Importance of Completing Hybrid Cardiac Rehabilitation for Long-Term Outcomes: A Real-World Evaluation
Abstract
1. Introduction
2. Methods
2.1. Cardiac Rehabilitation (CR) Particpation Study
2.2. Study Design and Data Sources
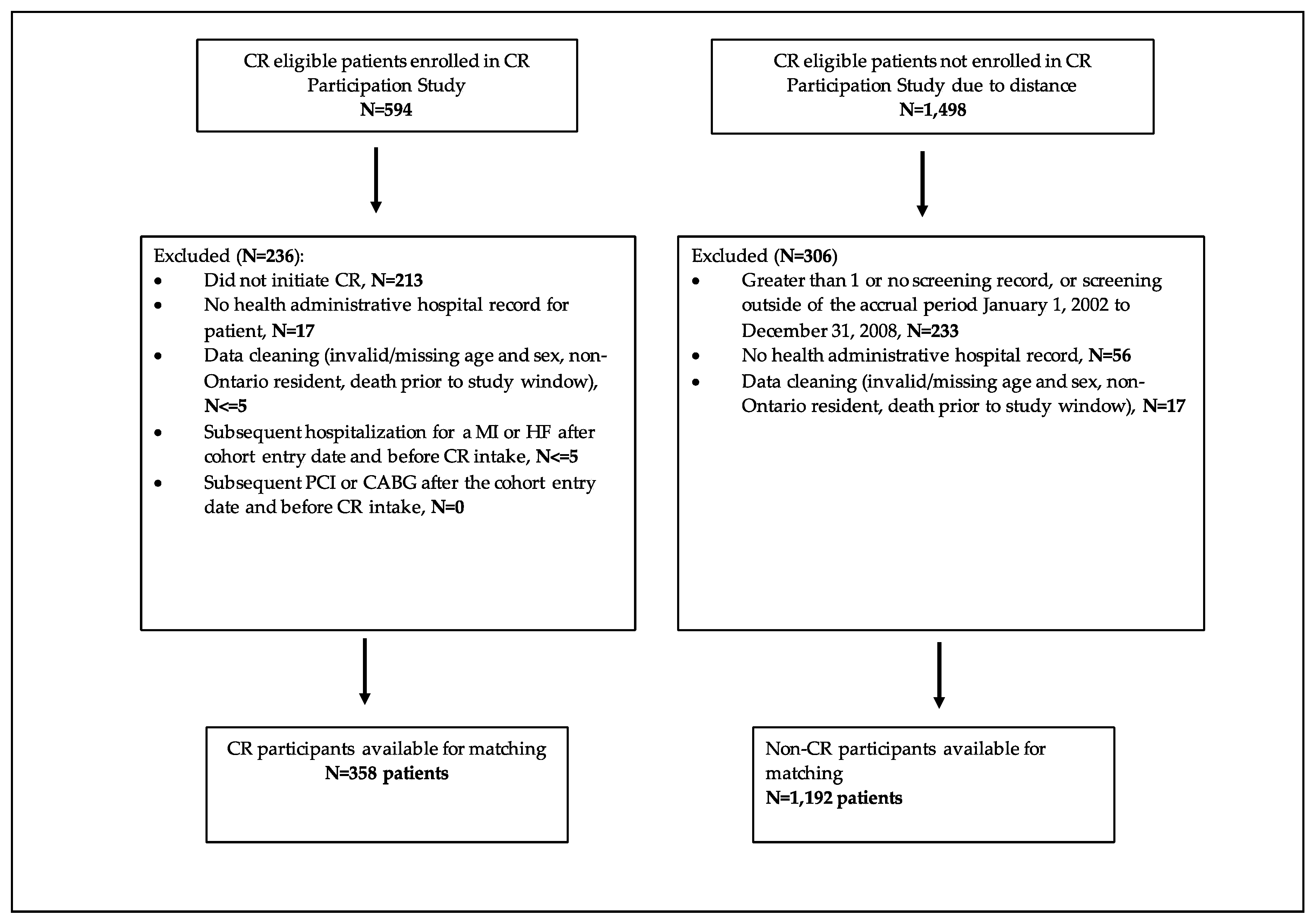
2.3. Study Sample
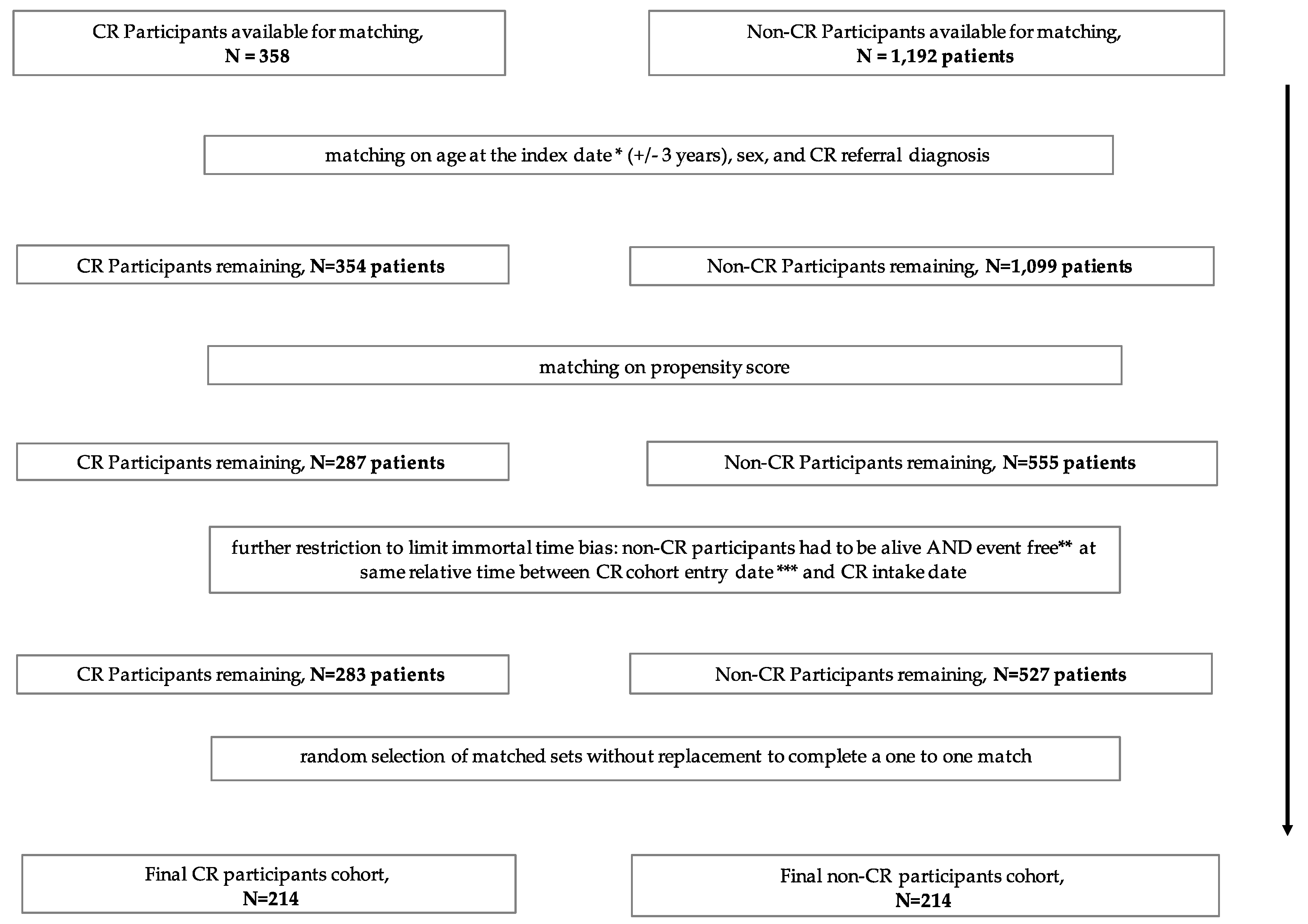
2.4. Matching Criteria
2.5. Hybrid CR Program Completion
2.6. Outcomes
2.7. Statistical Analysis
3. Results
3.1. Baseline Characteristics
3.2. Outcomes
4. Discussion
Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- O’Gara, P.T.; Kushner, F.G.; Ascheim, D.D.; Casey, D.E.; Chung, M.K.; de Lemos, J.A.; Ettinger, S.M.; Fang, J.C.; Fesmire, F.M.; Franklin, B.A.; et al. 2013 ACCF/AHA Guideline for the Management of ST-Elevation Myocardial Infarction: Executive Summary: A Report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Catheter. Cardiovasc. Interv. 2013, 82, E1–E27. [Google Scholar] [CrossRef] [PubMed]
- Levine, G.N.; Bates, E.R.; Blankenship, J.C.; Bailey, S.R.; Bittl, J.A.; Cercek, B.; Chambers, C.E.; Ellis, S.G.; Guyton, R.A.; Hollenberg, S.M.; et al. 2011 ACCF/AHA/SCAI Guideline for Percutaneous Coronary Intervention A Report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines and the Society for Cardiovascular Angiography and Interventions. J. Am. Coll.Cardiol. 2011, 58, E44–E122. [Google Scholar] [CrossRef]
- Hillis, L.D.; Smith, P.K.; Anderson, J.L.; Bittl, J.A.; Bridges, C.R.; Byrne, J.G.; Cigarroa, J.E.; DiSesa, V.J.; Hiratzka, L.F.; Hutter, A.M.; et al. 2011 ACCF/AHA Guideline for Coronary Artery Bypass Graft Surgery A Report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines Developed in Collaboration With the American Association for Thoracic Surgery, Society of Cardiovascular Anesthesiologists, and Society of Thoracic Surgeons. J. Am. Coll.Cardiol. 2011, 58, E123–E210. [Google Scholar] [PubMed]
- Grace, S.L.; Chessex, C.; Arthur, H.; Chan, S.; Cyr, C.; Dafoe, W.; Juneau, M.; Oh, P.; Suskin, N. Systematizing Inpatient Referral to Cardiac Rehabilitation 2010: Canadian Association of Cardiac Rehabilitation and Canadian Cardiovascular Society Joint Position Paper. Can. J. Cardiol. 2011, 27, 192–199. [Google Scholar] [CrossRef] [PubMed]
- Arena, R.; Williams, M.; Forman, D.E.; Cahalin, L.P.; Coke, L.; Myers, J.; Hamm, L.; Kris-Etherton, P.; Humphrey, R.; Bittner, V.; et al. Increasing referral and participation rates to outpatient cardiac rehabilitation: The valuable role of healthcare professionals in the inpatient and home health settings: A science advisory from the american heart association. Circulation 2012, 125, 1321–1329. [Google Scholar] [CrossRef] [PubMed]
- Piepoli, M.F.; Hoes, A.W.; Agewall, S.; Albus, C.; Brotons, C.; Catapano, A.L.; Cooney, M.T.; Corra, U.; Cosyns, B.; Deaton, C.; et al. 2016 European Guidelines on cardiovascular disease prevention in clinical practice: The Sixth Joint Task Force of the European Society of Cardiology and Other Societies on Cardiovascular Disease Prevention in Clinical Practice (constituted by representatives of 10 societies and by invited experts). Eur. J. Prev. Cardiol. 2016, 23, NP1–NP96. [Google Scholar] [PubMed]
- Candido, E.; Richards, J.A.; Oh, P.; Suskin, N.; Arthur, H.M.; Fair, T.; Alter, D.A. The Relationship between Need and Capacity for Multidisciplinary Cardiovascular Risk-Reduction Programs in Ontario. Can. J. Cardiol. 2011, 27, 200–207. [Google Scholar] [CrossRef] [PubMed]
- Sandesara, P.B.; Lambert, C.T.; Gordon, N.F.; Fletcher, G.F.; Franklin, B.A.; Wenger, N.K.; Sperling, L. Cardiac Rehabilitation and Risk Reduction: Time to “Rebrand and Reinvigorate”. J. Am. Coll. Cardiol. 2015, 65, 389–395. [Google Scholar] [CrossRef] [PubMed]
- Clark, A.M.; Hartling, L.; Vandermeer, B.; Lissel, S.L.; McAlister, F.A. Secondary prevention programmes for coronary heart disease: A meta-regression showing the merits of shorter, generalist, primary care-based interventions. Eur. J. Cardiovasc. Prev. Rehabil. 2007, 14, 538–546. [Google Scholar] [CrossRef] [PubMed]
- Clark, A.M.; Haykowsky, M.; Kryworuchko, J.; MacClure, T.; Scott, J.; DesMeules, M.; Luo, W.; Liang, Y.; McAlister, F.A. A meta-analysis of randomized control trials of home-based secondary prevention programs for coronary artery disease. Eur. J. Cardiovasc. Prev. Rehabil. 2010, 17, 261–270. [Google Scholar] [CrossRef] [PubMed]
- Clark, R.A.; Conway, A.; Poulsen, V.; Keech, W.; Tirimacco, R.; Tideman, P. Alternative models of cardiac rehabilitation: A systematic review. Eur. J. Prev. Cardiol. 2015, 22, 35–74. [Google Scholar] [CrossRef] [PubMed]
- Suskin, N.; Irvine, J.; Arnold, J.M.O.; Turner, R.; Zandri, J.; Prior, P.; Unsworth, K. Improving cardiac rehabilitation (CR) participation in women and men, the CR participation study. J. Cardiopul. Rehabil. 2007, 27, 342. [Google Scholar] [CrossRef]
- Canadian Association of Cardiac Rehabilitation. Canadian guidelines for cardiac rehabilitation and cardiovascular disease prevention: Enhancing the science, refining the art, 2nd ed.; Canadian Association of Cardiac Rehabilitation: Winnipeg, MB, Canada, 2004; pp. xii, 318. [Google Scholar]
- Zigmond, A.S.; Snaith, R.P. The hospital anxiety and depression scale. Acta Psychiatr. Scand. 1983, 67, 361–370. [Google Scholar] [CrossRef] [PubMed]
- Franklin, B.A.; Brinks, J.; Berra, K.; Lavie, C.J.; Gordon, N.F.; Sperling, L.S. Using Metabolic Equivalents in Clinical Practice. Am. J. Cardiol. 2018, 121, 382–387. [Google Scholar] [CrossRef] [PubMed]
- Alter, D.A.; Iron, K.; Austin, P.C.; Naylor, C.D.; Grp, S.S. Socioeconomic status, service patterns, and perceptions of care among survivors of acute myocardial infarction in Canada. JAMA 2004, 291, 1100–1107. [Google Scholar] [CrossRef] [PubMed]
- Ko, D.T.; Mamdani, M.; Alter, D.A. Lipid-lowering therapy with statins in high-risk elderly patients—The treatment-risk paradox. JAMA 2004, 291, 1864–1870. [Google Scholar] [CrossRef] [PubMed]
- Hux, J.E.; Ivis, F.; Flintoft, V.; Bica, A. Diabetes in Ontario: determination of prevalence and incidence using a validated administrative data algorithm. Diabetes Care 2002, 25, 512–516. [Google Scholar] [CrossRef] [PubMed]
- Tu, K.; Campbell, N.R.; Chen, Z.L.; Cauch-Dudek, K.J.; McAlister, F.A. Accuracy of administrative databases in identifying patients with hypertension. Open Med. 2007, 1, e18–e26. [Google Scholar] [PubMed]
- Dhoore, W.; Sicotte, C.; Tilquin, C. Risk adjustment in outcome assessment - the charlson comorbidity index. Methods Inf. Med. 1993, 32, 382–387. [Google Scholar]
- Austin, P.C. Optimal caliper widths for propensity-score matching when estimating differences in means and differences in proportions in observational studies. Pharm. Stat. 2011, 10, 150–161. [Google Scholar] [CrossRef] [PubMed]
- Grace, S.L.; Poirier, P.; Norris, C.M.; Oakes, G.H.; Somanader, D.S.; Suskin, N.; Canadian Association of Cardiac Rehabilitation. Pan-Canadian Development of Cardiac Rehabilitation and Secondary Prevention Quality Indicators. Can. J. Cardiol. 2014, 945–948. [Google Scholar] [CrossRef] [PubMed]
- Pescatello, L.S.; Franklin, B.A.; Fagard, R.; Farquhar, W.B.; Kelley, G.A.; Ray, C.A. American College of Sports Medicine position stand. Exercise and hypertension. Med. Sci. Sports Exerc. 2004, 36, 533–553. [Google Scholar] [CrossRef] [PubMed]
- Austin, P.A. Using the Standardized Difference to Compare the Prevalence of a Binary Variable Between Two Groups in Observational Research. Commun. Stat. Simul. Comput. 2009, 38, 1228–1234. [Google Scholar] [CrossRef]
- Martin, B.J.; Hauer, T.; Arena, R.; Austford, L.D.; Galbraith, P.D.; Lewin, A.M.; Knudtson, M.L.; Ghali, W.A.; Stone, J.A.; Aggarwal, S.G. Cardiac Rehabilitation Attendance and Outcomes in Coronary Artery Disease Patients. Circulation 2012, 126, 677–687. [Google Scholar] [CrossRef] [PubMed]
- Alter, D.A.; Oh, P.I.; Chong, A. Relationship between cardiac rehabilitation and survival after acute cardiac hospitalization within a universal health care system. Eur. J. Cardiovasc. Prev. Rehabil. 2009, 16, 102–113. [Google Scholar] [CrossRef] [PubMed]
- Taylor, R.S.; Brown, A.; Ebrahim, S.; Jolliffe, J.; Noorani, H.; Rees, K.; Skidmore, B.; Stone, J.A.; Thompson, D.R.; Oldridge, N. Exercise-based rehabilitation for patients with coronary heart disease: Systematic review and meta-analysis of randomized controlled trials. Am. J. Med. 2004, 116, 682–692. [Google Scholar] [CrossRef] [PubMed]
- Suskin, N.; Frisbee, S.; Stranges, S.; Pierce, A.; Ricci, J. AN integrated, region-wide cardiac rehabilitation system lowers mortality and rehospitalization. Can. J. Cardiol. 2018, 34, S30. [Google Scholar] [CrossRef]
- West, R.R.; Jones, D.A.; Henderson, A.H. Rehabilitation after myocardial infarction trial (RAMIT): Multi-centre randomised controlled trial of comprehensive cardiac rehabilitation in patients following acute myocardial infarction. Heart 2012, 98, 637–644. [Google Scholar] [CrossRef] [PubMed]
- Members, B.E.C. RAMIT presents an outdated version of cardiac rehabilitation. Heart 2012, 98, 672, author reply 673–674. [Google Scholar]
- Tran, M.; Pesah, E.; Turk-Adawi, K.; Supervia, M.; Jimenez, F.L.; Oh, P.; Baer, C.; Grace, S.L. Cardiac Rehabilitation Availability and Delivery in Canada: How Does It Compare With Other High-Income Countries? Can. J. Cardiol. 2018, 34, S252–S262. [Google Scholar] [CrossRef] [PubMed]
- Lavie, C.J.; Arena, R.; Franklin, B.A. Cardiac Rehabilitation and Healthy Life-Style Interventions Rectifying Program Deficiencies to Improve Patient Outcomes. J. Am. Coll. Cardiol. 2016, 67, 13–15. [Google Scholar] [CrossRef] [PubMed]
- Anderson, L.; Oldridge, N.; Thompson, D.R.; Zwisler, A.D.; Rees, K.; Martin, N.; Taylor, R.S. Exercise-Based Cardiac Rehabilitation for Coronary Heart Disease Cochrane Systematic Review and Meta-Analysis. J. Am. Coll. Cardiol. 2016, 67, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Reid, R.D.; Morrin, L.I.; Beaton, L.J.; Papadakis, S.; Kocourek, J.; McDonnell, L.; Slovinec D’Angelo, M.E.; Tulloch, H.; Suskin, N.; Unsworth, K.; et al. Randomized trial of an Internet-based computer-tailored expert system for physical activity in patients with heart disease. Eur. J. Prev. Cardiol. 2012, 19, 1357–1364. [Google Scholar] [CrossRef] [PubMed]
- Lear, S.A. The Delivery of Cardiac Rehabilitation Using Communications Technologies: The “Virtual” Cardiac Rehabilitation Program. Can. J. Cardiol. 2018, 34, S278–S283. [Google Scholar] [CrossRef] [PubMed]
- Marzolini, S.; Oh, P.I.; Alter, D.; Stewart, D.E.; Grace, S.L.; Investigators, C.R.C.C.t.A.R.E. Musculoskeletal comorbidities in cardiac patients: prevalence, predictors, and health services utilization. Arch. Phys. Med. Rehabil. 2012, 93, 856–862. [Google Scholar] [CrossRef] [PubMed]
- Ricci, J.; Suskin, N.; Stranges, S.; Pierce, A.; Fair, T.; Appasamy, T.; Williams, T.; Frisbee, S. Health care system redesign to achieve universal access to cardiac rehabilitation services: Implications completion and participation rates. Clin. Cardiol. 2018, 41, 10–11. [Google Scholar]
- Vigorito, C.; Abreu, A.; Ambrosetti, M.; Belardinelli, R.; Corra, U.; Cupples, M.; Davos, C.H.; Hoefer, S.; Iliou, M.C.; Schmid, J.P.; et al. Frailty and cardiac rehabilitation: A call to action from the EAPC Cardiac Rehabilitation Section. Eur. J. Prev. Cardiol. 2017, 24, 577–590. [Google Scholar] [CrossRef] [PubMed]
| Non-CR Participants (n = 214) | CR Participants (n = 214) | Total (n = 428) | Standardized Difference 1 | |
|---|---|---|---|---|
| Demographics | ||||
| Age (years), mean (SD) | 60.89 ± 9.15 | 60.86 ± 9.07 | 60.87 ± 9.10 | 0 |
| Female sex, n (%) | 56 (26.2%) | 56 (26.2%) | 112 (26.2%) | 0 |
| Income quintile, n (%) | ||||
| Quintile 1 | 30 (14.0%) | 32 (15.0%) | 62 (14.5%) | 0.03 |
| Quintile 2 | 55 (25.7%) | 45 (21.0%) | 100 (23.4%) | 0.11 |
| Quintile 3 | 36 (16.8%) | 41 (19.2%) | 77 (18.0%) | 0.06 |
| Quintile 4 | 47 (22.0%) | 39 (18.2%) | 86 (20.1%) | 0.09 |
| Quintile 5 | 46 (21.5%) | 57 (26.6%) | 103 (24.1%) | 0.12 |
| Rural, yes, n (%) | 59 (27.6%) | 15 (7.0%) | 74 (17.3%) | 0.56 |
| Year of cohort entry, n (%) | ||||
| 2003 | 25 (11.7%) | 15 (7.0%) | 40 (9.3%) | 0.16 |
| 2004 | 109 (50.9%) | 91 (42.5%) | 200 (46.7%) | 0.17 |
| 2005 | ≤80 | ≤70 | 144 (33.6%) | 0.14 |
| 2006 | ≤5 | ≤45 | 44 (10.3%) | 0.68 |
| Index cardiac event, n (%) | ||||
| Myocardial infarction | 43 (20.1%) | 43 (20.1%) | 86 (20.1%) | 0 |
| Unstable angina | 16 (7.5%) | 16 (7.5%) | 32 (7.5%) | 0 |
| Percutaneous coronary intervention | 69 (32.2%) | 69 (32.2%) | 138 (32.2%) | 0 |
| Coronary artery bypass graft surgery | 86 (40.2%) | 86 (40.2%) | 172 (40.2%) | 0 |
| Time between hospital discharge and start of CR (days) | ||||
| Mean (SD) | 103.28 (47.67) | 103.28 (47.67) | 103.28 (47.62) | 0 |
| Prior cardiac events in the previous 5 years, n (%) | ||||
| Myocardial infarction | 26 (12.1%) | 22 (10.3%) | 48 (11.2%) | 0.06 |
| Unstable angina | 22 (10.3%) | 21 (9.8%) | 43 (10.0%) | 0.02 |
| Percutaneous coronary intervention | 7 (3.3%) | 11 (5.1%) | 18 (4.2%) | 0.09 |
| Coronary artery bypass graft surgery | 0 | 0 | 0 | |
| Heart failure | 9 (4.2%) | 9 (4.2%) | 18 (4.2%) | 0 |
| Comorbidities in the previous 5 years, n (%) | ||||
| Atrial fibrillation/flutter | ≤5 | ≤5 | ≤10 | 0 |
| Hypertension | 51 (23.8%) | 53 (24.8%) | 104 (24.3%) | 0.02 |
| Hyperlipidemia | 9 (4.2%) | 6 (2.8%) | 15 (3.5%) | 0.08 |
| Haemorrhagic stroke | 0 (0.0%) | ≤5 | ≤5 | 0.1 |
| Ischemic stroke | 0 (0.0%) | ≤5 | ≤5 | 0.1 |
| Transient ischemic stroke | ≤5 | ≤5 | ≤5 | 0 |
| Chronic kidney disease | 11 (5.1%) | 13 (6.1%) | 24 (5.6%) | 0.04 |
| Diabetes mellitus | 24 (11.2%) | 18 (8.4%) | 42 (9.8%) | 0.09 |
| Peripheral vascular disease | ≤10 | ≤5 | 11 (2.6%) | 0.15 |
| Chronic lung disease (including chronic obstructive pulmonary disease) | 45 (21.0%) | 44 (20.6%) | 89 (20.8%) | 0.01 |
| Major cancers | 16 (7.5%) | 14 (6.5%) | 30 (7.0%) | 0.04 |
| Alcoholism | ≤5 | ≤5 | ≤5 | 0.06 |
| Obesity | ≤5 | ≤5 | ≤10 | 0 |
| Charlson comorbidity index [20] | ||||
| 0,1 | 63 (29.4%) | 63 (29.4%) | 126 (29.4%) | 0 |
| 2 | 10 (4.7%) | 12 (5.6%) | 22 (5.1%) | 0.04 |
| 3+ | 14 (6.5%) | 9 (4.2%) | 23 (5.4%) | 0.1 |
| No hospitalizations | 127 (59.3%) | 130 (60.7%) | 257 (60.0%) | 0.03 |
| Healthcare system utilization, n (%) | ||||
| Hospital episodes | ||||
| 0 | 127 (59.3%) | 130 (60.7%) | 257 (60.0%) | 0.03 |
| 1–5 | 84 (39.3%) | 82 (38.3%) | 166 (38.8%) | 0.02 |
| 6+ | ≤5 | ≤5 | ≤5 | 0.04 |
| Visits to a cardiologist | ||||
| 0 | 100 (46.7%) | 103 (48.1%) | 203 (47.4%) | 0.03 |
| 1+ | 114 (53.3%) | 111 (51.9%) | 225 (52.6%) | 0.03 |
| Visits to an internist | ||||
| 0 | 57 (26.6%) | 49 (22.9%) | 106 (24.8%) | 0.09 |
| 1+ | 157 (73.4%) | 165 (77.1%) | 322 (75.2%) | 0.09 |
| Non-CR Participants | CR Participants | p-Value 1 | |
|---|---|---|---|
| Primary analysis 2 | |||
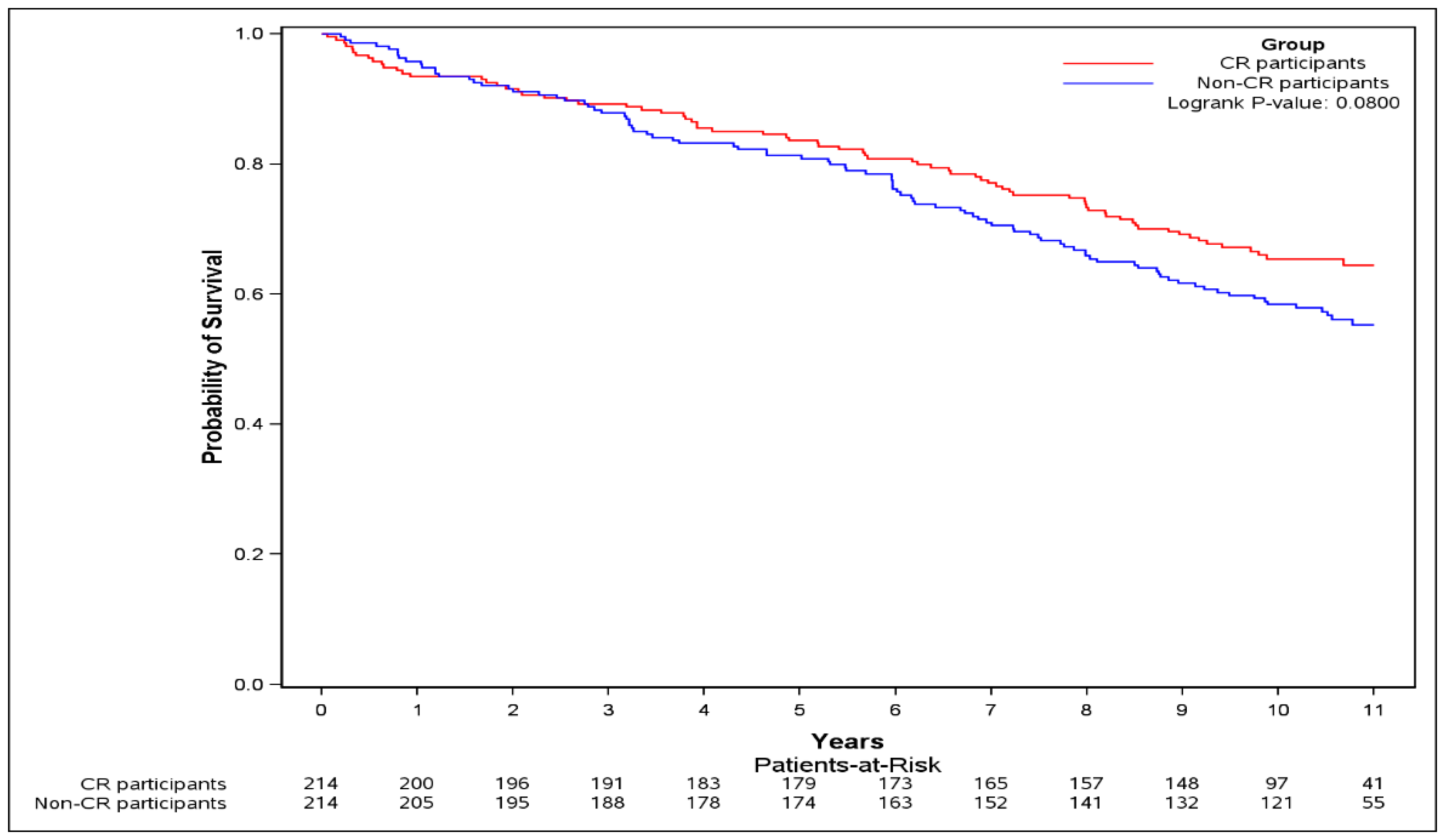
| Number of subjects | 214 | 214 | - |
| Follow-up duration in years Median (IQR) | 10.38 (6.17–11.02) | 9.88 (7.81–10.84) | - |
| Events N (%) | 97 (45.3) | 76 (35.5) | - |
| HR 3 unadjusted (95% CI) | - | 0.84 (0.60–1.17) | 0.3 |
| HR 4 adjusted (95% CI) | - | 0.86 (0.60–1.22) | 0.39 |
| Secondary analysis (event-free at 1-year) 5 | |||
| Number of subjects (%) | 192 (matched) | 192 (89.7%) | - |
| Follow-up duration in years Median (IQR) | 9.48 (6.20–10.05) | 8.92 (7.49–9.85) | |
| Events N (%) | 79 (41.1%) | 61 (31.8) | - |
| HR 4 unadjusted (95% CI) | - | 0.75 (0.52–1.09) | 0.13 |
| Sensitivity analysis (event-free at successful CR completion) 6 | |||
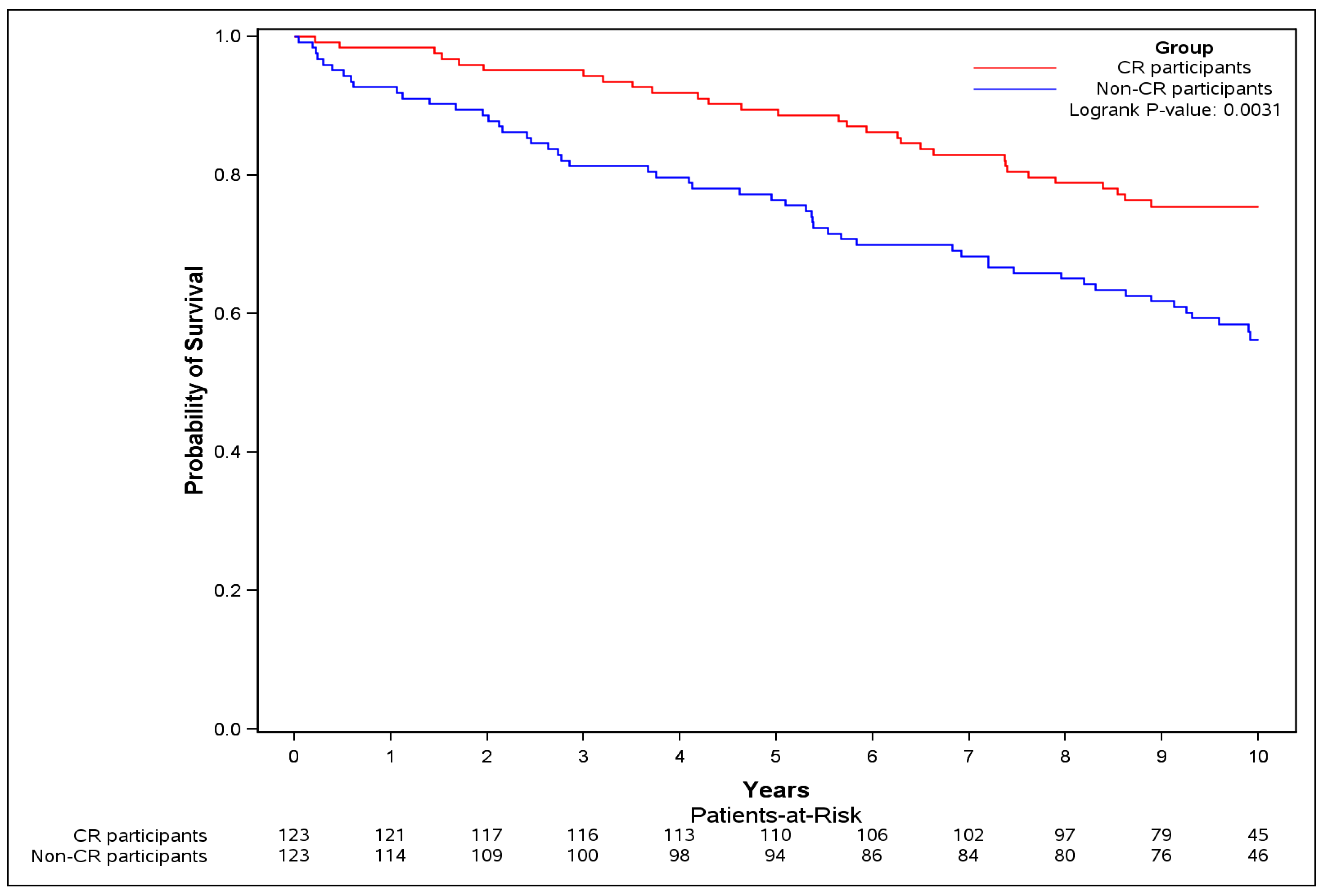
| Number of subjects (%) | 123 (matched) | 123 (57.5%) | - |
| Follow-up duration in years Median (IQR) | 9.72 (5.30–10.37) | 9.51 (8.54–10.32) | |
| Events N (%) | 54 (43.9) | 32 (26.0) | - |
| HR 4 unadjusted (95% CI) | - | 0.49 (0.29–0.81) | 0.006 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Suskin, N.G.; Shariff, S.Z.; Garg, A.X.; Reid, J.; Unsworth, K.; Prior, P.L.; Alter, D. Importance of Completing Hybrid Cardiac Rehabilitation for Long-Term Outcomes: A Real-World Evaluation. J. Clin. Med. 2019, 8, 290. https://doi.org/10.3390/jcm8030290
Suskin NG, Shariff SZ, Garg AX, Reid J, Unsworth K, Prior PL, Alter D. Importance of Completing Hybrid Cardiac Rehabilitation for Long-Term Outcomes: A Real-World Evaluation. Journal of Clinical Medicine. 2019; 8(3):290. https://doi.org/10.3390/jcm8030290
Chicago/Turabian StyleSuskin, Neville G., Salimah Z. Shariff, Amit X. Garg, Jennifer Reid, Karen Unsworth, Peter L. Prior, and David Alter. 2019. "Importance of Completing Hybrid Cardiac Rehabilitation for Long-Term Outcomes: A Real-World Evaluation" Journal of Clinical Medicine 8, no. 3: 290. https://doi.org/10.3390/jcm8030290
APA StyleSuskin, N. G., Shariff, S. Z., Garg, A. X., Reid, J., Unsworth, K., Prior, P. L., & Alter, D. (2019). Importance of Completing Hybrid Cardiac Rehabilitation for Long-Term Outcomes: A Real-World Evaluation. Journal of Clinical Medicine, 8(3), 290. https://doi.org/10.3390/jcm8030290





