Nutrition in Cancer Patients
Abstract
1. Introduction
2. Results
2.1. Nutritional Screening and Assessment
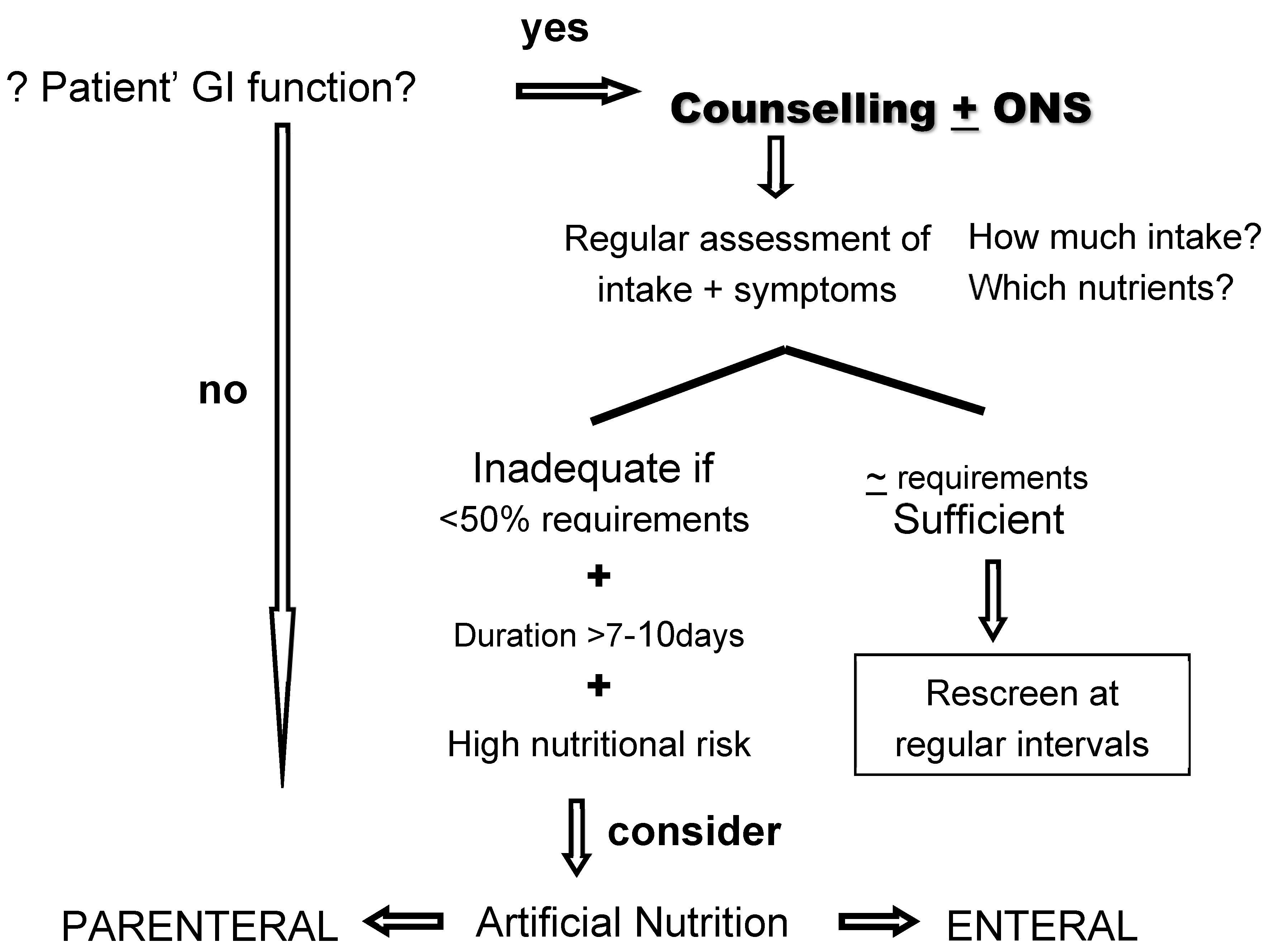
2.2. Nutritional Intervention
2.2.1. Individualised Nutritional Counselling
2.2.2. Artificial Nutrition
2.2.3. Surgery
2.2.4. Radiotherapy and Chemotherapy
2.3. Specific Nutrients
2.3.1. Protein
2.3.2. Eicosapentaenoic Acid and Fish Oil
2.3.3. Micronutrients
3. Discussion
4. Conclusions
Conflicts of Interest
References
- Fearon, K.; Barber, M. Moses A: The cancer cachexia syndrome. Surg. Oncol. Clin. N. Am. 2001, 10, 109–126. [Google Scholar] [CrossRef]
- Mattox, T.W. Cancer Cachexia: Cause, Diagnosis, and Treatment. Nutr. Clin. Pract. 2017, 32, 599–606. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.C.; Caan, B.J.; Meyerhardt, J.A.; Weltzien, E.; Xiao, J.; Feliciano, E.M.C.; Kroenke, C.H.; Castillo, A.; Kwan, M.L.; Prado, C.M. The deterioration of muscle mass and radiodensity is prognostic of poor survival in stage I–III colorectal cancer: A population-based cohort study (C-SCANS). J. Cachex Sarcopenia Muscle 2018, 9, 664–672. [Google Scholar] [CrossRef] [PubMed]
- Demark-Wahnefried, W.; Peterson, B.L.; Winer, E.P.; Marks, L.; Aziz, N.; Marcom, P.K.; Blackwell, K.; Rimer, B.K. Changes in Weight, Body Composition, and Factors Influencing Energy Balance Among Premenopausal Breast Cancer Patients Receiving Adjuvant Chemotherapy. J. Clin. Oncol. 2001, 19, 2381–2389. [Google Scholar] [CrossRef] [PubMed]
- De Wys, W.D.; Begg, C.; Lavin, P.T.; Band, P.R.; Bennett, J.M.; Bertino, J.R.; Cohen, M.H.; Douglass, H.O., Jr.; Engstrom, P.F.; Ezdinli, E.Z.; et al. Prognostic effect of weight loss prior to chemotherapy in cancer patients. Am. J. Med. 1980, 69, 491–497. [Google Scholar] [CrossRef]
- Wigmore, S.J.; Plester, C.E.; Ross, J.A.; Fearon, K.C.H.; Wigmore, S. Contribution of anorexia and hypermetabolism to weight loss in anicteric patients with pancreatic cancer. BJS 1997, 84, 196–197. [Google Scholar] [CrossRef]
- Belghiti, J.; Langonnet, F.; Bourstyn, E.; Fekete, F. Surgical implications of malnutrition and immunodeficiency in patients with carcinoma of the esophagus. Br. J. Surg. 1983, 70, 339–341. [Google Scholar] [CrossRef]
- Mantzorou, M.; Koutelidakis, A.; Theocharis, S.; Giaginis, C. Clinical Value of Nutritional Status in Cancer: What is its Impact and how it Affects Disease Progression and Prognosis? Nutr. Cancer 2017, 69, 1–26. [Google Scholar] [CrossRef]
- Ravasco, P.; Grillo, I.M.; Vidal, P.; Camilo, M. Nutritional Deterioration in Cancer: The Role of Disease and Diet. Clin. Oncol. 2003, 15, 443–450. [Google Scholar] [CrossRef]
- Prado, C.M.; Cushen, S.J.; Orsso, C.E.; Ryan, A.M. Sarcopenia and cachexia in the era of obesity: Clinical and nutritional impact. Proc. Nutr. Soc. 2016, 75, 188–198. [Google Scholar] [CrossRef]
- Bazzan, A.J.; Newberg, A.B.; Cho, W.C.; Monti, D.A. Diet and nutrition in cancersurvivorship and palliative care. Evid. Based Complement. Alternat. Med. 2013, 2013, 917647. [Google Scholar] [CrossRef] [PubMed]
- Orell-Kotikangas, H.; Österlund, P.; Mäkitie, O.; Saarilahti, K.; Ravasco, P.; Schwab, U.; Mäkitie, A.A. Cachexia at diagnosis is associated with poor survival in head and neck cancer patients. Acta Oto-Laryngol. 2017, 137, 778–785. [Google Scholar] [CrossRef] [PubMed]
- Fearon, K.; Strasser, F.; Anker, S.D.; Bosaeus, I.; Bruera, E.; Fainsinger, R.L.; Jatoi, A.; Loprinzi, C.; MacDonald, N.; Mantovani, G.; et al. Definition and classification of cancer cachexia: An international consensus statement. Lancet Oncol. 2011, 12, 489–495. [Google Scholar] [CrossRef]
- Donaldson, S. Nutritional consequences of radiotherapy. Cancer Res. 1997, 37, 2407–2413. [Google Scholar]
- Chao, K.S.C.; Perez, C.A.; Brady, L.W. (Eds.) Fundamentals of patient management. In Radiation Oncology: Management Decisions; Lippincot-Raven: Philadelphia, PA, USA, 2014; pp. 1–13. [Google Scholar]
- Blauwhoff-Buskermolen, S.; Versteeg, K.S.; De Van Der Schueren, M.A.; Braver, N.R.D.; Berkhof, J.; Langius, J.A.; Verheul, H.M. Loss of Muscle Mass During Chemotherapy Is Predictive for Poor Survival of Patients with Metastatic Colorectal Cancer. J. Clin. Oncol. 2016, 34, 1339–1344. [Google Scholar] [CrossRef]
- Van der Schueren, M.A.; Van Leeuwen, P.A.; Sauerwein, H.P.; Kuik, D.J.; Snow, G.B.; Quak, J.J. Assessment of malnutrition parameters in head and neck cancer patients and their relation to postoperative complications. Head Neck 1997, 19, 419–425. [Google Scholar] [CrossRef]
- Weimann, A.; Braga, M.; Carli, F.; Higashiguchi, T.; Hübner, M.; Klek, S.; Laviano, A.; Ljungqvist, O.; Lobo, D.N.; Martindale, R.; et al. ESPEN guideline: Clinical nutrition in surgery. Clin. Nutr. 2017, 36, 623–650. [Google Scholar] [CrossRef]
- Arends, J.; Bachmann, P.; Baracos, V.; Barthelemy, N.; Bertz, H.; Bozzetti, F.; Fearon, K.; Hütterer, E.; Isenring, E.; Kaasa, S.; et al. ESPEN guidelines on nutrition in cancer patients. Clin. Nutr. 2017, 36, 11–48. [Google Scholar] [CrossRef]
- Arends, J.; Baracos, V.; Bertz, H.; Bozzetti, F.; Calder, P.; Deutz, N.; Erickson, N.; Laviano, A.; Lisanti, M.; Lobo, D.; et al. ESPEN expert group recommendations for action against cancer-related malnutrition. Clin. Nutr. 2017, 36, 1187–1196. [Google Scholar] [CrossRef]
- De Las Peñas, R.; Majem, M.; Perez-Altozano, J.; Virizuela, J.A.; Cancer, E.; Diz, P.; Donnay, O.; Hurtado, A.; Jimenez-Fonseca, P.; Ocon, M.J. SEOM clinical guidelines on nutrition in cancer patients (2018). Clin. Transl. Oncol. 2019, 21, 87–93. [Google Scholar] [CrossRef]
- Raspé, C.; Flöther, L.; Schneider, R.; Bucher, M.; Piso, P. Best practice for perioperative management of patients with cytoreductive surgery and HIPEC. Eur. J. Surg. Oncol. 2017, 43, 1013–1027. [Google Scholar] [CrossRef]
- French Speaking Society of Clinical Nutrition and Metabolism (SFNEP). Clinical nutrition guidelines of the French Speaking Society of Clinical Nutrition and Metabolism (SFNEP): Summary of recommendations for adults undergoing non-surgical anticancer treatment. Dig. Liver Dis. 2014, 46, 667–674. [Google Scholar] [CrossRef]
- Prado, C.M.; Sawyer, M.B.; Ghosh, S.; Lieffers, J.R.; Esfandiari, N.; Antoun, S.; Baracos, V. Central tenet of cancer cachexia therapy: Do patients with advanced cancer have exploitable anabolic potential? Am. J. Clin. Nutr. 2013, 98, 1012–1019. [Google Scholar] [CrossRef]
- Benoist, S.; Brouquet, A. Nutritional assessment and screening for malnutrition. J. Visc. Surg. 2015, 152 (Suppl. 1), S3–S7. [Google Scholar] [CrossRef]
- Thompson, K.L.; Elliott, L.; Fuchs-Tarlovsky, V.; Levin, R.M.; Voss, A.C.; Piemonte, T. Rd Oncology Evidence-Based Nutrition Practice Guideline for Adults. J. Acad. Nutr. Diet. 2017, 117, 297–310. [Google Scholar] [CrossRef]
- Talwar, B.; Donnelly, R.; Skelly, R.; Donaldson, M. Nutritional management in head and neck cancer: United Kingdom National Multidisciplinary Guidelines. J. Laryngol. Otol. 2016, 130, S32–S40. [Google Scholar] [CrossRef]
- Orell-Kotikangas, H.; Österlund, P.; Saarilahti, K.; Ravasco, P.; Schwab, U.; Mäkitie, A.A. NRS-2002 for pre-treatment nutritional risk screening and nutritional status assessment in head and neck cancer patients. J. Support. Care Cancer 2015, 23, 1495–1502. [Google Scholar] [CrossRef]
- Boléo-Tomé, C.; Monteiro Grillo, I.; Camilo, M.E.; Ravasco, P. Validation of the Malnutrition Universal Screening Tool (MUST) in cancer. Br. J. Nutr. 2012, 108, 343–348. [Google Scholar] [CrossRef]
- Boléo-Tomé, C.; Chaves, M.; Monteiro-Grillo, I.; Camilo, M.; Ravasco, P. Teaching Nutrition Integration: MUST Screening in Cancer. Oncologist 2011, 16, 239–245. [Google Scholar] [CrossRef][Green Version]
- Ottery, F. Definition of standardised nutritional assessment and interventional pathways in oncology. Nutrition 1996, 12, s15–s19. [Google Scholar] [CrossRef]
- Ortiz, C. Analysis of clinical guidelines in oncology. Nutr. Hosp. 2016, 33 (Suppl. 1), 40–49. [Google Scholar]
- Ravasco, P.; Monteiro-Grillo, I.; Camilo, M.E. Does nutrition influence quality of life in cancer patients undergoing radiotherapy? Radiother. Oncol. 2003, 67, 213–220. [Google Scholar] [CrossRef]
- Monteiro-Grillo, I.; Vidal, P.M.; Camilo, M.E.; Ravasco, P. Cancer: Disease and nutrition are key determinants of patients’ quality of life. Support. Care Cancer 2004, 12, 246–252. [Google Scholar] [CrossRef]
- Ravasco, P.; Monteiro Grillo, I.; Marques Vidal, P.; Camilo, M.E. Dietary conseling improves patient outcomes: A prospective, randomized, controlled trial in colorectal cancer patients undergoing radiotherapy. J. Clin. Oncol. 2005, 23, 1431–1438. [Google Scholar] [CrossRef]
- Ravasco, P.; Vidal, P.M.; Camilo, M.E.; Monteiro-Grillo, I.; Monteiro-Grillo, I. Impact of nutrition on outcome: A prospective randomized controlled trial in patients with head and neck cancer undergoing radiotherapy. Head Neck 2005, 27, 659–668. [Google Scholar] [CrossRef]
- Martin, L.; Senesse, P.; Gioulbasanis, I.; Antoun, S.; Bozzetti, F.; Deans, C.; Strasser, F.; Thoresen, L.; Jagoe, R.T.; Chasen, M.; et al. Diagnostic criteria for the classification of cancer-associated weight loss. J. Clin. Oncol. 2015, 33, 90–99. [Google Scholar] [CrossRef]
- Von Haehling, S.; Morley, J.E.; Anker, S.D. An overview of sarcopenia: Facts and numbers on prevalence and clinical impact. J. Cachexia Sarcopenia Muscle 2010, 1, 129–133. [Google Scholar] [CrossRef]
- Orell-Kotikangas, H.; Österlund, P.; Saarilahti, K.; Ravasco, P.; Schwab, U.; Mäkitie, A.A. Nutritional Counseling for Head and Neck Cancer Patients Undergoing (Chemo) Radiotherapy-A Prospective Randomized Trial. Front. Nutr. 2019, 18, 22. [Google Scholar] [CrossRef]
- Van Bokhorst-de van der Schueren, M.A. Nutritional support strategies for malnourished cancer patients. Eur. J. Oncol. Nurs. 2005, 9 (Suppl. 2), S74–S83. [Google Scholar] [CrossRef]
- Bauer, J.; Isenring, E.; Ferguson, M. Dietary counseling: Evidence in chemotherapy patients. Support. Oncol. 2008, 6, 354–355. [Google Scholar]
- Isenring, E.A.; Bauer, J.D.; Capra, S. Nutrition Support Using the American Dietetic Association Medical Nutrition Therapy Protocol for Radiation Oncology Patients Improves Dietary Intake Compared with Standard Practice. J. Am. Diet. Assoc. 2007, 107, 404–412. [Google Scholar] [CrossRef]
- Ravasco, P.; Monteiro Grillo, I.; Camilo, M. Dietary individualized counseling benefits in colorectal cancer: The long term follow-up of a randomized controlled trial of nutritional therapy. Am. J. Clin. Nutr. 2012, 96, 1346–1353. [Google Scholar] [CrossRef]
- Staun, M.; Pironi, L.; Bozzetti, F.; Baxter, J.; Forbes, A.; Joly, F.; Jeppesena, P.; Morenog, J.; Hebuterne, X.; Pertkiewicz, M.; et al. ESPEN Guidelines on Parenteral Nutrition: Home Parenteral Nutrition (HPN) in Adult Patients. Available online: http://espen.info/documents/0909/Home Parenteral Nutrition in adults.pdf (accessed on 17 July 2019).
- Souza, N.C.S.; Simões, B.P.; Júnior, A.A.J.; Chiarello, P.G. Changes in Intestinal Permeability and Nutritional Status after Cytotoxic Therapy in Patients with Cancer. Nutr. Cancer 2014, 66, 576–582. [Google Scholar] [CrossRef]
- O’Reilly, D.; Fou, L.; Hasler, E.; Hawkins, J.; O’Connell, S.; Pelone, F.; Callaway, M.; Campbell, F.; Capel, M.; Charnley, R.; et al. Diagnosis and management of pancreatic cancer in adults: A summary of guidelines from the UK National Institute for Health and Care Excellence. Pancreatology 2018, 18, 962–970. [Google Scholar] [CrossRef]
- Lis, C.G.; Gupta, D.; Lammersfeld, C.A.; Markman, M.; Vashi, P.G. Role of nutritional status in predicting quality of life outcomes in cancer—A systematic review of the epidemiological literature. Nutr. J. 2012, 11, 27. [Google Scholar] [CrossRef]
- Nightingale, J.; Young, A.; Hawthorne, B.; McKee, R.; McKinlay, A.; Rafferty, G.; Protheroe, S.; Culkin, A.; Eastwood, J.; Farrer, K.; et al. Position Statement from BIFA Committee. Available online: https://www.bapen.org.uk/nutrition-support/parenteral-nutrition/position-statement-from-bifa-committee (accessed on 17 July 2019).
- Bozzetti, F.; Bozzetti, V. Is the intravenous supplementation of amino acid to cancer patients adequate? A critical appraisal of literature. Clin. Nutr. 2013, 32, 142–146. [Google Scholar] [CrossRef]
- Wolfe, R.R. The 2017 Sir David P Cuthbertson lecture. Amino acids and muscle protein metabolism in critical care. Clin. Nutr. 2018, 37, 1093–1100. [Google Scholar] [CrossRef]
- Körber, J.; Pricelius, S.; Heidrich, M.; Muller, M.J. Increased lipid utilization in weight losing and weight stable cancer patients with normal body weight. Eur. J. Clin. Nutr. 1999, 53, 740–745. [Google Scholar] [CrossRef]
- Cao, D.-X.; Wu, G.-H.; Zhang, B.; Quan, Y.-J.; Wei, J.; Jin, H.; Jiang, Y.; Yang, Z.-A. Resting energy expenditure and body composition in patients with newly detected cancer. Clin. Nutr. 2010, 29, 72–77. [Google Scholar] [CrossRef]
- Finocchiaro, C.; Gervasio, S.; Agnello, E.; Appiano, S.; Bertetto, O.; Ciuffreda, L.; Montrucchio, G.; Luisa, A.M.; D’andrea, F.; Domeniconi, D.; et al. Multicentric study on home parenteral nutrition in advanced cancer patients. Riv. Ital. Nutr. Parenter. Enter. 2002, 20, 98–107. [Google Scholar]
- Seys, P.; Tadmouri, A.; Senesse, P.; Radji, A.; Rotarski, M.; Balian, A.; Culine, S.; Dufour, P.; Chambrier, C. Home parenteral nutrition in elderly patients with cancer: An observational prospective study. Bull. Cancer 2014, 101, 243–249. [Google Scholar] [CrossRef]
- Culine, S.; Chambrier, C.; Tadmouri, A.; Senesse, P.; Seys, P.; Radji, A.; Rotarski, M.; Balian, A.; Dufour, P. Home parenteral nutrition improves quality of life and nutritional status in patients with cancer: A French observational multicentre study. Support. Care Cancer 2014, 22, 1867–1874. [Google Scholar] [CrossRef]
- Vashi, P.G.; Dahlk, S.; Popiel, B.; Lammersfeld, C.A.; Ireton-Jones, C.; Gupta, D. A longitudinal study investigating quality of life and nutritional outcomes in advanced cancer patients receiving home parenteral nutrition. BMC Cancer 2014, 14, 593. [Google Scholar] [CrossRef]
- Girke, J.; Seipt, C.; Markowski, A.; Luettig, B.; Schettler, A.; Momma, M.; Schneider, A.S. Quality of Life and Nutrition Condition of Patients Improve Under Home Parenteral Nutrition: An Exploratory Study. Nutr. Clin. Pract. 2016, 31, 659–665. [Google Scholar] [CrossRef]
- Keane, N.; Fragkos, K.C.; Patel, P.S.; Bertsch, F.; Mehta, S.J.; Di Caro, S.; Rahman, F. Performance status, prognostic scoring, and parenteral nutrition requirements predict survival in patients with advanced cancer receiving home parenteral nutrition. Nutr. Cancer 2018, 70, 73–82. [Google Scholar] [CrossRef]
- Bozzetti, F.; Arends, J.; Lundholm, K.; Micklewright, A.; Zürcher, G.; Muscaritoli, M. ESPEN Guidelines on Parenteral Nutrition: Non-surgical oncology. Clin. Nutr. 2009, 28, 445–454. [Google Scholar] [CrossRef]
- Scolapio, J.; Picco, M.; Tarrosa, V. Enteral versus parenteral nutrition: The patient’s preference. J. Parenter. Enter. Nutr. 2002, 26, 248–250. [Google Scholar] [CrossRef]
- Obermair, A.; Simunovic, M.; Isenring, L.; Janda, M. Nutrition interventions in patients with gynecological cancers requiring surgery. Gynecol. Oncol. 2017, 145, 192–199. [Google Scholar] [CrossRef]
- Baldwin, C.; Weekes, C.E. Dietary counselling with or without oral nutritional supplements in the management of malnourished patients: A systematic review and meta-analysis of randomised controlled trials. J. Hum. Nutr. Diet. 2012, 25, 411–426. [Google Scholar] [CrossRef]
- Deutz, N.E.; Safar, A.; Schutzler, S.; Memelink, R.; Ferrando, A.; Spencer, H.; Van Helvoort, A.; Wolfe, R.R. Muscle protein synthesis in cancer patients can be stimulated with a specially formulated medical food. Clin. Nutr. 2011, 30, 759–768. [Google Scholar] [CrossRef]
- Omlin, A.; Blum, D.; Wierecky, J.; Haile, S.R.; Ottery, F.D.; Strasser, F. Nutrition impact symptoms in advanced cancer patients: Frequency and specific interventions, a case–control study. J. Cachex Sarcopenia Muscle 2013, 4, 55–61. [Google Scholar] [CrossRef]
- Van Blarigan, E.L.; Fuchs, C.S.; Niedzwiecki, D.; Zhang, S.; Saltz, L.B.; Mayer, R.J.; Mowat, R.B.; Whittom, R.; Hantel, A.; Benson, A.; et al. Association of Survival with Adherence to the American Cancer Society Nutrition and Physical Activity Guidelines for Cancer Survivors After Colon Cancer Diagnosis: The CALGB 89803/Alliance Trial. JAMA Oncol. 2018, 4, 783–790. [Google Scholar] [CrossRef]
- Papallardo, G.; Almeida, A.; Ravasco, P. Eicosapentaenoic acid in cancer: Does it improve body composition and modulate metabolism? Nutrition 2015, 31, 549–555. [Google Scholar] [CrossRef]
- Ravasco, P.; Lavriv, D.; Neves, P. Should omega-3 be used in cancer cachexia? Clin. Nutr. ESPEN 2018, 25, 18–25. [Google Scholar]
- Hering, J.; Garrean, S.; Dekoj, T.R.; Razzak, A.; Saied, A.; Trevino, J.; Babcock, T.A.; Espat, N.J. Inhibition of Proliferation by Omega-3 Fatty Acids in Chemoresistant Pancreatic Cancer Cells. Ann. Surg. Oncol. 2007, 14, 3620–3628. [Google Scholar] [CrossRef]
- Murphy, R.A.; Mourtzakis, M.; Mazurak, V.C. n-3 polyunsaturated fatty acids: The potential role for supplementation in cancer. Curr. Opin. Clin. Nutr. Metab. Care 2012, 15, 246–251. [Google Scholar] [CrossRef]
- Geisler, C.; Prado, C.; Müller, M. Inadequacy of Body Weight-Based Recommendations for Individual Protein Intake—Lessons from Body Composition Analysis. Nutrients 2017, 9, 23. [Google Scholar] [CrossRef]
- Norman, H.A.; Butrum, R.R.; Feldman, E.; Picciano, M.F.; Rivlin, R.; Simopoulos, A.; Wargovich, M.J.; Weisburger, E.K.; Zeisel, S.H.; Heber, D.; et al. The Role of Dietary Supplements during Cancer Therapy. J. Nutr. 2003, 133, 3794S–3799S. [Google Scholar] [CrossRef]
- Rock, C.L.; Doyle, C.; Demark-Wahnefried, W.; Meyerhardt, J.; Courneya, K.S.; Schwartz, A.L.; Bandera, E.V.; Hamilton, K.K.; Grant, B.; McCullough, M.; et al. Nutrition and physical activity guidelines for cancer survivors. CA Cancer J. Clin. 2012, 62, 242–274. [Google Scholar] [CrossRef]
- Stene, G.B.; Helbostad, J.L.; Amundsen, T.; Sørhaug, S.; Hjelde, H.; Kaasa, S.; Grønberg, B.H. Changes in skeletal muscle mass during palliative chemotherapy in patients with advanced lung cancer. Acta Oncol. 2015, 54, 340–348. [Google Scholar] [CrossRef]
- Lieffers, J.R.; Bathe, O.F.; Fassbender, K.; Winget, M.; Baracos, V.E. Sarcopenia is associated with postoperative infection and delayed recovery from colorectal cancer resection surgery. Br. J. Cancer 2012, 107, 931–936. [Google Scholar] [CrossRef]
|
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ravasco, P. Nutrition in Cancer Patients. J. Clin. Med. 2019, 8, 1211. https://doi.org/10.3390/jcm8081211
Ravasco P. Nutrition in Cancer Patients. Journal of Clinical Medicine. 2019; 8(8):1211. https://doi.org/10.3390/jcm8081211
Chicago/Turabian StyleRavasco, Paula. 2019. "Nutrition in Cancer Patients" Journal of Clinical Medicine 8, no. 8: 1211. https://doi.org/10.3390/jcm8081211
APA StyleRavasco, P. (2019). Nutrition in Cancer Patients. Journal of Clinical Medicine, 8(8), 1211. https://doi.org/10.3390/jcm8081211





