1. Introduction
Alcohol remains a first-order global health problem, with 15 million affected people in the EU [
1]. Recent publications in the US also warn about an increase in the prevalence of alcohol use disorders during the last decade [
2]. The impact it has on both individuals and society is of an enormous dimension, both medically and economically [
3,
4]. Several strategies have been applied to decrease the burden of the problem, ranging from public health intervention to individualized psychosocial and pharmacological treatment.
In the arena of pharmacological treatments, one of the latest incorporations has been nalmefene. The lack of real-world data, the need to assess adverse events in clinical populations, and the critiques surrounding the drug since its approval led to the need of a phase-IV trial. A single-arm, observational, phase-IV study of nalmefene, including 110 patients, was started in four different sites in Barcelona, Spain, in 2015.
Baseline and one month results [
5] suggested nalmefene was effective in reducing alcohol consumption. They also showed that it was well-tolerated with no serious adverse events, and the satisfaction with the drug of both professionals and patients was high.
The study, which was designed to last 12 months, consisted of four assessment points in time (baseline, 1, 6 and 12 months). In this paper we report the results of the main effectiveness analysis which was scheduled at 6 months.
2. Methods
A full description of the methods has been described elsewhere [
5]. As a summary, this was an observational, multisite, single-arm, phase-IV study, conducted among adult alcohol-dependent outpatients taking nalmefene for the first time. The study consisted of 4 visits: Baseline, 4 weeks, 6 months, and 12 months.
The main outcome variables of the study were:
(1) Reduction in drinking parameters, measured as a change from baseline in heavy drinking days and total alcohol consumption (units in the previous 28 days). The data were derived from the previous month’s timeline follow-back. As the study was conducted in Spain, one drink was considered to contain 10 g of pure ethanol.
(2) Patient and clinician satisfaction, as measured by the Medication Satisfaction Questionnaire (MSQ) [
6,
7]. Secondary outcomes included changes in drinking risk-level according to the WHO definitions (very high-risk: More than 100 g of alcohol per day in men and more than 60 in women; high-risk: 60–100 g per day in men, 40–60 g per day in women; medium-risk: 40–60 g per day in men, 20–40 g per day in women; low-risk: 1–40 g per day in men, 1–20 g per day in women). Liver enzymes were also analyzed.
Other collected variables at baseline included previous history of drug use, psychiatric history, family history of drug and alcohol use, and concomitant or changes in psychiatric medication during the study period. At the study visit, the number of days that patients took nalmefene was also recorded.
Responders to medication were defined in the same manner as in the 1-month publication (a reduction in daily alcohol consumption of at least 70% or downshift of two categories in the drinking risk-level, according to the World Health Organization (WHO), or a shift to low-risk category)
For effectiveness analysis, the repeated measures linear mixed procedure was used for both heavy-drinking days and total alcohol consumption as main outcomes. Age, sex, and number of days taking nalmefene were entered as fixed effects. We also included the presence of any psychiatric or addictive comorbidity as covariates. For each main outcome, regression coefficients (b), t-values and p-values were calculated. Statistical significance was set at 0.05. Missing data at 6 months for outcome variables was addressed with the conservative approach of baseline observation carried forward (BOCF). Days of medication intake were imputed to 0 for missing values. A descriptive analysis of the Medication Satisfaction Questionnaire (MSQ) was conducted. A satisfaction analysis and a logistic regression analysis for the detection of significant predictors of medication responders were conducted. Included variables were sex, age, number of days taking nalmefene, presence of comorbid drug use, and presence of psychiatric comorbidity. A descriptive analysis of adverse events was also conducted.
The study protocol, final approved informed consent document, and all supporting information were submitted to and approved by the institutional review boards of all participating centers. All participants provided written informed consent before taking part in study procedures. The study was conducted in accordance with the International Conference on Harmonization and Good Clinical Practice and the principles of the Declaration of Helsinki.
3. Results
3.1. Sample
From a total of 110 patients included at baseline, 47 were lost to the follow-up at six months, leaving a total of 63 patients reporting drinking outcomes at six months. Of these, 34 patients were still taking nalmefene. Accordingly, drug interruption was reported by 29 patients (eight due to low efficacy, four due to the achievement of the reduction aim, one due to adverse reactions, two due to the change to an abstinence aim, and 14 due to other reasons). No overdoses were reported. Of the 34 patients still on nalmefene, complete abstinence was reported by six subjects.
In patients still on nalmefene, the mean number of days of patients taking the drug over the previous month was 19.3 (SD = 11.6). More than half of the patients (57%) took it on a daily basis. Basal characteristics of participants can be observed in
Table 1.
3.2. Efficacy
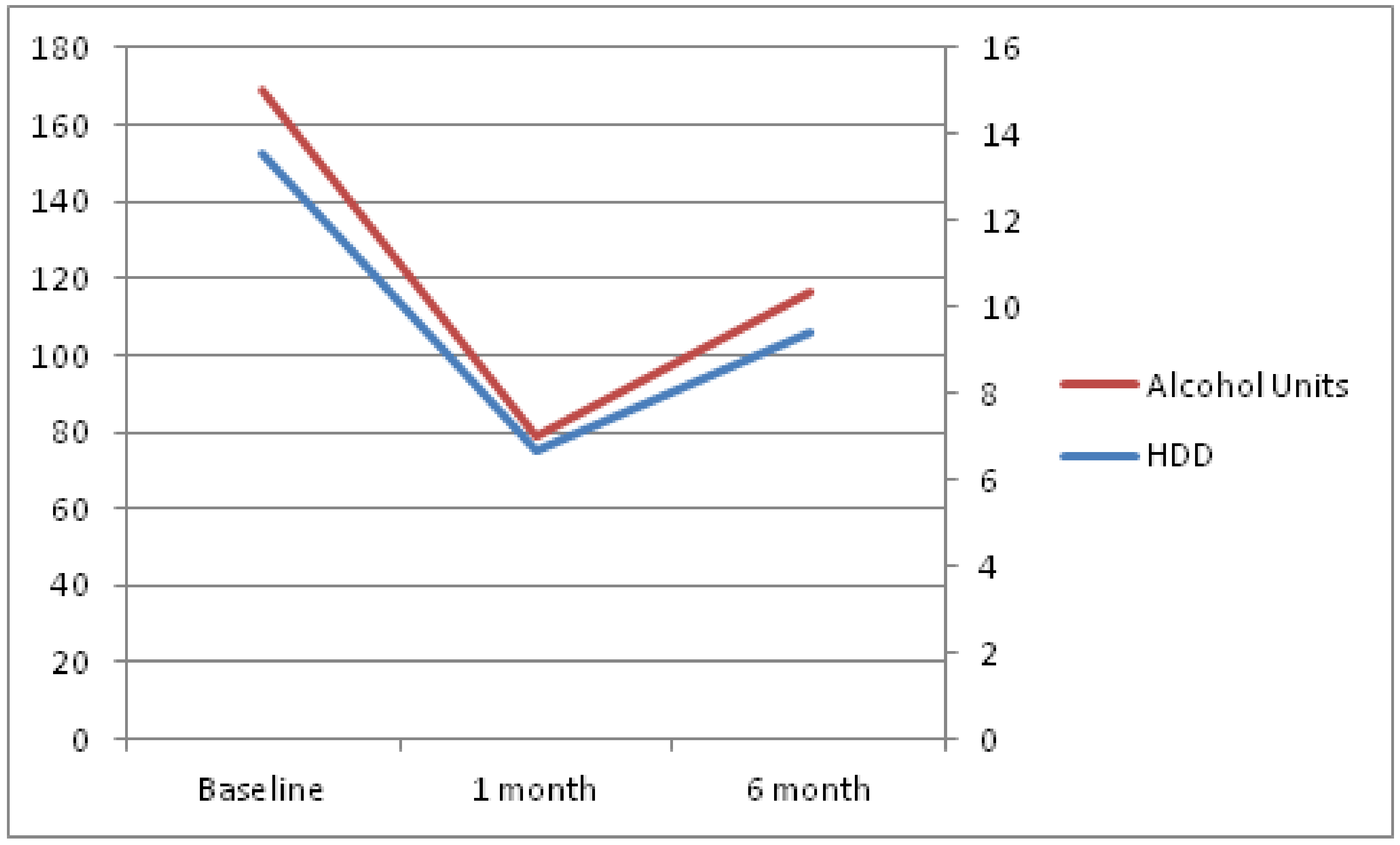
Both drinking outcomes increased, compared to month 1, but were still significantly reduced when compared to baseline values. The mean number of heavy-drinking days over the last 4 weeks was 9.4 (SD = 10.8) and the total alcohol consumption in units, also over the last 4 weeks, was 116.4 (SD = 171.8). Evolution over time of these parameters can be seen in
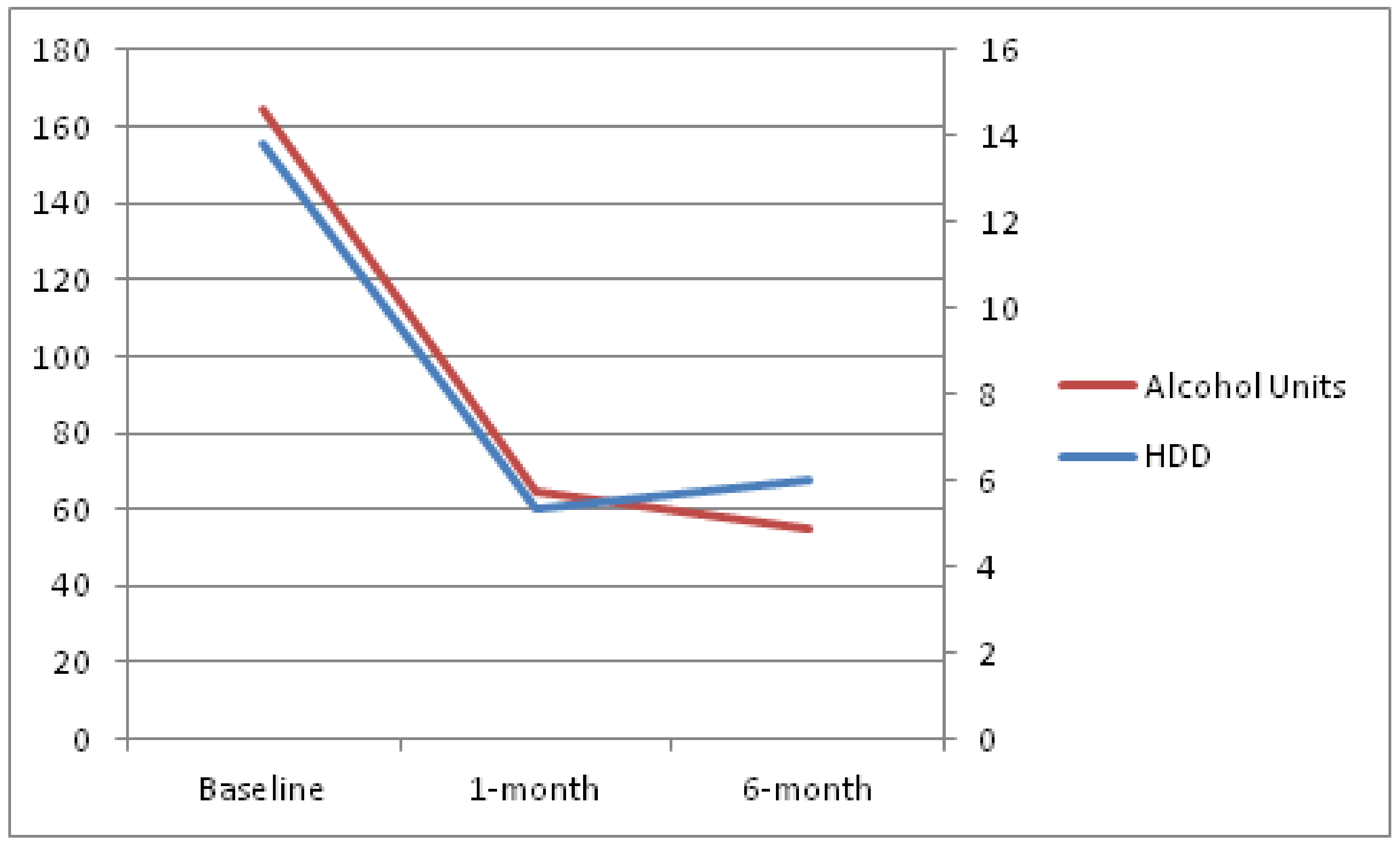
Figure 1. As a sensitivity analysis, evolution over time, according to per protocol analysis, is provided in
Figure 2. Per protocol analysis was conducted using only available data, that is, patients lost to follow-up were not included in this analysis.
The repeated measures linear mixed model revealed a significant effect in both outcomes at time 1 (first month). Given the increase in both outcomes at time 2 (month 6), no further significant changes were observed. The rest of the covariates were not statistically significant.
A total of 23 patients (21%) were considered medication responders. The only significant predictor found was the number of days with medication intake (OR = 1.058, CI 95% 1.001–1.118).
3.3. Satisfaction
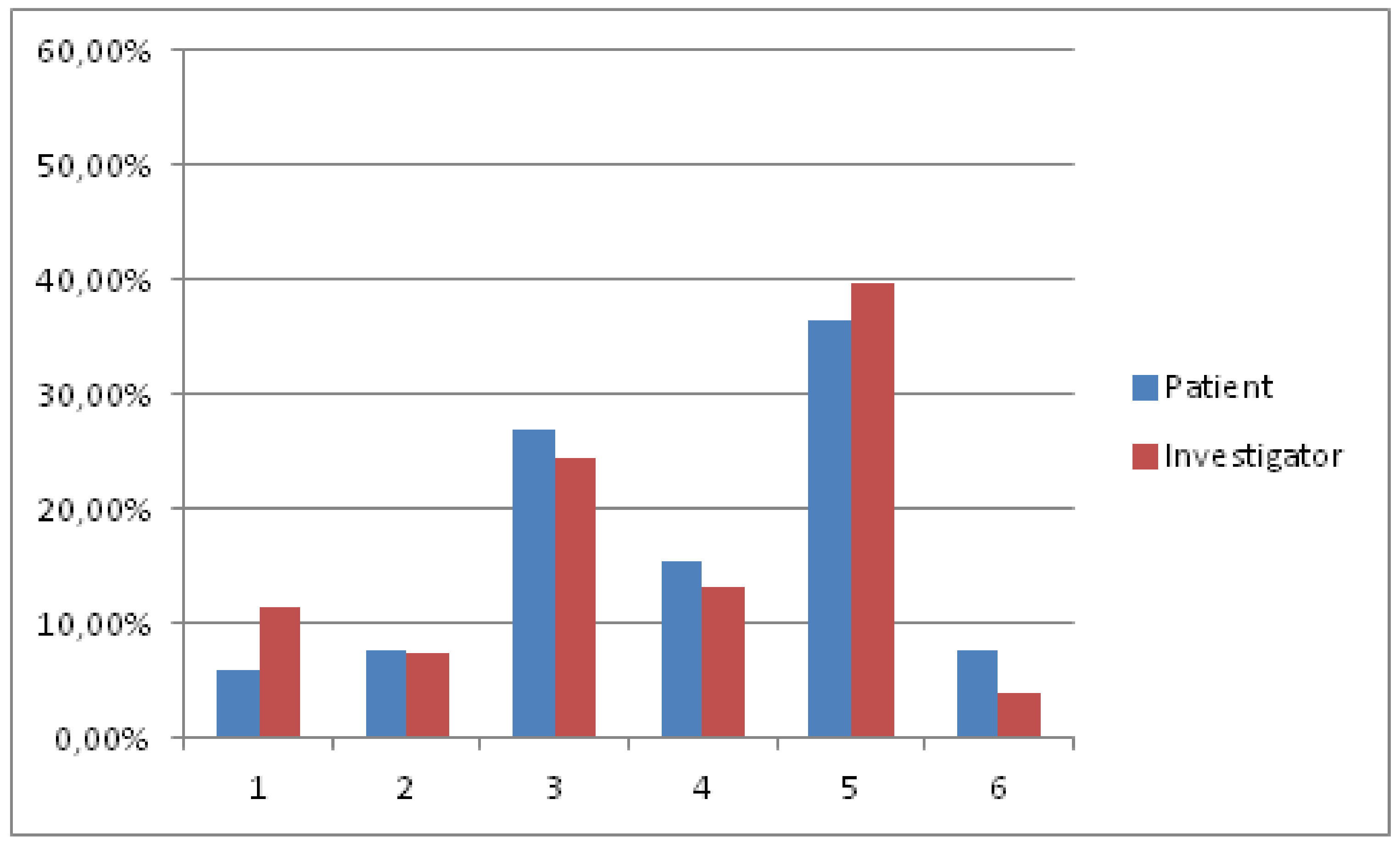
Figure 3 displays the satisfaction of professionals and patients with nalmefene at six months, as recorded by the MSQ. In the logistic regression analysis, the the number of days taking study medication revealed a significant effect upon patient satisfaction (OR = 1.074, CI 95% 1.021–1.130).
3.4. Safety
At six months, no new drug-related adverse events were notified. That left a total of 29 patients with medication-related adverse events during the first month of treatment. Most events were mild, and no serious adverse events were recorded. Additionally, no overdose was observed or notified.
4. Discussion
The main 6 month effectiveness analysis of this phase-IV trial suggests that nalmefene is effective in reducing alcohol-use when used in real-world, clinical settings. Similar to the 1 month results, nalmefene was well-tolerated and no significant, severe, or life-threatening reactions were observed. There are, however, relevant observations to be made in comparison with previous 1 month results.
Both heavy-drinking days and total alcohol consumption increased for the whole sample, suggesting nalmefene loses some efficacy over time. It is important to bear in mind that this is, in fact, a common phenomenon to many addiction treatments, whether pharmacological or psychosocial. On the other hand, we took the conservative approach of baseline observation carried forward (BOCF) to deal with missing data, a fact that could have decreased our statistical power.
It is important to note the relatively small number of patients still taking nalmefene at six months (31% approximately). While low-efficacy and adverse reactions might explain an important share of medication drop-outs, it is also possible that patients who are offered nalmefene are more prone to treatment abandonment, since it has been shown that patients still drinking, and those who aim at reduction objectives, are at greater risk of treatment drop-outs [
8,
9,
10].
Taken together, we believe these observations should remind professionals that patients who are prescribed nalmefene are especially prone to abandon treatment, and that efforts should also be directed toward increasing treatment retention.
Worth mentioning is a similar, recent study [
11] conducted among outpatients in routine settings that showed a significant decrease of drinking outcomes at 24 weeks. Similar to our sample, and other previous experimental studies with nalmefene [
12], psychiatric comorbidity was high. Interestingly, and contrary to our findings, improvements were seen over the six-month period. All taken together, recent evidence suggests that nalmefene is indeed effective for alcohol use disorder patients in routine settings, where comorbidity is frequent.
In trying to find differential characteristics between responders and non-responders to treatment, as measured by reductions in alcohol consumption parameters and changes in drinking risk categories, only the number of days taking nalmefene yielded significant effects.
Regarding satisfaction data, a slight decrease in comparison to one month results was observed, probably mimicking the decreased effectiveness at six months. Interestingly, the number of days taking medication was the only covariate associated with increased satisfaction. While it could be interpreted as a consequence, rather than a cause of increased satisfaction, and similar to the regression analysis, which was conducted in order to find predictors of treatment response, we believe this finding suggests that nalmefene might work better over the long-run if taken daily or with a high degree of frequency, rather than sporadically. Another hypothesis worth considering when analyzing this data could be that a higher degree of medication intake is, indeed, a reflection of higher motivation in patients. Therefore, the results obtained in this study are probably not to be entirely attributed to pharmacological effects. It is also fundamental to comment on the fact that we imputed as 0 the days of medication intake in the cases where this variable was missing. While we consider this imputation not unlikely, especially given that nalmefene requires ongoing medical prescription in Spain, it is also true that, in combination with the BOCF imputation for missing drinking outcomes, it could have biased the results obtained, regarding days of medication intake as a significant predictor of both medication response and satisfaction.
Finally, several other limitations apply to this study, such as its observational design and lack of control group, the reduced sample size, and the limited geographical area where the study was conducted.
5. Conclusions
Nalmefene seems to provide further effectiveness at 6 months, in spite of it being reduced, as compared to the first month, after initiating treatment. Our results suggest that a more frequent intake might be related to better outcomes, both in terms of satisfaction and reduced drinking.
Author Contributions
Conceptualization, A.G. and P.B.; methodology, A.G. and P.B.; formal analysis, P.B.; investigation, P.B., C.R., J.G., L.Y. and L.O.; writing—original draft preparation, P.B.; writing—review and editing, P.B.,C.R.,L.O., L.Y. and J.G.; funding acquisition, A.G.
Funding
This study was funded by Lundbeck. The sponsor was involved in the study design, but not in data collection, analysis, manuscript writing, or decision of submitting the article for publication.
Conflicts of Interest
P.B., C.R., J.G., and A.G. have received honoraria from Lundbeck. P.B. has also received honoraria from Pfizer. C.R. has also received honoraria from Janssen-cilag, Otsuka, Server, GSK, Rovi, Astra, MSD and Sanofi. L.Y. L.O. have no conflict of interest to declare.
References
- Wittchen, H.U.; Jacobi, F.; Rehm, J.; Gustavsson, A.; Svensson, M.; Jönsson, B.; Olesen, J.; Allgulander, C.; Alonso, J.; Faravelli, C.; et al. The size and burden of mental disorders and other disorders of the brain in Europe 2010. Eur. Neuropsychopharmacol. 2011, 21, 655–679. [Google Scholar] [CrossRef] [PubMed]
- Grant, B.F.; Chou, S.P.; Saha, T.D.; Pickering, R.P.; Kerridge, B.T.; Ruan, W.J.; Huang, B.; Jung, J.; Zhang, H.; Fan, A.; et al. Prevalence of 12-Month Alcohol Use, High-Risk Drinking, and DSM-IV Alcohol Use Disorder in the United States, 2001–2002 to 2012–2013. JAMA Psychiatry 2017, 74, 911. [Google Scholar] [CrossRef] [PubMed]
- Barrio, P.; Reynolds, J.; García-Altés, A.; Gual, A.; Anderson, P. Social costs of illegal drugs, alcohol and tobacco in the European Union: A systematic review. Drug. Alcohol Rev. 2017. [Google Scholar] [CrossRef] [PubMed]
- Miquel, L.; Rehm, J.; Shield, K.D.; Vela, E.; Bustins, M.; Segura, L.; Colom, J.; Anderson, P.; Gual, A. Alcohol, tobacco and health care costs: A population-wide cohort study (n = 606947 patients) of current drinkers based on medical and administrative health records from Catalonia. Eur. J. Public Health 2018. [Google Scholar] [CrossRef] [PubMed]
- Barrio, P.; Ortega, L.; Guardia, J.; Roncero, C.; Yuguero, L.; Gual, A. Who Receives Nalmefene and How Does It Work in the Real World? A Single-Arm, Phase IV Study of Nalmefene in Alcohol Dependent Outpatients: Baseline and 1-Month Results. Clin. Drug. Investig. 2017. [Google Scholar] [CrossRef] [PubMed]
- Kalali, A. Patient Satisfaction with, and Acceptability of, Atypical Antipsychotics. Curr. Med. Res. Opin. 1999, 15, 135–137. [Google Scholar] [CrossRef] [PubMed]
- Vernon, M.K.; Revicki, D.A.; Awad, A.G.; Dirani, R.; Panish, J.; Canuso, C.M.; Grinspan, A.; Mannix, S.; Kalali, A.H. Psychometric evaluation of the Medication Satisfaction Questionnaire (MSQ) to assess satisfaction with antipsychotic medication among schizophrenia patients. Schizophr. Res. 2010, 118, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Haug, S.; Schaub, M.P. Treatment outcome, treatment retention, and their predictors among clients of five outpatient alcohol treatment centres in Switzerland. BMC Public Health 2016, 16, 581. [Google Scholar] [CrossRef] [PubMed]
- Haug, S.; Eggli, P.; Schaub, M.P. Drinking Goals and Their Association With Treatment Retention and Treatment Outcomes Among Clients in Outpatient Alcohol Treatment. Subst. Use Misuse 2016, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Meyer, A.; Wapp, M.; Strik, W.; Moggi, F. Association Between Drinking Goal and Alcohol Use One Year After Residential Treatment: A Multicenter Study. J. Addict. Dis. 2014, 33, 234–242. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Di Nicola, M.; De Filippis, S.; Martinotti, G.; De Risio, L.; Pettorruso, M.; De Persis, S.; Giovanni, A.; Maremmani, I.; di Giannantonio, M.; Janiri, L. Nalmefene in Alcohol Use Disorder Subjects with Psychiatric Comorbidity: A Naturalistic Study. Adv. Ther. 2017, 34, 1636–1649. [Google Scholar] [CrossRef] [PubMed]
- Van den Brink, W.; Sørensen, P.; Torup, L.; Mann, K.; Gual, A.; SENSE Study Group. Long-term efficacy, tolerability and safety of nalmefene as-needed in patients with alcohol dependence: A 1-year, randomised controlled study. J. Psychopharmacol. 2014, 28, 733–744. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).