Sleep after Heavy Alcohol Consumption and Physical Activity Levels during Alcohol Hangover
Abstract
1. Introduction
1.1. Self-Report
1.2. Real-Time Assessments
2. Methods
2.1. Design
2.2. GENEactiv Accelerometer Assessments of Sleep and Activity
2.3. Self-Reported Sleep (Next Morning)
2.4. Assessments of Alcohol Consumption (Real-Time and Retrospective)
2.5. Assessments of Hangover Severity (Next Morning)
2.6. Statistical Analysis
3. Results
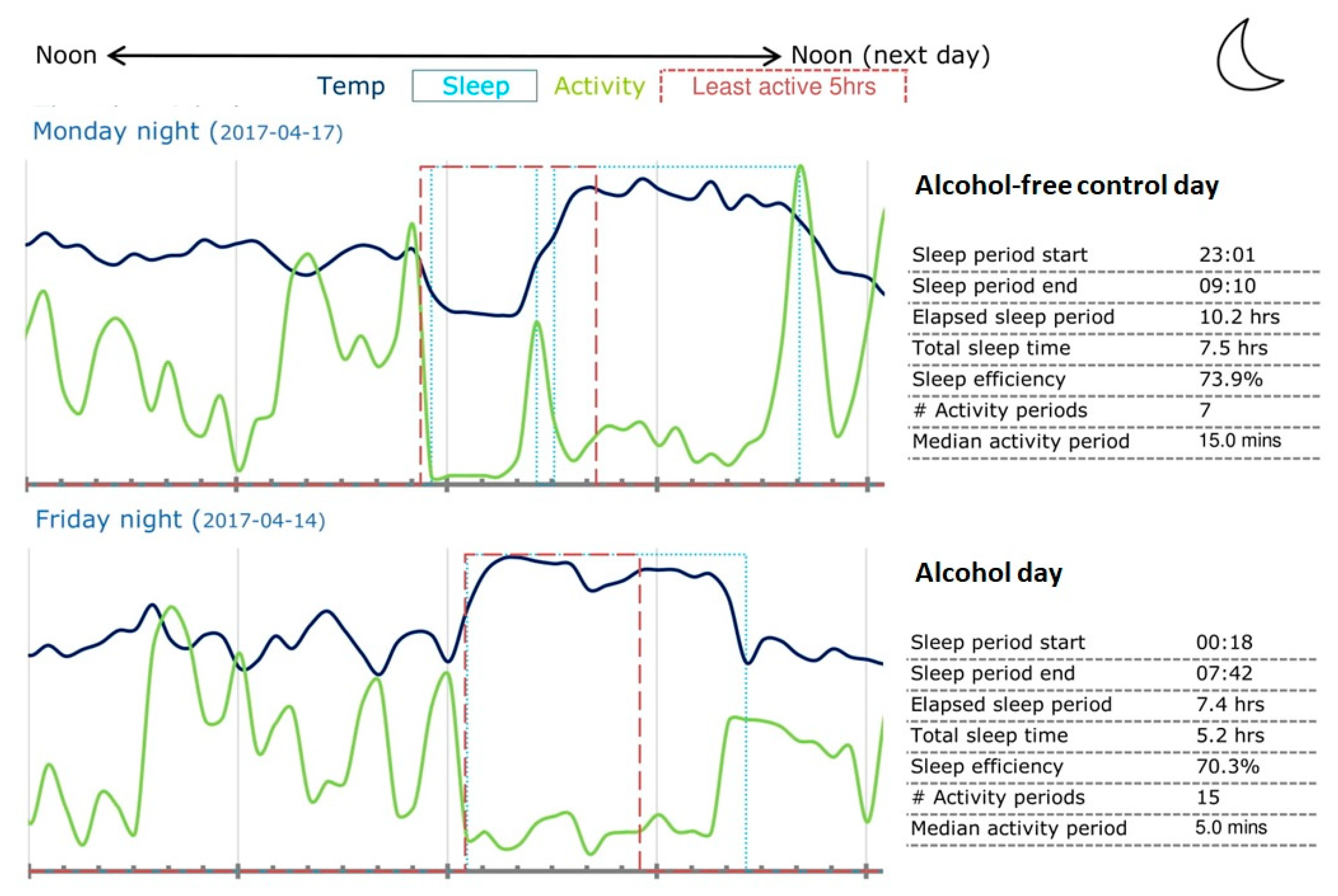
3.1. GENEActiv Sleep Assessments
3.2. Self-Reported Sleep
3.3. Correspondence between Objective and Subjective Sleep Assessments
3.4. Correspondence between Sleep Assessments, Alcohol Consumption, and Hangover Severity
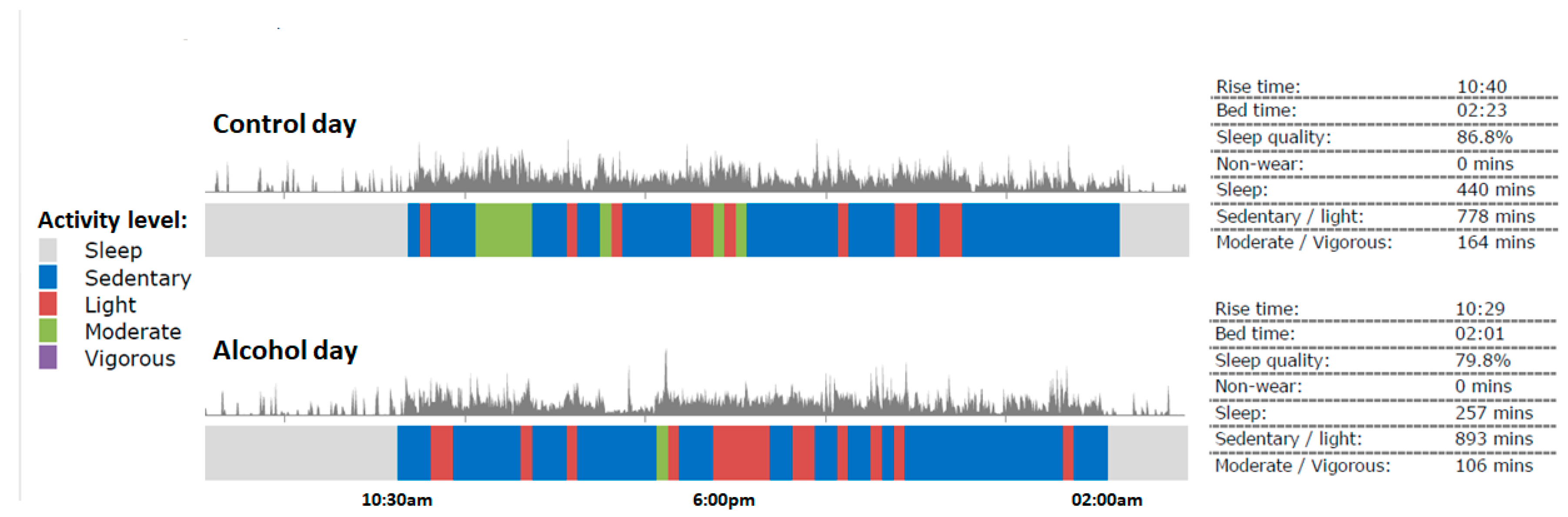
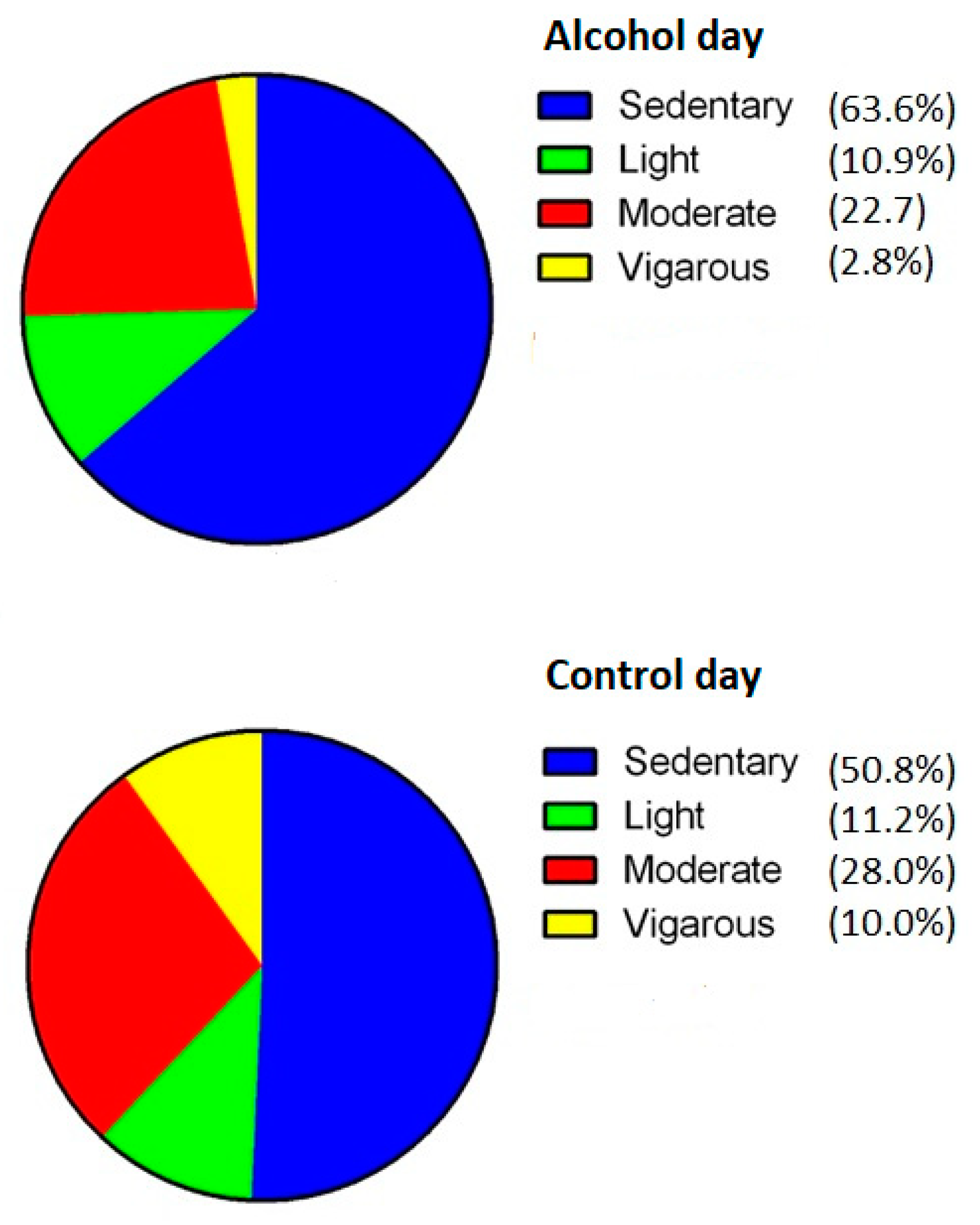
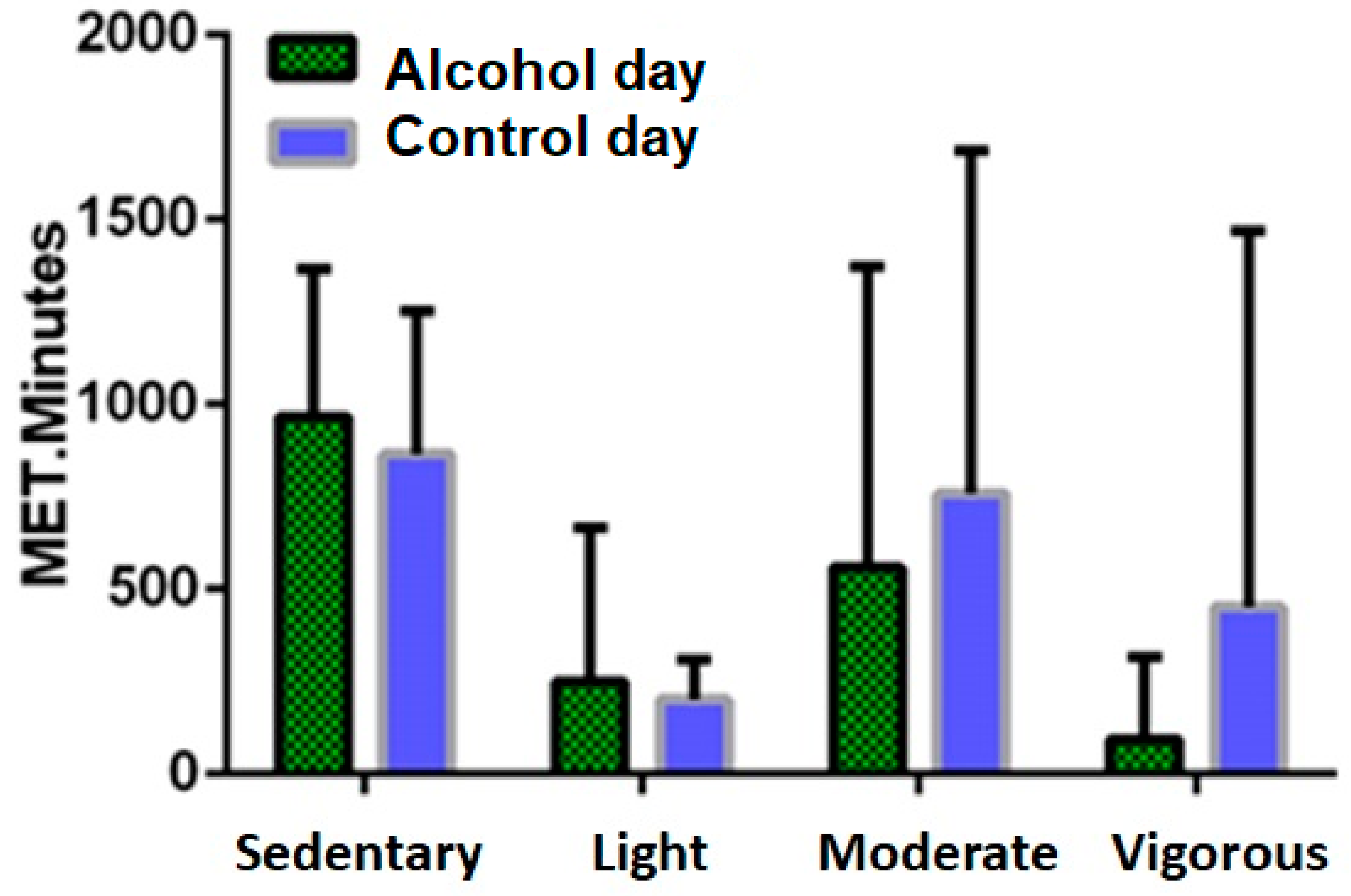
3.5. Physical Activity
3.6. Sleep and Drinking Variables Associated with Percentual Changes in Activity
4. Discussion
4.1. Objective Versus Subjective Assessments
4.2. Recall Bias
4.3. Daytime Activity Levels
4.4. Limitations and Objectives for Future Research
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Van Schrojenstein Lantman, M.; Mackus, M.; van de Loo, A.J.A.E.; Verster, J.C. Development of a definition for the alcohol hangover: Consumer descriptions and expert consensus. Curr. Drug Abuse Rev. 2016, 9, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Verster, J.C.; Bervoets, A.C.; de Klerk, S.; Vreman, R.A.; Olivier, B.; Roth, T.; Brookhuis, K.A. Effects of alcohol hangover on simulated highway driving performance. Psychopharmacology 2014, 231, 2999–3008. [Google Scholar] [CrossRef] [PubMed]
- Jongen, S.; Perrier, J.; Vuurman, E.F.; Ramaekers, J.G.; Vermeeren, A. Sensitivity and validity of psychometric tests for assessing driving impairment: Effects of sleep deprivation. PLoS ONE 2015, 10, e0117045. [Google Scholar] [CrossRef]
- Ebrahim, I.O.; Shapiro, C.M.; Williams, A.J.; Fenwick, P.B. Alcohol and sleep I: effects on normal sleep. Alcohol. Clin. Exp. Res. 2013, 37, 539–549. [Google Scholar] [CrossRef]
- Bajaj, L.; Singh, R. Alcohol hangover-its effects on human body. J. Addict. Clin. Res. 2018, 2, 14–16. [Google Scholar] [CrossRef]
- Hendler, R.A.; Ramchandani, V.A.; Gilman, J.; Hommer, D.W. Stimulant and sedative effects of alcohol. In Behavioral Neurobiology of Alcohol Addiction; Springer: Heidelberg, Germany, 2011; pp. 489–509. [Google Scholar]
- Martin, C.S.; Earleywine, M.; Musty, R.E.; Perrine, M.W.; Swift, R.M. Development and validation of the biphasic alcohol effects scale. Alcohol. Clin. Exp. Res. 1993, 17, 140–146. [Google Scholar] [CrossRef] [PubMed]
- Simpson, C.W.; Resch, G.E.; Millington, W.R.; Myers, R.D. Glycyl-L-glutamine injected centrally suppresses alcohol drinking in P rats. Alcohol 1998, 16, 101–107. [Google Scholar] [CrossRef]
- Roehrs, T.; Yoon, J.; Roth, T. Nocturnal and next-day effects of ethanol and basal level of sleepiness. Hum. Psychopharmacol. Clin. Exp. 1991, 6, 307–311. [Google Scholar] [CrossRef]
- Roehrs, T.; Roth, T. Sleep, sleepiness, and alcohol use. Alcohol Res. Health 2001, 25, 101–109. [Google Scholar] [PubMed]
- Finnigan, F.; Hammersley, R.; Cooper, T. An examination of next-day hangover effects after a 100 mg/100 mL dose of alcohol in heavy social drinkers. Addiction 1998, 93, 1829–1838. [Google Scholar] [CrossRef] [PubMed]
- McKinney, A.; Coyle, K. Alcohol hangover effects on measures of affect the morning after a normal night’s drinking. Alcohol Alcohol. 2006, 41, 54–60. [Google Scholar] [CrossRef]
- Hogewoning, A.; Van de Loo, A.J.A.E.; Mackus, M.; Raasveld, S.J.; De Zeeuw, R.; Bosma, E.R.; Bouwmeester, N.H.; Brookhuis, K.A.; Garssen, J.; Verster, J.C. Characteristics of social drinkers with and without a hangover after heavy alcohol consumption. Subst. Abuse Rehabil. 2016, 7, 161–167. [Google Scholar] [CrossRef]
- Rohsenow, D.J.; Howland, J.; Minsky, S.J.; Arnedt, J.T. Effects of heavy drinking by maritime academy cadets on hangover, perceived sleep, and next-day ship power plant operation. J. Stud. Alcohol 2006, 67, 406–415. [Google Scholar] [CrossRef]
- Van Schrojenstein Lantman, M.; Mackus, M.; Roth, T.; Verster, J.C. Total sleep time, alcohol consumption and the duration and severity of alcohol hangover. Nat. Sci. Sleep 2017, 9, 181–186. [Google Scholar] [CrossRef]
- Van Schrojenstein Lantman, M.; Roth, T.; Roehrs, T.; Verster, J.C. Alcohol hangover, sleep quality, and daytime sleepiness. Sleep Vigil. 2017, 1, 37–41. [Google Scholar] [CrossRef]
- Penning, R.; McKinney, A.; Verster, J.C. Alcohol hangover symptoms and their contribution to overall hangover severity. Alcohol Alcohol. 2012, 47, 248–252. [Google Scholar] [CrossRef]
- Van Schrojenstein Lantman, M.; Mackus, M.; van de Loo, A.J.A.E.; Verster, J.C. The impact of alcohol hangover symptoms on cognitive and physical functioning, and mood. Hum. Psychopharmacol. 2017, 32. [Google Scholar] [CrossRef]
- Rohsenow, D.J.; Howland, J.; Arnedt, J.T.; Almeida, A.B.; Greece, J.; Minsky, S.; Kempler, C.S.; Sales, S. Intoxication with bourbon versus vodka: Effects on hangover, sleep, and next-day neurocognitive performance in young adults. Alcohol Clin. Exp. Res. 2010, 34, 509–518. [Google Scholar] [CrossRef]
- Roehrs, T.; Papineau, K.; Rosenthal, L.; Roth, T. Ethanol as a hypnotic in insomniacs: Self administration and effects on sleep and mood. Neuropsychopharmacol 1999, 20, 279–286. [Google Scholar] [CrossRef]
- Feige, B.; Gann, H.; Brueck, R.; Hornyak, M.; Litsch, S.; Hohagen, F.; Riemann, D. Effects of alcohol on polysomnographically recorded sleep in healthy subjects. Alcohol Clin. Exp. Res. 2006, 30, 1527–1537. [Google Scholar] [CrossRef]
- Wilkinson, A.N.; Afshar, M.; Ali, O.; Bhatti, W.; Hasday, J.D.; Netzer, G.; Verceles, A.C. Effects of binge alcohol consumption on sleep and inflammation in healthy volunteers. J. Int. Med. Res. 2018. [Google Scholar] [CrossRef]
- Rohsenow, D.J.; Howland, J.; Minsky, S.J.; Greece, J.; Almeida, A.; Roehrs, T.A. The acute hangover scale: A new measure of immediate hangover symptoms. Addict. Behav. 2007, 32, 1314–1320. [Google Scholar] [CrossRef] [PubMed]
- Huntley, G.; Treloar, H.; Blanchard, A.; Monti, P.M.; Carey, K.B.; Rohsenow, D.J.; Miranda, R., Jr. An event-level investigation of hangovers’ relationship to age and drinking. Exp. Clin. Psychopharmacol. 2015, 23, 314. [Google Scholar] [CrossRef] [PubMed]
- Riordan, B.C.; Conner, T.S.; Flett, J.A.; Scarf, D. A brief orientation week ecological momentary intervention to reduce university student alcohol consumption. J. Stud. Alcohol Drugs 2015, 76, 525–529. [Google Scholar] [CrossRef]
- Selzer, M.L.; Vinokur, A.; van Rooijen, L. A self-administered Short Michigan Alcoholism Screening Test (SMAST). J. Stud. Alcohol 1975, 36, 117–126. [Google Scholar] [CrossRef]
- ActivInsights. The GENEActiv Product Range. Available online: https://www.activinsights.com/products/geneactiv (accessed on 1 August 2018).
- GENEActiv. Open Platform. Available online: https://open.geneactiv.org/ (accessed on 1 August 2018).
- Esliger, D.W.; Rowlands, A.V.; Hurst, T.L.; Catt, M.; Murray, P.; Eston, R.G. Validation of the GENEA Accelerometer. Med. Sci. Sports Exerc. 2011, 43, 1085–1093. [Google Scholar] [CrossRef]
- Jette, M.; Sidney, K.; Blümchen, G. Metabolic equivalents (METS) in exercise testing, exercise prescription, and evaluation of functional capacity. Clin. Cardiol. 1990, 13, 555–565. [Google Scholar] [CrossRef] [PubMed]
- Verster, J.C.; Roth, T. Drivers can poorly predict their own driving impairment: A comparison between measurements of subjective and objective driving quality. Psychopharmacol 2012, 219, 775–781. [Google Scholar] [CrossRef]
- Baldwin, W. Information no one else knows: The value of self-report. In The Science of Self-Report; Psychology Press: Hove, UK, 1999; pp. 15–20. [Google Scholar]
- Jobe, J.B.; Herrmann, D.J. Implications of models of survey cognition for memory theory. Basic Appl. Mem. Res. 1996, 2, 193–205. [Google Scholar]
- Godden, D.R.; Baddeley, A.D. Context-dependent memory in two natural environments: On land and underwater. Br. J. Psychol. 1975, 66, 325–331. [Google Scholar] [CrossRef]
- McKinney, A.; Coyle, K. Next day effects of a normal night’s drinking on memory and psychomotor performance. Alcohol Alcohol. 2004, 39, 509–513. [Google Scholar] [CrossRef]
- Verster, J.C.; Van Duin, D.; Volkerts, E.R.; Schreuder, A.H.C.M.L.; Verbaten, M.N. Alcohol hangover effects on memory functioning and vigilance performance after an evening of binge drinking. Neuropsychopharmacol 2003, 28, 740–746. [Google Scholar] [CrossRef]
- White, A.M. What happened? Alcohol, memory blackouts, and the brain. Alcohol Res. Health 2003, 27, 186–197. [Google Scholar]
- Verster, J.C.; Tiplady, B.; McKinney, A. Mobile technology and naturalistic study designs in addiction research. Curr. Drug Abuse Rev. 2012, 5, 169–171. [Google Scholar] [CrossRef]
- Dulin, P.L.; Alvarado, C.E.; Fitterling, J.M.; Gonzalez, V.M. Comparisons of alcohol consumption by timeline follow back vs. smartphone-based daily interviews. Addict. Res. Theory 2017, 25, 195–200. [Google Scholar] [CrossRef]
- Collins, R.L.; Kashdan, T.B.; Gollnisch, G. The feasibility of using cellular phones to collect ecological momentary assessment data: Application to alcohol consumption. Exp. Clin. Psychopharmacol. 2003, 11, 73. [Google Scholar] [CrossRef]
- Monk, R.L.; Heim, D.; Qureshi, A.; Price, A. I have no clue what I drunk last night using Smartphone technology to compare in-vivo and retrospective self-reports of alcohol consumption. PLoS ONE 2015, 10, e0126209. [Google Scholar] [CrossRef]
- Luczak, S.E.; Rosen, I.G.; Wall, T.L. Development of a real-time repeated-measures assessment protocol to capture change over the course of a drinking episode. Alcohol Alcohol. 2015, 50, 180–187. [Google Scholar] [CrossRef]
- Krenek, M.; Lyons, R.; Simpson, T.L. Degree of correspondence between daily monitoring and retrospective recall of alcohol use among men and women with comorbid AUD and PTSD. Am. J. Addict. 2016, 25, 145–151. [Google Scholar] [CrossRef]
- Howland, J.; Rohsenow, D.J.; McGeary, J.E.; Streeter, C.; Verster, J.C. Proceedings of the 2010 symposium on hangover and other residual alcohol effects: Predictors and consequences. Open Addict. J. 2010, 3, 131–132. [Google Scholar] [CrossRef][Green Version]
| Mean (SD) | |
|---|---|
| Demographics | |
| n | 25 |
| Male/Female | 12/13 |
| Age (years) | 26.0 (7.1) |
| Age of first drink (years) | 14.9 (1.7) |
| Usual Total sleep time (TST) (h:min) | 6:33 (1:55) |
| Alcohol consumption on study night | |
| Reported units of alcohol consumed (real-time) | 11.4 (3.8) |
| Reported units of alcohol consumed (retrospective) | 8.0 (2.7) |
| Start time drinking (h:min) | 20:48 (3:47) |
| Stop time drinking (h:min) | 01:17 (1:12) |
| Duration of alcohol consumption (min) | 269 (149.2) |
| Consumed more alcohol than planned (Yes/No) | 10/15 |
| Mean (SD) hangover severity | 2.2 (0.9) |
| Sleep Outcomes | Alcohol Day | Control Day | |
|---|---|---|---|
| Mean (SD) | Mean (SD) | p-Value | |
| Sleep start time | 02:41 (1:17) | 00.41 (1.16) | 0.00 * |
| Wake-up time | 9:46 (1:37) | 8.56 (1:53) | 0.07 |
| Time in bed (h:min) | 9:27 (2:46) | 9:22 (2:14) | 0.85 |
| Total sleep time (TST) (h:min) | 6:34 (3:45) | 7:59 (4:42) | 0.16 |
| Sleep efficiency (%) | 69.0 (16.7) | 80.0 (15.2) | 0.04 * |
| Number of nightly activity periods | 8.4 (5.5) | 8.0 (6.1) | 0.81 |
| Median duration nightly activity (min) | 27.5 (59.9) | 33.5 (79.0) | 0.46 |
| Alcohol Day | Control Day | ||
|---|---|---|---|
| Mean (SD) | Mean (SD) | p-Value | |
| Start time sleeping | 02:28 (1:14) | 00:23 (1:11) | 0.00 * |
| Sleep onset latency (min) | 29 (41) | 49 (62) | 0.22 |
| Total sleep time (TST) | 06:40 (1:53) | 7:01 (1:50) | 0.44 |
| Wake-up time | 09:00 (2:25) | 8:25 (1:14) | 0.00 * |
| Sleep efficiency (%) | 94.5% (17.0%) | 91.6% (8.6%) | 0.55 |
| Sleep quality 1 | |||
| Good-Bad | 3.7 (1.6) | 2.8 (1.3) | 0.046 * |
| Satisfying-Not Satisfying | 4.0 (1.6) | 3.4 (1.3) | 0.07 |
| Refreshing-Not Refreshing | 4.2 (1.4) | 3.1 (1.2) | 0.01 * |
| Restful-Not Restful | 4.6 (1.3) | 3.4 (1.4) | 0.01 * |
| Light-Deep Sleep | 5.0 (1.7) | 5.0 (1.2) | 0.92 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Devenney, L.E.; Coyle, K.B.; Roth, T.; Verster, J.C. Sleep after Heavy Alcohol Consumption and Physical Activity Levels during Alcohol Hangover. J. Clin. Med. 2019, 8, 752. https://doi.org/10.3390/jcm8050752
Devenney LE, Coyle KB, Roth T, Verster JC. Sleep after Heavy Alcohol Consumption and Physical Activity Levels during Alcohol Hangover. Journal of Clinical Medicine. 2019; 8(5):752. https://doi.org/10.3390/jcm8050752
Chicago/Turabian StyleDevenney, Lydia E., Kieran B. Coyle, Thomas Roth, and Joris C. Verster. 2019. "Sleep after Heavy Alcohol Consumption and Physical Activity Levels during Alcohol Hangover" Journal of Clinical Medicine 8, no. 5: 752. https://doi.org/10.3390/jcm8050752
APA StyleDevenney, L. E., Coyle, K. B., Roth, T., & Verster, J. C. (2019). Sleep after Heavy Alcohol Consumption and Physical Activity Levels during Alcohol Hangover. Journal of Clinical Medicine, 8(5), 752. https://doi.org/10.3390/jcm8050752






