Current State and Future Directions of Technology-Based Ecological Momentary Assessment and Intervention for Major Depressive Disorder: A Systematic Review
Abstract
1. Introduction
1.1. Ecological Momentary Assessment
1.2. Ecological Momentary Intervention
1.3. Objectives
2. Methods
2.1. Search Strategy
2.2. Selection Criteria
2.3. Quality Assessment and Data Abstraction
3. Results
3.1. Ecological Momentary Assessment in MDD
3.1.1. Electronic Devices and Use of Sensors
3.1.2. Sampling Methods
3.1.3. Compliance and Dropout Rates
3.1.4. Contribution of EMA to the Study of MDD
Recall Bias
Symptom Monitoring
Cortisol Secretion
Sleep Patterns
Physical Activity
Rumination
Affect and Emotional Reactivity
3.2. Ecological Momentary Intervention in MDD
3.2.1. General Overview of the Interventions
3.2.2. Effectiveness of the Intervention
3.2.3. Compliance and Dropout Rates
3.2.4. Participants’ Feedback and Satisfaction
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- World Health Organization. Depression and Other Common Mental Disorders: Global Health Estimates; World Health Organization: Geneva, Switzerland, 2017; pp. 1–24. [Google Scholar]
- Katon, W.; Ciechanowski, P. Impact of major depression on chronic medical illness. J. Psychosom. Res. 2002, 53, 859–863. [Google Scholar] [CrossRef]
- Simon, G.E. Social and economic burden of mood disorders. Biol. Psychiatry 2003, 54, 208–215. [Google Scholar] [CrossRef]
- Sullivan, L.E.; Fiellin, D.A.; O’Connor, P.G. The prevalence and impact of alcohol problems in major depression: A systematic review. Am. J. Med. 2005, 118, 330–341. [Google Scholar] [CrossRef] [PubMed]
- Swendsen, J.D.; Merikangas, K.R. The comorbidity of depression and substance use disorders. Clin. Psychol. Rev. 2000, 20, 173–189. [Google Scholar] [CrossRef]
- McConville, C.; Cooper, C. Mood variability and the intensity of depressive states. Curr. Psychol. 1996, 14, 329–338. [Google Scholar] [CrossRef]
- Peeters, F.; Berkhof, J.; Delespaul, P.; Rottenberg, J.; Nicolson, N.A. Diurnal mood variation in major depressive disorder. Emotion 2006, 6, 383–391. [Google Scholar] [CrossRef] [PubMed]
- Ben-Zeev, D.; Young, M.A.; Madsen, J.W. Retrospective recall of affect in clinically depressed individuals and controls. Cogn. Emot. 2009. [Google Scholar] [CrossRef]
- Chamberlain, S.R.; Sakakian, B.J. The neuropsychology of mood disorders. Curr. Psychiatry Rep. 2006, 8, 458–463. [Google Scholar] [CrossRef] [PubMed]
- Möller, H.J.; von Zerssen, D. Self-rating procedures in the evaluation of antidepressants: Review of the literature and results of our studies. Psychopathology 1995, 28, 291–306. [Google Scholar] [CrossRef]
- Gotlib, I.H.; Joormann, J. Cognition and Depression: Current Status and Future Directions. Annu. Rev. Clin. Psychol. 2010, 6, 285–312. [Google Scholar] [CrossRef]
- Csikszentmihalyi, M.; Larson, R. Validity and Reliability of the Experience- Sampling Method. J. Nerv. Ment. Dis. 1987, 175, 526–536. [Google Scholar] [CrossRef] [PubMed]
- Shiffman, S.; Stone, A.A.; Hufford, M.R. Ecological Momentary Assessment. Annu. Rev. Clin. Psychol. 2008, 4, 1–32. [Google Scholar] [CrossRef] [PubMed]
- Stone, A.A.; Shiffman, S.; Atienza, A.A.; Nebeling, A. Historical roots and rationale of ecological momentary assessment (EMA). In The Science of Real-Time Data Capture: Self-Reports in Health Research; Oxford University Press: Oxford, MI, USA, 2007; pp. 3–10. [Google Scholar]
- Aan het Rot, M.; Hogenelst, K.; Schoevers, R.A. Mood disorders in everyday life: A systematic review of experience sampling and ecological momentary assessment studies. Clin. Psychol. Rev. 2012, 32, 510–523. [Google Scholar] [CrossRef] [PubMed]
- Ebner-Priemer, U.W.; Trull, T.J. Ecological momentary assessment of mood disorders and mood dysregulation. Psychol. Assess. 2009, 21, 463–475. [Google Scholar] [CrossRef] [PubMed]
- Marzano, L.; Bardill, A.; Fields, B.; Herd, K.; Veale, D.; Grey, N.; Moran, P. The application of mHealth to mental health: Opportunities and challenges. Lancet Psychiatry 2015, 2, 942–948. [Google Scholar] [CrossRef]
- Mohr, D.C.; Zhang, M.; Schueller, S.M. Personal Sensing: Understanding Mental Health Using Ubiquitous Sensors and Machine Learning. Annu. Rev. Clin. Psychol. 2017, 13, 23–47. [Google Scholar] [CrossRef] [PubMed]
- van de Ven, P.; O’Brien, H.; Henriques, R.; Klein, M.; Msetfi, R.; Nelson, J.; Rocha, A.; Ruwaard, J.; O’Sullivan, D.; Riper, H. ULTEMAT: A mobile framework for smart ecological momentary assessments and interventions. Internet Interv. 2017, 9, 74–81. [Google Scholar] [CrossRef] [PubMed]
- Henderson, C.; Evans-Lacko, S.; Thornicroft, G. Mental illness stigma, help seeking, and public health programs. Am. J. Public Health 2013, 103, 777–780. [Google Scholar] [CrossRef] [PubMed]
- Kazdin, A.E.; Blase, S.L. Rebooting Psychotherapy Research and Practice to Reduce the Burden of Mental Illness. Perspect. Psychol. Sci. 2011, 6, 21–37. [Google Scholar] [CrossRef] [PubMed]
- Heron, K.E.; Smyth, J.M. Ecological momentary interventions: Incorporating mobile technology into psychosocial and health behaviour treatments. Br. J. Health Psychol. 2010, 15, 1–39. [Google Scholar] [CrossRef] [PubMed]
- Donker, T.; Petrie, K.; Proudfoot, J.; Clarke, J.; Birch, M.R.; Christensen, H. Smartphones for smarter delivery of mental health programs: A systematic review. J. Med. Internet Res. 2013, 15, e247. [Google Scholar] [CrossRef]
- Cuijpers, P.; Donker, T.; Van Straten, A.; Li, J.; Andersson, G. Is guided self-help as effective as face-to-face psychotherapy for depression and anxiety disorders? A systematic review and meta-analysis of comparative outcome studies. Psychol. Med. 2010, 40, 1943–1957. [Google Scholar] [CrossRef] [PubMed]
- Asada, H.H.; Shaltis, P.; Reisner, A.; Rhee, S.; Hutchinson, R.C. Mobile Monitoring with Wearable Photoplethysmographic Biosensors. IEEE Eng. Med. Biol. Mag. 2003, 22, 28–40. [Google Scholar] [CrossRef] [PubMed]
- Torous, J.; Friedman, R.; Keshvan, M. Smartphone ownership and interest in mobile applications to monitor symptoms of mental health conditions. J. Med. Internet Res. 2014, 16. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; PRISMA Group. Preferred reporting items for systematic reviews and meta analyses: The Prisma Statement. PLoS Med. 2009, 6, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Colombo, D.; Palacios, A.G.; Alvarez, J.F.; Patané, A.; Semonella, M.; Cipresso, P.; Kwiatkowska, M.; Riva, G.; Botella, C. Current state and future directions of technology-based ecological momentary assessments and interventions for major depressive disorder: Protocol for a systematic review. Syst. Rev. 2018, 7, 233. [Google Scholar] [CrossRef] [PubMed]
- Delgadillo, J.; de Jong, K.; Lucock, M.; Lutz, W.; Rubel, J.; Gilbody, S.; Ali, S.; Aguirre, E.; Appleton, M.; Nevin, J.; et al. Feedback-informed treatment versus usual psychological treatment for depression and anxiety: A multisite, open-label, cluster randomised controlled trial. Lancet Psychiatry 2018. [Google Scholar] [CrossRef]
- Downs, S.H.; Black, N. The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non-randomised studies of health care interventions. J. Epidemiol. Community Health 1998, 52, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Conrad, A.; Wilhelm, F.H.; Roth, W.T.; Spiegel, D.; Taylor, C.B. Circadian affective, cardiopulmonary, and cortisol variability in depressed and nondepressed individuals at risk for cardiovascular disease. J. Psychiatr. Res. 2008. [Google Scholar] [CrossRef]
- Kim, J.; Nakamura, T.; Kikuchi, H.; Yoshiuchi, K.; Sasaki, T.; Yamamoto, Y. Covariation of Depressive Mood and Spontaneous Physical Activity in Major Depressive Disorder: Toward Continuous Monitoring of Depressive Mood. IEEE J. Biomed. Heal. Inform. 2015, 19, 1347–1355. [Google Scholar] [CrossRef]
- Littlewood, D.L.; Kyle, S.D.; Carter, L.-A.; Peters, S.; Pratt, D.; Gooding, P. Short sleep duration and poor sleep quality predict next-day suicidal ideation: An ecological momentary assessment study. Psychol. Med. 2018. [Google Scholar] [CrossRef] [PubMed]
- Adams, Z.W.; McClure, E.A.; Gray, K.M.; Danielson, C.K.; Treiber, F.A.; Ruggiero, K.J. Mobile devices for the remote acquisition of physiological and behavioral biomarkers in psychiatric clinical research. J. Psychiatr. Res. 2017. [Google Scholar] [CrossRef] [PubMed]
- Ottaviani, C.; Shahabi, L.; Tarvainen, M.; Cook, I.; Abrams, M.; Shapiro, D. Cognitive, behavioral, and autonomic correlates of mind wandering and perseverative cognition in major depression. Front. Neurosci. 2015. [Google Scholar] [CrossRef] [PubMed]
- Booij, S.H.; Bos, E.H.; Bouwmans, M.E.J.; Van Faassen, M.; Kema, I.P.; Oldehinkel, A.J.; De Jonge, P. Cortisol and α-amylase secretion patterns between and within depressed and non-depressed individuals. PLoS ONE 2015. [Google Scholar] [CrossRef] [PubMed]
- Stavrakakis, N.; Booij, S.H.; Roest, A.M.; de Jonge, P.; Oldehinkel, A.J.; Bos, E.H. Temporal dynamics of physical activity and affect in depressed and nondepressed individuals. Health Psychol. 2015, 34S, 1268–1277. [Google Scholar] [CrossRef] [PubMed]
- Bouwmans, M.E.J.; Oude Oosterik, N.A.M.; Bos, E.H.; de Groot, I.W.; Oldehinkel, A.J.; de Jonge, P. The Temporal Order of Changes in Physical Activity and Subjective Sleep in Depressed Versus Nondepressed Individuals: Findings From the MOOVD Study. Behav. Sleep Med. 2018. [Google Scholar] [CrossRef] [PubMed]
- Husky, M.M.; Gindre, C.; Mazure, C.M.; Brebant, C.; Nolen-Hoeksema, S.; Sanacora, G.; Swendsen, J. Computerized ambulatory monitoring in mood disorders: Feasibility, compliance, and reactivity. Psychiatry Res. 2010. [Google Scholar] [CrossRef]
- Schaffer, A.; Kreindler, D.; Reis, C.; Levitt, A.J. Use of Mental Health Telemetry to Enhance Identification and Predictive Value of Early Changes During Augmentation Treatment of Major Depression. J. Clin. Psychopharmacol. 2013. [Google Scholar] [CrossRef] [PubMed]
- Hung, S.; Li, M.-S.; Chen, Y.-L.; Chiang, J.-H.; Chen, Y.-Y.; Hung, G.C.-L. Smartphone-based ecological momentary assessment for Chinese patients with depression: An exploratory study in Taiwan. Asian J. Psychiatr. 2016, 23, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Stetler, C.; Dickerson, S.S.; Miller, G.E. Uncoupling of social zeitgebers and diurnal cortisol secretion in clinical depression. Psychoneuroendocrinology 2004, 29, 1250–1259. [Google Scholar] [CrossRef]
- Stetler, C.; Miller, G.E. Blunted cortisol response to awakening in mild to moderate depression: Regulatory influences of sleep patterns and social contacts. J. Abnorm. Psychol. 2005, 114, 697–705. [Google Scholar] [CrossRef] [PubMed]
- Huffziger, S.; Ebner-Priemer, U.; Zamoscik, V.; Reinhard, I.; Kirsch, P.; Kuehner, C. Effects of mood and rumination on cortisol levels in daily life: An ambulatory assessment study in remitted depressed patients and healthy controls. Psychoneuroendocrinology 2013, 38, 2258–2267. [Google Scholar] [CrossRef] [PubMed]
- Booij, S.H.; Bos, E.H.; de Jonge, P.; Oldehinkel, A.J. The temporal dynamics of cortisol and affective states in depressed and non-depressed individuals. Psychoneuroendocrinology 2016. [Google Scholar] [CrossRef] [PubMed]
- Bower, B.; Bylsma, L.M.; Morris, B.H.; Rottenberg, J. Poor reported sleep quality predicts low positive affect in daily life among healthy and mood-disordered persons: Sleep quality and positive affect. J. Sleep Res. 2010, 19, 323–332. [Google Scholar] [CrossRef] [PubMed]
- O’Leary, K.; Small, B.J.; Panaite, V.; Bylsma, L.M.; Rottenberg, J. Sleep quality in healthy and mood-disordered persons predicts daily life emotional reactivity. Cogn. Emot. 2017, 31, 435–443. [Google Scholar] [CrossRef] [PubMed]
- Bouwmans, M.E.J.; Bos, E.H.; Hoenders, H.J.R.; Oldehinkel, A.J.; de Jonge, P. Sleep quality predicts positive and negative affect but not vice versa. An electronic diary study in depressed and healthy individuals. J. Affect. Disord. 2017, 207, 260–267. [Google Scholar] [CrossRef] [PubMed]
- Bouwmans, M.E.J.; Beltz, A.M.; Bos, E.H.; Oldehinkel, A.J.; de Jonge, P.; Molenaar, P.C.M. The person-specific interplay of melatonin, affect, and fatigue in the context of sleep and depression. Personal. Individ. Differ. 2018. [Google Scholar] [CrossRef]
- Thompson, R.J.; Mata, J.; Jaeggi, S.M.; Buschkuehl, M.; Jonides, J.; Gotlib, I.H. The everyday emotional experience of adults with major depressive disorder: Examining emotional instability, inertia, and reactivity. J. Abnorm. Psychol. 2012, 121, 819–829. [Google Scholar] [CrossRef] [PubMed]
- Putnam, K.M.; McSweeney, L.B. Depressive symptoms and baseline prefrontal EEG alpha activity: A study utilizing Ecological Momentary Assessment. Biol. Psychol. 2008. [Google Scholar] [CrossRef]
- Ruscio, A.M.; Gentes, E.L.; Jones, J.D.; Hallion, L.S.; Coleman, E.S.; Swendsen, J. Rumination Predicts Heightened Responding to Stressful Life Events in Major Depressive Disorder and Generalized Anxiety Disorder. J. Abnorm. Psychol. 2015, 124, 17–26. [Google Scholar] [CrossRef]
- Kircanski, K.; Thompson, R.J.; Sorenson, J.; Sherdell, L.; Gotlib, I.H. The everyday dynamics of rumination and worry: Precipitant events and affective consequences. Cogn. Emot. 2018, 32, 1424–1436. [Google Scholar] [CrossRef] [PubMed]
- Husky, M.M.; Mazure, C.M.; MacIejewski, P.K.; Swendsen, J.D. Past depression and gender interact to influence emotional reactivity to daily life stress. Cognit. Ther. Res. 2009, 33, 264–271. [Google Scholar] [CrossRef]
- Bylsma, L.M.; Taylor-Clift, A.; Rottenberg, J. Emotional reactivity to daily events in major and minor depression. J. Abnorm. Psychol. 2011. [Google Scholar] [CrossRef]
- Kohling, J.; Moessner, M.; Ehrenthal, J.C.; Bauer, S.; Cierpka, M.; Kammerer, A.; Schauenburg, H.; Dinger, U.; Köhling, J.; Moessner, M.; et al. Affective Instability and Reactivity in Depressed Patients With and Without Borderline Pathology. J. Personal. Disord. 2015, 30, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Slofstra, C.; Nauta, M.H.; Holmes, E.A.; Bos, E.H.; Wichers, M.; Batalas, N.; Klein, N.S.; Bockting, C.L.H. Exploring the relation between visual mental imagery and affect in the daily life of previously depressed and never depressed individuals. Cogn. Emot. 2018, 32, 1131–1138. [Google Scholar] [CrossRef] [PubMed]
- Hepp, J.; Lane, S.P.; Carpenter, R.W.; Niedtfeld, I.; Brown, W.C.; Trull, T.J. Interpersonal Problems and Negative Affect in Borderline Personality and Depressive Disorders in Daily Life. Clin. Psychol. Sci. 2017. [Google Scholar] [CrossRef] [PubMed]
- Quilty, L.C.; Watson, C.; Toneatto, T.; Bagby, R.M. A Prospective Investigation of Affect, the Desire to Gamble, Gambling Motivations and Gambling Behavior in the Mood Disorders. J. Gambl. Stud. 2017. [Google Scholar] [CrossRef] [PubMed]
- Fisher, A.J.; Reeves, J.W.; Lawyer, G.; Medaglia, J.D.; Rubel, J.A. Exploring the idiographic dynamics of mood and anxiety via network analysis. J. Abnorm. Psychol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Burns, M.N.; Begale, M.; Duffecy, J.; Gergle, D.; Karr, C.J.; Giangrande, E.; Mohr, D.C. Harnessing context sensing to develop a mobile intervention for depression. J. Med. Internet Res. 2011, 13. [Google Scholar] [CrossRef]
- Kramer, I.; Simons, C.J.P.; Hartmann, J.A.; Menne-Lothmann, C.; Viechtbauer, W.; Peeters, F.; Schruers, K.; van Bemmel, A.L.; Myin-Germeys, I.; Delespaul, P.; et al. A therapeutic application of the experience sampling method in the treatment of depression: A randomized controlled trial. World Psychiatry 2014, 13, 68–77. [Google Scholar] [CrossRef]
- Simons, C.J.P.; Hartmann, J.A.; Kramer, I.; Menne-Lothmann, C.; Höhn, P.; van Bemmel, A.L.; Myin-Germeys, I.; Delespaul, P.; van Os, J.; Wichers, M. Effects of momentary self-monitoring on empowerment in a randomized controlled trial in patients with depression. Eur. Psychiatry 2015. [Google Scholar] [CrossRef]
- Hartmann, J.A.; Wichers, M.; Menne-Lothmann, C.; Kramer, I.; Viechtbauer, W.; Peeters, F.; Schruers, K.R.J.; Van Bemmel, A.L.; Myin-Germeys, I.; Delespaul, P.; et al. Experience sampling-based personalized feedback and Positive affect: A randomized controlled trial in depressed patients. PLoS ONE 2015, 10. [Google Scholar] [CrossRef] [PubMed]
- Mohr, D.C.; Stiles-Shields, C.; Brenner, C.; Palac, H.; Montague, E.; Kaiser, S.M.; Carty-Fickes, E.; Duffecy, J. MedLink: A Mobile Intervention to Address Failure Points in the Treatment of Depression in General Medicine. Int. Conf. Pervasive Comput. Technol. Healthc. 2015, 2015, 100–107. [Google Scholar] [PubMed]
- Burton, C.; Szentagotai Tatar, A.; McKinstry, B.; Matheson, C.; Matu, S.; Moldovan, R.; Macnab, M.; Farrow, E.; David, D.; Pagliari, C.; et al. Pilot randomised controlled trial of Help4Mood, an embodied virtual agent-based system to support treatment of depression. J. Telemed. Telecare 2016, 22, 348–355. [Google Scholar] [CrossRef]
- Snippe, E.; Simons, C.J.P.; Hartmann, J.A.; Menne-Lothmann, C.; Kramer, I.; Booij, S.H.; Viechtbauer, W.; Delespaul, P.; Myin-Germeys, I.; Wichers, M. Change in daily life behaviors and depression: Within-person and between-person associations. Health Psychol. 2016, 35, 433–441. [Google Scholar] [CrossRef] [PubMed]
- Widdershoven, R.L.A.; Wichers, M.; Kuppens, P.; Hartmann, J.A.; Menne-Lothmann, C.; Simons, C.J.P.; Bastiaansen, J.A. Effect of self-monitoring through experience sampling on emotion differentiation in depression. J. Affect. Disord. 2019. [Google Scholar] [CrossRef] [PubMed]
- Torous, J.; Staples, P.; Shanahan, M.; Lin, C.; Peck, P.; Keshavan, M.; Onnela, J.-P. Utilizing a Personal Smartphone Custom App to Assess the Patient Health Questionnaire-9 (PHQ-9) Depressive Symptoms in Patients With Major Depressive Disorder. JMIR Ment. Health 2015, 2, e8. [Google Scholar] [CrossRef]
- Nilsen, W.J.; Pavel, M. Moving behavioral theories into the 21st century: Technological advancements for improving quality of life. IEEE Pulse 2013, 4, 25–28. [Google Scholar] [CrossRef]
- Ericsson Consumer Lab Eurupe Ericsson Mobility Report Appendix. Ericsson Mobil. Rep. 2015, 1–8. [CrossRef]
- Ben-Zeev, D.; Davis, K.E.; Kaiser, S.; Krzsos, I.; Drake, R.E. Mobile technologies among people with serious mental illness: Opportunities for future services. Adm. Policy Ment. Health 2013, 40, 340–343. [Google Scholar] [CrossRef]
- Colombo, D.; Cipresso, P.; Fernández Alvarez, J.; Garcia Palacios, A.; Riva, G.; Botella, C. An Overview of Factors Associated with Adherence and Dropout to Ecological Momentary Assessments in Depression. Annu. Rev. CyberTherapy Telemed. 2018, in press. [Google Scholar]
- Karyotaki, E.; Kleiboer, A.; Smit, F.; Turner, D.T.; Pastor, A.M.; Andersson, G.; Berger, T.; Botella, C.; Breton, J.M.; Carlbring, P.; et al. Predictors of treatment dropout in self-guided web-based interventions for depression: An “individual patient data” meta-analysis. Psychol. Med. 2015. [Google Scholar] [CrossRef] [PubMed]
- Kuppens, P.; Verduyn, P. Emotion dynamics. Curr. Opin. Psychol. 2017, 17, 22–26. [Google Scholar] [CrossRef] [PubMed]
- Boswell, J.F.; Kraus, D.R.; Miller, S.D.; Lambert, M.J. Implementing routine outcome monitoring in clinical practice: Benefits, challenges, and solutions. Psychother. Res. 2015. [Google Scholar] [CrossRef] [PubMed]
- Mikus, A.; Hoogendoorn, M.; Rocha, A.; Gama, J.; Ruwaard, J.; Riper, H. Predicting short term mood developments among depressed patients using adherence and ecological momentary assessment data. Internet Interv. 2018, 12, 105–110. [Google Scholar] [CrossRef]
- Nuij, C.; van Ballegooijen, W.; Ruwaard, J.; de Beurs, D.; Mokkenstorm, J.; van Duijn, E.; de Winter, R.F.P.; O’Connor, R.C.; Smit, J.H.; Riper, H.; et al. Smartphone-based safety planning and self-monitoring for suicidal patients: Rationale and study protocol of the CASPAR (Continuous Assessment for Suicide Prevention And Research) study. Internet Interv. 2018. [Google Scholar] [CrossRef] [PubMed]
- Saranummi, N.; Spruijt-Metz, D.; Intille, S.S.; Korhonen, I.; Nilsen, W.J.; Pavel, M. Moving the science of behavioral change into the 21st century: Part 2. IEEE Pulse 2013, 4, 32–33. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.; O’Neill, N.; van Woerden, H.; Eslambolchilar, P.; Jones, M.; John, A. Gamification and Adherence to Web-Based Mental Health Interventions: A Systematic Review. JMIR Ment. Health 2016, 3, e39. [Google Scholar] [CrossRef] [PubMed]
- Majumder, S.; Mondal, T.; Deen, M. Wearable Sensors for Remote Health Monitoring. Sensors 2017, 17, 130. [Google Scholar] [CrossRef] [PubMed]
- Aziz, O.; Atallah, L.; Lo, B.; ElHelw, M.; Wang, L.; Yang, G.Z.; Darzi, A. A pervasive body sensor network for measuring postoperative recovery at home. Surg. Innov. 2007. [Google Scholar] [CrossRef]
- Patel, S.; Park, H.; Bonato, P.; Chan, L.; Rodgers, M. A review of wearable sensors and systems with application in rehabilitation. J. Neuroeng. Rehabil. 2012, 9, 21. [Google Scholar] [CrossRef]
- Ohta, S.; Nakamoto, H.; Shinagawa, Y.; Tanikawa, T. A health monitoring system for elderly people living alone. J. Telemed. Telecare 2002. [Google Scholar] [CrossRef]
- Seppälä, J.; De Vita, I.; Jämsä, T.; Miettunen, J.; Isohanni, M.; Rubinstein, K.; Feldman, Y.; Grasa, E.; Corripio, I.; Berdun, J.; et al. Mobile Phone and Wearable Sensor-Based mHealth Approaches for Psychiatric Disorders and Symptoms: Systematic Review. JMIR Ment. Health 2019, 6, e9819. [Google Scholar] [CrossRef]
- Lisetti, C.L.; Nasoz, F. Using noninvasive wearable computers to recognize human emotions from physiological signals. EURASIP J. Appl. Signal Process. 2004, 2004, 1672–1687. [Google Scholar] [CrossRef]
- Choi, K.H.; Kim, J.; Kwon, O.S.; Kim, M.J.; Ryu, Y.H.; Park, J.E. Is heart rate variability (HRV) an adequate tool for evaluating human emotions? A focus on the use of the International Affective Picture System (IAPS). Psychiatry Res. 2017. [Google Scholar] [CrossRef]
- Kocielnik, R.; Sidorova, N.; Maggi, F.M.; Ouwerkerk, M.; Westerink, J.H.D.M. Smart technologies for long-term stress monitoring at work. In Proceedings of the CBMS 2013—26th IEEE International Symposium on Computer-Based Medical Systems, Porto, Portugal, 20–22 June 2013. [Google Scholar]
- Shen, N.; Levitan, M.-J.; Johnson, A.; Bender, J.L.; Hamilton-Page, M.; Jadad, A.A.R.; Wiljer, D. Finding a Depression App: A Review and Content Analysis of the Depression App Marketplace. JMIR Mhealth Uhealth 2015, 3, e16. [Google Scholar] [CrossRef]
- Lin, X.; Mermelstein, R.J.; Hedeker, D. A 3-level Bayesian mixed effects location scale model with an application to ecological momentary assessment data. Stat. Med. 2018. [Google Scholar] [CrossRef]
- Goldfried, M.R. The future of psychotherapy integration: Closing the gap between research and practice. J. Psychother. Integr. 2010, 20, 386–396. [Google Scholar] [CrossRef]
- Fernández-Álvarez, J.; Fernández-Álvarez, H.; Castonguay, L.G. Summarizing Novel Efforts to Integrate Practice and Research from a Practice Oriented Research Perspective. Rev. Argent. Clín. Psicol. 2018, 27, 353–362. [Google Scholar] [CrossRef]
- Walz, L.C.; Nauta, M.H.; aan het Rot, M. Experience sampling and ecological momentary assessment for studying the daily lives of patients with anxiety disorders: A systematic review. J. Anxiety Disord. 2014, 28, 925–937. [Google Scholar] [CrossRef]
- Schueller, S.M.; Aguilera, A.; Mohr, D.C. Ecological momentary interventions for depression and anxiety. Depress. Anxiety 2017, 34, 540–545. [Google Scholar] [CrossRef] [PubMed]
- Yoshiuchi, K.; Yamamoto, Y.; Akabayashi, A. Application of ecological momentary assessment in stress-related diseases. Biopsychosoc. Med. 2008, 2, 13. [Google Scholar] [CrossRef] [PubMed]
- Gee, B.L.; Griffiths, K.M.; Gulliver, A. Effectiveness of mobile technologies delivering Ecological Momentary Interventions for stress and anxiety: A systematic review. J. Am. Med. Inform. Assoc. 2016. [Google Scholar] [CrossRef]
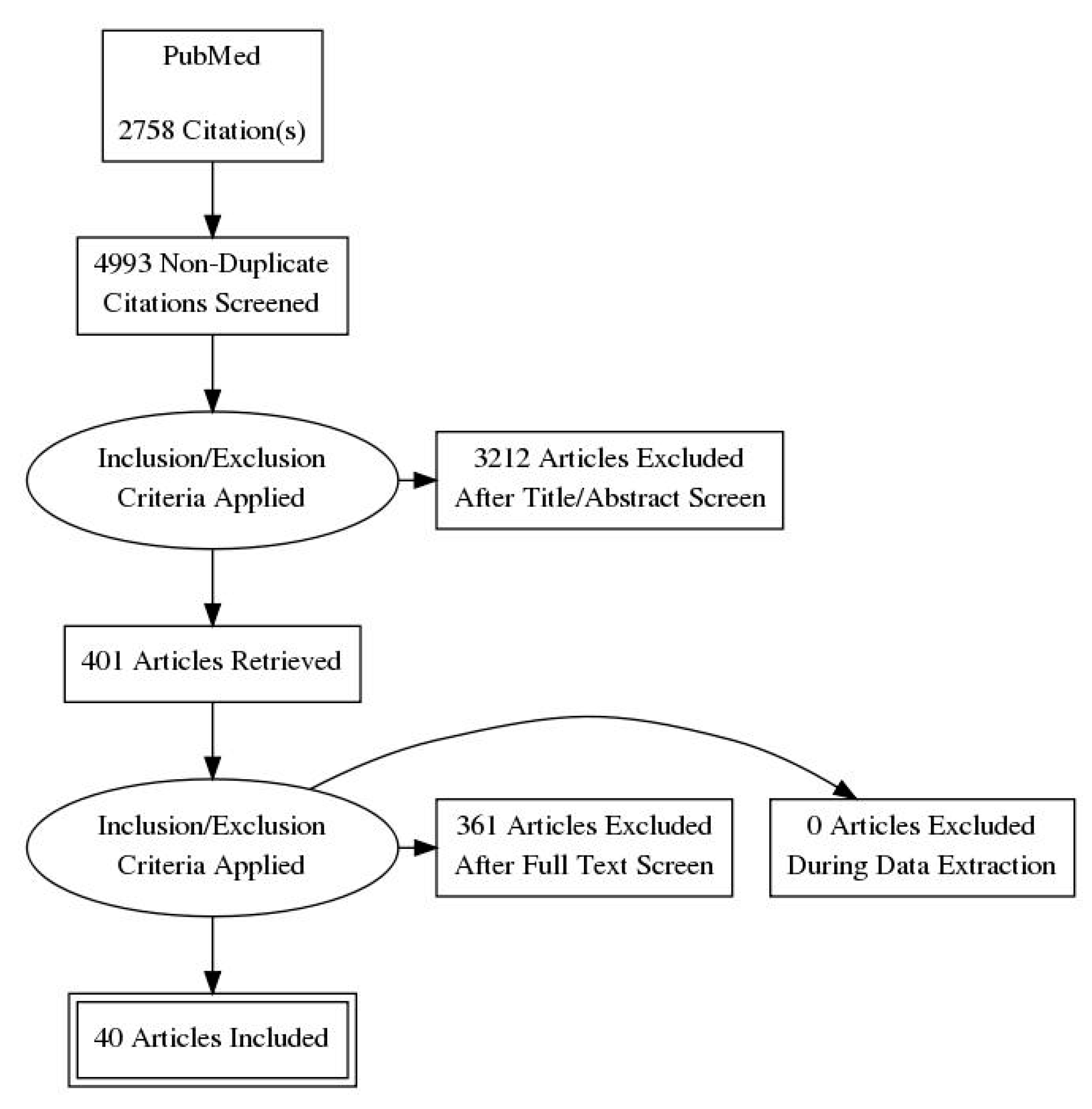
| Process | Results |
|---|---|
| PubMed/Medline | 2758 |
| Web of Science | 2235 |
| Total | 4993 |
| Not duplicated | 3613 |
| Excluded (after reading title and abstract) | 3212 |
| Retrieved | 401 |
| Excluded (after applying inclusion criteria) | 361 |
| Excluded (missing experimental data) | 0 |
| Final included articles | 40 |
| Authors | Sample | Variables | Device(s) | Duration | Prompts Per Day | Sampling Schema | Compliance | Sensor(s) | Primary Outcome(s) | |
|---|---|---|---|---|---|---|---|---|---|---|
| Recall bias | [8] | MDD (n = 26), and HCG (n = 25) | Affect | Palm Tungsten E2 | 7 days | 8 | Semi-randomized | 89% | No | Both depressed and non-depressed participants overestimate the retrospective recall of PA and NA. Depressed patients are more inaccurate in recalling NA. |
| [26] | MDD (n = 13) | Randomized items from PHQ-9 questionnaire | “Mindful Moods” mobile application | 29/30 days | 3 | Randomized | 78% | No | Even if strongly correlated, the PHQ-9 scores collected through the mobile application are significantly higher than those obtained though the retrospective paper-and-pencil PHQ-9. | |
| Symptoms monitoring | [39] | MDD (n = 20), and BD (n = 21) | Affect; stressors; behaviours; environment; social context | PDA | 3 days | 5 | Fixed sampling scheme | 85.7% | No | High rates of acceptance and compliance are observed among both samples. Participants show a practice effect, i.e., faster responses over the course of the study. |
| [40] | MDD (n = 26) | Depressive and anxiety symptoms | Palm Treo 650 Smartphone (Mental Health Telemetry mobile application) | 14 days | 1 | Selected by the patient | Not specified | No | Self-reported ratings of improvement at day 7 predict response to the treatment. | |
| [41] | MDD (n = 59) | Symptoms, sleep patterns, cognitive functioning | iHOPE smartphone application | 8 weeks | 2 (symptoms) | Not specified | Not specified | No | Baseline depression scores evaluated with HAM-D are associated with scores of PHQ-9, VAS for depression and anxiety symptoms collected with the application. | |
| 1 (sleep duration and quality) | ||||||||||
| Cortisol | [42] | MDD (n = 32), mD (n = 18), and HCG (n = 50) | Daily activities (frequency, social contacts); cortisol | Palm Pilot M100 | 4 days (over a maximum period of 7 days) | 4 (saliva samples) | Fixed sampling scheme | Not specified | No | In the control sample, daily activities are negatively associated with cortisol levels. This association is not observed in depressed patients. |
| 1 (self-report) | ||||||||||
| [43] | MDD (n = 37), and HCG (n = 36) | Sleep patterns; social contacts; cortisol | Palm Pilot M100 | 3 non-consecutive days (over a maximum period of 7 days) | 3 (saliva samples) | Fixed sampling scheme | 93% | No | Depressed patients show lower cortisol awakening response, lower sleep quality, and more negative social interactions. | |
| 1 (self-report) | ||||||||||
| [31] | MDD (n = 46) and HCG (n = 19) | Physiological indices (HR, respiration, accelerometer, cortisol); mood | LifeShirt System, with an integrated hand-held computer | 1 day | 6 (self-reports) | Fixed sampling scheme | 91% | LifeShirt System (HR, respiration, actigraphy) | Cortisol level and HRV do not differ between the two groups. Interestingly, NA is negatively correlated with HRV only in the control sample. | |
| 5 (saliva samples) | ||||||||||
| [44] | Remitted MDD (n = 31) and HCG (n = 32) | Mood; ruminative self-focus; stressful events; cortisol | Palm Tungsten E2 | 2 consecutive days | 10 | Semi-randomized | 94% | No | Rumination and low mood are associated with increased activation of the HPAA. In remitted patients, HPAA is less responsive to subtle emotional events. | |
| [36] | MDD (n = 15), and HCG (n = 15) | Affect; cognition; daily activities; cortisol | PsyMate | 30 days | 3 | Fixed sampling scheme | 92.5% | ActiCal (Respironics, Bend, OR, USA) | Compared to healthy participants, depressed patients report higher cortisol levels, higher α-amylase levels, and a greater ratio of α-amylase over cortisol. This latter association, however, disappears when correction for lifestyle factors is applied. | |
| [45] | MDD (n = 15), and HCG (n = 15) | Affect; cortisol | PsyMate | 30 days | 3 | Fixed sampling scheme | 92.5% | No | PA and NA are bidirectionally associated with cortisol levels. Nevertheless, the direction, sign, and timing of this association show great variability among subjects. | |
| Sleep patterns | [46] | MDD (n = 35), mD (n = 25), and HCG (n = 36) | Positive and negative affects | PDA | 3 days | 10 | Semi-randomized | 65% | No | Sleep quality predicts lower PA, but not NA. Low PA is associated with poor subjective sleep quality and self-reported daily dysfunction. |
| [47] | MDD and mD (n = 60), and HCG (n = 35) | Positive and negative affects; events appraisal | Palm Pilot Zire 22 | 3 days | 10 | Semi-randomized | 65% | No | In the non-clinical sample, sleep disturbances are associated with enhanced NA in response to negative events. Considering depressed patients, sleep disturbances negatively influence the emotional reactivity to both neutral and negative events. | |
| [48] | MDD (n = 27), and HCG (n = 27) | PA and NA, sleep quality; tiredness; rumination | PsyMate | 30 days | 3 | Fixed sampling scheme | 96% | No | Sleep quality directly influences PA and NA experienced during the following day, but not vice versa. Tiredness is a mediator. | |
| [49] | MDD (n = 14) and HCG (n = 15) | PA; NA; fatigue; sleep; activities; cognition; melatonin | PsyMate | 30 days | 3 | Fixed sampling scheme | 93% | No | Melatonin is associated with changes in affect and fatigue. However, changes in affect and fatigue are also predictors of melatonin levels. Individuals that do not show this association report higher depression severity and worse sleep quality. | |
| [38] | MDD (n = 27), and HCG (n = 27) | Sleep patterns | PsyMate | 30 days | 3 | Fixed sampling scheme | 96% | ActiCal (Respironics, Bend, OR, USA) | Sleep duration affects next-day physical activity. Depression does not moderate this association. | |
| [33] | MDD (n = 51) | Sleep patterns and quality; suicide ideation; entrapment perception | PRO-Diary actigraph watch (CamNtech) | 7 days | 6 | Semi-randomized | 89% | Accelerometer | Poor sleep quality, both objectively and subjectively evaluated, is associated with higher next-day suicide ideations. Suicide ideation does not influence sleep patterns and quality. | |
| Physical activity | [50] | MDD (n = 53), and HCG (n = 53) | Physical activity; positive and negative affects | Palm Pilot Zire 22 | 7 days | 8 | Randomized | 75% | No | Both samples show higher PA following physical activity. More specifically, depressed patients show a significantly higher increase in experienced PA levels after physical activity. |
| [32] | MDD (n = 14) and HCG (n = 43) | Mood; physical symptoms; physical activity | Ruputer ECOLOG | Average: 37.43 days (range:18–67 days) | 4 | Semi-randomized | 93% | Ambulatory Monitors Inc.—actigraph | Depressive mood is associated with increased intermittency of locomotor activity. | |
| [37] | MDD (n = 10), and HCG (n = 10) | Mood; cognition; daily activities; physical activity | PsyMate | 30 days | 3 | Fixed sampling scheme | 91% | ActiCal Respironics—actigraph | Despite the observation of large interindividual differences, results show a positive effect of physical activity on PA in all participants. | |
| Rumination | [51] | MDD (n = 6) and HCG (n = 7) | Context; mood; depressive symptoms; EEG (at baseline) | Palm Pilot and EEG (at baseline) | 7 days | 5 | Not specified | Not specified | No | Lower activation of bilateral PFC predicts higher rates of rumination, whereas higher levels of self-esteem are associated with lower right PFC activity. |
| [35] | MDD (n = 18) and HCG (n = 18) | Thoughts; disturbing events; feelings; possible influencing factors; feelings; HR | Electronic diary implemented on a smartphone | 1 day | Not reported | Semi-randomized | Not specified | RS 800CX; Bodyguard2 (HR and HRV) | Depressed participants show higher rates of perseverative cognition, which are associated with lower HRV. | |
| [52] | MDD (n = 38), GAD (n = 36), MDD with GAD comorbidity (n = 38), and HCG (n = 33) | Events stressfulness; rumination | Palm Pilot Zire 22 | 7 days | 8 | Randomized | 72% | No | MDD and GAD participants show the same level of rumination, which is even more severe in comorbid cases. Higher rates of rumination are predictive of worse affect, more maladaptive behaviours, and more severe symptoms. | |
| [53] | MDD (n = 16), GAD (n = 15), MDD with GAD comorbidity (n = 20), and HCG (n = 19) | Rumination; worry; PA and NA; significant events | Palm Pilot Zire 22 | 6 to7 days | 8 | Semi-randomized | 65% | No | Levels of rumination among all the clinical samples are higher in response to significant events. Decreased PA and increased NA are associated with higher momentary rumination. | |
| Emotional reaction | [54] | Remitted MDD (n = 55) and HCG (n = 55) | Perceived stress; mood | Hand held Psion “Revo” computer | 7 days | 5 | Randomized | 90% | No | Past episodes of depression are likely to increase the vulnerability to stressful events, especially in male participants. |
| [55] | MDD (n = 35), mD (n = 26), and HCG (n = 38) | Context; mood; events (nature of the event; location; people involved; affective rating) | Palm Pilot Zire 22 | 3 non-consecutive days (over a period of 5 days) | 10 | Semi-randomized | 65% | No | Both MDD and mD patients show lower levels of positive affect and rate events as more stressful and unpleasant than the control group. Furthermore, they show a higher reduction in negative feelings after positive events. | |
| [50] | MDD (n = 53), and HCG (n = 53) | Affect; significant events | Palm Pilot Zire 22 | 7/8 days | 8 | Randomized | 78% | No | Results point out greater emotional instability with respect to NA in depressed patients. No differences are observed in terms of reactivity, inertia, and instability in PA. | |
| [56] | MDD (n = 21) and MDD with BPD comorbidity (n = 20) | Affect and mood; events; subjective affective reactivity | Smartphone to access a web platform | 7 days | 5 | Randomized | 94% | No | Comorbidity with BPD does not imply major affective instability, but it is associated with lower subjective perception of affective reactivity. | |
| [57] | Remitted MDD (n = 10) and HCG (n = 11) | Mood; PA and NA; visual mental imagery | “Imagine your Mood”, smartphone application | 3 days a week, for 8 weeks | 10 | Semi-randomized | Not specified | No | In both samples, higher levels of visual imagery-based processing are associated with higher levels of PA and better mood, regardless of the valence of the imagery content. Elevated levels of visual imagery-based processing are not associated with daily affective reactivity. | |
| [58] | MDD (n = 51), and BPD (n = 80) | PA, NA, fear; hostility; sadness; interpersonal events | Palm Pilot Zire 31 | 28 days | 6 | Semi-randomized | 86% | No | Rejection and disagreement increase NA (especially hostility and sadness) both at a momentary and daily level, regardless of the diagnosis. The association between rejection/disagreement and hostility is stronger in BPD patients. | |
| [59] | MDD (n = 12), DD (n = 3), BD (n = 15) | Affect; location; social context; gambling desire/motivation/activities | Palm Pilot Zire 22 | 30 days | 3 | Randomized | 73% | No | High levels of sadness and arousal are predictive of gambling desire, regardless of the diagnosis. Depressed individuals are likely to gamble to increase PA or for social reasons. | |
| [60] | MDD (n = 15), GAD (n = 25) | Symptoms, PA, NA, rumination, behavioural avoidance, reassurance seeking | Web-based survey | 30 days | 4 | Not specified | Not specified | No | Using a person-by-person approach, results show that moment-to-moment symptomatology is mainly driven by positive mood, hopelessness, anger, and irritability, but not depressed mood, anhedonia, or worry. |
| Authors | Name | Sample | Intervention | Duration | Prompts | Sampling Schema | Sensor(s) | Primary Outcome(s) |
|---|---|---|---|---|---|---|---|---|
| [61] | Mobylize! | MDD (n = 7), with different comorbidities | Mobylize! is a context-aware system, composed of three main elements: (1) A mobile application for the collection of self-reports; (2) a website with feedback and theoretical lessons; (3) periodic contacts with trained coaches | 8 weeks | 5/day | Randomized | 38 concurrent sensors integrated in the phone | Mobylize! significantly reduced depressive symptoms. Predictive models did not reach high levels of accuracy, especially for mood. |
| [62] | PsyMate | MDD (n = 102): Experimental condition (n = 33), pseudo-experimental condition (n = 36), control condition (n = 33) | Daily assessment of self-reports and weekly EMA-derived feedback through face-to-face sessions | 3 days per week, for 6 weeks | 10/day | Semi-randomized | No | The use of EMA-derived feedback as a complementary intervention to pharmacological treatment significantly decreased depressive symptoms. These improvements were also maintained over time. |
| [63] | PsyMate | MDD (n = 102): Experimental condition (n = 33), pseudo-experimental condition (n = 36), control condition (n = 33) | Daily assessment of self-reports and weekly EMA-derived feedback through face-to-face sessions | 3 days per week, for 6 weeks | 10/day | Semi-randomized | No | The use of Psymate as a technique of self-monitoring could improve patients’ feelings of empowerment. |
| [64] | PsyMate | MDD (n = 102): Experimental condition (n = 33), pseudo-experimental condition (n = 36), control condition (n = 33) | Daily assessment of self-reports and weekly EMA-derived feedback through face-to-face sessions | 3 days per week, for 6 weeks | 10/day | Semi-randomized | No | Face-to-face EMA-derived feedback sessions did not increase the rate of PA experienced during or shortly after the intervention. |
| [65] | Medlink | MDD (n = 8) | Medlink is a mobile application delivering psychological support to MDD patients. The application provides users with: (1) psychoeducation; (2) weekly symptom assessment; (3) medication adherence monitoring; (4) monthly communication with a professional based on the previous points | 4 weeks | 1/week | Not specified | No | Medlink was positively evaluated by participants, especially regarding the weekly psychoeducation lessons. Depression severity of participants significantly decreased over the course of the experiment. |
| [66] | Help4Mood | MDD (n = 28): Experimental condition (n = 14) and control condition (n = 14) | Web platform providing daily assessment of symptoms, self-monitoring, and tailored activities. The delivered content is created in response to the user’s actions through a virtual agent | About 5 weeks | CESD- VAS-VA: 1/day | Not specified | Accelerometer and acoustic speech analysis | Only half of the participants used Help4Mood regularly. Significant changes in depressive symptoms were observed only among regular users. |
| PHQ-9: 1/week | ||||||||
| [67] | PsyMate | MDD (n = 102): Experimental condition (n = 33), pseudo-experimental condition (n = 36), control condition (n = 33) | Daily assessment of self-reports and weekly EMA-derived feedback through face-to-face sessions | 3 days per week, for 6 weeks | 10/day | Semi-randomized | No | The use of EMA-derived feedback decreased depressive symptoms and improved maladaptive behaviours. |
| [68] | PsyMate | MDD (n = 79): Experimental condition (n = 25), pseudo-experimental condition (n = 30), control condition (n = 24) | Daily assessment of self-reports and weekly EMA-derived feedback through face-to-face sessions | 3 days per week, for 6 weeks | 10/day | Semi-randomized | No | The use of a self-monitoring EMA improves negative emotion differentiation. |
| Field of Application | Retrieved Articles | Aim | Advantages |
|---|---|---|---|
| Recall bias | [8,26] | Experimental | No retrospective bias; control over backfilling; repeated momentary measurements. |
| Symptoms monitoring | [39,40,41] | Clinical | Continuous monitoring (symptoms assessment, treatment progress); real-time feedback to clinicians (e.g., crisis plan) and users (e.g., patterns visualization). |
| Cortisol dysregulation | [31,36,42,43,44,45] | Experimental | Role of contextual variables; temporal relationship between physiological measures and self-reports. |
| Sleep patterns | [33,38,46,47,48,49] | Experimental | Control over backfilling; no retrospective bias; integration of self-reports with passive data supplied by sensors and biosensors. |
| Physical activity | [32,37,50] | Experimental | Role of contextual variables; integration of self-reports with passive data supplied by sensors; temporal relationship between physiological measures and self-reports. |
| Rumination | [35,51,52,53] | Experimental | Role of contextual variables; rumination deployment across time. |
| Affect and emotional reactivity | [50,54,55,56,57,58,59,60], | Experimental | Role of contextual variables; temporal deployment of affect and emotional reactivity. |
| Advantages | Implications | |
|---|---|---|
| 1 | Real-time assessments | Reduction in retrospective bias and increase in accuracy. |
| 2 | Repeated measurements | Better comprehension of time-dependent processes and dynamic changes in symptoms. |
| 3 | Multimodal assessments | Contemporary analysis of behaviours, physiological signals, and subjective experiences. |
| 4 | Context-specific information | Assessment of symptoms as context-dependent. |
| 5 | Interactive assessments | Real-time customizable and interactive feedback. |
| 6 | Generalizability | Higher ecological validity and collection of more representative data. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Colombo, D.; Fernández-Álvarez, J.; Patané, A.; Semonella, M.; Kwiatkowska, M.; García-Palacios, A.; Cipresso, P.; Riva, G.; Botella, C. Current State and Future Directions of Technology-Based Ecological Momentary Assessment and Intervention for Major Depressive Disorder: A Systematic Review. J. Clin. Med. 2019, 8, 465. https://doi.org/10.3390/jcm8040465
Colombo D, Fernández-Álvarez J, Patané A, Semonella M, Kwiatkowska M, García-Palacios A, Cipresso P, Riva G, Botella C. Current State and Future Directions of Technology-Based Ecological Momentary Assessment and Intervention for Major Depressive Disorder: A Systematic Review. Journal of Clinical Medicine. 2019; 8(4):465. https://doi.org/10.3390/jcm8040465
Chicago/Turabian StyleColombo, Desirée, Javier Fernández-Álvarez, Andrea Patané, Michelle Semonella, Marta Kwiatkowska, Azucena García-Palacios, Pietro Cipresso, Giuseppe Riva, and Cristina Botella. 2019. "Current State and Future Directions of Technology-Based Ecological Momentary Assessment and Intervention for Major Depressive Disorder: A Systematic Review" Journal of Clinical Medicine 8, no. 4: 465. https://doi.org/10.3390/jcm8040465
APA StyleColombo, D., Fernández-Álvarez, J., Patané, A., Semonella, M., Kwiatkowska, M., García-Palacios, A., Cipresso, P., Riva, G., & Botella, C. (2019). Current State and Future Directions of Technology-Based Ecological Momentary Assessment and Intervention for Major Depressive Disorder: A Systematic Review. Journal of Clinical Medicine, 8(4), 465. https://doi.org/10.3390/jcm8040465







