Occurrence of Antithrombotic Related Adverse Events in Hospitalized Patients: Incidence and Clinical Context between 2008 and 2016
Abstract
1. Introduction
2. Materials and Methods
2.1. Design and Setting
- An unintended physical or mental injury
- The injury resulted in prolongation of hospital stay, temporary or permanent disability or death
- The injury was caused by healthcare management rather than the patient’s underlying disease
2.2. Identification of Antithrombotic Related Adverse Events
2.3. Outcomes
2.4. Statistical Analyses
3. Results
3.1. Study Population
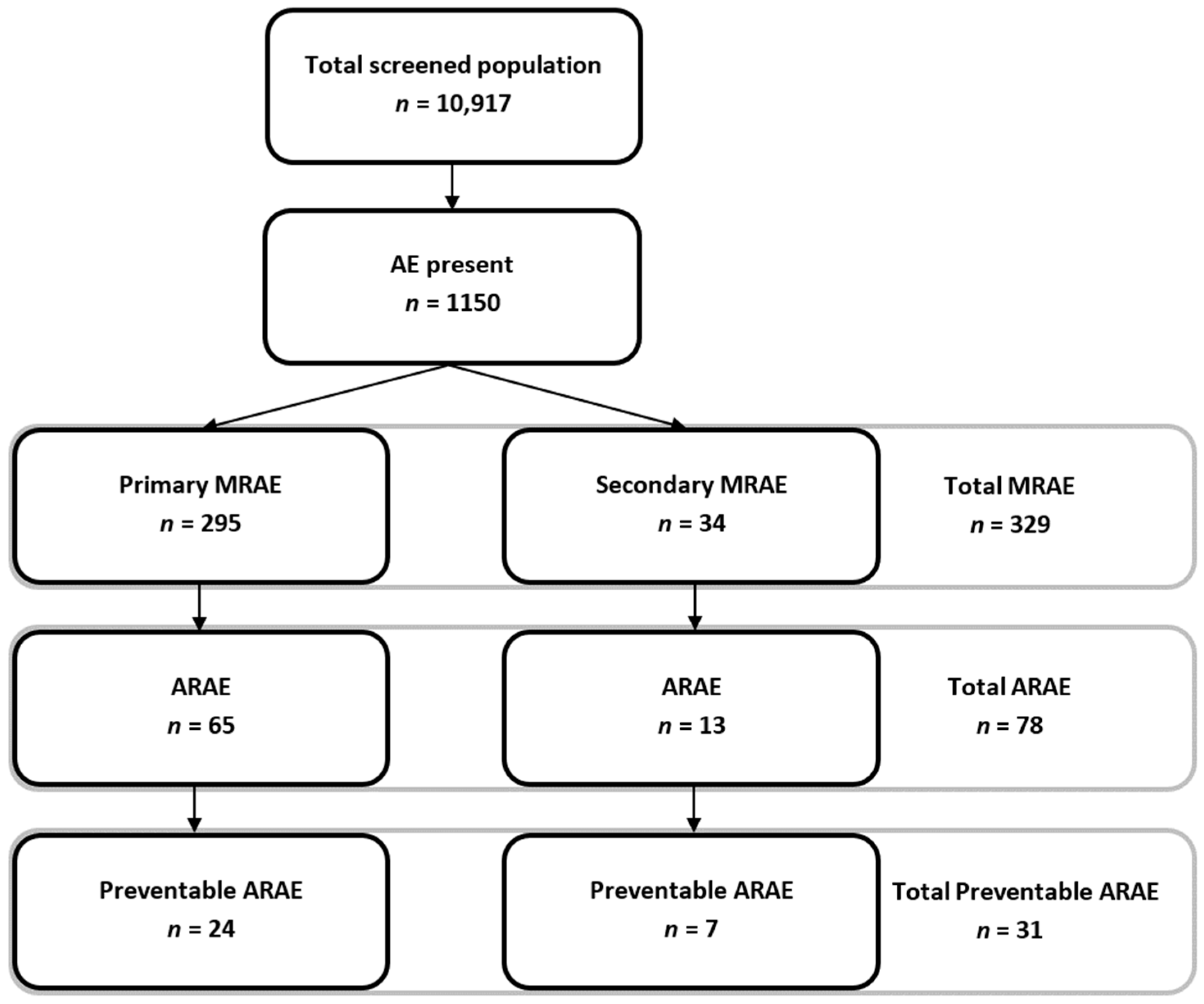
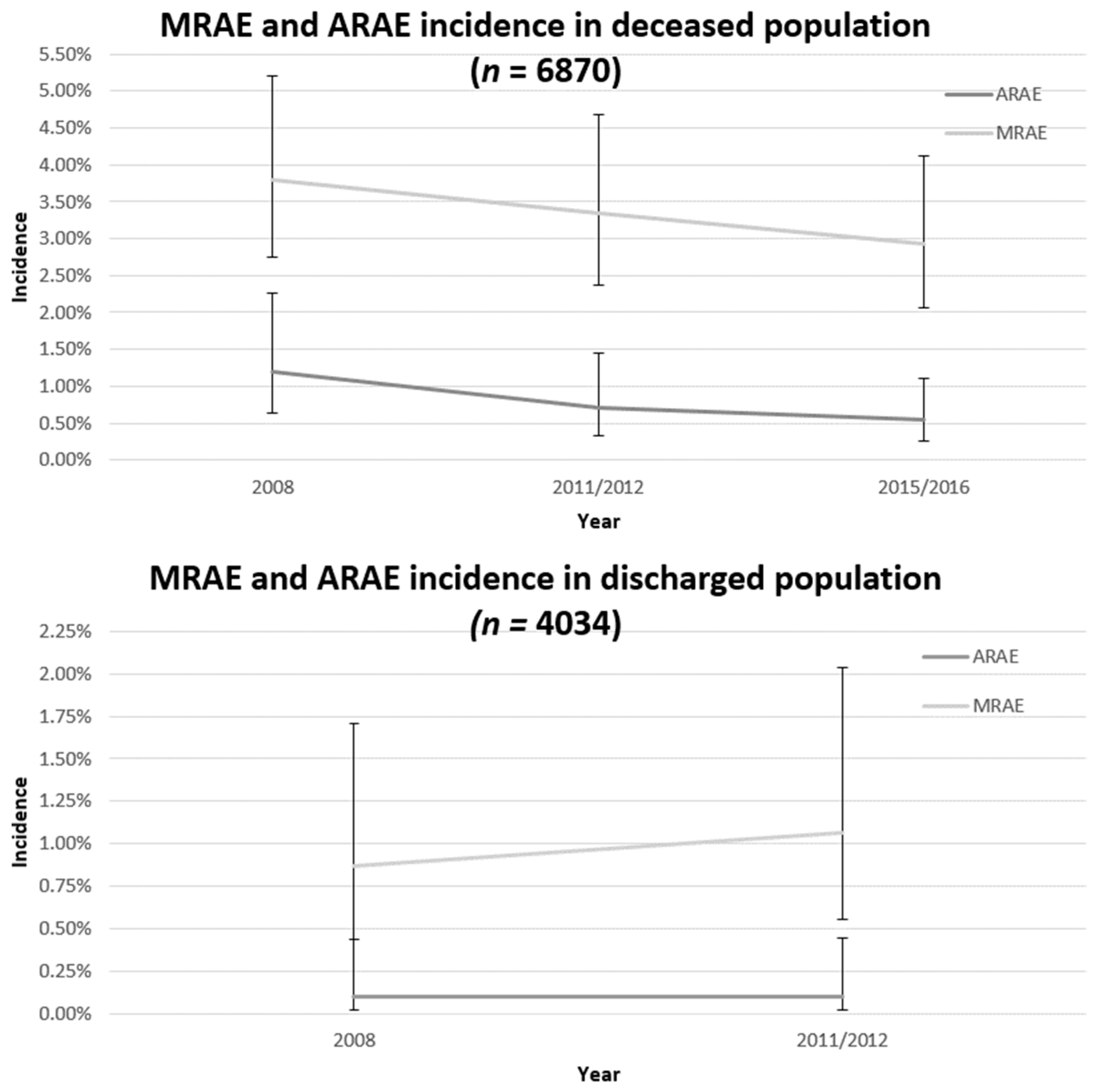
3.2. Antithrombotic Related Adverse Event Incidence
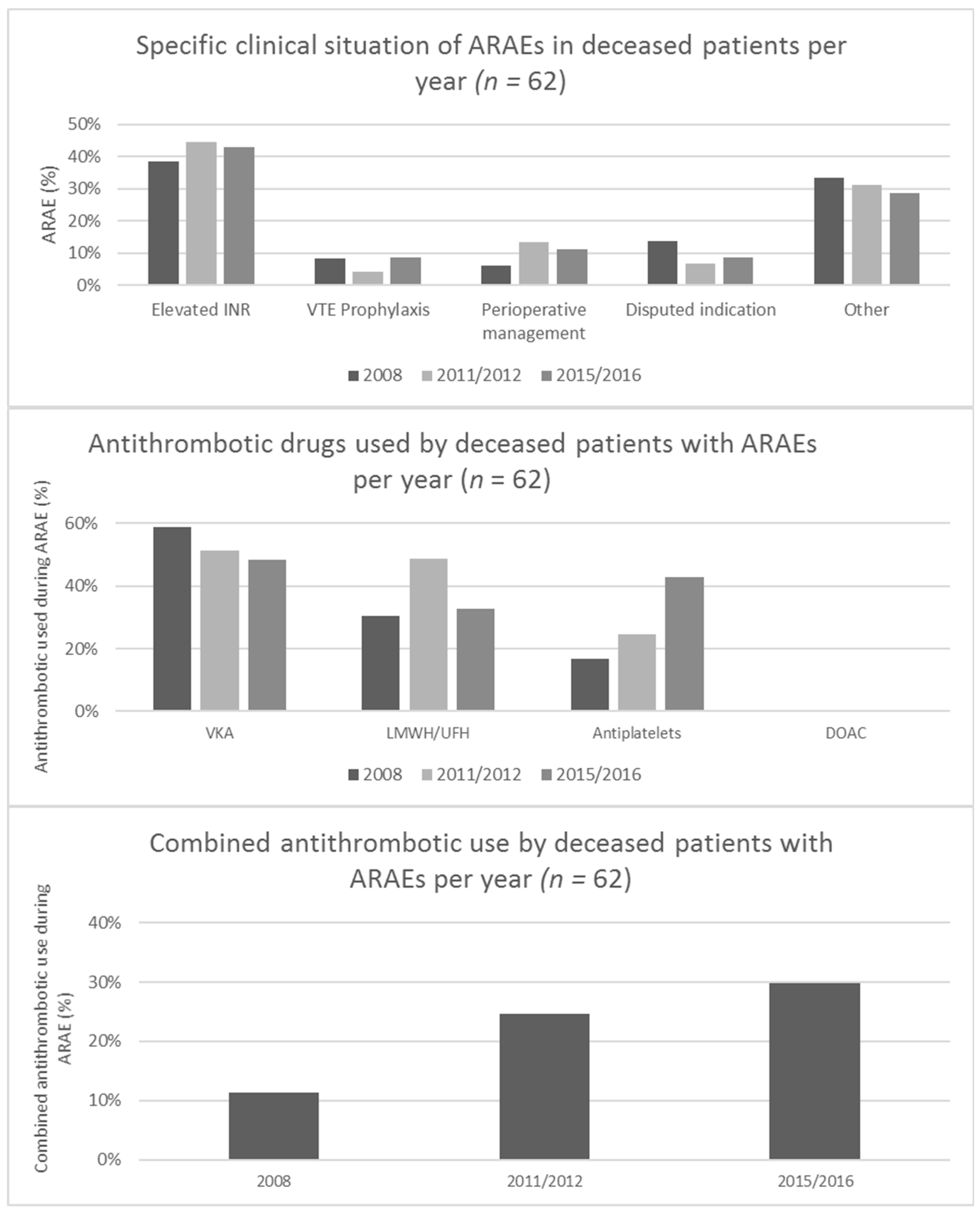
3.3. Clinical Context of Antithrombotic Related Adverse Events
3.4. Changes in the Clinical Context of Antithrombotic Related Adverse Events
3.5. Qualitative Antithrombotic Related Adverse Event Summaries
4. Discussion
4.1. Main Findings
4.2. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Patriquin, C.; Crowther, M. 26—Antithrombotic Agents A2—Kitchens, Craig, S. In Consultative Hemostasis and Thrombosis, 3rd ed.; Kessler, C.M., Konkle, B.A., Eds.; W.B. Saunders: Philadelphia, PA, USA, 2013; pp. 477–495. [Google Scholar]
- Mega, J.L.; Simon, T. Pharmacology of antithrombotic drugs: an assessment of oral antiplatelet and anticoagulant treatments. Lancet 2015, 386, 281–291. [Google Scholar] [CrossRef]
- Rohla, M.; Weiss, T.W.; Wojta, J.; Niessner, A.; Huber, K. Double or triple antithrombotic combination therapy in patients who need anticoagulation and antiplatelet therapy in parallel. Eur. Hear. J. Cardiovasc. Pharmacother. 2015, 1, 191–197. [Google Scholar] [CrossRef] [PubMed]
- Dowd, M. Concomitant antiplatelet and anticoagulation therapy: Indications, controversies and practical advice. Thromb. Res. 2008, 123, S11–S15. [Google Scholar] [CrossRef] [PubMed]
- Wiviott, S.D.; Braunwald, E.; McCabe, C.H.; Montalescot, G.; Ruzyllo, W.; Gottlieb, S.; Neumann, F.-J.; Ardissino, D.; De Servi, S.; Murphy, S.A.; et al. Prasugrel versus Clopidogrel in Patients with Acute Coronary Syndromes. N. Engl. J. Med. 2007, 357, 2001–2015. [Google Scholar] [CrossRef] [PubMed]
- Yusuf, S.; Zhao, F.; Mehta, S.R.; Chrolavicius, S.; Tognoni, G.; Fox, K.K. Effects of Clopidogrel in Addition to Aspirin in Patients with Acute Coronary Syndromes without ST-Segment Elevation. N. Engl. J. Med. 2001, 345, 494–502. [Google Scholar] [PubMed]
- Wysowski, D.K.; Nourjah, P.; Swartz, L. Bleeding complications with warfarin use: A prevalent adverse effect resulting in regulatory action. Arch. Intern. Med. 2007, 167, 1414–1419. [Google Scholar] [CrossRef]
- Kirchhof, P.; Benussi, S.; Kotecha, D.; Ahlsson, A.; Atar, D.; Casadei, B.; Castellà, M.; Diener, H.-C.; Heidbuchel, H.; Hendriks, J.; et al. 2016 ESC Guidelines for the management of atrial fibrillation developed in collaboration with EACTS. Kardiologia Polska 2016, 74, 1359–1469. [Google Scholar] [CrossRef]
- La Regina, M.; Orlandini, F.; Marchini, F.; Marinaro, A.; Bonacci, R.; Bonanni, P.; Corsini, F.; Ceraudo, A.M.; Pacetti, E.; Scuotri, L.; et al. Combined assessment of thrombotic and haemorrhagic risk in acute medical patients. Thromb. Haemost. 2016, 115, 392–398. [Google Scholar] [CrossRef]
- Beest, F.P.-V.; Van Meegen, E.; Rosendaal, F.R.; Stricker, B.H.C. Characteristics of Anticoagulant Therapy and Comorbidity Related to Overanticoagulation. Thromb. Haemost. 2001, 86, 569–574. [Google Scholar] [CrossRef]
- Holbrook, A.; Schulman, S.; Witt, D.M.; Vandvik, P.O.; Fish, J.; Kovacs, M.J.; Svensson, P.J.; Veenstra, D.L.; Crowther, M.; Guyatt, G.H. Evidence-based management of anticoagulant therapy: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest 2012, 141 (Suppl. 2), e152S–s184S. [Google Scholar] [CrossRef]
- Douketis, J.D.; Spyropoulos, A.C.; Spencer, F.A.; Mayr, M.; Jaffer, A.K.; Eckman, M.H.; Dunn, A.S.; Kunz, R. Perioperative management of antithrombotic therapy: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest 2012, 141 (Suppl. 2), e326S–e350S. [Google Scholar] [CrossRef] [PubMed]
- Zaidenstein, R.; Eyal, S.; Efrati, S.; Akivison, L.; Michowitz, M.K.; Nagornov, V.; Golik, A. Adverse drug events in hospitalized patients treated with cardiovascular drugs and anticoagulants. Pharmacoepidemiol. Drug Saf. 2002, 11, 235–238. [Google Scholar] [CrossRef] [PubMed]
- Hoonhout, L.H.; de Bruijne, M.C.; Wagner, C.; Asscheman, H.; van der Wal, G.; van Tulder, M.W. Nature, occurrence and consequences of medication-related adverse events during hospitalization: A retrospective chart review in The Netherlands. Drug Safety 2010, 33, 853–864. [Google Scholar] [CrossRef] [PubMed]
- Marquet, K.; Claes, N.; De Troy, E.; Kox, G.; Droogmans, M.; Vleugels, A. A multicenter record review of in-hospital adverse drug events requiring a higher level of care. Acta Clin. Belg. 2017, 72, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Ebbesen, J.; Buajordet, I.; Erikssen, J.; Brørs, O.; Hilberg, T.; Svaar, H.; Sandvik, L. Drug-Related Deaths in a Department of Internal Medicine. Arch. Intern. Med. 2001, 161, 2317. [Google Scholar] [CrossRef] [PubMed]
- Saedder, E.A.; Brock, B.; Nielsen, L.P.; Bonnerup, D.K.; Lisby, M. Identifying high-risk medication: a systematic literature review. Eur. J. Clin. Pharmacol. 2014, 70, 637–645. [Google Scholar] [CrossRef] [PubMed]
- Laatikainen, O.; Miettunen, J.; Sneck, S.; Lehtiniemi, H.; Tenhunen, O.; Turpeinen, M. The prevalence of medication-related adverse events in inpatients—a systematic review and meta-analysis. Eur. J. Clin. Pharmacol. 2017, 73, 1539–1549. [Google Scholar] [CrossRef] [PubMed]
- Spector, W.D.; Limcangco, R.; Furukawa, M.F.; Encinosa, W.E. The Marginal Costs of Adverse Drug Events Associated With Exposures to Anticoagulants and Hypoglycemic Agents During Hospitalization. Med Care 2017, 55, 856–863. [Google Scholar] [CrossRef]
- Hug, B.L.; Witkowski, D.J.; Sox, C.M.; Keohane, C.A.; Seger, D.L.; Yoon, C.; Matheny, M.E.; Bates, D.W. Adverse drug event rates in six community hospitals and the potential impact of computerized physician order entry for prevention. J. Gen. Intern. Med. 2010, 25, 31–81. [Google Scholar] [CrossRef]
- Poon, E.G.; Keohane, C.A.; Yoon, C.S.; Ditmore, M.; Bane, A.; Levtzion-Korach, O.; Moniz, T.; Rothschild, J.M.; Kachalia, A.B.; Hayes, J.; et al. Effect of Bar-Code Technology on the Safety of Medication Administration. Obstet. Gynecol. Surv. 2010, 65, 629–630. [Google Scholar] [CrossRef]
- Baines, R.; Langelaan, M.; De Bruijne, M.; Spreeuwenberg, P.; Wagner, C. How effective are patient safety initiatives? A retrospective patient record review study of changes to patient safety over time. BMJ Qual. Saf. 2015, 24, 561–571. [Google Scholar] [CrossRef] [PubMed]
- Baines, R.J.; Langelaan, M.; De Bruijne, M.C.; Asscheman, H.; Spreeuwenberg, P.; Van De Steeg, L.; Siemerink, K.M.; Van Rosse, F.; Broekens, M.; Wagner, C. Changes in adverse event rates in hospitals over time: a longitudinal retrospective patient record review study. BMJ Qual. Saf. 2013, 22, 290–298. [Google Scholar] [CrossRef] [PubMed]
- Langelaan, M.; Broekens, M.A.; Bruijne de, M.C.; Groot de, J.F.; Moesker, M.J.; Porte, P.J.; Schutijser, B.; Singotani, R.; Smits, M.; Zwaan, L.; et al. Monitor zorggerelateerde schade 2015/2016: Dossieronderzoek bij overleden patiënten in Nederlandse ziekenhuizen; Netherlands Institute for Health Services Research(NIVEL): Utrecht, The Netherlands, 2017. [Google Scholar]
- Klopotowska, J.; Schutijser, B.; Bruijne, M.D.; Wagner, C. Tweede evaluatie van het VMS veilgheidprogramma; NIVEL en EMGO+ instituut: Utrecht, The Netherlands, 2016. [Google Scholar]
- Samenvatting Medische Jaarverslagen 2017; Federatie Nederlandse Trombosediensten: Leiden, The Netherlands, 2018.
- Verdubbeling van gebruik nieuwe antistollingsmiddelen. Pharmaceutisch Weekblad 2016, 151, 14.
- Holbrook, A.M.; Pereira, J.A.; Labiris, R.; McDonald, H.; Douketis, J.D.; Crowther, M.; Wells, P.S. Systematic Overview of Warfarin and Its Drug and Food Interactions. Arch. Intern. Med. 2005, 165, 1095–1106. [Google Scholar] [CrossRef] [PubMed]
- Meegaard, P.M.; Holck, L.H.V.; Pottegård, A.; Madsen, H.; Hallas, J. Excessive anticoagulation with warfarin or phenprocoumon may have multiple causes. Dan. Med J. 2012, 59, 4383. [Google Scholar]
- Frank, C. Deprescribing: a new word to guide medication review. Can. Med Assoc. J. 2014, 186, 407–408. [Google Scholar] [CrossRef] [PubMed]
- Farrell, B.; Pottie, K.; Rojas-Fernandez, C.H.; Bjerre, L.M.; Thompson, W.; Welch, V. Methodology for Developing Deprescribing Guidelines: Using Evidence and GRADE to Guide Recommendations for Deprescribing. PLoS ONE 2016, 11, e0161248. [Google Scholar] [CrossRef] [PubMed]
- Bell, C.M.; Redelmeier, D.A. Mortality among Patients Admitted to Hospitals on Weekends as Compared with Weekdays. N. Engl. J. Med. 2001, 345, 663–668. [Google Scholar] [CrossRef]
- Hardmeier, B.; Braunschweig, S.; Cavallaro, M.; Roos, M.; Pauli-Magnus, C.; Giger, M.; Meier, P.J.; Fattinger, K. Adverse drug events caused by medication errors in medical inpatients. Swiss Med Wkly. 2004, 134, 664–670. [Google Scholar]
- Henriksen, J.N.; Nielsen, L.P.; Hellebek, A.; Poulsen, B.K. Medication errors involving anticoagulants: Data from the Danish patient safety database. Pharmacol. Res. Perspect. 2017, 5, e00307. [Google Scholar] [CrossRef] [PubMed]
| Study Period and Discharge Status | |||||
|---|---|---|---|---|---|
| Discharged | Deceased | ||||
| Hospital Characteristics | 2008 | 2011/2012 | 2008 | 2011/2012 | 2015/2016 |
| Number of admissions, n | 2016 | 2023 | 2007 | 2025 | 2846 |
| General hospital, n (%) | 1013 (50.25) | 794 (39.25) | 1015 (50.57) | 813 (40.15) | 1197 (42.06) |
| Tertiary teaching hospital, n (%) | 608 (30.16) | 822 (40.63) | 593 (29.55) | 820 (40.49) | 1052 (36.96) |
| University hospital, n (%) | 395 (19.59) | 407 (20.12) | 399 (19.88) | 392 (19.36) | 597 (20.98) |
| Discharged a | Deceased a | ||||
| Patient Characteristics | 2008 | 2011/2012 | 2008 | 2011/2012 | 2015/2016 |
| Male sex, % | 49.69 | 50.09 | 53.26 | 52.12 | 53.27 |
| Age (years), median (IQR) | 62 (47–75) | 63 (48–75) | 77 (67–84) | 77 (68–84) b | 77 (68–85) b |
| 1–65, % | 56.08 | 55.44 | 22.84 | 21.13 | 19.80 |
| 66–79, % | 28.29 | 28.87 | 37.13 | 37.23 | 36.39 |
| 80 and older, % | 15.63 | 15.70 | 39.95 | 41.64 | 43.78 |
| Length of stay (days): median (IQR) | 4 (2–8) | 3 (2–7) b | 7 (3–14) | 6 (2–13) b | 4 (1–11) b c |
| Non-elective admission, % | 52.44 | 53.36 | 86.21 | 88.50 b | 88.64 b |
| Department of admission, % | b d | b d | |||
| Surgery | 23.98 | 23.53 | 13.75 | 11.55 | 11.23 |
| Cardiology | 15.09 | 13.68 | 15.37 | 12.35 | 12.85 |
| Internal medicine | 17.98 | 17.62 | 29.41 | 29.36 | 31.59 |
| Orthopaedics | 11.57 | 11.62 | 1.50 | 1.38 | 1.10 |
| Neurology | 7.48 | 6.66 | 11.16 | 9.55 | 9.54 |
| Lung diseases | 5.75 | 6.52 | 13.33 | 15.26 | 12.87 |
| Urology | 5.34 | 5.36 | 0.87 | 1.32 | 0.86 |
| Other | 12.8 | 15.02 | 14.61 | 19.23 | 19.96 |
| Underwent surgical procedure, % | 45.48 | 45.17 | 20.52 | 19.04 | 15.07 b c |
| ICD 9 main diagnostic groups, % | b d | b d | b c d | ||
| Infection and parasitic diseases | 1.40 | 3.37 | 3.31 | 5.44 | 4.90 |
| Neoplasms | 11.76 | 11.15 | 19.06 | 19.16 | 12.44 |
| Endocrinic | 2.17 | 2.42 | 2.61 | 1.25 | 1.17 |
| Heart and vascular diseases | 19.93 | 17.20 | 33.64 | 29.69 | 24.14 |
| Respiratory diseases | 8.24 | 8.54 | 15.2 | 13.52 | 15.49 |
| Gastrointestinal diseases | 10.87 | 9.97 | 7.19 | 7.34 | 6.55 |
| Urogenital diseases | 6.41 | 6.30 | 2.81 | 2.59 | 3.43 |
| Signs and symptoms ill defined | 6.33 | 5.31 | 4.89 | 4.44 | 5.02 |
| Injury and poisoning | 9.67 | 9.17 | 5.94 | 6.12 | 6.68 |
| Other | 22.48 | 21.29 | 4.91 | 5.00 | 3.68 |
| Missing | 0.74 | 5.30 | 0.45 | 5.46 | 16.5 |
| Adverse event presence | |||||
| Adverse event present, n (%) | 152 (7.57) | 144 (6.92) | 315 (15.60) | 246 (11.93) b | 293 (9.86) b c |
| MRAE present, n (%) | 35 (1.76) | 36 (1.72) | 84 (4.08) | 73 (3.62) | 101 (3.44) |
| ARAE present, n (%) | 8 (0.51) | 9 (0.46) | 28 (1.35) | 16 (0.79) | 17 (0.54) b |
| Antithrombotic Used During Admission c | Patients Exposed to Antithrombotic Drugs During Admission (n = 1772) n (%, weighted) a | Patients with ARAE (n = 16) n | ARAE Incidence, % (95% CI) b | Odds Ratio ARAE (95% CI) b |
|---|---|---|---|---|
| VKA | 476 (27.59) | 9 | 0.61 (0.14–2.61) | 6.06 (2.02–18.14) |
| LMWH | 1162 (65.01) | 5 | 0.14 (0.03–0.74) | 1.37 (0.46–4.08) |
| Antiplatelet | 650 (36.95) | 6 | 0.43 (0.09–2.00) | 4.21 (1.41–12.57) |
| UFH | 170 (8.43) | 0 | - | - |
| DOAC | 35 (1.73) | 0 | - | - |
| All ARAEs (n = 79) %, Weighted a b | Preventable ARAEs (n = 32) %, Weighted a b c | |
|---|---|---|
| Hospital type | ||
| General hospital | 29.2 | 29.3 |
| Tertiary teaching hospital | 66.4 | 69.3 |
| University hospital | 4.4 | 1.4 |
| VKA use | 50.5 | 77.0 |
| LMWH/UFH use | 23.5 | 44.2 |
| DOAC use | 0 | - |
| Antiplatelet use | 45.0 | 2.5 |
| Combined antithrombotic use (2 or more) | 29.3 | 23.7 |
| Antithrombotic administered or omitted | ||
| Administered | 98.5 | 97.3 |
| Omitted | 1.5 | 2.7 |
| Specific clinical situation | ||
| Elevated INR | 34.6 | 50.6 |
| VTE prophylaxis | 1.0 | - |
| Perioperative/periprocedural antithrombotic management | 14.5 | 2.6 |
| Disputed antithrombotic indication | 19.0 | 43.2 |
| Adverse drug reaction | 6.6 | - |
| Patient related | 0.3 | - |
| Other | 24.1 | 0 |
| Type | ||
| Bleeding event | 91.7 | 95.7 |
| Thromboembolic event | 1.6 | 3.6 |
| Other | 6.8 | - |
| Medical specialty responsible for treatment during ARAE occurrence | ||
| Surgical speciality | 21.6 | 43.0 |
| Non-surgical specialty | 78.4 | 57.0 |
| Number of medical specialists involved in treatment | ||
| 1 | 36.2 | 2.7 |
| 2 | 36.0 | 64.9 |
| ≥3 | 27.8 | 32.4 |
| Admission department | ||
| Surgery | 6.0 | 0.7 |
| Cardiology | 18.8 | 6.7 |
| Internal medicine | 28.8 | 24.4 |
| Orthopaedics | 0.9 | - |
| Neurology | 7.2 | 0 |
| Lung diseases | 16.6 | 23.6 |
| Urology | 19.8 | 40.0 |
| Other | 1.8 | 3.0 |
| ARAE onset during weekend/holiday | 40.3 | 59.2 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moesker, M.J.; Schutijser, B.C.F.M.; de Groot, J.F.; Langelaan, M.; Spreeuwenberg, P.; Huisman, M.V.; de Bruijne, M.C.; Wagner, C. Occurrence of Antithrombotic Related Adverse Events in Hospitalized Patients: Incidence and Clinical Context between 2008 and 2016. J. Clin. Med. 2019, 8, 839. https://doi.org/10.3390/jcm8060839
Moesker MJ, Schutijser BCFM, de Groot JF, Langelaan M, Spreeuwenberg P, Huisman MV, de Bruijne MC, Wagner C. Occurrence of Antithrombotic Related Adverse Events in Hospitalized Patients: Incidence and Clinical Context between 2008 and 2016. Journal of Clinical Medicine. 2019; 8(6):839. https://doi.org/10.3390/jcm8060839
Chicago/Turabian StyleMoesker, Marco J., Bernadette C.F.M. Schutijser, Janke F. de Groot, Maaike Langelaan, Peter Spreeuwenberg, Menno V. Huisman, Martine C. de Bruijne, and Cordula Wagner. 2019. "Occurrence of Antithrombotic Related Adverse Events in Hospitalized Patients: Incidence and Clinical Context between 2008 and 2016" Journal of Clinical Medicine 8, no. 6: 839. https://doi.org/10.3390/jcm8060839
APA StyleMoesker, M. J., Schutijser, B. C. F. M., de Groot, J. F., Langelaan, M., Spreeuwenberg, P., Huisman, M. V., de Bruijne, M. C., & Wagner, C. (2019). Occurrence of Antithrombotic Related Adverse Events in Hospitalized Patients: Incidence and Clinical Context between 2008 and 2016. Journal of Clinical Medicine, 8(6), 839. https://doi.org/10.3390/jcm8060839






