Long Daytime Napping Is Associated with Increased Adiposity and Type 2 Diabetes in an Elderly Population with Metabolic Syndrome
Abstract
1. Introduction
2. Methods
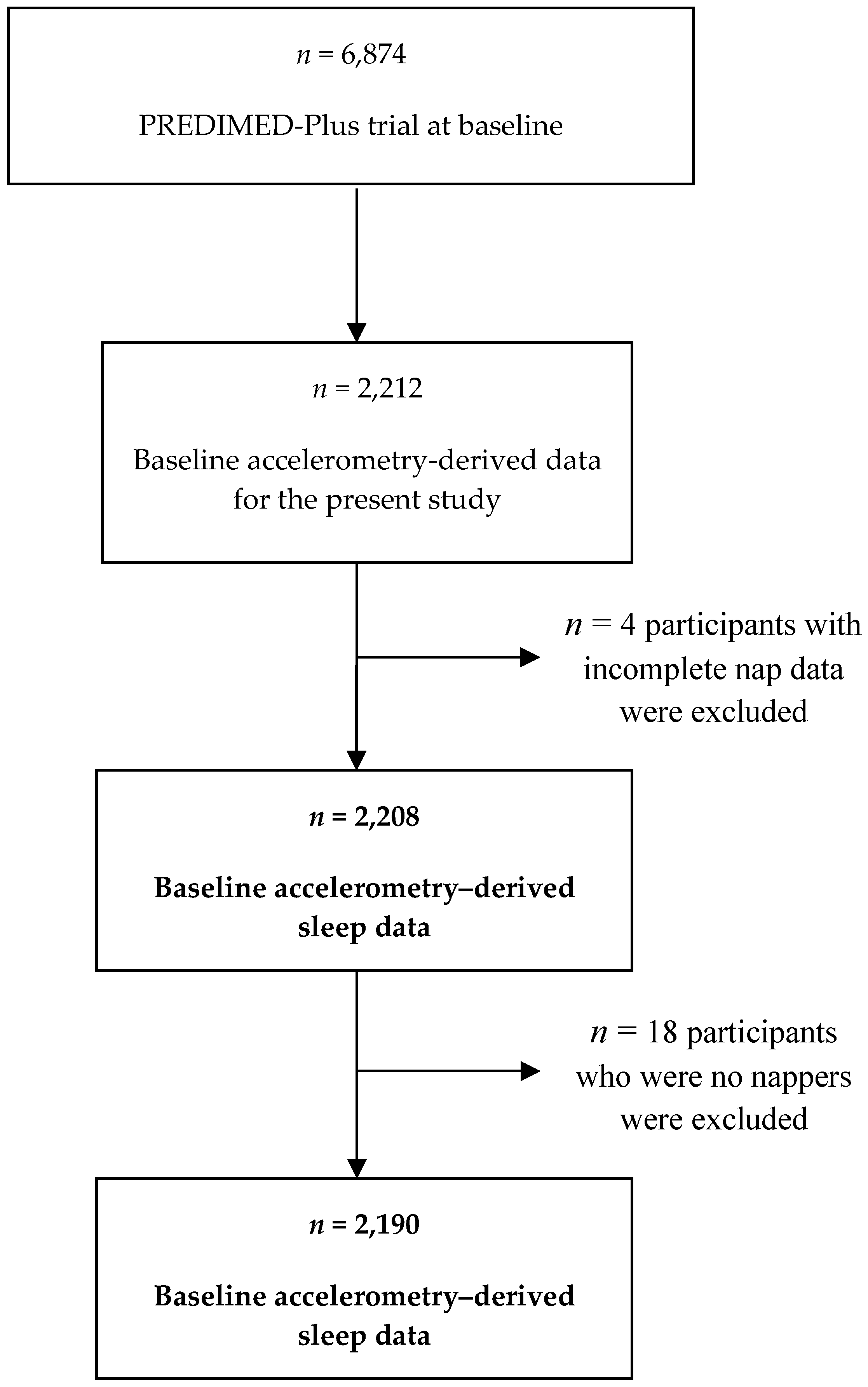
2.1. Study Design and Sample
2.2. Sleep Assessment by Accelerometry
2.3. T2D Prevalence
2.4. Adiposity Measures and Other Covariates
2.5. Statistical Analysis
3. Results
4. Discussion
5. Limitations
6. Conclusions
Author Contributions
Funding
Availability of Data and Materials
Acknowledgments
Conflicts of Interest
References
- Pressman, M.R.; Fry, J.M. What is normal sleep in the elderly? Clin. Geriatr. Med. 1988, 4, 71–82. [Google Scholar] [CrossRef]
- Yoon, I.Y.; Kripke, D.F.; Youngstedt, S.D.; Elliott, J.A. Actigraphy suggests age-related differences in napping and nocturnal sleep. J. Sleep Res. 2003, 12, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Picarsic, J.L.; Glynn, N.W.; Taylor, C.A.; Katula, J.A.; Goldman, S.E.; Studenski, S.A.; Newman, A.B. Self-reported napping and duration and quality of sleep in the lifestyle interventions and independence for elders pilot study. J. Am. Geriatr. Soc. 2008, 56, 1674–1680. [Google Scholar] [CrossRef] [PubMed]
- Dhand, R.; Sohal, H. Good sleep, bad sleep! The role of daytime naps in healthy adults. Curr. Opin. Pulm. Med. 2006, 12, 379–382. [Google Scholar] [CrossRef] [PubMed]
- Naska, A.; Oikonomou, E.; Trichopoulou, A.; Psaltopoulou, T.; Trichopoulos, D. Siesta in healthy adults and coronary mortality in the general population. Arch. Intern. Med. 2007, 167, 296–301. [Google Scholar] [CrossRef] [PubMed]
- Yamada, T.; Hara, K.; Shojima, N.; Yamauchi, T.; Kadowaki, T. Daytime Napping and the Risk of Cardiovascular Disease and All-Cause Mortality: A Prospective Study and Dose-Response Meta-Analysis. Sleep 2015, 38, 1945–1953. [Google Scholar] [CrossRef]
- Lin, D.; Sun, K.; Li, F.; Qi, Y.; Ren, M.; Huang, C.; Tang, J.; Xue, S.; Li, Y.; Yan, L. Association between habitual daytime napping and metabolic syndrome: A population-based study. Metabolism 2014, 63, 1520–1527. [Google Scholar] [CrossRef]
- Yamada, T.; Shojima, N.; Yamauchi, T.; Kadowaki, T. J-curve relation between daytime nap duration and type 2 diabetes or metabolic syndrome: A dose-response meta-analysis. Sci. Rep. 2016, 6, 38075. [Google Scholar] [CrossRef]
- Rana, J.S.; Li, T.Y.; Manson, J.E.; Hu, F.B. Adiposity compared with physical inactivity and risk of type 2 diabetes in women. Diabetes Care 2007, 30, 53–58. [Google Scholar] [CrossRef][Green Version]
- Sayón-Orea, C.; Bes-Rastrollo, M.; Carlos, S.; Beunza, J.J.; Basterra-Gortari, F.J.; Martínez-González, M.A. Association between sleeping hours and siesta and the risk of obesity: The SUN Mediterranean Cohort. Obes. Facts 2013, 6, 337–347. [Google Scholar] [CrossRef]
- Patel, S.R.; Hayes, A.L.; Blackwell, T.; Evans, D.S.; Ancoli-Israel, S.; Wing, Y.K.; Stone, K.L. Osteoporotic Fractures in Men (MrOS); Study of Osteoporotic Fractures (SOF) Research Groups. The association between sleep patterns and obesity in older adults. Int. J. Obes. 2014, 38, 1159–1164. [Google Scholar] [CrossRef]
- Loredo, J.S.; Weng, J.; Ramos, A.R.; Sotres-Alvarez, D.; Simonelli, G.; Talavera, G.A.; Patel, S.R. Sleep Patterns and Obesity: Hispanic Community Health Study/Study of Latinos Sueño Ancillar Study. Chest 2019. [Google Scholar] [CrossRef]
- Martínez-González, M.A.; Buil-Cosiales, P.; Corella, D.; Bulló, M.; Fitó, M.; Vioque, J.; Romaguera, D.; Martínez, J.A.; Wärnberg, J.; López-Miranda, J.; et al. Cohort Profile: Design and methods of the PREDIMED-Plus randomized trial. Int. J. Epidemiol. 2018. [Google Scholar] [CrossRef]
- Alberti, K.G.; Eckel, R.H.; Grundy, S.M.; Zimmet, P.Z.; Cleeman, J.I.; Donato, K.A.; Fruchart, J.C.; James, W.P.; Loria, C.M.; Smith, S.C., Jr. Harmonizing the metabolic syndrome: A joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation 2009, 120, 1640–1645. [Google Scholar]
- GGIR: Raw Accelerometer Data Analysis. Available online: https://cran.r-project.org/web/packages/GGIR/index.html (accessed on 1 April 2019).
- Van Hees, V.T.; Fang, Z.; Langford, J.; Assah, F.; Mohammad, A.; da Silva, I.C.; Trenell, M.I.; White, T.; Wareham, N.J.; Brage, S. Autocalibration of accelerometer data for free-living physical activity assessment using local gravity and temperature: An evaluation on four continents. J. Appl. Physiol. 2014, 117, 738–744. [Google Scholar] [CrossRef]
- Van Hees, V.T.; Sabia, S.; Anderson, K.N.; Denton, S.J.; Oliver, J.; Catt, M.; Abell, J.G.; Kivimäki, M.; Trenell, M.I.; Singh-Manoux, A. A Novel, Open Access Method to Assess Sleep Duration Using a Wrist-Worn Accelerometer. PLoS ONE 2015, 10, e0142533. [Google Scholar] [CrossRef]
- American Diabetes Association. Standards of medical care in diabetes-2014. Diabetes Care 2014, 37, S14–S80. [Google Scholar] [CrossRef]
- Menai, M.; van Hees, V.T.; Elbaz, A.; Kivimaki, M.; Singh-Manoux, A.; Sabia, S. Accelerometer assessed moderate-to-vigorous physical activity and successful ageing: Results from the Whitehall II study. Sci. Rep. 2017, 8, 45772. [Google Scholar] [CrossRef]
- World Health Organization. Global Recommendations on Physical Activity for Health. 2010. Available online: http://www.who.int/dietphysicalactivity/factsheet_olderadults/en/ (accessed on 3 April 2018).
- Barros, A.J.; Hirakata, V.N. Alternatives for logistic regression in cross-sectional studies: An empirical comparison of models that directly estimate the prevalence ratio. BMC Med. Res. Methodol. 2003, 3, 21. [Google Scholar] [CrossRef]
- Lee, J. Odds ratio or relative risk for cross-sectional data? Int. J. Epidemiol. 1994, 23, 201–203. [Google Scholar] [CrossRef]
- Smolensky, M.H.; Hermida, R.C.; Castriotta, R.J.; Portaluppi, F. Role of sleep-wake cycle on blood pressure circadian rhythms and hypertension. Sleep Med. 2007, 8, 668–680. [Google Scholar] [CrossRef]
- Rasch, B.; Dodt, C.; Mölle, M.; Born, J. Sleep-stage-specific regulation of plasma catecholamine concentration. Psychoneuroendocrinology 2007, 32, 884–891. [Google Scholar] [CrossRef]
- Devine, J.K.; Wolf, J.M. Determinants of cortisol awakening responses to naps and nighttime sleep. Psychoneuroendocrinology 2016, 63, 128–134. [Google Scholar] [CrossRef]
- Anagnostis, P.; Athyros, V.G.; Tziomalos, K.; Karagiannis, A.; Mikhailidis, D.P. Clinical review: The pathogenetic role of cortisol in the metabolic syndrome: A hypothesis. J. Clin. Endocrinol. Metab. 2009, 94, 2692–2701. [Google Scholar] [CrossRef]
- Jakubowski, K.P.; Boylan, J.M.; Cundiff, J.M.; Matthews, K.A. Poor sleep moderates the relationship between daytime napping and inflammation in Black and White men. Sleep Health 2017, 3, 328–335. [Google Scholar] [CrossRef]
- Leng, Y.; Ahmadi-Abhari, S.; Wainwright, N.W.; Cappuccio, F.P.; Surtees, P.G.; Luben, R.; Brayne, C.; Khaw, K.T. Daytime napping, sleep duration and serum C reactive protein: A population-based cohort study. BMJ Open 2014, 4, e006071. [Google Scholar] [CrossRef]
- Wang, X.; Bao, W.; Liu, J.; Ouyang, Y.Y.; Wang, D.; Rong, S.; Xiao, X.; Shan, Z.L.; Zhang, Y.; Yao, P.; et al. Inflammatory markers and risk of type 2 diabetes: A systematic review and meta-analysis. Diabetes Care 2013, 36, 166–175. [Google Scholar] [CrossRef]
- Goldman, S.E.; Hall, M.; Boudreau, R.; Matthews, K.A.; Cauley, J.A.; Ancoli-Israel, S.; Stone, K.L.; Rubin, S.M.; Satterfield, S.; Simonsick, E.M.; et al. Association between nighttime sleep and napping in older adults. Sleep 2008, 31, 733–740. [Google Scholar] [CrossRef]
- Jackson, S.E.; Kirschbaum, C.; Steptoe, A. Hair cortisol and adiposity in a population-based sample of 2527 men and women aged 54 to 87 years. Obesity 2017, 25, 539–544. [Google Scholar] [CrossRef]
- Fardet, L.; Fève, B. Systemic glucocorticoid therapy: A review of its metabolic and cardiovascular adverse events. Drugs 2014, 74, 1731–1745. [Google Scholar] [CrossRef]
- Engström, G.; Hedblad, B.; Stavenow, L.; Lind, P.; Janzon, L.; Lindgärde, F. Inflammation-sensitive plasma proteins are associated with future weight gain. Diabetes 2003, 52, 2097–2101. [Google Scholar] [CrossRef]
- Duncan, B.B.; Schmidt, M.I.; Chambless, L.E.; Folsom, A.R.; Carpenter, M.; Heiss, G. Fibrinogen, other putative markers of inflammation, and weight gain in middle-aged adults-the ARIC study. Atherosclerosis Risk in Communities. Obes. Res. 2000, 8, 279–286. [Google Scholar] [CrossRef]
| Categories of Daytime Napping (min) | |||||||
|---|---|---|---|---|---|---|---|
| Total n = 2190 | 5 to <30 min n = 344 | 30 to <60 min n = 692 | 60 to <90 min n = 562 | ≥90 min n = 592 | P-Value | ||
| Sleep parameters | |||||||
| Napping duration, median interquartile range, min | 62.4 (39.0; 92.4) | 20.4 (14.4; 25.2) | 44.4 (37.2;52.2) | 73.2 (66.6;79.8) | 115.8 (100.8; 139.2) | <0.001 | |
| Napping duration, min, max | 5.4, 275.4 | 5.4, 29.4 | 30.0, 59.4 | 60.0, 89.4 | 90.0, 275.4 | ||
| Sleep duration, mean ± SD, h | 6.2 ± 1.2 | 6.4 ± 1.0 | 6.3 ± 1.0 | 6.1 ± 1.2 | 5.9 ± 1.5 | <0.001 | |
| Age, mean ± SD, y | 65 ± 5 | 64 ± 5 | 65 ± 5 | 65 ± 5 | 65 ± 5 | 0.001 | |
| Male, n (%) | 1163(53) | 145(40) | 361(52) | 300(53) | 362(61) | <0.001 | |
| BMI, mean ± SD, kg/m2 | 32.6 ± 3.4 | 32.0 ± 3.3 | 32.6 ± 3.3 | 32.7 ± 3.5 | 32.9 ± 3.5 | 0.001 | |
| WC, mean ± SD, cm | 107.4 ± 9.5 | 104.8 ± 9.4 | 107.2 ± 9.4 | 107.4 ± 9.4 | 109.1 ± 9.6 | <0.001 | |
| Type 2 diabetes, n (%) | 720(33) | 89(24) | 216(31) | 191(34) | 228(38) | <0.001 | |
| Sleep apnea, n (%) | 283(13) | 47(13) | 85(12) | 74(13) | 79(13) | 0.944 | |
| Depression, n (%) | 482(22) | 70(19) | 140(20) | 125(22) | 152(25) | 0.049 | |
| Sedative treatment, n (%) | 530(24) | 90(25) | 153(22) | 138(24) | 156(26) | 0.365 | |
| Smoking, n (%) | |||||||
| Never | 940(43) | 195(54) | 317(46) | 230(41) | 207(35) | <0.001 | |
| Former | 989(45) | 141(39) | 309(45) | 270(48) | 277(47) | ||
| Current | 253(11) | 26(7) | 62(9) | 59(10) | 107(18) | ||
| Adherence to energy-restricted MedDiet (score from 0 to 17 item), mean ± SD | 8.5 ± 2.7 | 8.9 ± 2.7 | 8.4 ± 2.6 | 8.5 ± 2.9 | 8.6 ± 2.6 | 0.083 | |
| Compliance of MVPA recommendations a, n (%) | 755(34) | 130(36) | 252(36) | 211(37) | 169(28) | 0.005 | |
| Education status, n (%) | |||||||
| Primary education | 1071(49) | 174(48) | 341(49) | 265(47) | 303(52) | 0.088 | |
| Secondary education | 610(28) | 107(30) | 203(29) | 147(26) | 158(27) | ||
| Academic/graduate | 479(22) | 78(21) | 141(20) | 139(25) | 122(21) | ||
| Employment status, n (%) | |||||||
| Working | 439(20) | 93(26) | 127(21) | 115(21) | 86(15) | <0.001 | |
| Non-working | 535(24) | 102(28) | 197(28) | 121(21) | 135(23) | ||
| Retired | 1215(55) | 165(45) | 365(53) | 322(58) | 365(62) | ||
| Marital status, n (%) | |||||||
| Single/divorced | 323(14) | 62(17) | 83(12) | 84(15) | 94(16) | 0.716 | |
| Married | 1630(75) | 262(72) | 536(77) | 415(74) | 432(73) | ||
| Widower | 228(12) | 37(10) | 72(10) | 59(10) | 62(10) | ||
| Categories of Daytime Napping (min) | 10 min Increment in Daytime Napping (ln Transformed) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 5 to <30 min | 30 to <60 min | 60 to <90 min | ≥90 min | P-Value 2 vs. 1 | P-Value 3 vs. 1 | P-Value 4 vs. 1 | Continuous | P-Value | |
| n | 344 | 692 | 562 | 592 | 2190 | ||||
| T2D (%) n | (24) 89 | (31) 216 | (34) 191 | (38) 228 | |||||
| Model 1 | 1 (ref.) | 1.24 (0.96, 1.59) | 1.34 (1.04, 1.74) | 1.50 (1.17, 1.93) | 0.096 | 0.025 | 0.002 | 1.22 (1.09, 1.37) | 0.001 |
| Model 2 | 1 (ref.) | 1.25 (0.97, 1.61) | 1.29 (0.99, 1.68) | 1.40 (1.08, 1.82) | 0.086 | 0.054 | 0.011 | 1.17 (1.04, 1.32) | 0.008 |
| Model 3 | 1 (ref.) | 1.24 (0.96, 1.60) | 1.28 (0.98, 1.66) | 1.37 (1.06, 1.78) | 0.101 | 0.064 | 0.017 | 1.16 (1.03, 1.31) | 0.012 |
| Categories of Daytime Napping (min) | 10 min Increment in Daytime Napping (ln Transformed) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 5 to <30 min | 30 to <60 min | 60 to <90 min | ≥90 min | P-Value 2 vs. 1 | P-Value 3 vs. 1 | P-Value 4 vs. 1 | Continuous | P-Value | |
| BMI, kg/m2 | |||||||||
| n | 344 | 692 | 562 | 592 | 2190 | ||||
| Model 1 | 0 (ref.) | 0.70 (0.26, 1.15) | 0.82 (0.35, 1.28) | 1.15 (0.69, 1.61) | 0.002 | 0.001 | <0.001 | 0.50 (0.28, 0.71) | <0.001 |
| Model 2 | 0 (ref.) | 0.73 (0.29, 1.16) | 0.84 (0.38, 1.29) | 1.11 (0.65, 1.58) | 0.001 | <0.001 | <0.001 | 0.48 (0.27, 0.70) | <0.001 |
| WC, cm | |||||||||
| n | 344 | 692 | 562 | 592 | 2190 | ||||
| Model 1 | 0 (ref.) | 1.66 (0.50, 2.81) | 1.75 (0.55, 2.95) | 2.95 (1.75, 4.15) | 0.005 | 0.004 | <0.001 | 1.31 (0.75, 1.86) | <0.001 |
| Model 2 | 0 (ref.) | 1.52 (0.39, 2.66) | 1.51 (0.33, 2.70) | 2.43 (1.23, 3.64) | 0.009 | 0.012 | <0.001 | 1.04 (0.47, 1.60) | <0.001 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Papandreou, C.; Díaz-López, A.; Babio, N.; Martínez-González, M.A.; Bulló, M.; Corella, D.; Fitó, M.; Romaguera, D.; Vioque, J.; Alonso-Gómez, Á.M.; et al. Long Daytime Napping Is Associated with Increased Adiposity and Type 2 Diabetes in an Elderly Population with Metabolic Syndrome. J. Clin. Med. 2019, 8, 1053. https://doi.org/10.3390/jcm8071053
Papandreou C, Díaz-López A, Babio N, Martínez-González MA, Bulló M, Corella D, Fitó M, Romaguera D, Vioque J, Alonso-Gómez ÁM, et al. Long Daytime Napping Is Associated with Increased Adiposity and Type 2 Diabetes in an Elderly Population with Metabolic Syndrome. Journal of Clinical Medicine. 2019; 8(7):1053. https://doi.org/10.3390/jcm8071053
Chicago/Turabian StylePapandreou, Christopher, Andrés Díaz-López, Nancy Babio, Miguel A. Martínez-González, Mónica Bulló, Dolores Corella, Montse Fitó, Dora Romaguera, Jesús Vioque, Ángel M. Alonso-Gómez, and et al. 2019. "Long Daytime Napping Is Associated with Increased Adiposity and Type 2 Diabetes in an Elderly Population with Metabolic Syndrome" Journal of Clinical Medicine 8, no. 7: 1053. https://doi.org/10.3390/jcm8071053
APA StylePapandreou, C., Díaz-López, A., Babio, N., Martínez-González, M. A., Bulló, M., Corella, D., Fitó, M., Romaguera, D., Vioque, J., Alonso-Gómez, Á. M., Wärnberg, J., Martínez, A. J., Serra-Majem, L., Estruch, R., Fernández-García, J. C., Lapetra, J., Pintó, X., Tur, J. A., Garcia-Rios, A., ... Salas-Salvadó, J. (2019). Long Daytime Napping Is Associated with Increased Adiposity and Type 2 Diabetes in an Elderly Population with Metabolic Syndrome. Journal of Clinical Medicine, 8(7), 1053. https://doi.org/10.3390/jcm8071053





















