Cardiac Autonomic Derangement Is Associated with Worse Neurological Outcome in the Very Early Phases of Ischemic Stroke
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Protocol
2.2. Data Analysis
2.2.1. Spectral Analysis
2.2.2. Symbolic Analysis
2.3. Statistical Analysis
3. Results
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Krishnamurthi, R.V.; Feigin, V.L.; Forouzanfar, M.H.; Mensah, G.A.; Connor, M.; Bennett, D.A.; Moran, A.E.; Sacco, R.L.; Anderson, L.M.; Truelsen, T.; et al. Global and regional burden of first–ever ischaemic and haemorrhagic stroke during 1990–2010: findings from the Global Burden of Disease Study 2010. Lancet Glob. Heal. 2013, e259–e281. [Google Scholar] [CrossRef]
- Adams, H.P.; Davis, P.H.; Leira, E.C.; Chang, K.C.; Bendixen, B.H.; Clarke, W.R.; Woolson, R.F.; Hansen, M.D. Baseline NIH Stroke Scale score strongly predicts outcome after stroke: A report of the Trial of Org 10172 in Acute Stroke Treatment (TOAST). Neurology. 1999, 53, 126–131. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Venkat, P.; Seyfried, D.; Chopp, M.; Yan, T.; Chen, J. Brain–Heart Interaction: Cardiac Complications After Stroke. Circ. Res. 2017, 121, 451–468. [Google Scholar] [CrossRef] [PubMed]
- Bieber, M.; Werner, R.A.; Tanai, E.; Hofmann, U.; Higuchi, T.; Schuh, K.; Heuschmann, P.U.; Frantz, S.; Ritter, O.; Kraft, P.; et al. Stroke–induced chronic systolic dysfunction driven by sympathetic overactivity. Ann. Neurol. 2017, 82, 729–743. [Google Scholar] [CrossRef]
- Laowattana, S.; Zeger, S.L.; Lima, J.A.C.; Goodman, S.N.; Wittstein, I.S.; Oppenheimer, S.M. Left insular stroke is associated with adverse cardiac outcome. Neurology 2006, 66, 477–483, discussion 463. [Google Scholar] [CrossRef] [PubMed]
- Micieli, G.; Cavallini, A. The autonomic nervous system and ischemic stroke: a reciprocal interdependence. Clin. Auton. Res. 2008, 18, 308–317. [Google Scholar] [CrossRef] [PubMed]
- Go, A.S.; Mozaffarian, D.; Roger, V.L.; Benjamin, E.J.; Berry, J.D.; Blaha, M.J.; Dai, S.; Ford, E.S.; Fox, C.S.; Franco, S.; et al. Heart Disease and Stroke Statistics—2014 Update. Circulation. 2014, 129, e28–e292. [Google Scholar] [CrossRef] [PubMed]
- Powers, W.J.; Derdeyn, C.P.; Biller, J.; Coffey, C.S.; Hoh, B.L.; Jauch, E.C.; Johnston, K.C.; Johnston, S.C.; Khalessi, A.A.; Kidwell, C.S.; et al. 2015 American Heart Association/American Stroke Association Focused Update of the 2013 Guidelines for the Early Management of Patients with Acute Ischemic Stroke Regarding Endovascular Treatment. Stroke 2015, 46, 3020–3035. [Google Scholar] [CrossRef]
- Montano, N.; Porta, A.; Cogliati, C.; Costantino, G.; Tobaldini, E.; Casali, K.R.; Iellamo, F. Heart rate variability explored in the frequency domain: A tool to investigate the link between heart and behavior. Neurosci. Biobehav. Rev. 2009, 33, 71–80. [Google Scholar] [CrossRef]
- Barron, S.A.; Rogovski, Z.; Hemli, J. Autonomic consequences of cerebral hemisphere infarction. Stroke 1994, 25, 113–116. [Google Scholar] [CrossRef]
- Tokgozoglu, S.L.; Batur, M.K.; Topcuoglu, M.A.; Saribas, O.; Kes, S.; Oto, A. Effects of Stroke Localization on Cardiac Autonomic Balance and Sudden Death. Stroke 1999, 30, 1307–1311. [Google Scholar] [CrossRef] [PubMed]
- Chidambaram, H.; Gnanamoorthy, K.; Suthakaran, P.K.; Rajendran, K.; Pavadai, C. Assessment of Autonomic Dysfunction in Acute Stroke Patients at a Tertiary Care Hospital. J. Clin. Diagn. Res. 2017, 11, OC28–OC31. [Google Scholar] [CrossRef] [PubMed]
- Colivicchi, F.; Bassi, A.; Santini, M.; Caltagirone, C. Prognostic implications of right–sided insular damage, cardiac autonomic derangement, and arrhythmias after acute ischemic stroke. Stroke 2005, 36, 1710–1715. [Google Scholar] [CrossRef] [PubMed]
- Bassi, A.; Colivicchi, F.; Santini, M.; Caltagirone, C. Cardiac autonomic dysfunction and functional outcome after ischaemic stroke. Eur. J. Neurol. 2007, 14, 917–922. [Google Scholar] [CrossRef] [PubMed]
- Arad, M.; Abboud, S.; Radai, M.M.; Adunsky, A. Heart rate variability parameters correlate with functional independence measures in ischemic stroke patients. J. Electrocardiol. 2002, 35, 243–246. [Google Scholar] [CrossRef] [PubMed]
- Nayani, S.; Sreedharan, S.E.; Namboodiri, N.; Sarma, P.S.; Sylaja, P.N. Autonomic dysfunction in first ever ischemic stroke: Prevalence, predictors and short term neurovascular outcome. Clin. Neurol. Neurosurg. 2016, 150, 54–58. [Google Scholar] [CrossRef]
- Hachinski, V.C.; Oppenheimer, S.M.; Wilson, J.X.; Guiraudon, C.; Cechetto, D.F. Asymmetry of Sympathetic Consequences of Experimental Stroke. Arch. Neurol. 1992, 49, 697–702. [Google Scholar] [CrossRef]
- Colivicchi, F.; Bassi, A.; Santini, M.; Caltagirone, C. Cardiac autonomic derangement and arrhythmias in right–sided stroke with insular involvement. Stroke 2004, 35, 2094–2098. [Google Scholar] [CrossRef]
- Constantinescu, V.; Matei, D.; Cuciureanu, D.; Corciova, C.; Ignat, B.; Popescu, C.D. Cortical modulation of cardiac autonomic activity in ischemic stroke patients. Acta Neurol. Belg. 2016, 116, 473–480. [Google Scholar] [CrossRef]
- Cechetto, D.F. Experimental cerebral ischemic lesions and autonomic and cardiac effects in cats and rats. Stroke 1993, 24, I6-9. [Google Scholar]
- Schlegel, D.; Kolb, S.J.; Luciano, J.M.; Tovar, J.M.; Cucchiara, B.L.; Liebeskind, D.S.; Kasner, S.E. Utility of the NIH stroke scale as a predictor of hospital disposition. Stroke 2003, 34, 134–137. [Google Scholar] [CrossRef] [PubMed]
- Bamford, J.M.; Sandercock, P.A.; Warlow, C.P.; Slattery, J. Interobserver agreement for the assessment of handicap in stroke patients. Stroke 1989, 20, 828. [Google Scholar] [CrossRef]
- Broderick, J.P.; Adeoye, O.; Elm, J. Comments and Opinions Evolution of the Modified Rankin Scale and Its Use in Future Stroke Trials. Stroke 2017, 48, 2007–2012. [Google Scholar] [CrossRef] [PubMed]
- Powers, W.J.; Rabinstein, A.A.; Ackerson, T.; Adeoye, O.M.; Bambakidis, N.C.; Becker, K.; Biller, J.; Brown, M.; Demaerschalk, B.M.; Hoh, B.; et al. 2018 Guidelines for Management of Acute Ischemic Stroke e47. Stroke 2018. [Google Scholar] [CrossRef]
- SPREAD–Stroke Prevention and Educational Awareness Diffusion Ictus cerebrale: Linee guida italiane di prevenzione e trattamento. 2016:296. Available online: http://www.iso-spread.it/capitoli/LINEE_GUIDA_SPREAD_8a_EDIZIONE.pdf (accessed on 25 February 2019).
- Jauch, E.C.; Saver, J.L.; Adams, H.P.; Bruno, A.; Connors, J.J.; Demaerschalk, B.M.; Khatri, P.; McMullan, P.W., Jr.; Qureshi, A.I.; Rosenfield, K.; et al. Guidelines for the early management of patients with acute ischemic stroke: A guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2013, 44, 870–947. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, N.; Steiner, T.; Caso, V.; Wahlgren, N. Recommendations from the ESO–Karolinska Stroke Update Conference, Stockholm 13–15 November 2016. Eur. Stroke J 2017, 2, 95–102. [Google Scholar] [CrossRef] [PubMed]
- European Stroke Organisation (ESO) Executive Committee; ESO Writing Committee. Guidelines for Management of Ischaemic Stroke and Transient Ischaemic Attack 2008. Cerebrovasc. Dis. 2008, 25, 457–507. [Google Scholar] [CrossRef] [PubMed]
- Porta, A.; Tobaldini, E.; Guzzetti, S.; Furlan, R.; Montano, N.; Gnecchi–Ruscone, T. Assessment of cardiac autonomic modulation during graded head–up tilt by symbolic analysis of heart rate variability. Am. J. Physiol. Circ. Physiol. 2007, 293, 702–708. [Google Scholar] [CrossRef] [PubMed]
- Tobaldini, E.; Toschi–Dias, E.; Trimarchi, P.D.; Brena, N.; Comanducci, A.; Casarotto, S.; Montano, N.; Devalle, G. Cardiac autonomic responses to nociceptive stimuli in patients with chronic disorders of consciousness. Clin. Neurophysiol. 2018, 129, 1083–1089. [Google Scholar] [CrossRef]
- Tobaldini, E.; Covassin, N.; Calvin, A.; Singh, P.; Bukartyk, J.; Wang, S.; Montano, N.; Somers, V.K. Cardiac autonomic control and complexity during sleep are preserved after chronic sleep restriction in healthy subjects. Physiol. Rep. 2017, 5, e13197. [Google Scholar] [CrossRef]
- Sörös, P.; Hachinski, V. Cardiovascular and neurological causes of sudden death after ischaemic stroke. Lancet Neurol. 2012, 11, 179–188. [Google Scholar] [CrossRef]
- Graff, B.; Gąsecki, D.; Rojek, A.; Boutouyrie, P.; Nyka, W.; Laurent, S.; Narkiewicz, K. Heart rate variability and functional outcome in ischemic stroke: a multiparameter approach. J. Hypertens. 2013, 31, 1629–1636. [Google Scholar] [CrossRef] [PubMed]
- Aliberti, S.; Tobaldini, E.; Giuliani, F.; Nunziata, V.; Casazza, G.; Suigo, G.; D’Adda, A.; Bonaiti, G.; Roveda, A.; Queiroz, A.; et al. Cardiovascular autonomic alterations in hospitalized patients with community–acquired pneumonia. Respir. Res. 2016, 17, 98. [Google Scholar] [CrossRef] [PubMed]
- Naver, H.K.; Blomstrand, C.; Wallin, B.G. Reduced heart rate variability after right–sided stroke. Stroke 1996, 27, 247–251. [Google Scholar] [CrossRef] [PubMed]
- Algra, A.; Gates, P.C.; Fox, A.J.; Hachinski, V.; Barnett, H.J. Side of brain infarction and long–term risk of sudden death in patients with symptomatic carotid disease. Stroke 2003, 34, 2871–2875. [Google Scholar] [CrossRef]
- Christensen, H.; Boysen, G.; Christensen, A.F.; Johannesen, H.H. Insular lesions, ECG abnormalities, and outcome in acute stroke. J. Neurol. Neurosurg. Psychiatry. 2005, 76, 269–271. [Google Scholar] [CrossRef] [PubMed]
- Tobaldini, E.; Proserpio, P.; Sambusida, K.; Lanza, A.; Redaelli, T.; Frigerio, P.; Fratticci, L.; Rosa, S.; Casali, K.R.; Somers, V.K.; et al. Preserved cardiac autonomic dynamics during sleep in subjects with spinal cord injuries. Sleep Med. 2015, 16, 779–784. [Google Scholar] [CrossRef]
| Demographic and Clinical Data | Male (28) | Female (13) | All (41) |
|---|---|---|---|
| Age (years) | 67.8 ± 12.6 | 68.4 ± 13.2 | 68.0 ± 12.8 |
| Active smoking | 10 (35.7) | 2 (15.4) | 12 (29.3) |
| Hypercholesterolemia | 14 (50) | 7 (53.8) | 21 (51.2) |
| History of transient ischemic attack or stroke | 8 (28.6) | 7 (53.8) | 15 (36.6) |
| Hypertension | 19 (67.9) | 9 (69.2) | 28 (68.3) |
| Diabetes mellitus | 8 (28.6) | 1 (7.7) | 9 (22) |
| History of atrial fibrillation or flutter | 1 (3.6) | 2 (15.4) | 3 (7.3) |
| Heart failure | 2 (7.1) | 1 (7.7) | 3 (7.3) |
| Beta-blockers medication | 9 (32.1) | 3 (23.1) | 12 (29.3) |
| Systolic blood pressure | 156.8 ± 25.7 | 145.4 ± 23 | 153.4 ± 25.2 |
| Diastolic blood pressure | 89.8 ± 16.5 | 81.2 ± 19.8 | 87.3 ± 17.7 |
| Autonomic Parameters | NIHSS <14 (29) | NIHSS ≥14 (12) | p |
|---|---|---|---|
| HR (bpm) | 72.5 (61.7–83.7) | 64.4 (59.5–71.9) | ns |
| Total Power (ms2) | 612.8 (301.6–1334.6) | 804.3 (308.8–1045.6) | ns |
| LF (n.u.) | 56.7 (38.4–83.2) | 49.6 (29.4–81.8) | ns |
| HF (n.u.) | 25 (10.1–49.2) | 27 (15.6–45.2) | ns |
| LF/HF | 1.9 (0.7–7.6) | 2.1 (0.8–4.7) | ns |
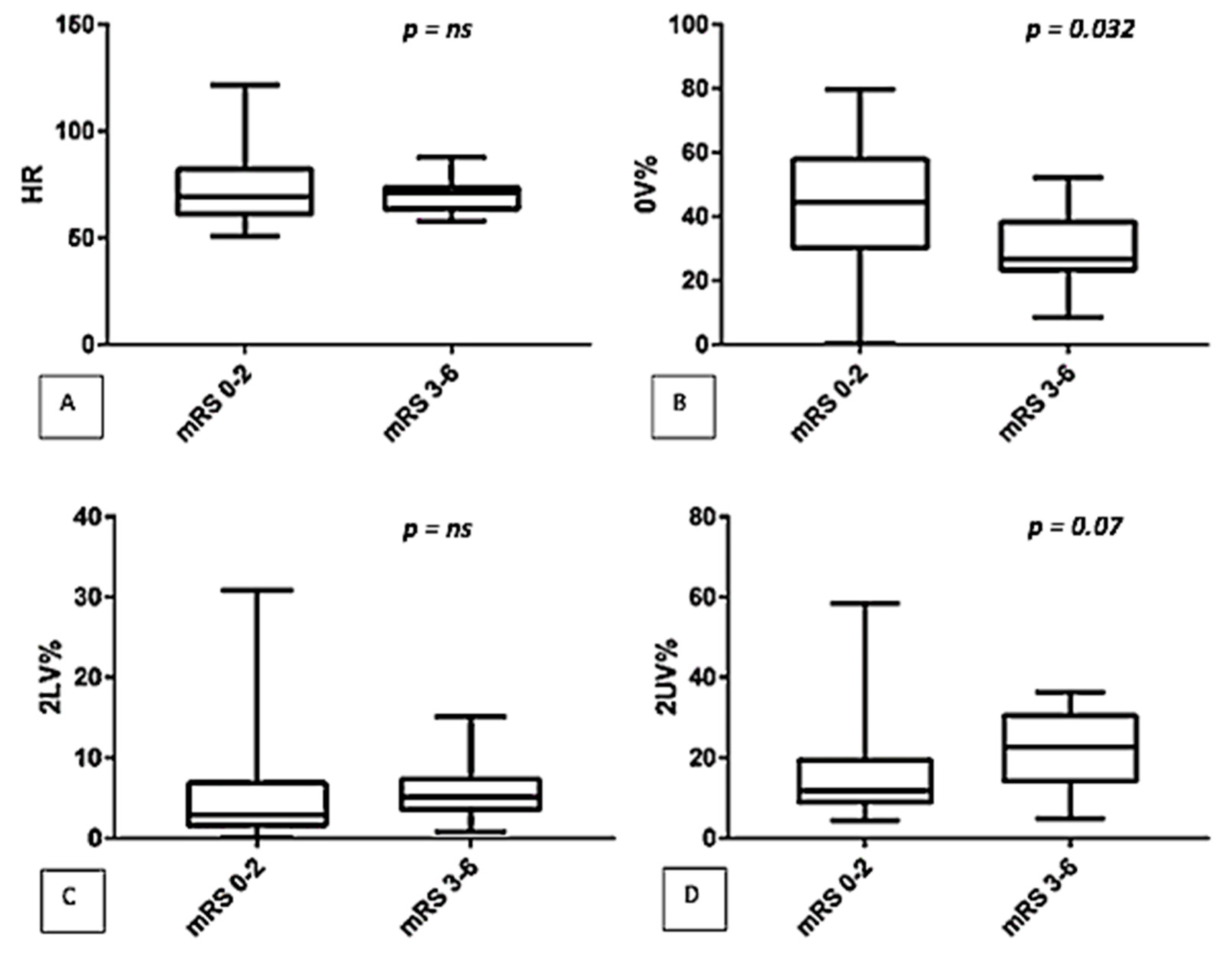
| 0V% | 38.9 (22.7–51.8) | 26.2 (22.9–50.5) | ns |
| 1V% | 42 (32.8–48.6) | 39.1 (34.3–46.6) | ns |
| 2LV% | 3.3 (1.3–8.5) | 5 (2.7–7.1) | ns |
| 2UV% | 12.6 (8.8–20.2) | 20.7 (13–32.2) * | 0.04 |
| Autonomic Parameters | Right Hemispheric Strokes (21) | Left Hemispheric Strokes (19) | p | Anterior Ischemic Lesions (33) | Posterior Ischemic Lesions (8) | p | Insular Ischemic Lesions (13) | Non-Insular Ischemic Lesions (28) | p |
|---|---|---|---|---|---|---|---|---|---|
| HR (bpm) | 68.2 (61.7–77.8) | 71.8 (60.2–84.4) | ns | 71.8 (62.1–83.7) | 61.2 (56.2–72.4) | ns | 68.6 (62.3–86.5) | 70.5 (58.5–79.7) | ns |
| Total Power (ms2) | 941 (274.9–1720.4) | 585.9 (272.9–940.8) | ns | 612.8 (260–1334.6) | 863.4 (540.5–976) | ns | 896.2 (260–1632.8) | 599.4 (333.2–1213.9) | ns |
| LF (n.u.) | 53.1 (39–80.7) | 56.7 (27.6–82.7) | ns | 53.1 (30.1–83.3) | 61.7 (33.1–80.4) | ns | 58.1 (28.1–86) | 54.9 (33.1–81.3) | ns |
| HF (n.u.) | 27.7 (15.1–50.2) | 26.7 (9.4–46.6) | ns | 26.7 (10.1–47.6) | 23.9 (12–52.2) | ns | 27.3 (10.2–53.1) | 25.8 (11.3–45.3) | ns |
| LF/HF | 1.8 (0.8–4.4) | 2.1 (0.7–8.7) | ns | 1.9 (0.8–7.6) | 2.1 (0.9–6.4) | ns | 2.1 (0.8–8.5) | 2 (0.8–6.4) | ns |
| 0V% | 31.9 (20.8–44.4) | 44.1 (25.5–53.4) | ns | 37.7 (21.7–49.6) | 35.8 (25.2–51.3) | ns | 37.3 (21.4–43.3) | 37.1 (23.1–55.8) | ns |
| 1V% | 42.7 (34–47.9) | 39.9 (33.1–49) | ns | 42 (33.6–47.2) | 43.9 (34.8–48.3) | ns | 42 (35.3–47.5) | 42.1 (30.8–48.2) | ns |
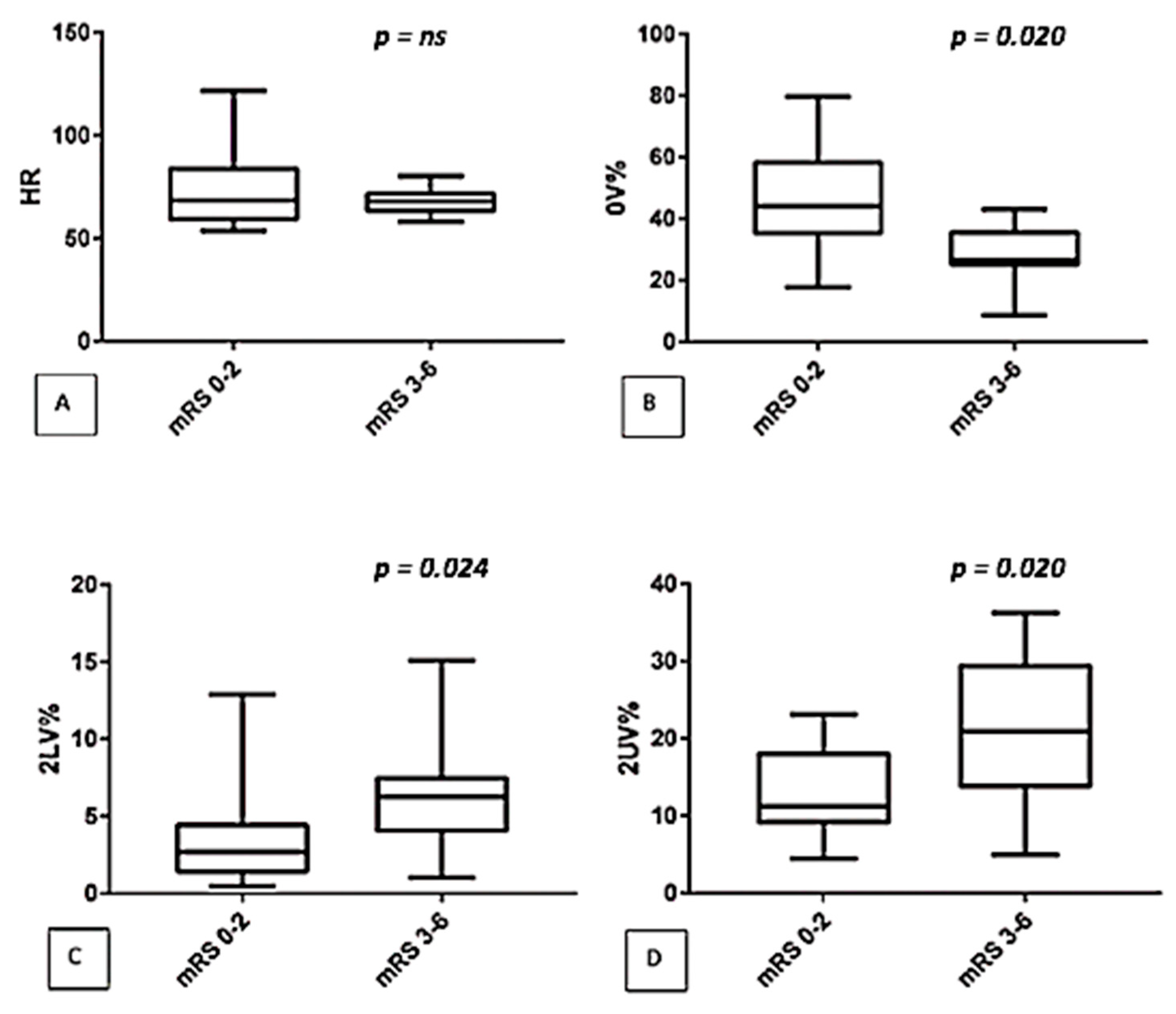
| 2LV% | 5.7 (2.7–9.7) | 2.9 (1.2–4.4) * | 0.022 | 4 (2.2–7.8) | 3.4 (1.2–6.7) | ns | 4.5 (1.2–7.8) | 3.5 (2.1–7.3) | ns |
| 2UV% | 16.5 (9.8–23.4) | 12.6 (9.4–20.1) | ns | 15.1 (9.2–22.5) | 12.7 (11.4–19.6) | ns | 18.2 (10.9–24.9) | 12.7 (8.9–20.3) | ns |
| Autonomic Parameters | T0 (19) | T1 (19) | p | T0 (15) | T7 (15) | p |
|---|---|---|---|---|---|---|
| HR (bpm) | 66 (58.8–82.5) | 65.8 (61.9–79.6) | ns | 73 (57.9–84.4) | 67.5 (57.5–74.4) | ns |
| Total Power (ms2) | 712.4 (439.8–1266.6) | 606.1 (247.6–1124.9) | ns | 585.9 (434.9–1227.6) | 400.9 (235.1–681.2) | ns |
| LF (n.u.) | 42.5 (28.6–82.7) | 50.9 (27.4–71.7) | ns | 46.9 (31.7–82.7) | 29.9 (13.5–64.2) | ns |
| HF (n.u.) | 25 (10.9–51.9) | 18.9 (13.3–33.5) | ns | 15.3 (9.4–51.9) | 30.9 (15.1–55.9) | ns |
| LF/HF | 1.8 (0.7–6.8) | 2.5 (0.8–6.2) | ns | 3.1 (0.7–8.8) | 1 (0.5–2.5) | ns |
| 0V% | 37.3 (26.3–45.7) | 34.8 (22.3–48.8) | ns | 37.7 (33–53.4) | 36.5 (26.1–49.5) | ns |
| 1V% | 44.6 (35–46.3) | 42.3 (36.3–46) | ns | 45.2 (33.1–49) | 42.5 (32.4–47.1) | ns |
| 2LV% | 3 (1.2–5.7) | 3.6 (1.3–6.1) | ns | 2.9 (1.2–5.7) | 2.8 (1.3–6) | ns |
| 2UV% | 12.8 (9.5–20.5) | 18.1 (11–26.1) | ns | 12.3 (9–20.1) | 17.7 (13–26.9) | ns |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tobaldini, E.; Sacco, R.M.; Serafino, S.; Tassi, M.; Gallone, G.; Solbiati, M.; Costantino, G.; Montano, N.; Torgano, G. Cardiac Autonomic Derangement Is Associated with Worse Neurological Outcome in the Very Early Phases of Ischemic Stroke. J. Clin. Med. 2019, 8, 852. https://doi.org/10.3390/jcm8060852
Tobaldini E, Sacco RM, Serafino S, Tassi M, Gallone G, Solbiati M, Costantino G, Montano N, Torgano G. Cardiac Autonomic Derangement Is Associated with Worse Neurological Outcome in the Very Early Phases of Ischemic Stroke. Journal of Clinical Medicine. 2019; 8(6):852. https://doi.org/10.3390/jcm8060852
Chicago/Turabian StyleTobaldini, Eleonora, Roberto M. Sacco, Serena Serafino, Michele Tassi, Gianluca Gallone, Monica Solbiati, Giorgio Costantino, Nicola Montano, and Giuseppe Torgano. 2019. "Cardiac Autonomic Derangement Is Associated with Worse Neurological Outcome in the Very Early Phases of Ischemic Stroke" Journal of Clinical Medicine 8, no. 6: 852. https://doi.org/10.3390/jcm8060852
APA StyleTobaldini, E., Sacco, R. M., Serafino, S., Tassi, M., Gallone, G., Solbiati, M., Costantino, G., Montano, N., & Torgano, G. (2019). Cardiac Autonomic Derangement Is Associated with Worse Neurological Outcome in the Very Early Phases of Ischemic Stroke. Journal of Clinical Medicine, 8(6), 852. https://doi.org/10.3390/jcm8060852






