Effect of Iron Supplementation on the Outcome of Non-Progressive Pulmonary Mycobacterium tuberculosis Infection
Abstract
:1. Introduction
2. Materials and Methods
2.1. Bacteria and Chemicals
2.2. Experimental Design
2.3. Rabbit Infection and Treatment
2.4. Histology Staining
2.5. Measurement of Blood Parameters
2.6. Measurement of Plasma and Lung Tissue Iron
2.7. RNA Isolation from Rabbit Lung and Blood
2.8. Quantitative Real-Time PCR Analysis (qPCR)
2.9. Statistical Analysis
3. Results
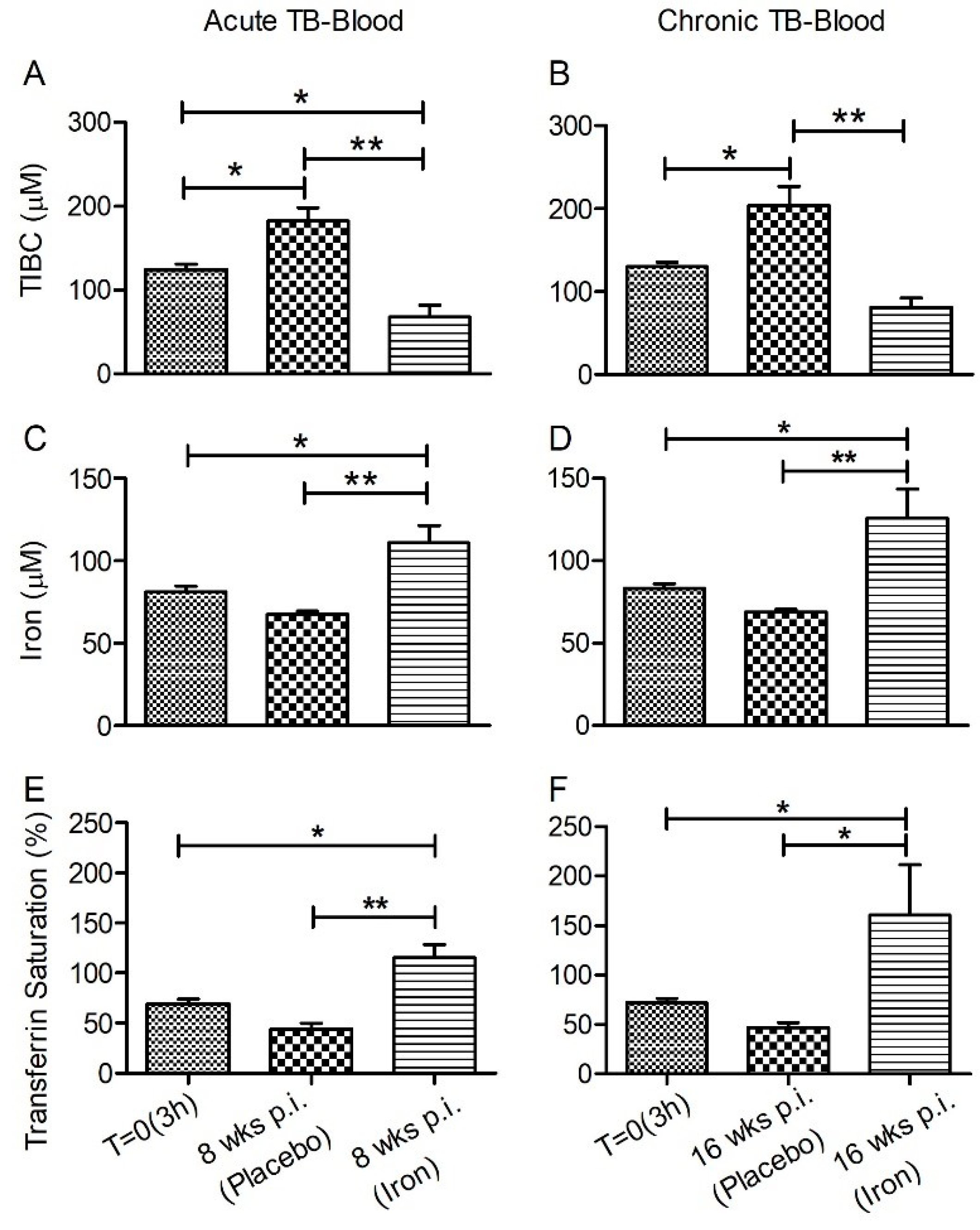
3.1. Iron Parameters in Fe-Supplemented Rabbits during Acute or Chronic Mtb Infection
3.2. Systemic and Lung Fe Levels in Mtb-Infected Rabbits with or without Fe-Supplementation
3.3. Fe-Supplementation Does Not Affect Systemic Host Iron-Responsive Gene Expression during Mtb Infection
3.4. Fe-Supplementation Perturbs Expression of Host Iron-Responsive Genes in the Lungs of Mtb-Infected Rabbits
3.5. Fe-Supplementation Affects Systemic Expression of Host Immune Response Genes in Mtb-Infected Rabbits
3.6. Fe-Supplementation Perturbs Expression of Host Immune Response Genes in the Lungs of Mtb-Infected Rabbits
3.7. Fe-Supplementation Did Not Affect the Lung Bacillary Load in Mtb-Infected Rabbits
3.8. Fe-Supplementation Did Not Affect Disease Pathology in Rabbit Lungs Infected with Mtb
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Dye, C.; Williams, B.G. The population dynamics and control of tuberculosis. Science 2010, 328, 856–861. [Google Scholar] [CrossRef] [PubMed]
- Behr, M.A.; Edelstein, P.H.; Ramakrishnan, L. Revisiting the timetable of tuberculosis. BMJ 2018, 362, k2738. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- O’Garra, A.; Redford, P.S.; McNab, F.W.; Bloom, C.I.; Wilkinson, R.J.; Berry, M.P. The immune response in tuberculosis. Annu. Rev. Immunol. 2013, 31, 475–527. [Google Scholar] [CrossRef] [PubMed]
- Ernst, J.D. The immunological life cycle of tuberculosis. Nat. Rev. Immunol. 2012, 12, 581–591. [Google Scholar] [CrossRef] [PubMed]
- Ganz, T.; Nemeth, E. Iron homeostasis in host defence and inflammation. Nat. Rev. Immunol. 2015, 15, 500–510. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Michels, K.R.; Zhang, Z.; Bettina, A.M.; Cagnina, R.E.; Stefanova, D.; Burdick, M.D.; Vaulont, S.; Nemeth, E.; Ganz, T.; Mehrad, B. Hepcidin-mediated iron sequestration protects against bacterial dissemination during pneumonia. JCI Insight 2017, 2, e92002. [Google Scholar] [CrossRef] [PubMed]
- Stefanova, D.; Raychev, A.; Arezes, J.; Ruchala, P.; Gabayan, V.; Skurnik, M.; Dillon, B.J.; Horwitz, M.A.; Ganz, T.; Bulut, Y.; et al. Endogenous hepcidin and its agonist mediate resistance to selected infections by clearing non-transferrin-bound iron. Blood 2017, 130, 245–257. [Google Scholar] [CrossRef]
- Chao, A.; Sieminski, P.J.; Owens, C.P.; Goulding, C.W. Iron acquisition in Mycobacterium tuberculosis. Chem. Rev. 2019, 119, 1193–1220. [Google Scholar] [CrossRef] [PubMed]
- Clemens, D.L.; Horwitz, M.A. The Mycobacterium tuberculosis phagosome interacts with early endosomes and is accessible to exogenously administered transferrin. J. Exp. Med. 1996, 184, 1349–1355. [Google Scholar] [CrossRef]
- Boelaert, J.R.; Vandecasteele, S.J.; Appelberg, R.; Gordeuk, V.R. The effect of the host’s iron status on tuberculosis. J. Infect. Dis. 2007, 195, 1745–1753. [Google Scholar] [CrossRef]
- Ratledge, C. Iron, mycobacteria and tuberculosis. Tuberculosis 2004, 84, 110–130. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, G.M. Control of iron metabolism in Mycobacterium tuberculosis. Trends Microbiol. 2006, 14, 320–327. [Google Scholar] [CrossRef] [PubMed]
- Kurthkoti, K.; Amin, H.; Marakalala, M.J.; Ghanny, S.; Subbian, S.; Sakatos, A.; Livny, J.; Fortune, S.M.; Berney, M.; Rodriguez, G.M. The capacity of Mycobacterium tuberculosis to survive iron starvation might enable it to persist in iron-deprived microenvironments of human granulomas. MBio 2017, 8, e01092-17. [Google Scholar] [CrossRef] [PubMed]
- Tufariello, J.M.; Chapman, J.R.; Kerantzas, C.A.; Wong, K.W.; Vilchèze, C.; Jones, C.M.; Cole, L.E.; Tinaztepe, E.; Thompson, V.; Fenyö, D.; et al. Separable roles for Mycobacterium tuberculosis ESX-3 effectors in iron acquisition and virulence. Proc. Natl. Acad. Sci. USA 2016, 113, E348–E357. [Google Scholar] [CrossRef] [PubMed]
- Madigan, C.A.; Martinot, A.J.; Wei, J.R.; Madduri, A.; Cheng, T.Y.; Young, D.C.; Layre, E.; Murry, J.P.; Rubin, E.J.; Moody, D.B. Lipidomic analysis links mycobactin synthase K to iron uptake and virulence in M. tuberculosis. PLoS Pathog. 2015, 11, e1004792. [Google Scholar] [CrossRef] [PubMed]
- Kochan, I. The role of iron in bacterial infections, with special consideration of host-tubercle bacillus interaction. In Current Topics in Microbiology and Immunology; Springer: Berlin, Heidelberg, Germany, 1973; pp. 1–30. [Google Scholar]
- Murray, M.J.; Murray, A.B.; Murray, M.B.; Murray, C.J. The adverse effect of iron repletion on the course of certain infections. Br. Med. J. 1978, 2, 1113–1115. [Google Scholar] [CrossRef]
- Lounis, N.; Maslo, C.; Truffot-Pernot, C.; Grosset, J.; Boelaert, R.J. Impact of iron loading on the activity of isoniazid or ethambutol in the treatment of murine tuberculosis. Int. J. Tuberc. Lung Dis. 2003, 7, 575–579. [Google Scholar] [PubMed]
- Subbian, S.; Tsenova, L.; O’Brien, P.; Yang, G.; Kushner, N.L.; Parsons, S.; Peixoto, B.; Fallows, D.; Kaplan, G. Spontaneous latency in a rabbit model of pulmonary tuberculosis. Am. J. Pathol. 2012, 181, 1711–1724. [Google Scholar] [CrossRef]
- Rashtchizadeh, N.; Ettehad, S.; DiSilvestro, R.A.; Mahdavi, R. Antiatherogenic effects of zinc are associated with copper in iron-overloaded hypercholesterolemic rabbits. Nutr. Res. 2008, 28, 98–105. [Google Scholar] [CrossRef]
- Deschemin, J.C.; Mathieu, J.R.R.; Zumerle, S.; Peyssonnaux, C.; Vaulont, S. Pulmonary iron homeostasis in hepcidin knockout mice. Front. Physiol. 2017, 8, 804. [Google Scholar] [CrossRef]
- Subbian, S.; Eugenin, E.; Kaplan, G. Detection of Mycobacterium tuberculosis in latently infected lungs by immunohistochemistry and confocal microscopy. J. Med. Microbiol. 2014, 63, 1432–1435. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Avecilla, S.T.; Marionneaux, S.M.; Leiva, T.D.; Tonon, J.A.; Chan, V.T.; Moung, C.; Meagher, R.C.; Maslak, P. Comparison of manual hematocrit determinations versus automated methods for hematopoietic progenitor cell apheresis products. Transfusion 2016, 56, 528–532. [Google Scholar] [CrossRef] [PubMed]
- Subbian, S.; O’Brien, P.; Kushner, N.L.; Yang, G.; Tsenova, L.; Peixoto, B.; Bandyopadhyay, N.; Bader, J.S.; Karakousis, P.C.; Fallows, D.; et al. Molecular immunologic correlates of spontaneous latency in a rabbit model of pulmonary tuberculosis. Cell Commun. Signal. 2013, 11, 16. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez Martinez, A.; Niemela, O.; Parkkila, S. Hepatic and extrahepatic expression of the new iron regulatory protein hemojuvelin. Haematologica 2004, 89, 1441–1445. [Google Scholar] [PubMed]
- D’Alessio, F.; Hentze, M.W.; Muckenthaler, M.U. The hemochromatosis proteins HFE, TfR2, and HJV form a membrane-associated protein complex for hepcidin regulation. J. Hepatol. 2012, 57, 1052–1060. [Google Scholar] [CrossRef]
- Babitt, J.L.; Huang, F.W.; Wrighting, D.M.; Xia, Y.; Sidis, Y.; Samad, T.A.; Campagna, J.A.; Chung, R.T.; Schneyer, A.L.; Woolf, C.J.; et al. Bone morphogenetic protein signaling by hemojuvelin regulates hepcidin expression. Nat. Genet. 2006, 38, 531–539. [Google Scholar] [CrossRef] [PubMed]
- Sabelli, M.; Montosi, G.; Garuti, C.; Caleffi, A.; Oliveto, S.; Biffo, S.; Pietrangelo, A. Human macrophage ferroportin biology and the basis for the ferroportin disease. Hepatology 2017, 65, 1512–1525. [Google Scholar] [CrossRef]
- Arosio, P.; Elia, L.; Poli, M. Ferritin, cellular iron storage and regulation. IUBMB Life 2017, 69, 414–422. [Google Scholar] [CrossRef]
- El-Rifaie, A.A.; Sabry, D.; Doss, R.W.; Kamal, M.A.; Abd El Hassib, D.M. Heme oxygenase and iron status in exosomes of psoriasis patients. Arch. Dermatol. Res. 2018, 310, 651–656. [Google Scholar] [CrossRef]
- Kasai, S.; Mimura, J.; Ozaki, T.; Itoh, K. Emerging regulatory role of Nrf2 in iron, heme, and hemoglobin metabolism in physiology and disease. Front. Vet. Sci. 2018, 5, 242. [Google Scholar] [CrossRef]
- Flo, T.H.; Smith, K.D.; Sato, S.; Rodriguez, D.J.; Holmes, M.A.; Strong, R.K.; Akira, S.; Aderem, A. Lipocalin 2 mediates an innate immune response to bacterial infection by sequestrating iron. Nature 2004, 432, 917–921. [Google Scholar] [CrossRef] [PubMed]
- Wareham, A.S.; Tree, J.A.; Marsh, P.D.; Butcher, P.D.; Dennis, M.; Sharpe, S.A. Evidence for a role for interleukin-17, Th17 cells and iron homeostasis in protective immunity against tuberculosis in cynomolgus macaques. PLoS ONE 2014, 9, e88149. [Google Scholar] [CrossRef] [PubMed]
- Harrington-Kandt, R.; Stylianou, E.; Eddowes, L.A.; Lim, P.J.; Stockdale, L.; Pinpathomrat, N.; Bull, N.; Pasricha, J.; Ulaszewska, M.; Beglov, Y.; et al. Hepcidin deficiency and iron deficiency do not alter tuberculosis susceptibility in a murine M.tb infection model. PLoS ONE 2018, 13, e0191038. [Google Scholar]
- McDermid, J.M.; Prentice, A.M. Iron and infection: Effects of host iron status and the iron-regulatory genes haptoglobin and NRAMP1 (SLC11A1) on host-pathogen interactions in tuberculosis and HIV. Clin. Sci. 2006, 110, 503–524. [Google Scholar] [CrossRef] [PubMed]
- Thom, R.E.; Elmore, M.J.; Williams, A.; Andrews, S.C.; Drobniewski, F.; Marsh, P.D.; Tree, J.A. The expression of ferritin, lactoferrin, transferrin receptor and solute carrier family 11A1 in the host response to BCG-vaccination and Mycobacterium tuberculosis challenge. Vaccine 2012, 30, 3159–3168. [Google Scholar] [CrossRef] [PubMed]
- Agoro, R.; Benmerzoug, S.; Rose, S.; Bouyer, M.; Gozzelino, R.; Garcia, I.; Ryffel, B.; Quesniaux, V.F.J.; Mura, C. An iron-rich diet decreases the Mycobacterial burden and correlates with hepcidin upregulation, lower levels of proinflammatory mediators, and increased T-cell recruitment in a model of Mycobacterium bovis Bacille Calmette-Guerin infection. J. Infect. Dis. 2017, 216, 907–918. [Google Scholar] [CrossRef] [PubMed]
- Abreu, R.; Quinn, F.; Giri, P.K. Role of the hepcidin-ferroportin axis in pathogen-mediated intracellular iron sequestration in human phagocytic cells. Blood Adv. 2018, 2, 1089–1100. [Google Scholar] [CrossRef] [PubMed]
- Paesano, R.; Natalizi, T.; Berlutti, F.; Valenti, P. Body iron delocalization: The serious drawback in iron disorders in both developing and developed countries. Pathog. Glob. Health 2012, 106, 200–216. [Google Scholar] [CrossRef]
- Domingo-Gonzalez, R.; Prince, O.; Cooper, A.; Khader, S.A. Cytokines and chemokines in Mycobacterium tuberculosis infection. Microbiol. Spectr. 2016, 4. [Google Scholar] [CrossRef]
- Serafin-Lopez, J.; Chacon-Salinas, R.; Munoz-Cruz, S.; Enciso-Moreno, J.A.; Estrada-Parra, S.A.; Estrada-Garcia, I. The effect of iron on the expression of cytokines in macrophages infected with Mycobacterium tuberculosis. Scand. J. Immunol. 2004, 60, 329–337. [Google Scholar] [CrossRef]
- Byrd, T.F. Tumor necrosis factor alpha (TNFalpha) promotes growth of virulent Mycobacterium tuberculosis in human monocytes iron-mediated growth suppression is correlated with decreased release of TNFalpha from iron-treated infected monocytes. J. Clin. Invest. 1997, 99, 2518–2529. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, R.; Jung, C.L.; Gabayan, V.; Deng, J.C.; Ganz, T.; Nemeth, E.; Bulut, Y. Hepcidin induction by pathogens and pathogen-derived molecules is strongly dependent on interleukin-6. Infect. Immun. 2014, 82, 745–752. [Google Scholar] [CrossRef] [PubMed]
- Armitage, A.E.; Eddowes, L.A.; Gileadi, U.; Cole, S.; Spottiswoode, N.; Selvakumar, T.A.; Ho, L.P.; Townsend, A.R.; Drakesmith, H. Hepcidin regulation by innate immune and infectious stimuli. Blood 2011, 118, 4129–4139. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nairz, M.; Dichtl, S.; Schroll, A.; Haschka, D.; Tymoszuk, P.; Theurl, I.; Weiss, G. Iron and innate antimicrobial immunity-depriving the pathogen, defending the host. J. Trace Elem. Med. Biol. 2018, 48, 118–133. [Google Scholar] [CrossRef] [PubMed]
- Schaible, U.E.; Collins, H.L.; Priem, F.; Kaufmann, S.H. Correction of the iron overload defect in beta-2-microglobulin knockout mice by lactoferrin abolishes their increased susceptibility to tuberculosis. J. Exp. Med. 2002, 196, 1507–1513. [Google Scholar] [CrossRef]
- Miles, A.A.; Khimji, P.L.; Maskell, J. The variable response of bacteria to excess ferric iron in host tissues. J. Med. Microbiol. 1979, 12, 17–28. [Google Scholar] [CrossRef]
- Punnonen, K.; Irjala, K.; Rajamaki, A. Iron-deficiency anemia is associated with high concentrations of transferrin receptor in serum. Clin. Chem. 1994, 40, 774–776. [Google Scholar]
- Das, B.S.; Devi, U.; Mohan Rao, C.; Srivastava, V.K.; Rath, P.K. Effect of iron supplementation on mild to moderate anaemia in pulmonary tuberculosis. Br. J. Nutr. 2003, 90, 541–550. [Google Scholar] [Green Version]
- Fleck, A.; Myers, M.A. Diagnostic and prognostic significance of acute phase proteins. In The Acute Phase Response to Injury and Infection; Gordon, A.H., Ed.; Elsevier Science Publishers: Amsterdam, The Netherlands, 1985; pp. 249–271. [Google Scholar]
- Gangaidzo, I.T.; Moyo, V.M.; Mvundura, E.; Aggrey, G.; Murphree, N.L.; Khumalo, H.; Saungweme, T.; Kasvosve, I.; Gomo, Z.A.; Rouault, T.; et al. Association of pulmonary tuberculosis with increased dietary iron. J. Infect. Dis. 2001, 184, 936–939. [Google Scholar] [CrossRef]
- Tanner, R.; O’Shea, M.K.; White, A.D.; Muller, J.; Harrington-Kandt, R.; Matsumiya, M.; Dennis, M.J.; Parizotto, E.A.; Harris, S.; Stylianou, E.; et al. The influence of haemoglobin and iron on in vitro mycobacterial growth inhibition assays. Sci. Rep. 2017, 7, 43478. [Google Scholar] [CrossRef]
- Iannotti, L.L.; Tielsch, J.M.; Black, M.M.; Black, R.E. Iron supplementation in early childhood: Health benefits and risks. Am. J. Clin. Nutr. 2006, 84, 1261–1276. [Google Scholar] [CrossRef] [PubMed]
- Adetifa, I.; Okomo, U. Iron supplementation for reducing morbidity and mortality in children with HIV. Cochrane Database Syst. Rev. 2009, 1, CD006736. [Google Scholar] [CrossRef] [PubMed]
- Minchella, P.A.; Donkor, S.; McDermid, J.M.; Sutherland, J.S. Iron homeostasis and progression to pulmonary tuberculosis disease among household contacts. Tuberculosis 2015, 95, 288–293. [Google Scholar] [CrossRef] [PubMed]
- Takenami, I.; Loureiro, C.; Machado, A.; Emodi, K., Jr.; Riley, L.W.; Arruda, S. Blood cells and interferon-gamma levels correlation in latent tuberculosis infection. ISRN Pulmonol. 2013, 2013, 256148. [Google Scholar] [CrossRef] [PubMed]
- Minchella, P.A.; Donkor, S.; Owolabi, O.; Sutherland, J.S.; McDermid, J.M. Complex anemia in tuberculosis: The need to consider causes and timing when designing interventions. Clin. Infect. Dis. 2015, 60, 764–772. [Google Scholar] [CrossRef]
- Baer, A.N.; Dessypris, E.N.; Krantz, S.B. The pathogenesis of anemia in rheumatoid arthritis: A clinical and laboratory analysis. Semin. Arthr. Rheum. 1990, 19, 209–223. [Google Scholar] [CrossRef]








© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kolloli, A.; Singh, P.; Rodriguez, G.M.; Subbian, S. Effect of Iron Supplementation on the Outcome of Non-Progressive Pulmonary Mycobacterium tuberculosis Infection. J. Clin. Med. 2019, 8, 1155. https://doi.org/10.3390/jcm8081155
Kolloli A, Singh P, Rodriguez GM, Subbian S. Effect of Iron Supplementation on the Outcome of Non-Progressive Pulmonary Mycobacterium tuberculosis Infection. Journal of Clinical Medicine. 2019; 8(8):1155. https://doi.org/10.3390/jcm8081155
Chicago/Turabian StyleKolloli, Afsal, Pooja Singh, G. Marcela Rodriguez, and Selvakumar Subbian. 2019. "Effect of Iron Supplementation on the Outcome of Non-Progressive Pulmonary Mycobacterium tuberculosis Infection" Journal of Clinical Medicine 8, no. 8: 1155. https://doi.org/10.3390/jcm8081155
APA StyleKolloli, A., Singh, P., Rodriguez, G. M., & Subbian, S. (2019). Effect of Iron Supplementation on the Outcome of Non-Progressive Pulmonary Mycobacterium tuberculosis Infection. Journal of Clinical Medicine, 8(8), 1155. https://doi.org/10.3390/jcm8081155






