mg/day) for the past 5 months | Sudden dizziness and right hemiparesis | Cerebral CT, MRI, conventional and magnetic resonance angiography, transesophageal echocardiography, cervical Doppler duplex ultrasound, transcranial Doppler, and ECG | Acute ischemic stroke | [156] |
| Male, 39, bodybuilder, 3 months earlier sudden loss of vision in the left eye, weakness and numbness in the left upper and lower limbs, lasting less than 1 h, refused admission to hospital | Intramuscular injections of nandrolone twice weekly for the past three years | Dizziness and expressive aphasia for the last 6 h | Brain CT and MRI, ECG, chest X-ray, echocardiography, and magnetic resonance angiography | Dilated cardiomyopathy with LV thrombus formation; embolic stroke and peripheral vascular disease as a complication of the former | [141] |
| Male, 31, kickboxer | Nandrolone, testosterone clenbuterol since the age of 16; cocaine, ecstasy and alcohol abuser for three years | Patient disoriented in space, mild dysarthria without aphasic elements, oculocephalic preference to right, left homonymous hemianopsia, paresis (3/5), hemicorporal anesthesia on the left side and somatoagnosia | Cranial CT, cerebral arteriography, transesophageal and transthoracic echocardiography, and magnetic resonance angiography | Acute ischemic stroke: Cerebral infarction due to occlusion of the artery cerebral media of unknown etiology | [172] |
| Male | Injectable (nandrolone decanoate) and oral (methandrostenolone/danabol) three months prior to the incidence Previous intravenous (heroin), and inhaled (marijuana) drug use | Visual disturbances and left-sided weakness commencing 24 h prior to presentation Homonymous hemianopia, mild left-sided weakness in his upper limbs and ataxia in his left upper limb, and high hemoglobin (200 g/L) | Brain magnetic resonance, magnetic resonance angiography, transthoracic echocardiogram, and 24-h Holter monitoring, extensive hematological screening, and thrombophilia screening | Cerebral infarction: Extensive region of acute infarction in the right posterior cerebral artery territory and ongoing occlusion in his right posterior cerebral artery Polycythemia | [173] |
| Drugs of Abuse | Ischemic Stroke | Hemorrhagic Stroke | |
|---|---|---|---|
| Amphetamines | + | +++ | |
| Amphetamine derivatives | + | +++ Risk in young people without comorbidities | |
| Cocaine | In those with a history of use | In active users | |
| Cocaine | Hydrochloride | + | +++ |
| Crack | ++ | ++ | |
| Cannabis | ++ | + In recent case reports | |
| Synthetic cannabinoids | ++ | +In recent case reports | |
| Opiates/Heroin | ++ | +In recent case reports | |
| Anabolic androgenic steroids | ++ | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsatsakis, A.; Docea, A.O.; Calina, D.; Tsarouhas, K.; Zamfira, L.-M.; Mitrut, R.; Sharifi-Rad, J.; Kovatsi, L.; Siokas, V.; Dardiotis, E.; et al. A Mechanistic and Pathophysiological Approach for Stroke Associated with Drugs of Abuse. J. Clin. Med. 2019, 8, 1295. https://doi.org/10.3390/jcm8091295
Tsatsakis A, Docea AO, Calina D, Tsarouhas K, Zamfira L-M, Mitrut R, Sharifi-Rad J, Kovatsi L, Siokas V, Dardiotis E, et al. A Mechanistic and Pathophysiological Approach for Stroke Associated with Drugs of Abuse. Journal of Clinical Medicine. 2019; 8(9):1295. https://doi.org/10.3390/jcm8091295
Chicago/Turabian StyleTsatsakis, Aristides, Anca Oana Docea, Daniela Calina, Konstantinos Tsarouhas, Laura-Maria Zamfira, Radu Mitrut, Javad Sharifi-Rad, Leda Kovatsi, Vasileios Siokas, Efthimios Dardiotis, and et al. 2019. "A Mechanistic and Pathophysiological Approach for Stroke Associated with Drugs of Abuse" Journal of Clinical Medicine 8, no. 9: 1295. https://doi.org/10.3390/jcm8091295
APA StyleTsatsakis, A., Docea, A. O., Calina, D., Tsarouhas, K., Zamfira, L.-M., Mitrut, R., Sharifi-Rad, J., Kovatsi, L., Siokas, V., Dardiotis, E., Drakoulis, N., Lazopoulos, G., Tsitsimpikou, C., Mitsias, P., & Neagu, M. (2019). A Mechanistic and Pathophysiological Approach for Stroke Associated with Drugs of Abuse. Journal of Clinical Medicine, 8(9), 1295. https://doi.org/10.3390/jcm8091295













