Motor Imagery and Action Observation of Specific Neck Therapeutic Exercises Induced Hypoalgesia in Patients with Chronic Neck Pain: A Randomized Single-Blind Placebo Trial
Abstract
1. Introduction
2. Methods
2.1. Study Design
2.2. Recruitment of Participants
2.3. Randomization
2.4. Blinding
2.5. Interventions
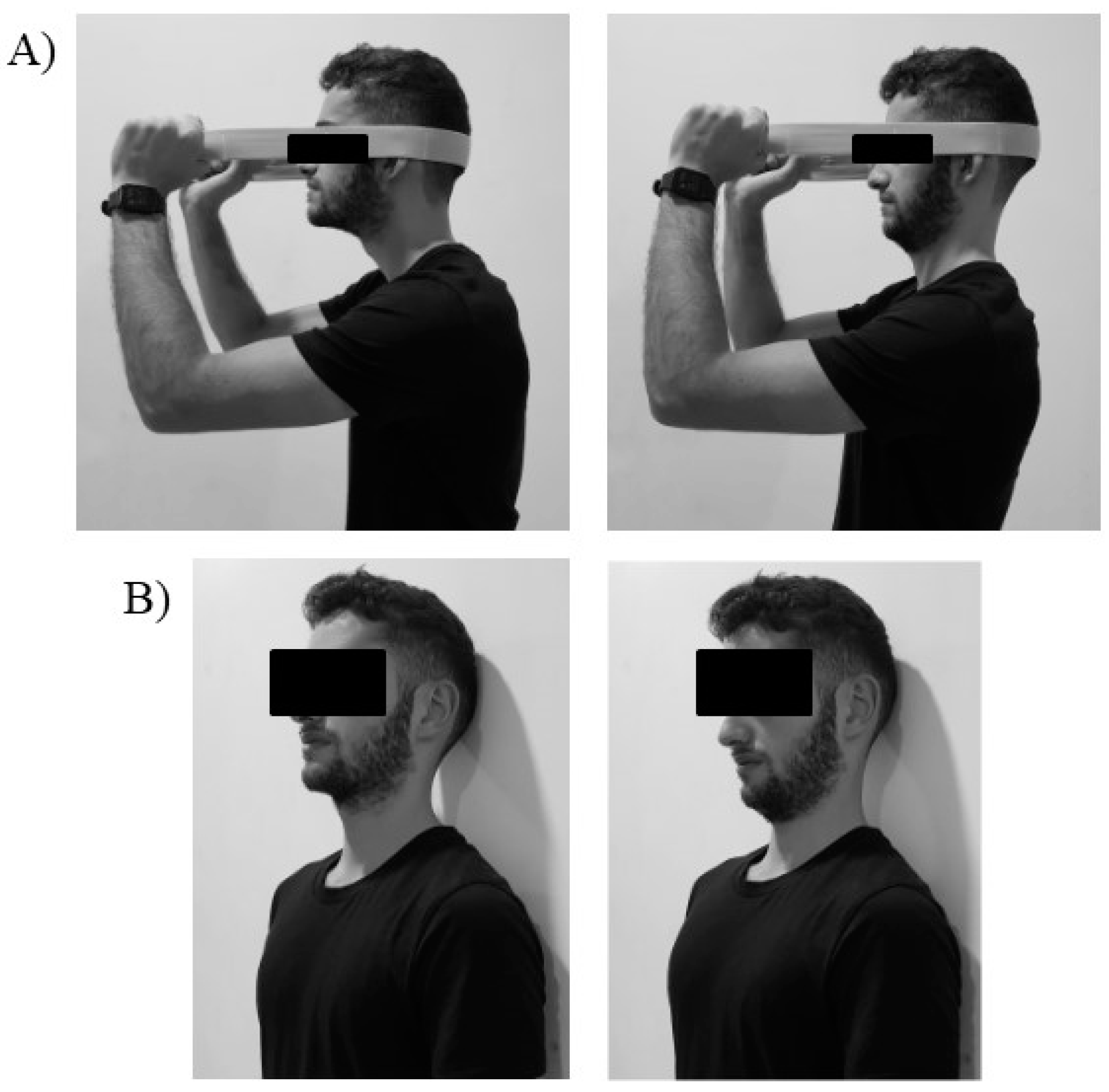
2.5.1. Action Observation Group
2.5.2. Motor Imagery Group
2.5.3. Placebo Observation Group
2.6. Outcomes
2.6.1. Primary Outcomes
Pressure Pain Thresholds
2.6.2. Secondary Outcomes
Heart Rate
Motor Imagery Ability
Mental Chronometry
Pain Catastrophizing
Neck Disability
Physical Activity Level
Visual Analogue Scale
2.7. Procedures
2.8. Statistical Analysis
3. Results
3.1. Primary Outcomes
3.1.1. Pressure Pain Threshold
C2/C3
Right Trapezius Muscle
Left Trapezius Muscle
Lateral Epicondyle
3.2. Secondary Outcomes
Heart Rate
4. Discussion
4.1. Clinical Implications
4.2. Limitations
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Vos, T.; Flaxman, A.D.; Naghavi, M.; Lozano, R.; Michaud, C.; Ezzati, M.; Shibuya, K.; Salomon, J.A.; Abdalla, S.; Aboyans, V.; et al. Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990–2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012, 380, 2163–2196. [Google Scholar] [CrossRef]
- Manchikanti, L.; Singh, V.; Datta, S.; Cohen, S.P.; Hirsch, J.A. Comprehensive review of epidemiology, scope, and impact of spinal pain. Pain Phys. 2009, 12, E35–E70. [Google Scholar]
- Falla, D.; Farina, D. Neuromuscular adaptation in experimental and clinical neck pain. J. Electromyogr. Kinesiol. 2008, 18, 255–261. [Google Scholar] [CrossRef] [PubMed]
- Falla, D.; Jull, G.; Hodges, P.W. Feedforward activity of the cervical flexor muscles during voluntary arm movements is delayed in chronic neck pain. Exp. Brain Res. 2004, 157, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Martin-Gomez, C.; Sestelo-Diaz, R.; Carrillo-Sanjuan, V.; Navarro-Santana, M.J.; Bardon-Romero, J.; Plaza-Manzano, G. Motor control using cranio-cervical flexion exercises versus other treatments for non-specific chronic neck pain: A systematic review and meta-analysis. Musculoskelet. Sci. Pract. 2019, 42, 52–59. [Google Scholar] [CrossRef] [PubMed]
- O’Leary, S.; Falla, D.; Hodges, P.W.; Jull, G.; Vicenzino, B. Specific Therapeutic Exercise of the Neck Induces Immediate Local Hypoalgesia. J. Pain 2007, 8, 832–839. [Google Scholar] [CrossRef] [PubMed]
- Koltyn, K.F. Analgesia following exercise: A review. Sports Med. 2000, 29, 85–98. [Google Scholar] [CrossRef] [PubMed]
- Koltyn, K.F.; Brellenthin, A.G.; Cook, D.B.; Sehgal, N.; Hillard, C. Mechanisms of Exercise-Induced Hypoalgesia. J. Pain 2014, 15, 1294–1304. [Google Scholar] [CrossRef]
- Jeannerod, M. Neural Simulation of Action: A Unifying Mechanism for Motor Cognition. NeuroImage 2001, 14, S103–S109. [Google Scholar] [CrossRef]
- Mulder, T. Motor imagery and action observation: Cognitive tools for rehabilitation. J. Neural Transm. 2007, 114, 1265–1278. [Google Scholar] [CrossRef]
- Decety, J. The neurophysiological basis of motor imagery. Behav. Brain Res. 1996, 77, 45–52. [Google Scholar] [CrossRef]
- Buccino, G. Action observation treatment: A novel tool in neurorehabilitation. Philos. Trans. R. Soc. B Boil. Sci. 2014, 369, 20130185. [Google Scholar] [CrossRef] [PubMed]
- Wright, D.J.; Williams, J.; Holmes, P.S. Combined action observation and imagery facilitates corticospinal excitability. Front. Hum. Neurosci. 2014, 8, 951. [Google Scholar] [CrossRef] [PubMed]
- Lotze, M.; Montoya, P.; Erb, M.; Hülsmann, E.; Flor, H.; Klose, U.; Birbaumer, N.; Grodd, W. Activation of Cortical and Cerebellar Motor Areas during Executed and Imagined Hand Movements: An fMRI Study. J. Cogn. Neurosci. 1999, 11, 491–501. [Google Scholar] [CrossRef] [PubMed]
- Decety, J. Do imagined and executed actions share the same neural substrate? Cogn. Brain Res. 1996, 3, 87–93. [Google Scholar] [CrossRef]
- Collet, C.; Di Rienzo, F.; El Hoyek, N.; Guillot, A.; Hoyek, N. Autonomic nervous system correlates in movement observation and motor imagery. Front. Hum. Neurosci. 2013, 7, 415. [Google Scholar] [CrossRef] [PubMed]
- Decety, J.; Jeannerod, M.; Durozard, D.; Baverel, G. Central activation of autonomic effectors during mental simulation of motor actions in man. J. Physiol. 1993, 461, 549–563. [Google Scholar] [CrossRef]
- Bolliet, O.; Collet, C.; Dittmar, A. Observation of action and autonomic nervous system responses 1. Percept. Mot. Skill 2005, 101, 195–202. [Google Scholar] [CrossRef]
- Maciver, K.; Lloyd, D.M.; Kelly, S.; Roberts, N.; Nurmikko, T. Phantom limb pain, cortical reorganization and the therapeutic effect of mental imagery. Brain 2008, 131, 2181–2191. [Google Scholar] [CrossRef]
- Hoyek, N.; Di Rienzo, F.; Collet, C.; Hoyek, F.; Guillot, A. The therapeutic role of motor imagery on the functional rehabilitation of a stage II shoulder impingement syndrome. Disabil. Rehabil. 2014, 36, 1113–1119. [Google Scholar] [CrossRef]
- Moseley, G.L.; Moseley, L. Graded motor imagery for pathologic pain: A randomized controlled trial. Neurology 2006, 67, 2129–2134. [Google Scholar] [CrossRef] [PubMed]
- Bang, D.-H.; Shin, W.-S.; Kim, S.-Y.; Choi, J.-D. The effects of action observational training on walking ability in chronic stroke patients: A double-blind randomized controlled trial. Clin. Rehabil. 2013, 27, 1118–1125. [Google Scholar] [CrossRef] [PubMed]
- Buccino, G.; Arisi, D.; Gough, P.; Aprile, D.; Ferri, C.; Serotti, L.; Tiberti, A.; Fazzi, E. Improving upper limb motor functions through action observation treatment: A pilot study in children with cerebral palsy. Dev. Med. Child Neurol. 2012, 54, 822–828. [Google Scholar] [CrossRef] [PubMed]
- Chesterton, L.S.; Sim, J.; Wright, C.C.; Foster, N.E. Interrater Reliability of Algometry in Measuring Pressure Pain Thresholds in Healthy Humans, Using Multiple Raters. Clin. J. Pain 2007, 23, 760–766. [Google Scholar] [CrossRef] [PubMed]
- Takala, E.P. Pressure pain threshold on upper trapezius and levator scapulae muscles. Repeatability and relation to subjective symptoms in a working population. Scand. J. Rehabil. Med. 1990, 22, 63–68. [Google Scholar] [PubMed]
- Campos, A.; González, M.Á. Spanish version of the revised movement image questionnaire (MIQ-R): Psychometric properties and validation. Rev. Psicol. Deporte 2010, 19, 265–275. [Google Scholar]
- Guillot, A.; Collet, C. Duration of Mentally Simulated Movement: A Review. J. Mot. Behav. 2005, 37, 10–20. [Google Scholar] [CrossRef]
- Malouin, F.; Richards, C.L.; Durand, A.; Doyon, J. Reliability of Mental Chronometry for Assessing Motor Imagery Ability After Stroke. Arch. Phys. Med. Rehabil. 2008, 89, 311–319. [Google Scholar] [CrossRef]
- Williams, S.E.; Guillot, A.; Di Rienzo, F.; Cumming, J. Comparing self-report and mental chronometry measures of motor imagery ability. Eur. J. Sport Sci. 2015, 15, 703–711. [Google Scholar] [CrossRef]
- Guillot, A.; Hoyek, N.; Louis, M.; Collet, C. Understanding the timing of motor imagery: Recent findings and future directions. Int. Rev. Sport Exerc. Psychol. 2012, 5, 3–22. [Google Scholar] [CrossRef]
- Gómez-Pérez, L.; López-Martínez, A.E.; Ruiz-Párraga, G.T. Psychometric Properties of the Spanish Version of the Tampa Scale for Kinesiophobia (TSK). J. Pain 2011, 12, 425–435. [Google Scholar] [CrossRef] [PubMed]
- Campayo, J.G.; Rodero, B.; Alda, M.; Sobradiel, N.; Montero, J.; Moreno, S. [Validation of the Spanish version of the Pain Catastrophizing Scale in fibromyalgia]. Med. Clín. 2008, 131, 487–492. [Google Scholar]
- Ortega, J.A.A.; Martínez, A.D.D.; Ruiz, R.A. Validación de una versión española del Índice de Discapacidad Cervical. Med. Clín. 2008, 130, 85–89. [Google Scholar] [CrossRef]
- Roman-Viñas, B.; Serra-Majem, L.; Hagströmer, M.; Ribas-Barba, L.; Sjöström, M.; Segura-Cardona, R. International Physical Activity Questionnaire: Reliability and validity in a Spanish population. Eur. J. Sport Sci. 2010, 10, 297–304. [Google Scholar] [CrossRef]
- Bijur, P.E.; Silver, W.; Gallagher, E.J. Reliability of the Visual Analog Scale for Measurement of Acute Pain. Acad. Emerg. Med. 2001, 8, 1153–1157. [Google Scholar] [CrossRef] [PubMed]
- Ostelo, R.W.; Deyo, R.A.; Stratford, P.; Waddell, G.; Croft, P.; Von Korff, M.; Bouter, L.M.; Henrica, C. Interpreting Change Scores for Pain and Functional Status in Low Back Pain. Spine 2008, 33, 90–94. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences; Lawrence Erlbaum Associates: Hillsdale, NJ, USA, 1988. [Google Scholar]
- Hardwick, R.M.; Caspers, S.; Eickhoff, S.B.; Swinnen, S.P. Neural correlates of action: Comparing meta-analyses of imagery, observation, and execution. Neurosci. Biobehav. Rev. 2018, 94, 31–44. [Google Scholar] [CrossRef]
- Hétu, S.; Grégoire, M.; Saimpont, A.; Coll, M.-P.; Eugène, F.; Michon, P.-E.; Jackson, P.L. The neural network of motor imagery: An ALE meta-analysis. Neurosci. Biobehav. Rev. 2013, 37, 930–949. [Google Scholar] [CrossRef]
- Beinert, K.; Sofsky, M.; Trojan, J. Train the brain! Immediate sensorimotor effects of mentally-performed flexor exercises in patients with neck pain. A pilot study. Eur. J. Phys. Rehabil. Med. 2019, 55, 63–70. [Google Scholar] [CrossRef]
- Beinert, K.; Preiss, S.; Huber, M.; Taube, W. Cervical joint position sense in neck pain. Immediate effects of muscle vibration versus mental training interventions: A RCT. Eur. J. Phys. Rehabil. Med. 2015, 51, 825–832. [Google Scholar]
- Beinert, K.; Mouthon, A.; Keller, M.; Mouthon, M.; Annoni, J.M.; Taube, W. Neural Correlates of Maladaptive Pain Behavior in Chronic Neck Pain—A Single Case Control fMRI Study. Pain Physician 2017, 20, E115–E125. [Google Scholar] [PubMed]
- Forkmann, K.; Wiech, K.; Ritter, C.; Sommer, T.; Rose, M.; Bingel, U. Pain-Specific Modulation of Hippocampal Activity and Functional Connectivity during Visual Encoding. J. Neurosci. 2013, 33, 2571–2581. [Google Scholar] [CrossRef] [PubMed]
- Meier, M.L.; Stämpfli, P.; Humphreys, B.K.; Vrana, A.; Seifritz, E.; Schweinhardt, P. The impact of pain-related fear on neural pathways of pain modulation in chronic low back pain. PAIN Rep. 2017, 2, 1. [Google Scholar] [CrossRef] [PubMed]
- De-La-Puente-Ranea, L.; García-Calvo, B.; La Touche, R.; Fernández-Carnero, J.; Gil-Martínez, A. Influence of the actions observed on cervical motion in patients with chronic neck pain: A pilot study. J. Exerc. Rehabil. 2016, 12, 346–354. [Google Scholar] [CrossRef]
- Hayashi, K.; Aono, S.; Shiro, Y.; Ushida, T. Effects of Virtual Reality-Based Exercise Imagery on Pain in Healthy Individuals. BioMed Res. Int. 2019, 2019, 5021914. [Google Scholar] [CrossRef] [PubMed]
- Peerdeman, K.; Van Laarhoven, A.; Bartels, D.; Peters, M.; Evers, A.; Laarhoven, A. Placebo-like analgesia via response imagery. Eur. J. Pain 2017, 21, 1366–1377. [Google Scholar] [CrossRef] [PubMed]
- Paungmali, A.; O’Leary, S.; Souvlis, T.; Vicenzino, B. Hypoalgesic and Sympathoexcitatory Effects of Mobilization with Movement for Lateral Epicondylalgia. Phys. Ther. 2003, 83, 374–383. [Google Scholar] [CrossRef] [PubMed]
- Vicenzino, B.; Collins, D.; Benson, H.; Wright, A. An investigation of the interrelationship between manipulative therapy-induced hypoalgesia and sympathoexcitation. J. Manip. Physiol. Ther. 1998, 21, 448–453. [Google Scholar]
- Schabrun, S.M.; Elgueta-Cancino, E.L.; Hodges, P.W. Smudging of the Motor Cortex is Related to the Severity of Low Back Pain. Spine 2017, 42, 1172–1178. [Google Scholar] [CrossRef] [PubMed]
- Le Pera, D.; Graven-Nielsen, T.; Valeriani, M.; Oliviero, A.; Di Lazzaro, V.; Tonali, P.A.; Arendt-Nielsen, L. Inhibition of motor system excitability at cortical and spinal level by tonic muscle pain. Clin. Neurophysiol. 2001, 112, 1633–1641. [Google Scholar] [CrossRef]
- Caspers, S.; Zilles, K.; Laird, A.R.; Eickhoff, S.B. ALE meta-analysis of action observation and imitation in the human brain. Neuroimage 2010, 50, 1148–1167. [Google Scholar] [CrossRef] [PubMed]
- Buccino, G.; Binkofski, F.; Fink, G.R.; Fadiga, L.; Fogassi, L.; Gallese, V.; Seitz, R.J.; Zilles, K.; Rizzolatti, G.; Freund, H.J. Action observation activates premotor and parietal areas in a somatotopic manner: An fMRI study. Eur. J. Neurosci. 2001, 13, 400–404. [Google Scholar] [CrossRef] [PubMed]
- Larsen, D.B.; Graven-Nielsen, T.; Boudreau, S.A. Pain-induced reduction in corticomotor excitability is counteracted by combined action-observation and motor imagery. J. Pain 2019. [Google Scholar] [CrossRef] [PubMed]
- Castillo Saavedra, L.; Mendonca, M.; Fregni, F. Role of the primary motor cortex in the maintenance and treatment of pain in fibromyalgia. Med. Hypotheses 2014, 83, 332–336. [Google Scholar] [CrossRef] [PubMed]
- Volz, M.S.; Suarez-Contreras, V.; Portilla, A.L.S.; Illigens, B.; Bermpohl, F.; Fregni, F. Movement observation-induced modulation of pain perception and motor cortex excitability. Clin. Neurophysiol. 2015, 126, 1204–1211. [Google Scholar] [CrossRef] [PubMed]
- Shimo, K.; Ueno, T.; Younger, J.; Nishihara, M.; Inoue, S.; Ikemoto, T.; Taniguchi, S.; Ushida, T. Visualization of Painful Experiences Believed to Trigger the Activation of Affective and Emotional Brain Regions in Subjects with Low Back Pain. PLoS ONE 2011, 6, e26681. [Google Scholar] [CrossRef] [PubMed]
- La Touche, R.; Pérez-González, A.; Suso-Martí, L.; Paris-Alemany, A.; Cuenca-Martínez, F. Observing neck movements evokes an excitatory response in the sympathetic nervous system associated with fear of movement in patients with chronic neck pain. Somatosens. Mot. Res. 2018, 35, 162–169. [Google Scholar] [CrossRef] [PubMed]
- Patterson, D.R.; Hoffman, H.G.; Palacios, A.G.; Jensen, M.J. Analgesic effects of posthypnotic suggestions and virtual reality distraction on thermal pain. J. Abnorm. Psychol. 2006, 115, 834–841. [Google Scholar] [CrossRef] [PubMed]
- Decety, J.; Jeannerod, M.; Germain, M.; Pastene, J. Vegetative response during imagined movement is proportional to mental effort. Behav. Brain Res. 1991, 42, 1–5. [Google Scholar] [CrossRef]
- Cuenca-Martínez, F.; Suso-Martí, L.; Grande-Alonso, M.; Paris-Alemany, A.; La Touche, R. Combining motor imagery with action observation training does not lead to a greater autonomic nervous system response than motor imagery alone during simple and functional movements: A randomized controlled trial. PeerJ 2018, 6, e5142. [Google Scholar] [CrossRef]
- Butler, R.K.; Finn, D.P. Stress-induced analgesia. Prog. Neurobiol. 2009, 88, 184–202. [Google Scholar] [CrossRef] [PubMed]
- Breckenridge, J.D.; Ginn, K.A.; Wallwork, S.B.; McAuley, J.H. Do People With Chronic Musculoskeletal Pain Have Impaired Motor Imagery? A Meta-analytical Systematic Review of the Left/Right Judgment Task. J. Pain 2019, 20, 119–132. [Google Scholar] [CrossRef] [PubMed]
| Measures | AO Group (n = 10) | MI Group (n = 10) | PO Group (n = 10) | p Value |
|---|---|---|---|---|
| Age | 33.5 ± 14.25 | 30.6 ± 11.53 | 27.70 ± 6.39 | 0.520 |
| Height (cm) | 171.9 ± 0.80 | 173.10 ± 0.70 | 174 ± 0.40 | 0.798 |
| Weight (kg) | 66.7 ± 7.97 | 68.70 ± 4.8 | 69.5 ± 8.26 | 0.672 |
| Pain intensity (VAS) | 68.9 ± 13.95 | 75 ± 7.73 | 70.8 ± 9.36 | 0.437 |
| Pain duration (month) | 27.9 ± 17.99 | 26.2 ± 12.45 | 17.4 ± 10.05 | 0.212 |
| Sex | 0.875 | |||
| Male | 5 (50) | 5 (50) | 4 (40) | |
| Female | 5 (50) | 5 (50) | 6 (60) | |
| Educational Level | 0.03 | |||
| Secondary education | 3 (30) | 5 (50) | 0 (00) | |
| College education | 7 (70) | 5 (50) | 10 (100) | |
| Marital Status | 0.136 | |||
| Single | 7 (70) | 3 (30) | 5 (50) | |
| Married | 3 (30) | 4 (40) | 4 (40) | |
| Divorced | 0 (0) | 3 (30) | 1 (0) | |
| Pain Location | 0.530 | |||
| Right | 5 (50) | 2 (20) | 2 (20) | |
| Left | 3 (30) | 5 (50) | 4 (40) | |
| Both | 2 (20) | 3 (30) | 4 (40) |
| Measures | AO Group (n = 10) | MI Group (n = 10) | PO Group (n = 10) | p Value |
|---|---|---|---|---|
| PCS | 31 ± 5.9 | 32.2 ± 6.71 | 33.1 ± 5.65 | 0.745 |
| TSK-11 | 32.3 ± 6 | 33 ± 4.85 | 31.3 ± 3.93 | 0.633 |
| NDI | 30.5 ± 3.62 | 29.8 ± 3.82 | 32.1 ± 4.48 | 0.430 |
| IPAQ | 1760.6 ± 483.51 | 1713.85 ± 500.3 | 1785.7 ± 659.17 | 0.958 |
| MIQ-R | 47.4 ± 4.77 | 47.3 ± 7.86 | 48 ± 4.52 | 0.960 |
| MC | 3.65 ± 3.96 | 4.39 ± 5.7 | 4.71 ± 4.52 | 0.879 |
| Measure | Group | Mean ± SD | Mean Difference (95% CI); Effect Size (d) (a) Pre–Post 1 (b) Pre–Post 2 (c) Post 1–Post 2 | ||
|---|---|---|---|---|---|
| Pre | Post-1 | Post-2 | |||
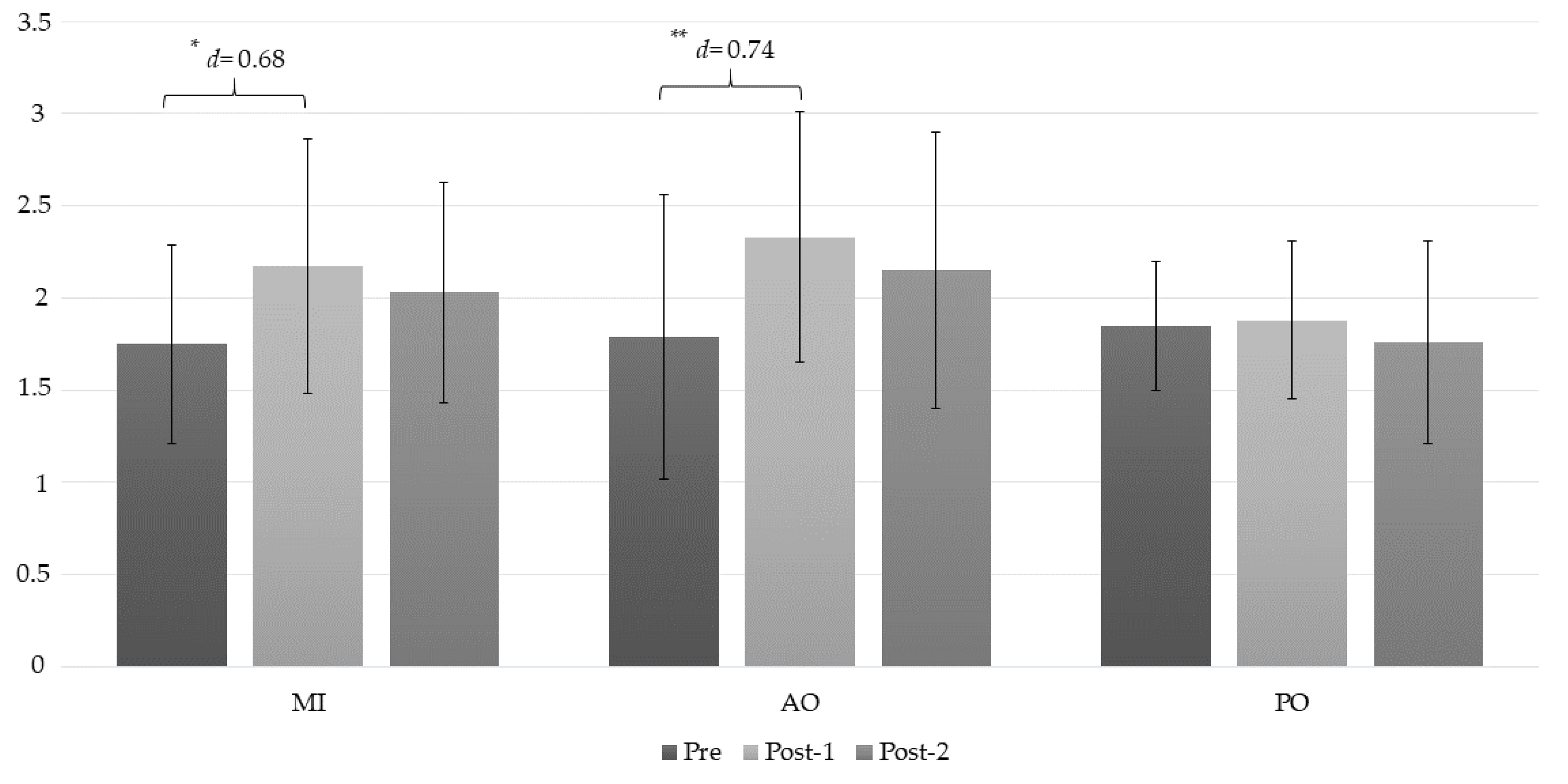
| PPT (C2/C3) | MI | 1.75 ± 0.54 | 2.17 ± 0.69 | 2.03 ± 0.60 | (a) −0.41 * (−0.71 to 0.12); d = 0.68 (b) −0.27 (−0.66 to 0.11); d = 0.49 (c) 0.14 (−0.83 to 0.37); d = 0.21 |
| AO | 1.79 ± 0.77 | 2.33 ± 0.68 | 2.15 ± 0.75 | (a) −0.54 ** (−0.84 to −0.25); d = 0.74 (b) −0.36 (−0.75 to 0.02); d = 0.47 (c) 0.18 (−0.05 to 0.40); d = 0.25 | |
| PO | 1.85 ± 0.35 | 1.88 ± 0.43 | 1.76 ± 0.55 | (a) −0.02 (−0.02 to 0.75); d = 0.07 (b) 0.09 (0.29 to 0.48); d = 0.19 (c) 0.11 (−0.11 to 0.33); d = 0.24 | |
| Mean difference (95% CI) Effect size (d) | |||||
| MI-AO | −0.03 (−0.69 to 0.63); d = 0.06 | −0.16 (−0.86 to 0.54); d = 0.23 | −0.13 (− 0.85 to 0.60); d = 0.17 | ||
| MI-PO | −0.10 (−0.76 to 0.56); d = 0.22 | 0.29 (−0.41 to 0.99); d = 0.5 | 0.27 (−0.46 to 0.99); d = 0.47 | ||
| AO-PO | −0.07 (−0.73 to 0.59); d = 0.1 | 0.46 (−0.24 to 1.11); d = 0.79 | −0.39 (−0.34 to 1.12); d = 0.59 | ||
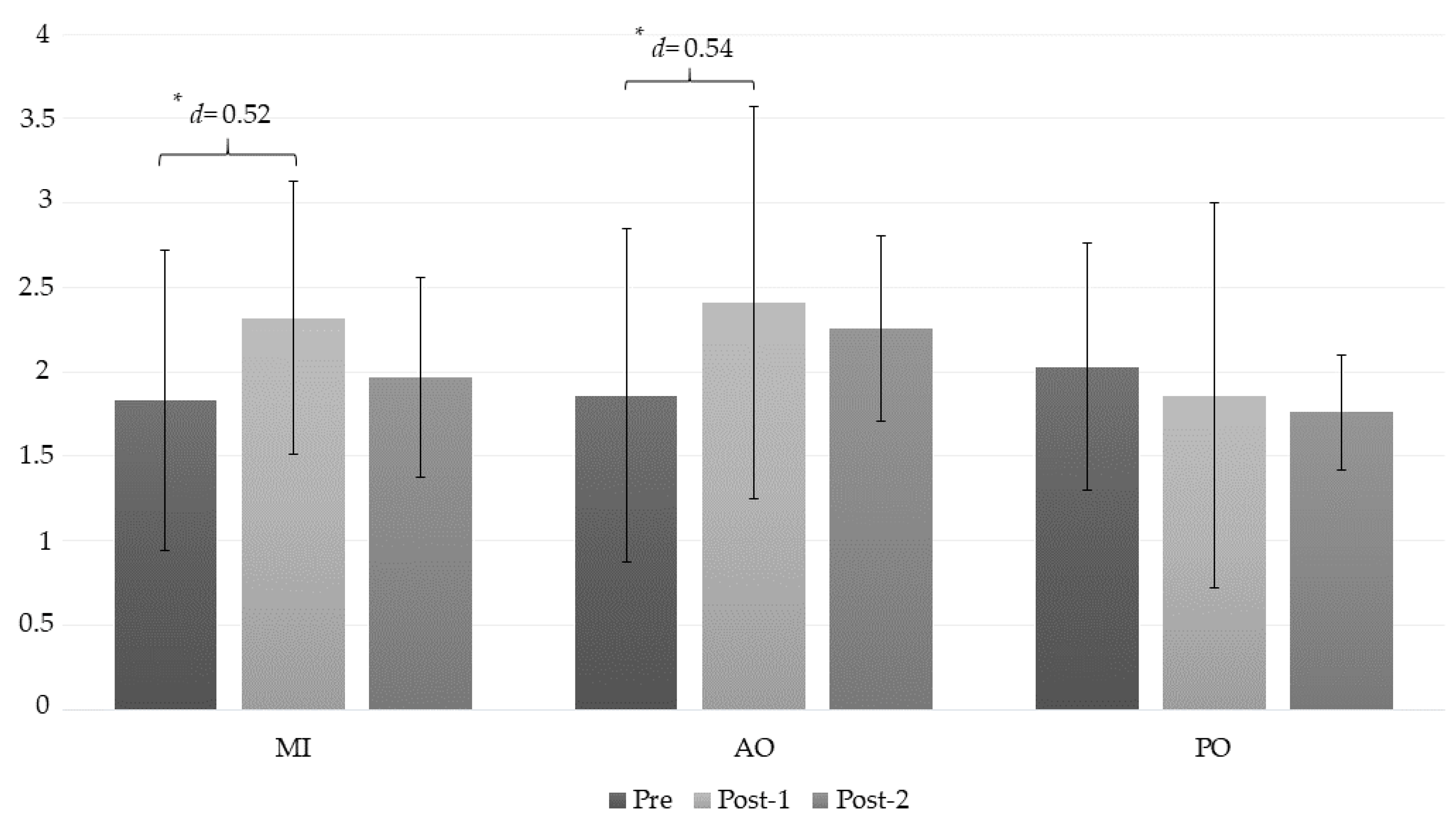
| PPT (RT) | MI | 1.83 ± 0.89 | 2.32 ± 0.99 | 1.97 ± 0.73 | (a) −0.49 * (−0.93 to −0.04); d = 0.52 (b) −0.14 (−0.60 to 0.32); d = 0.17 (c) 0.35 (−0.22 to 0.72); d = 0.40 |
| AO | 1.86 ± 0.81 | 2.41 ± 1.16 | 2.26 ± 0.1.14 | (a) −0.55 * (−0.99 to −0.11); d = 0.54 (b) −0.40 (−0.86 to 0.07); d = 0.40 (c) 0.16 (−0.21 to 0.52); d = 0.13 | |
| PO | 2.03 ± 0.59 | 1.86 ± 0.55 | 1.76 ± 0.34 | (a) 0.17 (−0.28 to 0.61); d = 0.29 (b) 0.27 (−0.19 to 0.74); d = 0.56 (c) 0.11 (−0.26 to 0.47); d = 0.21 | |
| Mean difference (95% CI) Effect size (d) | |||||
| MI-AO | −0.02 (−0.91 to 0.86); d = 0.03 | −0.09 (−1.15 to 0.98); d = 0.08 | 0.28 (−1.2 to 0.63); d = −0.40 | ||
| MI-PO | −0.20 (−1.08 to 0.68); d = 0.26 | 0.46 (−0.61 to 1.52); d = 0.57 | 0.21 (−0.70 to 1.13); d = 0.36 | ||
| AO-PO | −0.18 (−1.06 to 0.71); d = 0.23 | 0.54 (−0.52 to 1.61); d = 0.6 | 0.49 (−0.42 to 1.41); d = 0.59 | ||
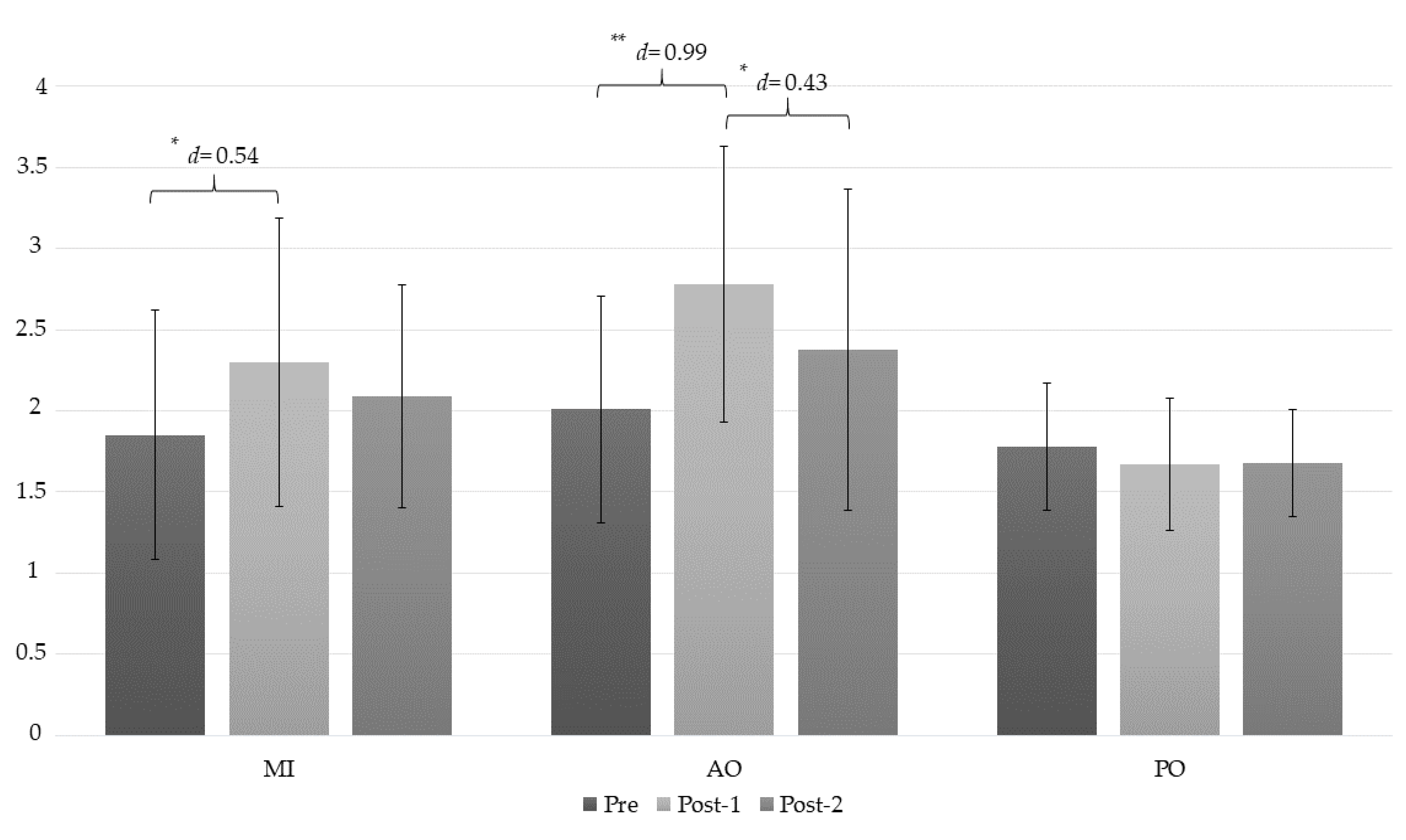
| PPT (LT) | MI | 1.85 ± 0.77 | 2.30 ± 0.89 | 2.09 ± 0.69 | (a) −0.46 * (−0.85 to −0.07); d = 0.54 (b) −0.24 (−0.66 to 0.17); d = 0.32 (c) 0.21 (−0.17 to 0.60); d = 0.26 |
| AO | 2.01 ± 0.70 | 2.78 ± 0.85 | 2.38 ± 0.99 | (a) −0.78 ** (−1.16 to −0.39); d = 0.99 (b) −0.37 (−0.79 to 0.04); d = 0.43 (c) −0.40 * (0.02 to 0.79); d = 0.43 | |
| PO | 1.78 ± 0.39 | 1.67 ± 0.41 | 1.68 ± 0.33 | (a) 0.10 (−0.28 to 0.49); d = 0.27 (b) 0.09 (−0.32 to 0.51); d = 0.27 (c) 0.01 (−0.39 to 0.38); d = 0.02 | |
| Mean difference (95% CI) Effect size (d) | |||||
| MI-AO | −0.16 (−0.90 to 0.57); d = 0.21 | −0.48 (−1.34 to 0.38); d = 0.55 | −0.29 (−1.12 to 0.53) d =0.33 | ||
| MI-PO | −0.06 (−0.67 to 0.80) d = 0.11 | 0.63 (−0.23 to 1.49), d = 0.90 | 0.40 (−0.42 to 1.22); d = 0.75 | ||
| AO-PO | 0.23 (0.51 to 0.96); d = 0.40 | 1.11 ** (0.25 to 1.96); d = 1.66 | 0.69 (−0.13 to 1.51); d = 0.94 | ||
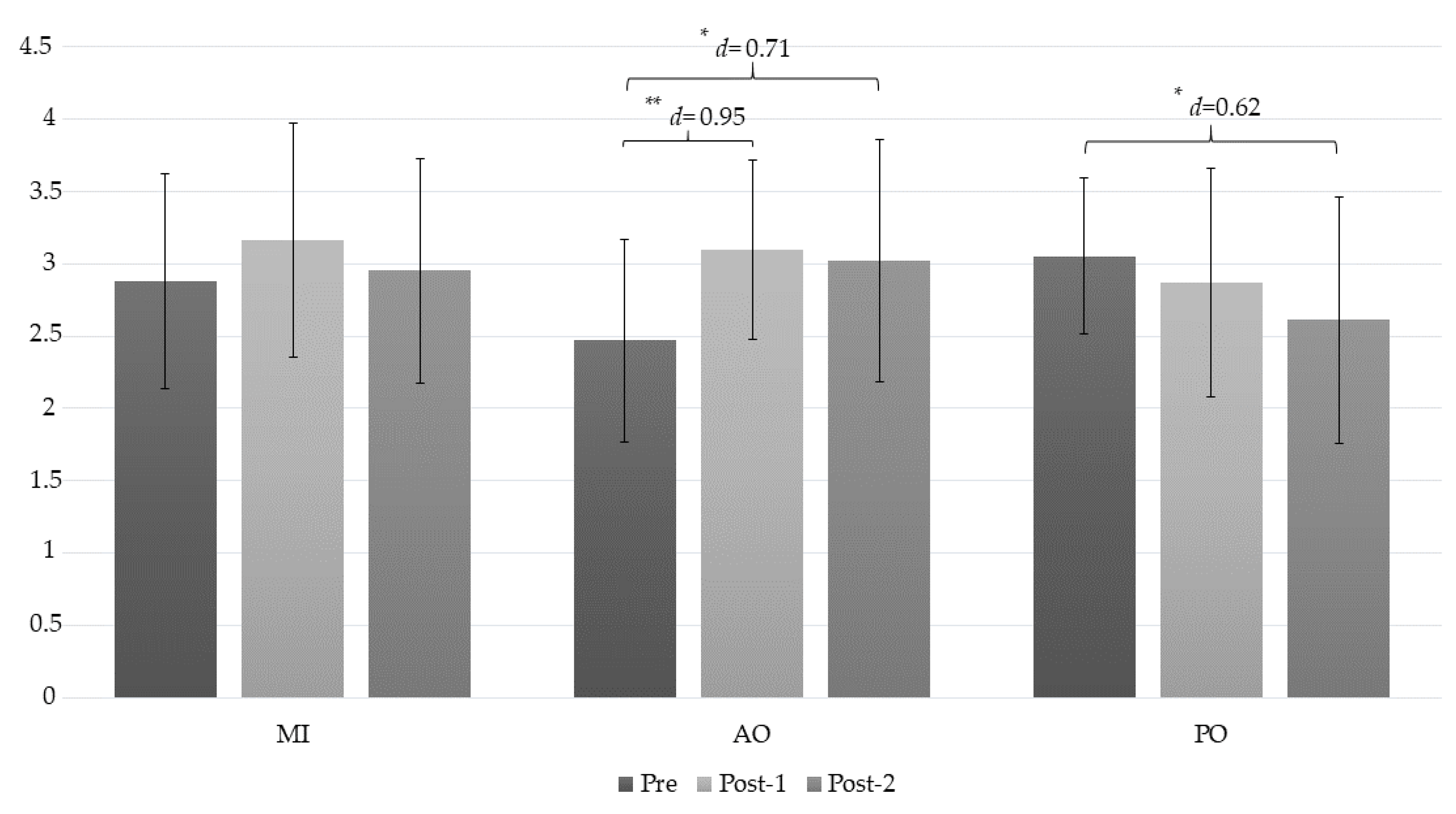
| PPT (Epicondyle) | MI | 2.88 ± 0.74 | 3.16 ± 0.81 | 2.95 ± 0.78 | (a) −0.29 (−0.60 to −0.01); d = 0.36 (b) −0.08 (−0.21 to 0.62); d = 0.09 (c) 0.21 (−0.21 to 0.62); d = 0.26 |
| AO | 2.47 ± 0.70 | 3.1 ± 0.62 | 3.02 ± 0.84 | (a) −0.64 ** (−0.95 to −0.33); d = 0.95 (b) −0.56 * (−0.96 to −0.15); d = 0.71 (c) 0.07 (−0.49 to 0.34); d = 0.11 | |
| PO | 3.05 ± 0.54 | 2.87 ± 0.79 | 2.61 ± 0.85 | (a) 0.18 (−0.13 to 0.49); d = 0.26 (b) 0.44 * (0.03 to 0.84); d = 0.62 (c) 0.25 (−0.67 to 0.16); d = 0.31 | |
| Mean difference (95% CI) Effect size (d) | |||||
| MI-AO | 0.41 (−0.35 to 1.16); d = 0.56 | −0.06 (−0.79 to 0.91); d = 0.08 | −0.07 (−1.01 to 0.87) d = 0.08 | ||
| MI-PO | −0.18 (−0.94 to 0.58) d = 0.26 | 0.30 (−0.55 to 1.15), d= 0.36 | 0.34 (−0.59 to 1.28); d = 0.41 | ||
| AO-PO | −0.58 (−1.34 to 0.18); d = 0.92 | 0.24 (−0.61 to 1.09); d = 0.32 | 0.41 (−0.53 to 1.35); d = 0.48 | ||
| Measure | Group | Mean ± SD | Mean Difference (95% CI); Effect Size (d). (a) Pre-Intervention (b) Pre–Post (c) Intervention–Post | ||
|---|---|---|---|---|---|
| Pre | Intervention | Post | |||
| HR | MI | 72.3 ± 5.38 | 74.84 ± 4.99 | 77.3 ± 6.4 | (a) −2.54 ** (−4.09 to −0.97) d = 0.48 (b) −5 ** (−7.15 to −2.85); d = 0.84 (c) −2.47 ** (−3.74 to −1.2) d = 0.42 |
| AO | 75.7 ± 6.77 | 80.08 ± 6.24 | 84.8 ± 7.19 | (a) −4.38 ** (−5.94 to −2.82) d = 0.67 (b) −9.1 ** (−11.24 to −6.95); d = 1.3 (c) −4.72 ** (−5.99 to −3.45) d = −0.7 | |
| PO | 71.6 ± 5.42 | 72.12 ± 5.05 | 72.6 ± 5.4 | (a) −0.52 (−2.08 to 1.04) d = −0.09 (b) −1 (−0.73 to 2.73); d = −0.18 (c) −0.48 (−1.75 to 0.79); d = −0.09 | |
| Mean difference (95% CI) Effect size (d) | |||||
| MI-AO | −3.4 (−10.13 to 3.33); d = −0.55 | −5.24 (−11.48 to 0.99); d = −0.92 | −7.5 * (−14.77 to −0.23); d = 1.10 | ||
| MI-PO | −0.7 (−6.02 to 7.43); d = −0.12 | 2.72 (−3.52 to 8.95); d = −0.54 | 4.7 (−2.57 to 11.97); d = −0.79 | ||
| AO-PO | −4.1 (−2.63 to 10.82); d = −0.66 | 7.96 * (1.73 to 14.19); d = 1.4 | 12.2 ** (−19.47 to −4.93); d = 1.92 | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Suso-Martí, L.; León-Hernández, J.V.; La Touche, R.; Paris-Alemany, A.; Cuenca-Martínez, F. Motor Imagery and Action Observation of Specific Neck Therapeutic Exercises Induced Hypoalgesia in Patients with Chronic Neck Pain: A Randomized Single-Blind Placebo Trial. J. Clin. Med. 2019, 8, 1019. https://doi.org/10.3390/jcm8071019
Suso-Martí L, León-Hernández JV, La Touche R, Paris-Alemany A, Cuenca-Martínez F. Motor Imagery and Action Observation of Specific Neck Therapeutic Exercises Induced Hypoalgesia in Patients with Chronic Neck Pain: A Randomized Single-Blind Placebo Trial. Journal of Clinical Medicine. 2019; 8(7):1019. https://doi.org/10.3390/jcm8071019
Chicago/Turabian StyleSuso-Martí, Luis, Jose Vicente León-Hernández, Roy La Touche, Alba Paris-Alemany, and Ferran Cuenca-Martínez. 2019. "Motor Imagery and Action Observation of Specific Neck Therapeutic Exercises Induced Hypoalgesia in Patients with Chronic Neck Pain: A Randomized Single-Blind Placebo Trial" Journal of Clinical Medicine 8, no. 7: 1019. https://doi.org/10.3390/jcm8071019
APA StyleSuso-Martí, L., León-Hernández, J. V., La Touche, R., Paris-Alemany, A., & Cuenca-Martínez, F. (2019). Motor Imagery and Action Observation of Specific Neck Therapeutic Exercises Induced Hypoalgesia in Patients with Chronic Neck Pain: A Randomized Single-Blind Placebo Trial. Journal of Clinical Medicine, 8(7), 1019. https://doi.org/10.3390/jcm8071019









