A Culture-Independent Analysis of the Microbiota of Female Interstitial Cystitis/Bladder Pain Syndrome Participants in the MAPP Research Network
Abstract
1. Introduction
2. Experimental Section
2.1. Participants and Specimens
2.2. Specimen Handling
2.3. DNA Extraction and Ibis Eubacterial and Fungal Domain Assays on the PLEX-ID
2.4. Statistical Analysis
3. Results
3.1. Demographic Data
3.2. Clinical Data
3.3. Species Data
3.4. Genus Data
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A. NIH/NIDDK MAPP Network

Appendix B. Inclusion and Exclusion Criteria for MAPP 1. Adapted from [24]
Appendix B.1. Inclusion Criteria
Appendix B.2. Exclusion Criteria
Appendix B.3. Eligibility Criteria for Controls
Appendix C. Details of Methodology in Regard to PLEX-ID Analysis
References
- Hanno, P.M.; Burks, D.A.; Clemens, J.Q.; Dmochowski, R.R.; Erickson, D.; Fitzgerald, M.P.; Forrest, J.B.; Gordon, B.; Gray, M.; Mayer, R.D.; et al. AUA guideline for the diagnosis and treatment of interstitial cystitis/bladder pain syndrome. J. Urol. 2011, 185, 2162–2170. [Google Scholar] [CrossRef]
- Clemens, J.Q.; Meenan, R.T.; O’Keeffe Rosetti, M.C.; Brown, S.O.; Gao, S.Y.; Calhoun, E.A. Prevalence of interstitial cystitis symptoms in a managed care population. J. Urol. 2005, 174, 576–580. [Google Scholar] [CrossRef]
- Nickel, J.C.; Tripp, D.A.; Beiko, D.; Tolls, V.; Herschorn, S.; Carr, L.K.; Kelly, K.; Golda, N. The Interstitial Cystitis/Bladder Pain Syndrome Clinical Picture: A Perspective from Patient Life Experience. Urol. Pract. 2018, 5, 286–292. [Google Scholar] [CrossRef]
- Giannantoni, A.; Bini, V.; Dmochowski, R.; Hanno, P.; Nickel, J.C.; Proietti, S.; Wyndaele, J.J. Contemporary Management of the Painful Bladder. Eur. Urol. 2012, 61, 29–53. [Google Scholar] [CrossRef] [PubMed]
- Domingue, G.J.; Ghoniem, G.M. Dormant microbes in interstitial cystitis. J. Urol. 1995, 153, 1321–1326. [Google Scholar] [CrossRef]
- Heritz, D.M.; Lacroix, J.M.; Batra, S.D.; Jarvi, K.A.; Beheshti, B.; Mittelman, M.W. Detection of eubacteria in interstitial cystitis by 16S rDNA amplification. J. Urol. 1997, 158, 2291–2297. [Google Scholar] [CrossRef]
- Haarala, M.; Kiilholma, P.; Lehtonen, O.P. Urinary bacterial flora of women with urethral syndrome and interstitial cystitis. Gynecol. Obstet. Investig. 1999, 47, 42–44. [Google Scholar] [CrossRef] [PubMed]
- Haarala, M.; Kiiholma, P.; Nurmi, M.; Uksila, J.; Alanen, A. The role of Borrelia burgdorferi in interstitial cystitis. Eur. Urol. 2000, 37, 395–399. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, M.; Dixon, R.A. A study to detect Helicobacter pylori in fresh and archival specimens from patients with interstitial cystitis, using amplification methods. BJU Int. 2003, 91, 814–816. [Google Scholar] [CrossRef]
- Atug, F.; Turkeri, L.; Atug, O.; Cal, C. Detection of Helicobacter pylori in bladder biopsy specimens of patients with interstitial cystitis by polymerase chain reaction. Urol. Res. 2004, 32, 346–349. [Google Scholar] [CrossRef] [PubMed]
- Ehrlich, G.D.; Greenberg, S.J. PCR-Based Diagnostics in Infectious Disease; Blackwell Scientific Publications: Boston, MA, USA, 1994; 697p. [Google Scholar]
- Khasriya, R.; Sathiananthamoothy, S.; Ismail, S.; Kelsey, M.; Wilson, M.; Rohn, J.L.; Malone-Lee, J. Spectrum of bacterial colonization associated with urothelial cells from patients with chronic lower urinary tract symptoms. J. Clin. Microbiol. 2013, 51, 2054–2062. [Google Scholar] [CrossRef] [PubMed]
- Hilt, E.E.; McKinley, K.; Pearce, M.M.; Rosenfeld, A.B.; Zilliox, M.J.; Mueller, E.R.; Brubaker, L.; Gai, X.; Wolfe, A.J.; Schreckenberger, P.C. Bacterial Flora in the Adult Female Bladder Culture Techniques to Detect Resident Urine Is Not Sterile: Use of Enhanced Urine. J. Clin. Microbiol. 2014, 52, 871–876. [Google Scholar] [CrossRef] [PubMed]
- Whiteside, S.A.; Razvi, H.; Dave, S.; Reid, G.; Burton, J.P. The microbiome of the urinary tract—A role beyond infection. Nat. Rev. Urol. 2015, 12, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Thomas-White, K.; Brady, M.; Wolfe, A.J.; Mueller, E.R. The bladder is not sterile: History and current discoveries on the urinary microbiome. Curr. Bladder Dysfunct. Rep. 2016, 11, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Pearce, M.M.; Hilt, E.E.; Rosenfeld, A.B.; Zilliox, M.J.; Thomas-White, K.; Fok, C.; Kliethermes, S.; Schreckenberger, P.C.; Brubaker, L.; Gai, X.; et al. The female urinary microbiome: A comparison of women with and without urgency urinary incontinence. mBio 2014, 8, e01283-14. [Google Scholar] [CrossRef] [PubMed]
- Karstens, L.; Asquith, M.; Davin, S. Does the urinary microbiome play a role in urgency urinary incontinence and its severity. Front. Cell. Infect. Microbiol. 2016, 27, 78. [Google Scholar] [CrossRef] [PubMed]
- Thomas-White, K.J.; Kliethermes, S.; Rickey, L.; Lukacz, E.S.; Richter, H.E.; Moalli, P.; Zimmern, P.; Norton, P.; Kusek, J.W.; Wolfe, A.J.; et al. Evaluation of the urinary microbiota of women with uncomplicated stress urinary incontinence. Am. J. Obstet. Gynecol. 2017, 216, 55.e1–55.e16. [Google Scholar] [CrossRef] [PubMed]
- Al-Hadithi, H.N.; Williams, H.; Hart, C.A.; Frazer, M.; Adams, E.J.; Richmond, D.H.; Tincello, D.G. Absence of bacterial and viral DNA in bladder biopsies from patients with interstitial cystitis/chronic pelvic pain syndrome. J. Urol. 2005, 174, 151–154. [Google Scholar] [CrossRef] [PubMed]
- Ecker, D.J.; Sampath, R.; Massire, C.; Blyn, L.B.; Hall, T.A.; Eshoo, M.W.; Hofstadler, S.A. Ibis T5000: A universal biosensor approach for microbiology. Nat. Rev. Microbiol. 2008, 6, 553–558. [Google Scholar] [CrossRef]
- Nickel, J.C.; Stephens, A.; Landis, J.R.; Chen, J.; Mullins, C.; van Bokhoven, A.; Lucia, M.S.; Melton-Kreft, R.; Ehrlich, G.D.; MAPP Research Network. Search for Microorganisms in Men with Urologic Chronic Pelvic Pain Syndrome: A Culture-Independent Analysis in the MAPP Research Network. J. Urol. 2015, 194, 127–135. [Google Scholar] [CrossRef]
- Nickel, J.C.; Stephens, A.; Landis, J.R.; Mullins, C.; van Bokhoven, A.; Lucia, M.S.; Ehrlich, G.D.; MAPP Research Network. Assessment of the Lower Urinary Tract Microbiota during Symptom Flare in Women with Urologic Chronic Pelvic Pain Syndrome: A MAPP Network Study. J. Urol. 2016, 195, 356–362. [Google Scholar] [CrossRef]
- Clemens, J.Q.; Mullins, C.; Kusek, J.W.; Kirkali, Z.; Mayer, E.A.; Rodríguez, L.V.; Klumpp, D.J.; Schaeffer, A.J.; Kreder, K.J.; Buchwald, D.; et al. The MAPP research network: A novel study of urologic chronic pelvic pain syndromes. BMC Urol. 2014, 14, 57. [Google Scholar] [CrossRef] [PubMed]
- Landis, J.R.; Williams, D.A.; Lucia, M.S.; Clauw, D.J.; Naliboff, B.D.; Robinson, N.A.; van Bokhoven, A.; Sutcliffe, S.; Schaeffer, A.J.; Rodriguez, L.V.; et al. The MAPP research network: Design, patient characterization and operations. BMC Urol. 2014, 14, 58. [Google Scholar] [CrossRef] [PubMed]
- Chao, A. Non-parametric estimation of the number of classes in a population. Scand. J. Stat. 1984, 11, 265–270. [Google Scholar]
- Shannon, C.E.; Weaver, W. The Mathematical Theory of Communication; The University of Illinois Press: Urbana, IL, USA, 1949; 117p. [Google Scholar]
- Anderson, M.J. A new method for non-parametric multivariate analysis of variance. Aust. Ecol. 2001, 26, 32–46. [Google Scholar]
- Benjamini, Y.; Hochberg, Y. Controlling the false discovery rate: A practical and powerful approach to multiple testing. J. R. Stat. Soc. Ser. B 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Siddiqui, H.; Lagesen, K.; Nederbragt, A.J.; Jeansson, S.L.; Jakobsen, K.S. Alterations of microbiota in urine from women with interstitial cystitis. BMC Microbiol. 2012, 12, 205. [Google Scholar] [CrossRef]
- Brauundmeier-Fleming, A.; Russell, N.T.; Nas, M.Y.; Yaggie, R.E.; Berry, M.; Bachrach, L.; Flury, S.C.; Marko, D.S.; Bushell, C.B.; et al. Stool-based biomarkers of interstitial cystitis/bladder pain syndrome. Sci. Rep. 2016, 6, 26083. [Google Scholar] [CrossRef]
| Parameter | Category | *IC/BPS | Controls | Total | p |
|---|---|---|---|---|---|
| Number of Participants | n (%) | 181 | 182 | 363 | |
| Clinical Site | Northwestern U | 17 (9.4%) | 22 (12.1%) | 39 (10.7%) | 0.906 |
| UCLA | 25 (13.8%) | 24 (13.2%) | 49 (13.5%) | ||
| U of Iowa | 36 (19.9%) | 29 (15.9%) | 65 (17.9%) | ||
| U of Michigan | 33 (18.2%) | 33 (18.1%) | 66 (18.2%) | ||
| U of Washington | 23 (12.7%) | 29 (15.9%) | 52 (14.3%) | ||
| Wash U St. Louis | 39 (21.5%) | 37 (20.3%) | 76 (20.9%) | ||
| Stanford U | 8 (4.4%) | 8 (4.4%) | 16 (4.4%) | ||
| Age Group | <35 years | 76 (42.0%) | 81 (44.5%) | 157 (43.3%) | 0.869 |
| 35–50 years | 50 (27.6%) | 50 (27.5%) | 100 (27.5%) | ||
| 50+ years | 55 (30.4%) | 51 (28.0%) | 106 (29.2%) | ||
| Race | White | 165 (91.2%) | 137 (75.3%) | 302 (83.2%) | <0.001 |
| Black | 5 (2.8%) | 25 (13.7%) | 30 (8.3%) | ||
| Asian | 2 (1.1%) | 10 (5.5%) | 12 (3.3%) | ||
| Multi Race | 3 (1.7%) | 5 (2.7%) | 8 (2.2%) | ||
| Other | 5 (2.8%) | 4 (2.2%) | 9 (2.5%) | ||
| Unknown | 1 (0.6%) | 1 (0.5%) | 2 (0.6%) | ||
| Ethnicity | Hispanic | 11 (6.1%) | 11 (6.0%) | 22 (6.1%) | 1.000 |
| Non-Hispanic | 170 (93.9%) | 171 (94.0%) | 341 (93.9%) |
| Parameter | Category | IC/BPS | Controls | Total | p |
|---|---|---|---|---|---|
| Number of Participants | n (%) | 181 | 182 | 363 | |
| Self-reported IC diagnosis | No | 24 (13.3%) | 169 (92.9%) | 193 (53.2%) | <0.001 |
| Yes | 157 (86.7%) | 2 (1.1%) | 159 (43.8%) | ||
| Missing | 11 (6.0%) | 11 (3.0%) | |||
| Meet MAPP IC/BPS Criteria | Yes | 181 (100.0%) | 181 (49.9%) | ||
| Missing | 182 (100.0%) | 182 (50.1%) | |||
| IC diagnosis from Rice form | No | 64 (35.4%) | 172 (94.5%) | 236 (65.0%) | <0.001 |
| Yes | 117 (64.6%) | 10 (5.5%) | 127 (35.0%) | ||
| Associated Chronic Pain Syndrome | None | 105 (58.0%) | 110 (60.4%) | 215 (59.2%) | 0.670 |
| Any Syndrome | 76 (42.0%) | 72 (39.6%) | 148 (40.8%) | ||
| Interstitial Cystitis Symptom Index (ICSI) | 10.9 (4.4) | 3.0 (3.2) | 7.0 (5.5) | <0.001 | |
| Genitourinary Pain Index (GUPI) | 26.6 (8.7) | 4.7 (7.4) | 15.7 (13.6) | <0.001 | |
| Meds for urologic or pelvic pain symptoms | No | 37 (20.4%) | 176 (96.7%) | 213 (58.7%) | <0.001 |
| Yes | 144 (79.6%) | 6 (3.3%) | 150 (41.3%) | ||
| Pain medication class | None | 36 (19.9%) | 121 (66.5%) | 157 (43.3%) | <0.001 |
| Peripheral | 42 (23.2%) | 24 (13.2%) | 66 (18.2%) | ||
| Central | 81 (44.8%) | 28 (15.4%) | 109 (30.0%) | ||
| Opioid | 22 (12.2%) | 9 (4.9%) | 31 (8.5%) |
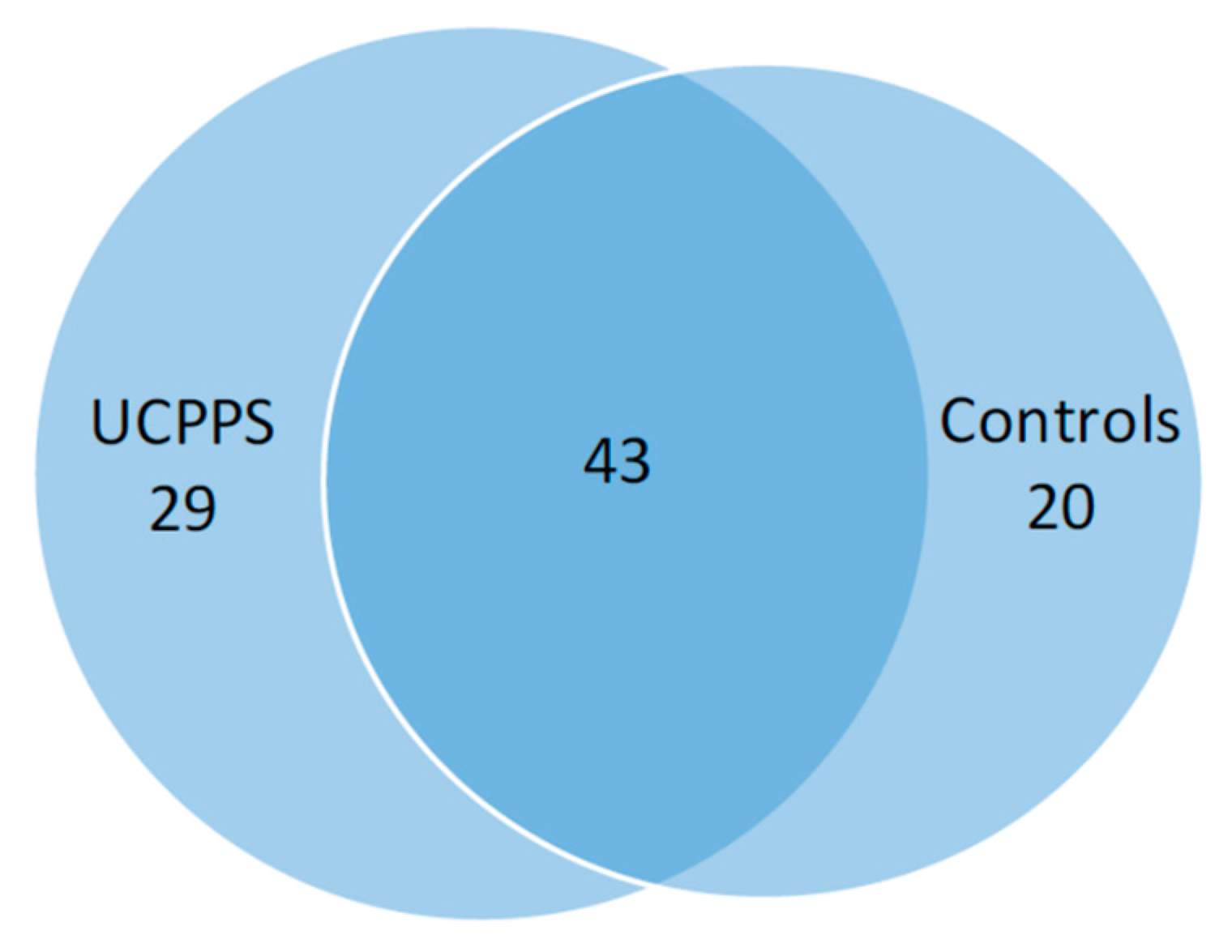
| Species unique to IC/BPS | Species unique to Controls | Species found in both: |
|---|---|---|
| 1. Acinetobacter grimontii | 1. Bordetella parapertussis | 1. Bacteroides ureolyticus |
| 2. Akkermansia muciniphila | 2. Burkholderia sp. | 2. Bifidobacterium inopinatum |
| 3. Bacillus sp. | 3. Clostridium sp. | 3. Bifidobacterium longum |
| 4. Bifidobacterium bifidum | 4. Enterococcus faecium | 4. Bifidobacterium subtile |
| 5. Bifidobacterium infantis | 5. Haemophilus influenzae | 5. Bordetella bronchiseptica |
| 6. Borrelia turicatae | 6. Klebsiella oxytoca | 6. Burkholderia cenocepacia |
| 7. Candida dubliniensis | 7. Klebsiella pneumoniae | 7. Candida albicans |
| 8. Clostridium perfringens | 8. Lactobacillus collinoides | 8. Candida glabrata |
| 9. Escherichia coli | 9. Ochrobactrum anthropi | 9. Corynebacterium diphtheriae |
| 10. Helicobacter hepaticus | 10. Pasteurella multocida | 10. Corynebacterium jeikeium |
| 11. Lactobacillus casei | 11. Pediococcus pentosaceus | 11. Corynebacterium pseudodiphtheriticum |
| 12. Lactobacillus helveticus | 12. Pseudomonas stutzeri | |
| 13. Lactobacillus reuteri | 13. Serratia marcescens | |
| 14. Lactococcus lactis | 14. Streptococcus mutans | |
| 15. Proteus mirabilis | 15. Streptococcus sanguinis | |
| 16. Pseudomonas aeruginosa | 16. Treponema denticola | |
| 17. Salmonella enterica | 17. Ureaplasma urealyticum | |
| 18. Staphylococcus intermedius | 18. Francisella philomiragia | |
| 19. Streptococcus dysgalactiae | 19. Corynebacterium striatum | |
| 20. Streptococcus porcinus | 20. Microbacterium sp. | |
| 21. Streptococcus pyogenes | ||
| 22. Bacteroides vulgatus | ||
| 23. Bifidobacterium pseudocatenulatum | ||
| 24. Francisella tularensis | ||
| 25. Mycoplasma hyorhinis | ||
| 26. Paracoccus denitrificans | ||
| 27. Staphylococcus sp. | ||
| 28. Micrococcus lylae | ||
| 29. Tetragenococcus halophilus |
| Taxonomic Level | Index | Controls | UCPPS | p1 | ||
|---|---|---|---|---|---|---|
| n | Mean (SD) | n | Mean (SD) | |||
| Species | Chao1 | 182 | 2.3 (1.3) | 181 | 2.5 (1.5) | 0.18 |
| Shannon | 182 | 0.2 (0.3) | 181 | 0.3 (0.3) | 0.15 | |
| Genus | Chao1 | 182 | 1.9 (1.1) | 181 | 2 (1.1) | 0.41 |
| Shannon | 182 | 0.2 (0.3) | 181 | 0.2 (0.3) | 0.38 | |
| Controls | UCPPS | Associate: Prevalence | Association: Relative Abundance | |||||
|---|---|---|---|---|---|---|---|---|
| taxa | Prevalence | Mean (SD) Relative Abundance | Prevalence | Mean (SD) Relative Abundance | OR (95% CI) | p1 | Mean Difference | p2 |
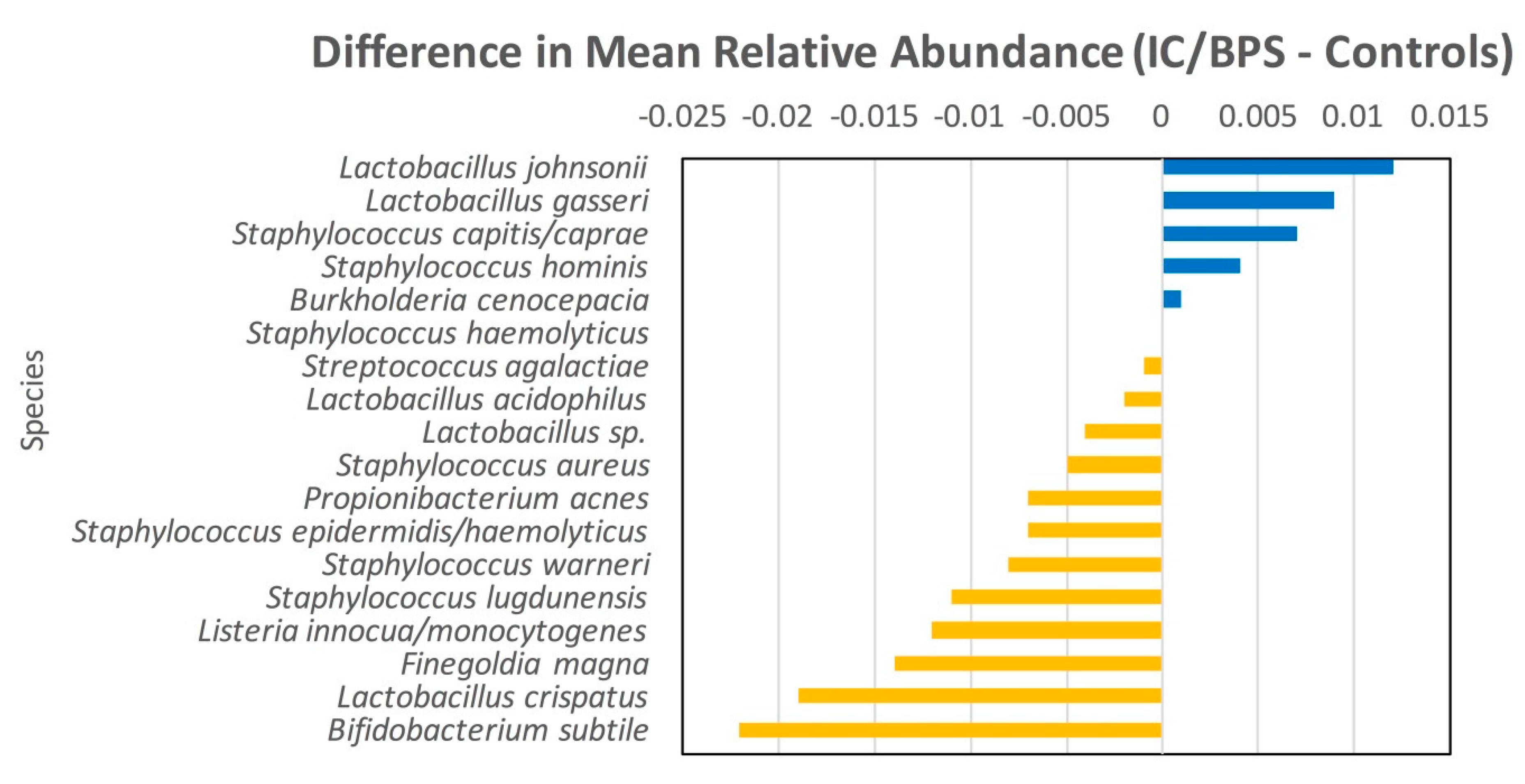
| Staphylococcus hominis | 20/182 (11%) | 0.01 (0.055) | 29/181 (16%) | 0.014 (0.091) | 1.55 (0.84,2.85) | 0.163 | 0.004 | 0.216 |
| Staphylococcus lugdunensis | 6/182 (3.3%) | 0.012 (0.106) | 6/181 (3.3%) | 0.002 (0.016) | 1.01 (0.32,3.18) | 0.992 | −0.011 | 0.996 |
| Staphylococcus warneri | 10/182 (5.5%) | 0.011 (0.091) | 4/181 (2.2%) | 0.003 (0.043) | 0.39 (0.12,1.26) | 0.116 | −0.008 | 0.098 |
| Streptococcus agalactiae | 11/182 (6%) | 0.009 (0.068) | 11/181 (6.1%) | 0.008 (0.078) | 1.01 (0.42,2.38) | 0.989 | −0.001 | 0.972 |
| Bifidobacterium subtile | 26/182 (14.3%) | 0.128 (0.322) | 26/181 (14.4%) | 0.106 (0.282) | 1.01 (0.56,1.81) | 0.982 | −0.022 | 0.872 |
| Burkholderia cenocepacia | 12/182 (6.6%) | 0.018 (0.128) | 8/181 (4.4%) | 0.019 (0.128) | 0.66 (0.26,1.64) | 0.367 | 0.001 | 0.389 |
| Finegoldia magna | 10/182 (5.5%) | 0.018 (0.113) | 12/181 (6.6%) | 0.004 (0.029) | 1.22 (0.51,2.9) | 0.651 | −0.014 | 0.703 |
| Lactobacillus acidophilus | 21/182 (11.5%) | 0.062 (0.223) | 20/181 (11%) | 0.06 (0.22) | 0.95 (0.5,1.82) | 0.883 | −0.002 | 0.878 |
| Lactobacillus crispatus | 54/182 (29.7%) | 0.277 (0.434) | 51/181 (28.2%) | 0.258 (0.419) | 0.93 (0.59,1.46) | 0.753 | −0.019 | 0.415 |
| Lactobacillus gasseri | 5/182 (2.7%) | 0.015 (0.11) | 12/181 (6.6%) | 0.024 (0.122) | 2.51 (0.87,7.29) | 0.090 | 0.009 | 0.084 |
| Lactobacillus johnsonii | 45/182 (24.7%) | 0.027 (0.127) | 51/181 (28.2%) | 0.038 (0.146) | 1.19 (0.75,1.91) | 0.456 | 0.012 | 0.329 |
| Lactobacillus sp. | 11/182 (6%) | 0.059 (0.233) | 11/181 (6.1%) | 0.055 (0.218) | 1.01 (0.42,2.38) | 0.989 | −0.004 | 0.96 |
| Listeria innocua/monocytogenes | 8/182 (4.4%) | 0.013 (0.098) | 5/181 (2.8%) | 0.001 (0.016) | 0.62 (0.2,1.93) | 0.407 | −0.012 | 0.39 |
| Propionibacterium acnes | 10/182 (5.5%) | 0.036 (0.173) | 12/181 (6.6%) | 0.03 (0.145) | 1.22 (0.51,2.9) | 0.651 | −0.007 | 0.686 |
| Staphylococcus aureus | 6/182 (3.3%) | 0.006 (0.051) | 4/181 (2.2%) | 0.001 (0.008) | 0.66 (0.18,2.39) | 0.560 | −0.005 | 0.524 |
| Staphylococcus capitis/caprae | 8/182 (4.4%) | 0.009 (0.077) | 7/181 (3.9%) | 0.016 (0.108) | 0.88 (0.31,2.47) | 0.801 | 0.007 | 0.814 |
| Staphylococcus epidermidis/haemolyticus | 57/182 (31.3%) | 0.05 (0.184) | 61/181 (33.7%) | 0.042 (0.171) | 1.11 (0.72,1.73) | 0.628 | −0.007 | 0.816 |
| Staphylococcus haemolyticus | 10/182 (5.5%) | 0.004 (0.037) | 11/181 (6.1%) | 0.004 (0.031) | 1.11 (0.46,2.69) | 0.812 | 0.000 | 0.819 |
| Controls | UCPPS | Associate: Prevalence | Association: Relative Abundance | |||||
|---|---|---|---|---|---|---|---|---|
| Taxa | Prevalence | Mean (SD) Relative Abundance | Prevalence | Mean (SD) Relative Abundance | OR (95% CI) | p1 | Mean Difference | p2 |
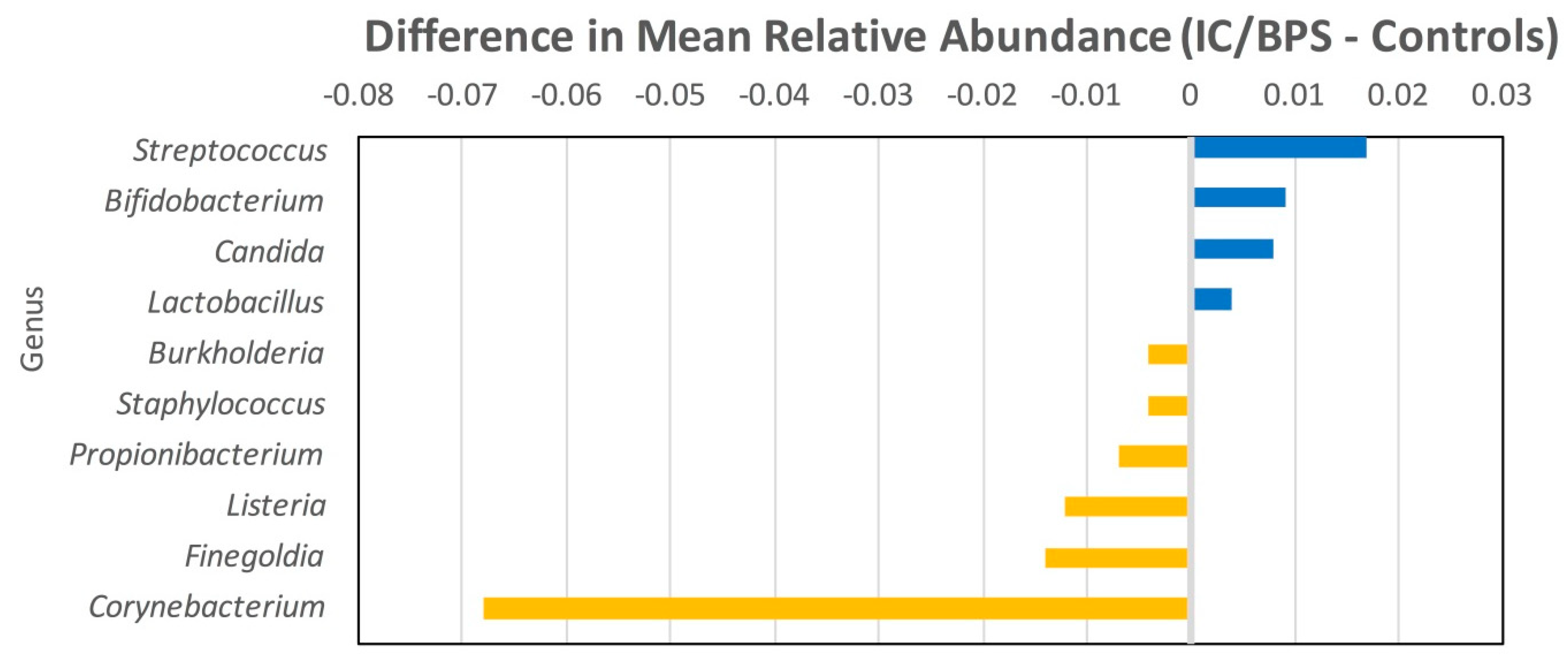
| Bifidobacterium | 28/182 (15.4%) | 0.138 (0.333) | 35/181 (19.3%) | 0.148 (0.327) | 1.32 (0.76,2.28) | 0.321 | 0.009 | 0.461 |
| Burkholderia | 13/182 (7.1%) | 0.023 (0.147) | 8/181 (4.4%) | 0.019 (0.128) | 0.6 (0.24,1.49) | 0.271 | −0.004 | 0.283 |
| Candida | 5/182 (2.7%) | 0 (0.004) | 11/181 (6.1%) | 0.009 (0.078) | 2.29 (0.78,6.73) | 0.132 | 0.008 | 0.119 |
| Corynebacterium | 23/182 (12.6%) | 0.08 (0.248) | 6/181 (3.3%) | 0.012 (0.085) | 0.24 (0.09,0.6) | 0.002* | −0.068 | 0.001 * |
| Finegoldia | 10/182 (5.5%) | 0.018 (0.113) | 12/181 (6.6%) | 0.004 (0.029) | 1.22 (0.51,2.9) | 0.651 | −0.014 | 0.703 |
| Lactobacillus | 104/182 (57.1%) | 0.457 (0.473) | 109/181 (60.2%) | 0.461 (0.461) | 1.14 (0.75,1.72) | 0.552 | 0.004 | 0.822 |
| Listeria | 8/182 (4.4%) | 0.013 (0.098) | 5/181 (2.8%) | 0.001 (0.016) | 0.62 (0.2,1.93) | 0.407 | −0.012 | 0.39 |
| Propionibacterium | 10/182 (5.5%) | 0.036 (0.173) | 12/181 (6.6%) | 0.03 (0.145) | 1.22 (0.51,2.9) | 0.651 | −0.007 | 0.686 |
| Staphylococcus | 101/182 (55.5%) | 0.107 (0.258) | 99/181 (54.7%) | 0.103 (0.263) | 0.97 (0.64,1.46) | 0.879 | −0.004 | 0.511 |
| Streptococcus | 17/182 (9.3%) | 0.018 (0.099) | 26/181 (14.4%) | 0.035 (0.156) | 1.63 (0.85,3.12) | 0.141 | 0.017 | 0.141 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nickel, J.C.; Stephens-Shields, A.J.; Landis, J.R.; Mullins, C.; van Bokhoven, A.; Lucia, M.S.; Henderson, J.P.; Sen, B.; Krol, J.E.; Ehrlich, G.D.; et al. A Culture-Independent Analysis of the Microbiota of Female Interstitial Cystitis/Bladder Pain Syndrome Participants in the MAPP Research Network. J. Clin. Med. 2019, 8, 415. https://doi.org/10.3390/jcm8030415
Nickel JC, Stephens-Shields AJ, Landis JR, Mullins C, van Bokhoven A, Lucia MS, Henderson JP, Sen B, Krol JE, Ehrlich GD, et al. A Culture-Independent Analysis of the Microbiota of Female Interstitial Cystitis/Bladder Pain Syndrome Participants in the MAPP Research Network. Journal of Clinical Medicine. 2019; 8(3):415. https://doi.org/10.3390/jcm8030415
Chicago/Turabian StyleNickel, J. Curtis, Alisa J. Stephens-Shields, J. Richard Landis, Chris Mullins, Adrie van Bokhoven, M. Scott Lucia, Jeffrey P. Henderson, Bhaswati Sen, Jaroslaw E. Krol, Garth D. Ehrlich, and et al. 2019. "A Culture-Independent Analysis of the Microbiota of Female Interstitial Cystitis/Bladder Pain Syndrome Participants in the MAPP Research Network" Journal of Clinical Medicine 8, no. 3: 415. https://doi.org/10.3390/jcm8030415
APA StyleNickel, J. C., Stephens-Shields, A. J., Landis, J. R., Mullins, C., van Bokhoven, A., Lucia, M. S., Henderson, J. P., Sen, B., Krol, J. E., Ehrlich, G. D., & The MAPP Research Network. (2019). A Culture-Independent Analysis of the Microbiota of Female Interstitial Cystitis/Bladder Pain Syndrome Participants in the MAPP Research Network. Journal of Clinical Medicine, 8(3), 415. https://doi.org/10.3390/jcm8030415






