Higher Serum Testosterone Levels Associated with Favorable Prognosis in Enzalutamide- and Abiraterone-Treated Castration-Resistant Prostate Cancer
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Selection and Clinical Variables
2.2. Definition of PSA Progression
2.3. Definition of High-Volume Tumor
2.4. Institutional Approval
2.5. Statistical Analysis
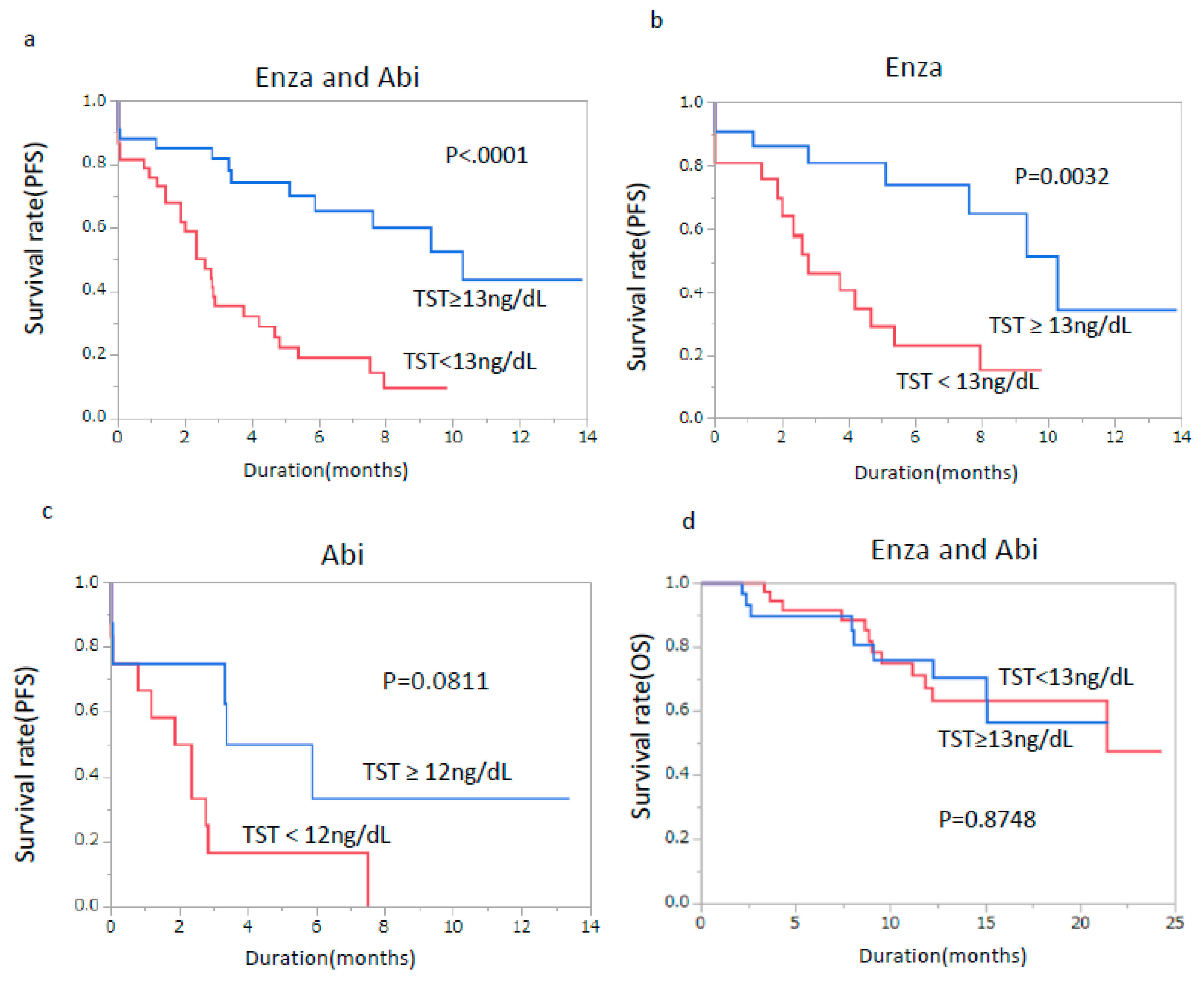
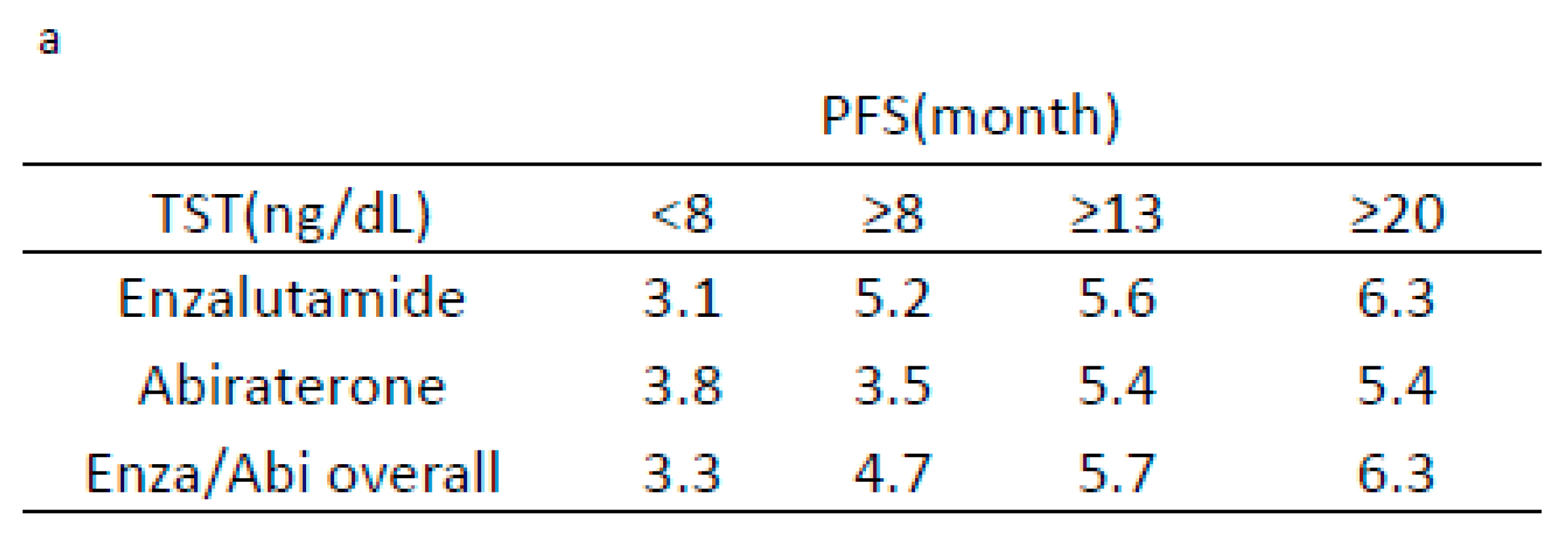
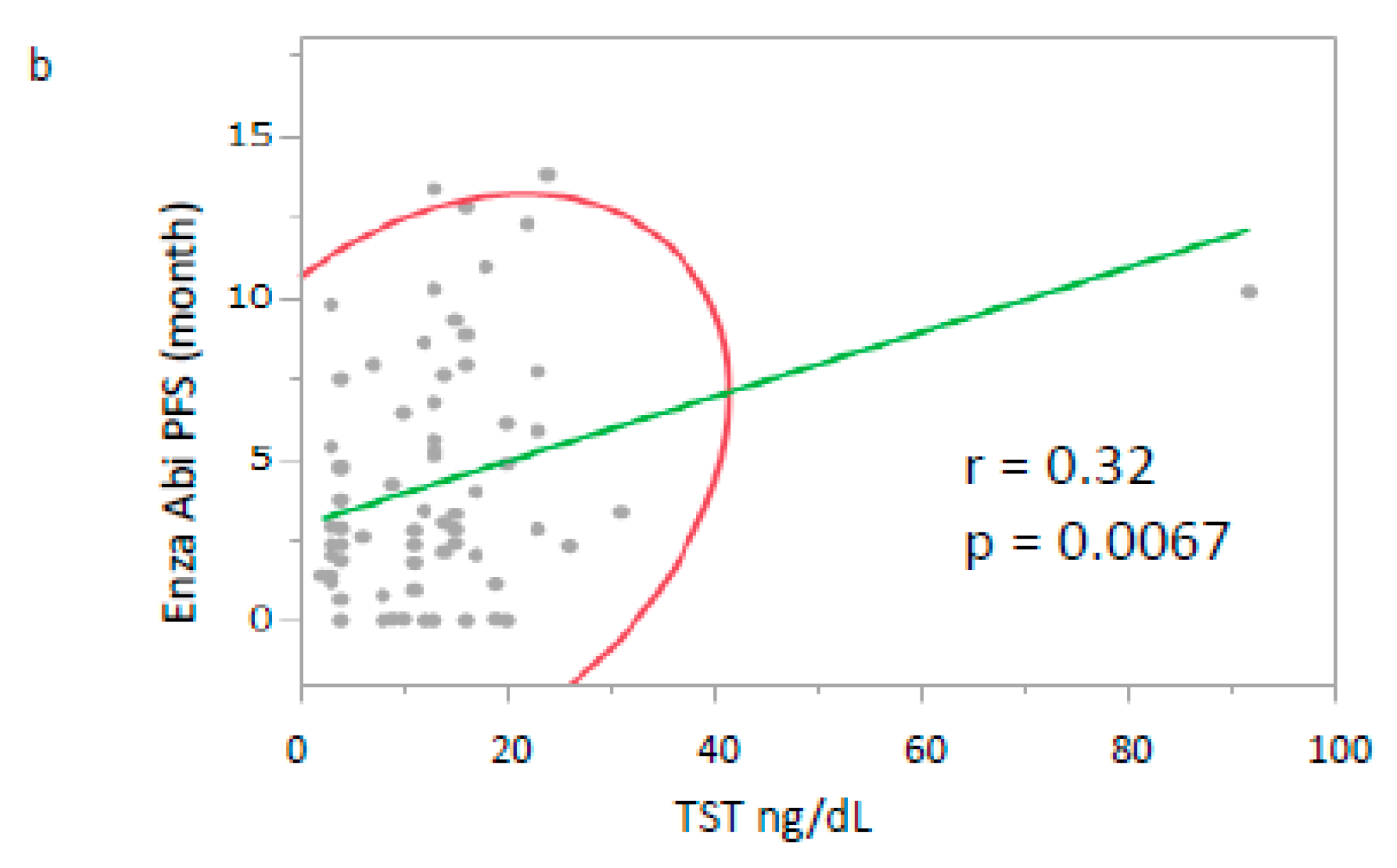
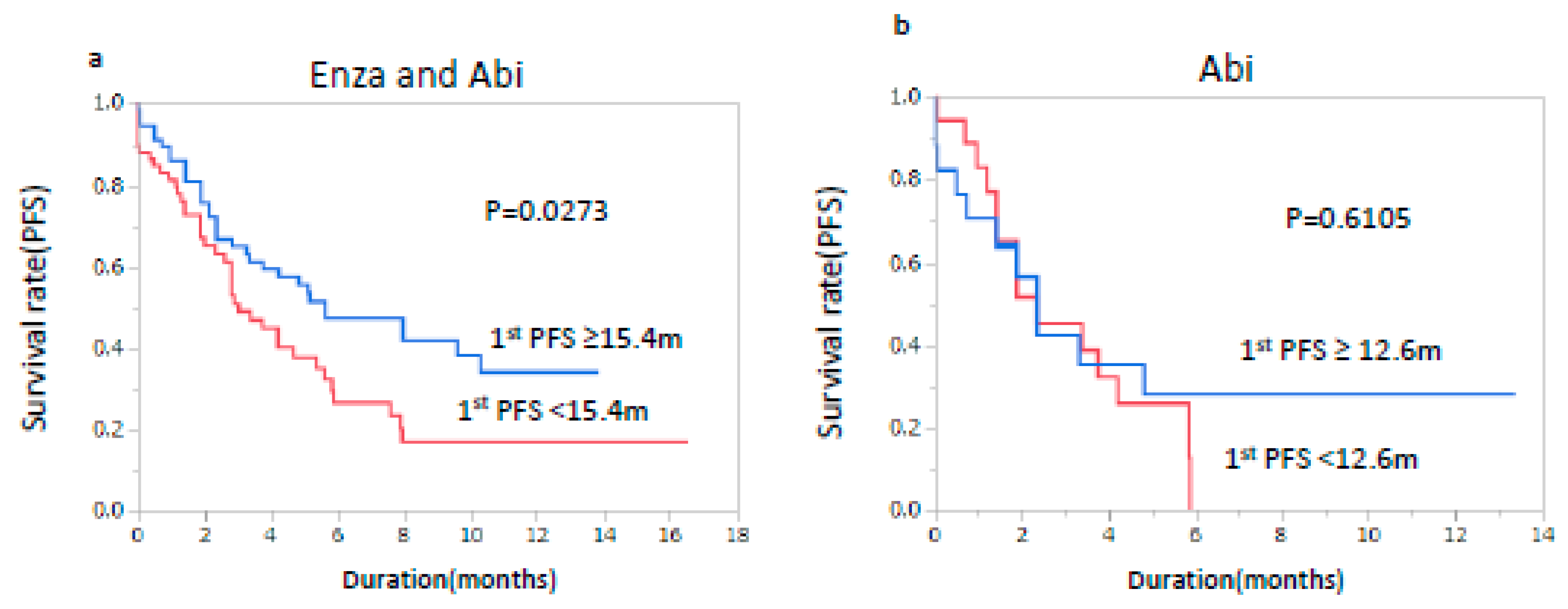
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| PSA | prostate-specific antigen |
| CRPC | castration-resistant prostate cancer |
| TST | testosterone |
| AR | androgen receptor |
| LH-RH | luteinizing hormone-releasing hormone |
| ALP | alkaline phosphatase |
| OS | overall survival |
| PFS | progression free survival |
| ADT | androgen deprivation therapy |
References
- Sakamoto, S.; McCann, R.O.; Dhir, R.; Kyprianou, N. Talin1 promotes tumor invasion and metastasis via focal adhesion signaling and anoikis resistance. Cancer Res. 2010, 70, 1885–1895. [Google Scholar] [CrossRef] [PubMed]
- Huggins, C.; Hodges, C.V. Studies on prostatic cancer. I. The effect of castration, of estrogen and androgen injection on serum phosphatases in metastatic carcinoma of the prostate. CA A Cancer J. Clin. 1972, 22, 232–240. [Google Scholar] [CrossRef]
- Gomella, L.G. Effective testosterone suppression for prostate cancer: Is there a best castration therapy? Rev. Urol. 2009, 11, 52–60. [Google Scholar]
- Kamada, S.; Sakamoto, S.; Ando, K.; Muroi, A.; Fuse, M.; Kawamura, K.; Imamoto, T.; Suzuki, H.; Nagata, M.; Nihei, N.; et al. Nadir testosterone after long-term followup predicts prognosis in patients with prostate cancer treated with combined androgen blockade. J. Urol. 2015, 194, 1264–1270. [Google Scholar] [CrossRef]
- Imamura, Y.; Sadar, M.D. Androgen receptor targeted therapies in castration-resistant prostate cancer: Bench to clinic. Int. J. Urol. 2016, 23, 654–665. [Google Scholar] [CrossRef]
- Sakamoto, S. Editorial comment to androgen receptor targeted therapies in castration-resistant prostate cancer: Bench to clinic. Int. J. Urol. 2016, 23, 666. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, S. Editorial comment to androgen receptor splice variant 7 in castration-resistant prostate cancer: Clinical considerations. Int. J. Urol. 2016, 23, 653. [Google Scholar] [CrossRef]
- Di Zazzo, E.; Galasso, G.; Giovannelli, P.; Di Donato, M.; Castoria, G. Estrogens and their receptors in prostate cancer: Therapeutic implications. Front. Oncol. 2018, 8, 2. [Google Scholar] [CrossRef] [PubMed]
- Di Zazzo, E.; Galasso, G.; Giovannelli, P.; Di Donato, M.; Di Santi, A.; Cernera, G.; Rossi, V.; Abbondanza, C.; Moncharmont, B.; Sinisi, A.A.; et al. Prostate cancer stem cells: The role of androgen and estrogen receptors. Oncotarget 2016, 7, 193–208. [Google Scholar] [CrossRef] [PubMed]
- Chi, K.; Hotte, S.J.; Joshua, A.M.; North, S.; Wyatt, A.W.; Collins, L.L.; Saad, F. Treatment of mCRPC in the AR-axis-targeted therapy-resistant state. Ann. Oncol. 2015, 26, 2044–2056. [Google Scholar] [CrossRef]
- Sweeney, C.J.; Chen, Y.H.; Carducci, M.; Liu, G.; Jarrard, D.F.; Eisenberger, M.; Wong, Y.N.; Hahn, N.; Kohli, M.; Cooney, M.M.; et al. Chemohormonal therapy in metastatic hormone-sensitive prostate cancer. N. Engl. J. Med. 2015, 373, 737–746. [Google Scholar] [CrossRef]
- Sakamoto, S. Editorial comment to current status of primary pharmacotherapy and future perspectives toward upfront therapy for metastatic hormone-sensitive prostate cancer. Int. J. Urol. 2016, 23, 370. [Google Scholar] [CrossRef][Green Version]
- Shiota, M.; Eto, M. Current status of primary pharmacotherapy and future perspectives toward upfront therapy for metastatic hormone-sensitive prostate cancer. Int. J. Urol. 2016, 23, 360–369. [Google Scholar] [CrossRef]
- James, N.D.; Sydes, M.R.; Clarke, N.W.; Mason, M.D.; Dearnaley, D.P.; Spears, M.R.; Ritchie, A.W.; Parker, C.C.; Russell, J.M.; Attard, G.; et al. Addition of docetaxel, zoledronic acid, or both to first-line long-term hormone therapy in prostate cancer (STAMPEDE): Survival results from an adaptive, multiarm, multistage, platform randomised controlled trial. Lancet 2016, 387, 1163–1177. [Google Scholar] [CrossRef]
- Fizazi, K.; Tran, N.; Fein, L.; Matsubara, N.; Rodriguez-Antolin, A.; Alekseev, B.Y.; Ozguroglu, M.; Ye, D.; Feyerabend, S.; Protheroe, A.; et al. Abiraterone plus prednisone in metastatic, castration-sensitive prostate cancer. N. Engl. J. Med. 2017, 377, 352–360. [Google Scholar] [CrossRef]
- James, N.D.; de Bono, J.S.; Spears, M.R.; Clarke, N.W.; Mason, M.D.; Dearnaley, D.P.; Ritchie, A.W.S.; Amos, C.L.; Gilson, C.; Jones, R.J.; et al. Abiraterone for prostate cancer not previously treated with hormone therapy. N. Engl. J. Med. 2017, 377, 338–351. [Google Scholar] [CrossRef]
- Ryan, C.J.; Molina, A.; Li, J.; Kheoh, T.; Small, E.J.; Haqq, C.M.; Grant, R.P.; de Bono, J.S.; Scher, H.I. Serum androgens as prognostic biomarkers in castration-resistant prostate cancer: Results from an analysis of a randomized phase III trial. J. Clin. Oncol. 2013, 31, 2791–2798. [Google Scholar] [CrossRef]
- Knudsen, K.E.; Penning, T.M. Partners in crime: Deregulation of AR activity and androgen synthesis in prostate cancer. Trends Endocrinol. Metab. 2010, 21, 315–324. [Google Scholar] [CrossRef]
- Morote, J.; Orsola, A.; Planas, J.; Trilla, E.; Raventos, C.X.; Cecchini, L.; Catalan, R. Redefining clinically significant castration levels in patients with prostate cancer receiving continuous androgen deprivation therapy. J. Urol. 2007, 178, 1290–1295. [Google Scholar] [CrossRef]
- Bertaglia, V.; Tucci, M.; Fiori, C.; Aroasio, E.; Poggio, M.; Buttigliero, C.; Grande, S.; Saini, A.; Porpiglia, F.; Berruti, A. Effects of serum testosterone levels after 6 months of androgen deprivation therapy on the outcome of patients with prostate cancer. Clin. Genitourin. Cancer 2013, 11, 325–330. [Google Scholar] [CrossRef]
- Klotz, L.; O’Callaghan, C.; Ding, K.; Toren, P.; Dearnaley, D.; Higano, C.S.; Horwitz, E.; Malone, S.; Goldenberg, L.; Gospodarowicz, M.; et al. Nadir testosterone within first year of androgen-deprivation therapy (ADT) predicts for time to castration-resistant progression: A secondary analysis of the PR-7 trial of intermittent versus continuous ADT. J. Clin. Oncol. 2015, 33, 1151–1156. [Google Scholar] [CrossRef]
- Yamamoto, S.; Sakamoto, S.; Minhui, X.; Tamura, T.; Otsuka, K.; Sato, K.; Maimaiti, M.; Kamada, S.; Takei, A.; Fuse, M.; et al. Testosterone reduction of >/= 480 ng/dL predicts favorable prognosis of japanese men with advanced prostate cancer treated with androgen-deprivation therapy. Clin. Genitourin. Cancer 2017, 15, e1107–e1115. [Google Scholar] [CrossRef]
- Loriot, Y.; Eymard, J.C.; Patrikidou, A.; Ileana, E.; Massard, C.; Albiges, L.; Di Palma, M.; Escudier, B.; Fizazi, K. Prior long response to androgen deprivation predicts response to next-generation androgen receptor axis targeted drugs in castration resistant prostate cancer. Eur. J. Cancer 2015, 51, 1946–1952. [Google Scholar] [CrossRef]
- Bellmunt, J.; Kheoh, T.; Yu, M.K.; Smith, M.R.; Small, E.J.; Mulders, P.F.; Fizazi, K.; Rathkopf, D.E.; Saad, F.; Scher, H.I.; et al. Prior endocrine therapy impact on abiraterone acetate clinical efficacy in metastatic castration-resistant prostate cancer: Post-hoc analysis of randomised phase 3 studies. Eur. Urol. 2016, 69, 924–932. [Google Scholar] [CrossRef]
- Tran, C.; Ouk, S.; Clegg, N.J.; Chen, Y.; Watson, P.A.; Arora, V.; Wongvipat, J.; Smith-Jones, P.M.; Yoo, D.; Kwon, A.; et al. Development of a second-generation antiandrogen for treatment of advanced prostate cancer. Science 2009, 324, 787–790. [Google Scholar] [CrossRef]
| Value | Range/% | |
|---|---|---|
| Enza as initial therapy | 82 | |
| Abi as initial therapy | 25 | Total 107 |
| Enza as second-line therapy | 7 | |
| Abi as second-line therapy | 21 | Total 28 |
| Median age | 73.0 | 54-88 |
| Median BMI (kg/m2) | 23.4 | 16.09-34.06 |
| Median TST at biopsy (ng/dL) | 457.5 | 228-847 |
| Median PSA at biopsy (ng/mL) | 79.5 | 3.43-15332 |
| Median PSA at Enza/Abi/total (ng/mL) | 30.1/41.1/34.1 | 0.59–5942.62/3.52–13296/0.59–13296 |
| Median TST at Enza/Abi/total (ng/dL) | 13/12/13 | 2–92/3–31/2–92 |
| Median previous treatment course number | 3 | 1 to 5 |
| Median EOD score 0/1/2/3/4 | 2 | 7/23/18/28/15 |
| Median follow-up period (month) | 68.3 | 11.81–241.60 |
| Median Enza/Abi PFS period (month) | 3.9/2.1 | 0–16.50/0–13.37 |
| Median first-line PFS (month) | 15.9 | 0.50–171.40 |
| Gleason Score sum (N) | ||
| ≤6 | 4 | 4.17% |
| 7 | 15 | 15.63% |
| 8 | 25 | 26.04% |
| ≥9 | 52 | 54.17% |
| Bone mets | 89 | 83.18% |
| Lymph mets | 34 | 31.78% |
| Lung mets | 9 | 8.41% |
| Liver mets | 6 | 5.61% |
| No mets | 12 | 11.21% |
| Patients who died | 23 | 21.50% |
| Pre-/post-docetaxel | 43/64 | 40.19/59.81% |
| Steroid use | 43 | 40.19% |
| Estramustine use | 50 | 46.73% |
| Enzalutamide dose | 160 mg/80 mg | 84/5 |
| Abiraterone dose | 1000 mg/750 mg | 45/1 |
| Univariate Analysis | Multivariate Analysis | ||||||
|---|---|---|---|---|---|---|---|
| Cut off | HR | COI | P | HR | COI | P | |
| Age | 72 | 0.75 | 0.48–1.16 | 0.1932 | |||
| GS | 9 | 0.93 | 0.59–1.48 | 0.7654 | |||
| First-line PFS (m) | 15.4 | 0.60 | 0.37–0.95 | 0.0309 | 0.85 | 0.215–3.25 | 0.8126 |
| Previous docetaxel | +/− | 2.44 | 1.575–3.81 | <0.0001 | 1.50 | 0.43–5.36 | 0.5222 |
| Liver mets | +/− | 1.69 | 0.70–3.46 | 0.2186 | |||
| Visceral mets | +/− | 1.51 | 0.83–2.58 | 0.1682 | |||
| Lymph mets | 0 | 1.67 | 1.07–2.63 | 0.0249 | 2.32 | 0.71–7.90 | 0.1607 |
| High volume | +/− | 1.73 | 1.09–2.83 | 0.0198 | 2.47 | 0.45–13.74 | 0.2927 |
| EOD score | 2 | 2.79 | 1.52–5.47 | 0.0004 | 2.79 | 0.65–14.57 | 0.1721 |
| ALP (ng/dL) | 254 | 1.20 | 0.78–1.86 | 0.4057 | |||
| ICTP (ng/mL) | 6.6 | 1.40 | 0.71–2.78 | 0.3307 | |||
| Hb (g/dL) | 11.9 | 0.65 | 0.42–1.01 | 0.0538 | |||
| LDH (mg/dL) | 212 | 0.98 | 0.64–1.52 | 0.9446 | |||
| Alb (g/dL) | 3.9 | 0.85 | 0.54–1.35 | 0.5001 | |||
| CRP (mg/dL) | 0.15 | 1.87 | 1.11–3.30 | 0.0173 | 0.67 | 0.17–3.10 | 0.5871 |
| NLR (ng/dL) | 2.6 | 1.13 | 0.66–1.94 | 0.6593 | |||
| PSA (ng/mL) | 34.1 | 1.73 | 1.12–2.71 | 0.0143 | 1.24 | 0.44–3.66 | 0.6899 |
| TST (ng/dL) | 13 | 0.26 | 0.13–0.51 | <0.0001 | 0.31 | 0.10–0.93 | 0.0365 * |
| Steroid use | +/− | 2.24 | 1.44–3.54 | 0.0003 | 1.10 | 0.40–2.95 | 0.8484 |
| Estramustine use | +/− | 1.69 | 1.09–2.65 | 0.0179 | 1.41 | 0.46–4.39 | 0.5442 |
| TST < 13 | TST ≥ 13 | P-Value | |
|---|---|---|---|
| Median (Average) | Median (Average) | ||
| Age | 71.50(68.74) | 70.00(70.88) | 0.2298 † |
| 1st-line PFS (month) | 29.40(36.33) | 14.63(32.755) | 0.3576 †† |
| Pre-docetaxel | 42.11%(16/38) | 23.53%(8/34) | 0.1336 ††† |
| GS≥9 | 41.94%(13/31) | 50%(16/30) | 0.6159 ††† |
| Lymph mets | 54.29%(19/35) | 38.24%(13/34) | 0.2301 ††† |
| Bone mets | 92.11%(35/38) | 88.24%(30/34) | 0.7002 ††† |
| Liver mets | 5.56%(2/36) | 14.71%(5/34) | 0.2533 ††† |
| Visceral mets | 8.33%(3/36) | 23.53%(8/34) | 0.1062 ††† |
| High volume | 65.71%(23/35) | 50.00%(17/34) | 0.2270 ††† |
| EOD score | 2(2.24) | 2(1.97) | 0.3965 † |
| BSI | 0.48(2.16) | 0.59(2.33) | 0.9342 †† |
| ALP (ng/dL) | 209(647.87) | 263.50(603.50) | 0.0158 *,†† |
| PSA (ng/mL) | 33.34(710.73) | 39.66(110.72) | 0.8391 †† |
| CRP (mg/dL) | 0.42(1.09) | 0.50(1.53) | 0.7866 †† |
| PSA at biopsy (ng/mL) | 58.57(473.01) | 72.95(563.79) | 0.3903 †† |
| TST at biopsy (ng/dL) | 4.42(4.22) | 3.41(3.32) | 0.1544 † |
| TST nadir at 1st line (ng/dL) | 9(38.69) | 12(13.23) | 0.8974 †† |
| Univariate Analysis | Multivariate Analysis | ||||||
|---|---|---|---|---|---|---|---|
| Cut off | HR | COI | P | HR | COI | P | |
| Age | 72 | 0.94 | 0.54–1.66 | 0.8371 | |||
| GS | 9 | 0.81 | 0.45–1.46 | 0.4801 | |||
| First-line PFS (m) | 15 | 0.70 | 0.36–1.23 | 0.2134 | |||
| Previous docetaxel | +/− | 2.38 | 1.35–4.31 | 0.0025 | 3.04 | 1.41–7.175 | 0.0038 * |
| Liver mets | +/− | 4.35 | 1.87–8.92 | 0.0014 | 0.35 | 0.07–1.905 | 0.2079 |
| EOD score | 2 | 2.87 | 1.305–7.56 | 0.0069 | 1.64 | 0.56–5.58 | 0.3778 |
| Visceral mets | +/− | 2.90 | 1.45–5.40 | 0.0038 | 7.88 | 1.62–29.52 | 0.0139 * |
| Lymph mets | +/− | 2.13 | 1.21–3.80 | 0.0091 | 1.60 | 0.77–3.29 | 0.2041 |
| High volume | +/− | 2.65 | 1.38–5.63 | 0.0028 | 1.60 | 0.64–4.50 | 0.3266 |
| ALP (ng/dL) | 254 | 3.04 | 1.71–5.66 | 0.0001 | 2.51 | 1.25–5.265 | 0.0090 * |
| ICTP (ng/mL) | 6.6 | 2.26 | 1.87–6.51 | 0.0941 | |||
| Hb (g/dL) | 11.9 | 0.49 | 0.27–0.85 | 0.0109 | 0.87 | 0.39–1.86 | 0.7144 |
| LDH (mg/dL) | 212 | 1.92 | 1.10–3.43 | 0.0210 | 2.95 | 1.43–6.40 | 0.0033 * |
| Alb (g/dL) | 3.9 | 0.61 | 0.33–1.10 | 0.0990 | |||
| CRP (mg/dL) | 0.15 | 1.93 | 0.97–4.28 | 0.0607 | |||
| NLR (ng/dL) | 2.6 | 1.93 | 0.93–4.18 | 0.0790 | |||
| PSA (ng/mL) | 34.1 | 3.11 | 1.72–5.97 | 0.0001 | 1.76 | 0.88–3.69 | 0.1101 |
| TST (ng/dL) | 13 | 0.99 | 0.44–2.55 | 0.8750 | |||
| Steroid use | +/− | 1.11 | 0.64–1.95 | 0.7011 | |||
| Estramustine use | +/− | 0.95 | 0.55–1.66 | 0.8567 | |||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sakamoto, S.; Maimaiti, M.; Xu, M.; Kamada, S.; Yamada, Y.; Kitoh, H.; Matsumoto, H.; Takeuchi, N.; Higuchi, K.; Uchida, H.A.; et al. Higher Serum Testosterone Levels Associated with Favorable Prognosis in Enzalutamide- and Abiraterone-Treated Castration-Resistant Prostate Cancer. J. Clin. Med. 2019, 8, 489. https://doi.org/10.3390/jcm8040489
Sakamoto S, Maimaiti M, Xu M, Kamada S, Yamada Y, Kitoh H, Matsumoto H, Takeuchi N, Higuchi K, Uchida HA, et al. Higher Serum Testosterone Levels Associated with Favorable Prognosis in Enzalutamide- and Abiraterone-Treated Castration-Resistant Prostate Cancer. Journal of Clinical Medicine. 2019; 8(4):489. https://doi.org/10.3390/jcm8040489
Chicago/Turabian StyleSakamoto, Shinichi, Maihulan Maimaiti, Minhui Xu, Shuhei Kamada, Yasutaka Yamada, Hiroki Kitoh, Hiroaki Matsumoto, Nobuyoshi Takeuchi, Kosuke Higuchi, Haruhito A. Uchida, and et al. 2019. "Higher Serum Testosterone Levels Associated with Favorable Prognosis in Enzalutamide- and Abiraterone-Treated Castration-Resistant Prostate Cancer" Journal of Clinical Medicine 8, no. 4: 489. https://doi.org/10.3390/jcm8040489
APA StyleSakamoto, S., Maimaiti, M., Xu, M., Kamada, S., Yamada, Y., Kitoh, H., Matsumoto, H., Takeuchi, N., Higuchi, K., Uchida, H. A., Komiya, A., Nagata, M., Nakatsu, H., Matsuyama, H., Akakura, K., & Ichikawa, T. (2019). Higher Serum Testosterone Levels Associated with Favorable Prognosis in Enzalutamide- and Abiraterone-Treated Castration-Resistant Prostate Cancer. Journal of Clinical Medicine, 8(4), 489. https://doi.org/10.3390/jcm8040489







